Extended Distal Pancreatectomy for Cancer of the Body and Tail of the Pancreas: Analysis of Early and Late Results
Abstract
:1. Introduction
2. Materials and Methods
3. Results
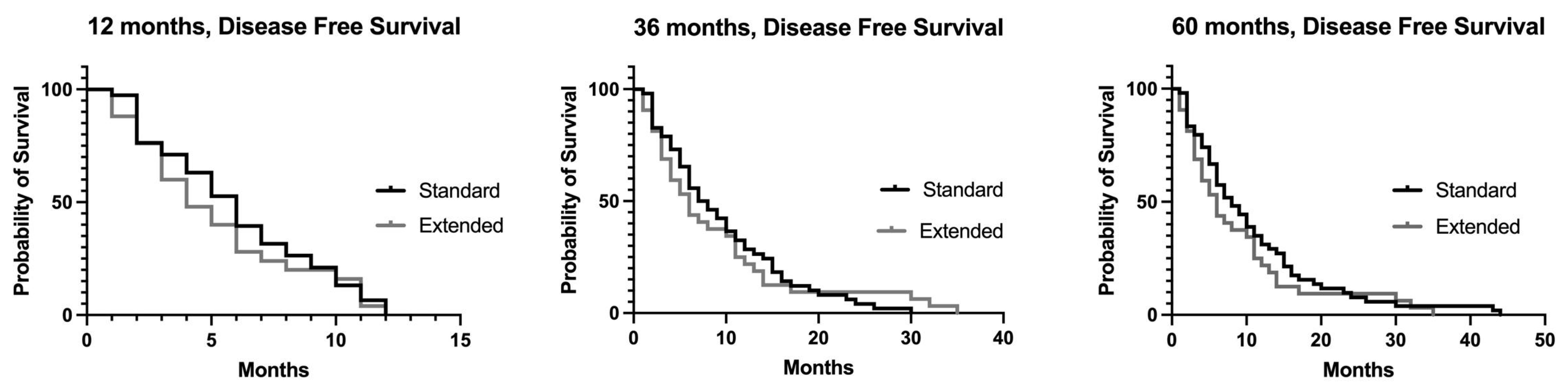
3.1. Recurrence and Disease-Free Survival
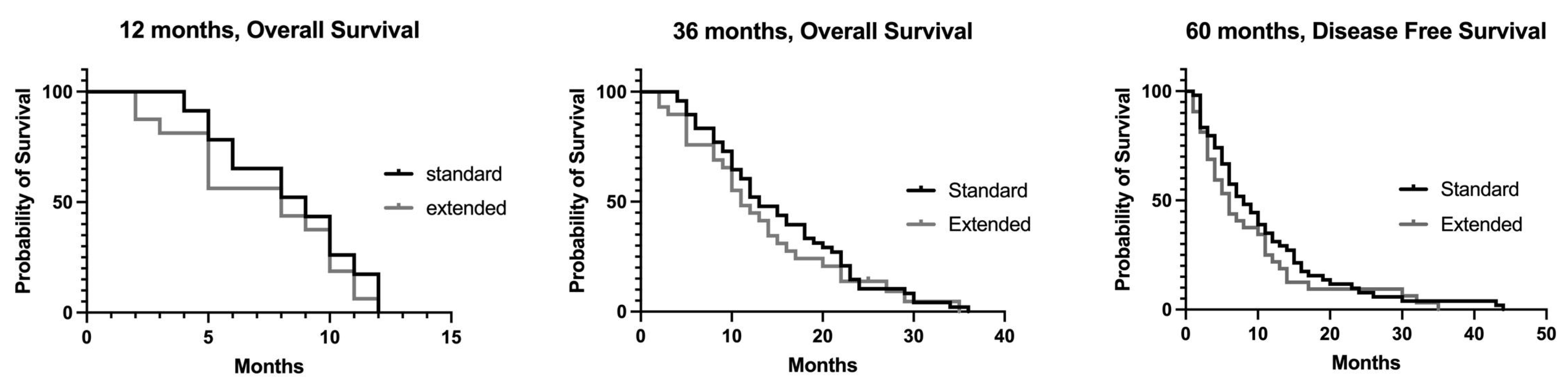
3.2. Overall Survival
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Artinyan, A.; Soriano, P.A.; Prendergast, C.; Low, T.; Ellenhorn, J.D.; Kim, J. The anatomic location of pancreatic cancer is a prognostic factor for survival. HPB 2008, 10, 371–376. [Google Scholar] [CrossRef] [PubMed]
- Van Erning, F.N.; Mackay, T.M.; van der Geest, L.G.M.; Groot Koerkamp, B.; van Laarhoven, H.W.M.; Bonsing, B.A.; Wilmink, J.W.; van Santvoort, H.C.; de Vos-Geelen, J.; van Eijck, C.H.J.; et al. Association of the location of pancreatic ductal adenocarcinoma (head, body, tail) with tumor stage, treatment, and survival: A population-based analysis. Acta Oncol. 2018, 57, 1655–1662. [Google Scholar] [CrossRef] [PubMed]
- Ruess, D.A.; Makowiec, F.; Chikhladze, S.; Sick, O.; Riediger, H.; Hopt, U.T.; Wittel, U.A. The prognostic influence of intrapancreatic tumor location on survival after resection of pancreatic ductal adenocarcinoma. BMC Surg. 2015, 15, 123. [Google Scholar] [CrossRef] [PubMed]
- Tessman, D.; Chou, J.; Shebrain, S.; Munene, G. Surgical outcome of distal pancreatectomy in elderly patients. Am. Surg. 2022, 88, 115–119. [Google Scholar] [CrossRef] [PubMed]
- Paye, F.; Micelli Lupinacci, R.; Bachellier, P.; Boher, J.M.; Delpero, J.R.; French Surgical Association (AFC). Distal pancreatectomy for pancreatic carcinoma in the era of multimodal treatment. Br. J. Surg. 2015, 102, 229–236. [Google Scholar] [CrossRef]
- Probst, P.; Hüttner, F.J.; Meydan, Ö.; Abu Hilal, M.; Adham, M.; Barreto, S.G.; Besselink, M.G.; Busch, O.R.; Bockhorn, M.; Del Chiaro, M.; et al. Evidence Map of Pancreatic Surgery—A living systematic review with meta-analyses by the International Study Group of Pancreatic Surgery (ISGPS). Surgery 2021, 170, 1517–1524. [Google Scholar] [CrossRef] [PubMed]
- Tonini, V.; Zanni, M. Pancreatic cancer in 2021: What you need to know to win. World J. Gastroenterol. 2021, 27, 5851–5889. [Google Scholar] [CrossRef]
- Hartwig, W.; Vollmer, C.M.; Fingerhut, A.; Yeo, C.J.; Neoptolemos, J.P.; Adham, M.; Andrén-Sandberg, A.; Asbun, H.J.; Bassi, C.; Bockhorn, M.; et al. Extended pancreatectomy in pancreatic ductal adenocarcinoma: Definition and consensus of the International Study Group for Pancreatic Surgery (ISGPS). Surgery 2014, 156, 1–14. [Google Scholar] [CrossRef]
