Assessment of Prevalence and Risk Factors for Central Sensitization Related to Shoulder Osteoarthritis and Rotator Cuff Tears Using the Central Sensitization Inventory: A Cross-Sectional Study in Shoulder Surgery Patients
Abstract
:1. Introduction
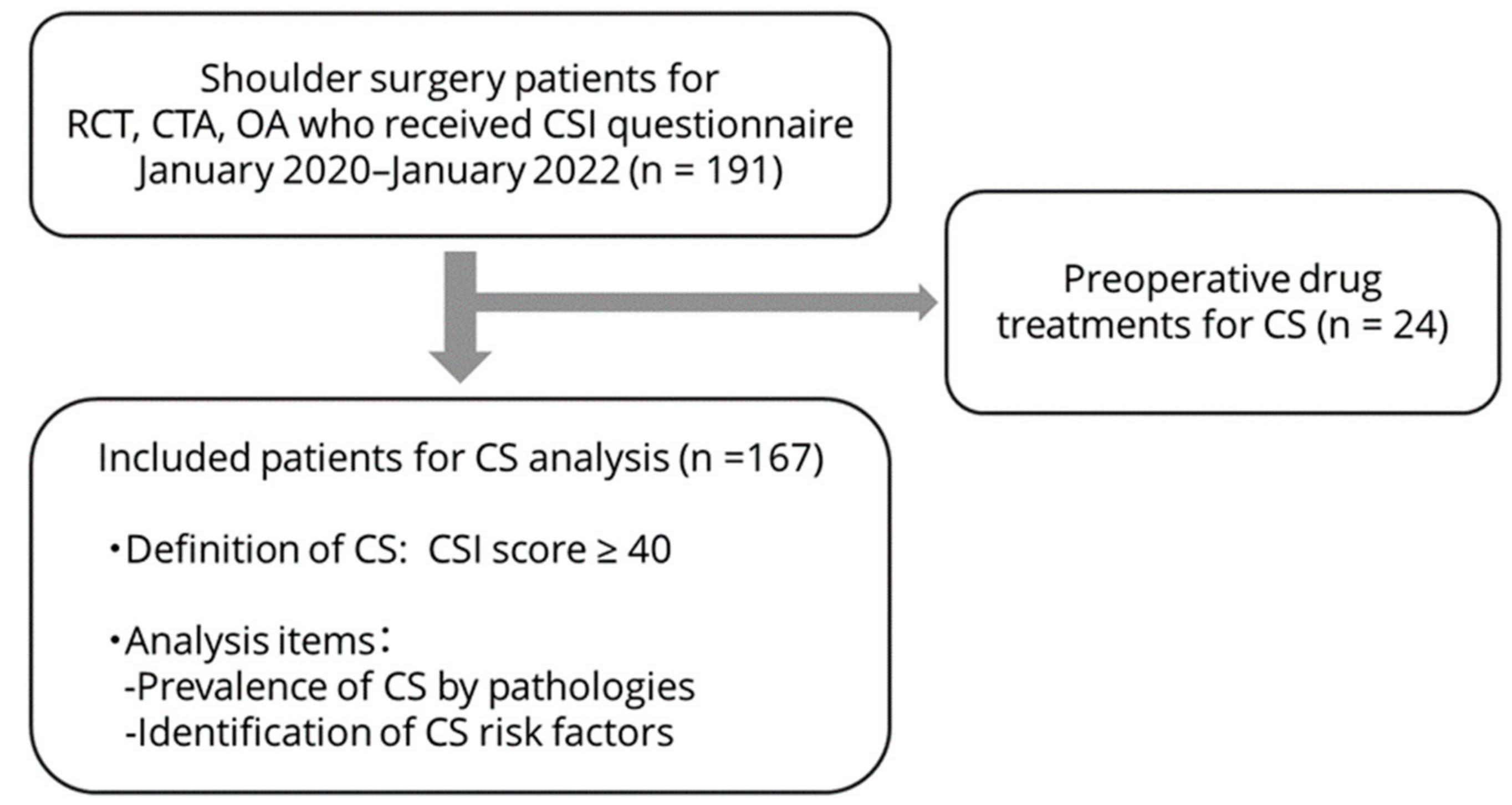
2. Materials and Methods
2.1. Patients
2.2. Evaluation of Central Sensitization
2.3. Statistical Analysis
3. Results
3.1. Demographic Data
3.2. Incidence and Severity of Central Sensitization
3.3. Association of Central Sensitization with Glenohumeral Osteoarthritis and Rotator Cuff Tears
3.4. Factors Associated with Central Sensitization
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Meislin, R.; Sperling, J.; Stitik, T. Persistent shoulder pain: Epidemiology, pathophysiology, and diagnosis. Am. J. Orthop. 2005, 34, 5–9. [Google Scholar] [PubMed]
- Harkness, E.F.; Macfarlane, G.J.; Nahit, E.S.; Silman, A.J.; McBeth, J. Mechanical and psychosocial factors predict new onset shoulder pain: A prospective cohort study of newly employed workers. Occup. Environ. Med. 2003, 60, 850–857. [Google Scholar] [CrossRef] [PubMed]
- Hviid Andersen, J.; Kaergaard, A.; Frost, P.; Frølund Thomsen, J.; Peter Bonde, J.; Fallentin, N.; Borg, V.; Mikkelsen, S. Physical, psychosocial, and individual risk factors for neck/shoulder pain with pressure tenderness in the muscles among workers performing monotonous, repetitive work. Spine 2002, 27, 660–667. [Google Scholar] [CrossRef] [PubMed]
- Latremoliere, A.; Woolf, C.J. Central sensitization: A generator of pain hypersensitivity by central neural plasticity. J. Pain 2009, 10, 895–926. [Google Scholar] [CrossRef] [PubMed]
- Gaskin, D.J.; Richard, P. The economic costs of pain in the United States. J. Pain 2012, 13, 715–724. [Google Scholar] [CrossRef] [PubMed]
- Weaver, K.R.; Griffioen, M.A.; Klinedinst, N.J.; Galik, E.; Duarte, A.C.; Colloca, L.; Resnick, B.; Dorsey, S.G.; Renn, C.L. Quantitative sensory testing across chronic pain conditions and use in special populations. Front. Pain Res. 2022, 2, 779068. [Google Scholar] [CrossRef] [PubMed]
- Rolke, R.; Baron, R.; Maier, C.; Tölle, T.R.; Treede, R.D.; Beyer, A.; Binder, A.; Birbaumer, N.; Birklein, F.; Bötefür, I.C.; et al. Quantitative sensory testing in the German Research Network on Neuropathic Pain (DFNS): Standardized protocol and reference values. Pain 2006, 123, 231–243. [Google Scholar] [CrossRef] [PubMed]
- Zambreanu, L.; Wise, R.G.; Brooks, J.C.W.; Iannetti, G.D.; Tracey, I. A role for the brainstem in central sensitisation in humans. Evidence from functional magnetic resonance imaging. Pain 2005, 114, 397–407. [Google Scholar] [CrossRef]
