The Diagnostic Accuracy of Procalcitonin, Soluble Urokinase-Type Plasminogen Activator Receptors, and C-Reactive Protein in Diagnosing Urinary Tract Infections in the Emergency Department—A Diagnostic Accuracy Study
Abstract
:1. Introduction
1.1. Background
1.2. Aim and Objectives
2. Materials and Methods
2.1. Study Design
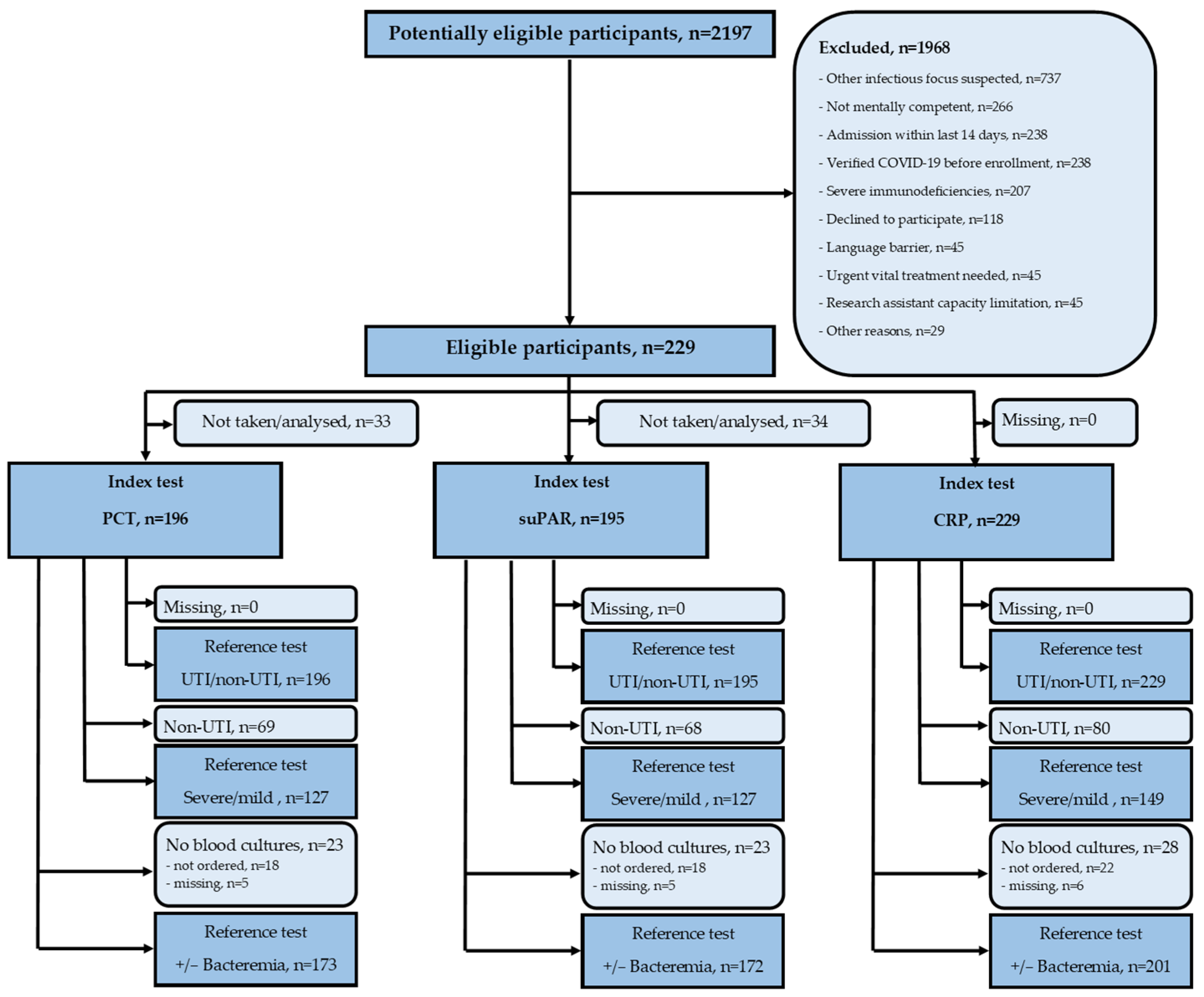
2.2. Participants
2.3. Blood Sample Procedure
2.4. Tests and Variables
2.4.1. Procalcitonin—Index Test
2.4.2. Soluble Urokinase-Type Plasminogen Activator Receptor—Index Test
2.4.3. C-Reactive Protein—Index Test
2.4.4. Urinary Tract Infection Diagnosis and Severity—Reference Tests
2.4.5. Blood Cultures—Reference Test
2.4.6. Other Variables
2.5. Statistical Analysis
3. Results
3.1. Participants
3.2. Diagnostic Precision of Procalcitonin
3.3. Diagnostic Precision of Soluble Urokinase-Type Plasminogen Activator Receptor
3.4. Diagnostic Accuracy of C-Reactive Protein
4. Discussion
4.1. Key Results
4.2. Study Limitations
4.3. Implications for Practice
4.3.1. Procalcitonin
4.3.2. Soluble Urokinase-Type Plasminogen Activator Receptor
4.3.3. C-Reactive Protein
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| AUROC | area under receiver operator characteristics curve |
| CRP | C-reactive protein |
| DA | diagnostic accuracy |
| ED | emergency department |
| NPV | negative predictive value |
| PCT | procalcitonin |
| PPV | positive predictive value |
| suPAR | soluble urokinase-type plasminogen activator receptor |
| UTI | urinary tract infection |
References
- Foxman, B. Urinary tract infection syndromes: Occurrence, recurrence, bacteriology, risk factors, and disease burden. Infect. Dis. Clin. N. Am. 2014, 28, 1–13. [Google Scholar] [CrossRef]
- Schappert, S.M.; Rechtsteiner, E.A. Ambulatory medical care utilization estimates for 2007. Vital Health Stat. 13 2011, 13, 1–38. [Google Scholar]
- Foxman, B. The epidemiology of urinary tract infection. Nat. Rev. Urol. 2010, 7, 653–660. [Google Scholar] [CrossRef] [PubMed]
- Wolfertz, N.; Bohm, L.; Keitel, V.; Hannappel, O.; Kumpers, P.; Bernhard, M.; Michael, M. Epidemiology, management, and outcome of infection, sepsis, and septic shock in a German emergency department (EpiSEP study). Front. Med. 2022, 9, 997992. [Google Scholar] [CrossRef] [PubMed]
- Danmarks_Statistik. Danmarks Statistik, Statistikbanken.dk. Available online: https://statistikbanken.dk/ind04 (accessed on 24 May 2023).
- Pedersen, G.; Schonheyder, H.C.; Sorensen, H.T. Source of infection and other factors associated with case fatality in community-acquired bacteremia--a Danish population-based cohort study from 1992 to 1997. Clin. Microbiol. Infect. 2003, 9, 793–802. [Google Scholar] [CrossRef]