- Kaiser, J.; Hackert, T.; Büchler, M.W. Extended Pancreatectomy: Does It Have a Role in the Contemporary Management of Pancreatic Adenocarcinoma? Dig Surg. 2017, 34, 441–446. [Google Scholar] [CrossRef]
- Kawarada, Y. New classification of pancreatic carcinoma—Japan Pancreas Society. Nihon Shokakibyo Gakkai Zasshi 2003, 100, 974–980. [Google Scholar]
- Menon, K.V.; Gomez, D.; Smith, A.M.; Anthoney, A.; Verbeke, C.S. Impact of margin status on survival following pancreatoduodenectomy for cancer: The Leeds Pathology Protocol (LEEPP). HPB 2009, 11, 18–24. [Google Scholar] [CrossRef] [PubMed]
- Brierley, J.D.; Gospodarowiez, M.K.; Wittekind, C. TNM Classification of Malignant Tumors, 8th ed.; UICC: Geneva, Switzerland, 2017. [Google Scholar]
- Dindo, D.; Demartines, N.; Clavien, P.A. Classification of surgical complications: A new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann. Surg. 2004, 240, 205–213. [Google Scholar] [CrossRef] [PubMed]
- De Oliveira, M.L.; Winter, J.M.; Schafer, M.; Cunningham, S.C.; Cameron, J.L.; Yeo, C.J.; Clavien, P.A. Assessment of complications after pancreatic surgery: A novel grading system applied to 633 patients undergoing pancreaticoduodenectomy. Ann. Surg. 2006, 244, 931–937. [Google Scholar] [CrossRef] [PubMed]
- Bassi, C.; Dervenis, C.; Butturini, G.; Fingerhut, A.; Yeo, C.; Izbicki, J.; Neoptolemos, J.; Sarr, M.; Traverso, W.; Buchler, M.; et al. Postoperative pancreatic fistula: An international study group (ISGPF) definition. Surgery 2005, 138, 8–13. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Z.; Wang, M.; Tan, C.; Chen, Y.; Ping, J.; Wang, R.; Liu, X. Disparities in survival by stage after surgery between pancreatic head and body/tail in patients with nonmetastatic pancreatic cancer. PLoS ONE 2019, 14, e0226726. [Google Scholar] [CrossRef] [PubMed]
- Birnbaum, D.J.; Bertucci, F.; Finetti, P.; Birnbaum, D.; Mamessier, E. Head and Body/Tail Pancreatic Carcinomas Are Not the Same Tumors. Cancers 2019, 11, 497. [Google Scholar] [CrossRef] [PubMed]
- Goh, B.K.; Tan, Y.M.; Cheow, P.C.; Chung, Y.F.; Chow, P.K.; Wong, W.K.; Ooi, L.L. Outcome of distal pancreatectomy for pancreatic adenocarcinoma. Dig. Surg. 2008, 25, 32–38. [Google Scholar] [CrossRef]
