Abstract
Background: The rate of in-stent restenosis (ISR) is decreasing; however, it is still a challenge for contemporary invasive cardiologists. Therapeutic methods, including drug-eluting balloons (DEBs), intravascular lithotripsy, excimer laser coronary atherectomy, and imaging-guided percutaneous coronary intervention (PCI) with drug-eluting stents (DES), have been implemented. Patients with diabetes mellitus (DM) are burdened with a higher risk of ISR than the general population. Aims: DM-Dragon is aimed at evaluating the clinical outcomes of ISR treatment with DEBs vs. DES, focusing on patients with co-existing diabetes mellitus. Methods: The DM-Dragon registry is a retrospective study comprising data from nine high-volume PCI centers in Poland. A total of 1117 patients, of whom 473 individuals had DM and were treated with PCI due to ISR, were included. After propensity-score matching (PSM), 198 pairs were created for further analysis. The primary outcome of the study was target lesion revascularization (TLR). Results: In DM patients after PSM, TLR occurred in 21 (10.61%) vs. 20 (10.1%) in non-diabetic patients, p = 0.8690. Rates of target vessel revascularization (TVR), target vessel myocardial infarction, device-oriented composite endpoint (DOCE), and cardiac death did not differ significantly. Among diabetic patients, the risk of all-cause mortality was significantly lower in the DEB group (2.78% vs. 11.11%, HR 3.67 (95% confidence interval, CI) [1.01–13.3), p = 0.0483). Conclusions: PCI with DEBs is almost as effective as DES implantation in DM patients treated for ISR. In DM-Dragon, the rate of all-cause death was significantly lower in patients treated with DEBs. Further large-scale, randomized clinical trials would be needed to support these findings.
1. Introduction
The rapid development of interventional cardiology and construction of new-generation drug-eluting stents (DES) has significantly reduced the rate of in-stent restenosis (ISR) in treated vessels. However, its rate still ranges from 5 to 10% [1]. Due to its unpredictable clinical consequences, including death, acute coronary syndromes (ACS), unscheduled revascularization or readmissions, large-scale efforts have been made to establish effective methods of both prevention and treatment of ISR [2]. Currently proposed approaches of ISR therapy comprise usage of drug eluting balloons (DEB), vascular brachytherapy, excimer laser coronary atherectomy, intravascular lithotripsy, another DES implantation all guided by intravascular imaging with intravascular ultrasound or optical coherent tomography. If none of these methods are applicable, coronary artery by-pass grafting (CABG) can be performed [3]. The rates of ISR and restenosis after balloon angioplasty have been found to be significantly higher in diabetic patients than in the general population (55% vs. 20%, p = 0.001) [4]. Consistently, in another study, stent-edge restenosis occurred more frequently in individuals with diabetes (20.3% vs. 9.2%, p = 0.019) [5]. Several potential mechanisms of ISR in diabetes mellitus (DM) patients have been proposed. Among them, the abnormal activity of vascular smooth muscle cells; more aggressive and rapidly progressing atherosclerosis; the impairment of particular glycoproteins, such as plasminogen activator inhibitor-1; increased platelet aggregation resulting from insulin-resistance; and hyperinsulinemia need to be mentioned due to their most pronounced impact on the risk of ISR [6]. The optimal treatment of ISR in the general population was evaluated in the DEB-Dragon registry, a large-scale multicenter study including 1117 participants, which showed comparable clinical outcomes, i.e., cardiac mortality, rate of recurrent myocardial infarctions (MIs), target lesion revascularization (TLR) or a device-oriented composite endpoint between patients treated with thin DES vs. drug-eluting balloons (DEBs) [7]. The DM-Dragon is a sub-analysis of the DEB-Dragon registry (ClinicalTrials.gov Identifier: NCT 04415216) aimed at evaluating the safety and efficacy of DEBs vs. thin-strut DES in diabetic patients. The DM-Dragon study focuses on the clinical outcomes of percutaneous coronary interventions (PCIs) with DEBs and DES in diabetic patients in comparison with non-DM patients. Additional analyses evaluating the safety of DEB vs. DES were conducted in study subpopulations including active smokers, history of chronic kidney disease (CKD), males vs. females, index hospitalization myocardial infarction (MI) or preselected procedural aspects such as location of the treated lesion, bifurcation angioplasty etc. were performed.
