Local Serpin Treatment via Chitosan-Collagen Hydrogel after Spinal Cord Injury Reduces Tissue Damage and Improves Neurologic Function
Abstract
1. Introduction
2. Experimental Section
2.1. Ethical Considerations
2.2. Serp-1 Protein Expression and Purification
2.3. Preparation of Chitosan-Collagen Hydrogels
2.4. Neurosurgery: Dorsal Column Fine Forceps Crush Model
2.5. Post-Surgical Period
2.6. Neurological Tests
2.7. H&E and Immunohistochemistry
2.8. Pathology Imaging and Analysis
2.9. Statistics
3. Results
3.1. Treatment with Serp-1 Loaded into Chitosan-Collagen Hydrogel Improves Clinical Scores in SCI Rats
3.2. Treatment with Chitosan-Collagen Hydrogel with Serp-1 Reduces the Extent of Spinal Cord Damage
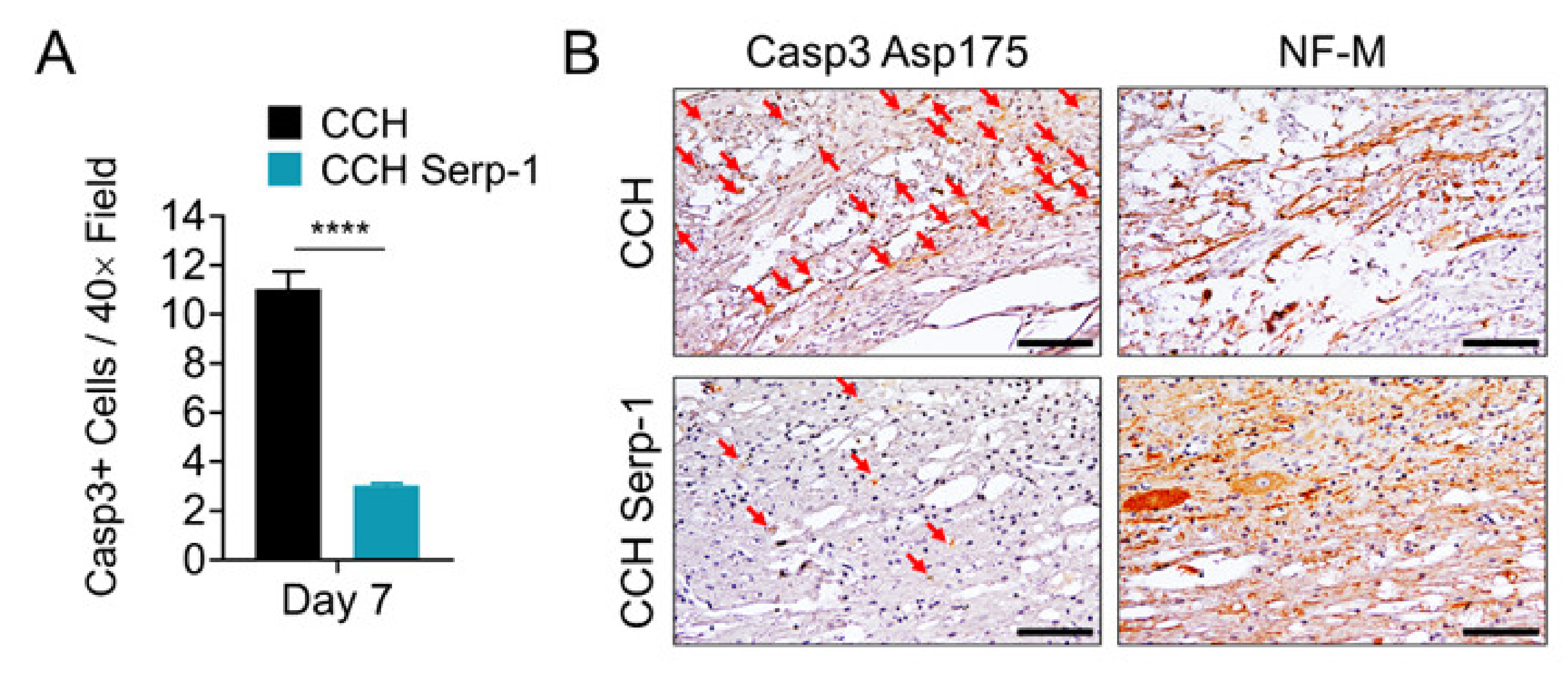
3.3. Treatment with Chitosan-Collagen Hydrogel Containing Serp-1 Reduces Apoptosis in Rat Spinal Cord after SCI
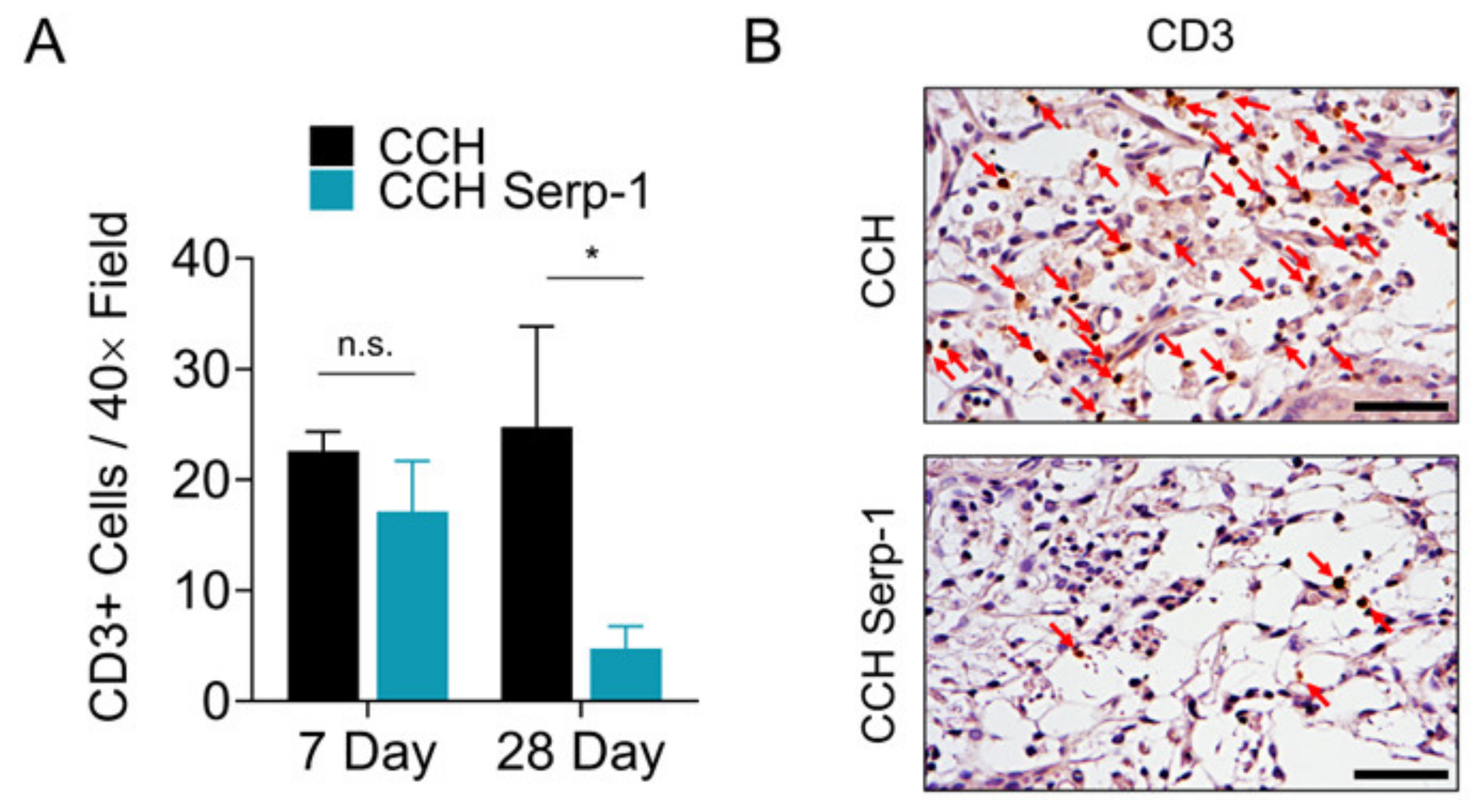
3.4. Treatment with Chitosan-Collagen Hydrogel with Serp-1 Reduces T Cell Infiltration in Spinal Cord in SCI Rats
3.5. Treatment with Chitosan-Collagen Hydrogel with Serp-1 Promotes Earlier Astrogliosis in the Rat Spinal Cord after SCI
4. Discussion and Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- James, S.L.; Theadom, A.; Ellenbogen, R.G.; Bannick, M.S.; Montjoy-Venning, W.; Lucchesi, L.R.; Abbasi, N.; Abdulkader, R.; Abraha, H.N.; Adsuar, J.C.; et al. Global, regional, and national burden of traumatic brain injury and spinal cord injury, 1990–2016: A systematic analysis for the Global Burden of Disease Study 2016. Lancet Neurol. 2019, 18, 56–87. [Google Scholar] [CrossRef]
- Bradbury, E.J.; Burnside, E.R. Moving beyond the glial scar for spinal cord repair. Nat. Commun. 2019, 10, 3879. [Google Scholar] [CrossRef]
- Kwiecien, J.M.; Dabrowski, W.; Dąbrowska-Bouta, B.; Sulkowski, G.; Oakden, W.; Kwiecien-Delaney, C.J.; Yaron, J.R.; Zhang, L.; Schutz, L.; Marzec-Kotarska, B.; et al. Prolonged inflammation leads to ongoing damage after spinal cord injury. PLoS ONE 2020, 15, e0226584. [Google Scholar] [CrossRef] [PubMed]
- Bowes, A.L.; Yip, P.K. Modulating inflammatory cell responses to spinal cord injury: All in good time. J. Neurotrauma 2014, 31, 1753–1766. [Google Scholar] [CrossRef] [PubMed]
- Bracken, M.B.; Shepard, M.J.; Collins, W.F.; Holford, T.R.; Young, W.; Baskin, D.S.; Eisenberg, H.M.; Flamm, E.; Leo-Summers, L.; Maroon, J. A randomized, controlled trial of methylprednisolone or naloxone in the treatment of acute spinal-cord injury. Results of the Second National Acute Spinal Cord Injury Study. N. Engl. J. Med. 1990, 322, 1405–1411. [Google Scholar] [CrossRef] [PubMed]
- Markandaya, M.; Stein, D.M.; Menaker, J. Acute Treatment Options for Spinal Cord Injury. Curr. Treat. Options Neurol. 2012, 14, 175–187. [Google Scholar] [CrossRef]