- Irani, J.L.; Ashley, S.W.; Brooks, D.C.; Osteen, R.T.; Raut, C.P.; Russell, S.; Swanson, R.S.; Whang, E.E.; Zinner, M.J.; Clancy, T.E. Distal pancreatectomy is not associated with increased perioperative morbidity when performed as part of a multivisceral resection. J. Gastrointest. Surg. 2008, 12, 2177–2182. [Google Scholar] [CrossRef]
- Bergenfeldt, M.; Moesgaard, F.; Burcharth, F. Curative resection for left-sided pancreatic malignancy. HPB 2006, 8, 211–215. [Google Scholar] [CrossRef]
- Malinka, T.; Klein, F.; Andreou, A.; Pratschke, J.; Bahra, M. Distal Pancreatectomy Combined with Multivisceral Resection Is Associated with Postoperative Complication Rates and Survival Comparable to Those After Standard Procedures. J. Gastrointest. Surg. 2018, 22, 1549–1556. [Google Scholar] [CrossRef]
- Panzeri, F.; Marchegiani, G.; Malleo, G.; Malpaga, A.; Maggino, L.; Marchese, T.; Salvia, R.; Bassi, C.; Butturini, G. Distal pancreatectomy associated with multivisceral resection: Results from a single centre experience. Langenbecks. Arch. Surg. 2017, 402, 457–464. [Google Scholar] [CrossRef] [PubMed]
- Burdelski, C.M.; Reeh, M.; Bogoevski, D.; Gebauer, F.; Tachezy, M.; Vashist, Y.K.; Cataldegirmen, G.; Yekebas, E.; Izbicki, J.R.; Bockhorn, M. Multivisceral resections in pancreatic cancer: Identification of risk factors. World J. Surg. 2011, 35, 2756–2763. [Google Scholar] [CrossRef] [PubMed]
- Ramia, J.M.; Del Río-Martín, J.V.; Blanco-Fernández, G.; Cantalejo-Díaz, M.; Rotellar-Sastre, F.; Sabater-Orti, L.; Carabias-Hernandez, A.; Manuel-Vázquez, A.; Hernández-Rivera, P.J.; Jaén-Torrejimeno, I.; et al. Distal pancreatectomy with multivisceral resection: A retrospective multicenter study—Case series. Int. J. Surg. 2020, 82, 123–129. [Google Scholar] [CrossRef] [PubMed]
- Siripong, A.; Chung, M.; Rich, S.E. Multivisceral Pancreatic Resections: Worth the Risk? Clin. Surg. 2018, 3, 1869. [Google Scholar]
- Beetz, O.; Sarisin, A.; Kaltenborn, A.; Klempnauer, J.; Winkler, M.; Grannas, G. Multivisceral resection for adenocarcinoma of the pancreatic body and tail-a retrospective single-center analysis. World J. Surg. Oncol. 2020, 18, 218. [Google Scholar] [CrossRef] [PubMed]
- Tangtawee, P.; Mingphruedhi, S.; Rungsakulkij, N.; Suragul, W.; Vassanasiri, W.; Muangkaew, P. Comparative outcomes of extended distal pancreatectomy and distal pancreatectomy. Asian J. Surg. 2020, in press. [Google Scholar] [CrossRef] [PubMed]