- Mayer, T.G.; Neblett, R.; Cohen, H.; Howard, K.J.; Choi, Y.H.; Williams, M.J.; Perez, Y.; Gatchel, R.J. The development and psychometric validation of the central sensitization inventory. Pain Pract. 2012, 12, 276–285. [Google Scholar] [CrossRef]
- Tanaka, K.; Nishigami, T.; Mibu, A.; Manfuku, M.; Yono, S.; Shinohara, Y.; Tanabe, A.; Ono, R. Validation of the Japanese version of the central sensitization inventory in patients with musculoskeletal disorders. PLoS ONE 2017, 12, e018871. [Google Scholar] [CrossRef]
- Scerbo, T.; Colasurdo, J.; Dunn, S.; Unger, J.; Nijs, J.; Cook, C. Measurement properties of the central sensitization inventory: A systematic review. Pain Pract. 2018, 18, 544–554. [Google Scholar] [CrossRef] [PubMed]
- Roussel, N.A.; Nijs, J.; Meeus, M.; Mylius, V.; Fayt, C.; Oostendorp, R. Central sensitization and altered central pain processing in chronic low back pain: Fact or myth? Clin. J. Pain 2013, 29, 625–638. [Google Scholar] [CrossRef] [PubMed]
- Aoyagi, K.; He, J.; Nicol, A.L.; Clauw, D.J.; Kluding, P.M.; Jernigan, S.; Sharma, N.K. A subgroup of chronic low back pain patients with central sensitization. Clin. J. Pain 2019, 35, 869–879. [Google Scholar] [CrossRef] [PubMed]
- Ohashi, Y.; Fukushima, K.; Inoue, G.; Uchida, K.; Koyama, T.; Tsuchiya, M.; Uchiyama, K.; Takahira, N.; Takaso, M. Central sensitization inventory scores correlate with pain at rest in patients with hip osteoarthritis: A retrospective study. BMC Musculoskelet. Disord. 2020, 21, 595. [Google Scholar] [CrossRef] [PubMed]
- Hattori, T.; Shimo, K.; Niwa, Y.; Tokiwa, Y.; Matsubara, T. Association of chronic pain with radiologic severity and central sensitization in hip osteoarthritis patients. J. Pain Res. 2021, 14, 1153–1160. [Google Scholar] [CrossRef] [PubMed]
- Bjurström, M.F.; Blennow, K.; Zetterberg, H.; Bodelsson, M.; Waldén, M.; Dietz, N.; Hall, S.; Hansson, O.; Irwin, M.R.; Mattsson-Carlgren, N. Central nervous system monoaminergic activity in hip osteoarthritis patients with disabling pain: Associations with pain severity and central sensitization. Pain Rep. 2022, 7, e988. [Google Scholar] [CrossRef]
- Fingleton, C.; Smart, K.; Moloney, N.; Fullen, B.M.; Doody, C. Pain sensitization in people with knee osteoarthritis: A systematic review and meta-analysis. Osteoarthr. Cartil. 2015, 23, 1043–1056. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.S.; Kim, J.J.; Kang, K.H.; Kim, M.J.; In, Y. Diagnosis of central sensitization and its effects on postoperative outcomes following total knee arthroplasty: A systematic review and meta-analysis. Diagnostics 2022, 12, 1248. [Google Scholar] [CrossRef]
