Inorganic Phosphate Solubilization by a Novel Isolated Bacterial Strain Enterobacter sp. ITCB-09 and Its Application Potential as Biofertilizer
Abstract
:1. Introduction
2. Materials and Methods
2.1. Isolation of Inorganic Phosphate Solubilizing Bacteria from Agroecosystems Soils
2.2. Determination of Phosphate-Solubilizing Activity and Selection of Strain ITCB-09
2.3. 16S rRNA Gene Sequence Analysis
2.4. Detection of Phosphate Solubilization Mechanisms
2.4.1. Siderophores
2.4.2. Phosphatase Activity
2.4.3. Production and Characterization of Extracellular Polymeric Substances (EPS)
2.5. Effect of Enterobacter sp. ITCB-09 on Habanero Pepper (Capsicum chinense Jacq.) Seedling Growth
2.5.1. Cultivation of Enterobacter sp. ITCB-09 for Inoculation
2.5.2. Preparation of Substrates for Habanero Pepper (Capsicum chinense Jacq.) Cultivation
2.5.3. Inoculation and Cultivation of Habanero Pepper
2.6. Data Analysis
3. Results
3.1. Isolation of Inorganic Phosphate Solubilizing Bacteria
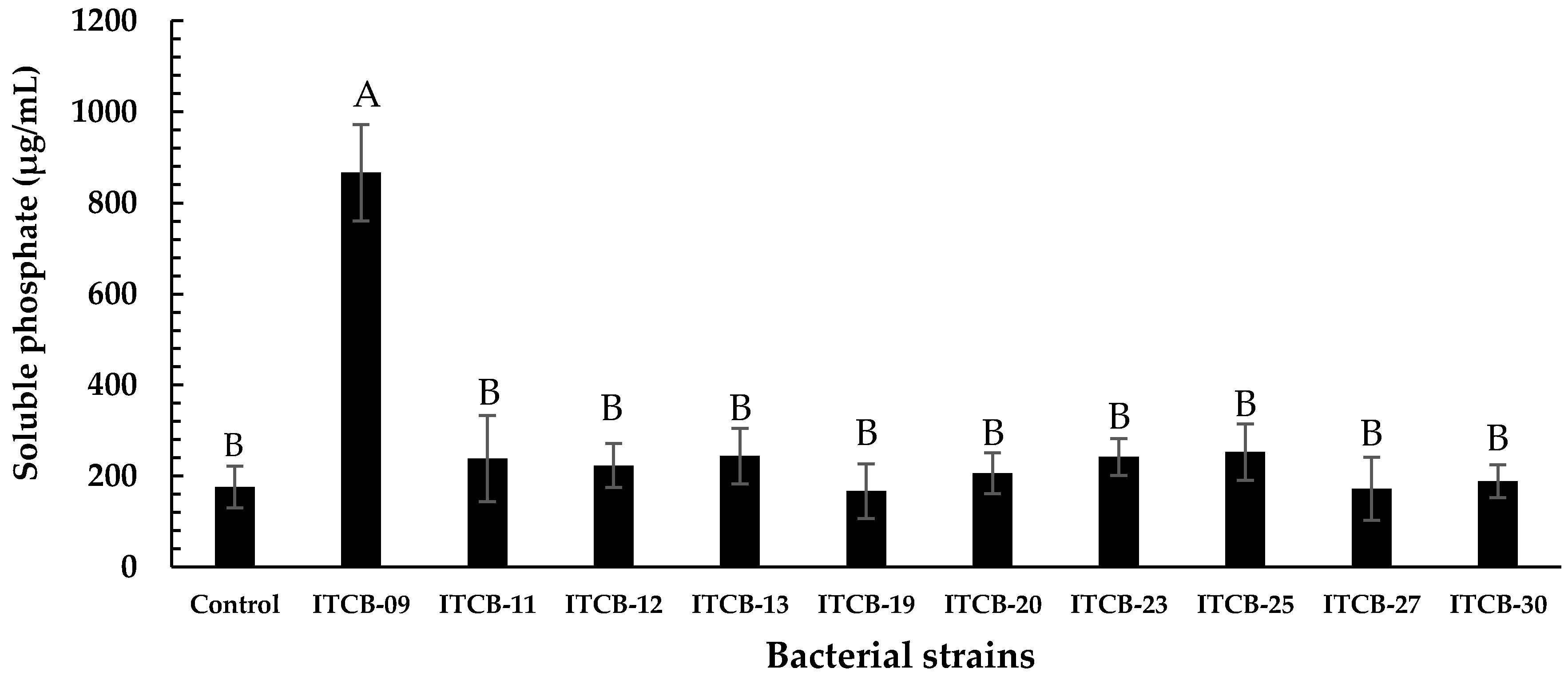
3.2. Determination of Phosphate-Solubilizing Ability and Selection of the Strain ITCB-09
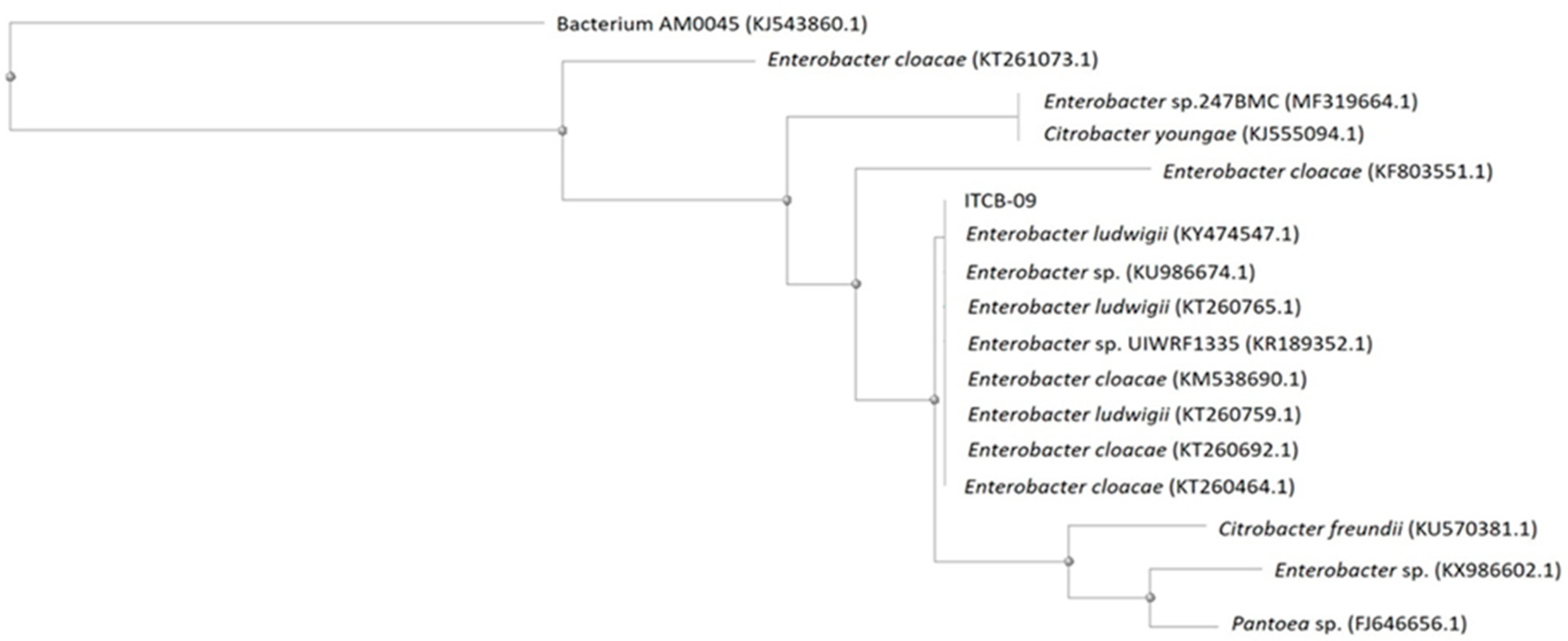
3.3. Identification of Bacterial Strain ITCB-09
3.4. Mechanisms of Phosphate Solubilization of Enterobacter sp. ITCB-09
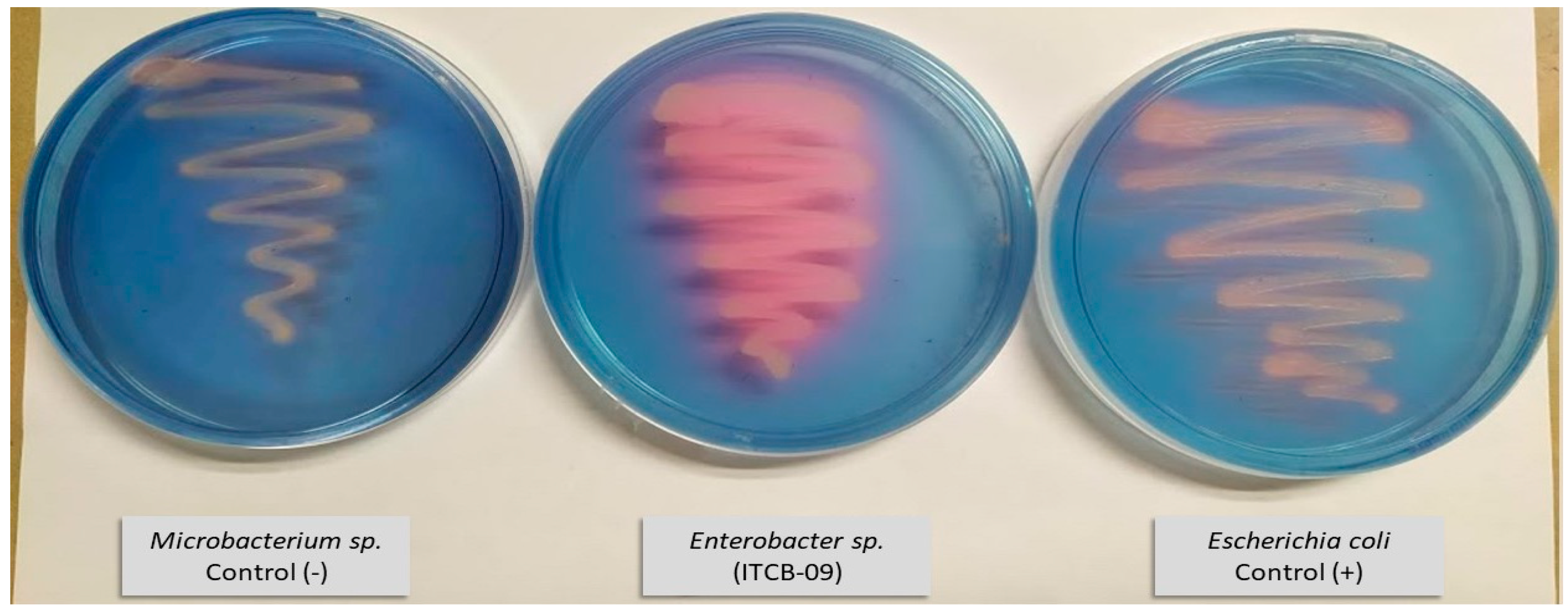
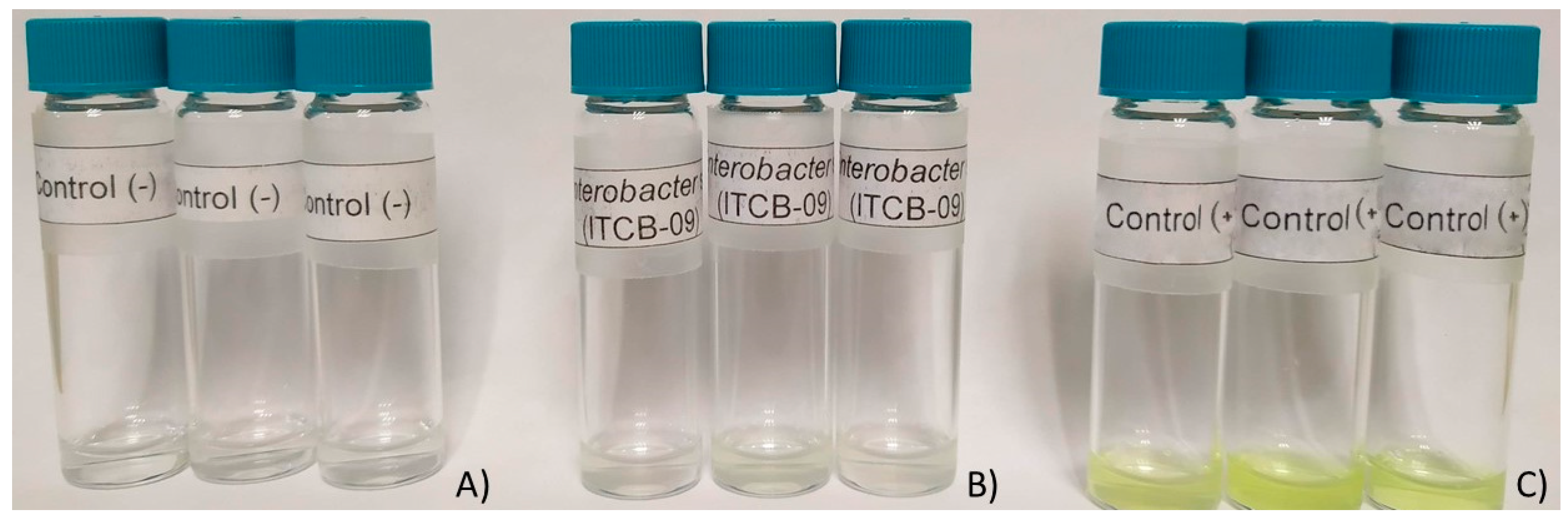
3.4.1. Siderophores
3.4.2. Phosphatase Activity
3.4.3. EPS
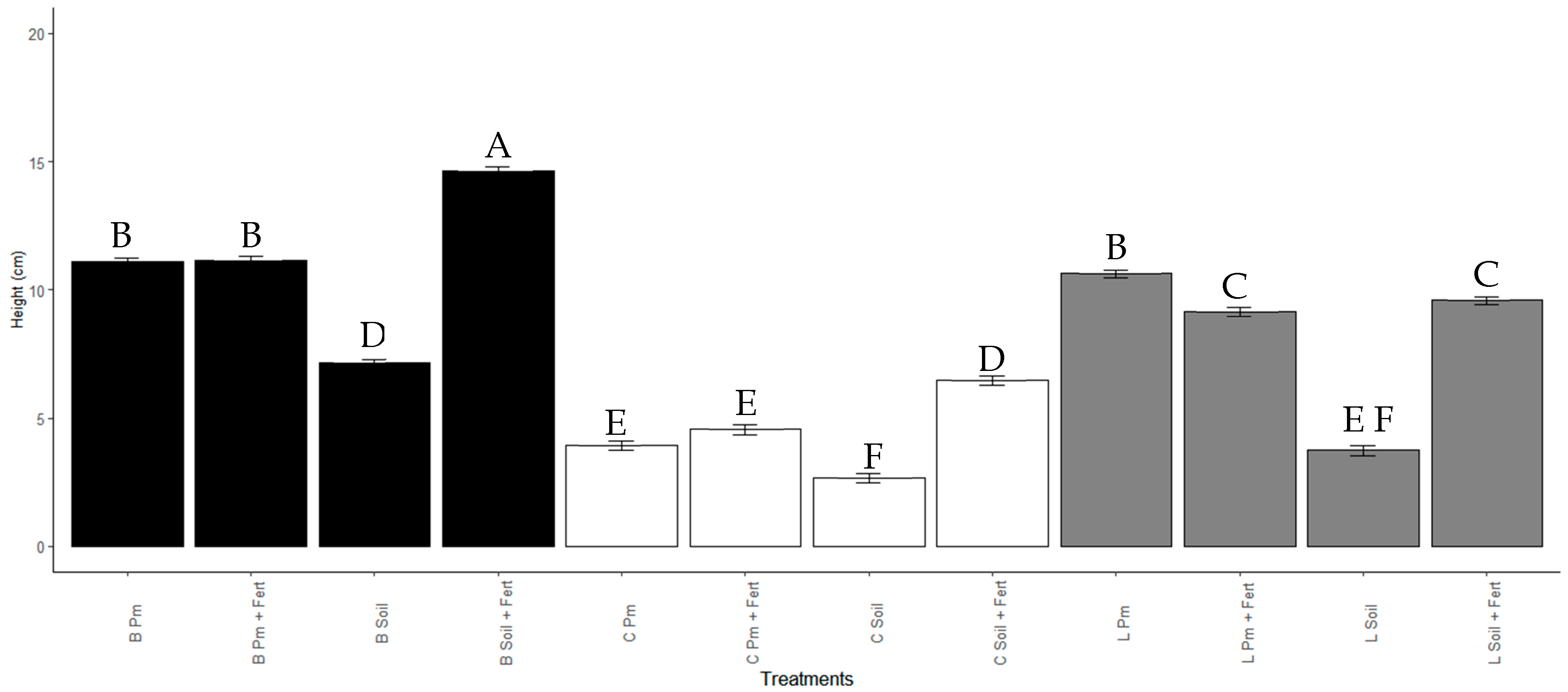
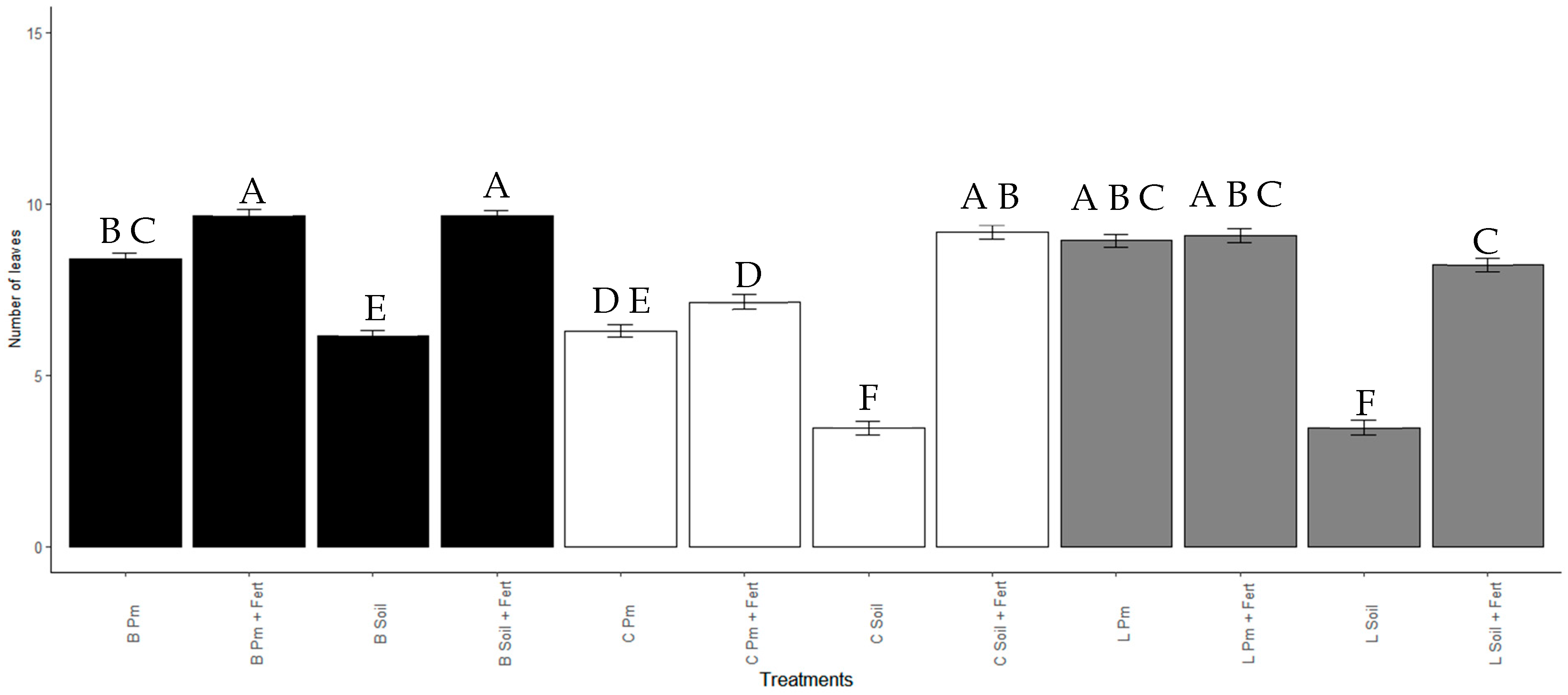
3.5. Effect of Enterobacter sp. ITCB-09 on the Growth of Habanero Pepper (Capsicum chinense Jacq.) Seedlings
