Evidence-Based Analysis of the Critical Steps of Radical Cystectomy for Bladder Cancer
Abstract
:1. Introduction
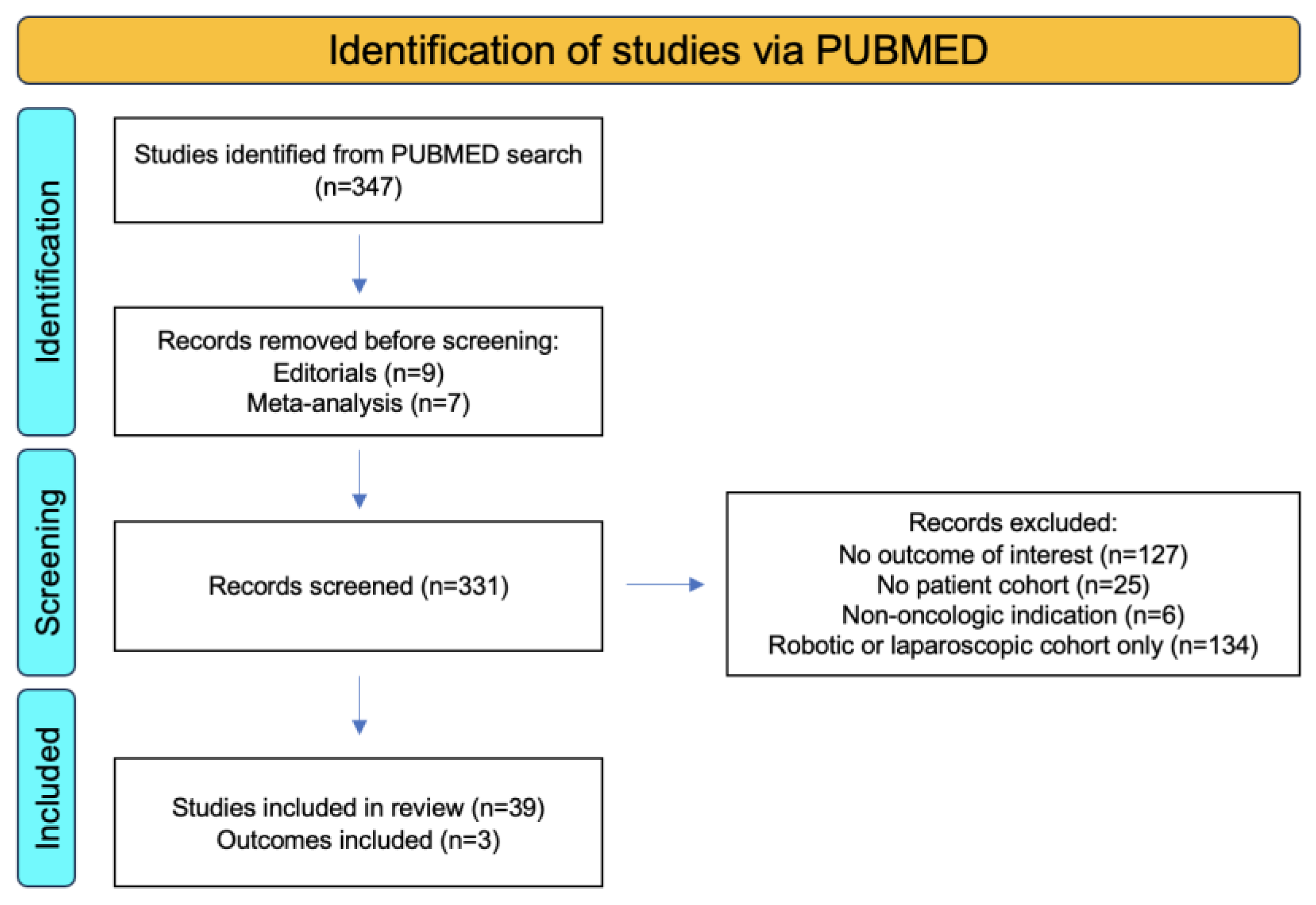
2. Materials and Methods
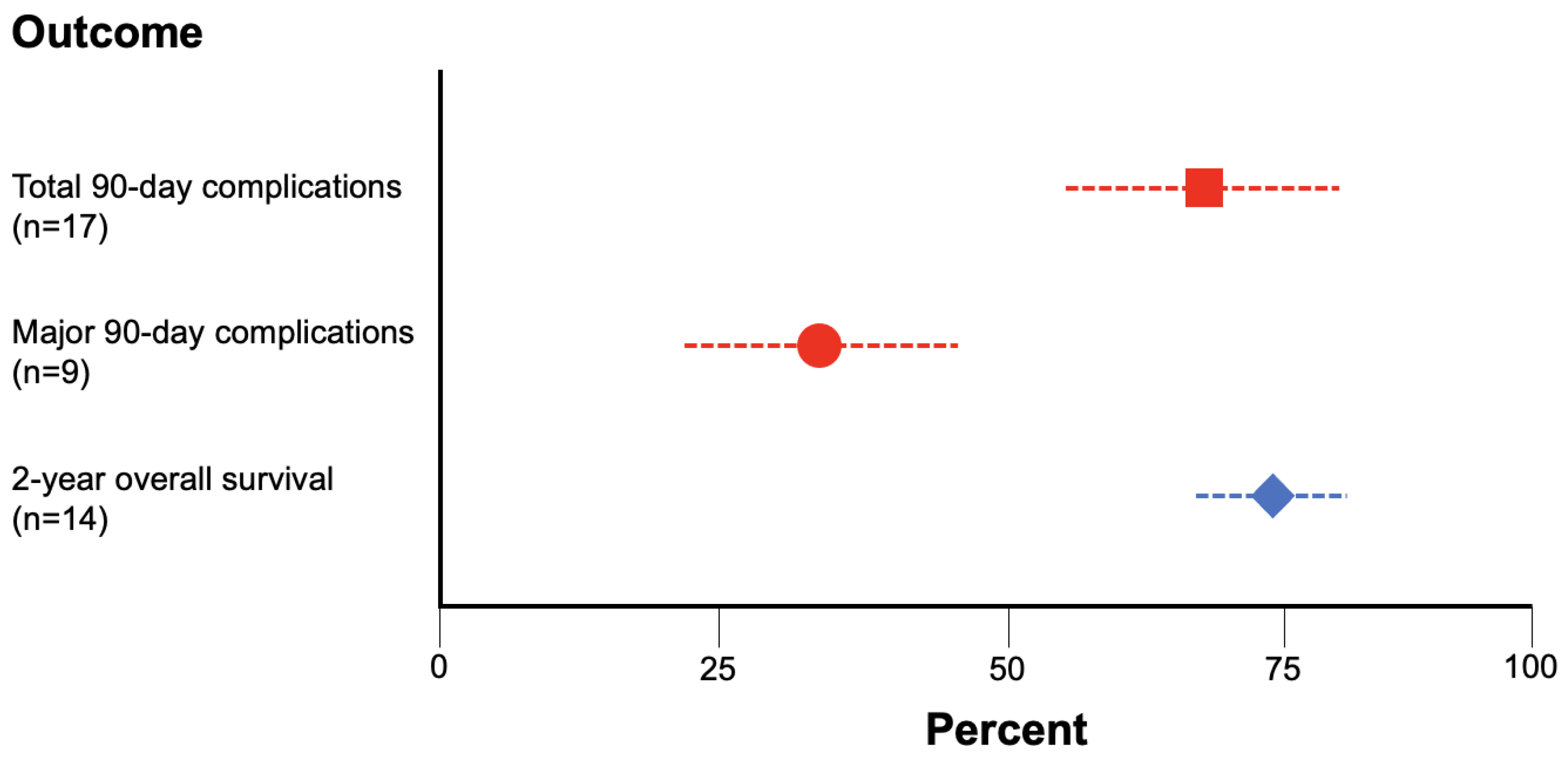
3. Results
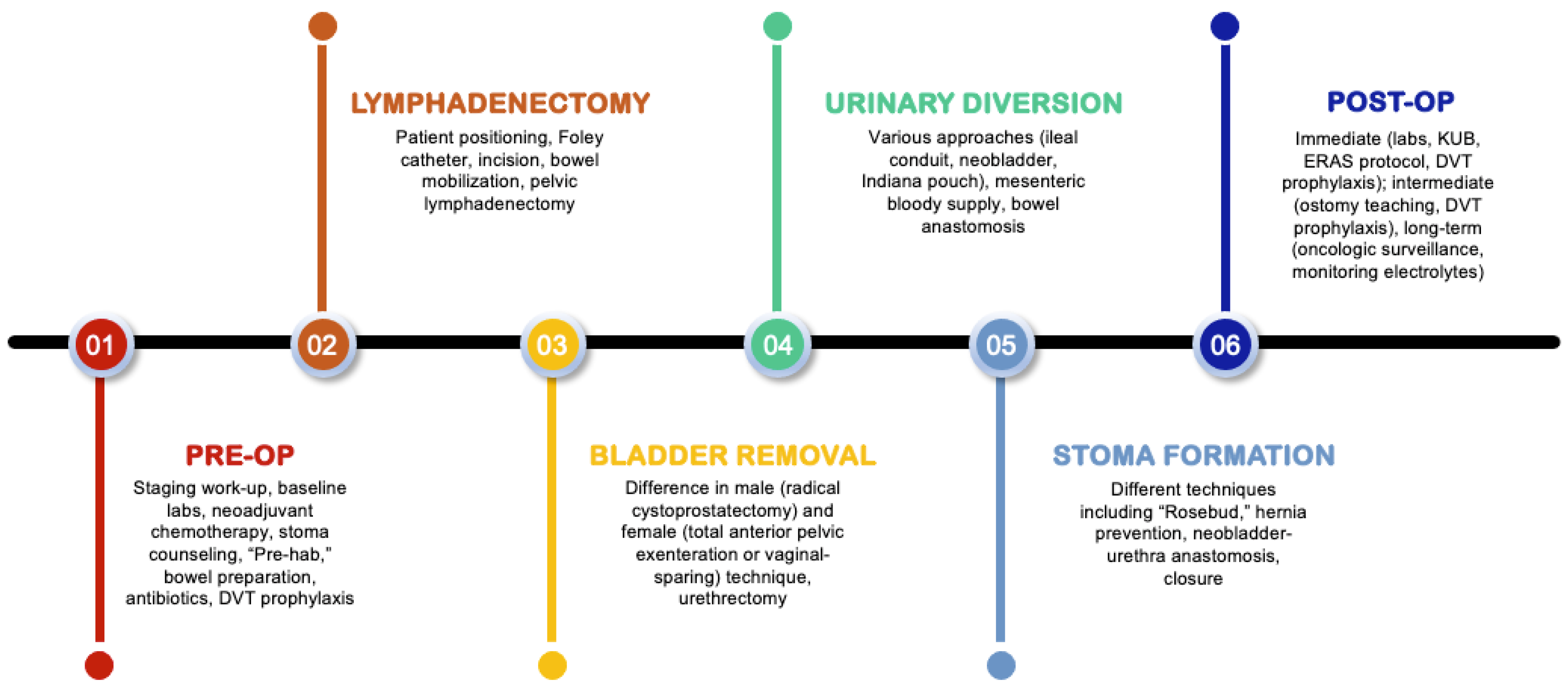
3.1. Preoperative Considerations
3.2. Pelvic Lymphadenectomy
3.3. Bladder Removal
3.4. Urinary Diversion
3.5. Stoma Formation
3.6. Postoperative Considerations
4. Discussion
5. Conclusions
Funding
Data Availability Statement
Conflicts of Interest
References
- Bray, F.; Ferlay, J.; Soerjomataram, I.; Siegel, R.L.; Torre, L.A.; Jemal, A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2018, 68, 394–424. [Google Scholar] [CrossRef]
- Siegel, R.L.; Miller, K.D.; Wagle, N.S.; Jemal, A. Cancer statistics, 2023. CA Cancer J. Clin. 2023, 73, 17–48. [Google Scholar] [CrossRef] [PubMed]
- Jubber, I.; Ong, S.; Bukavina, L.; Black, P.C.; Compérat, E.; Kamat, A.M.; Kiemeney, L.; Lawrentschuk, N.; Lerner, S.P.; Meeks, J.J.; et al. Epidemiology of Bladder Cancer in 2023: A Systematic Review of Risk Factors. Eur. Urol. 2023, 84, 176–190. [Google Scholar] [CrossRef] [PubMed]
- Bilano, V.; Gilmour, S.; Moffiet, T. Global trends and projections for tobacco use, 1990–2025: An analysis of smoking indicators from the WHO Comprehensive Information Systems for Tobacco Control. Lancet 2015, 385, 966–976. [Google Scholar] [CrossRef] [PubMed]
- Cumberbatch, M.G.; Cox, A.; Teare, D.; Catto, J.W. Contemporary occupational carcinogen exposure and bladder cancer: A systematic review and meta-analysis. JAMA Oncol. 2015, 1, 1282–1290. [Google Scholar] [CrossRef] [PubMed]
- Sanli, O.; Dobruch, J.; Knowles, M.A.; Burger, M.; Alemozaffar, M.; Nielsen, M.E.; Lotan, Y. Bladder cancer. Nat. Rev. Dis. Primers 2017, 3, 17022. [Google Scholar] [CrossRef] [PubMed]
