Coronary Microvascular Dysfunction
Abstract
:1. Introduction
2. Coronary Microvascular Circulation
2.1. Distribution of Blood Flow
2.2. Heterogeneity of Coronary Microcirculation
2.3. Collaterals
3. Pathophysiology of CMD
3.1. Endothelial Dysfunction
3.2. Autonomic Nervous System
4. Clinical Presentation
4.1. Gender Differences in CMD
4.2. Clinical Classification
5. CMD without Atherosclerosis
6. CMD with Non-Obstructive Atherosclerosis
6.1. CMD in the Development of HFpEF
6.2. Microvascular Disease of the Brain
6.3. CMD in Diabetes
6.4. CMD in Hypertensive Heart Disease
6.5. CMD in Hypertrophic Cardiomyopathy (HCM)
6.6. CMD in Aortic Valve Stenosis
6.7. CMD in Chronic Kidney Disease (CKD)
6.8. CMD in Chronic Inflammatory and Autoimmune Diseases
7. CMD with Obstructive Atherosclerosis
7.1. Epicardial Effects on Microcirculation
7.2. Coronary No-Reflow
7.3. Microvascular Effects on Epicardial Coronary Arteries
8. Diagnosis of CMD
8.1. Invasive Diagnosis
8.2. Non-Invasive Diagnosis
9. Treatment of CMD
10. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Timmis, A.; Townsend, N.; Gale, C.P.; Torbica, A.; Lettino, M.; Petersen, S.E.; Mossialos, E.A.; Maggioni, A.P.; Kazakiewicz, D.; May, H.T.; et al. European Society of Cardiology: Cardiovascular Disease Statistics 2019. Eur. Heart J. 2020, 41, 12–85. [Google Scholar] [CrossRef] [PubMed]
- Thygesen, K.; Alpert, J.S.; Jaffe, A.S.; Chaitman, B.R.; Bax, J.J.; Morrow, D.A.; White, H.D.; Group, E.S.D. Fourth universal definition of myocardial infarction (2018). Eur. Heart J. 2019, 40, 237–269. [Google Scholar] [CrossRef] [Green Version]
- Knuuti, J.; Wijns, W.; Saraste, A.; Capodanno, D.; Barbato, E.; Funck-Brentano, C.; Prescott, E.; Storey, R.F.; Deaton, C.; Cuisset, T.; et al. 2019 ESC Guidelines for the diagnosis and management of chronic coronary syndromes: The Task Force for the diagnosis and management of chronic coronary syndromes of the European Society of Cardiology (ESC). Eur. Heart J. 2020, 41, 407–477. [Google Scholar] [CrossRef] [PubMed]
- Agewall, S.; Beltrame, J.F.; Reynolds, H.R.; Niessner, A.; Rosano, G.; Caforio, A.L.P.; De Caterina, R.; Zimarino, M.; Roffi, M.; Kjeldsen, K.; et al. ESC working group position paper on myocardial infarction with non-obstructive coronary arteries. Eur. Heart J. 2017, 38, 143–153. [Google Scholar] [CrossRef]
- Shaw, L.J.; Shaw, R.E.; Merz, C.N.B.; Brindis, R.G.; Klein, L.W.; Nallamothu, B.; Douglas, P.S.; Krone, R.J.; McKay, C.R.; Block, P.C.; et al. Impact of Ethnicity and Gender Differences on Angiographic Coronary Artery Disease Prevalence and In-Hospital Mortality in the American College of Cardiology-National Cardiovascular Data Registry. Circulation 2008, 117, 1787–1801. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Patel, M.R.; Peterson, E.D.; Dai, D.; Brennan, J.M.; Redberg, R.F.; Anderson, H.V.; Brindis, R.G.; Douglas, P.S. Low diagnostic yield of elective coronary angiography. N. Engl. J. Med. 2010, 362, 886–895. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Johnston, N.; Schenck-Gustafsson, K.; Lagerqvist, B. Are we using cardiovascular medications and coronary angiography appropriately in men and women with chest pain? Eur. Heart J. 2011, 32, 1331–1336. [Google Scholar] [CrossRef] [Green Version]
- Lindahl, B.; Baron, T.; Erlinge, D.; Hadziosmanovic, N.; Nordenskjöld, A.; Gard, A.; Jernberg, T. Medical Therapy for Secondary Prevention and Long-Term Outcome in Patients With Myocardial Infarction With Nonobstructive Coronary Artery Disease. Circulation 2017, 135, 1481–1489. [Google Scholar] [CrossRef]
- Schindler, T.H.; Dilsizian, V. Coronary Microvascular Dysfunction Clinical Considerations and Noninvasive Diagnosis. JACC Cardiovasc. Imaging 2019, 13, 140–155. [Google Scholar] [CrossRef]
- Jespersen, L.; Hvelplund, A.; Abildstrøm, S.Z.; Pedersen, F.; Galatius, S.; Madsen, J.K.; Jørgensen, E.; Kelbæk, H.; Prescott, E. Stable angina pectoris with no obstructive coronary artery disease is associated with increased risks of major adverse cardiovascular events. Eur. Heart J. 2012, 33, 734–744. [Google Scholar] [CrossRef]
- Patel, M.R.; Dai, D.; Hernandez, A.F.; Douglas, P.S.; Messenger, J.; Garratt, K.N.; Maddox, T.M.; Peterson, E.D.; Roe, M.T. Prevalence and predictors of nonobstructive coronary artery disease identified with coronary angiography in contemporary clinical practice. Am. Heart J. 2014, 167, 846–852. [Google Scholar] [CrossRef] [PubMed]
- Gehrie, E.R.; Reynolds, H.R.; Chen, A.Y.; Neelon, B.H.; Roe, M.T.; Gibler, W.B.; Ohman, E.M.; Newby, L.K.; Peterson, E.D.; Hochman, J.S. Characterization and outcomes of women and men with non–ST-segment elevation myocardial infarction and nonobstructive coronary artery disease: Results from the Can Rapid Risk Stratification of Unstable Angina Patients Suppress Adverse Outcomes with Early Implementation of the ACC/AHA Guidelines (CRUSADE) Quality Improvement Initiative. Am. Heart J. 2009, 158, 688–694. [Google Scholar] [CrossRef] [PubMed]
- Bainey, K.R.; Welsh, R.C.; Alemayehu, W.; Westerhout, C.M.; Traboulsi, D.; Anderson, T.; Brass, N.; Armstrong, P.W.; Kaul, P. Population-level incidence and outcomes of myocardial infarction with non-obstructive coronary arteries (MINOCA): Insights from the Alberta contemporary acute coronary syndrome patients invasive treatment strategies (COAPT) study. Int. J. Cardiol. 2018, 264, 12–17. [Google Scholar] [CrossRef] [PubMed]
- Maddox, T.M.; Stanislawski, M.A.; Grunwald, G.K.; Bradley, S.M.; Ho, P.M.; Tsai, T.T.; Patel, M.R.; Sandhu, A.; Valle, J.; Magid, D.J.; et al. Nonobstructive Coronary Artery Disease and Risk of Myocardial Infarction. JAMA 2014, 312, 1754–1763. [Google Scholar] [CrossRef] [PubMed]
- Ong, P.; Athanasiadis, A.; Borgulya, G.; Mahrholdt, H.; Kaski, J.C.; Sechtem, U. High Prevalence of a Pathological Response to Acetylcholine Testing in Patients With Stable Angina Pectoris and Unobstructed Coronary Arteries: The ACOVA Study (Abnormal COronary VAsomotion in patients with stable angina and unobstructed coronary arteries). J. Am. Coll. Cardiol. 2012, 59, 655–662. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sipahi, I.; Tuzcu, E.M.; Schoenhagen, P.; Nicholls, S.J.; Chen, M.S.; Crowe, T.; Loyd, A.B.; Kapadia, S.; Nissen, S.E. Paradoxical increase in lumen size during progression of coronary atherosclerosis: Observations from the REVERSAL trial. Atherosclerosis 2006, 189, 229–235. [Google Scholar] [CrossRef] [PubMed]
- Sara, J.D.; Widmer, R.J.; Matsuzawa, Y.; Lennon, R.J.; Lerman, L.O.; Lerman, A. Prevalence of Coronary Microvascular Dysfunction Among Patients With Chest Pain and Nonobstructive Coronary Artery Disease. JACC Cardiovasc. Interv. 2015, 8, 1445–1453. [Google Scholar] [CrossRef]
- Ong, P.; Camici, P.G.; Beltrame, J.F.; Crea, F.; Shimokawa, H.; Sechtem, U.; Kaski, J.C.; Bairey Merz, C.N. International standardization of diagnostic criteria for microvascular angina. Int. J. Cardiol. 2018, 250, 16–20. [Google Scholar] [CrossRef]
- Pepine, C.J.; Ferdinand, K.C.; Shaw, L.J.; Light-McGroary, K.A.; Shah, R.U.; Gulati, M.; Duvernoy, C.; Walsh, M.N.; Bairey Merz, C.N.; Committee, A.C.i.W. Emergence of Nonobstructive Coronary Artery Disease: A Woman’s Problem and Need for Change in Definition on Angiography. J. Am. Coll. Cardiol. 2015, 66, 1918–1933. [Google Scholar] [CrossRef]
- Padro, T.; Manfrini, O.; Bugiardini, R.; Canty, J.; Cenko, E.; De Luca, G.; Duncker, D.J.; Eringa, E.C.; Koller, A.; Tousoulis, D.; et al. ESC Working Group on Coronary Pathophysiology and Microcirculation position paper on ‘coronary microvascular dysfunction in cardiovascular disease’. Cardiovasc. Res. 2020, 116, 741–755. [Google Scholar] [CrossRef] [Green Version]
- Bugiardini, R.; Manfrini, O.; De Ferrari, G.M. Unanswered Questions for Management of Acute Coronary Syndrome: Risk Stratification of Patients With Minimal Disease or Normal Findings on Coronary Angiography. Arch. Intern. Med. 2006, 166, 1391–1395. [Google Scholar] [CrossRef]
- Planer, D.; Mehran, R.; Ohman, E.M.; White, H.D.; Newman, J.D.; Xu, K.; Stone, G.W. Prognosis of Patients With Non-ST-Segment-Elevation Myocardial Infarction and Nonobstructive Coronary Artery Disease. Circ. Cardiovasc. Interv. 2014, 7, 285–293. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Camici, P.G.; Crea, F. Coronary Microvascular Dysfunction. N. Engl. J. Med. 2007, 356, 830–840. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- John, W.P.; Johnson, B.D.; Kevin, E.K.; Anderson, R.D.; Eileen, M.H.; Barry, S.; Puja, K.M.; Sheryl, F.K.; Merz, C.N.B.; Carl, J.P. TIMI frame count and adverse events in women with no obstructive coronary disease: A pilot study from the NHLBI-sponsored Women’s Ischemia Syndrome Evaluation (WISE). PLoS ONE 2014, 9, e96630. [Google Scholar] [CrossRef] [Green Version]
- Gulati, M.; Cooper-DeHoff, R.M.; McClure, C.; Johnson, B.D.; Shaw, L.J.; Handberg, E.M.; Zineh, I.; Kelsey, S.F.; Arnsdorf, M.F.; Black, H.R.; et al. Adverse Cardiovascular Outcomes in Women With Nonobstructive Coronary Artery Disease: A Report From the Women’s Ischemia Syndrome Evaluation Study and the St James Women Take Heart Project. Arch. Intern. Med. 2009, 169, 843–850. [Google Scholar] [CrossRef] [Green Version]
- Brainin, P.; Frestad, D.; Prescott, E. The prognostic value of coronary endothelial and microvascular dysfunction in subjects with normal or non-obstructive coronary artery disease: A systematic review and meta-analysis. Int. J. Cardiol. 2018, 254, 1–9. [Google Scholar] [CrossRef]
- Herscovici, R.; Sedlak, T.; Wei, J.; Pepine, C.J.; Handberg, E.; Merz, C.N.B. Ischemia and No Obstructive Coronary Artery Disease (INOCA): What Is the Risk? J. Am. Heart Assoc. 2018, 7, e008868. [Google Scholar] [CrossRef]
- Lind, L.; Berglund, L.; Larsson, A.; Sundström, J. Endothelial Function in Resistance and Conduit Arteries and 5-Year Risk of Cardiovascular Disease. Circulation 2011, 123, 1545–1551. [Google Scholar] [CrossRef] [Green Version]
- Lee, F.B.-K.; Lim, S.H.-S.; Fearon, P.W.; Yong, C.A.; Yamada, A.R.; Tanaka, A.S.; Lee, A.D.; Yeung, A.A.; Tremmel, A.J. Invasive Evaluation of Patients With Angina in the Absence of Obstructive Coronary Artery Disease. Circulation 2015, 131, 1054–1060. [Google Scholar] [CrossRef] [Green Version]
- Khuddus, M.A.; Pepine, C.J.; Handberg, E.M.; Bairey Merz, C.N.; Sopko, G.; Bavry, A.A.; Denardo, S.J.; McGorray, S.P.; Smith, K.M.; Sharaf, B.L.; et al. An Intravascular Ultrasound Analysis in Women Experiencing Chest Pain in the Absence of Obstructive Coronary Artery Disease: A Substudy from the National Heart, Lung and Blood Institute–Sponsored Women’s Ischemia Syndrome Evaluation (WISE). J. Interv. Cardiol. 2010, 23, 511–519. [Google Scholar] [CrossRef]