- Dubbs, S.B.; Sommerkamp, S.K. Evaluation and Management of Urinary Tract Infection in the Emergency Department. Emerg. Med. Clin. N. Am. 2019, 37, 707–723. [Google Scholar] [CrossRef]
- Long, B.; Koyfman, A. The Emergency Department Diagnosis and Management of Urinary Tract Infection. Emerg. Med. Clin. N. Am. 2018, 36, 685–710. [Google Scholar] [CrossRef]
- Chernaya, A.; Soborg, C.; Midttun, M. Validity of the urinary dipstick test in the diagnosis of urinary tract infections in adults. Dan. Med. J. 2021, 69, A07210607. [Google Scholar]
- Chu, C.M.; Lowder, J.L. Diagnosis and treatment of urinary tract infections across age groups. Am. J. Obstet. Gynecol. 2018, 219, 40–51. [Google Scholar] [CrossRef]
- Caterino, J.M.; Stevenson, K.B. Disagreement between emergency physician and inpatient physician diagnosis of infection in older adults admitted from the emergency department. Acad. Emerg. Med. 2012, 19, 908–915. [Google Scholar] [CrossRef]
- Gupta, A.; Petty, L.; Gandhi, T.; Flanders, S.; Hsaiky, L.; Basu, T.; Zhang, Q.; Horowitz, J.; Masood, Z.; Chopra, V.; et al. Overdiagnosis of urinary tract infection linked to overdiagnosis of pneumonia: A multihospital cohort study. BMJ Qual. Saf. 2022, 31, 383–386. [Google Scholar] [CrossRef]
- Evans, L.; Rhodes, A.; Alhazzani, W.; Antonelli, M.; Coopersmith, C.M.; French, C.; Machado, F.R.; McIntyre, L.; Ostermann, M.; Prescott, H.C.; et al. Surviving Sepsis Campaign: International Guidelines for Management of Sepsis and Septic Shock 2021. Crit. Care Med. 2021, 49, e1063–e1143. [Google Scholar] [CrossRef]
- Gupta, K.; Hooton, T.M.; Naber, K.G.; Wullt, B.; Colgan, R.; Miller, L.G.; Moran, G.J.; Nicolle, L.E.; Raz, R.; Schaeffer, A.J.; et al. International clinical practice guidelines for the treatment of acute uncomplicated cystitis and pyelonephritis in women: A 2010 update by the Infectious Diseases Society of America and the European Society for Microbiology and Infectious Diseases. Clin. Infect. Dis. 2011, 52, e103–e120. [Google Scholar] [CrossRef]
- Deftos, L.J.; Roos, B.A.; Parthemore, J.G. Calcium and skeletal metabolism. West. J. Med. 1975, 123, 447–458. [Google Scholar]
- Hoeboer, S.H.; van der Geest, P.J.; Nieboer, D.; Groeneveld, A.B. The diagnostic accuracy of procalcitonin for bacteraemia: A systematic review and meta-analysis. Clin. Microbiol. Infect. 2015, 21, 474–481. [Google Scholar] [CrossRef]
- Kapasi, A.J.; Dittrich, S.; Gonzalez, I.J.; Rodwell, T.C. Host Biomarkers for Distinguishing Bacterial from Non-Bacterial Causes of Acute Febrile Illness: A Comprehensive Review. PLoS ONE 2016, 11, e0160278. [Google Scholar] [CrossRef]