- Pettiford, J.N.; Bikhchandani, J.; Ostlie, D.J.; St. Peter, S.D.; Sharp, R.J.; Juang, D. A review: The role of high dose methylprednisolone in spinal cord trauma in children. Pediatr. Surg. Int. 2012, 28, 287–294. [Google Scholar] [CrossRef]
- Schoenfeld, A.J.; Laughlin, M.D.; McCriskin, B.J.; Bader, J.O.; Waterman, B.R.; Belmont, P.J. Spinal Injuries in United States Military Personnel Deployed to Iraq and Afghanistan: an epidemiological investigation involving 7877 combat casualties from 2005 to 2009. Spine 2013, 38, 1770–1778. [Google Scholar] [CrossRef]
- Shank, C.D.; Walters, B.C.; Hadley, M.N. Current Topics in the Management of Acute Traumatic Spinal Cord Injury. Neurocrit. Care 2019, 30, 261–271. [Google Scholar] [CrossRef]
- Ahuja, C.S.; Martin, A.R.; Fehlings, M. Recent advances in managing a spinal cord injury secondary to trauma. F1000Research 2016, 5, 1017. [Google Scholar] [CrossRef]
- Guizar-Sahagun, G.; Martinez-Cruz, A.; Franco-Bourland, R.E.; Cruz-García, E.; Corona-Juarez, A.; Diaz-Ruiz, A.; Grijalva, I.; Reyes-Alva, H.J.; Madrazo, I. Creation of an intramedullary cavity by hemorrhagic necrosis removal 24 h after spinal cord contusion in rats for eventual intralesional implantation of restorative materials. PLoS ONE 2017, 12, e0176105. [Google Scholar] [CrossRef] [PubMed]
- Kwiecien, J.M.; Jarosz, B.; Urdzikova, L.M.; Rola, R.; Dabrowski, W. Subdural infusion of dexamethasone inhibits leukomyelitis after acute spinal cord injury in a rat model. Folia Neuropathol. 2015, 1, 41–51. [Google Scholar] [CrossRef] [PubMed]
- Kwiecien, J.M.; Dabrowski, W.; Marzec-Kotarska, B.; Kwiecien-Delaney, C.J.; Yaron, J.R.; Zhang, L.; Schutz, L.; Lucas, A.R. Myxoma virus derived immune modulating proteins, M-T7 and Serp-1, reduce early inflammation after spinal cord injury in the rat model. Folia Neuropathol. 2019, 57, 41–50. [Google Scholar] [CrossRef]
- Sayer, F.T.; Kronvall, E.; Nilsson, O.G. Methylprednisolone treatment in acute spinal cord injury: The myth challenged through a structured analysis of published literature. Spine J. 2006, 6, 335–343. [Google Scholar] [CrossRef] [PubMed]
- Evaniew, N.; Belley-Côté, E.P.; Fallah, N.; Noonan, V.K.; Rivers, C.S.; Dvorak, M.F. Methylprednisolone for the treatment of patients with acute spinal cord injuries: A systematic review and meta-analysis. J. Neurotrauma 2016, 33, 468–481. [Google Scholar] [CrossRef] [PubMed]
- Hurlbert, R.J.; Hadley, M.N.; Walters, B.C.; Aarabi, B.; Dhall, S.S.; Gelb, D.E.; Rozzelle, C.J.; Ryken, T.C.; Theodore, N. Pharmacological therapy for acute spinal cord injury. Neurosurgery 2013, 72, 93–105. [Google Scholar] [CrossRef]
- Rust, R.; Kaiser, J. Insights into the dual role of inflammation after spinal cord injury. J. Neurosci. 2017, 37, 4658–4660. [Google Scholar] [CrossRef]