- Koh, I.J.; Kang, B.M.; Kim, M.S.; Choi, K.Y.; Sohn, S.; In, Y. How does preoperative central sensitization affect quality of life following total knee arthroplasty? J. Arthroplast. 2020, 35, 2044–2049. [Google Scholar] [CrossRef]
- Ohashi, Y.; Fukushima, K.; Uchida, K.; Koyama, T.; Tsuchiya, M.; Saito, H.; Uchiyama, K.; Takahira, N.; Inoue, G.; Takaso, M. Adverse effects of higher preoperative pain at rest, a central sensitization-related symptom, on outcomes after total hip arthroplasty in patients with osteoarthritis. J. Pain Res. 2021, 14, 3345–3352. [Google Scholar] [CrossRef]
- Arendt-Nielsen, L.; Nie, H.; Laursen, M.B.; Laursen, B.S.; Madeleine, P.; Simonsen, O.H.; Graven-Nielsen, T. Sensitization in patients with painful knee osteoarthritis. Pain 2010, 149, 573–581. [Google Scholar] [CrossRef] [PubMed]
- Smith, M.T.; Remeniuk, B.; Finan, P.H.; Speed, T.J.; Tompkins, D.A.; Robinson, M.; Gonzalez, K.; Bjurstrom, M.F.; Irwin, M.R. Sex differences in measures of central sensitization and pain sensitivity to experimental sleep disruption: Implications for sex differences in chronic. Pain Sleep 2019, 42, zsy209. [Google Scholar] [CrossRef] [PubMed]
- Sanchis, M.N.; Lluch, E.; Nijs, J.; Struyf, F.; Kangasperko, M. The role of central sensitization in shoulder pain: A systematic literature review. Semin. Arthritis Rheum. 2015, 44, 710–716. [Google Scholar] [CrossRef] [PubMed]
- Bilika, P.; Nijs, J.; Fandridis, E.; Dimitriadis, Z.; Strimpakos, N.; Kapreli, E. In the shoulder or in the brain? Behavioral, psychosocial and cognitive characteristics of unilateral chronic shoulder pain with symptoms of central sensitization. Healthcare 2022, 10, 1658. [Google Scholar] [CrossRef]
- Mertens, M.G.; Struyf, F.; Lluch Girbes, E.; Dueñas, L.; Verborgt, O.; Meeus, M. Autonomic nervous system function and central pain processing in people with frozen shoulder: A case-control study. Clin. J. Pain 2022, 38, 659–669. [Google Scholar] [CrossRef] [PubMed]
- Neblett, R.; Hartzell, M.M.; Mayer, T.G.; Cohen, H.; Gatchel, R.J. Establishing clinically relevant severity levels for the central sensitization inventory. Pain Pract. 2017, 17, 166–175. [Google Scholar] [CrossRef]
- Nijs, J.; Torres-Cueco, R.; Paul Van Wilgen, C.; Girbés, E.L.; Struyf, F.; Roussel, N.; Van Oosterwijck, J.; Daenen, L.; Kuppens, K.; Vanderweeën, L.; et al. Applying modern pain neuroscience in clinical practice: Criteria for the classification of central sensitization Pain. Pain Physician 2014, 17, 447–457. [Google Scholar] [CrossRef] [PubMed]