2. Materials and Methods
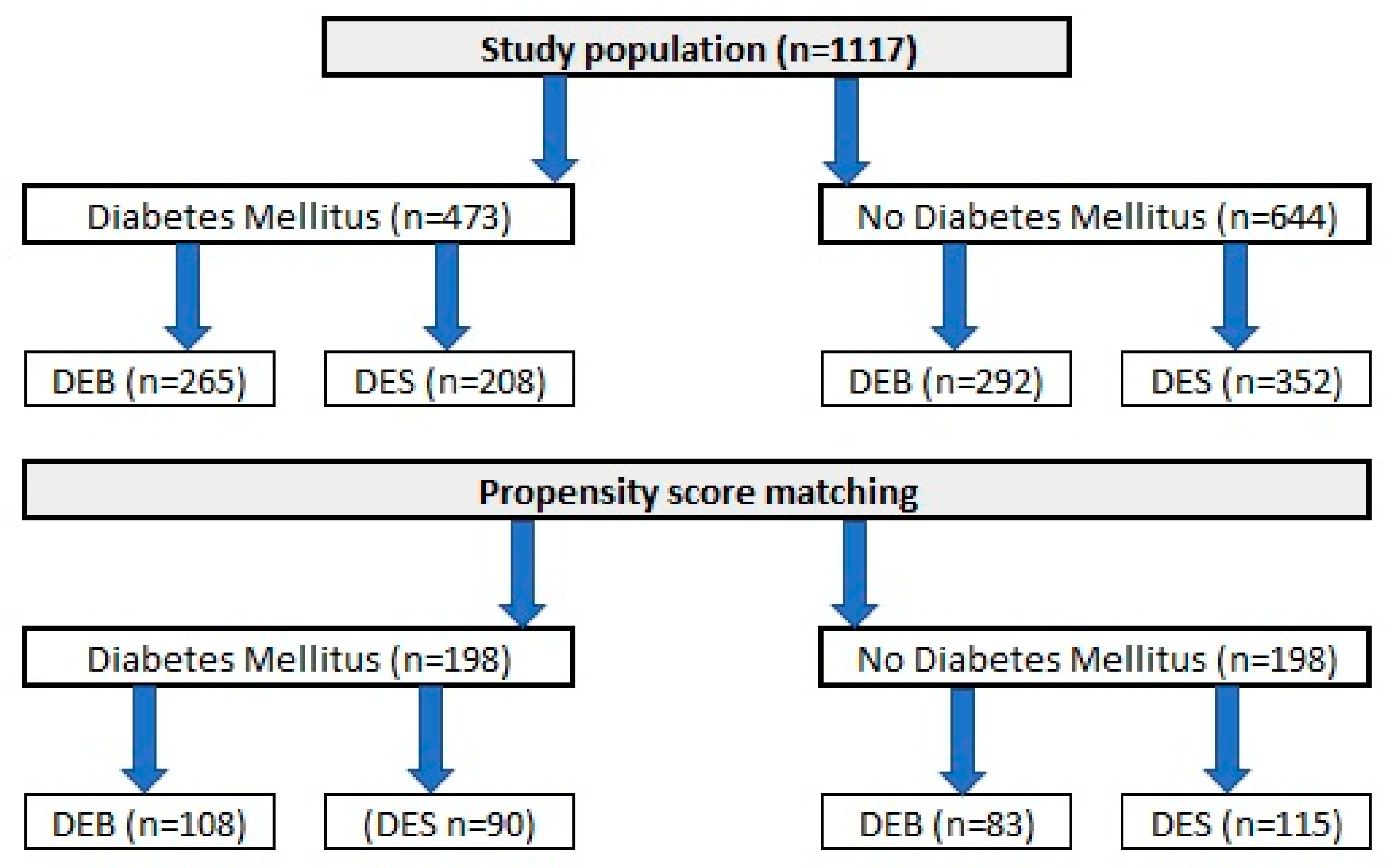
A total of 1117 patients, who underwent PCI due to ISR in one of nine high-volume PCI centers in Poland, were included in the analysis. Similar to the original DEB-Dragon registry [7], patients were divided into study arms as follows: (1) patients treated with thin DES, defined as DES with a strut thickness below 100 µm (n = 560), and (2) patients treated with DEBs (n = 557). A complete list of thin DES and DEBs used in the study is presented in Appendix A. Only patients who underwent PCI in native arteries and were not treated with both methods in the same procedure were included. Moreover, patients with repeated PCI with stent implantation causing the formation of multiple stent layers were also excluded from analysis. Data on clinical outcomes throughout a 3-year follow-up period were obtained from each center’s clinical documentation, and we used the databases of the National Health Fund and follow-up phone calls if needed. The design of the study was approved by The Bioethics Committee of The Medical University of Silesia (approval number PCN/0022/KB/171/20). Because of the retrospective character of analysis, no written informed consent was required. Data of each study participant were anonymized. The study flow chart is presented in Figure 1.
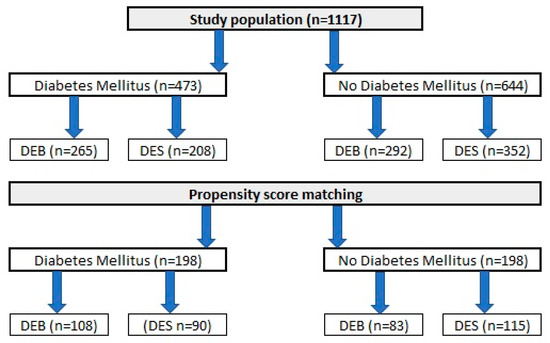
Figure 1.
A DM-Dragon study flowchart.
3. Endpoints
Study endpoints in the DM-Dragon registry were defined identically to those used in the main trial, DEB-Dragon [7]; however, all outcomes were analyzed in diabetic vs. non-diabetic participants. Target lesion revascularization (TLR) was the primary endpoint of the study. A composite of cardiac death, TLR, target vessel MI (device-oriented composite endpoint—DOCE), as well as target lesion revascularization (TVR), MI and cardiac death were defined as secondary endpoints.
4. Statistical Analysis
The statistical analysis was carried out using the Statistica 13.0 package (TIBCO Software Inc, Palo Alto, CA, USA). Continuous data were presented as medians with inter-quartile ranges (first quartile (Q1)–third quartile (Q3). Qualitative data were presented as values and percentages. The normality of data distribution was assessed with the Shapiro–Wilk test. Since the variables were not normally distributed, comparisons of continuous variables between groups were performed with the Mann–Whitney unpaired rank sum test. Categorical variables were compared using the χ2 or Fisher exact test. Kaplan–Meier survival curves were created for time-to-event data, and data were compared with the log-rank test. Due to the inhomogeneity of the population enrolled in the study, propensity-score matching (PSM) was performed. The variables included in the PSM were age, gender, CKD, dialysis, hypertension, hyperlipidemia, chronic obstructive pulmonary disease (COPD), atrial fibrillation (AF), active smoking, previous MI, previous coronary artery bypass surgery (CABG), family history of coronary artery disease (CAD), peripheral artery disease (PAD), stable angina on admission, STEMI on admission, left ventricle ejection fraction, three-vessel disease, left main PCI, circumflex artery PCI, bifurcation lesion, thrombus, calcifications, and residual stenosis post-PCI. Due to the fact that DM-Dragon focused on the clinical outcomes of DEB vs. DES therapy in DM patients, the diagnosis of DM was not included in the PSM, contrary to the original DEB-Dragon registry [7]. After PSM, a total of 198 pairs were generated. Cox regression analysis was performed for long-term follow-up event rates. The results of the Cox regression analysis were presented as hazard ratios (HRs) and 95% confidence intervals (CIs). p values < 0.05 were considered significant.
5. Results
Taking into account the baseline characteristics of the enrolled participants (Table 1), significant differences were observed in body weight and its derivative, body mass index. Diabetic patients more commonly suffered from CKD, arterial hypertension, PAD and AF. Moreover, in diabetic patients, UA was diagnosed more often. In invasive diagnostics, the rate of a three-vessel disease was significantly higher in diabetic patients. Also, the percentage of patients with bifurcation lesions was significantly higher in cases of co-existing diabetes, and lesions were treated with longer stents.

Table 1.
Baseline characteristics of the population of DM-Dragon trial.