- Kwiecien, J.M.; Jarosz, B.; Oakden, W.; Klapec, M.; Stanisz, G.J.; Delaney, K.H.; Kotlinska-Hasiec, E.; Janik, R.; Rola, R.; Dabrowski, W. An in vivo model of anti-inflammatory activity of subdural dexamethasone following the spinal cord injury. Neurol. Neurochir. Pol. 2016, 50, 7–15. [Google Scholar] [CrossRef]
- Chedly, J.; Soares, S.; Montembault, A.; von Boxberg, Y.; Veron-Ravaille, M.; Mouffle, C.; Benassy, M.N.; Taxi, J.; David, L.; Nothias, F. Physical chitosan microhydrogels as scaffolds for spinal cord injury restoration and axon regeneration. Biomaterials 2017, 138, 91–107. [Google Scholar] [CrossRef]
- Yao, Z.A.; Chen, F.J.; Cui, H.L.; Lin, T.; Guo, N.; Wu, H.G. Efficacy of chitosan and sodium alginate scaffolds for repair of spinal cord injury in rats. Neural Regen. Res. 2018, 13, 502–509. [Google Scholar]
- Rao, J.-S.; Zhao, C.; Zhang, A.; Duan, H.; Hao, P.; Wei, R.-H.; Shang, J.; Zhao, W.; Liu, Z.; Yu, J.; et al. NT3-chitosan enables de novo regeneration and functional recovery in monkeys after spinal cord injury. Proc. Natl. Acad. Sci. USA 2018, 115, E5595–E5604. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Zheng, D.; Abbott, J.; Liu, L.; Bartee, M.Y.; Long, M.; Davids, J.; Williams, J.; Feldmann, H.; Strong, J.; et al. Myxomavirus-derived serpin prolongs survival and reduces inflammation and hemorrhage in an unrelated lethal mouse viral infection. Antimicrob. Agents Chemother. 2013, 57, 4114–4127. [Google Scholar] [CrossRef]
- Ambadapadi, S.; Munuswamy-Ramanujam, G.; Zheng, D.; Sullivan, C.; Dai, E.; Morshed, S.; McFadden, B.; Feldman, E.; Pinard, M.; McKenna, R.; et al. Reactive Center Loop (RCL) peptides derived from serpins display independent coagulation and immune modulating activities. J. Biol. Chem. 2016, 291, 2874–2887. [Google Scholar] [CrossRef] [PubMed]
- Viswanathan, K.; Richardson, J.; Togonu-Bickersteth, B.; Dai, E.; Liu, L.; Vatsya, P.; Sun, Y.; Yu, J.; Munuswamy-Ramanujam, G.; Baker, H.; et al. Myxoma viral serpin, Serp-1, inhibits human monocyte adhesion through regulation of actin-binding protein filamin B. J. Leukoc. Biol. 2009, 85, 418–426. [Google Scholar] [CrossRef] [PubMed]
- Brahn, E.; Lee, S.; Lucas, A.; McFadden, G.; Macaulay, C. Suppression of collagen-induced arthritis with a serine proteinase inhibitor (serpin) derived from myxoma virus. Clin. Immunol. 2014, 153, 254–263. [Google Scholar] [CrossRef] [PubMed]
- Tardif, J.C.; L’Allier, P.L.; Grégoire, J.; Ibrahim, R.; McFadden, G.; Kostuk, W.; Knudtson, M.; Labinaz, M.; Labinaz, R.W.; Pepine, C.J.; et al. A randomized controlled, phase 2 trial of the viral serpin serp-1 in patients with acute coronary syndromes undergoing percutaneous coronary intervention. Circ. Cardiovasc. Interv. 2010, 3, 543–548. [Google Scholar] [CrossRef]
- Li, Z.; Liu, F.; Zhang, L.; Cao, Y.; Shao, Y.; Wang, X.; Jiang, X.; Chen, Z. Neuroserpin restores autophagy and promotes functional recovery after acute spinal cord injury in rats. Mol. Med. Rep. 2018, 17, 2957–2963. [Google Scholar] [CrossRef]