- Agency for Healthcare Research and Quality (AHRQ). Healthcare Cost and Utilization Project (HCUP). 2017. Available online: https://www.ahrq.gov/research/data/hcup/index.html (accessed on 27 September 2023).
- Vetterlein, M.W.; Klemm, J.; Gild, P.; Bradtke, M.; Soave, A.; Dahlem, R.; Fisch, M.; Rink, M. Improving Estimates of Perioperative Morbidity After Radical Cystectomy Using the European Association of Urology Quality Criteria for Standardized Reporting and Introducing the Comprehensive Complication Index. Eur. Urol. 2020, 77, 55–65. [Google Scholar] [CrossRef] [PubMed]
- Nix, J.; Smith, A.; Kurpad, R.; Nielsen, M.E.; Wallen, E.M.; Pruthi, R.S. Prospective randomized controlled trial of robotic versus open radical cystectomy for bladder cancer: Perioperative and pathologic results. Eur. Urol. 2010, 57, 196–201. [Google Scholar] [CrossRef] [PubMed]
- Bochner, B.H.; Dalbagni, G.; Sjoberg, D.D.; Silberstein, J.; Keren Paz, G.E.; Donat, S.M.; Coleman, J.A.; Mathew, S.; Vickers, A.; Schnorr, G.C.; et al. Comparing Open Radical Cystectomy and Robot-assisted Laparoscopic Radical Cystectomy: A Randomized Clinical Trial. Eur. Urol. 2015, 67, 1042–1050. [Google Scholar] [CrossRef]
- Khan, M.S.; Gan, C.; Ahmed, K.; Ismail, A.F.; Watkins, J.; Summers, J.A.; Peacock, J.L.; Rimington, P.; Dasgupta, P. A Single-centre Early Phase Randomised Controlled Three-arm Trial of Open, Robotic, and Laparoscopic Radical Cystectomy (CORAL). Eur. Urol. 2016, 69, 613–621. [Google Scholar] [CrossRef]
- Bochner, B.H.; Dalbagni, G.; Marzouk, K.H.; Sjoberg, D.D.; Lee, J.; Donat, S.M.; Coleman, J.A.; Vickers, A.; Herr, H.W.; Laudone, V.P. Randomized Trial Comparing Open Radical Cystectomy and Robot-assisted Laparoscopic Radical Cystectomy: Oncologic Outcomes. Eur. Urol. 2018, 74, 465–471. [Google Scholar] [CrossRef] [PubMed]