Of the 1117 patients included in the analysis, 473 (42.3%) participants had diabetes. A 3-year follow-up (Table 2) showed no significant difference in the primary endpoint between the study arms in the unselected cohort (57 [12.05%] vs. 70 [18.87%], p = 0.5389) or post-PSM (20 [10.1%] vs. 21 [10.61%], p = 0.8690) for DM and non-DM patients, respectively. The rates of MI and all-cause death were higher among diabetic patients in the unselected cohort (70 [14.8%] vs. 52 [8.07%], p = 0.0004 and 43 [9.09%] vs. 28 [4.35%], p = 0.0013 for MI and all-cause death, respectively). However, differences in these outcomes turned out to be insignificant after PSM. The remaining outcomes, including DOCE, TVR, cardiac death and target vessel MI, did not differ significantly between the study arms neither in the unselected population nor after PSM.

Table 2.
Long-term clinical outcomes in DM-Dragon trial.
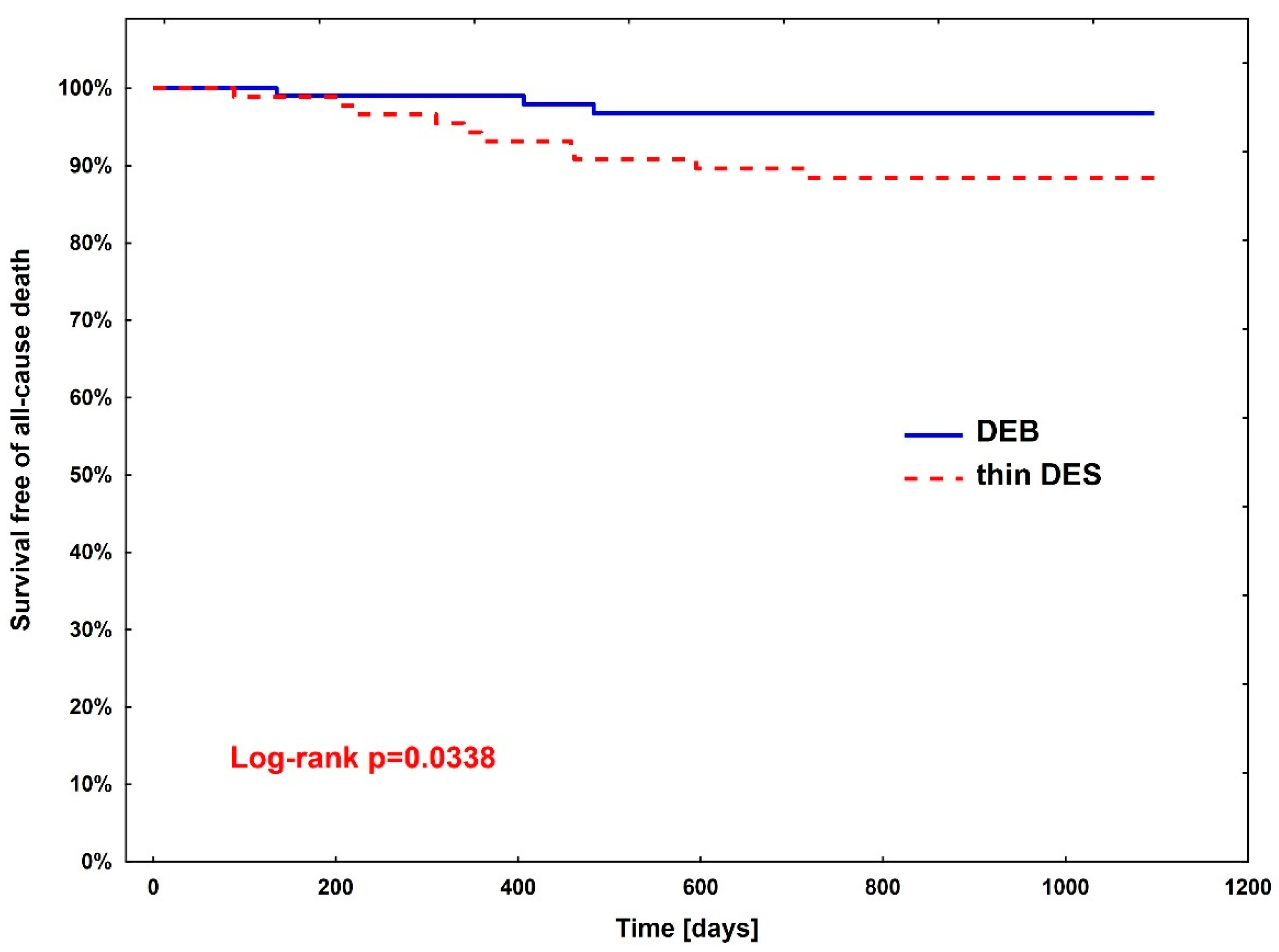
Rates of clinical endpoints (Table 3) were compared between patients treated with DEBs vs. DES after PSM. A significant difference was observed only in all-cause death, where the superiority of DEBs over DES was shown (hazard ratio (HR)—3.67, 95% CI (1.01–13.3; p = 0.0483)). The Kaplan–Meier curve for all-cause death is presented in Figure 2.

Table 3.
Summary of clinical endpoints in the DM-Dragon study.
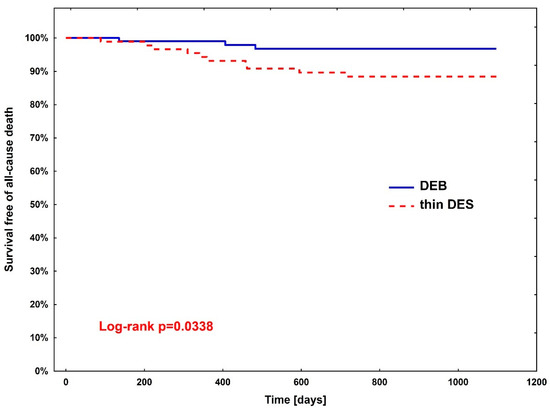
Figure 2.
Kaplan–Meier curve for all-cause death between diabetic patients treated with DEBs (group 1) vs. DES (group 2).