- Bartee, M.Y.; Chen, H.; Dai, E.; Liu, L.Y.; Davids, J.A.; Lucas, A. Defining the anti-inflammatory activity of a potent myxomaviral chemokine modulating protein, M-T7, through site directed mutagenesis. Cytokine 2014, 65, 79–87. [Google Scholar] [CrossRef]
- Mothe, A.J.; Tam, R.Y.; Zahir, T.; Tator, C.H.; Shoichet, M.S. Repair of the injured spinal cord by transplantation of neural stem cells in a hyaluronan-based hydrogel. Biomaterials 2013, 34, 3775–3783. [Google Scholar] [CrossRef]
- White, F.A.; Jung, H.; Miller, R.J. Chemokines and the pathophysiology of neuropathic pain. Proc. Natl. Acad. Sci. USA. 2007, 104, 20151–20158. [Google Scholar] [CrossRef]
- Mahon, B.P.; Ambadapadi, S.; Yaron, J.R.; Lomelino, C.L.; Pinard, M.A.; Keinan, S.; Kurnikov, I.; Macaulay, C.; Zhang, L.; Reeves, W.; et al. Crystal Structure of Cleaved Serp-1, a Myxomavirus-Derived Immune Modulating Serpin: Structural Design of Serpin Reactive Center Loop Peptides with Improved Therapeutic Function. Biochemistry 2018, 57, 1096–1107. [Google Scholar] [CrossRef] [PubMed]
- Lucas, A.; Yaron, J.R.; Zhang, L.; Ambadapadi, S. Overview of serpins and their roles in biological systems. In Serpins; Lucas, A.R., Ed.; Humana Press; New York, NY, USA, 2018; Volume 1826, pp. 255–265. [Google Scholar]
- Chen, H.; Ambadapadi, S.; Dai, E.; Liu, L.; Yaron, J.R.; Zhang, L.; Lucas, A. Analysis of In Vivo Serpin Functions in Models of Inflammatory Vascular Disease. In Serpins; Lucas, A.R., Ed.; Humana Press: New York, NY, USA, 2018; Volume 1826, pp. 157–182. [Google Scholar]
- Strickland, D.K.; Muratoglu, S.C.; Antalis, T.M. Serpin-Enzyme Receptors: LDL Receptor-Related Protein 1, 1st ed.; Elsevier: Amsterdam, The Netherlands, 2011; Volume 499, ISBN 9780123864710. [Google Scholar]
- Kounnas, M.Z.; Church, F.C.; Argraves, W.S.; Strickland, D.K. Cellular internalization and degradation of antithrombin III-thrombin, heparin cofactor II-thrombin, and α1-antitrypsin-trypsin complexes is mediated by the low density lipoprotein receptor-related protein. J. Biol. Chem. 1996, 271, 6523–6529. [Google Scholar] [CrossRef] [PubMed]
- Gelderblom, M.; Neumann, M.; Ludewig, P.; Bernreuther, C.; Krasemann, S.; Arunachalam, P.; Gerloff, C.; Glatzel, M.; Magnus, T. Deficiency in Serine Protease Inhibitor Neuroserpin Exacerbates Ischemic Brain Injury by Increased Postischemic Inflammation. PLoS ONE 2013, 8, e63118. [Google Scholar] [CrossRef] [PubMed]
- Lemarchant, S.; Pruvost, M.; Hébert, M.; Gauberti, M.; Hommet, Y.; Briens, A.; Maubert, E.; Gueye, Y.; Féron, F.; Petite, D.; et al. tPA promotes ADAMTS-4-induced CSPG degradation, thereby enhancing neuroplasticity following spinal cord injury. Neurobiol. Dis. 2014, 66, 28–42. [Google Scholar] [CrossRef] [PubMed]