- Masajtis-Zagajewska, A.; Kurnatowska, I.; Wajdlich, M.; Nowicki, M. Utility of copeptin and standard inflammatory markers in the diagnostics of upper and lower urinary tract infections. BMC Urol. 2015, 15, 67. [Google Scholar] [CrossRef]
- Choi, J.J.; McCarthy, M.W.; Meltzer, K.K.; Cornelius-Schecter, A.; Jabri, A.; Reshetnyak, E.; Banerjee, S.; Westblade, L.F.; Mehta, S.; Simon, M.S.; et al. The Diagnostic Accuracy Of Procalcitonin for Urinary Tract Infection in Hospitalized Older Adults: A Prospective Study. J. Gen. Intern. Med. 2022, 37, 3663–3669. [Google Scholar] [CrossRef]
- Levine, A.R.; Tran, M.; Shepherd, J.; Naut, E. Utility of initial procalcitonin values to predict urinary tract infection. Am. J. Emerg. Med. 2018, 36, 1993–1997. [Google Scholar] [CrossRef]
- Xu, R.Y.; Liu, H.W.; Liu, J.L.; Dong, J.H. Procalcitonin and C-reactive protein in urinary tract infection diagnosis. BMC Urol. 2014, 14, 45. [Google Scholar] [CrossRef]
- Thuno, M.; Macho, B.; Eugen-Olsen, J. suPAR: The molecular crystal ball. Dis. Markers 2009, 27, 157–172. [Google Scholar] [CrossRef]
- Behrendt, N.; Ploug, M.; Patthy, L.; Houen, G.; Blasi, F.; Danø, K. The ligand-binding domain of the cell surface receptor for urokinase-type plasminogen activator. J. Biol. Chem. 1991, 266, 7842–7847. [Google Scholar] [CrossRef]
- Ni, W.; Han, Y.; Zhao, J.; Cui, J.; Wang, K.; Wang, R.; Liu, Y. Serum soluble urokinase-type plasminogen activator receptor as a biological marker of bacterial infection in adults: A systematic review and meta-analysis. Sci. Rep. 2016, 6, 39481. [Google Scholar] [CrossRef]
- Velissaris, D.; Zareifopoulos, N.; Karamouzos, V.; Pierrakos, C.; Karanikolas, M. Soluble urokinase plasminogen activator receptor (suPAR) in the emergency department: An update. Caspian J. Intern. Med. 2022, 13, 650–665. [Google Scholar] [CrossRef]
- Wittenhagen, P.; Andersen, J.B.; Hansen, A.; Lindholm, L.; Ronne, F.; Theil, J.; Tvede, M.; Eugen-Olsen, J. Plasma soluble urokinase plasminogen activator receptor in children with urinary tract infection. Biomark. Insights 2011, 6, 79–82. [Google Scholar] [CrossRef]
- Gewurz, H.; Mold, C.; Siegel, J.; Fiedel, B. C-reactive protein and the acute phase response. Adv. Intern. Med. 1982, 27, 345–372. [Google Scholar] [CrossRef]
- Tillett, W.S.; Francis, T. Serological Reactions in Pneumonia with a Non-Protein Somatic Fraction of Pneumococcus. J. Exp. Med. 1930, 52, 561–571. [Google Scholar] [CrossRef]
- Richards, D.; Toop, L.; Chambers, S.; Fletcher, L. Response to antibiotics of women with symptoms of urinary tract infection but negative dipstick urine test results: Double blind randomised controlled trial. BMJ 2005, 331, 143. [Google Scholar] [CrossRef]
