Metabolomic Profiles Predict Diabetes Remission after Bariatric Surgery
Abstract
1. Introduction
2. Methods
2.1. Study Participants
2.2. Surgical Procedures
2.3. Management of Nutrition and Blood Glucose
2.4. Measurements of Serum AAMs
2.5. Outcome Measures
2.6. Statistical Analysis
3. Results
3.1. Patients’ Baseline Characteristics
3.2. Patients’ Characteristics at 3 and 12 Months Postoperatively
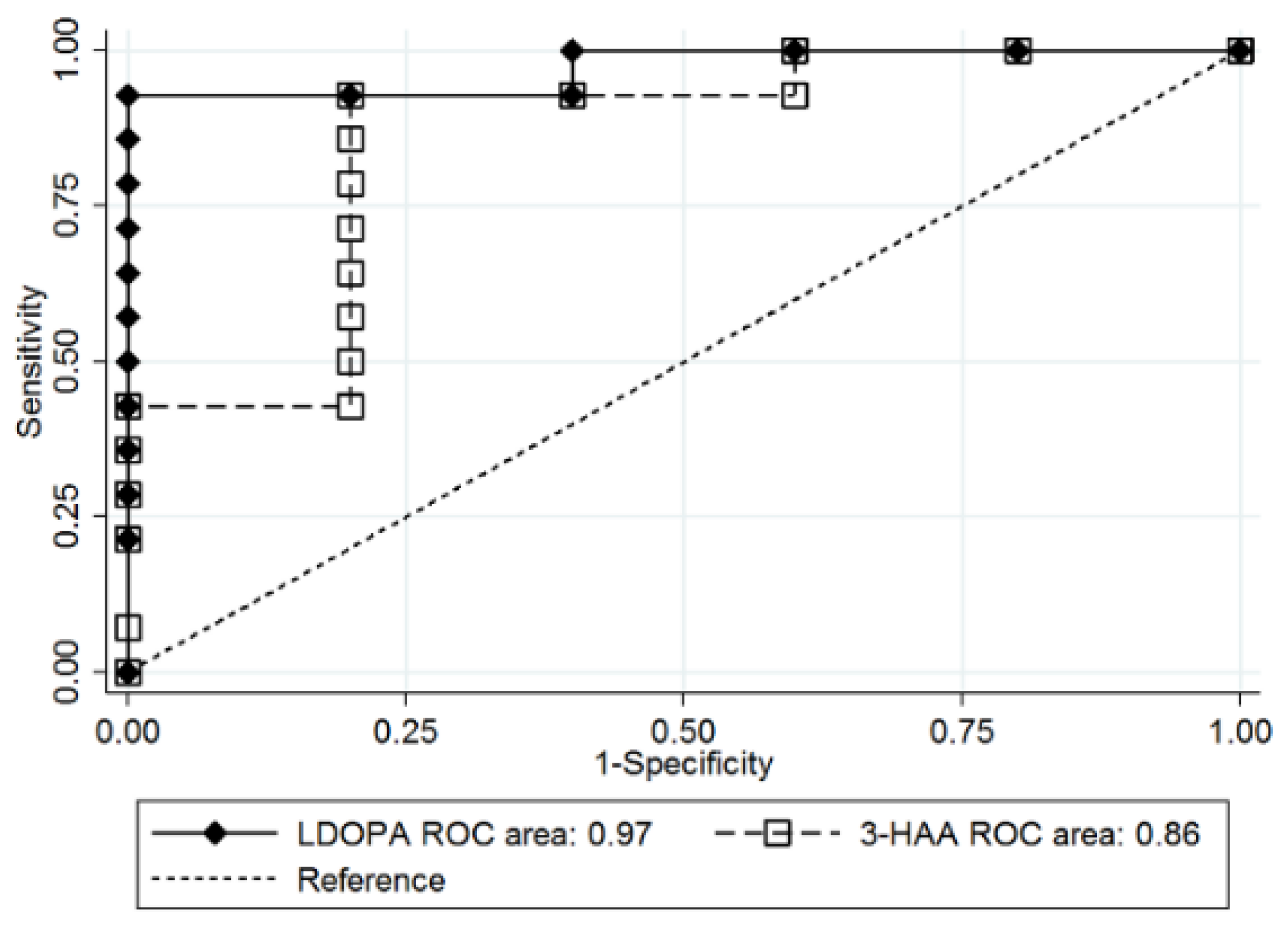
3.3. Prediction of T2D Remission after Bariatric Surgery
3.4. Postoperative Changes in L-DOPA and 3-HAA
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Mingrone, G.; Panunzi, S.; De Gaetano, A.; Guidone, C.; Iaconelli, A.; Leccesi, L.; Nanni, G.; Pomp, A.; Castagneto, M.; Ghirlanda, G.; et al. Bariatric surgery versus conventional medical therapy for type 2 diabetes. N. Engl. J. Med. 2012, 366, 1577–1585. [Google Scholar] [CrossRef] [PubMed]
- Schauer, P.R.; Kashyap, S.R.; Wolski, K.; Brethauer, S.A.; Kirwan, J.P.; Pothier, C.E.; Thomas, S.; Abood, B.; Nissen, S.E.; Bhatt, D.L. Bariatric surgery versus intensive medical therapy in obese patients with diabetes. N. Engl. J. Med. 2012, 366, 1567–1576. [Google Scholar] [CrossRef] [PubMed]
- American Diabetes Association. 8. Obesity Management for the Treatment of Type 2 Diabetes: Standards of Medical Care in Diabetes-2019. Diabetes Care 2019, 42, S81–S89. [Google Scholar] [CrossRef] [PubMed]
- Rubino, F.; Nathan, D.M.; Eckel, R.H.; Schauer, P.R.; Alberti, K.G.; Zimmet, P.Z.; Del Prato, S.; Ji, L.; Sadikot, S.M.; Herman, W.H.; et al. Metabolic Surgery in the Treatment Algorithm for Type 2 Diabetes: A Joint Statement by International Diabetes Organizations. Diabetes Care 2016, 39, 861–877. [Google Scholar] [CrossRef]
- Schauer, P.R.; Bhatt, D.L.; Kirwan, J.P.; Wolski, K.; Aminian, A.; Brethauer, S.A.; Navaneethan, S.D.; Singh, R.P.; Pothier, C.E.; Nissen, S.E.; et al. Bariatric Surgery versus Intensive Medical Therapy for Diabetes—5-Year Outcomes. N. Engl. J. Med. 2017, 376, 641–651. [Google Scholar] [CrossRef]
- Sjostrom, L.; Peltonen, M.; Jacobson, P.; Ahlin, S.; Andersson-Assarsson, J.; Anveden, A.; Bouchard, C.; Carlsson, B.; Karason, K.; Lonroth, H.; et al. Association of bariatric surgery with long-term remission of type 2 diabetes and with microvascular and macrovascular complications. JAMA 2014, 311, 2297–2304. [Google Scholar] [CrossRef]