- Hoef, T.P.V.d.; Lavieren, M.A.V.; Damman, P.; Delewi, R.; Piek, M.A.; Chamuleau, S.A.J.; Voskuil, M.; Henriques, J.P.S.; Koch, K.T.; Winter, R.J.d.; et al. Physiological Basis and Long-Term Clinical Outcome of Discordance Between Fractional Flow Reserve and Coronary Flow Velocity Reserve in Coronary Stenoses of Intermediate Severity. Circ. Cardiovasc. Interv. 2014, 7, 301–311. [Google Scholar] [CrossRef] [Green Version]
- Rahman, H.; Corcoran, D.; Aetesam-ur-Rahman, M.; Hoole, S.P.; Berry, C.; Perera, D. Diagnosis of patients with angina and non-obstructive coronary disease in the catheter laboratory. Heart 2019, 105, 1536–1542. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gould, K.L.; Johnson, N.P.; Bateman, T.M.; Beanlands, R.S.; Bengel, F.M.; Bober, R.; Camici, P.G.; Cerqueira, M.D.; Chow, B.J.W.; Di Carli, M.F.; et al. Anatomic Versus Physiologic Assessment of Coronary Artery Disease: Role of Coronary Flow Reserve, Fractional Flow Reserve, and Positron Emission Tomography Imaging in Revascularization Decision-Making. J. Am. Coll. Cardiol. 2013, 62, 1639–1653. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Herrmann, J.; Kaski, J.C.; Lerman, A. Coronary microvascular dysfunction in the clinical setting: From mystery to reality. Eur. Heart J. 2012, 33, 2771–2783. [Google Scholar] [CrossRef] [Green Version]
- Patel, B.; Fisher, M. Therapeutic advances in myocardial microvascular resistance: Unravelling the enigma. Pharmacol. Ther. 2010, 127, 131–147. [Google Scholar] [CrossRef]
- Duncker, D.J.; Koller, A.; Merkus, D.; Canty, J.M. Regulation of Coronary Blood Flow in Health and Ischemic Heart Disease. Prog. Cardiovasc. Dis. 2015, 57, 409–422. [Google Scholar] [CrossRef]
- Camici, P.G.; d’Amati, G.; Rimoldi, O. Coronary microvascular dysfunction: Mechanisms and functional assessment. Nat. Rev. Cardiol. 2015, 12, 48–62. [Google Scholar] [CrossRef]
- Deussen, A.; Ohanyan, V.; Jannasch, A.; Yin, L.; Chilian, W. Mechanisms of metabolic coronary flow regulation. J. Mol. Cell. Cardiol. 2012, 52, 794–801. [Google Scholar] [CrossRef]
- Camici, P.G.; Olivotto, I.; Rimoldi, O.E. The coronary circulation and blood flow in left ventricular hypertrophy. J. Mol. Cell. Cardiol. 2012, 52, 857–864. [Google Scholar] [CrossRef] [Green Version]
- Østergaard, L.; Kristiansen, S.B.; Angleys, H.; Frøkiær, J.; Michael Hasenkam, J.; Jespersen, S.N.; Bøtker, H.E. The role of capillary transit time heterogeneity in myocardial oxygenation and ischemic heart disease. Basic Res. Cardiol. 2014, 109, 409. [Google Scholar] [CrossRef] [Green Version]
- Deussen, A. Blood flow heterogeneity in the heart. Basic Res. Cardiol. 1998, 93, 430–438. [Google Scholar] [CrossRef] [PubMed]
- Pries, A.R.; Secomb, T.W. Origins of heterogeneity in tissue perfusion and metabolism. Cardiovasc. Res. 2009, 81, 328–335. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schulz, R.; Heusch, G. The relationship between regional blood flow and contractile function in normal, ischemic, and reperfused myocardium. Basic Res. Cardiol. 1998, 93, 455–462. [Google Scholar] [CrossRef] [PubMed]
- Pries, A.R.; Badimon, L.; Bugiardini, R.; Camici, P.G.; Dorobantu, M.; Duncker, D.J.; Escaned, J.; Koller, A.; Piek, J.J.; de Wit, C. Coronary vascular regulation, remodelling, and collateralization: Mechanisms and clinical implications on behalf of the working group on coronary pathophysiology and microcirculation. Eur. Heart J. 2015, 36, 3134–3146. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wieneke, H.; Schmermund, A.; Ge, J.; Altmann, C.; Haude, M.; von Birgelen, C.; Baumgart, D.; Dirsch, O.; Erbel, R. Increased heterogeneity of coronary perfusion in patients with early coronary atherosclerosis. Am. Heart J. 2001, 142, 691–697. [Google Scholar] [CrossRef]
- Seiler, C. The human coronary collateral circulation. Eur. J. Clin. Investig. 2010, 40, 465–476. [Google Scholar] [CrossRef] [Green Version]
- Buschmann, I.; Schaper, W. The pathophysiology of the collateral circulation (arteriogenesis). J. Pathol. 2000, 190, 338–342. [Google Scholar] [CrossRef]
- Zimarino, M.; D’Andreamatteo, M.; Waksman, R.; Epstein, S.E.; De Caterina, R. The dynamics of the coronary collateral circulation. Nat. Rev. Cardiol. 2014, 11, 191–197. [Google Scholar] [CrossRef]
- van Royen, N.; Piek, J.J.; Buschmann, I.; Hoefer, I.; Voskuil, M.; Schaper, W. Stimulation of arteriogenesis; a new concept for the treatment of arterial occlusive disease. Cardiovasc. Res. 2001, 49, 543–553. [Google Scholar] [CrossRef]
- Wustmann, K.; Zbinden, S.; Windecker, S.; Meier, B.; Seiler, C. Is There Functional Collateral Flow During Vascular Occlusion in Angiographically Normal Coronary Arteries? Circulation 2003, 107, 2213–2220. [Google Scholar] [CrossRef] [Green Version]
- Werner, G.S.; Ferrari, M.; Betge, S.; Gastmann, O.; Richartz, B.M.; Figulla, H.R. Collateral Function in Chronic Total Coronary Occlusions Is Related to Regional Myocardial Function and Duration of Occlusion. Circulation 2001, 104, 2784–2790. [Google Scholar] [CrossRef] [Green Version]
- Carmeliet, P. Mechanisms of angiogenesis and arteriogenesis. Nat. Med. 2000, 6, 389–395. [Google Scholar] [CrossRef] [PubMed]
- Khand, A.; Fisher, M.; Jones, J.; Patel, B.; Perry, R.; Mitsudo, K. The collateral circulation of the heart in coronary total arterial occlusions in man: Systematic review of assessment and pathophysiology. Am. Heart J. 2013, 166, 941–952. [Google Scholar] [CrossRef] [PubMed]
- Meier, P.; Hemingway, H.; Lansky, A.J.; Knapp, G.; Pitt, B.; Seiler, C. The impact of the coronary collateral circulation on mortality: A meta-analysis. Eur. Heart J. 2012, 33, 614–621. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Konst, R.E.; Guzik, T.J.; Kaski, J.-C.; Maas, A.H.E.M.; Elias-Smale, S.E. The pathogenic role of coronary microvascular dysfunction in the setting of other cardiac or systemic conditions. Cardiovasc. Res. 2020, 116, 817–828. [Google Scholar] [CrossRef] [Green Version]
- Lanza, G.A.; Crea, F. Primary Coronary Microvascular Dysfunction. Circulation 2010, 121, 2317–2325. [Google Scholar] [CrossRef] [Green Version]
- Aird, W.C. Phenotypic Heterogeneity of the Endothelium. II. Representative Vascular Beds. Circ. Res. 2007, 100, 174–190. [Google Scholar] [CrossRef] [Green Version]
- Brutsaert, D.L. Cardiac Endothelial-Myocardial Signaling: Its Role in Cardiac Growth, Contractile Performance, and Rhythmicity. Physiol. Rev. 2003, 83, 59–115. [Google Scholar] [CrossRef]
- Gutiérrez, E.; Flammer, A.J.; Lerman, L.O.; Elízaga, J.; Lerman, A.; Fernández-Avilés, F. Endothelial dysfunction over the course of coronary artery disease. Eur. Heart J. 2013, 34, 3175–3181. [Google Scholar] [CrossRef] [Green Version]
- Kinlay, S.; Libby, P.; Ganz, P. Endothelial function and coronary artery disease. Curr. Opin. Lipidol. 2001, 12, 383–389. [Google Scholar] [CrossRef]
- Huige, L.; Ulrich, F. Prevention of Atherosclerosis by Interference with the Vascular Nitric Oxide System. Curr. Pharm. Des. 2009, 15, 3133–3145. [Google Scholar] [CrossRef] [Green Version]
- Rubinshtein, R.; Yang, E.H.; Rihal, C.S.; Prasad, A.; Lennon, R.J.; Best, P.J.; Lerman, L.O.; Lerman, A. Coronary microcirculatory vasodilator function in relation to risk factors among patients without obstructive coronary disease and low to intermediate Framingham score. Eur. Heart J. 2010, 31, 936–942. [Google Scholar] [CrossRef] [PubMed]
- Granger, D.N.; Rodrigues, S.F.; Yildirim, A.; Senchenkova, E.Y. Microvascular Responses to Cardiovascular Risk Factors. Microcirculation 2010, 17, 192–205. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wessel, T.R.; Arant, C.B.; McGorray, S.P.; Sharaf, B.L.; Reis, S.E.; Kerensky, R.A.; von Mering, G.O.; Smith, K.M.; Pauly, D.F.; Handberg, E.M.; et al. Coronary microvascular reactivity is only partially predicted by atherosclerosis risk factors or coronary artery disease in women evaluated for suspected ischemia: Results from the NHLBI Women’s Ischemia Syndrome Evaluation (WISE). Clin. Cardiol. 2007, 30, 69–74. [Google Scholar] [CrossRef]
- Bonetti, P.O.; Lerman, L.O.; Lerman, A. Endothelial dysfunction: A marker of atherosclerotic risk. Arterioscler. Thromb. Vasc. Biol. 2003, 23, 168–175. [Google Scholar] [CrossRef]
- Aird, C.W. Endothelium as an organ system. Crit. Care Med. 2004, 32, S271–S279. [Google Scholar] [CrossRef]
- Tomai, F.; Ribichini, F.; Ghini, A.S.; Ferrero, V.; Andò, G.; Vassanelli, C.; Romeo, F.; Crea, F.; Chiariello, L. Elevated C-reactive protein levels and coronary microvascular dysfunction in patients with coronary artery disease. Eur. Heart J. 2005, 26, 2099–2105. [Google Scholar] [CrossRef] [Green Version]
- Recio-Mayoral, A.; Rimoldi, O.E.; Camici, P.G.; Kaski, J.C. Inflammation and Microvascular Dysfunction in Cardiac Syndrome X Patients Without Conventional Risk Factors for Coronary Artery Disease. JACC Cardiovasc. Imaging 2013, 6, 660–667. [Google Scholar] [CrossRef] [Green Version]
- Meng-Yu, W.; Chia-Jung, L.; Ming-Feng, H.; Pei-Yi, C. New Insights into the Role of Inflammation in the Pathogenesis of Atherosclerosis. Int J. Mol. Sci. 2017, 18, 2034. [Google Scholar] [CrossRef]
- Adukauskienė, D.; Čiginskienė, A.; Adukauskaitė, A.; Pentiokinienė, D.; Šlapikas, R.; Čeponienė, I. Clinical relevance of high sensitivity C-reactive protein in cardiology. Medicina 2016, 52, 1–10. [Google Scholar] [CrossRef]
- Singh, T.P.; Morris, D.R.; Smith, S.; Moxon, J.V.; Golledge, J. Systematic Review and Meta-Analysis of the Association Between C-Reactive Protein and Major Cardiovascular Events in Patients with Peripheral Artery Disease. Eur. J. Vasc. Endovasc. Surg. 2017, 54, 220–233. [Google Scholar] [CrossRef]
- Twine, C.P. The Relationship Between CRP and MACE: Controversial and Confounded. Eur. J. Vasc. Endovasc. Surg. 2017, 54, 234. [Google Scholar] [CrossRef] [PubMed]
- Kwak, B.R.; Bäck, M.; Bochaton-Piallat, M.-L.; Caligiuri, G.; Daemen, M.J.A.P.; Davies, P.F.; Hoefer, I.E.; Holvoet, P.; Jo, H.; Krams, R.; et al. Biomechanical factors in atherosclerosis: Mechanisms and clinical implications†. Eur. Heart J. 2014, 35, 3013–3020. [Google Scholar] [CrossRef] [PubMed]
- Helderman, F.; Segers, D.; de Crom, R.; Hierck, B.P.; Poelmann, R.E.; Evans, P.C.; Krams, R. Effect of shear stress on vascular inflammation and plaque development. Curr. Opin. Lipidol. 2007, 18, 527–533. [Google Scholar] [CrossRef] [PubMed]
- Papafaklis, M.I.; Takahashi, S.; Antoniadis, A.P.; Coskun, A.U.; Tsuda, M.; Mizuno, S.; Andreou, I.; Nakamura, S.; Makita, Y.; Hirohata, A.; et al. Effect of the local hemodynamic environment on the de novo development and progression of eccentric coronary atherosclerosis in humans: Insights from PREDICTION. Atherosclerosis 2015, 240, 205–211. [Google Scholar] [CrossRef]