- Woolf, C.J. Central sensitization: Implications for the diagnosis and treatment of Pain. Pain 2011, 152, S2–S15. [Google Scholar] [CrossRef]
- Fernández-Carnero, J.; Fernández-De-Las-Peñas, C.; De La Llave-Rincón, A.I.; Ge, H.Y.; Arendt-Nielsen, L. Widespread mechanical pain hypersensitivity as sign of central sensitization in unilateral epicondylalgia: A blinded, controlled study. Clin. J. Pain 2009, 25, 555–561. [Google Scholar] [CrossRef]
- Zanette, G.; Cacciatori, C.; Tamburin, S. Central sensitization in carpal tunnel syndrome with extraterritorial spread of sensory symptoms. Pain 2010, 148, 227–236. [Google Scholar] [CrossRef]
- Gwilym, S.E.; Oag, H.C.L.; Tracey, I.; Carr, A.J. Evidence that central sensitisation is present in patients with shoulder impingement syndrome and influences the outcome after surgery. J. Bone Jt. Surg. Br. 2011, 93, 498–502. [Google Scholar] [CrossRef] [PubMed]
- Koh, I.J.; Kim, M.S.; Sohn, S.; Song, K.Y.; Choi, N.Y.; In, Y. Duloxetine reduces pain and improves quality of recovery following total knee arthroplasty in centrally sensitized patients: A prospective, randomized controlled study. J. Bone Jt. Surg. Am. 2019, 101, 64–73. [Google Scholar] [CrossRef] [PubMed]
- Ohashi, Y.; Uchida, K.; Fukushima, K.; Inoue, G.; Takaso, M. Mechanisms of peripheral and central sensitization in osteoarthritis. Pain Cureus 2023, 15, e35331. [Google Scholar] [CrossRef] [PubMed]
- Dunn, W.R.; Kuhn, J.E.; Sanders, R.; An, Q.; Baumgarten, K.M.; Bishop, J.Y.; Brophy, R.H.; Carey, J.L.; Holloway, G.B.; Jones, G.L.; et al. Symptoms of pain do not correlate with rotator cuff tear severity: A cross-sectional study of 393 patients with a symptomatic atraumatic full-thickness rotator cuff tear. J. Bone Jt. Surg. Am. 2014, 96, 793–800. [Google Scholar] [CrossRef] [PubMed]
- Curry, E.J.; Matzkin, E.E.; Dong, Y.; Higgins, L.D.; Katz, J.N.; Jain, N.B. Structural characteristics are not associated with pain and function in rotator cuff tears: The ROW cohort study. Orthop. J. Sports Med. 2015, 3, 2325967115584596. [Google Scholar] [CrossRef] [PubMed]
- Nijs, J.; Malfliet, A.; Nishigami, T. Nociplastic pain and central sensitization in patients with chronic pain conditions: A terminology update for clinicians. Braz. J. Phys. Ther. 2023, 27, 100518. [Google Scholar] [CrossRef] [PubMed]
- Nijs, J.; Lahousse, A.; Kapreli, E.; Bilika, P.; Saraçoğlu, İ.; Malfliet, A.; Coppieters, I.; De Baets, L.; Leysen, L.; Roose, E.; et al. Nociplastic pain criteria or recognition of central sensitization? Pain phenotyping in the past, present and future. J. Clin. Med. 2021, 10, 3203. [Google Scholar] [CrossRef]
| All Patients (n = 167) | |
|---|---|