- Parekh, D.J.; Reis, I.M.; Castle, E.P.; Gonzalgo, M.L.; Woods, M.E.; Svatek, R.S.; Weizer, A.Z.; Konety, B.R.; Tollefson, M.; Krupski, T.L.; et al. Robot-assisted radical cystectomy versus open radical cystectomy in patients with bladder cancer (RAZOR): An open-label, randomised, phase 3, non-inferiority trial. Lancet 2018, 391, 2525–2536. [Google Scholar] [CrossRef] [PubMed]
- Khan, M.S.; Omar, K.; Ahmed, K.; Gan, C.; Van Hemelrijck, M.; Nair, R.; Thurairaja, R.; Rimington, P.; Dasgupta, P. Long-term Oncological Outcomes from an Early Phase Randomised Controlled Three-arm Trial of Open, Robotic, and Laparoscopic Radical Cystectomy (CORAL). Eur. Urol. 2020, 77, 110–118. [Google Scholar] [CrossRef] [PubMed]
- Catto, J.W.F.; Khetrapal, P.; Ricciardi, F. Effect of Robot-Assisted Radical Cystectomy with Intracorporeal Urinary Diversion vs. Open Radical Cystectomy on 90-Day Morbidity and Mortality Among Patients with Bladder Cancer: A Randomized Clinical Trial. JAMA 2022, 327, 2092–2103. [Google Scholar] [CrossRef] [PubMed]
- Korkes, F.; Timóteo, F.; Martins, S.; Nascimento, M.; Monteiro, C.; Santiago, J.H.; Baccaglini, W.; Silveira, M.A.; Pedroso, E.F.; Gava, M.M.; et al. Dramatic Impact of Centralization and a Multidisciplinary Bladder Cancer Program in Reducing Mortality: The CABEM Project. JCO Glob. Oncol. 2021, 7, 1547–1555. [Google Scholar] [CrossRef] [PubMed]
- Leow, J.J.; Reese, S.; Trinh, Q.D.; Bellmunt, J.; Chung, B.I.; Kibel, A.S.; Chang, S.L. Impact of surgeon volume on the morbidity and costs of radical cystectomy in the USA: A contemporary population-based analysis. BJU Int. 2015, 115, 713–721. [Google Scholar] [CrossRef] [PubMed]