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Li, Y.; Liu, X.; Hao, T.; Chen, S. Colonization and maize growth promotion induced by phosphate solubilizing bacterial isolates. Int. J. Mol. Sci. 2017, 18, 1253. [Google Scholar]
- Mpanga, I.K.; Nkebiwe, P.M.; Kuhkmann, M.; Cozzolino, V.; Piccolo, A.; Geistlinger, J.; Berger, N.; Ludewig, U.; Neumann, G. The form of N supply determines plant growth promotion by P-solubilizing microorganisms in maize. Microorganisms 2019, 7, 38. [Google Scholar] [CrossRef] [Green Version]
- Khan, S.; Zaidi, A.; Ahmad, E. Mechanism of phosphate solubilization and physiological functions of phosphate-solubilizing microorganisms. In Phosphate Solubilizing Microorganisms. Principles and Application of Microphos Technology, 1st ed.; Khan, S., Zaidi, A., Musarrat, J., Eds.; Springer: Berlin/Heidelberg, Germany, 2014; pp. 31–62. [Google Scholar]
- Goldstein, A.H. Recent progress in understanding the molecular genetics and biochemistry of calcium phosphate solubilization by gram negative bacteria. Biol. Agric. Hortic 1995, 12, 185–193. [Google Scholar] [CrossRef]
- Kim, K.Y.; McDonald, G.A.; Jordan, D. Solubilization of hydroxyapatite by Enterobacter agglomerans and cloned Escherichia coli in culture medium. Biol. Fertil. Soils 1997, 24, 347–352. [Google Scholar] [CrossRef]
- Zhu, J.; Li, M.; Whelan, M. Phosphorus activators contribute to legacy phosphorus availability in agricultural soils: A review. Sci. Total Environ. 2018, 612, 522–537. [Google Scholar] [CrossRef] [Green Version]