| Age (years) | 70.0 [63.0–75.0] |
| Sex—Male, n (%) | 84 (50.3) |
| Sex—Female, n (%) | 83 (49.7) |
| Affected side—Right, n (%) | 102 (61.1) |
| Affected side—Left, n (%) | 65 (38.9) |
| CSI | 18.0 [10.0–32.0] |
| CSI < 40, n (%) | 146 (87.4) |
| CSI ≥ 40, n (%) | 21 (12.6) |
| Pain VAS (mm) | 35.0 [11.0–64.0] |
| Pain duration (year) | 0.5 [0.3–1.2] |
| Active flexion (degrees) | 125.0 [80.0–150.0] |
| Active abduction (degrees) | 100.0 [60.0–152.5] |
| Active external rotation (degrees) | 35.0 [20.0–52.5] |
| Active internal rotation (Points, based on Constant score) | 6.0 [4.0–8.0] |
| Rotator cuff tear without osteoarthritis of GH joint, n (%) | 131 (78.4) |
| Osteoarthritis of GH joint with rotator cuff tear, n (%) | 23 (13.8) |
| Osteoarthritis of GH joint without rotator cuff tear, n (%) | 13 (7.8) |
| History of trauma, n (%) | 40 (24.0) |
| History of shoulder surgery | 20 (12.0) |
| CSI Score | n (%) |
|---|---|
| Subclinical (0–29) | 120 (71.6) |
| Mild (30–39) | 26 (15.6) |
| Moderate (40–49) | 14 (8.4) |
| Severe (50–59) | 5 (3.9) |
| Extreme (60–100) | 2 (1.2) |
| OA Patients (with RCT and without RCT) (n = 36) | Non-OA Patients (with RCT) (n = 131) | p-Value | |
|---|---|---|---|
| CSI | 27.5 [10.8–40.5] | 18.0 [10.0–27.5] | 0.043 * |
| CSI ≥ 40, n (%) | 10 (27.8) | 11 (8.4) | 0.004 * |
| Age (years) | 74.5 [68.8–79.0] | 69.0 [60.5–74.0] | 0.001 * |
| Sex—Male/Female, n | 15/21 | 69/62 | 0.26 |
| Affected side— Right/Left, n | 25/11 | 77/54 | 0.34 |
| Pain VAS (mm) | 32.5 [9.8–63.5] | 74.5 [13.0–62.5] | 0.52 |
| Pain duration (year) | 0.7 [0.3–2.0] | 0.5 [0.3–1.0] | 0.24 |
| OA Patients with RCT (n = 23) | OA Patients without RCT (n = 13) | p-Value | |
|---|---|---|---|
| CSI | 25.7 ± 16.9 | 31.2 ± 21.5 | 0.41 |
| CSI ≥ 40, n (%) | 6 (26.1) | 4 (30.8) | 1.0 |
| Age (years) | 69.0 [68.0–71.0] | 76.0 [73.5–79.0] | 0.007 * |
| Sex—Male/Female, n | 10/13 | 5/8 | 1.0 |
| Affected side— Right/Left, n | 16/7 | 9/4 | 1.0 |
| Pain VAS (mm) | 25.0 [9.5–59.0] | 40.0 [10.0–70.0] | 0.79 |
| Pain duration (year) | 1.0 [0.2–2.3] | 0.6 [0.4–1.3] | 0.54 |
| Partial-Thickness Tear (n = 19) | S and M Size Tear (n = 60) | L and Ma Size Tear (n = 52) | p-Value | |
|---|---|---|---|---|
| CSI | 25.1 ± 13.5 | 18.4 ± 11.2 | 19.7 ± 13.6 | 0.12 |
| CSI ≥ 40, n (%) | 4 (21.1) | 2 (3.3) | 5 (9.6) | 0.038 * |
| Age (years) | 62.0 [58.5–65.5] | 67.5 [55.8–73.0] | 72.5 [68.0–77.0] | <0.001 * |
| Sex— Male/Female, n | 10/9 | 30/30 | 29/23 | 0.83 |
| Affected side— Right/Left, n | 9/10 | 39/21 | 29/23 | 0.34 |
| Pain VAS (mm) | 40.0 [26.0–71.5] | 35.0 [14.3–59.3] | 35.5 [10.8–61.3] | 0.61 |