- Shallcross, L.; Gaskell, K.; Fox-Lewis, A.; Bergstrom, M.; Noursadeghi, M. Mismatch between suspected pyelonephritis and microbiological diagnosis: A cohort study from a UK teaching hospital. J. Hosp. Infect. 2018, 98, 219–222. [Google Scholar] [CrossRef]
- Vaughn, V.M.; Gupta, A.; Petty, L.A.; Malani, A.N.; Osterholzer, D.; Patel, P.K.; Younas, M.; Bernstein, S.J.; Burdick, S.; Ratz, D.; et al. A Statewide Quality Initiative to Reduce Unnecessary Antibiotic Treatment of Asymptomatic Bacteriuria. JAMA Intern. Med. 2023, 183, 933–941. [Google Scholar] [CrossRef]
- Bilsen, M.P.; Jongeneel, R.M.H.; Schneeberger, C.; Platteel, T.N.; van Nieuwkoop, C.; Mody, L.; Caterino, J.M.; Geerlings, S.E.; Koves, B.; Wagenlehner, F.; et al. Definitions of Urinary Tract Infection in Current Research: A Systematic Review. Open Forum Infect. Dis. 2023, 10, ofad332. [Google Scholar] [CrossRef]
- Skjot-Arkil, H.; Heltborg, A.; Lorentzen, M.H.; Cartuliares, M.B.; Hertz, M.A.; Graumann, O.; Rosenvinge, F.S.; Petersen, E.R.B.; Ostergaard, C.; Laursen, C.B.; et al. Improved diagnostics of infectious diseases in emergency departments: A protocol of a multifaceted multicentre diagnostic study. BMJ Open 2021, 11, e049606. [Google Scholar] [CrossRef]
- Bossuyt, P.M.; Reitsma, J.B.; Bruns, D.E.; Gatsonis, C.A.; Glasziou, P.P.; Irwig, L.; Lijmer, J.G.; Moher, D.; Rennie, D.; de Vet, H.C.; et al. STARD 2015: An updated list of essential items for reporting diagnostic accuracy studies. BMJ 2015, 351, h5527. [Google Scholar] [CrossRef]
- Shi, J.; Zhan, Z.S.; Zheng, Z.S.; Zhu, X.X.; Zhou, X.Y.; Zhang, S.Y. Correlation of procalcitonin and c-reactive protein levels with pathogen distribution and infection localization in urinary tract infections. Sci. Rep. 2023, 13, 17164. [Google Scholar] [CrossRef]
- Caffarini, E.M.; DeMott, J.; Patel, G.; Lat, I. Determining the Clinical Utility of an Absolute Procalcitonin Value for Predicting a Positive Culture Result. Antimicrob. Agents Chemother. 2017, 61, e02007-16. [Google Scholar] [CrossRef]
- Ha, Y.E.; Kang, C.I.; Wi, Y.M.; Chung, D.R.; Kang, E.S.; Lee, N.Y.; Song, J.H.; Peck, K.R. Diagnostic usefulness of procalcitonin as a marker of bacteremia in patients with acute pyelonephritis. Scand. J. Clin. Lab. Investig. 2013, 73, 444–448. [Google Scholar] [CrossRef]
- Julian-Jimenez, A.; Gutierrez-Martin, P.; Lizcano-Lizcano, A.; Lopez-Guerrero, M.A.; Barroso-Manso, A.; Heredero-Galvez, E. Usefulness of procalcitonin and C-reactive protein for predicting bacteremia in urinary tract infections in the emergency department. Actas Urol. Esp. 2015, 39, 502–510. [Google Scholar] [CrossRef]