- Floegel, A.; Stefan, N.; Yu, Z.; Muhlenbruch, K.; Drogan, D.; Joost, H.G.; Fritsche, A.; Haring, H.U.; Hrabe de Angelis, M.; Peters, A.; et al. Identification of serum metabolites associated with risk of type 2 diabetes using a targeted metabolomic approach. Diabetes 2013, 62, 639–648. [Google Scholar] [CrossRef]
- Roberts, L.D.; Koulman, A.; Griffin, J.L. Towards metabolic biomarkers of insulin resistance and type 2 diabetes: Progress from the metabolome. Lancet Diabetes Endocrinol. 2014, 2, 65–75. [Google Scholar] [CrossRef]
- Wang, T.J.; Larson, M.G.; Vasan, R.S.; Cheng, S.; Rhee, E.P.; McCabe, E.; Lewis, G.D.; Fox, C.S.; Jacques, P.F.; Fernandez, C.; et al. Metabolite profiles and the risk of developing diabetes. Nat. Med. 2011, 17, 448–453. [Google Scholar] [CrossRef]
- Wurtz, P.; Soininen, P.; Kangas, A.J.; Ronnemaa, T.; Lehtimaki, T.; Kahonen, M.; Viikari, J.S.; Raitakari, O.T.; Ala-Korpela, M. Branched-chain and aromatic amino acids are predictors of insulin resistance in young adults. Diabetes Care 2013, 36, 648–655. [Google Scholar] [CrossRef]
- Le Floc’h, N.; Otten, W.; Merlot, E. Tryptophan metabolism, from nutrition to potential therapeutic applications. Amino Acids 2011, 41, 1195–1205. [Google Scholar] [CrossRef] [PubMed]
- Oxenkrug, G. Insulin resistance and dysregulation of tryptophan-kynurenine and kynurenine-nicotinamide adenine dinucleotide metabolic pathways. Mol. Neurobiol. 2013, 48, 294–301. [Google Scholar] [CrossRef] [PubMed]
- Pedersen, E.R.; Tuseth, N.; Eussen, S.J.; Ueland, P.M.; Strand, E.; Svingen, G.F.; Midttun, O.; Meyer, K.; Mellgren, G.; Ulvik, A.; et al. Associations of plasma kynurenines with risk of acute myocardial infarction in patients with stable angina pectoris. Arterioscler. Thromb. Vasc. Biol. 2015, 35, 455–462. [Google Scholar] [CrossRef] [PubMed]
- Yabut, J.M.; Crane, J.D.; Green, A.E.; Keating, D.J.; Khan, W.I.; Steinberg, G.R. Emerging Roles for Serotonin in Regulating Metabolism: New Implications for an Ancient Molecule. Endocr. Rev. 2019, 40, 1092–1107. [Google Scholar] [CrossRef] [PubMed]
- Nash, A.I. Crosstalk between insulin and dopamine signaling: A basis for the metabolic effects of antipsychotic drugs. J. Chem. Neuroanat. 2017, 83–84, 59–68. [Google Scholar] [CrossRef] [PubMed]
- Ustione, A.; Piston, D.W.; Harris, P.E. Minireview: Dopaminergic regulation of insulin secretion from the pancreatic islet. Mol. Endocrinol. 2013, 27, 1198–1207. [Google Scholar] [CrossRef]