- Tesauro, M.; Mauriello, A.; Rovella, V.; Annicchiarico-Petruzzelli, M.; Cardillo, C.; Melino, G.; Di Daniele, N. Arterial ageing: From endothelial dysfunction to vascular calcification. J. Intern. Med. 2017, 281, 471–482. [Google Scholar] [CrossRef] [Green Version]
- Stone, P.H.; Maehara, A.; Coskun, A.U.; Maynard, C.C.; Zaromytidou, M.; Siasos, G.; Andreou, I.; Fotiadis, D.; Stefanou, K.; Papafaklis, M.; et al. Role of Low Endothelial Shear Stress and Plaque Characteristics in the Prediction of Nonculprit Major Adverse Cardiac Events: The PROSPECT Study. JACC Cardiovasc. Imaging 2018, 11, 462–471. [Google Scholar] [CrossRef]
- Taqueti, V.R.; Di Carli, M.F. Coronary Microvascular Disease Pathogenic Mechanisms and Therapeutic Options: JACC State-of-the-Art Review. J. Am. Coll. Cardiol. 2018, 72, 2625–2641. [Google Scholar] [CrossRef]
- Goligorsky, M.S. Microvascular rarefaction. Organogenesis 2010, 6, 1–10. [Google Scholar] [CrossRef] [Green Version]
- O’Riordan, E.; Mendelev, N.; Patschan, S.; Patschan, D.; Eskander, J.; Cohen-Gould, L.; Chander, P.; Goligorsky, M.S. Chronic NOS inhibition actuates endothelial-mesenchymal transformation. Am. J. Physiol. Heart Circ. Physiol. 2007, 292, H285–H294. [Google Scholar] [CrossRef] [Green Version]
- Vita, J.A. Endothelial Function. Circulation 2011, 124, e906–e912. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Huang, A.L.; Vita, J.A. Effects of Systemic Inflammation on Endothelium-Dependent Vasodilation. Trends Cardiovasc. Med. 2006, 16, 15–20. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zannad, F.; Radauceanu, A. Effect of MR Blockade on Collagen Formation and Cardiovascular Disease with a Specific Emphasis on Heart Failure. Heart Fail. Rev. 2005, 10, 71–78. [Google Scholar] [CrossRef] [PubMed]
- González, A.; Ravassa, S.; Beaumont, J.; López, B.; Díez, J. New Targets to Treat the Structural Remodeling of the Myocardium. J. Am. Coll. Cardiol. 2011, 58, 1833–1843. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Westermann, D.; Lindner, D.; Kasner, M.; Zietsch, C.; Savvatis, K.; Escher, F.; Schlippenbach, J.v.; Skurk, C.; Steendijk, P.; Riad, A.; et al. Cardiac Inflammation Contributes to Changes in the Extracellular Matrix in Patients With Heart Failure and Normal Ejection Fraction. Circ. Heart Fail. 2011, 4, 44–52. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lindemann, H.; Petrovic, I.; Hill, S.; Athanasiadis, A.; Mahrholdt, H.; Schaufele, T.; Klingel, K.; Sechtem, U.; Ong, P. Biopsy-confirmed endothelial cell activation in patients with coronary microvascular dysfunction. Coron. Artery Dis. 2018, 29, 216–222. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, H.; Takeyama, Y.; Koba, S.; Suwa, Y.; Katagiri, T. Small vessel pathology and coronary hemodynamics in patients with microvascular angina. Int. J. Cardiol. 1994, 43, 139–150. [Google Scholar] [CrossRef]
- Mohammed, S.F.; Hussain, S.; Mirzoyev, S.A.; Edwards, W.D.; Maleszewski, J.J.; Redfield, M.M. Coronary Microvascular Rarefaction and Myocardial Fibrosis in Heart Failure With Preserved Ejection Fraction. Circulation 2015, 131, 550–559. [Google Scholar] [CrossRef] [Green Version]
- Tsagalou, E.P.; Anastasiou-Nana, M.; Agapitos, E.; Gika, A.; Drakos, S.G.; Terrovitis, J.V.; Ntalianis, A.; Nanas, J.N. Depressed Coronary Flow Reserve Is Associated With Decreased Myocardial Capillary Density in Patients With Heart Failure Due to Idiopathic Dilated Cardiomyopathy. J. Am. Coll. Cardiol. 2008, 52, 1391–1398. [Google Scholar] [CrossRef] [Green Version]
- Escaned, J.; Flores, A.; García-Pavía, P.; Segovia, J.; Jimenez, J.; Aragoncillo, P.; Salas, C.; Alfonso, F.; Hernández, R.; Angiolillo, D.J.; et al. Assessment of Microcirculatory Remodeling With Intracoronary Flow Velocity and Pressure Measurements. Circulation 2009, 120, 1561–1568. [Google Scholar] [CrossRef] [Green Version]
- Hong, H.; Aksenov, S.; Guan, X.; Fallon, J.T.; Waters, D.; Chen, C. Remodeling of Small Intramyocardial Coronary Arteries Distal to a Severe Epicardial Coronary Artery Stenosis. Arterioscler. Thromb. Vasc. Biol. 2002, 22, 2059–2065. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Heusch, G.; Skyschally, A.; Kleinbongard, P. Coronary microembolization and microvascular dysfunction. Int. J. Cardiol. 2018, 258, 17–23. [Google Scholar] [CrossRef] [PubMed]
- Kleinbongard, P.; Konorza, T.; Böse, D.; Baars, T.; Haude, M.; Erbel, R.; Heusch, G. Lessons from human coronary aspirate. J. Mol. Cell. Cardiol. 2012, 52, 890–896. [Google Scholar] [CrossRef] [PubMed]
- Duncker, D.J.; Bache, R.J.; Merkus, D. Regulation of coronary resistance vessel tone in response to exercise. J. Mol. Cell. Cardiol. 2012, 52, 802–813. [Google Scholar] [CrossRef] [PubMed]
- Heusch, G.; Baumgart, D.; Camici, P.; Chilian, W.; Gregorini, L.; Hess, O.; Indolfi, C.; Rimoldi, O. α-Adrenergic Coronary Vasoconstriction and Myocardial Ischemia in Humans. Circulation 2000, 101, 689–694. [Google Scholar] [CrossRef] [Green Version]
- Heusch, G. The paradox of α-adrenergic coronary vasoconstriction revisited. J. Mol. Cell. Cardiol. 2011, 51, 16–23. [Google Scholar] [CrossRef]
- Levy, B.I.; Heusch, G.; Camici, P.G. The many faces of myocardial ischaemia and angina. Cardiovasc. Res. 2019, 115, 1460–1470. [Google Scholar] [CrossRef]
- Merz, C.N.B.; Pepine, C.J.; Walsh, M.N.; Fleg, J.L.; Camici, P.G.; Chilian, W.M.; Clayton, J.A.; Cooper, L.S.; Crea, F.; Carli, M.D.; et al. Ischemia and No Obstructive Coronary Artery Disease (INOCA). Circulation 2017, 135, 1075–1092. [Google Scholar] [CrossRef]
- Shaw, J.; Anderson, T. Coronary endothelial dysfunction in non-obstructive coronary artery disease: Risk, pathogenesis, diagnosis and therapy. Vasc. Med. 2016, 21, 146–155. [Google Scholar] [CrossRef] [Green Version]
- Kunadian, V.; Chieffo, A.; Camici, P.G.; Berry, C.; Escaned, J.; Maas, A.H.E.M.; Prescott, E.; Karam, N.; Appelman, Y.; Fraccaro, C.; et al. An EAPCI Expert Consensus Document on Ischaemia with Non-Obstructive Coronary Arteries in Collaboration with European Society of Cardiology Working Group on Coronary Pathophysiology & Microcirculation Endorsed by Coronary Vasomotor Disorders International Study Group. Eur. Heart J. 2020. [Google Scholar] [CrossRef]
- Ford, T.J.; Berry, C. How to Diagnose and Manage Angina Without Obstructive Coronary Artery Disease: Lessons from the British Heart Foundation CorMicA Trial. Interv. Cardiol. 2019, 14, 76–82. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ford, T.J.; Yii, E.; Sidik, N.; Good, R.; Rocchiccioli, P.; McEntegart, M.; Watkins, S.; Eteiba, H.; Shaukat, A.; Lindsay, M.; et al. Ischemia and No Obstructive Coronary Artery Disease. Circ. Cardiovasc. Interv. 2019, 12, e008126. [Google Scholar] [CrossRef] [PubMed]
- Scalone, G.; Niccoli, G.; Crea, F. Editor’s Choice- Pathophysiology, diagnosis and management of MINOCA: An update. Eur. Heart J. Acute Cardiovasc. Care 2018, 8, 54–62. [Google Scholar] [CrossRef] [PubMed]
- Niccoli, G.; Camici, P.G. Myocardial infarction with non-obstructive coronary arteries: What is the prognosis? Eur. Heart J. Suppl. 2020, 22, E40–E45. [Google Scholar] [CrossRef]
- Pasupathy, S.; Tavella, R.; Beltrame, J.F. Myocardial Infarction With Nonobstructive Coronary Arteries (MINOCA). Circulation 2017, 135, 1490–1493. [Google Scholar] [CrossRef]
- Niccoli, G.; Scalone, G.; Lerman, A.; Crea, F. Coronary microvascular obstruction in acute myocardial infarction. Eur. Heart J. 2016, 37, 1024–1033. [Google Scholar] [CrossRef] [Green Version]
- Collste, O.; Sörensson, P.; Frick, M.; Agewall, S.; Daniel, M.; Henareh, L.; Ekenbäck, C.; Eurenius, L.; Guiron, C.; Jernberg, T.; et al. Myocardial infarction with normal coronary arteries is common and associated with normal findings on cardiovascular magnetic resonance imaging: Results from the Stockholm Myocardial Infarction with Normal Coronaries study. J. Intern. Med. 2013, 273, 189–196. [Google Scholar] [CrossRef]
- Pasupathy, S.; Air, T.; Dreyer, R.P.; Tavella, R.; Beltrame, J.F. Systematic Review of Patients Presenting With Suspected Myocardial Infarction and Nonobstructive Coronary Arteries. Circulation 2015, 131, 861–870. [Google Scholar] [CrossRef] [Green Version]
- Pacheco Claudio, C.; Quesada, O.; Pepine, C.J.; Noel Bairey Merz, C. Why names matter for women: MINOCA/INOCA (myocardial infarction/ischemia and no obstructive coronary artery disease). Clin. Cardiol. 2018, 41, 185–193. [Google Scholar] [CrossRef]
- Montone, R.A.; Niccoli, G.; Fracassi, F.; Russo, M.; Gurgoglione, F.; Cammà, G.; Lanza, G.A.; Crea, F. Patients with acute myocardial infarction and non-obstructive coronary arteries: Safety and prognostic relevance of invasive coronary provocative tests. Eur. Heart J. 2017, 39, 91–98. [Google Scholar] [CrossRef]
- Mahajan, A.M.; Gandhi, H.; Smilowitz, N.R.; Roe, M.T.; Hellkamp, A.S.; Chiswell, K.; Gulati, M.; Reynolds, H.R. Seasonal and circadian patterns of myocardial infarction by coronary artery disease status and sex in the ACTION Registry-GWTG. Int. J. Cardiol. 2019, 274, 16–20. [Google Scholar] [CrossRef] [PubMed]
- Aziz, A.; Hansen, H.S.; Sechtem, U.; Prescott, E.; Ong, P. Sex-Related Differences in Vasomotor Function in Patients With Angina and Unobstructed Coronary Arteries. J. Am. Coll. Cardiol. 2017, 70, 2349–2358. [Google Scholar] [CrossRef] [PubMed]
- Taqueti, V.R.; Shaw, L.J.; Cook, N.R.; Murthy, V.L.; Shah, N.R.; Foster, C.R.; Hainer, J.; Blankstein, R.; Dorbala, S.; Carli, M.F.D. Excess Cardiovascular Risk in Women Relative to Men Referred for Coronary Angiography Is Associated With Severely Impaired Coronary Flow Reserve, Not Obstructive Disease. Circulation 2017, 135, 566–577. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Smilowitz, N.R.; Sampson, B.A.; Abrecht, C.R.; Siegfried, J.S.; Hochman, J.S.; Reynolds, H.R. Women have less severe and extensive coronary atherosclerosis in fatal cases of ischemic heart disease: An autopsy study. Am. Heart J. 2011, 161, 681–688. [Google Scholar] [CrossRef] [PubMed]
- Waheed, N.; Elias-Smale, S.; Malas, W.; Maas, A.H.; Sedlak, T.L.; Tremmel, J.; Mehta, P.K. Sex differences in non-obstructive coronary artery disease. Cardiovasc. Res. 2020, 116, 829–840. [Google Scholar] [CrossRef] [PubMed]
- Murthy, V.L.; Naya, M.; Foster, C.R.; Gaber, M.; Hainer, J.; Klein, J.; Dorbala, S.; Blankstein, R.; Carli, M.F.D. Association Between Coronary Vascular Dysfunction and Cardiac Mortality in Patients With and Without Diabetes Mellitus. Circulation 2012, 126, 1858–1868. [Google Scholar] [CrossRef] [Green Version]
- Tamis-Holland, J.E.; Jneid, H.; Reynolds, H.R.; Agewall, S.; Brilakis, E.S.; Brown, T.M.; Lerman, A.; Cushman, M.; Kumbhani, D.J.; Arslanian-Engoren, C.; et al. Contemporary Diagnosis and Management of Patients With Myocardial Infarction in the Absence of Obstructive Coronary Artery Disease: A Scientific Statement From the American Heart Association. Circulation 2019, 139, e891–e908. [Google Scholar] [CrossRef]