- Varela-Patino, M.; Lopez-Izquierdo, R.; Velayos-Garcia, P.; Alvarez-Manzanares, J.; Ramos-Sanchez, C.; Carbajosa-Rodriguez, V.; Martin-Rodriguez, F.; Eiros, J.M. Usefulness of infection biomarkers for diagnosing bacteremia in patients with a sepsis code in the emergency department. Infez. Med. 2020, 28, 29–36. [Google Scholar]
- Kofoed, K.; Andersen, O.; Kronborg, G.; Tvede, M.; Petersen, J.; Eugen-Olsen, J.; Larsen, K. Use of plasma C-reactive protein, procalcitonin, neutrophils, macrophage migration inhibitory factor, soluble urokinase-type plasminogen activator receptor, and soluble triggering receptor expressed on myeloid cells-1 in combination to diagnose infections: A prospective study. Crit. Care 2007, 11, R38. [Google Scholar] [CrossRef]
- Latour, K.; De Lepeleire, J.; Catry, B.; Buntinx, F. Nursing home residents with suspected urinary tract infections: A diagnostic accuracy study. BMC Geriatr. 2022, 22, 187. [Google Scholar] [CrossRef]
- Shaikh, N.; Borrell, J.L.; Evron, J.; Leeflang, M.M. Procalcitonin, C-reactive protein, and erythrocyte sedimentation rate for the diagnosis of acute pyelonephritis in children. Cochrane Database Syst. Rev. 2015, 1, CD009185. [Google Scholar] [CrossRef] [PubMed]
- Shigemura, K.; Tanaka, K.; Osawa, K.; Arakawa, S.; Miyake, H.; Fujisawa, M. Clinical factors associated with shock in bacteremic UTI. Int. Urol. Nephrol. 2013, 45, 653–657. [Google Scholar] [CrossRef] [PubMed]
- Yilmaz, G.; Koksal, I.; Karahan, S.C.; Mentese, A. The diagnostic and prognostic significance of soluble urokinase plasminogen activator receptor in systemic inflammatory response syndrome. Clin. Biochem. 2011, 44, 1227–1230. [Google Scholar] [CrossRef] [PubMed]
| Expert Panel Diagnosis | UTI | Non-UTI |
|---|---|---|
| no. (%) | 149 (65.1%) | 80 (34.9%) |
| Clinical characteristics | ||
| Age, years, median (IQR), n = 229 | 75 (17) | 76.5 (18.5) |
| Sex, male, no. (%), n = 229 | 88 (59.1%) | 43 (53.8%) |
| UTI symptoms, no. (%), n = 217 | 106 (75.2%) | 47 (61.8%) |
| Positive urine culture, no. (%), n = 216 | 104 (74.8%) | 30 (39%) |
| UTI symptoms & positive urine culture, no. (%), n = 206 | 70 (52.6%) | 15 (20.6%) |
| Laboratory results | ||
| PCT, median (IQR), μg/L, n = 196 | 0.53 (2.3) | 0.14 (0.36) |
| suPAR, median (IQR), μg/L, n = 195 | 7.2 (4.7) | 6.3 (5.3) |
| CRP, median (IQR), mg/L, n = 229 | 136 (126) | 50 (118) |
| Severity of disease (only UTI), n = 149 | ||
| Mild (cystitis), no. (%) | 29 (19.5%) | - |
| Severe (pyelonephritis or urosepsis, no. (%) | 120 (80.5%) | - |
| Blood cultures, n = 201 | ||
| Positive, no. (%) | 39 (28.7%) | 8 (12.3%) |
| Negative, no. (%) | 97 (71.3%) | 57 (87.7%) |