- Smelser, W.W.; Tallman, J.E.; Gupta, V.K.; Al Hussein Al Awamlh, B.; Johnsen, N.V.; Barocas, D.A.; Kline-Quiroz, C.; Tomlinson, C.A.; McEvoy, M.D.; Hamilton-Reeves, J.; et al. Implementation of a comprehensive prehabilitation program for patients undergoing radical cystectomy. Urol. Oncol. 2023, 41, 108. [Google Scholar] [CrossRef]
- Minnella, E.M.; Awasthi, R.; Bousquet-Dion, G.; Ferreira, V.; Austin, B.; Audi, C.; Tanguay, S.; Aprikian, A.; Carli, F.; Kassouf, W. Multimodal Prehabilitation to Enhance Functional Capacity Following Radical Cystectomy: A Randomized Controlled Trial. Eur. Urol. Focus 2021, 7, 132–138. [Google Scholar] [CrossRef]
- Navai, N.; Dinney, C. Surgical Management of Bladder Cancer: Transurethral, Open, and Robotic. Chapter 138. In Campbell Walsh Wein Urology, 12th ed.; Elsevier: Amsterdam, The Netherlands, 2021; pp. 3133–3159. [Google Scholar]
- Herr, H.W. Surgical factors influence bladder cancer outcomes: A cooperative group report. J. Clin. Oncol. 2004, 22, 2781–2789. [Google Scholar] [CrossRef]
- Wang, Y.C.; Wu, J.; Dai, B. Extended versus non-extended lymphadenectomy during radical cystectomy for patients with bladder cancer: A meta-analysis of the effect on long-term and short-term outcomes. World J. Surg. Oncol. 2019, 17, 225. [Google Scholar] [CrossRef]
- Lerner, S.P.; Tangen, C.; Svatek, R.S.; Daneshmand, S.; Pohar, K.S.; Skinner, E.C.; Schuckman, A.K.; Sagalowsky, A.I.; Smith, N.D.; Kamat, A.M.; et al. SWOG S1011: A phase III surgical trial to evaluate the benefit of a standard versus an extended lymphadenectomy performed at time of radical cystectomy for muscle invasive urothelial cancer. J. Clin. Oncol. 2023, 41, 4508. [Google Scholar] [CrossRef]