- Oliveira-Rita, J.; Gama-Rodrigues, A.; Gama-Rodrigues, E.; Costa-Zaia, F.; Duarte-Nunes, D. Mineralization of organic phosphorus in soil size fractions under different vegetation covers in the north of Rio de Janeiro. Rev. Bras. Cienc Solo 2013, 37, 1207–1215. [Google Scholar] [CrossRef] [Green Version]
- Oliveira, C.; Alves, V.; Marriel, I.; Gomes, E.; Scotti, M.; Carneiro, N.; Sá, N. Phosphate solubilizing microorganisms isolated from rhizosphere of maize cultivated in an oxisol of the Brazilian Cerrado Biome. Soil Biol. Biochem. 2009, 41, 1782–1787. [Google Scholar] [CrossRef]
- Aseri, G.K.; Jain, N.; Tarafdar, J.C. Hydrolysis of organic phosphate forms by phosphatases and phytase producing fungi of arid and semi-arid soils of India. Am.-Eurasian JAES 2009, 5, 564–570. [Google Scholar]
- Maougal, R.T.; Brauman, A.; Plassard, C.; Abadie, J.; Djekoun, A.; Drevon, J.J. Bacterial capacities to mineralize phytate increase in the rhizosphere of nodulated common bean (Phaseolus vulgaris) under P deficiency. Eur. J. Soil Biol. 2014, 62, 8–14. [Google Scholar] [CrossRef]
- Sharma, S.B.; Sayyed, R.Z.; Trivedi, M.H.; Gobi, T.A. Phosphate solubilizing microbes: Sustainable approach for managing phosphorus deficiency in agricultural soils. SpringerPlus 2013, 2, 587. [Google Scholar] [CrossRef] [Green Version]
- Flemming, H.C.; Wingender, J. Relevance of microbial extracellular polymeric substances (EPSs)—Part I: Structural and ecological aspects. Water Sci. Technol. 2001, 43, 1–8. [Google Scholar] [CrossRef]