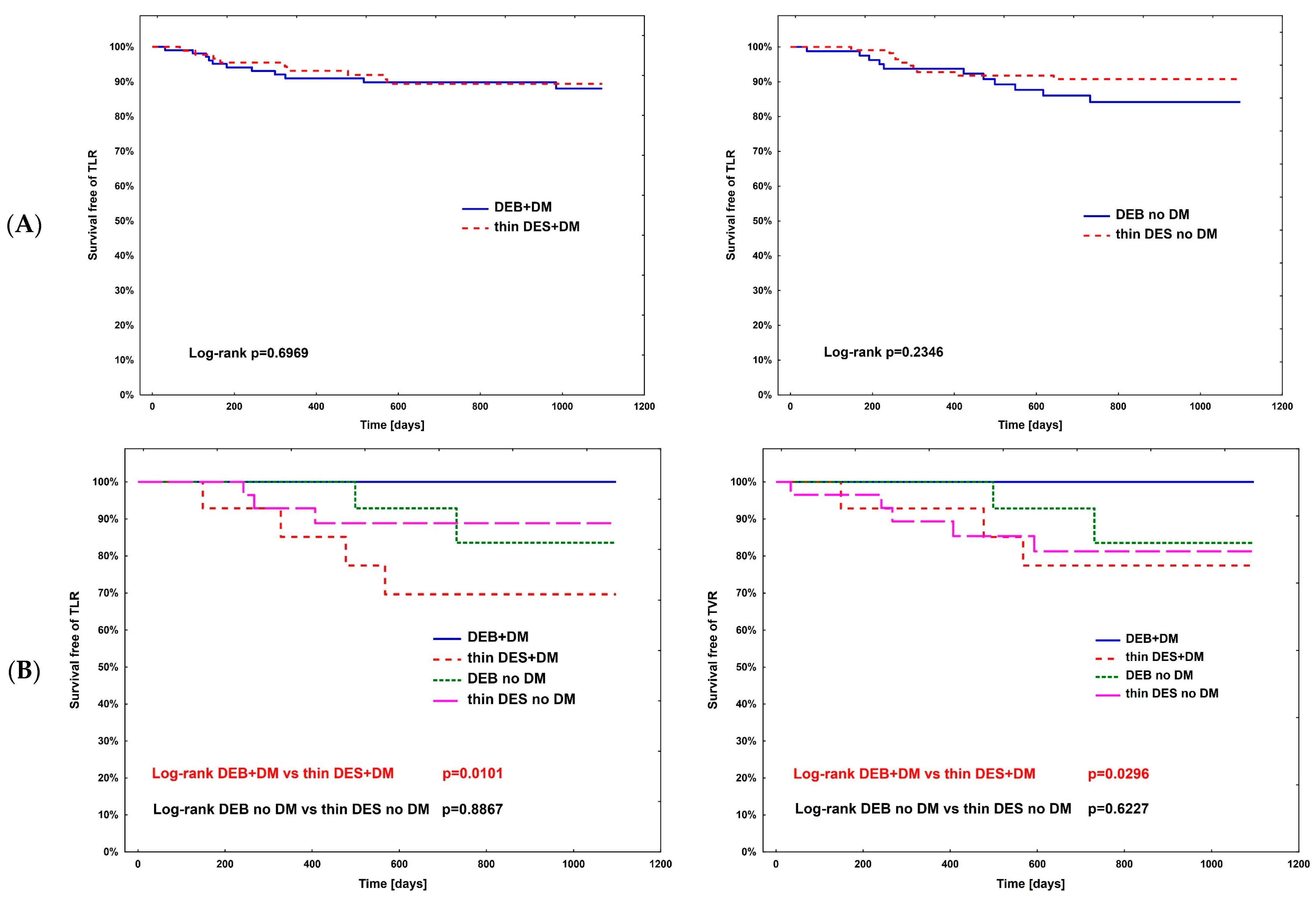
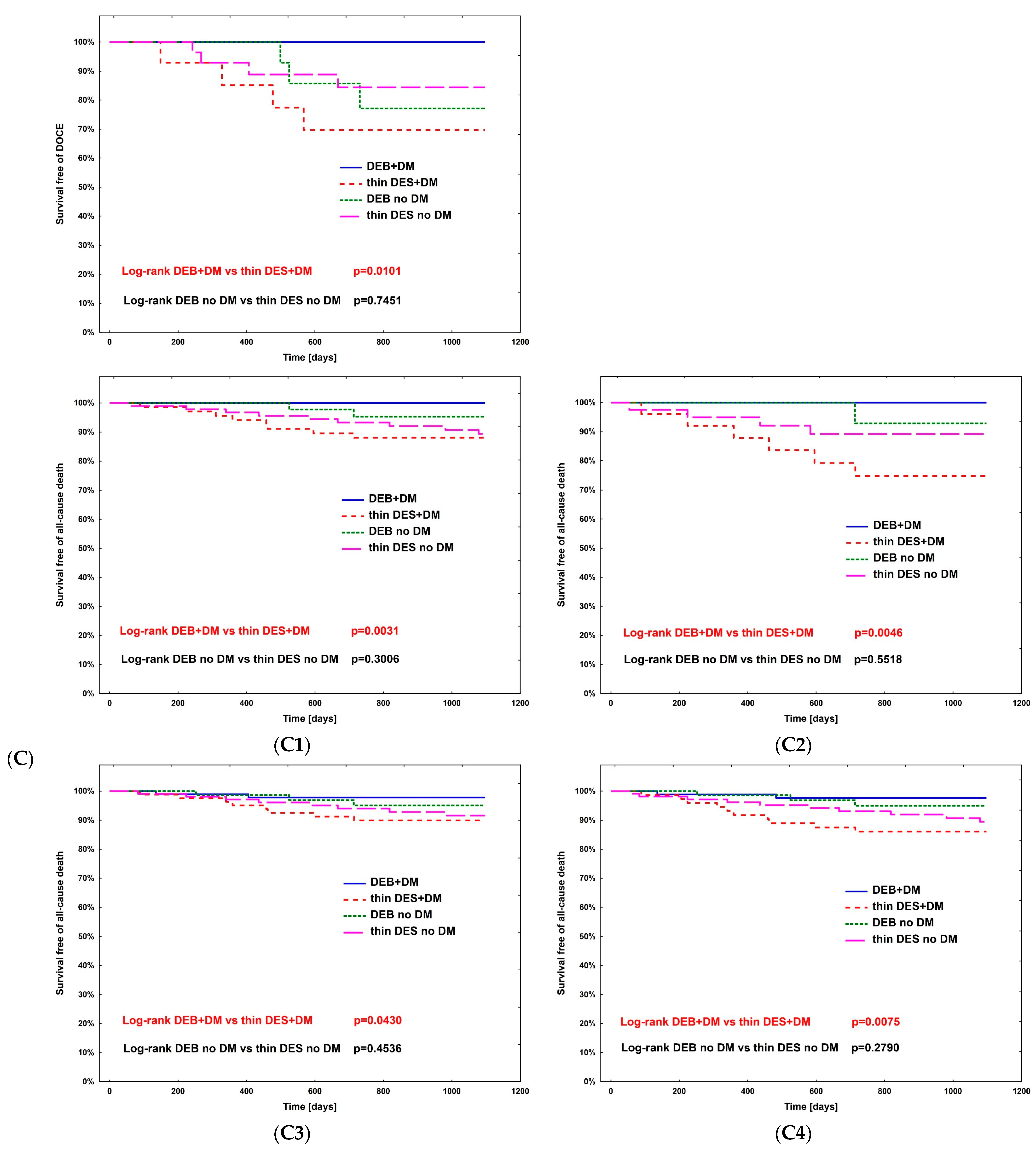
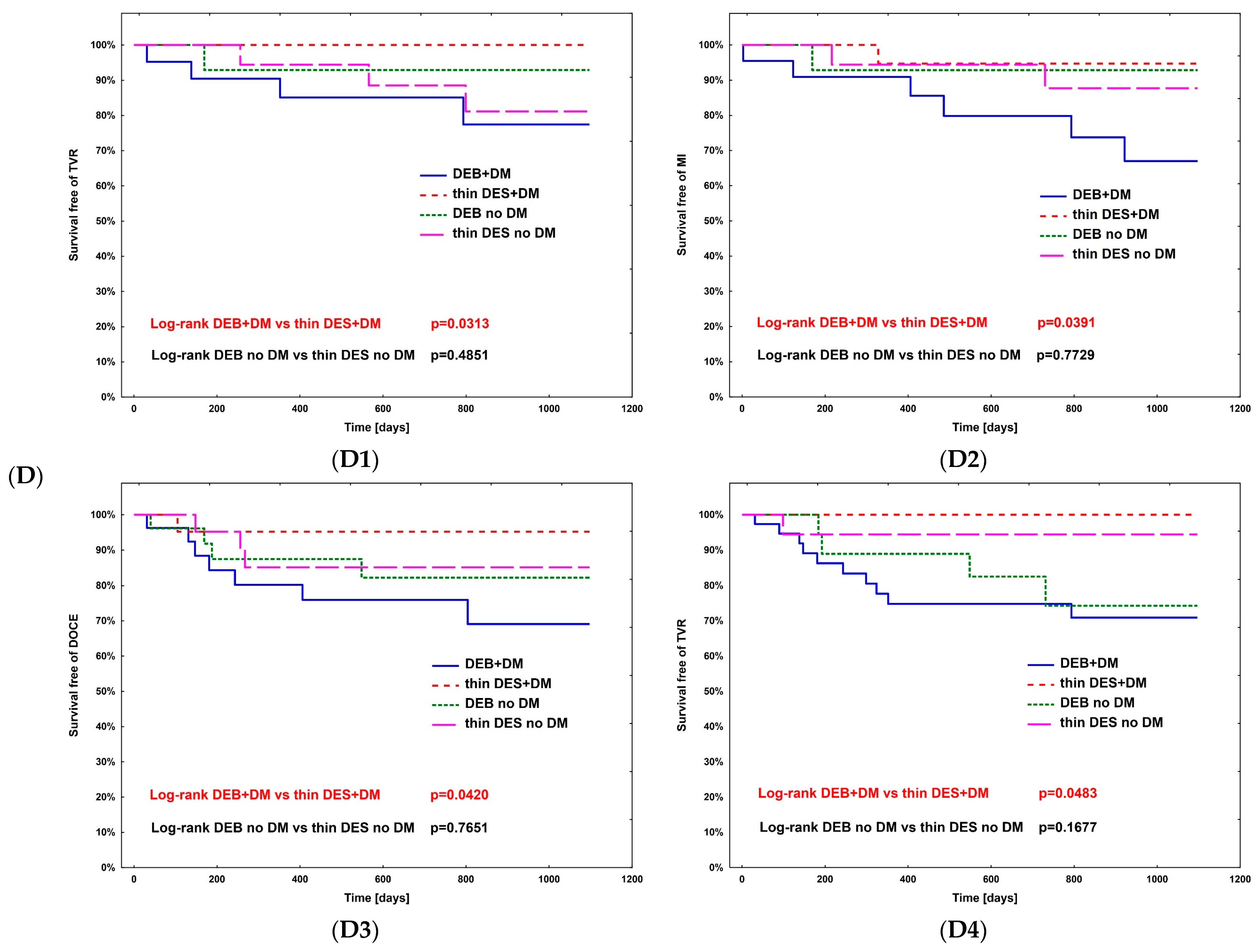
The hazard ratio for TLR (Table 4) was calculated using Cox regression analysis in all subpopulations; however, no significance was observed. The complete comparison of DEBs vs. DES in diabetic and non-diabetic subgroups is presented in Table 5. After PSM, the rates of study outcomes between DES vs. DEBs in diabetic and non-diabetic subgroups including current MI vs. no MI, current ACS vs. CCS, males vs. females, CKD vs. no-CKD, hypertension vs. no hypertension, active smoking vs. non-smokers, bifurcation lesion vs. no-bifurcation, and LM PCI vs. no-LM PCI revealed no significant differences in the non-diabetic population. In DM patients, in terms of the primary endpoint of the study, the superiority of DEBs over DES was observed only in active cigarette smokers (p = 0.0101). Actively smoking DM patients more often experienced TVR and DOCE (p = 0.0296 and p = 0.0101, respectively). Also, more favorable outcomes were associated with the use of DEBs with respect to all-cause death in several analyzed subgroups, including females, non-smokers, and patients without bifurcation lesions, without LM PCI and who did not present with an acute MI. On the other hand, the superiority of DES over DEBs was observed in terms of TVR in CKD patients (p = 0.0483), as well as TVR and MI in bifurcation lesions (p = 0.0313 and p = 0.0391, respectively). Long-term observation also revealed less episodes of DOCE in patients treated for MI (p = 0.0420); however, as mentioned above, the superiority of DEBs over DES was significant in active smokers. Kaplan–Meier curves for the primary endpoints of the study and selected significant differences between study arms are presented in Figure 3A–D.

Table 4.