- Gschwend, J.E.; Heck, M.M.; Lehmann, J.; Rübben, H.; Albers, P.; Wolff, J.M.; Frohneberg, D.; de Geeter, P.; Heidenreich, A.; Kälble, T.; et al. Extended Versus Limited Lymph Node Dissection in Bladder Cancer Patients Undergoing Radical Cystectomy: Survival Results from a Prospective, Randomized Trial. Eur. Urol. 2019, 75, 604–611. [Google Scholar] [CrossRef] [PubMed]
- Salem, H.K. Genital Organs-Sparing Radical Cystectomy in Female Patients with Muscle Invasive Urothelial Carcinoma of the Bladder. In Bladder Cancer—Management of NMI and Muscle-Invasive Cancer; InTech: London, UK, 2017. [Google Scholar]
- Patel, S.H.; Wang, S.; Metcalf, M.R.; Gupta, N.; Gabrielson, A.; Lee, E.; Rostom, M.; Pierorazio, P.; Smith, A.; Hahn, N.; et al. Safety and Efficacy of Reproductive Organ-Sparing Radical Cystectomy in Women with Variant Histology and Advanced Stage. Clin. Genitourin. Cancer 2022, 20, 60–68. [Google Scholar] [CrossRef] [PubMed]
- Wintner, A.; Dahl, D. Use of Intestinal Segments in Urinary Diversion. Chapter 139. In Campbell Walsh Wein Urology, International Edition, 12th ed.; Elsevier: Amsterdam, The Netherlands, 2021; pp. 3160–3205. [Google Scholar]
- Singh, V.; Yadav, R.; Sinha, R.J.; Gupta, D.K. Prospective comparison of quality-of-life outcomes between ileal conduit urinary diversion and orthotopic neobladder reconstruction after radical cystectomy: A statistical model. BJU Int. 2014, 113, 726–732. [Google Scholar] [CrossRef] [PubMed]
- Clements, M.B.; Atkinson, T.M.; Dalbagni, G.M.; Li, Y.; Vickers, A.J.; Herr, H.W.; Donat, S.M.; Sandhu, J.S.; Sjoberg, D.S.; Tin, A.L.; et al. Health-related Quality of Life for Patients Undergoing Radical Cystectomy: Results of a Large Prospective Cohort. Eur. Urol. 2022, 81, 294–304. [Google Scholar] [CrossRef] [PubMed]
- Dewulf, M.; Hildebrand, N.D.; Bouwense, S.A.W. Parastomal hernias after cystectomy and ileal conduit urinary diversion: Surgical treatment and the use of prophylactic mesh: A systematic review. BMC Surg. 2022, 22, 118. [Google Scholar] [CrossRef] [PubMed]