- Costa, O.; Raaijmakers, J.M.; Kuramae, E.E. Microbial extracellular polymeric substances: Ecological function and impact on soil aggregation. Front. Microbiol. 2018, 9, 1636. [Google Scholar] [CrossRef] [Green Version]
- Yi, Y.; Huang, W.; Ge, Y. Exopolysaccharide: A novel important factor in the microbial dissolution of tricalcium phosphate. World J. Microbiol. Biotechnol. 2008, 24, 1059–1065. [Google Scholar] [CrossRef]
- Chaudhary, I.; Neeraj, A.; Siddiqui, M.; Singh, V. Nutrient Management Technologies and the Role of Organic Matrix-Based Slow-Release Biofertilizers for Agricultural Sustainability: A Review. Agric. Rev. 2020, 41, 1–13. [Google Scholar] [CrossRef] [Green Version]
- Glick, B. Plant growth-promoting bacteria: Mechanisms and applications. Scientifica 2012, 2012, 963401. [Google Scholar] [CrossRef] [Green Version]
- Sarikhani, M.; Aliasgharzad, N.; Khoshru, B. P Solubilizing Potential of Some Plant Growth Promoting Bacteria Used as Ingredient in Phosphatic Biofertilizers with Emphasis on Growth Promotion of Zea mays L. Geomicrobiol. J. 2020, 37, 327–335. [Google Scholar] [CrossRef]
- Ribaudo, C.; Zaballa, J.; Golluscio, R. Effect of the Phosphorus-Solubilizing Bacterium Enterobacter ludwigii on Barley Growth Promotion. Am. Sci. Res. J. Eng. Technol. Sci. 2020, 63, 144–157. [Google Scholar]
- Lee, K.; Adhikari, A.; Kang, S.; You, Y.; Joo, G.; Kim, J.; Lee, I. Isolation and characterization of the high silicate and phosphate solubilizing novel strain Enterobacter ludwigii GAK2 that promotes growth in rice plants. Agronomy 2019, 9, 144. [Google Scholar] [CrossRef] [Green Version]