- Park, Y.S.; Park, D.J.; Lee, J.H.; Lee, H.J.; Ha, T.K.; Kim, Y.J.; Ryu, S.W.; Han, S.M.; Yoo, M.W.; Park, S.; et al. Korean OBEsity Surgical Treatment Study (KOBESS): Protocol of a prospective multicentre cohort study on obese patients undergoing laparoscopic sleeve gastrectomy and Roux-en-Y gastric bypass. BMJ Open 2017, 7, e018044. [Google Scholar] [CrossRef]
- Parrott, J.; Frank, L.; Rabena, R.; Craggs-Dino, L.; Isom, K.A.; Greiman, L. American Society for Metabolic and Bariatric Surgery Integrated Health Nutritional Guidelines for the Surgical Weight Loss Patient 2016 Update: Micronutrients. Surg. Obes. Relat. Dis. 2017, 13, 727–741. [Google Scholar] [CrossRef]
- Lee, W.J.; Almulaifi, A.; Chong, K.; Chen, S.C.; Tsou, J.J.; Ser, K.H.; Lee, Y.C.; Chen, J.C. The Effect and Predictive Score of Gastric Bypass and Sleeve Gastrectomy on Type 2 Diabetes Mellitus Patients with BMI < 30 kg/m(2). Obes. Surg. 2015, 25, 1772–1778. [Google Scholar] [CrossRef]
- Still, C.D.; Wood, G.C.; Benotti, P.; Petrick, A.T.; Gabrielsen, J.; Strodel, W.E.; Ibele, A.; Seiler, J.; Irving, B.A.; Celaya, M.P.; et al. Preoperative prediction of type 2 diabetes remission after Roux-en-Y gastric bypass surgery: A retrospective cohort study. Lancet Diabetes Endocrinol. 2014, 2, 38–45. [Google Scholar] [CrossRef]
- Aminian, A.; Brethauer, S.A.; Andalib, A.; Nowacki, A.S.; Jimenez, A.; Corcelles, R.; Hanipah, Z.N.; Punchai, S.; Bhatt, D.L.; Kashyap, S.R.; et al. Individualized Metabolic Surgery Score: Procedure Selection Based on Diabetes Severity. Ann. Surg. 2017, 266, 650–657. [Google Scholar] [CrossRef] [PubMed]
- Schwarcz, R. The kynurenine pathway of tryptophan degradation as a drug target. Curr. Opin. Pharmacol. 2004, 4, 12–17. [Google Scholar] [CrossRef] [PubMed]
- Brandacher, G.; Hoeller, E.; Fuchs, D.; Weiss, H.G. Chronic immune activation underlies morbid obesity: Is IDO a key player? Curr. Drug Metab. 2007, 8, 289–295. [Google Scholar] [CrossRef] [PubMed]
- Oxenkrug, G.; van der Hart, M.; Summergrad, P. Elevated anthranilic acid plasma concentrations in type 1 but not type 2 diabetes mellitus. Integr. Mol. Med. 2015, 2, 365–368. [Google Scholar] [CrossRef][Green Version]
- Muzik, O.; Burghardt, P.; Yi, Z.; Kumar, A.; Seyoum, B. Successful metformin treatment of insulin resistance is associated with down-regulation of the kynurenine pathway. Biochem. Biophys. Res. Commun. 2017, 488, 29–32. [Google Scholar] [CrossRef]