- Liedberg, F.; Kollberg, P.; Allerbo, M.; Baseckas, G.; Brändstedt, J.; Gudjonsson, S.; Hagberg, O.; Håkansson, U.; Jerlström, T.; Löfgren, A.; et al. Preventing Parastomal Hernia After Ileal Conduit by the Use of a Prophylactic Mesh: A Randomised Study. Eur. Urol. 2020, 78, 757–763. [Google Scholar] [CrossRef]
- Tinoco, C.L.; Lima, E. Urinary diversions for radical cystectomy: A review of complications and their management. Mini-Invasive Surg. 2021, 5, 28. [Google Scholar] [CrossRef]
- Lee, C.T.; Chang, S.S.; Kamat, A.M.; Amiel, G.; Beard, T.L.; Fergany, A.; Karnes, R.J.; Kurz, A.; Menon, V.; Sexton, W.J.; et al. Alvimopan accelerates gastrointestinal recovery after radical cystectomy: A multicenter randomized placebo-controlled trial. Eur. Urol. 2014, 66, 265–272. [Google Scholar] [CrossRef]
- Peerbocus, M.; Wang, Z.J. Enhanced Recovery After Surgery and Radical Cystectomy: A Systematic Review and Meta-Analysis. Res. Rep. Urol. 2021, 13, 535–547. [Google Scholar] [CrossRef]
- Blackwell, R.H.; Kothari, A.N.; Shah, A.; Gange, W.; Quek, M.L.; Luchette, F.A.; Flanigan, R.C.; Kuo, P.C.; Gupta, G.N. Adhesive Bowel Obstruction Following Urologic Surgery: Improved Outcomes with Early Intervention. Curr. Urol. 2018, 11, 175–181. [Google Scholar] [CrossRef]


| Study | Year | Total Patients | Primary Outcome(s) | Observation between Robotic and Open RC |
|---|---|---|---|---|
| Nix et al. [9] | 2010 | 41 | Lymph node yield | No difference |
| Bochner et al. [10] | 2015 | 118 | 90-day complication rate | No difference |
| CORAL [11] | 2016 | 164 | 30- and 90-day complication rate | Favored robotic vs. open for 30-day complication rate No difference for 90-day complication rate |