- Abe, Y.; Nakamura, H.; Yoshino, O.; Oya, T.; Kimura, T. Decreased neural damage after spinal cord injury in tPA-deficient mice. J. Neurotrauma 2003, 20, 43–57. [Google Scholar] [CrossRef]
- Li, X.; Yang, Z.; Zhang, A.; Wang, T.; Chen, W. Repair of thoracic spinal cord injury by chitosan tube implantation in adult rats. Biomaterials 2009, 30, 1121–1132. [Google Scholar] [CrossRef]
- Shrestha, B.; Coykendall, K.; Li, Y.; Moon, A.; Priyadarshani, P.; Yao, L. Repair of injured spinal cord using biomaterial scaffolds and stem cells. Stem Cell Res. Ther. 2014, 5, 1–11. [Google Scholar] [CrossRef]
- Yang, Z.; Zhang, A.; Duan, H.; Zhang, S.; Hao, P.; Ye, K.; Sun, Y.E.; Li, X. NT3-chitosan elicits robust endogenous neurogenesis to enable functional recovery after spinal cord injury. Proc. Natl. Acad. Sci. USA 2015, 112, 13354–13359. [Google Scholar] [CrossRef]
- Zhang, L.Q.; Yaron, J.R.; Tafoya, A.M.; Wallace, S.E.; Kilbourne, J.; Haydel, S.; Rege, K.; McFadden, G.; Lucas, A.R. A Virus-Derived Immune Modulating Serpin Accelerates Wound Closure with Improved Collagen Remodeling. J. Clin. Med. 2019, 8, 1626. [Google Scholar] [CrossRef]
- Shi, S.; Cheng, X.; Wang, J.; Zhang, W.; Peng, L.; Zhang, Y. RhBMP-2 microspheres-loaded chitosan/collagen scaffold enhanced osseointegration: An experiment in dog. J. Biomater. Appl. 2009, 23, 331–346. [Google Scholar] [PubMed]
- Filous, A.R.; Tran, A.; Howell, C.J.; Busch, S.A.; Evans, T.A.; Stallcup, W.B.; Kang, S.H.; Bergles, D.E.; Lee, S.; Levine, J.M.; et al. Entrapment via Synaptic-Like Connections between NG2 Proteoglycan+ Cells and Dystrophic Axons in the Lesion Plays a Role in Regeneration Failure after Spinal Cord Injury. J. Neurosci. 2014, 34, 16369–16384. [Google Scholar] [CrossRef] [PubMed]
- Kwiecien, J.M. Methods for Assessing Serpins as Neuroprotective Therapeutics. In Serpins; Lucas, A.R., Ed.; Springer: New York, NY, USA, 2018; Volume 1826, pp. 223–235. [Google Scholar]
- Basso, D.M.; Beattie, M.S.; Bresnahan, J.C. A Sensitive and Reliable Locomotor Rating Scale for Open Field Testing in Rats. J. Neurotrauma 1995, 12, 1–21. [Google Scholar] [CrossRef] [PubMed]
- Guízar-Sahagún, G.; Grijalva, I.; Salgado-Ceballos, H.; Espitia, A.; Orozco, S.; Ibarra, A.; Martínez, A.; Franco-Bourland, R.E.; Madrazo, I. Spontaneous and induced aberrant sprouting at the site of injury is irrelevant to motor function outcome in rats with spinal cord injury. Brain Res. 2004, 1013, 143–151. [Google Scholar] [CrossRef] [PubMed]
- Basso, D.M. Behavioral Testing after Spinal Cord Injury: Congruities, Complexities, and Controversies. J. Neurotrauma 2004, 21, 395–404. [Google Scholar] [CrossRef] [PubMed]
- Gomez, R.M.; Ghotme, K.; Nino, J.J.; Quiroz-Padilla, M.; Vargas, D.; Dominguez, A.R.; Barreto, G.E.; Sanchez, M.Y. Combined Strategy for a Reliable Evaluation of Spinal Cord Injury Using an in vivo Model. Cent. Nerv. Syst. Agents Med. Chem. 2018, 18, 49–57. [Google Scholar] [CrossRef] [PubMed]