- Teng, Z.; Chen, Z.; Zhang, Q.; Yao, Y.; Song, M.; Li, M. Isolation and characterization of phosphate solubilizing bacteria from rhizosphere soils of the Yeyahu Wetland in Beijing, China. Environ. Sci. Pollut. Res. 2019, 26, 33976–33987. [Google Scholar] [CrossRef]
- Widdig, M.; Schleuss, P.; Weig, A.; Guhr, A.; Biederman, L.; Borer, E.; Spohn, M. Nitrogen and phosphorus additions alter the abundance of phosphorus-solubilizing bacteria and phosphatase activity in grassland soils. Front. Environ. Sci. 2019, 7, 185–200. [Google Scholar] [CrossRef] [Green Version]
- Bendaha, M.E.; Belaouni, H. Effect of the endophytic plant growth promoting Enterobacter ludwigii EB4B on tomato growth. Hell. Plant. Prot. J. 2020, 13, 54–65. [Google Scholar] [CrossRef]
- Vyas, R.; Panpatte, D.; Jhala, Y.; Shelat, H. Wonders of microbes in agriculture for productivity and sustainability. In Microorganisms for Green Revolution, 1st ed.; Kumar, N., Ed.; Springer: Berlin/Heidelberg, Germany, 2017; Volume 1, pp. 1–24. [Google Scholar]
- Keneni, A.; Assefa, F.; Prabu, P.C. Isolation of phosphate solubilizing bacteria from the rhizosphere of faba bean of Ethiopia and their abilities on solubilizing insoluble phosphates. J. Agric. Sci. Technol. 2010, 12, 79–89. [Google Scholar]
- Singh, N.K.; Patel, D.B.; Chaudhari, S.R.; Morad, B.G.; Rabari, S.M. Characterization of phosphate-solubilizing isolates of Bacillus from cumin rhizosphere. IJTA 2016, 34, 1469–1480. [Google Scholar]
- National Center for Biotechnology Information. Available online: http://www.ncbi.nlm.nih.gov (accessed on 1 June 2020).