- Oxenkrug, G.F. Increased Plasma Levels of Xanthurenic and Kynurenic Acids in Type 2 Diabetes. Mol. Neurobiol. 2015, 52, 805–810. [Google Scholar] [CrossRef]
- Christensen, M.H.E.; Fadnes, D.J.; Rost, T.H.; Pedersen, E.R.; Andersen, J.R.; Vage, V.; Ulvik, A.; Midttun, O.; Ueland, P.M.; Nygard, O.K.; et al. Inflammatory markers, the tryptophan-kynurenine pathway, and vitamin B status after bariatric surgery. PLoS ONE 2018, 13, e0192169. [Google Scholar] [CrossRef]
- Krause, D.; Suh, H.S.; Tarassishin, L.; Cui, Q.L.; Durafourt, B.A.; Choi, N.; Bauman, A.; Cosenza-Nashat, M.; Antel, J.P.; Zhao, M.L.; et al. The tryptophan metabolite 3-hydroxyanthranilic acid plays anti-inflammatory and neuroprotective roles during inflammation: Role of hemeoxygenase-1. Am. J. Pathol. 2011, 179, 1360–1372. [Google Scholar] [CrossRef]
- Farino, Z.J.; Morgenstern, T.J.; Maffei, A.; Quick, M.; De Solis, A.J.; Wiriyasermkul, P.; Freyberg, R.J.; Aslanoglou, D.; Sorisio, D.; Inbar, B.P.; et al. New roles for dopamine D2 and D3 receptors in pancreatic beta cell insulin secretion. Mol. Psychiatry 2019. [Google Scholar] [CrossRef]
- Rubi, B.; Ljubicic, S.; Pournourmohammadi, S.; Carobbio, S.; Armanet, M.; Bartley, C.; Maechler, P. Dopamine D2-like receptors are expressed in pancreatic beta cells and mediate inhibition of insulin secretion. J. Biol. Chem. 2005, 280, 36824–36832. [Google Scholar] [CrossRef]
- Shankar, E.; Santhosh, K.T.; Paulose, C.S. Dopaminergic regulation of glucose-induced insulin secretion through dopamine D2 receptors in the pancreatic islets in vitro. IUBMB Life 2006, 58, 157–163. [Google Scholar] [CrossRef] [PubMed]
- Simpson, N.; Maffei, A.; Freeby, M.; Burroughs, S.; Freyberg, Z.; Javitch, J.; Leibel, R.L.; Harris, P.E. Dopamine-mediated autocrine inhibitory circuit regulating human insulin secretion in vitro. Mol. Endocrinol. 2012, 26, 1757–1772. [Google Scholar] [CrossRef] [PubMed]
- Rubino, F.; R’Bibo, S.L.; del Genio, F.; Mazumdar, M.; McGraw, T.E. Metabolic surgery: The role of the gastrointestinal tract in diabetes mellitus. Nat. Rev. Endocrinol. 2010, 6, 102–109. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Eisenhofer, G.; Aneman, A.; Friberg, P.; Hooper, D.; Fandriks, L.; Lonroth, H.; Hunyady, B.; Mezey, E. Substantial production of dopamine in the human gastrointestinal tract. J. Clin. Endocrinol. Metab. 1997, 82, 3864–3871. [Google Scholar] [CrossRef]