- Tunc, E.; Eve, A.A.; Madak-Erdogan, Z. Coronary Microvascular Dysfunction and Estrogen Receptor Signaling. Trends Endocrinol. Metab. 2020, 31, 228–238. [Google Scholar] [CrossRef]
- Crea, F.; Camici, P.G.; Bairey Merz, C.N. Coronary microvascular dysfunction: An update. Eur. Heart J. 2014, 35, 1101–1111. [Google Scholar] [CrossRef] [Green Version]
- Sechtem, U.; Brown, D.; Godo, S.; Lanza, G.A.; Shimokawa, H.; Sidik, N. Coronary microvascular dysfunction in stable ischaemic heart disease (non-obstructive coronary artery disease and obstructive coronary artery disease). Cardiovasc. Res. 2020, 116, 771–786. [Google Scholar] [CrossRef] [Green Version]
- Hamasaki, S.; Al Suwaidi, J.; Higano, S.T.; Miyauchi, K.; Holmes, D.R.; Lerman, A. Attenuated coronary flow reserve and vascular remodeling in patients with hypertension and left ventricular hypertrophy. J. Am. Coll. Cardiol. 2000, 35, 1654–1660. [Google Scholar] [CrossRef] [Green Version]
- Dweck, M.R.; Boon, N.A.; Newby, D.E. Calcific Aortic Stenosis: A Disease of the Valve and the Myocardium. J. Am. Coll. Cardiol. 2012, 60, 1854–1863. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Majmudar, M.D.; Murthy, V.L.; Shah, R.V.; Kolli, S.; Mousavi, N.; Foster, C.R.; Hainer, J.; Blankstein, R.; Dorbala, S.; Sitek, A.; et al. Quantification of coronary flow reserve in patients with ischaemic and non-ischaemic cardiomyopathy and its association with clinical outcomes. Eur. Heart J. Cardiovasc. Imaging 2015, 16, 900–909. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cecchi, F.; Olivotto, I.; Gistri, R.; Lorenzoni, R.; Chiriatti, G.; Camici, P.G. Coronary Microvascular Dysfunction and Prognosis in Hypertrophic Cardiomyopathy. N. Engl. J. Med. 2003, 349, 1027–1035. [Google Scholar] [CrossRef] [PubMed]
- Dorbala, S.; Vangala, D.; Bruyere, J.; Quarta, C.; Kruger, J.; Padera, R.; Foster, C.; Hanley, M.; Di Carli, M.F.; Falk, R. Coronary Microvascular Dysfunction Is Related to Abnormalities in Myocardial Structure and Function in Cardiac Amyloidosis. JACC Heart Fail. 2014, 2, 358–367. [Google Scholar] [CrossRef]
- Pelliccia, F.; Kaski, J.C.; Crea, F.; Camici, P.G. Pathophysiology of Takotsubo Syndrome. Circulation 2017, 135, 2426–2441. [Google Scholar] [CrossRef]
- Paulus, W.J.; Tschöpe, C. A Novel Paradigm for Heart Failure With Preserved Ejection Fraction: Comorbidities Drive Myocardial Dysfunction and Remodeling Through Coronary Microvascular Endothelial Inflammation. J. Am. Coll. Cardiol. 2013, 62, 263–271. [Google Scholar] [CrossRef] [Green Version]
- Shah, S.J.; Lam, C.S.P.; Svedlund, S.; Saraste, A.; Hage, C.; Tan, R.-S.; Beussink-Nelson, L.; Ljung Faxén, U.; Fermer, M.L.; Broberg, M.A.; et al. Prevalence and correlates of coronary microvascular dysfunction in heart failure with preserved ejection fraction: PROMIS-HFpEF. Eur. Heart J. 2018, 39, 3439–3450. [Google Scholar] [CrossRef]
- Lee, J.F.; Barrett-O’Keefe, Z.; Garten, R.S.; Nelson, A.D.; Ryan, J.J.; Nativi, J.N.; Richardson, R.S.; Wray, D.W. Evidence of microvascular dysfunction in heart failure with preserved ejection fraction. Heart 2016, 102, 278–284. [Google Scholar] [CrossRef] [Green Version]
- Taqueti, V.R.; Solomon, S.D.; Shah, A.M.; Desai, A.S.; Groarke, J.D.; Osborne, M.T.; Hainer, J.; Bibbo, C.F.; Dorbala, S.; Blankstein, R.; et al. Coronary microvascular dysfunction and future risk of heart failure with preserved ejection fraction. Eur. Heart J. 2018, 39, 840–849. [Google Scholar] [CrossRef]
- Crea, F.; Bairey Merz, C.N.; Beltrame, J.F.; Kaski, J.C.; Ogawa, H.; Ong, P.; Sechtem, U.; Shimokawa, H.; Camici, P.G.; Coronary Vasomotion Disorders International Study Group (COVADIS). The parallel tales of microvascular angina and heart failure with preserved ejection fraction: A paradigm shift. Eur. Heart J. 2017, 38, 473–477. [Google Scholar] [CrossRef] [PubMed]
- Camici, P.G.; Tschöpe, C.; Di Carli, M.F.; Rimoldi, O.; Van Linthout, S. Coronary microvascular dysfunction in hypertrophy and heart failure. Cardiovasc. Res. 2020, 116, 806–816. [Google Scholar] [CrossRef] [PubMed]
- Schiattarella, G.G.; Sequeira, V.; Ameri, P. Distinctive patterns of inflammation across the heart failure syndrome. Heart Fail. Rev. 2020, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Lakhani, I.; Wong, M.V.; Hung, J.K.F.; Gong, M.; Waleed, K.B.; Xia, Y.; Lee, S.; Roever, L.; Liu, T.; Tse, G.; et al. Diagnostic and prognostic value of serum C-reactive protein in heart failure with preserved ejection fraction: A systematic review and meta-analysis. Heart Fail. Rev. 2020, 1–10. [Google Scholar] [CrossRef] [Green Version]
- Bork, N.I.; Molina, C.E.; Nikolaev, V.O. cGMP signalling in cardiomyocyte microdomains. Biochem. Soc. Trans. 2019, 47, 1327–1339. [Google Scholar] [CrossRef]
- Heerebeek, L.v.; Hamdani, N.; Falcão-Pires, I.; Leite-Moreira, A.F.; Begieneman, M.P.V.; Bronzwaer, J.G.F.; Velden, J.v.d.; Stienen, G.J.M.; Laarman, G.J.; Somsen, A.; et al. Low Myocardial Protein Kinase G Activity in Heart Failure With Preserved Ejection Fraction. Circulation 2012, 126, 830–839. [Google Scholar] [CrossRef] [Green Version]
- Liu, G.; Ma, C.; Yang, H.; Zhang, P.-Y. Transforming growth factor β and its role in heart disease. Exp. Med. 2017, 13, 2123–2128. [Google Scholar] [CrossRef] [Green Version]
- Harvey, A.; Montezano, A.C.; Lopes, R.A.; Rios, F.; Touyz, R.M. Vascular Fibrosis in Aging and Hypertension: Molecular Mechanisms and Clinical Implications. Can. J. Cardiol. 2016, 32, 659–668. [Google Scholar] [CrossRef] [Green Version]
- Tschöpe, C.; Van Linthout, S. New Insights in (Inter)Cellular Mechanisms by Heart Failure with Preserved Ejection Fraction. Curr Heart Fail. Rep. 2014, 11, 436–444. [Google Scholar] [CrossRef] [Green Version]
- Franssen, C.; Chen, S.; Unger, A.; Korkmaz, H.I.; De Keulenaer, G.W.; Tschöpe, C.; Leite-Moreira, A.F.; Musters, R.; Niessen, H.W.M.; Linke, W.A.; et al. Myocardial Microvascular Inflammatory Endothelial Activation in Heart Failure With Preserved Ejection Fraction. JACC Heart Fail. 2016, 4, 312–324. [Google Scholar] [CrossRef]
- Tschöpe, C.; Post, H. Latent ischaemia as a trigger for a circulus vitiosus of inflammation, fibrosis, and stiffness in HFPEF. Eur. J. Heart Fail. 2015, 17, 1210–1212. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- van Heerebeek, L.; Borbély, A.; Niessen, H.W.M.; Bronzwaer, J.G.F.; van der Velden, J.; Stienen, G.J.M.; Linke, W.A.; Laarman, G.J.; Paulus, W.J. Myocardial Structure and Function Differ in Systolic and Diastolic Heart Failure. Circulation 2006, 113, 1966–1973. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Burchfield, J.S.; Xie, M.; Hill, J.A. Pathological Ventricular Remodeling. Circulation 2013, 128, 388–400. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sanders-van Wijk, S.; van Empel, V.; Davarzani, N.; Maeder, M.T.; Handschin, R.; Pfisterer, M.E.; Brunner-La Rocca, H.P.; TIME-CHF Investigators. Circulating biomarkers of distinct pathophysiological pathways in heart failure with preserved vs. reduced left ventricular ejection fraction. Eur. J. Heart Fail. 2015, 17, 1006–1014. [Google Scholar] [CrossRef]
- Berry, C.; Sidik, N.; Pereira, A.C.; Ford, T.J.; Touyz, R.M.; Kaski, J.-C.; Hainsworth, A.H. Small-Vessel Disease in the Heart and Brain: Current Knowledge, Unmet Therapeutic Need, and Future Directions. J. Am. Heart Assoc. 2019, 8, e011104. [Google Scholar] [CrossRef]
- Conijn, M.M.A.; Kloppenborg, R.P.; Algra, A.; Mali, W.P.T.M.; Kappelle, L.J.; Vincken, K.L.; Graaf, Y.v.d.; Geerlings, M.I. Cerebral Small Vessel Disease and Risk of Death, Ischemic Stroke, and Cardiac Complications in Patients With Atherosclerotic Disease. Stroke 2011, 42, 3105–3109. [Google Scholar] [CrossRef]
- Wardlaw, J.M.; Allerhand, M.; Doubal, F.N.; Valdes Hernandez, M.; Morris, Z.; Gow, A.J.; Bastin, M.; Starr, J.M.; Dennis, M.S.; Deary, I.J. Vascular risk factors, large-artery atheroma, and brain white matter hyperintensities. Neurology 2014, 82, 1331–1338. [Google Scholar] [CrossRef] [Green Version]
- Pantoni, L. Cerebral small vessel disease: From pathogenesis and clinical characteristics to therapeutic challenges. Lancet Neurol. 2010, 9, 689–701. [Google Scholar] [CrossRef]
- Wardlaw, J.M.; Smith, E.E.; Biessels, G.J.; Cordonnier, C.; Fazekas, F.; Frayne, R.; Lindley, R.I.; O’Brien, J.T.; Barkhof, F.; Benavente, O.R.; et al. Neuroimaging standards for research into small vessel disease and its contribution to ageing and neurodegeneration. Lancet Neurol. 2013, 12, 822–838. [Google Scholar] [CrossRef] [Green Version]
- Wardlaw, J.M.; Smith, C.; Dichgans, M. Mechanisms of sporadic cerebral small vessel disease: Insights from neuroimaging. Lancet Neurol. 2013, 12, 483–497. [Google Scholar] [CrossRef] [Green Version]
- Makin, S.D.J.; Turpin, S.; Dennis, M.S.; Wardlaw, J.M. Cognitive impairment after lacunar stroke: Systematic review and meta-analysis of incidence, prevalence and comparison with other stroke subtypes. J. Neurol. Neurosurg. Psychiatry 2013, 84, 893–900. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Marek, M.; Horyniecki, M.; Frączek, M.; Kluczewska, E. Leukoaraiosis—New concepts and modern imaging. Pol J. Radiol 2018, 83, e76–e81. [Google Scholar] [CrossRef] [PubMed]
- Wardlaw, J.M.; Makin, S.J.; Valdés Hernández, M.C.; Armitage, P.A.; Heye, A.K.; Chappell, F.M.; Muñoz-Maniega, S.; Sakka, E.; Shuler, K.; Dennis, M.S.; et al. Blood-brain barrier failure as a core mechanism in cerebral small vessel disease and dementia: Evidence from a cohort study. Alzheimer’s Dement. 2017, 13, 634–643. [Google Scholar] [CrossRef] [Green Version]
- Gorelick, P.B.; Scuteri, A.; Black, S.E.; DeCarli, C.; Greenberg, S.M.; Iadecola, C.; Launer, L.J.; Laurent, S.; Lopez, O.L.; Nyenhuis, D. Vascular contributions to cognitive impairment and dementia: A statement for healthcare professionals from the American Heart Association/American Stroke Association. Stroke 2011, 42, 2672–2713. [Google Scholar] [CrossRef] [PubMed]
- Wardlaw, J.M. Blood-brain barrier and cerebral small vessel disease. J. Neurol. Sci. 2010, 299, 66–71. [Google Scholar] [CrossRef]
- Knottnerus, I.L.H.; Ten Cate, H.; Lodder, J.; Kessels, F.; van Oostenbrugge, R.J. Endothelial Dysfunction in Lacunar Stroke: A Systematic Review. Cerebrovasc. Dis. 2009, 27, 519–526. [Google Scholar] [CrossRef]
- Horsburgh, K.; Wardlaw, J.M.; van Agtmael, T.; Allan, S.M.; Ashford, M.L.J.; Bath, P.M.; Brown, R.; Berwick, J.; Cader, M.Z.; Carare, R.O.; et al. Small vessels, dementia and chronic diseases–molecular mechanisms and pathophysiology. Clin. Sci. 2018, 132, 851–868. [Google Scholar] [CrossRef] [Green Version]