- Tamura, K.; Peterson, D.; Peterson, N.; Stecher, G.; Nei, M.; Kumar, S. MEGA 5: Molecular evolutionary genetics analysis using maximun likelihood, evolutionary distance, and maximum parsimony methods. Mol. Biol. Evol. 2011, 28, 2731–2739. [Google Scholar] [CrossRef] [Green Version]
- Louden, B.C.; Haarmann, D.; Lynne, A.M. Use of blue agar CAS assay for siderophore detection. J. Microbiol. Biol. Educ. 2011, 12, 51–53. [Google Scholar] [CrossRef] [Green Version]
- Shin, S.H.; Lim, Y.; Lee, S.E.; Yang, N.W.; Rhee, J.H. CAS agar diffusion assay for the measurement of siderophores in biological fluids. J. Microbiol. Meth. 2001, 44, 89–95. [Google Scholar] [CrossRef]
- Schiessl, K.T.; Janssen, E.M.; Kraemer, S.M.; McNeill, K.; Ackermann, M. Magnitude and mechanism of siderophore-mediated competition at low iron solubility in the Pseudomonas aeruginosa pyochelin system. Front. Microbiol. 2017, 8, 1964. [Google Scholar] [CrossRef] [Green Version]
- Dave, B.P.; Anshuman, K.; Hajela, P. Siderophores of halophilic archaea and their chemical characterization. Indian J. Exp. Biol. 2006, 44, 340–344. [Google Scholar]
- Bobadilla, C.; Rincón, S. Aislamiento y Producción de Bacterias Fosfato Solubilizadoras a Partir de Compost Obtenido de Residuos de Plaza. Bachelor’s Thesis, Facultad de Ciencias de la Universidad Javeriana, Bogotá, Colombia, June 2008. [Google Scholar]
- Malo, M.S.; Moaven, O.; Muhammad, N.; Biswas, B.; Alam, S.N.; Economopoulos, K.P.; Mohamed, M.M.R. Intestinal alkaline phosphatase promotes gut bacterial growth by reducing the concentration of luminal nucleotide triphosphates. Am. J. Physiol. Gastrointest. Liver Physiol. 2014, 306, 826–838. [Google Scholar] [CrossRef] [PubMed]
- Yang, K.; Metcalf, W.W. A new activity for an old enzyme: Escherichia coli bacterial alkaline phosphatase is a phosphite-dependent hydrogenase. PNAS 2004, 101, 7919–7924. [Google Scholar] [CrossRef] [Green Version]
- Chattopadhyay, M.K.; Uma-Devi, K.; Gopishankar, Y.; Shivaji, S. Thermolabile alkaline phosphatase from Sphingobacterium antarticus a psychrotrophic bacterium from Antarctica. Polar Biol 1995, 15, 215–219. [Google Scholar] [CrossRef]
- Ortega-Morales, B.O.; Santiago-García, J.L.; Chan-Bacab, M.J.; Moppert, X.; Miranda-Tello, E.; Fardeau, M.L.; Guezennec, J. Characterization of extracellular polymers synthesized by tropical intertidal biofilm bacteria. J. Appl. Microbiol. 2007, 102, 254–264. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Camacho-Chab, J.C.; Guézennec, J.; Chan-Bacab, M.J.; Ríos-Leal, E.; Sinquin, C.; Munñiz-Salazar, R.; Ortega-Morales, B.O. Emulsifying activity and stability of a non-toxic bioemulsifier synthesized by Microbacterium sp. MC3B-10. Int. J. Mol. Sci. 2013, 14, 18959–18972. [Google Scholar] [CrossRef] [Green Version]
- Dubois, M.; Gilles, K.; Hamilton, J.; Rebers, P.; Smith, F. Colorimetric method for determination of sugar and related substances. Anal. Chem. 1956, 15, 350–356. [Google Scholar] [CrossRef]
- Blumenkrantz, N.; Asboe-Hansen, G. New method for quantitative determination of uronic acids. Anal. Biochem. 1973, 54, 484–489. [Google Scholar] [CrossRef]
- Lowry, O.; Rosebrough, H.; Farr, A.; Randall, R. Protein measurement with the folin-phenol reagent. J. Biol. Chem. 1951, 193, 265–275. [Google Scholar]
- Colli-Camal, G. Efecto de la Cepa Solubilizadora de Fosfato ITCB-09 en el Crecimiento de Plántulas de Chile Habanero (Capsicum chinense) var. Jaguar. Bachelor’s Thesis, Instituto Tecnológico de Chiná, Chiná, Mexico, September 2017. [Google Scholar]
- Lee, K.Y.; Mooney, D.J. Alginate: Properties and biomedical applications. Prog. Polym. Sci. 2012, 37, 106–126. [Google Scholar] [CrossRef] [Green Version]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2018; Available online: https://www.r-project.org (accessed on 1 June 2020).