- Kwiecien, J.M. Cellular mechanisms of white matter regeneration in an adult dysmyelinated rat model. Folia Neuropathol. 2013, 51, 189–202. [Google Scholar] [CrossRef]
- Kwiecien, J.M. Tissue reaction to acellular implants in the acute spinal cord injury in the dysmyelinated rat. In Advances in Medicine and Biology; Berhardt, L., Ed.; Nova Science Publishers, Inc.: Hauppauge, NY, USA, 2016; pp. 111–123. [Google Scholar]
- Yacoub, A.; Hajec, M.C.; Stanger, R.; Wan, W.; Young, H.; Mathern, B.E. Neuroprotective effects of perflurocarbon (oxycyte) after contusive spinal cord injury. J. Neurotrauma 2014, 31, 256–267. [Google Scholar] [CrossRef]
- Lü, H.-Z.; Wang, Y.-X.; Zhou, J.-S.; Wang, F.-C.; Hu, J.-G. Cyclosporin A increases recovery after spinal cord injury but does not improve myelination by oligodendrocyte progenitor cell transplantation. BMC Neurosci. 2010, 11, 127. [Google Scholar] [CrossRef]
- Sperling, L.E.; Pires Reis, K.; Nicola, F.; Euzebio Teixeira, C.; Gulielmin Didó, G.; Garrido dos Santos, M.; Konrath, E.; Netto, C.A.; Pranke, P. Galantamine improves functional recovery and reduces lesion size in a rat model of spinal cord injury. Brain Res. 2019, 1724, 146424. [Google Scholar] [CrossRef]
- Horng, S.; Therattil, A.; Moyon, S.; Gordon, A.; Kim, K.; Argaw, A.T.; Hara, Y.; Mariani, J.N.; Sawai, S.; Flodby, P.; et al. Astrocytic tight junctions control inflammatory CNS lesion pathogenesis. J. Clin. Investig. 2017, 127, 3136–3151. [Google Scholar] [CrossRef] [PubMed]
- Wanner, I.B.; Anderson, M.A.; Song, B.; Levine, J.; Fernandez, A.; Gray-Thompson, Z.; Ao, Y.; Sofroniew, M.V. Glial scar borders are formed by newly proliferated, elongated astrocytes that interact to corral inflammatory and fibrotic cells via STAT3-dependent mechanisms after spinal cord injury. J. Neurosci. 2013, 33, 12870–12886. [Google Scholar] [CrossRef] [PubMed]
- Okada, S.; Nakamura, M.; Katoh, H.; Miyao, T.; Shimazaki, T.; Ishii, K.; Yamane, J.; Yoshimura, A.; Iwamoto, Y.; Toyama, Y.; et al. Conditional ablation of Stat3 or Socs3 discloses a dual role for reactive astrocytes after spinal cord injury. Nat. Med. 2006, 12, 829–834. [Google Scholar] [CrossRef] [PubMed]
- Satish Kumar, T.; Vijaya Ramu, D.; Sampath Kumar, N.S. Preparation and characterization of biodegradable collagen—Chitosan scaffolds. Mater. Today Proc. 2019, 19, 2587–2590. [Google Scholar] [CrossRef]
- Fu, F.; Zhu, X.; Qin, Z.; Wang, J.J.; Xu, C.; Wang, L.N.; Tu, Y.; Zhang, S.; Li, R.X.; Li, X.H.; et al. Differential degradation rate and underlying mechanism of a collagen/chitosan complex in subcutis, spinal cord and brain tissues of rat. J. Mater. Sci. Mater. Med. 2018, 29, 35. [Google Scholar] [CrossRef]