- Song, K.B.; Kwon, J.; Kim, Y.W.; Hwang, D.W.; Lee, J.H.; Hong, S.; Lee, J.W.; Hwang, K.; Yoo, D.; Kim, S.C. Prognostic value of adjacent organ resection in patients with left-sided pancreatic ductal adenocarcinoma following distal pancreatectomy. J. Hepatobiliary Pancreat. Sci. 2019, 26, 227–234. [Google Scholar] [CrossRef] [PubMed]
- Christein, J.D.; Kendrick, M.L.; Iqbal, C.W.; Nagorney, D.M.; Farnell, M.B. Distal pancreatectomy for resectable adenocarcinoma of the body and tail of the pancreas. J. Gastrointest. Surg. 2005, 9, 922–927. [Google Scholar] [CrossRef]
- Shoup, M.; Conlon, K.C.; Klimstra, D.; Brennan, M.F. Is extended resection for adenocarcinoma of the body or tail of the pancreas justified? J. Gastrointest. Surg. 2003, 7, 946–952. [Google Scholar] [CrossRef]
- Roch, A.M.; Singh, H.; Turner, A.P.; Ceppa, E.P.; House, M.G.; Zyromski, N.J.; Nakeeb, A.; Schmidt, C.M. Extended distal pancreatectomy for pancreatic adenocarcinoma with splenic vein thrombosis and/or adjacent organ invasion. Am. J. Surg. 2015, 209, 564–569. [Google Scholar] [CrossRef]
- Hartwig, W.; Hackert, T.; Hinz, U.; Hassenpflug, M.; Strobel, O.; Büchler, M.W.; Werner, J. Multivisceral resection for pancreatic malignancies: Risk-analysis and long-term outcome. Ann. Surg. 2009, 250, 81–87. [Google Scholar] [CrossRef]
- Chandrashekhar, S.H.; Ismail, S.; Jonnada, P.K.; Senhadipan, B.; Karunakaran, M.; Barreto, S.G. Extended distal pancreatectomy in pancreatic cancer. Is it justified? A systematic review of literature. Future Oncol. 2023. ahead of print. [Google Scholar] [CrossRef]
- Loos, M.; Khajeh, E.; Mehrabi, A.; Kinny-koster, B.; Al-Seedi, M.; Berchtold, C.; Hoffmann, K.; Schneider, M.; Eslami, P.; Feisst, M.; et al. Distal pancreatectomy with en-bolc celiac axis resection (DP-CAR) for locally advanced pancreatic cancer: A safe and effective procedure. Ann. Surg. 2023. ahead of print. [Google Scholar] [CrossRef]
- Yoon, S.J.; Park, S.-J.; Yoon, Y.-S.; Hong, T.-H.; Jang, J.-Y.; Kim, H.J.; Heo, J.S.; Hwang, D.W.; Han, I.W. 15-year experience of distal pancreatectomy with celiac axis resection (DP-CAR) for pancreatic cancer-a Korean nationwide investigation. Cancers 2023, 15, 3850. [Google Scholar] [CrossRef]
- Sakamoto, T.; Gocho, T.; Tsunematsu, M.; Shirai, Y.; Hamura, R.; Haruki, K.; Abe, K.; Okamoto, T.; Shiozaki, H.; Fujioka, S.; et al. D1 distal pancreatectomy for left-sided pancreatic ductal adenocarcinoma is justifiable: A propensity-score matched multicenter study. Anticancer Res. 2023, 43, 201–208. [Google Scholar] [CrossRef]


| Variables | ||
|---|---|---|
| Age, years, median (range) | 69 (41–87) | |