Hazard ratio for target lesion revascularization between patients treated with DEBs vs. DES in study sub-populations.

Table 5.
Clinical outcomes analysis in pre-selected subgroups of patients before and after PSM. (*)—indicates superiority of DEBs, (+)—indicates superiority of DES.
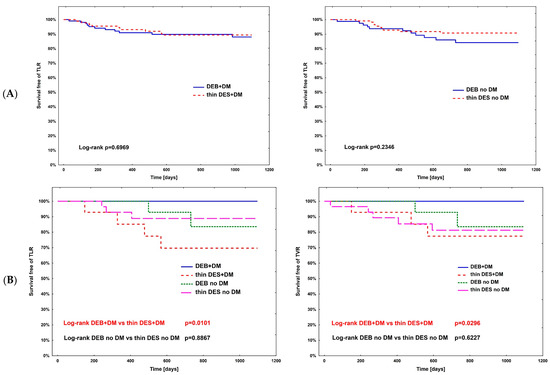
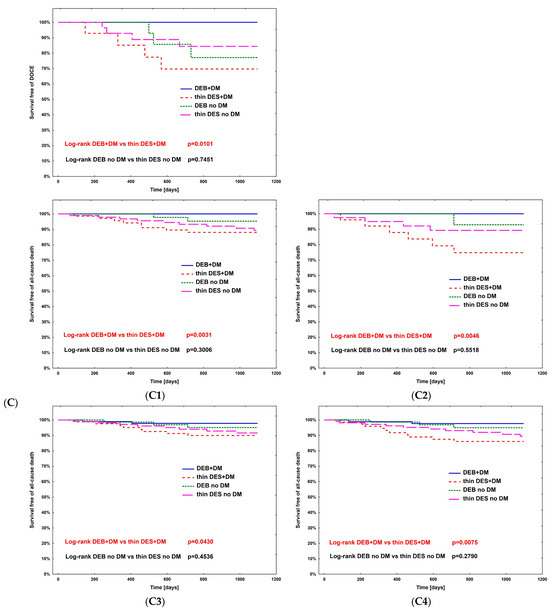
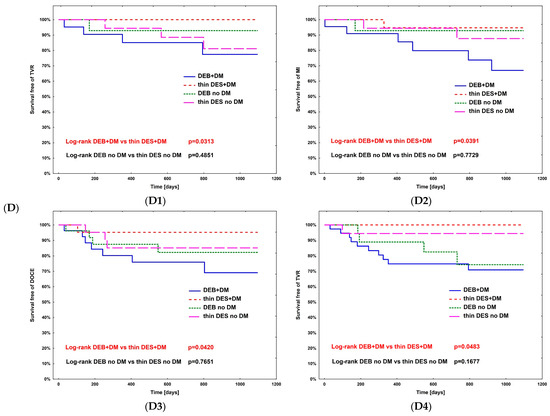
Figure 3.
Kaplan–Meier curves for primary endpoint of DM Dragon (A) and preselected subpopulation analysis of DEBs vs. DES use long-term outcomes (B–D). (B) Active smokers. (C) All-cause death—superiority of DEBs over DES: (C1) No MI; (C2) Female; (C3) Arterial hypertension; (C4) Other than LM PCI. (D) Superiority of DES over DEBs: (D1) TVR—bifurcation lesions; (D2) MI—bifurcation lesions; (D3) DOCE—current MI; (D4) TVR—CKD patients.
6. Discussion
As mentioned above, diabetic patients undergoing PCI are more predisposed to ISR when compared with the general population; therefore, the optimization of measures undertaken during invasive procedures is of a great importance. In the DM-Dragon study, active smokers diagnosed with DM had a significantly higher risk of TLR, TVR and DOCE if they were treated with DES implantation. On the other hand, in this study, diabetic patients undergoing the angioplasty of bifurcation lesions with DEBs were predisposed to TVR and MI when compared with DES implantation. The superiority of DES was also observed in CKD patients in terms of TVR rate and in terms of DOCE if an acute MI was currently diagnosed. Additionally, in the proposed study, rates of TLR did not differ significantly in DM vs. no DM patients treated with DEBs or DES due to ISR. In a subpopulation analysis, no significant difference was observed in terms of TLR rate in CKD vs. no CKD patients. All-cause mortality turned out to be significantly lower in DM patients treated with DEBs when compared with those who received DES.
A study by Alexandrescu et. al., which aimed to examine potential risk factors of ISR, proved the significant influence of active smoking (RR 1.63, 95% confidence interval [CI] 1.25–2.13, p = 0.001), arterial hypertension (RR 1.86, 95% CI 1.41–2.45, p = 0.001), DM (RR 1.83, 95% CI 1.42–2.36, p = 0.001) and CKD (RR 1.90, 95% CI 1.53–2.36, p = 0.001) on the rate of ISR post-PCI [8]. Also, compared to those who never smoked, individuals with a history of current or previous smoking turned out to have significantly higher risk of ISR in a study by Megaly et. al. (odds ratio [OR] 10.085, 95% CI 1.495–68.038, p = 0.018) [9]. In a study by Jonas et. al., patients with CKD who were treated with DEB had a higher risk of major adverse cardiac events (MACEs) (23.8% vs. 13.8%, p < 0.005 and MI 15.9% vs. 3.8%, p < 0.001) than those without CKD. Similar to the DM-Dragon study, the rate of TLR was similar in CKD vs. no-CKD patients [10]. In a large analysis of PCI outcomes (n = 3 187 404) in CKD patients (11%), there was a significant increase in in-hospital mortality, periprocedural hemorrhage, and in-hospital stay (p < 0.001 for each parameter). Another study, by Wang et. al., which compared outcomes of DEB vs. DES use in patients with ISR, found DM, as well as at least three stent layers and re-DES implantation, to be independent risk factors of MACEs for recurrent ISR. Contrary to DM-Dragon, the same work indicates that the risk of MACEs and TLR in recurrent ISR is significantly higher in patients treated with DES than in those who received DEBs (17.2% vs. 32.9%; p = 0.02 and 15.1% vs. 27.8%; p = 0.04, respectively) [11]. A recent review and meta-analysis by Murphy et. al. proved the utility of small-vessel angioplasty with DEBs. Patients treated with DEBs in comparison with DES had significantly lower rates of non-fatal MI at the 1-year timepoint, which was accompanied with the reduction of major bleeding episodes [12]. A DARE study by Claessen et. al. that compared clinical outcomes and angiographic parameters after PCI using paclitaxel-coated balloons (PCBs) vs. everolimus-eluting stents (EESs) showed similar major adverse events, including death, TVR and target vessel MI in diabetic patients between the study arms (MACE: 11.9% vs. 17.4% for PCBs and EESs, respectively, p = 0.44) [13]. Similar MACE rates at 1 year in both diabetic and non-diabetic patients treated with paclitaxel-eluting stents and PCBs were also observed in the BELLO trial by Giannini et al. (13.2% vs. 25%, p = 0.194 for DES and PCBs, respectively, in DM patients and 11.8% vs. 14.3%, p = 0.699 in no-DM ones) [14]. Another comparison of DEBs and DES in DM patients treated for ISR or de novo lesions performed by Verdoia et. al. [15] showed comparable clinical outcomes between study arms (MACE: 21.6% vs. 17.3%, HR (95% CI) 1.51 [0.46–4.93], for DEBs and DES, respectively, p = 0.50), but overall mortality was significantly lower in the DEB group (3.6% vs. 10.9%; HR (95% CI) 0.27 [0.08–0.91], p = 0.03). These results are consistent with findings from the current DM-Dragon study, where the superiority of DEBs vs. DES was observed for all-cause death in DM patients (2.78% vs. 11.11%, HR 3.67 (95% CI) [1.01–13.3), p = 0.0483). A meta-analysis of six clinical studies (a total of 847 diabetic patients) by Kui et. al. aimed to compare clinical outcomes of small-vessel PCI with DEBs vs. DES. Treatment with DEBs was associated with a lower risk of MACEs (RR, 0.60; 95% CI: 0.39–0.93; p = 0.02), MI (RR, 0.42; 95% CI, 0.19–0.94; p = 0.03), TLR (RR, 0.24; 95% CI, 0.08–0.69; p < 0.001) and TVR (RR, 0.33; 95% CI, 0.18–0.63; p < 0.001) [16].