- Citron, B.A.; Arnold, P.M.; Haynes, N.G.; Ameenuddin, S.; Farooque, M.; Santacruz, K.; Festoff, B.W. Neuroprotective effects of caspase-3 inhibition on functional recovery and tissue sparing after acute spinal cord injury. Spine 2008, 33, 2269–2277. [Google Scholar] [CrossRef]
- Viswanathan, K.; Liu, L.; Vaziri, S.; Dai, E.; Richardson, J.; Togonu-Bickersteth, B.; Vatsya, P.; Christov, A.; Lucas, A.R. Myxoma viral serpin, Serp-1, a unique interceptor of coagulation and innate immune pathways. Thromb. Haemost. 2006, 95, 499–510. [Google Scholar] [CrossRef]
- Wang, G.J.; Collinge, M.; Blasi, F.; Pardi, R.; Bender, J.R. Posttranscriptional regulation of urokinase plasminogen activator receptor messenger RNA levels by leukocyte integrin engagement. Proc. Natl. Acad. Sci. USA 1998, 95, 6296–6301. [Google Scholar] [CrossRef] [PubMed]
- Beschorner, R.; Schluesener, H.J.; Nguyen, T.D.; Magdolen, V.; Luther, T.; Pedal, I.; Mattern, R.; Meyermann, R.; Schwab, J.M. Lesion-associated accumulation of uPAR/CD87 expressing infiltrating granulocytes, activated microglial cells/macrophages and upregulation by endothelial cells following TBI and FCI in humans. Neuropathol. Appl. Neurobiol. 2000, 26, 522–527. [Google Scholar] [CrossRef]
- Ishii, H.; Jin, X.; Ueno, M.; Tanabe, S.; Kubo, T.; Serada, S.; Naka, T.; Yamashita, T. Adoptive transfer of Th1-conditioned lymphocytes promotes axonal remodeling and functional recovery after spinal cord injury. Cell Death Dis. 2012, 3, 1–10. [Google Scholar] [CrossRef][Green Version]
- Burda, J.E.; Sofroniew, M.V. Reactive gliosis and the multicellular response to CNS damage and disease. Neuron 2014, 81, 229–248. [Google Scholar] [CrossRef] [PubMed]
- Kjell, J.; Olson, L. Rat models of spinal cord injury: From pathology to potential therapies. DMM Dis. Model. Mech. 2016, 9, 1125–1137. [Google Scholar] [CrossRef] [PubMed]



| Treatment | Infusion | Days Follow-Up | Number of Rats |
|---|---|---|---|
| Chitosan hydrogel | No | 7 | 6 |
| Chitosan-Serp-1 low (10 µg) | No | 7 | 6 |
| Chitosan-Serp-1 high (100 µg) | No | 7 | 6 |
| Chitosan hydrogel | No | 28 | 7 a |
| Chitosan-Serp-1 low (10 µg) | No | 28 | 6 b |
| Chitosan-Serp-1 high (100 µg) | No | 28 | 6 |
| Chitosan hydrogel | Yes | 28 | 5 c |
| Chitosan-Serp-1 low (10 µg) | Yes | 28 | 5 d |
| Chitosan-Serp-1 high (100 µg) | Yes | 28 | 6 |
| Score | Description |
|---|---|
| 0 | Both hind legs have no motion. |
| 1 | One hind leg has flexing motion caudal to the level of the hip joint, with the plantar surface of the foot up, no weight support. |
| 2 | Both legs have flexing motion caudal to the hip, with the plantar surface of the foot up, no weight support or one leg has flexing motion beyond the hip, no body support, the other leg has no motion. |
| 3 | One leg has flexing motion beyond the hip, with the dorsal surface of the foot up, no weight support, the other leg has flexing motion caudal to the hip, with the plantar surface of the foot up; or one leg has flexing motion beyond the hip, with dorsal surface of the foot up, with body weight support but the other leg has no motion. |