| Sex, n (%) | Male | 48 (48) |
| Female | 53 (52) | |
| Lymph node, n (%) | Negative | 34 (34) |
| Positive | 67 (66) | |
| Grading, n (%) | Low Grade | 58 (57) |
| High Grade | 43 (43) | |
| Stage, n (%) | I–II | 70 (69) |
| III–IV | 31 (31) | |
| Extended, n (%) | No | 65 (63) |
| Yes | 36 (37) | |
| R0 resection, n (%) | Yes | 59 (58) |
| No | 42 (42) |
| Variables | Extended DP | Standard DP | p-Value | |
|---|---|---|---|---|
| Age, years, median (range) | 69 (41–83) | 67.5 (45–87) | 0.951 | |
| Sex, n (%) | Male | 16 (45.7) | 27 (45) | 0.947 |
| Female | 19 (54.3) | 33 (55) | ||
| Lymph node, n (%) | Negative | 12 (34.3) | 19 (31.7) | 0.795 |
| Positive | 23 (65.7) | 41 (67.3) | ||
| Grading, n (%) | Low Grade | 22 (62.9) | 31 (51.7) | 0.294 |
| High Grade | 13 (37.1) | 29 (48.3) | ||
| Stage, n (%) | I–II | 13 (37.1) | 53 (88.3) | <0.001 |
| III–IV | 22 (62.9) | 7 (11.7) | ||
| R0 Resection, n (%) | Yes | 18 (51.4) | 38 (63.3) | 0.260 |
| No | 17 (48.6) | 22 (36.7) | ||
| CA 19.9, n (%) | <37 U/L | 25 (69) | 40 (62) | 0.630 |
| >37 U/L | 10 (31) | 20 (38) | ||
| CA 19.9, U/L median (range) | 254 (50–1884) | 420 (80–1546) | 0.096 | |
| Morbidity, n (%) | 12 (33) | 19 (29) | 0.793 | |
| Mortality, n (%) | 1 (2.9) | 1 (1.7) | 0.697 | |
| Adjuvant CT, n (%) | 28 (78) | 44 (68) | 0.464 | |
| Neoadjuvant CT, n (%) | 4 (11) | 2 (3) | 0.118 |
| Extended DP | Standard DP | p-Value | |
|---|---|---|---|
| POPF, n | 5 (14) | 7 (11) | 0.711 |
| Abdominal collection, n (%) | 5 (14) | 5 (8) | 0.362 |
| Bleeding, n (%) | 1 (3) | 5 (8) | 0.290 |
| Colonic fistula, n (%) | 1 (3) | 1 (1.5) | 0.697 |
| Liver abscess, n (%) | 0 | 1 (1.5) | 0.443 |
| DVT, n (%) | 0 | 1 (1.5) | 0.443 |
| Pneumonia, n (%) | 0 | 1 (1.5) | 0.443 |
| DGE, n (%) | 1 (3) | 0 | 0.194 |
| MI, n (%) | 1 (3) | 0 | 0.194 |
| Re-operation, n (%) | 4 (11) | 4 (6) | 0.420 |
| Site | Extended DP | Standard DP | p-Value |
|---|---|---|---|
| Liver, n (%) | 14 (39) | 36 (55) | 0.060 |
| Local, n (%) | 10 (28) | 12 (18) | 0.339 |
| Peritoneum, n (%) | 12 (33) | 13 (20) | 0.178 |
| Lung, n (%) | 3 (8) | 4 (6) | 0.732 |
| Other, n (%) | 0 | 5 (8) | 0.079 |
| No recurrence, n (%) | 4 (11) | 4 (6) | 0.420 |
| Variables | HR (Univariate) | HR (Multivariate) |
|---|---|---|
| Age > 70 years | 0.87 (0.57–1.33, p = 0.527) | 0.77 (0.48–1.22, p = 0.261) |
| Sex | 1.08 (0.71–1.64, p = 0.722) | 0.91 (0.58–1.42, p = 0.672) |
| Lymph node | 1.88 (1.17–3.01, p = 0.009) | 1.51 (0.90–2.52, p = 0.118) |