- Ha, J.; Kwon, Y.; Kim, N.H.; Park, S.; Menzo, E.L.; Rosenthal, R.J. Discordance in prediction for prognosis of type 2 diabetes after metabolic surgery: Comparison of the ABCD, DiaRem, and individualized metabolic surgery models. Ann. Surg. Treat. Res. 2019, 97, 309–318. [Google Scholar] [CrossRef]
- Fontana, L.; Cummings, N.E.; Arriola Apelo, S.I.; Neuman, J.C.; Kasza, I.; Schmidt, B.A.; Cava, E.; Spelta, F.; Tosti, V.; Syed, F.A.; et al. Decreased Consumption of Branched-Chain Amino Acids Improves Metabolic Health. Cell Rep. 2016, 16, 520–530. [Google Scholar] [CrossRef]
- Zhang, Y.; Guo, K.; LeBlanc, R.E.; Loh, D.; Schwartz, G.J.; Yu, Y.H. Increasing dietary leucine intake reduces diet-induced obesity and improves glucose and cholesterol metabolism in mice via multimechanisms. Diabetes 2007, 56, 1647–1654. [Google Scholar] [CrossRef]


| Variables | Diabetes Status 1 Year after Bariatric Surgery | p Value | |
|---|---|---|---|
| Remission (n = 14) | Non-Remission (n = 10) | ||
| Age, y | 41.0 ± 8.7 | 54.8 ± 8.0 | 0.004 |
| Female sex, no. (%) | 12 (85.7) | 5 (50.0) | 0.058 |
| Body mass index, kg/m2 | 39.6 ± 7.9 | 33.9 ± 4.5 | 0.116 |
| Body weight, kg | 106.1 ± 31.2 | 89.7 ± 18.4 | 0.251 |
| Waist circumference, cm | 120.5 ± 19.9 | 112.1 ± 9.7 | 0.347 |
| Waist-to-hip ratio | 0.97 ± 0.05 | 1.03 ± 0.01 | 0.017 |
| Duration of diabetes, y | 2.2 ± 1.4 | 8.9 ± 8.6 | 0.009 |
| Use of insulin, no. (%) | 2 (14.3) | 3 (30.0) | 0.350 |
| Current smoker, no. (%) | 3 (21.4) | 2 (20.0) | 0.932 |
| Hypertension, no. (%) | 5 (35.7) | 5 (50.0) | 0.484 |
| Dyslipidemia, no. (%) | 9 (64.2) | 6 (60.0) | 0.831 |
| Surgical methods (RYGB/SG), no. (%) | 5/9 (35.7/64.3) | 5/5 (50.0/50.0) | 0.484 |
| ABCD score | 6.2 ± 2.0 | 3.3 ± 2.7 | 0.015 |
| DiaRem score | 4.7 ± 5.4 | 12.0 ± 5.3 | 0.013 |
| IMS score | 39.3 ± 22.9 | 88.2 ± 30.6 | 0.001 |
| Variables | Remission | Non-Remission | p Value for Baseline | p Value for 3 Months | p Value for 12 Months | ||||
|---|---|---|---|---|---|---|---|---|---|
| Baseline (n = 14) | 3 Months (n = 14) | 12 Months (n = 14) | Baseline (n = 10) | 3 Months (n = 10) | 12 Months (n = 10) | ||||
| Glycated hemoglobin, % | 7.2 ± 2.0 | 5.6 ± 0.3 | 5.4 ± 0.2 | 8.9 ± 1.1 | 7.3 ± 1.1 | 7.6 ± 1.0 | 0.067 | <0.001 | <0.001 |
| Fasting plasma glucose, mg/dL | 151.1 ± 65.3 | 98.3 ± 9.8 | 97.2 ± 7.8 | 146.1 ± 51.9 | 129 ± 22.9 | 131 ± 26.6 | 0.871 | <0.001 | <0.001 |