7. Study Limitations
The DM-Dragon is a retrospective analysis of clinical data obtained from each PCI center, National Health Fund databases and follow-up phone calls. Despite a relatively large baseline population (n = 1117), PSM resulted in a noticeable reduction of participants included in the final analysis of study outcomes (198 pairs). Therefore, the numbers of some analyzed events were low, which may have influenced the obtained results. Despite the fact that the study focuses on the analysis of clinical outcomes of ISR therapy with DEBs vs. DES in DM patients, no detailed data including the duration of diabetes or its current pharmacotherapy were available.
8. Conclusions
In DM patients treated for ISR, PCI with DEBs has a similar effectiveness to DES implantation in terms of the rate of adverse events, including TLR, TVR, MI or cardiac death. However, patients treated with DEBs had a lower risk of all-cause death when compared with those who underwent PCI with DES implantation. The results of the DM-Dragon study may be useful to lay the foundations for the further widespread use of such a therapeutic approach in DM patients undergoing PCI due to restenosis. PCI with DEBs allows us to avoid the multiplication of stent layers in the treated vessels, which undoubtedly makes the procedure beneficial for patients. Further development of large-scale randomized clinical trials on the efficacy of DEBs vs. DES in ISR in diabetic patients would be needed to support these findings. Taking into account the DM-Dragon results, accompanied by other worldwide data on the clinical utility of DEBs in coronary procedures, conducting further trials may be considered safe for study participants, which in turn may allow us to accelerate the enrollment of patients and reduce the potential concerns of both the patient and the investigator.
Author Contributions
Writing—original draft, P.N. and M.K. (Michał Kasprzak); Writing—review & editing, P.N., M.K. (Michał Kasprza), J.K., Ł.K., R.J., S.I., B.T., J.B., M.K. (Mariusz Kowalewski), M.J., M.W., K.R., S.D., S.B., M.L., M.G., R.W., A.W., R.G., B.C., F.D., W.W. (Wojciech Wojakowski) and W.W. (Wojciech Wańha); Supervision, J.K. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
The design of the study was approved by The Bioethics Committee of The Medical University of Silesia (approval number PCN/0022/KB/171/20).
Informed Consent Statement
Patient consent was waived due to the retrospective character of the data analysis.
Data Availability Statement
The original contributions presented in the study are included in the article, further inquiries can be directed to the corresponding author.
Acknowledgments
The authors would like to thank each center’s workers who participated in data collection.
Conflicts of Interest
The authors declare no conflicts of interest.
Abbreviations
| ACS | acute coronary syndrome |
| AF | atrial fibrillation |
| AH | arterial hypertension |
| CABG | coronary artery bypass grafting |
| CAD | coronary artery disease |
| CCS | chronic coronary syndrome |
| CI | confidence interval |
| CKD | chronic kidney disease |
| DEB | drug-eluting balloon |
| DES | drug-eluting stent |
| DM | diabetes mellitus |
| DOCE | device-oriented composite endpoint |
| HR | hazard ratio |
| ISR | in-stent restenosis |
| LM | left main trunk |
| MACE | major adverse cardiac event |
| MI | myocardial infarction |
| PAD | peripheral artery disease |
| PCI | percutaneous coronary intervention |
| PSM | Propensity-score matching |
| RR | risk ratio |
| TLR | target lesion revascularization |
| TVR | target vessel revascularization |
| UA | unstable angina |
Appendix A. The Complete List of Thin-Strut DES and Paclitaxel-Coated DEB Types Used in the Study
| Thin-Strut DES | Paclitaxel—DEB | ||
| Name | Manufacturer | Name | Manufacturer |
| Xience | Abbott Vascular Devices, Santa Clara, CA, USA | Agent | Boston Scientific, Natick, MA, USA |
| Resolute | (Medtronic CardioVascular, Santa Rosa, CA, USA | Elutax | Aachen Resonance GmbH, Aachen, Germany |
| Promus | Boston Scientific, Natick, MA, USA | Essentia | iVascular, Barcelona, Spain |
| Ultimaster | Terumo Corporation, Tokyo, Japan | In.Pact | Medtronic Vascular, Santa Clara, CA, USA |
| Synergy | Boston Scientific, Natick, MA, USA | Pantera Lux | Biotronik AG, Buulach, Switzerland |