- Obermeier, B.; Daneman, R.; Ransohoff, R.M. Development, maintenance and disruption of the blood-brain barrier. Nat. Med. 2013, 19, 1584–1596. [Google Scholar] [CrossRef]
- Low, A.; Mak, E.; Rowe, J.B.; Markus, H.S.; O’Brien, J.T. Inflammation and cerebral small vessel disease: A systematic review. Ageing Res. Rev. 2019, 53, 100916. [Google Scholar] [CrossRef]
- Rouhl, R.P.W.; Damoiseaux, J.G.; Lodder, J.; Theunissen, R.O.; Knottnerus, I.L.; Staals, J.; Henskens, L.H.; Kroon, A.A.; De Leeuw, P.W.; Tervaert, J.W.C.; et al. Vascular inflammation in cerebral small vessel disease. Neurobiol. Aging 2012, 33, 1800–1806. [Google Scholar] [CrossRef] [Green Version]
- Hassan, A.; Hunt, B.J.; O’Sullivan, M.; Parmar, K.; Bamford, J.M.; Briley, D.; Brown, M.M.; Thomas, D.J.; Markus, H.S. Markers of endothelial dysfunction in lacunar infarction and ischaemic leukoaraiosis. Brain 2003, 126, 424–432. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Walker, K.A.; Power, M.C.; Hoogeveen, R.C.; Folsom, A.R.; Ballantyne, C.M.; Knopman, D.S.; Windham, B.G.; Selvin, E.; Jack, C.R.; Gottesman, R.F. Midlife Systemic Inflammation, Late-Life White Matter Integrity, and Cerebral Small Vessel Disease. Stroke 2017, 48, 3196–3202. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.E.; Wong, S.M.; Van De Haar, H.J.; Staals, J.; Jansen, J.; Jeukens, C.R.; Hofman, P.A.M.; Van Oostenbrugge, R.J.; Backes, W. Blood–brain barrier leakage is more widespread in patients with cerebral small vessel disease. Neurology 2016, 88, 426–432. [Google Scholar] [CrossRef] [PubMed]
- Wong, S.M.; Jansen, J.F.A.; Zhang, C.E.; Hoff, E.I.; Staals, J.; van Oostenbrugge, R.J.; Backes, W.H. Blood-brain barrier impairment and hypoperfusion are linked in cerebral small vessel disease. Neurology 2019, 92, E1669–E1677. [Google Scholar] [CrossRef] [PubMed]
- Koizumi, K.; Wang, G.; Park, L. Endothelial Dysfunction and Amyloid-β-Induced Neurovascular Alterations. Cell. Mol. Neurobiol. 2016, 36, 155–165. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kloner, R.A.; King, K.S.; Harrington, M.G. No-reflow phenomenon in the heart and brain. Am. J. Physiol. Heart Circ. Physiol. 2018, 315, H550–H562. [Google Scholar] [CrossRef]
- Nie, X.; Pu, Y.; Zhang, Z.; Liu, X.; Duan, W.; Liu, L. Futile Recanalization after Endovascular Therapy in Acute Ischemic Stroke. Biomed Res. Int. 2018, 2018, 5879548. [Google Scholar] [CrossRef] [Green Version]
- del Zoppo, G.J. Virchow’s triad: The vascular basis of cerebral injury. Rev. Neurol. Dis. 2008, 5, S12–S21. [Google Scholar]
- Burrows, F.E.; Bray, N.; Denes, A.; Allan, S.M.; Schiessl, I. Delayed Reperfusion Deficits after Experimental Stroke Account for Increased Pathophysiology. J. Cereb. Blood Flow Metab. 2015, 35, 277–284. [Google Scholar] [CrossRef] [Green Version]
- Hall, C.N.; Reynell, C.; Gesslein, B.; Hamilton, N.B.; Mishra, A.; Sutherland, B.A.; O’Farrell, F.M.; Buchan, A.M.; Lauritzen, M.; Attwell, D. Capillary pericytes regulate cerebral blood flow in health and disease. Nature 2014, 508, 55–60. [Google Scholar] [CrossRef] [Green Version]
- Yemisci, M.; Gursoy-Ozdemir, Y.; Vural, A.; Can, A.; Topalkara, K.; Dalkara, T. Pericyte contraction induced by oxidative-nitrative stress impairs capillary reflow despite successful opening of an occluded cerebral artery. Nat. Med. 2009, 15, 1031–1037. [Google Scholar] [CrossRef]
- Kisler, K.; Nelson, A.R.; Rege, S.V.; Ramanathan, A.; Wang, Y.; Ahuja, A.; Lazic, D.; Tsai, P.S.; Zhao, Z.; Zhou, Y.; et al. Pericyte degeneration leads to neurovascular uncoupling and limits oxygen supply to brain. Nat. Neurosci. 2017, 20, 406–416. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Khatri, P.; Yeatts, S.D.; Mazighi, M.; Broderick, J.P.; Liebeskind, D.S.; Demchuk, A.M.; Amarenco, P.; Carrozzella, J.; Spilker, J.; Foster, L.D.; et al. Time to angiographic reperfusion and clinical outcome after acute ischaemic stroke: An analysis of data from the Interventional Management of Stroke (IMS III) phase 3 trial. Lancet Neurol. 2014, 13, 567–574. [Google Scholar] [CrossRef] [Green Version]
- del Zoppo, G.J.; Sharp, F.R.; Heiss, W.-D.; Albers, G.W. Heterogeneity in the penumbra. J. Cereb. Blood Flow Metab. 2011, 31, 1836–1851. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Di Carli, M.F.; Janisse, J.; Ager, J.; Grunberger, G. Role of chronic hyperglycemia in the pathogenesis of coronary microvascular dysfunction in diabetes. J. Am. Coll. Cardiol. 2003, 41, 1387–1393. [Google Scholar] [CrossRef] [Green Version]
- von Scholten, B.J.; Hasbak, P.; Christensen, T.E.; Ghotbi, A.A.; Kjaer, A.; Rossing, P.; Hansen, T.W. Cardiac 82Rb PET/CT for fast and non-invasive assessment of microvascular function and structure in asymptomatic patients with type 2 diabetes. Diabetologia 2016, 59, 371–378. [Google Scholar] [CrossRef] [Green Version]
- Sezer, M.; Kocaaga, M.; Aslanger, E.; Atici, A.; Demirkiran, A.; Bugra, Z.; Umman, S.; Umman, B. Bimodal Pattern of Coronary Microvascular Involvement in Diabetes Mellitus. J. Am. Heart Assoc. 2016, 5, e003995. [Google Scholar] [CrossRef] [Green Version]
- Wasserman, D.H.; Wang, T.J.; Brown, N.J. The Vasculature in Prediabetes. Circ. Res. 2018, 122, 1135–1150. [Google Scholar] [CrossRef]
- Osborne, M.T.; Bajaj, N.S.; Taqueti, V.R.; Gupta, A.; Bravo, P.E.; Hainer, J.; Bibbo, C.F.; Dorbala, S.; Blankstein, R.; Di Carli, M.F. Coronary Microvascular Dysfunction Identifies Patients at High Risk of Adverse Events Across Cardiometabolic Diseases. J. Am. Coll. Cardiol. 2017, 70, 2835–2837. [Google Scholar] [CrossRef]
- Mazurek, T.; Zhang, L.; Zalewski, A.; Mannion, J.D.; Diehl, J.T.; Arafat, H.; Sarov-Blat, L.; O’Brien, S.; Keiper, E.A.; Johnson, A.G.; et al. Human Epicardial Adipose Tissue Is a Source of Inflammatory Mediators. Circulation 2003, 108, 2460–2466. [Google Scholar] [CrossRef] [Green Version]
- Bettencourt, N.; Toschke, A.M.; Leite, D.; Rocha, J.; Carvalho, M.; Sampaio, F.; Xará, S.; Leite-Moreira, A.; Nagel, E.; Gama, V. Epicardial adipose tissue is an independent predictor of coronary atherosclerotic burden. Int. J. Cardiol. 2012, 158, 26–32. [Google Scholar] [CrossRef] [PubMed]
- Feihl, F.; Liaudet, L.; Waeber, B.; Levy, B.I. Hypertension: A Disease of the Microcirculation? Hypertension 2006, 48, 1012–1017. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Brush, J.E.; Cannon, R.O.; Schenke, W.H.; Bonow, R.O.; Leon, M.B.; Maron, B.J.; Epstein, S.E. Angina Due to Coronary Microvascular Disease in Hypertensive Patients without Left Ventricular Hypertrophy. N. Engl. J. Med. 1988, 319, 1302–1307. [Google Scholar] [CrossRef] [PubMed]
- Drazner, M.H. The Progression of Hypertensive Heart Disease. Circulation 2011, 123, 327–334. [Google Scholar] [CrossRef] [PubMed]
- Mehta, S.K.; Rame, J.E.; Khera, A.; Murphy, S.A.; Canham, R.M.; Peshock, R.M.; de Lemos, J.A.; Drazner, M.H. Left Ventricular Hypertrophy, Subclinical Atherosclerosis, and Inflammation. Hypertension 2007, 49, 1385–1391. [Google Scholar] [CrossRef] [Green Version]
- Drazner, M.H. The Transition From Hypertrophy to Failure. Circulation 2005, 112, 936–938. [Google Scholar] [CrossRef] [Green Version]
- Rosen, B.D.; Edvardsen, T.; Lai, S.; Castillo, E.; Pan, L.; Jerosch-Herold, M.; Sinha, S.; Kronmal, R.; Arnett, D.; Crouse, J.R.; et al. Left Ventricular Concentric Remodeling Is Associated With Decreased Global and Regional Systolic Function. Circulation 2005, 112, 984–991. [Google Scholar] [CrossRef] [Green Version]
- Zhou, W.; Brown, J.M.; Bajaj, N.S.; Chandra, A.; Divakaran, S.; Weber, B.; Bibbo, C.F.; Hainer, J.; Taqueti, V.R.; Dorbala, S.; et al. Hypertensive coronary microvascular dysfunction: A subclinical marker of end organ damage and heart failure. Eur. Heart J. 2020, 41, 2366–2375. [Google Scholar] [CrossRef] [Green Version]
- Tsujita, K.; Yamanaga, K.; Komura, N.; Sakamoto, K.; Miyazaki, T.; Ishii, M.; Tabata, N.; Akasaka, T.; Sueta, D.; Arima, Y.; et al. Impact of left ventricular hypertrophy on impaired coronary microvascular dysfunction. Int. J. Cardiol. 2015, 187, 411–413. [Google Scholar] [CrossRef]
- Landmesser, U.; Drexler, H. Endothelial function and hypertension. Curr. Opin. Cardiol. 2007, 22, 316–320. [Google Scholar] [CrossRef]
- Gimelli, A.; Schneider-Eicke, J.; Neglia, D.; Sambuceti, G.; Giorgetti, A.; Bigalli, G.; Parodi, G.; Pedrinelli, R.; Parodi, O. Homogeneously Reduced Versus Regionally Impaired Myocardial Blood Flow in Hypertensive Patients: Two Different Patterns of Myocardial Perfusion Associated With Degree of Hypertrophy. J. Am. Coll. Cardiol. 1998, 31, 366–373. [Google Scholar] [CrossRef] [Green Version]
- Escaned, J.; Lerman, L.O. Coronary microcirculation and hypertensive heart failure. Eur. Heart J. 2020, 41, 2376–2378. [Google Scholar] [CrossRef] [PubMed]
- Davies, J.E.; Whinnett, Z.I.; Francis, D.P.; Manisty, C.H.; Aguado-Sierra, J.; Willson, K.; Foale, R.A.; Malik, I.S.; Hughes, A.D.; Parker, K.H.; et al. Evidence of a Dominant Backward-Propagating “Suction” Wave Responsible for Diastolic Coronary Filling in Humans, Attenuated in Left Ventricular Hypertrophy. Circulation 2006, 113, 1768–1778. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Raphael, C.E.; Cooper, R.; Parker, K.H.; Collinson, J.; Vassiliou, V.; Pennell, D.J.; de Silva, R.; Hsu, L.Y.; Greve, A.M.; Nijjer, S.; et al. Mechanisms of Myocardial Ischemia in Hypertrophic Cardiomyopathy: Insights From Wave Intensity Analysis and Magnetic Resonance. J. Am. Coll. Cardiol. 2016, 68, 1651–1660. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Johansson, B.; Mörner, S.; Waldenström, A.; Stål, P. Myocardial capillary supply is limited in hypertrophic cardiomyopathy: A morphological analysis. Int. J. Cardiol. 2008, 126, 252–257. [Google Scholar] [CrossRef]
- Ismail, T.F.; Hsu, L.-Y.; Greve, A.M.; Gonçalves, C.; Jabbour, A.; Gulati, A.; Hewins, B.; Mistry, N.; Wage, R.; Roughton, M.; et al. Coronary microvascular ischemia in hypertrophic cardiomyopathy—A pixel-wise quantitative cardiovascular magnetic resonance perfusion study. J. Cardiovasc. Magn. Reson. 2014, 16, 49. [Google Scholar] [CrossRef] [PubMed]
- Olivotto, I.; Cecchi, F.; Gistri, R.; Lorenzoni, R.; Chiriatti, G.; Girolami, F.; Torricelli, F.; Camici, P.G. Relevance of Coronary Microvascular Flow Impairment to Long-Term Remodeling and Systolic Dysfunction in Hypertrophic Cardiomyopathy. J. Am. Coll. Cardiol. 2006, 47, 1043–1048. [Google Scholar] [CrossRef] [Green Version]
- Choudhury, L.; Rosen, S.D.; Patel, D.; Nihoyannopoulos, P.; Camici, P.G. Coronary vasodilator reserve in primary and secondary left ventricular hypertrophy: A study with positron emission tomography. Eur. Heart J. 1997, 18, 108–116. [Google Scholar] [CrossRef]
- Rajappan, K.; Rimoldi, O.E.; Dutka, D.P.; Ariff, B.; Pennell, D.J.; Sheridan, D.J.; Camici, P.G. Mechanisms of Coronary Microcirculatory Dysfunction in Patients With Aortic Stenosis and Angiographically Normal Coronary Arteries. Circulation 2002, 105, 470–476. [Google Scholar] [CrossRef] [Green Version]