| Body mass index, kg/m2 | 39.6 ± 7.9 | 32.3 ± 7.7 | 28.8 ± 6.6 | 33.9 ± 4.5 | 29.4 ± 4.5 | 28.9 ± 5.6 | 0.116 | 0.409 | 0.974 |
| Body weight, kg | 106.1 ± 31.2 | 87.7 ± 29.8 | 77.0 ± 24.9 | 89.7 ± 18.4 | 77.9 ± 17.6 | 76.7 ± 20.1 | 0.251 | 0.471 | 0.977 |
| % Excess weight loss | - | 62.9 ± 31.5 | 90.6 ± 38.3 | - | 59.4 ± 24.7 | 68.8 ± 32.4 | - | 0.818 | 0.240 |
| % Weight loss | - | 18.2 ± 4.1 | 27.6 ± 5.2 | - | 13.4 ± 2.7 | 15.1 ± 5.8 | - | 0.019 | <0.001 |
| Waist circumference, cm | 120.5 ± 19.9 | 104.6 ± 21.0 | 93.8 ± 16.5 | 112.1 ± 9.7 | 98.4 ± 12.4 | 98.1 ± 12.5 | 0.347 | 0.515 | 0.573 |
| Systolic blood pressure, mmHg | 135.7 ± 9.8 | 125.1 ± 12.1 | 120.1 ± 13.1 | 136.8 ± 8.4 | 124.1 ± 11.5 | 130 ± 10.2 | 0.812 | 0.870 | 0.120 |
| Diastolic blood pressure, mmHg | 84.5 ± 9.8 | 77.6 ± 10.9 | 74.5 ± 9.1 | 85.0 ± 4.9 | 73.6 ± 12.7 | 77.1 ± 11.40 | 0.908 | 0.496 | 0.585 |
| High-density lipoprotein cholesterol, mg/dL | 49.5 ± 9.8 | 48.6 ± 10.7 | 60.1 ± 12.2 | 46.6 ± 8.5 | 46.1 ± 13.1 | 52.5 ± 11.2 | 0.549 | 0.671 | 0.208 |
| Triglycerides, mg/dL | 204.4 ± 232.3 | 110.6 ± 38.7 | 94.5 ± 36.0 | 174.6 ± 140.6 | 149.5 ± 88.6 | 139 ± 77.5 | 0.776 | 0.193 | 0.091 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ha, J.; Jang, M.; Kwon, Y.; Park, Y.S.; Park, D.J.; Lee, J.-H.; Lee, H.-J.; Ha, T.K.; Kim, Y.-J.; Han, S.-M.; et al. Metabolomic Profiles Predict Diabetes Remission after Bariatric Surgery. J. Clin. Med. 2020, 9, 3897. https://doi.org/10.3390/jcm9123897
Ha J, Jang M, Kwon Y, Park YS, Park DJ, Lee J-H, Lee H-J, Ha TK, Kim Y-J, Han S-M, et al. Metabolomic Profiles Predict Diabetes Remission after Bariatric Surgery. Journal of Clinical Medicine. 2020; 9(12):3897. https://doi.org/10.3390/jcm9123897
Chicago/Turabian StyleHa, Jane, Mi Jang, Yeongkeun Kwon, Young Suk Park, Do Joong Park, Joo-Ho Lee, Hyuk-Joon Lee, Tae Kyung Ha, Yong-Jin Kim, Sang-Moon Han, and et al. 2020. "Metabolomic Profiles Predict Diabetes Remission after Bariatric Surgery" Journal of Clinical Medicine 9, no. 12: 3897. https://doi.org/10.3390/jcm9123897
APA StyleHa, J., Jang, M., Kwon, Y., Park, Y. S., Park, D. J., Lee, J.-H., Lee, H.-J., Ha, T. K., Kim, Y.-J., Han, S.-M., Han, S.-U., Heo, Y., & Park, S. (2020). Metabolomic Profiles Predict Diabetes Remission after Bariatric Surgery. Journal of Clinical Medicine, 9(12), 3897. https://doi.org/10.3390/jcm9123897