| Bochner et al. (update) [12] | 2018 | 118 | Recurrence; cancer-specific survival; overall survival | No difference |
| RAZOR [13] | 2019 | 350 | Two-year progression-free survival | No difference |
| CORAL (update) [14] | 2020 | 60 | Recurrence; cancer-specific survival; overall survival | No difference |
| iROC (intracorporeal diversion) [15] | 2022 | 317 | Median number of days alive and outside of the hospital within 90 days of surgery | Favored robotic (82 days) vs. open (80 days) approach |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
D’Andrea, V.D.; Melnick, K.; Yim, K.; Ernandez, J.; Onochie, N.; Clinton, T.N.; Steele, G.S.; Preston, M.A.; Kibel, A.S.; Mossanen, M. Evidence-Based Analysis of the Critical Steps of Radical Cystectomy for Bladder Cancer. J. Clin. Med. 2023, 12, 6845. https://doi.org/10.3390/jcm12216845
D’Andrea VD, Melnick K, Yim K, Ernandez J, Onochie N, Clinton TN, Steele GS, Preston MA, Kibel AS, Mossanen M. Evidence-Based Analysis of the Critical Steps of Radical Cystectomy for Bladder Cancer. Journal of Clinical Medicine. 2023; 12(21):6845. https://doi.org/10.3390/jcm12216845
Chicago/Turabian StyleD’Andrea, Vincent D., Kevin Melnick, Kendrick Yim, John Ernandez, Nnamdi Onochie, Timothy N. Clinton, Graeme S. Steele, Mark A. Preston, Adam S. Kibel, and Matthew Mossanen. 2023. "Evidence-Based Analysis of the Critical Steps of Radical Cystectomy for Bladder Cancer" Journal of Clinical Medicine 12, no. 21: 6845. https://doi.org/10.3390/jcm12216845
APA StyleD’Andrea, V. D., Melnick, K., Yim, K., Ernandez, J., Onochie, N., Clinton, T. N., Steele, G. S., Preston, M. A., Kibel, A. S., & Mossanen, M. (2023). Evidence-Based Analysis of the Critical Steps of Radical Cystectomy for Bladder Cancer. Journal of Clinical Medicine, 12(21), 6845. https://doi.org/10.3390/jcm12216845