- Fabián, D.; Guadarrama, P.; Hernadez-Cuevas, L.; Ramos-Zapata, J.A. Arbuscular mycorrhizal fungi in a coastal wetland in Yucatan, Mexico. Bot. Sci. 2018, 96, 24–34. [Google Scholar] [CrossRef] [Green Version]
- Saitou, N.; Nei, M. The neighbor-joining method: A new method for reconstructing phylogenetic trees. Mol. Biol. Evol. 1987, 4, 406–425. [Google Scholar]
- Shoebitz, M.; Ribaudo, C.M.; Pardo, M.A.; Cantore, M.L.; Ciampi, L.; Curá, J.A. Plant growth promoting properties of a strain of Enterobacter ludwigii isolated from Lolium perenne rhizosphere. Soil Biol. Biochem. 2009, 41, 1768–1774. [Google Scholar] [CrossRef]
- Hwangbo, H.; Park, R.D.; Kim, Y.W.; Rim, Y.S.; Park, K.H.; Kim, T.H.; Kim, K.Y. 2-Ketogluconic acid production and phosphate solubilization by Enterobacter intermedium. Curr. Microbiol. 2003, 47, 0087–0092. [Google Scholar]
- Vassilev, N.; Toro, M.; Vassileva, M.; Azcon, R.; Barea, J.M. Rock phosphate solubilization by immobilized cells of Enterobacter sp. in fermentation and soil conditions. Bioresour. Technol 1997, 61, 29–32. [Google Scholar] [CrossRef]
- Camacho-Chab, J.C.; Castañeda-Chávez, M.D.R.; Chan-Bacab, M.J.; Aguila-Ramírez, R.N.; Galaviz-Villa, I.; Bartolo-Pérez, P.; Ortega-Morales, B.O. Biosorption of cadmium by non-toxic extracellular polymeric substances (EPS) synthesized by bacteria from marine intertidal biofilms. Int. J. Environ. Res. Public Health 2018, 15, 314. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Behera, B.C.; Yadav, H.; Singh, S.K.; Mishra, R.R.; Sethi, B.K.; Dutta, S.K.; Thatoi, H.N. Phosphate solubilization and acid phosphatase activity of Serratia sp. isolated from mangrove soil of Mahanadi river delta, Odisha, India. J. Genet. Eng. Biotechnol. 2017, 15, 169–178. [Google Scholar] [CrossRef]
- Cordero-Elvia, J.; Ortega-Rodés, P.; Ortega, E. La inoculación de plantas con Pantoea sp., bacteria solubilizadora de fosfatos, incrementa la concentración de P en los tejidos foliares. Rev. Colomb. Biotecnol. 2008, 10, 111–121. [Google Scholar]
- Nowack, B. Environmental chemistry of phosphonates. Water Res. 2003, 37, 2533–2546. [Google Scholar] [CrossRef]
- Chen, Y.P.; Rekha, P.D.; Arun, A.B.; Shen, F.T.; Lai, W.A.; Young, C.C. Phosphate solubilizing bacteria from subtropical soil and their tricalcium phosphate solubilizing abilities. Appl. Soil Ecol. 2006, 34, 33–41. [Google Scholar] [CrossRef]
- Nannipieri, P.; Giagnoni, L.; Landi, L.; Renella, G. Role of phosphatase enzymes in soil. In Phosphorus in Action. Biological Processes in Soil Phosphorus Cycling, 1st ed.; Büneman, E.K., Oberson, A., Frossard, E., Eds.; Springer: Berlin/Heidelberg, Germany, 2011; pp. 215–243. [Google Scholar]
- Crowley, D. Microbial siderophores in the plan rhizosphere. In Iron Nutrition in Plants and Rhizospheric Microorganisms, 1st ed.; Barton, L., Abadia, J., Eds.; Springer Netherlands: Dordrecht, The Netherlands, 2007; pp. 169–198. [Google Scholar]
- Sasirekha, B.; Srividya, S. Siderophore production by Pseudomonas aeruginosa FP6, a biocontrol strain for Rhizoctonia solani and Colletotrichum gloeosporioides causing diseases in chilli. Agric. Nat. Resour. 2016, 50, 250–256. [Google Scholar] [CrossRef] [Green Version]
- Kumar, P.; Thakur, S.; Dhingra, G.K.; Singh, A.; Kumar-Pal, M.; Harshvardhan, K.; Dubey, R.C.; Maheshwari, D. Inoculation of siderophore producing rhizobacteria and their consortium for growth enhancement of wheat plant. Biocatal. Agric. Biotechnol. 2018, 15, 264–269. [Google Scholar] [CrossRef]
- Park, K.H.; Lee, C.; Son, H. Mechanism of insoluble phosphate solubilization by Pseudomonas fluorescens RAF15 isolated from ginseng rhizosphere and its plan growth-promoting activities. Lett. Appl. Microbiol. 2009, 49, 222–228. [Google Scholar] [CrossRef]