- Rajappan, K.; Rimoldi, O.E.; Camici, P.G.; Bellenger, N.G.; Pennell, D.J.; Sheridan, D.J. Functional Changes in Coronary Microcirculation After Valve Replacement in Patients With Aortic Stenosis. Circulation 2003, 107, 3170–3175. [Google Scholar] [CrossRef] [Green Version]
- Schwartzkopff, B.; Frenzel, H.; Diekerhoff, J.; Betz, P.; Flasshove, M.; Schulte, H.D.; Mundhenke, M.; Motz, W.; Strauer, B.E. Morphometric investigation of human myocardium in arterial hypertension and valvular aortic stenosis. Eur. Heart J. 1992, 13, 17–23. [Google Scholar] [CrossRef] [PubMed]
- Amann, K.; Wanner, C.; Ritz, E. Cross-Talk between the Kidney and the Cardiovascular System. J. Am. Soc. Nephrol. 2006, 17, 2112–2119. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rangaswami, J.; Bhalla, V.; Blair, J.E.A.; Chang, T.I.; Costa, S.; Lentine, K.L.; Lerma, E.V.; Mezue, K.; Molitch, M.; Mullens, W.; et al. Cardiorenal Syndrome: Classification, Pathophysiology, Diagnosis, and Treatment Strategies: A Scientific Statement From the American Heart Association. Circulation 2019, 139, e840–e878. [Google Scholar] [CrossRef] [PubMed]
- Gansevoort, R.T.; Correa-Rotter, R.; Hemmelgarn, B.R.; Jafar, T.H.; Heerspink, H.J.L.; Mann, J.F.; Matsushita, K.; Wen, C.P. Chronic kidney disease and cardiovascular risk: Epidemiology, mechanisms, and prevention. Lancet 2013, 382, 339–352. [Google Scholar] [CrossRef]
- Schiffrin, E.L.; Lipman, M.L.; Mann, J.F.E. Chronic Kidney Disease. Effects on the cardiovascular system. Circulation 2007, 116, 85–97. [Google Scholar] [CrossRef] [Green Version]
- Bajaj, N.S.; Singh, A.; Zhou, W.; Gupta, A.; Fujikura, K.; Byrne, C.; Harms, H.J.; Osborne, M.T.; Bravo, P.; Andrikopolou, E.; et al. Coronary Microvascular Dysfunction, Left Ventricular Remodeling, and Clinical Outcomes in Patients With Chronic Kidney Impairment. Circulation 2020, 141, 21–33. [Google Scholar] [CrossRef]
- Nelson, A.J.; Dundon, B.K.; Worthley, S.G.; Richardson, J.D.; Puri, R.; Wong, D.T.L.; Coates, P.T.; Faull, R.J.; Worthley, M.I. End-stage renal failure is associated with impaired coronary microvascular function. Coron. Artery Dis. 2019, 30, 520–527. [Google Scholar] [CrossRef]
- Charytan, D.M.; Skali, H.; Shah, N.R.; Veeranna, V.; Cheezum, M.K.; Taqueti, V.R.; Kato, T.; Bibbo, C.R.; Hainer, J.; Dorbala, S.; et al. Coronary flow reserve is predictive of the risk of cardiovascular death regardless of chronic kidney disease stage. Kidney Int. 2018, 93, 501–509. [Google Scholar] [CrossRef]
- Fliser, D.; Wiecek, A.; Suleymanlar, G.; Ortiz, A.; Massy, Z.; Lindholm, B.; Martinez-Castelao, A.; Agarwal, R.; Jager, K.J.; Dekker, F.W.; et al. The dysfunctional endothelium in CKD and in cardiovascular disease: Mapping the origin(s) of cardiovascular problems in CKD and of kidney disease in cardiovascular conditions for a research agenda. Kidney Int. Suppl. 2011, 1, 6–9. [Google Scholar] [CrossRef] [Green Version]
- Prommer, H.-U.; Maurer, J.; von Websky, K.; Freise, C.; Sommer, K.; Nasser, H.; Samapati, R.; Reglin, B.; Guimarães, P.; Pries, A.R.; et al. Chronic kidney disease induces a systemic microangiopathy, tissue hypoxia and dysfunctional angiogenesis. Sci. Rep. 2018, 8, 5317. [Google Scholar] [CrossRef]
- Charytan, D.M.; Padera, R.; Helfand, A.M.; Zeisberg, M.; Xu, X.; Liu, X.; Himmelfarb, J.; Cinelli, A.; Kalluri, R.; Zeisberg, E.M. Increased concentration of circulating angiogenesis and nitric oxide inhibitors induces endothelial to mesenchymal transition and myocardial fibrosis in patients with chronic kidney disease. Int. J. Cardiol. 2014, 176, 99–109. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cachofeiro, V.; Goicochea, M.; de Vinuesa, S.G.; Oubiña, P.; Lahera, V.; Luño, J. Oxidative stress and inflammation, a link between chronic kidney disease and cardiovascular disease. Kidney Int. 2008, 74, S4–S9. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Himmelfarb, J.; Stenvinkel, P.; Ikizler, T.A.; Hakim, R.M. The elephant in uremia: Oxidant stress as a unifying concept of cardiovascular disease in uremia. Kidney Int. 2002, 62, 1524–1538. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Prasad, M.; Hermann, J.; Gabriel, S.E.; Weyand, C.M.; Mulvagh, S.; Mankad, R.; Oh, J.K.; Matteson, E.L.; Lerman, A. Cardiorheumatology: Cardiac involvement in systemic rheumatic disease. Nat. Rev. Cardiol. 2015, 12, 168–176. [Google Scholar] [CrossRef]
- Ntusi, N.A.B.; Piechnik, S.K.; Francis, J.M.; Ferreira, V.M.; Matthews, P.M.; Robson, M.D.; Wordsworth, P.B.; Neubauer, S.; Karamitsos, T.D. Diffuse Myocardial Fibrosis and Inflammation in Rheumatoid Arthritis: Insights From CMR T1 Mapping. JACC Cardiovasc. Imaging 2015, 8, 526–536. [Google Scholar] [CrossRef] [Green Version]
- Gargiulo, P.; Marsico, F.; Parente, A.; Paolillo, S.; Cecere, M.; Casaretti, L.; Pellegrino, A.M.; Formisano, T.; Fabiani, I.; Soricelli, A.; et al. Ischemic heart disease in systemic inflammatory diseases. An appraisal. Int. J. Cardiol. 2014, 170, 286–290. [Google Scholar] [CrossRef]
- Mason, J.C.; Libby, P. Cardiovascular disease in patients with chronic inflammation: Mechanisms underlying premature cardiovascular events in rheumatologic conditions. Eur. Heart J. 2014, 36, 482–489. [Google Scholar] [CrossRef] [Green Version]
- Lira-Junior, R.; Figueredo, C.M.; Bouskela, E.; Fischer, R.G. Severe Chronic Periodontitis Is Associated With Endothelial and Microvascular Dysfunctions: A Pilot Study. J. Periodontol. 2014, 85, 1648–1657. [Google Scholar] [CrossRef]
- Caliskan, Z.; Gokturk, H.S.; Caliskan, M.; Gullu, H.; Ciftci, O.; Ozgur, G.T.; Guven, A.; Selcuk, H. Impaired coronary microvascular and left ventricular diastolic function in patients with inflammatory bowel disease. Microvasc. Res. 2015, 97, 25–30. [Google Scholar] [CrossRef]
- Liao, K.P.; Solomon, D.H. Traditional cardiovascular risk factors, inflammation and cardiovascular risk in rheumatoid arthritis. Rheumatology 2013, 52, 45–52. [Google Scholar] [CrossRef] [Green Version]
- Arida, A.; Protogerou, A.; Kitas, G.; Sfikakis, P. Systemic Inflammatory Response and Atherosclerosis: The Paradigm of Chronic Inflammatory Rheumatic Diseases. Int J. Mol. Sci 2018, 19, 1890. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bordy, R.; Totoson, P.; Prati, C.; Marie, C.; Wendling, D.; Demougeot, C. Microvascular endothelial dysfunction in rheumatoid arthritis. Nat. Rev. Rheumatol. 2018, 14, 404–420. [Google Scholar] [CrossRef] [PubMed]
- Steyers, C.; Miller, F. Endothelial Dysfunction in Chronic Inflammatory Diseases. Int J. Mol. Sci 2014, 15, 11324–11349. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Recio-Mayoral, A.; Mason, J.C.; Kaski, J.C.; Rubens, M.B.; Harari, O.A.; Camici, P.G. Chronic inflammation and coronary microvascular dysfunction in patients without risk factors for coronary artery disease. Eur. Heart J. 2009, 30, 1837–1843. [Google Scholar] [CrossRef] [Green Version]
- Ciftci, O.; Yilmaz, S.; Topcu, S.; Calıskan, M.; Gullu, H.; Erdogan, D.; Pamuk, B.O.; Yildirir, A.; Muderrisoglu, H. Impaired coronary microvascular function and increased intima-media thickness in rheumatoid arthritis. Atherosclerosis 2008, 198, 332–337. [Google Scholar] [CrossRef]
- Erre, G.L.; Buscetta, G.; Paliogiannis, P.; Mangoni, A.A.; Carru, C.; Passiu, G.; Zinellu, A. Coronary flow reserve in systemic rheumatic diseases: A systematic review and meta-analysis. Rheumatol. Int. 2018, 38, 1179–1190. [Google Scholar] [CrossRef]
- Sandhu, V.K.; Wei, J.; Thomson, L.E.J.; Berman, D.S.; Schapira, J.; Wallace, D.; Weisman, M.H.; Bairey Merz, C.N.; Ishimori, M.L. A Five-Year Follow up of Coronary Microvascular Dysfunction and Coronary Artery Disease in SLE: Results from a Community-Based Lupus Cohort. Arthritis Care Res. 2020, 72, 882–887. [Google Scholar] [CrossRef]
- Suda, A.; Takahashi, J.; Hao, K.; Kikuchi, Y.; Shindo, T.; Ikeda, S.; Sato, K.; Sugisawa, J.; Matsumoto, Y.; Miyata, S.; et al. Coronary Functional Abnormalities in Patients With Angina and Nonobstructive Coronary Artery Disease. J. Am. Coll. Cardiol. 2019, 74, 2350–2360. [Google Scholar] [CrossRef]
- De Bruyne, B.; Hersbach, F.; Pijls, N.H.J.; Bartunek, J.; Bech, J.-W.; Heyndrickx, G.R.; Gould, K.L.; Wijns, W. Abnormal Epicardial Coronary Resistance in Patients With Diffuse Atherosclerosis but “Normal” Coronary Angiography. Circulation 2001, 104, 2401–2406. [Google Scholar] [CrossRef] [Green Version]
- Sorop, O.; Merkus, D.; Beer, V.J.d.; Houweling, B.; Pistea, A.; McFalls, E.O.; Boomsma, F.; Beusekom, H.M.v.; Giessen, W.J.v.d.; VanBavel, E.; et al. Functional and Structural Adaptations of Coronary Microvessels Distal to a Chronic Coronary Artery Stenosis. Circ. Res. 2008, 102, 795–803. [Google Scholar] [CrossRef]
- Canty, J.M.; Suzuki, G. Myocardial perfusion and contraction in acute ischemia and chronic ischemic heart disease. J. Mol. Cell. Cardiol. 2012, 52, 822–831. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Niccoli, G.; Montone, R.A.; Lanza, G.A.; Crea, F. Angina after percutaneous coronary intervention: The need for precision medicine. Int. J. Cardiol. 2017, 248, 14–19. [Google Scholar] [CrossRef] [PubMed]
- Windecker, S.; Stortecky, S.; Stefanini, G.G.; daCosta, B.R.; Rutjes, A.W.; Di Nisio, M.; Siletta, M.G.; Maione, A.; Alfonso, F.; Clemmensen, P.M.; et al. Revascularisation versus medical treatment in patients with stable coronary artery disease: Network meta-analysis. BMJ Br. Med J. 2014, 348, g3859. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Stergiopoulos, K.; Boden, W.E.; Hartigan, P.; Möbius-Winkler, S.; Hambrecht, R.; Hueb, W.; Hardison, R.M.; Abbott, J.D.; Brown, D.L. Percutaneous Coronary Intervention Outcomes in Patients With Stable Obstructive Coronary Artery Disease and Myocardial Ischemia: A Collaborative Meta-analysis of Contemporary Randomized Clinical Trials. JAMA Intern. Med. 2014, 174. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- De Bruyne, B.; Fearon, W.F.; Pijls, N.H.J.; Barbato, E.; Tonino, P.; Piroth, Z.; Jagic, N.; Mobius-Winckler, S.; Rioufol, G.; Witt, N.; et al. Fractional Flow Reserve–Guided PCI for Stable Coronary Artery Disease. N. Engl. J. Med. 2014, 371, 1208–1217. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Johnson, N.P.; Kirkeeide, R.L.; Gould, K.L. Is Discordance of Coronary Flow Reserve and Fractional Flow Reserve Due to Methodology or Clinically Relevant Coronary Pathophysiology? JACC Cardiovasc. Imaging 2012, 5, 193–202. [Google Scholar] [CrossRef] [Green Version]
- Wong, D.T.L.; Puri, R.; Richardson, J.D.; Worthley, M.I.; Worthley, S.G. Myocardial ‘no-reflow’—Diagnosis, pathophysiology and treatment. Int. J. Cardiol. 2013, 167, 1798–1806. [Google Scholar] [CrossRef]