| 4 | Both legs have flexing motion beyond the hip, with the dorsal surface of the foot up, but no body weight support; or one leg with the flexing motion beyond the hip with body support and the other leg with flexing motion caudal to the hip, with the plantar surface of the foot up, but no body weight support. |
| 5 | One leg has flexing motion beyond the hip with body weight support, the other leg flexing motion beyond the hip, with the dorsal surface of the foot up, but no body support. |
| 6 | Normal gait, no apparent weakness or proprioceptive deficits. |
| Score | Description |
|---|---|
| 0 | No toe retraction |
| 1 | Weak, no jerking |
| 2 | Weak with jerking |
| 3 | Strong/normal with jerking |
| Score | Description |
|---|---|
| 3 | Distended with hemorrhagic urine (treated with 50 µL Baytril s.i.d I/M until clear) |
| 2 | Distended with some blood in urine at the beginning of voiding |
| 1 | Distended, clear urine |
| 0 | Normal function, not distended |
| Score | Description |
|---|---|
| 0 | No containment |
| 1 | Weak containment, <1 side of the injury |
| 2 | Mild containment, >1 and <2 sides of the injury |
| 3 | Moderate containment, >2 sides of the injury |
| 4 | Complete containment, all sides of the injury |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kwiecien, J.M.; Zhang, L.; Yaron, J.R.; Schutz, L.N.; Kwiecien-Delaney, C.J.; Awo, E.A.; Burgin, M.; Dabrowski, W.; Lucas, A.R. Local Serpin Treatment via Chitosan-Collagen Hydrogel after Spinal Cord Injury Reduces Tissue Damage and Improves Neurologic Function. J. Clin. Med. 2020, 9, 1221. https://doi.org/10.3390/jcm9041221
Kwiecien JM, Zhang L, Yaron JR, Schutz LN, Kwiecien-Delaney CJ, Awo EA, Burgin M, Dabrowski W, Lucas AR. Local Serpin Treatment via Chitosan-Collagen Hydrogel after Spinal Cord Injury Reduces Tissue Damage and Improves Neurologic Function. Journal of Clinical Medicine. 2020; 9(4):1221. https://doi.org/10.3390/jcm9041221
Chicago/Turabian StyleKwiecien, Jacek M., Liqiang Zhang, Jordan R. Yaron, Lauren N. Schutz, Christian J. Kwiecien-Delaney, Enkidia A. Awo, Michelle Burgin, Wojciech Dabrowski, and Alexandra R. Lucas. 2020. "Local Serpin Treatment via Chitosan-Collagen Hydrogel after Spinal Cord Injury Reduces Tissue Damage and Improves Neurologic Function" Journal of Clinical Medicine 9, no. 4: 1221. https://doi.org/10.3390/jcm9041221
APA StyleKwiecien, J. M., Zhang, L., Yaron, J. R., Schutz, L. N., Kwiecien-Delaney, C. J., Awo, E. A., Burgin, M., Dabrowski, W., & Lucas, A. R. (2020). Local Serpin Treatment via Chitosan-Collagen Hydrogel after Spinal Cord Injury Reduces Tissue Damage and Improves Neurologic Function. Journal of Clinical Medicine, 9(4), 1221. https://doi.org/10.3390/jcm9041221