| Grading | 1.60 (1.04–2.45, p = 0.031) | 1.46 (0.93–2.29, p = 0.099) |
| Stage | 2.99 (1.85–4.85, p < 0.001) | 1.94 (1.04–3.62, p = 0.037) |
| Radicality | 3.08 (1.92–4.95, p < 0.001) | 2.32 (1.35–3.99, p = 0.002) |
| Extended | 1.25 (0.81–1.93, p = 0.309) | 1.14 (0.66–1.98, p = 0.630) |
| Adjuvant Chemotherapy | 1.16 (0.75–1.81, p = 0.507) | 0.72 (0.43–1.20, p = 0.205) |
| Vascular resection | 1.64 (0.86–3.13, p = 0.132) | 0.83 (0.40–1.75, p = 0.627) |
| Variables | HR (Univariate) | HR (Multivariate) |
|---|---|---|
| Age > 70 years | 0.94 (0.62–1.42, p = 0.762) | 0.87 (0.56–1.37, p = 0.549) |
| Sex | 1.20 (0.79–1.82, p = 0.393) | 1.05 (0.68–1.63, p = 0.814) |
| Lymph node | 1.62 (1.02–2.56, p = 0.039) | 1.11 (0.67–1.82, p = 0.690) |
| Grading | 1.47 (0.96–2.25, p = 0.077) | 1.37 (0.88–2.13, p = 0.166) |
| Stage | 2.96 (1.84–4.76, p < 0.001) | 2.32 (1.21–4.45, p = 0.011) |
| Radicality | 3.50 (2.15–5.71, p < 0.001) | 2.57 (1.49–4.44, p = 0.001) |
| Extended | 1.11 (0.72–1.72, p = 0.631) | 0.75 (0.43–1.31, p = 0.315) |
| Adjuvant Chemotherapy | 1.23 (0.79–1.92, p = 0.348) | 1.01 (0.63–1.63, p = 0.960) |
| Vascular resection | 2.11 (1.09–4.05, p = 0.026) | 1.32 (0.62–2.80, p = 0.469) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sperti, C.; Serafini, S.; Friziero, A.; Todisco, M.; Tamponi, G.; Bassi, D.; Belluzzi, A. Extended Distal Pancreatectomy for Cancer of the Body and Tail of the Pancreas: Analysis of Early and Late Results. J. Clin. Med. 2023, 12, 5858. https://doi.org/10.3390/jcm12185858
Sperti C, Serafini S, Friziero A, Todisco M, Tamponi G, Bassi D, Belluzzi A. Extended Distal Pancreatectomy for Cancer of the Body and Tail of the Pancreas: Analysis of Early and Late Results. Journal of Clinical Medicine. 2023; 12(18):5858. https://doi.org/10.3390/jcm12185858
Chicago/Turabian StyleSperti, Cosimo, Simone Serafini, Alberto Friziero, Matteo Todisco, Giulia Tamponi, Domenico Bassi, and Amanda Belluzzi. 2023. "Extended Distal Pancreatectomy for Cancer of the Body and Tail of the Pancreas: Analysis of Early and Late Results" Journal of Clinical Medicine 12, no. 18: 5858. https://doi.org/10.3390/jcm12185858
APA StyleSperti, C., Serafini, S., Friziero, A., Todisco, M., Tamponi, G., Bassi, D., & Belluzzi, A. (2023). Extended Distal Pancreatectomy for Cancer of the Body and Tail of the Pancreas: Analysis of Early and Late Results. Journal of Clinical Medicine, 12(18), 5858. https://doi.org/10.3390/jcm12185858