| Pain duration (year) | 0.4 [0.3–0.8] | 0.6 [0.3–1.0] | 0.4 [0.3–1.0] | 0.64 |
| Post hoc test (Bonferroni) regarding CSI ≥ 40 | ||||
| Partial-thickness tear and S and M size tear | Partial-thickness tear and L and Ma size tear | S and M size tear and L and Ma size tear | ||
| 0.080 | 0.71 | 0.74 | ||
| Post hoc test (Steel-Dwass test) regarding age | ||||
| Partial-thickness tear and S and M size tear | Partial-thickness tear and L and Ma size tear | S and M size tear and L and Ma size tear | ||
| 0.21 | <0.001 * | <0.001 * | ||
| Risk Factor | Coefficients (95% CI) | SE | T-Value | p-Value |
|---|---|---|---|---|
| Age (years) | 0.11 (−0.10–0.32) | 0.11 | 1.0 | 0.31 |
| Sex—Female | 5.6 (1.6–9.6) | 2.0 | 2.7 | 0.007 * |
| Pain VAS (mm) | 0.14 (0.066–0.22) | 0.038 | 3.7 | <0.001 * |
| Pain duration (year) | 0.90 (0.019–1.8) | 0.45 | 2.0 | 0.045 * |
| Active flexion (degrees) | −0.048 (−0.13–0.004) | 0.033 | −1.9 | 0.065 |
| Active external rotation (degrees) | −0.046 (−0.15–0.059) | 0.053 | −0.87 | 0.39 |
| Active internal rotation (Points, based on Constant score) | 0.33 (−0.67–1.3) | 0.50 | 0.65 | 0.52 |
| Rotator cuff tear | −1.4 (−11–8.7) | 5.1 | −0.27 | 0.79 |
| Osteoarthritis of GH joint | 1.5 (−5.2–8.2) | 3.8 | 0.45 | 0.65 |
| History of trauma | −2.2 (−7.0–2.5) | 2.4 | −0.93 | 0.35 |
| History of shoulder surgery | −3.3 (−11–4.7) | 4.0 | −0.82 | 0.41 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Iio, R.; Manaka, T.; Nakazawa, K.; Hirakawa, Y.; Ito, Y.; Ogura, A.; Nakamura, H. Assessment of Prevalence and Risk Factors for Central Sensitization Related to Shoulder Osteoarthritis and Rotator Cuff Tears Using the Central Sensitization Inventory: A Cross-Sectional Study in Shoulder Surgery Patients. J. Clin. Med. 2023, 12, 5633. https://doi.org/10.3390/jcm12175633
Iio R, Manaka T, Nakazawa K, Hirakawa Y, Ito Y, Ogura A, Nakamura H. Assessment of Prevalence and Risk Factors for Central Sensitization Related to Shoulder Osteoarthritis and Rotator Cuff Tears Using the Central Sensitization Inventory: A Cross-Sectional Study in Shoulder Surgery Patients. Journal of Clinical Medicine. 2023; 12(17):5633. https://doi.org/10.3390/jcm12175633
Chicago/Turabian StyleIio, Ryosuke, Tomoya Manaka, Katsumasa Nakazawa, Yoshihiro Hirakawa, Yoichi Ito, Ayako Ogura, and Hiroaki Nakamura. 2023. "Assessment of Prevalence and Risk Factors for Central Sensitization Related to Shoulder Osteoarthritis and Rotator Cuff Tears Using the Central Sensitization Inventory: A Cross-Sectional Study in Shoulder Surgery Patients" Journal of Clinical Medicine 12, no. 17: 5633. https://doi.org/10.3390/jcm12175633