- Niccoli, G.; Burzotta, F.; Galiuto, L.; Crea, F. Myocardial No-Reflow in Humans. J. Am. Coll. Cardiol. 2009, 54, 281–292. [Google Scholar] [CrossRef]
- Sezer, M.; Royen, N.V.; Umman, B.; Bugra, Z.; Bulluck, H.; Hausenloy, D.J.; Umman, S. Coronary Microvascular Injury in Reperfused Acute Myocardial Infarction: A View From an Integrative Perspective. J. Am. Heart Assoc. 2018, 7, e009949. [Google Scholar] [CrossRef] [Green Version]
- Bulluck, H.; Foin, N.; Tan, J.W.; Low, A.F.; Sezer, M.; Hausenloy, D.J. Invasive Assessment of the Coronary Microcirculation in Reperfused ST-Segment–Elevation Myocardial Infarction Patients. Circ. Cardiovasc. Interv. 2017, 10, e004373. [Google Scholar] [CrossRef]
- de Waha, S.; Patel, M.R.; Granger, C.B.; Ohman, E.M.; Maehara, A.; Eitel, I.; Ben-Yehuda, O.; Jenkins, P.; Thiele, H.; Stone, G.W. Relationship between microvascular obstruction and adverse events following primary percutaneous coronary intervention for ST-segment elevation myocardial infarction: An individual patient data pooled analysis from seven randomized trials. Eur. Heart J. 2017, 38, 3502–3510. [Google Scholar] [CrossRef] [PubMed]
- Bolognese, L.; Carrabba, N.; Parodi, G.; Santoro, G.M.; Buonamici, P.; Cerisano, G.; Antoniucci, D. Impact of Microvascular Dysfunction on Left Ventricular Remodeling and Long-Term Clinical Outcome After Primary Coronary Angioplasty for Acute Myocardial Infarction. Circulation 2004, 109, 1121–1126. [Google Scholar] [CrossRef] [PubMed]
- Dhawan, S.S.; Corban, M.T.; Nanjundappa, R.A.; Eshtehardi, P.; McDaniel, M.C.; Kwarteng, C.A.; Samady, H. Coronary microvascular dysfunction is associated with higher frequency of thin-cap fibroatheroma. Atherosclerosis 2012, 223, 384–388. [Google Scholar] [CrossRef] [PubMed]
- Borlotti, A.; Jerosch-Herold, M.; Liu, D.; Viliani, D.; Bracco, A.; Alkhalil, M.; De Maria, G.L.; Channon, K.M.; Banning, A.P.; Choudhury, R.P.; et al. Acute Microvascular Impairment Post-Reperfused STEMI Is Reversible and Has Additional Clinical Predictive Value: A CMR OxAMI Study. JACC Cardiovasc. Imaging 2019, 12, 1783–1793. [Google Scholar] [CrossRef] [PubMed]
- Galiuto, L.; Lombardo, A.; Maseri, A.; Santoro, L.; Porto, I.; Cianflone, D.; Rebuzzi, A.G.; Crea, F. Temporal evolution and functional outcome of no reflow: Sustained and spontaneously reversible patterns following successful coronary recanalisation. Heart 2003, 89, 731–737. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lerman, A.; Holmes, D.R.; Herrmann, J.; Gersh, B.J. Microcirculatory dysfunction in ST-elevation myocardial infarction: Cause, consequence, or both? Eur. Heart J. 2007, 28, 788–797. [Google Scholar] [CrossRef] [Green Version]
- Stokes, K.Y.; Granger, D.N. The microcirculation: A motor for the systemic inflammatory response and large vessel disease induced by hypercholesterolaemia? J. Physiol. 2005, 562, 647–653. [Google Scholar] [CrossRef]
- Siasos, G.; Sara, J.D.; Zaromytidou, M.; Park, K.H.; Coskun, A.U.; Lerman, L.O.; Oikonomou, E.; Maynard, C.C.; Fotiadis, D.; Stefanou, K.; et al. Local Low Shear Stress and Endothelial Dysfunction in Patients With Nonobstructive Coronary Atherosclerosis. J. Am. Coll. Cardiol. 2018, 71, 2092–2102. [Google Scholar] [CrossRef]
- Samady, H.; Eshtehardi, P.; McDaniel, M.C.; Suo, J.; Dhawan, S.S.; Maynard, C.; Timmins, L.H.; Quyyumi, A.A.; Giddens, D.P. Coronary Artery Wall Shear Stress Is Associated With Progression and Transformation of Atherosclerotic Plaque and Arterial Remodeling in Patients With Coronary Artery Disease. Circulation 2011, 124, 779–788. [Google Scholar] [CrossRef] [Green Version]
- Siasos, G.; Tsigkou, V.; Zaromytidou, M.; Sara, J.D.; Varshney, A.; Coskun, A.U.; Lerman, A.; Stone, P.H. Role of local coronary blood flow patterns and shear stress on the development of microvascular and epicardial endothelial dysfunction and coronary plaque. Curr. Opin. Cardiol. 2018, 33, 638. [Google Scholar] [CrossRef]
- Usui, E.; Yonetsu, T.; Kanaji, Y.; Hoshino, M.; Yamaguchi, M.; Hada, M.; Fukuda, T.; Sumino, Y.; Ohya, H.; Hamaya, R.; et al. Optical Coherence Tomography–Defined Plaque Vulnerability in Relation to Functional Stenosis Severity and Microvascular Dysfunction. JACC Cardiovasc. Interv. 2018, 11, 2058–2068. [Google Scholar] [CrossRef] [PubMed]
- Ford, T.J.; Corcoran, D.; Berry, C. Stable coronary syndromes: Pathophysiology, diagnostic advances and therapeutic need. Heart 2018, 104, 284–292. [Google Scholar] [CrossRef] [PubMed]
- Neumann, F.-J.; Sousa-Uva, M.; Ahlsson, A.; Alfonso, F.; Banning, A.P.; Benedetto, U.; Byrne, R.A.; Collet, J.-P.; Falk, V.; Head, S.J.; et al. 2018 ESC/EACTS Guidelines on myocardial revascularization. Eur. Heart J. 2019, 40, 87–165. [Google Scholar] [CrossRef] [PubMed]
- Radico, F.; Cicchitti, V.; Zimarino, M.; De Caterina, R. Angina Pectoris and Myocardial Ischemia in the Absence of Obstructive Coronary Artery Disease: Practical Considerations for Diagnostic Tests. JACC Cardiovasc. Interv. 2014, 7, 453–463. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Beltrame, J.F.; Limaye, S.B.; Horowitz, J.D. The Coronary Slow Flow Phenomenon–A New Coronary Microvascular Disorder. Cardiology 2002, 97, 197–202. [Google Scholar] [CrossRef]
- Sharif-Yakan, A.; Divchev, D.; Trautwein, U.; Nienaber, C.A. The coronary slow flow phenomena or “cardiac syndrome Y”: A review. Rev. Vasc. Med. 2014, 2, 118–122. [Google Scholar] [CrossRef]
- Ong, P.; Safdar, B.; Seitz, A.; Hubert, A.; Beltrame, J.F.; Prescott, E. Diagnosis of coronary microvascular dysfunction in the clinic. Cardiovasc. Res. 2020, 116, 841–855. [Google Scholar] [CrossRef]
- Sanati, H.; Kiani, R.; Shakerian, F.; Firouzi, A.; Zahedmehr, A.; Peighambari, M.; Shokrian, L.; Ashrafi, P. Coronary Slow Flow Phenomenon Clinical Findings and Predictors. Res. Cardiovasc Med. 2016, 5, e30296. [Google Scholar] [CrossRef]
- Fineschi, M.; Bravi, A.; Gori, T. The “slow coronary flow” phenomenon: Evidence of preserved coronary flow reserve despite increased resting microvascular resistances. Int. J. Cardiol. 2008, 127, 358–361. [Google Scholar] [CrossRef]
- Beltrame, J.F. Defining the Coronary Slow Flow Phenomenon. Circ. J. 2012, 76, 818–820. [Google Scholar] [CrossRef] [Green Version]
- Gibson, C.M.; Cannon, C.P.; Daley, W.L.; Dodge, J.T.; Alexander, B.; Marble, S.J.; McCabe, C.H.; Raymond, L.; Fortin, T.; Poole, W.K.; et al. TIMI Frame Count: A quantitative method of assessing coronary artery flow. Circulation 1996, 93, 879–888. [Google Scholar] [CrossRef] [PubMed]
- Echavarria-Pinto, M.; Escaned, J.; Macías, E.; Medina, M.; Gonzalo, N.; Petraco, R.; Sen, S.; Jimenez-Quevedo, P.; Hernandez, R.; Mila, R.; et al. Disturbed Coronary Hemodynamics in Vessels With Intermediate Stenoses Evaluated With Fractional Flow Reserve. Circulation 2013, 128, 2557–2566. [Google Scholar] [CrossRef] [PubMed]
- Mejía-Rentería, H.; van der Hoeven, N.; van de Hoef, T.P.; Heemelaar, J.; Ryan, N.; Lerman, A.; van Royen, N.; Escaned, J. Targeting the dominant mechanism of coronary microvascular dysfunction with intracoronary physiology tests. Int. J. Cardiovasc. Imaging 2017, 33, 1041–1059. [Google Scholar] [CrossRef] [PubMed]
- Xaplanteris, P.; Fournier, S.; Keulards, D.C.J.; Adjedj, J.; Ciccarelli, G.; Milkas, A.; Pellicano, M.; Veer, M.V.T.; Barbato, E.; Pijls, N.H.J.; et al. Catheter-Based Measurements of Absolute Coronary Blood Flow and Microvascular Resistance. Circ. Cardiovasc. Interv. 2018, 11, e006194. [Google Scholar] [CrossRef] [PubMed]
- Park, S.-D.; Lee, M.-J.; Woo, S.-I.; Baek, Y.-S.; Shin, S.-H.; Kim, D.-H.; Kwan, J.; Park, K.-S. Epicardial Artery Stenosis with a High Index of Microcirculatory Resistance Is Frequently Functionally Insignificant as Estimated by Fractional Flow Reserve (FFR). Intern. Med. 2016, 55, 97–103. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lee, J.M.; Jung, J.-H.; Hwang, D.; Park, J.; Fan, Y.; Na, S.-H.; Doh, J.-H.; Nam, C.-W.; Shin, E.-S.; Koo, B.-K. Coronary Flow Reserve and Microcirculatory Resistance in Patients With Intermediate Coronary Stenosis. J. Am. Coll. Cardiol. 2016, 67, 1158–1169. [Google Scholar] [CrossRef]
- Ahn, S.G.; Suh, J.; Hung, O.Y.; Lee, H.S.; Bouchi, Y.H.; Zeng, W.; Gandhi, R.; Eshtehardi, P.; Gogas, B.D.; Samady, H. Discordance Between Fractional Flow Reserve and Coronary Flow Reserve: Insights From Intracoronary Imaging and Physiological Assessment. JACC Cardiovasc. Interv. 2017, 10, 999–1007. [Google Scholar] [CrossRef]
- Ford, T.J.; Stanley, B.; Good, R.; Rocchiccioli, P.; McEntegart, M.; Watkins, S.; Eteiba, H.; Shaukat, A.; Lindsay, M.; Robertson, K.; et al. Stratified Medical Therapy Using Invasive Coronary Function Testing in Angina: The CorMicA Trial. J. Am. Coll. Cardiol. 2018, 72, 2841–2855. [Google Scholar] [CrossRef]
- Sen, S.; Asrress, K.N.; Nijjer, S.; Petraco, R.; Malik, I.S.; Foale, R.A.; Mikhail, G.W.; Foin, N.; Broyd, C.; Hadjiloizou, N.; et al. Diagnostic Classification of the Instantaneous Wave-Free Ratio Is Equivalent to Fractional Flow Reserve and Is Not Improved With Adenosine Administration: Results of CLARIFY (Classification Accuracy of Pressure-Only Ratios Against Indices Using Flow Study). J. Am. Coll. Cardiol. 2013, 61, 1409–1420. [Google Scholar] [CrossRef] [Green Version]
- Götberg, M.; Christiansen, E.H.; Gudmundsdottir, I.J.; Sandhall, L.; Danielewicz, M.; Jakobsen, L.; Olsson, S.E.; Öhagen, P.; Olsson, H.; Omerovic, E.; et al. Instantaneous Wave-free Ratio versus Fractional Flow Reserve to Guide PCI. N. Engl. J. Med. 2017, 376, 1813–1823. [Google Scholar] [CrossRef] [Green Version]
- Davies, J.E.; Sen, S.; Dehbi, H.M.; Al-Lamee, R.; Petraco, R.; Nijjer, S.S.; Bhindi, R.; Lehman, S.J.; Walters, D.; Sapontis, J.; et al. Use of the Instantaneous Wave-free Ratio or Fractional Flow Reserve in PCI. N. Engl. J. Med. 2017, 376, 1824–1834. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vegsundvåg, J.; Holte, E.; Wiseth, R.; Hegbom, K.; Hole, T. Coronary Flow Velocity Reserve in the Three Main Coronary Arteries Assessed with Transthoracic Doppler: A Comparative Study with Quantitative Coronary Angiography. J. Am. Soc. Echocardiogr. 2011, 24, 758–767. [Google Scholar] [CrossRef] [PubMed]
- Gan, L.M.; Svedlund, S.; Wittfeldt, A.; Eklund, C.; Gao, S.; Matejka, G.; Jeppsson, A.; Albertsson, P.; Omerovic, E.; Lerman, A. Incremental Value of Transthoracic Doppler Echocardiography-Assessed Coronary Flow Reserve in Patients With Suspected Myocardial Ischemia Undergoing Myocardial Perfusion Scintigraphy. J. Am. Heart Assoc. 2017, 6, e004875. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cortigiani, L.; Rigo, F.; Gherardi, S.; Sicari, R.; Galderisi, M.; Bovenzi, F.; Picano, E. Additional prognostic value of coronary flow reserve in diabetic and nondiabetic patients with negative dipyridamole stress echocardiography by wall motion criteria. J. Am. Coll. Cardiol. 2007, 50, 1354–1361. [Google Scholar] [CrossRef] [Green Version]