| Index Test | Reference Test | n | Model AUROC | EEO AUROC | Non-Inf AUROC | Cut-Off | Sens | Spec | PPV | NPV | DA | TP | FP | FN | TN |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| PCT | UTI | 196 | 0.717 | 0.648 | 0.612 | 0.43 μg/L | 57.5% | 78.3% | 83.0% | 50.0% | 64.8% | 73 | 15 | 54 | 54 |
| Severity | 127 | 0.712 | 0.712 | - | 0.08 μg/L | 95.1% | 25.0% | 84.5% | 54.5% | 81.9% | 98 | 18 | 5 | 6 | |
| Bacteremia | 173 | 0.809 | 0.858 | 0.777 | 0.15 μg/L | 94.9% | 38.8% | 31.1% | 96.3% | 51.4% | 37 | 82 | 2 | 52 | |
| suPAR | UTI | 195 | 0.583 | 0.581 | 0.480 | 6.5 μg/L | 66.1% | 54.4% | 73.0% | 46.3% | 62.1% | 84 | 31 | 43 | 37 |
| Severity | 127 | 0.576 | 0.638 | - | 3.74 μg/L | 95.1% | 0.0% | 80.3% | 0.0% | 77.2% | 98 | 24 | 5 | 0 | |
| Bacteremia | 172 | 0.637 | 0.679 | 0.605 | 4.62 μg/L | 94.9% | 19.5% | 25.7% | 92.9% | 36.6% | 37 | 107 | 2 | 26 | |
| CRP | UTI | 229 | 0.723 | 0.771 | 0.599 | 71 mg/L | 77.9% | 58.8% | 77.9% | 58.8% | 71.2% | 116 | 33 | 33 | 47 |
| Severity | 149 | 0.676 | 0.778 | - | 19 mg/L | 95.0% | 20.7% | 83.2% | 50.0% | 80.5% | 114 | 23 | 6 | 6 | |
| Bacteremia | 201 | 0.689 | 0.782 | 0.646 | 14 mg/L | 95.7% | 13.6% | 25.3% | 91.3% | 32.8% | 45 | 133 | 2 | 21 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hertz, M.A.; Johansen, I.S.; Rosenvinge, F.S.; Brasen, C.L.; Andersen, E.S.; Heltborg, A.; Skovsted, T.A.; Petersen, E.R.B.; Cartuliares, M.B.; Nielsen, S.L.; et al. The Diagnostic Accuracy of Procalcitonin, Soluble Urokinase-Type Plasminogen Activator Receptors, and C-Reactive Protein in Diagnosing Urinary Tract Infections in the Emergency Department—A Diagnostic Accuracy Study. J. Clin. Med. 2024, 13, 1776. https://doi.org/10.3390/jcm13061776
Hertz MA, Johansen IS, Rosenvinge FS, Brasen CL, Andersen ES, Heltborg A, Skovsted TA, Petersen ERB, Cartuliares MB, Nielsen SL, et al. The Diagnostic Accuracy of Procalcitonin, Soluble Urokinase-Type Plasminogen Activator Receptors, and C-Reactive Protein in Diagnosing Urinary Tract Infections in the Emergency Department—A Diagnostic Accuracy Study. Journal of Clinical Medicine. 2024; 13(6):1776. https://doi.org/10.3390/jcm13061776
Chicago/Turabian StyleHertz, Mathias Amdi, Isik Somuncu Johansen, Flemming S. Rosenvinge, Claus Lohman Brasen, Eline Sandvig Andersen, Anne Heltborg, Thor Aage Skovsted, Eva Rabing Brix Petersen, Mariana Bichuette Cartuliares, Stig Lønberg Nielsen, and et al. 2024. "The Diagnostic Accuracy of Procalcitonin, Soluble Urokinase-Type Plasminogen Activator Receptors, and C-Reactive Protein in Diagnosing Urinary Tract Infections in the Emergency Department—A Diagnostic Accuracy Study" Journal of Clinical Medicine 13, no. 6: 1776. https://doi.org/10.3390/jcm13061776