| Orsiro | Biotronik AG, Bulach, Switzerland | Restore DEB | Cardionovum GmbH, Bonn, Germany |
| Alex | Balton, Warsaw, Poland | SeQuentPleaseNeo | B.Braun Interventional Group, Ltd., Melsulgen, Germany |
References
- Aoki, J.; Tanabe, K. Mechanisms of drug-eluting stent restenosis. Cardiovasc. Interv. Ther. 2021, 36, 23–29. [Google Scholar] [CrossRef] [PubMed]
- Magalhaes, M.A.; Minha, S.; Chen, F.; Torguson, R.; Omar, A.F.; Loh, J.P.; Escarcega, R.O.; Lipinski, M.J.; Baker, N.C.; Kitabata, H.; et al. Clinical presentation and outcomes of coronary in-stent restenosis across 3-stent generations. Circ. Cardiovasc. Interv. 2014, 7, 768–776. [Google Scholar] [CrossRef] [PubMed]
- Shlofmitz, E.; Iantorno, M.; Waksman, R. Restenosis of Drug-Eluting Stents: A New Classification System Based on Disease Mechanism to Guide Treatment and State-of-the-Art Review. Circ. Cardiovasc. Interv. 2019, 12, e007023. [Google Scholar] [CrossRef] [PubMed]
- Carrozza, J.P., Jr.; Kuntz, R.E.; Fishman, R.F.; Baim, D.S. Restenosis after arterial injury caused by coronary stenting in patients with diabetes mellitus. Ann. Intern. Med. 1993, 118, 344–349. [Google Scholar] [CrossRef] [PubMed]
- Paramasivam, G.; Devasia, T.; Jayaram, A.; Razak, A.; Rao, M.S.; Vijayvergiya, R.; Nayak, K. In-stent restenosis of drug-eluting stents in patients with diabetes mellitus: Clinical presentation, angiographic features, and outcomes. Anatol. J. Cardiol. 2020, 23, 28–34. [Google Scholar] [CrossRef] [PubMed]
- Wilson, S.; Mone, P.; Kansakar, U.; Jankauskas, S.S.; Donkor, K.; Adebayo, A.; Varzideh, F.; Eacobacci, M.; Gambardella, J.; Lombardi, A.; et al. Diabetes and restenosis. Cardiovasc. Diabetol. 2022, 21, 23. [Google Scholar] [CrossRef] [PubMed]
- Wańha, W.; Bil, J.; Januszek, R.; Gilis-Malinowska, N.; Figatowski, T.; Milewski, M.; Pawlik, A.; Staszczak, B.; Wybraniec, M.; Tomasiewicz, B.; et al. Long-Term Outcomes Following Drug-Eluting Balloons Versus Thin-Strut Drug-Eluting Stents for Treatment of In-Stent Restenosis (DEB-Dragon-Registry). Circ. Cardiovasc. Interv. 2021, 14, e010868. [Google Scholar] [CrossRef] [PubMed]
- Alexandrescu, D.M.; Mitu, O.; Costache, I.I.; Macovei, L.; Mitu, I.; Alexandrescu, A.; Georgescu, C.A. Risk factors associated with intra-stent restenosis after percutaneous coronary intervention. Exp. Ther. Med. 2021, 22, 1141. [Google Scholar] [CrossRef] [PubMed]
- Megaly, M.; Alani, F.; Cheng, C.I.; Ragina, N. Risk Factors for the Development of Carotid Artery In-Stent Restenosis: Multivariable Analysis. Cardiovasc. Revasc. Med. 2021, 24, 65–69. [Google Scholar] [CrossRef] [PubMed]
- Jonas, M.; Kagan, M.; Sella, G.; Haberman, D.; Chernin, G. Cardiovascular outcomes following percutaneous coronary intervention with drug-eluting balloons in chronic kidney disease: A retrospective analysis. BMC Nephrol. 2020, 21, 445. [Google Scholar] [CrossRef] [PubMed]
- Wang, G.; Zhao, Q.; Chen, Q.; Zhang, X.; Tian, L.; Zhang, X. Comparison of drug-eluting balloon with repeat drug-eluting stent for recurrent drug-eluting stent in-stent restenosis. Coron. Artery Dis. 2019, 30, 473–480. [Google Scholar] [CrossRef] [PubMed]
- Murphy, G.; Naughton, A.; Durand, R.; Heron, E.; McCaughey, C.; Murphy, R.T.; Pearson, I. Long-term Outcomes for Drug-eluting Balloons versus Drug-eluting Stents in the Treatment of Small Vessel Coronary Artery Disease: A Systematic Review and Meta-analysis. Interv. Cardiol. 2023, 18, e14. [Google Scholar] [CrossRef] [PubMed]
- Claessen, B.E.; Henriques, J.P.S.; Vendrik, J.; Boerlage-van Dijk, K.; van der Schaaf, R.J.; Meuwissen, M.; van Royen, N.; Gosselink, A.T.M.; van Wely, M.H.; Dirkali, A.; et al. Paclitaxel-eluting balloon versus everolimus-eluting stent in patients with diabetes mellitus and in-stent restenosis: Insights from the randomized DARE trial. Catheter. Cardiovasc. Interv. 2019, 93, 216–221. [Google Scholar] [CrossRef] [PubMed]
- Giannini, F.; Latib, A.; Jabbour, R.J.; Costopoulos, C.; Chieffo, A.; Carlino, M.; Montorfano, M.; Menozzi, A.; Castriota, F.; Micari, A.; et al. Comparison of paclitaxel drug-eluting balloon and paclitaxel-eluting stent in small coronary vessels in diabetic and nondiabetic patients—Results from the BELLO (balloon elution and late loss optimization) trial. Cardiovasc. Revasc. Med. 2017, 18, 4–9. [Google Scholar] [CrossRef] [PubMed]
- Verdoia, M.; Zilio, F.; Gioscia, R.; Viola, O.; Brancati, M.F.; Fanti, D.; Soldà, P.L.; Bonmassari, R.; Rognoni, A.; De Luca, G. Prognostic Impact of Drug-Coated Balloons in Patients with Diabetes Mellitus: A Propensity-Matched Study. Am. J. Cardiol. 2023, 206, 73–78. [Google Scholar] [CrossRef] [PubMed]
- Li, K.; Cui, K.; Dan, X.; Feng, J.; Pu, X. The comparative short-term efficacy and safety of drug-coated balloon vs. drug-eluting stent for treating small-vessel coronary artery lesions in diabetic patients. Front. Public Health 2022, 10, 1036766. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).