- Dilsizian, V.; Bacharach, S.L.; Beanlands, R.S.; Bergmann, S.R.; Delbeke, D.; Dorbala, S.; Gropler, R.J.; Knuuti, J.; Schelbert, H.R.; Travin, M.I. ASNC imaging guidelines/SNMMI procedure standard for positron emission tomography (PET) nuclear cardiology procedures. J. Nucl. Cardiol. Off. Publ. Am. Soc. Nucl. Cardiol. 2016, 23, 1187–1226. [Google Scholar] [CrossRef] [Green Version]
- Lanza, G.A.; Camici, P.G.; Galiuto, L.; Niccoli, G.; Pizzi, C.; Di Monaco, A.; Sestito, A.; Novo, S.; Piscione, F.; Tritto, I.; et al. Methods to investigate coronary microvascular function in clinical practice. J. Cardiovasc. Med. 2013, 14, 1–18. [Google Scholar] [CrossRef]
- Feher, A.; Sinusas, A.J. Quantitative Assessment of Coronary Microvascular Function: Dynamic Single-Photon Emission Computed Tomography, Positron Emission Tomography, Ultrasound, Computed Tomography, and Magnetic Resonance Imaging. Circ. Cardiovasc. Imaging 2017, 10. [Google Scholar] [CrossRef] [Green Version]
- Bravo, P.E.; Di Carli, M.F.; Dorbala, S. Role of PET to evaluate coronary microvascular dysfunction in non-ischemic cardiomyopathies. Heart Fail. Rev. 2017, 22, 455–464. [Google Scholar] [CrossRef]
- Schindler, T.H.; Schelbert, H.R.; Quercioli, A.; Dilsizian, V. Cardiac PET Imaging for the Detection and Monitoring of Coronary Artery Disease and Microvascular Health. JACC Cardiovasc. Imaging 2010, 3, 623–640. [Google Scholar] [CrossRef]
- Liu, A.; Wijesurendra, R.S.; Liu, J.M.; Forfar, J.C.; Channon, K.M.; Jerosch-Herold, M.; Piechnik, S.K.; Neubauer, S.; Kharbanda, R.K.; Ferreira, V.M. Diagnosis of Microvascular Angina Using Cardiac Magnetic Resonance. J. Am. Coll Cardiol 2018, 71, 969–979. [Google Scholar] [CrossRef]
- Liu, A.; Wijesurendra, R.S.; Liu, J.M.; Greiser, A.; Jerosch-Herold, M.; Forfar, J.C.; Channon, K.M.; Piechnik, S.K.; Neubauer, S.; Kharbanda, R.K.; et al. Gadolinium-Free Cardiac MR Stress T1-Mapping to Distinguish Epicardial From Microvascular Coronary Disease. J. Am. Coll Cardiol 2018, 71, 957–968. [Google Scholar] [CrossRef] [PubMed]
- Indorkar, R.; Kwong, R.Y.; Romano, S.; White, B.E.; Chia, R.C.; Trybula, M.; Evans, K.; Shenoy, C.; Farzaneh-Far, A. Global Coronary Flow Reserve Measured During Stress Cardiac Magnetic Resonance Imaging Is an Independent Predictor of Adverse Cardiovascular Events. JACC Cardiovasc. Imaging 2019, 12, 1686–1695. [Google Scholar] [CrossRef] [PubMed]
- Doltra, A.; Schneeweis, C.; Fleck, E.; Kelle, S. Cardiac magnetic resonance for prognostic assessment: Present applications and future directions. Expert Rev. Cardiovasc. 2014, 12, 771–782. [Google Scholar] [CrossRef] [PubMed]
- Doyle, M.; Weinberg, N.; Pohost, G.M.; Bairey Merz, C.N.; Shaw, L.J.; Sopko, G.; Fuisz, A.; Rogers, W.J.; Walsh, E.G.; Johnson, B.D.; et al. Prognostic value of global MR myocardial perfusion imaging in women with suspected myocardial ischemia and no obstructive coronary disease: Results from the NHLBI-sponsored WISE (Women’s Ischemia Syndrome Evaluation) study. JACC Cardiovasc. Imaging 2010, 3, 1030–1036. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Branch, K.R.; Haley, R.D.; Bittencourt, M.S.; Patel, A.R.; Hulten, E.; Blankstein, R. Myocardial computed tomography perfusion. Cardiovasc. Diagn. Ther. 2017, 7, 452–462. [Google Scholar] [CrossRef] [Green Version]
- Danad, I.; Szymonifka, J.; Schulman-Marcus, J.; Min, J.K. Static and dynamic assessment of myocardial perfusion by computed tomography. Eur. Heart J. Cardiovasc. Imaging 2016, 17, 836–844. [Google Scholar] [CrossRef] [Green Version]
- Bechsgaard, D.F.; Gustafsson, I.; Michelsen, M.M.; Mygind, N.D.; Raft, K.F.; Linde, J.J.; Kofoed, K.F.; Lin, F.Y.; Min, J.K.; Prescott, E.; et al. Evaluation of computed tomography myocardial perfusion in women with angina and no obstructive coronary artery disease. Int J. Cardiovasc. Imaging 2020, 36, 367–382. [Google Scholar] [CrossRef]
- Piepoli, M.F.; Hoes, A.W.; Agewall, S.; Albus, C.; Brotons, C.; Catapano, A.L.; Cooney, M.-T.; Corrà, U.; Cosyns, B.; Deaton, C.; et al. 2016 European Guidelines on cardiovascular disease prevention in clinical practice. The Sixth Joint Task Force of the European Society of Cardiology and Other Societies on Cardiovascular Disease Prevention in Clinical Practice. Eur. Heart J. 2016, 37, 2315–2381. [Google Scholar] [CrossRef]
- Bairey Merz, C.N.; Pepine, C.J.; Shimokawa, H.; Berry, C. Treatment of coronary microvascular dysfunction. Cardiovasc. Res. 2020, 116, 856. [Google Scholar] [CrossRef]
- AlBadri, A.; Bairey Merz, C.N.; Johnson, B.D.; Wei, J.; Mehta, P.K.; Cook-Wiens, G.; Reis, S.E.; Kelsey, S.F.; Bittner, V.; Sopko, G.; et al. Impact of Abnormal Coronary Reactivity on Long-Term Clinical Outcomes in Women. J. Am. Coll. Cardiol. 2019, 73, 684–693. [Google Scholar] [CrossRef]
- Marinescu, M.A.; Löffler, A.I.; Ouellette, M.; Smith, L.; Kramer, C.M.; Bourque, J.M. Coronary Microvascular Dysfunction, Microvascular Angina, and Treatment Strategies. JACC Cardiovasc. Imaging 2015, 8, 210–220. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Suhrs, H.E.; Michelsen, M.M.; Prescott, E. Treatment strategies in coronary microvascular dysfunction: A systematic review of interventional studies. Microcirculation 2019, 26, e12430. [Google Scholar] [CrossRef] [PubMed]
- Crea, F.; Lanza, G.A. Treatment of microvascular angina: The need for precision medicine. Eur. Heart J. 2016, 37, 1514–1516. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bairey Merz, C.N.; Handberg, E.M.; Shufelt, C.L.; Mehta, P.K.; Minissian, M.B.; Wei, J.; Thomson, L.E.J.; Berman, D.S.; Shaw, L.J.; Petersen, J.W.; et al. A randomized, placebo-controlled trial of late Na current inhibition (ranolazine) in coronary microvascular dysfunction (CMD): Impact on angina and myocardial perfusion reserve. Eur. Heart J. 2016, 37, 1504–1513. [Google Scholar] [CrossRef] [PubMed]
- Pauly, D.F.; Johnson, B.D.; Anderson, R.D.; Handberg, E.M.; Smith, K.M.; Cooper-DeHoff, R.M.; Sopko, G.; Sharaf, B.M.; Kelsey, S.F.; Merz, C.N.B.; et al. In women with symptoms of cardiac ischemia, nonobstructive coronary arteries, and microvascular dysfunction, angiotensin-converting enzyme inhibition is associated with improved microvascular function: A double-blind randomized study from the National Heart, Lung and Blood Institute Women’s Ischemia Syndrome Evaluation (WISE). Am. Heart J. 2011, 162, 678–684. [Google Scholar] [CrossRef] [Green Version]
- Villano, A.; Di Franco, A.; Nerla, R.; Sestito, A.; Tarzia, P.; Lamendola, P.; Di Monaco, A.; Sarullo, F.M.; Lanza, G.A.; Crea, F. Effects of Ivabradine and Ranolazine in Patients With Microvascular Angina Pectoris. Am. J. Cardiol. 2013, 112, 8–13. [Google Scholar] [CrossRef]
- Kramer, C.K.; Zinman, B. Sodium–Glucose Cotransporter–2 (SGLT-2) Inhibitors and the Treatment of Type 2 Diabetes. Annu. Rev. Med. 2019, 70, 323–334. [Google Scholar] [CrossRef] [PubMed]
- Lucas, A.R.; Korol, R.; Pepine, C.J. Inflammation in Atherosclerosis. Some thoughts about acute coronary syndromes. Circulation 2006, 113, e728–e732. [Google Scholar] [CrossRef] [Green Version]
- Ikonomidis, I.; Lekakis, J.P.; Nikolaou, M.; Paraskevaidis, I.; Andreadou, I.; Kaplanoglou, T.; Katsimbri, P.; Skarantavos, G.; Soucacos, P.N.; Kremastinos, D.T. Inhibition of Interleukin-1 by Anakinra Improves Vascular and Left Ventricular Function in Patients With Rheumatoid Arthritis. Circulation 2008, 117, 2662–2669. [Google Scholar] [CrossRef]
- Nidorf, S.M.; Thompson, P.L. Why Colchicine Should Be Considered for Secondary Prevention of Atherosclerosis: An Overview. Clin. Ther. 2019, 41, 41–48. [Google Scholar] [CrossRef] [Green Version]
- Verheye, S.; Jolicœur, E.M.; Behan, M.W.; Pettersson, T.; Sainsbury, P.; Hill, J.; Vrolix, M.; Agostoni, P.; Engstrom, T.; Labinaz, M.; et al. Efficacy of a Device to Narrow the Coronary Sinus in Refractory Angina. N. Engl. J. Med. 2015, 372, 519–527. [Google Scholar] [CrossRef] [PubMed]


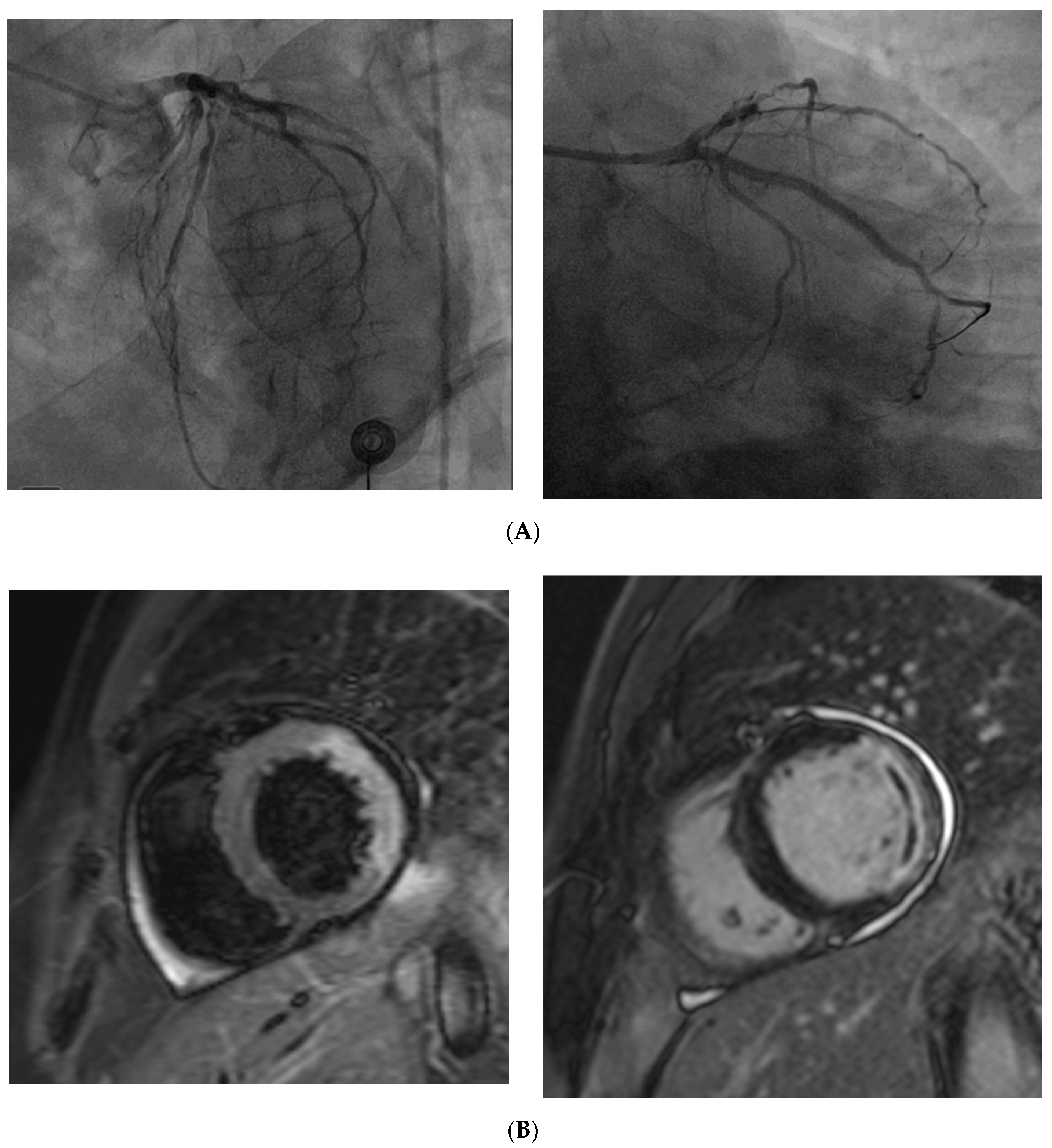

| 1. Symptoms of myocardial ischemia |
| -Effort and/or rest angina |
| -Angina equivalents (exertional dyspnea) |
| 2. Absence of obstructive CAD (<50% diameter reduction or FFR > 0.80) by coronary CTA or invasive angiography |
| 3. Objective evidence of myocardial ischemia |
| -Ischemic ECG changes during chest pain |
| -Stress-induced chest pain and/or ischemic ECG changes with or without transient |
| or reversible abnormal myocardial perfusion and/or wall motion abnormality |
| 4. Evidence of impaired coronary microvascular function |
| -Impaired coronary flow reserve ≤ 2.0 |
| -Coronary microvascular spasm, defined as symptoms and ischemic ECG changes but not epicardial spasm during acetylcholine testing |
| -Abnormal coronary microvascular resistance indices (IMR > 25) |
| -Coronary slow flow phenomenon, defined as TIMI frame count > 25 |
| A. CMD in chronic coronary syndrome |
| with non-obstructive chronic coronary syndrome |
| with obstructive chronic coronary syndrome |
| CMD in acute coronary syndrome (ACS) |
| with non-obstructive ACS |
| with obstructive ACS |
| with coronary no-reflow phenomenon |
| CMD following successful revascularization after MI |
| B. Group 1. CMD in the absence of obstructive CAD and myocardial disease |
| Group 2. CMD in the presence of myocardial disease |
| Group 3. CMD in the presence of obstructive CAD |
| Group 4. CMD after successful percutaneous coronary intervention |
| C. CMD without atherosclerosis |
| CMD with non-obstructive atherosclerosis |
| CMD with obstructive atherosclerosis |
| Clinical Feature | Dysfunction | Diagnostic Criteria |
|---|---|---|
| Microvascular angina | Impaired coronary vasodilatation | CFR < 2.0 |
| Increased microvascular resistance | IMR ≥ 25 units | |
| Microvascular spasm | angina symptoms after intracoronary Ach Ischemic ECG changes <90% diameter reduction | |
| Vasospastic angina | Epicardial spasm | Angina symptoms after intracoronary Ach Infusion and Ischemic ECG changes with >90% epicardial coronary constriction |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vancheri, F.; Longo, G.; Vancheri, S.; Henein, M. Coronary Microvascular Dysfunction. J. Clin. Med. 2020, 9, 2880. https://doi.org/10.3390/jcm9092880
Vancheri F, Longo G, Vancheri S, Henein M. Coronary Microvascular Dysfunction. Journal of Clinical Medicine. 2020; 9(9):2880. https://doi.org/10.3390/jcm9092880
Chicago/Turabian StyleVancheri, Federico, Giovanni Longo, Sergio Vancheri, and Michael Henein. 2020. "Coronary Microvascular Dysfunction" Journal of Clinical Medicine 9, no. 9: 2880. https://doi.org/10.3390/jcm9092880