- Mahmood, S.; Daur, I.; Al-Solaimani, S.G.; Ahmad, S.; Madkour, M.H.; Yasir, M.; Hirt, H.; Ali, H.; Ali, Z. Plant growth promoting rhizobacteria and silicon synergistically enhance salinity tolerance of mung bean. Front. Plant Sci. 2016, 7, 876–890. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pan, Q.; Shikano, I.; Hoover, K.; Liu, T.X.; Felton, G.W. Enterobacter ludwigii, isolated from the gut microbiota of Helicoverpa zea, promotes tomato plant growth and yield without compromising anti-herbivore defenses. Arthropod Plant Interact. 2019, 13, 271–278. [Google Scholar] [CrossRef]
- Borham, A.; Belal, E.; Metwaly, M. Phosphate Solubilization by Enterobacter cloacae and Its Impact on Growth and Yield of Wheat Plants. J. Sustain. Agric. Sci. 2017, 43, 89–103. [Google Scholar] [CrossRef] [Green Version]

| Treatments | Substrates | Nomenclature | |||
|---|---|---|---|---|---|
| 1. Control | Soil (leptosol) | Peat moss | Soil 50% + Peat Moss 50% | Peat Moss + Fertilizer (1 g/seed) | 1. C Soil 2. C Pm 3. C Soil + Pm 4. C Pm + Fert |
| 2. Enterobacter sp. ITCB-09 in liquid | Soil (leptosol) | Peat moss | Soil 50% + Peat Moss 50% | Peat Moss + Fertilizer (1 g/seed) | 1. L Soil 2. L Pm 3. L Soil + Pm 4. L Pm + Fert |
| 3. Enterobacter sp. ITCB-09 in beads | Soil (leptosol) | Peat moss | Soil 50% + Peat Moss 50% | Peat Moss + Fertilizer (1 g/seed) | 1. B Soil 2. B Pm 3. B Soil + Pm 4. B Pm + Fert |
| No. | Bacterium Isolated | Cell Morphology | Gram Staining | No. | Bacterium Isolated | Cell Morphology | Gram Staining |
|---|---|---|---|---|---|---|---|
| 1. | ITCB-09 | Rod | - | 12. | ITCB-23 | Rod | - |
| 2. | ITCB-10 | Rod | - | 13. | ITCB-24 | Rod | - |
| 3. | ITCB-11 | Rod | - | 14. | ITCB-25 | Rod | - |
| 4. | ITCB-12 | Rod | - | 15. | ITCB-26 | Rod | - |
| 5. | ITCB-13 | Rod | - | 16. | ITCB-27 | Rod | - |
| 6. | ITCB-17 | Coccus | - | 17. | ITCB-28 | Rod | - |
| 7. | ITCB-18 | Rod | - | 18. | ITCB-29 | Rod | - |
| 8. | ITCB-19 | Rod | - | 19. | ITCB-30 | Rod | - |
| 9. | ITCB-20 | Rod | - | 20. | ITCB-31 | Rod | - |
| 10. | ITCB-21 | Rod | + | 21. | ITCB-32 | Rod | - |
| 11. | ITCB-22 | Rod | - |
| Test Solutions | Absorbance |
|---|---|
| FeCl3 + FS | 2.568 |
| FS + metanol | 0.742 |
| FeCl3 + metanol | 0.469 |
| Sugars Total | Hexuronic Acids | Proteins |
|---|---|---|
| 42 ± 1.8% | 11 ± 1% | 36 ± 1.3% |
| Treatments/Substrate | Germination Percentage |
|---|---|
| Control/Soil | 75% |
| Control/Pm | 87.5% |
| Control/Soil + Pm | 82.5% |
| Control/Pm + Fert | 70% |
| Enterobacter sp. ITCB-09 in liquid/Soil | 65% |
| Enterobacter sp. ITCB-09 in liquid/Pm | 100% |
| Enterobacter sp. ITCB-09 in liquid/Soil + Pm | 92.5% |
| Enterobacter sp. ITCB-09 in liquid/Pm + Fert | 80% |
| Enterobacter sp. ITCB-09 in beads/Soil | 100% |
| Enterobacter sp. ITCB-09 in beads/Pm | 100% |
| Enterobacter sp. ITCB-09 in beads/Soil + Pm | 97.5% |
| Enterobacter sp. ITCB-09 in beads/Pm + Fert | 82.5% |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mendoza-Arroyo, G.E.; Chan-Bacab, M.J.; Aguila-Ramírez, R.N.; Ortega-Morales, B.O.; Canché Solís, R.E.; Chab-Ruiz, A.O.; Cob-Rivera, K.I.; Dzib-Castillo, B.; Tun-Che, R.E.; Camacho-Chab, J.C. Inorganic Phosphate Solubilization by a Novel Isolated Bacterial Strain Enterobacter sp. ITCB-09 and Its Application Potential as Biofertilizer. Agriculture 2020, 10, 383. https://doi.org/10.3390/agriculture10090383
Mendoza-Arroyo GE, Chan-Bacab MJ, Aguila-Ramírez RN, Ortega-Morales BO, Canché Solís RE, Chab-Ruiz AO, Cob-Rivera KI, Dzib-Castillo B, Tun-Che RE, Camacho-Chab JC. Inorganic Phosphate Solubilization by a Novel Isolated Bacterial Strain Enterobacter sp. ITCB-09 and Its Application Potential as Biofertilizer. Agriculture. 2020; 10(9):383. https://doi.org/10.3390/agriculture10090383
Chicago/Turabian StyleMendoza-Arroyo, Gustavo Enrique, Manuel Jesús Chan-Bacab, Ruth Noemi Aguila-Ramírez, Benjamín Otto Ortega-Morales, René Efraín Canché Solís, Antonio O. Chab-Ruiz, Katia I. Cob-Rivera, Benito Dzib-Castillo, Rodrigo Enrique Tun-Che, and Juan Carlos Camacho-Chab. 2020. "Inorganic Phosphate Solubilization by a Novel Isolated Bacterial Strain Enterobacter sp. ITCB-09 and Its Application Potential as Biofertilizer" Agriculture 10, no. 9: 383. https://doi.org/10.3390/agriculture10090383






