Abstract
Weeds remain a challenge in organic arable farming, as well as supply ecosystem services. The aim is to control weed densities while hosting a diverse and manageable weed community, preventing domination of few deleterious species. Therefore, we want to understand how specific species are stimulated, and which traits are selected for. This study focuses on crop diversity hypothesizing that (1) regions and (2) crop diversity function as filters for specific weed species traits. We conducted a weed monitoring in spring cereals over 2 years on organic farms in five northern European regions. Management and weed trait variables collected for the occurring species allowed an RLQ fourth-corner analysis. The weed communities were regionally specific, but trait selection was not observed, except in Latvia. Hence, the regional species pool provided different species with similar traits. Crop diversity within the management of spring cereals, such as undersowing and cereal frequency in the rotation, affected weed traits. The number of years under organic production selected no traits, although species numbers are known to increase. Hence, general weed species diversity increased, irrespective of traits. We conclude that organic management may support the agility within the weed community against selection of species and act as a buffer rather than as filter.
1. Introduction
During the last few decades, the floral diversity in arable fields has declined severely, driven among others by the use of fertilizers and herbicides [1,2]. The presence of weeds provides, however, a plethora of beneficial ecosystem services in arable fields [3]. Therefore, a more sustainable weed management is an important step toward ecological intensification [4]. On the other hand, weed pressure still remains the main production-limiting factor in agricultural systems, especially in those systems forgoing the use of herbicides [5,6,7]. Thus, the aim is to continue controlling weeds and, within the remaining weed community, host weakly competitive and manageable species in the absence of herbicides. There have been arguments made for balanced weed communities, in order to mitigate weed problems [8,9]. Furthermore, there are signs indicating that weed diversity and evenness are capable of decreasing total weed biomass, as well as mitigating crop yield losses [10]. High weed functional diversity, with more evenly distributed weed functional space, might lower competition pressure [11,12].
In order to form agronomically innocuous, diverse, and controllable weed communities, we need to understand how these communities are composed, how specific species are stimulated, and which traits are selected for. In the last few decades, the theory of community assembly has been successfully applied to describe the formation and development of weed populations [11,13,14,15]. Trait-based research is argued to be the next step forward in understanding weed diversity and competitiveness [9], as well as in exploring how environment and management can be modified to select for certain preferred traits in the community. Specific weed species can be promoted or discouraged through the selection in specific traits by repetitive and strong filters [13]. These filters include the timing of management, especially the timing of soil disturbance, such as tillage or harrowing, and the crop choice along with sowing time [13,15,16,17]. Crop diversity, temporal and spatial, such as the use of catch crops, intercropping, undersowing, and a diverse crop rotation, could all form another layer of filters, via additional competition for light, soil disturbance, and niche differentiation [11,18]. However, how specific traits are affected by crop diversity factors is still unclear.
From the research done so far, there appears to be a hierarchy of filters and disturbances from both environment (local conditions) and management (crop abiotic factors) [11,15]. As Navas (2012) [11] proposed, the local soil conditions and climate determine the local species pool available, as well as shape crop management. This larger influence of environmental factors on the weed community has been well studied [19,20,21,22,23]. Management factors, such as herbicide use, in addition to crop sowing date and tillage, are considered strong filters for the composition of weed communities, but strengths of the filters vary [11,16,17,24,25]. The crop present is often a much stronger filter than other elements in the rotation [11,20,26,27]. The present crop creates a specific ecological condition [28] and is associated with its crop specific management practices [29]. Rotational elements, such as the effect of previous crop types, use of grass clover ley, undersowing, intercropping, cover cropping, and general crop diversity seem inundated as a filter, taking place in the community already shaped by the stronger primary filters.
Our research focused on the weed community in organic spring cereals in northern European regions and, thus, farming systems without herbicides, in one crop type, with similar tillage regimes. We studied the possible selection of traits in the community by the filters of regional environment and crop rotational elements, with a specific interest in crop management and crop diversity. The weed traits of interest were related to growth (life form, growth form, Grime’s life strategy), life cycle (duration of flowering, germination), and physiology (specific leaf area, plant height, seed weight, affinity to soil nutrient conditions). Firstly, as the data were collected in five different regions (Denmark, Finland, Germany, Latvia, and Sweden), there was large variation in environmental conditions, climate, and geography. Previous studies have indicated effects of environmental factors on weed communities [20,21,30], but it remains unclear if the regions and their specific management were selecting for specific traits. Secondly, we were interested in the effect of crop diversity on weed diversity. In a previous study on the data, we observed selection for specific weed species across regions [18]. Thus, we expect trait selection to cause these reaction patterns. This led to the first hypothesis that regions, with their specific environment, are a strong filter for traits in the weed community. By collecting the data in one crop type, we bypassed the known strong filtering factor of different crops present, allowing for better insight into more subtle and historical cropping filters. Thus, within the chosen study crop spring cereals, we secondly hypothesize that crop diversity and crop management select for weeds with certain traits.
2. Materials and Methods
2.1. Vegetation Data
A weed survey was conducted on 58 organic farms in the northern European countries of Denmark, Finland, Germany, Latvia, and Sweden (Figure A1, Appendix A) [18]. The survey was conducted in spring cereals during 2015 and 2016. In total, 207 fields were monitored at the crop flowering stage (stages 61–69 of the BBCH scale) [31], after all physical weed control measures were completed. During the survey, weed densities and the number of weed species were documented for each field. Within these arable fields, three plots of 100 m2 were surveyed for the density of all individual weed species. To estimate the densities, a classification scale was used, which included 10 density classes, exponentially increasing from fewer than 0.2 individuals per m2 to more than 200 individuals per m2. To avoid edge effects, the plots were randomly located in the field, at least 10 m away from the nearest boundary. Several weed species were impossible to identify at time of the survey and, thus, only classified and recorded at the genus level, such as Vicia spp. To allow for analysis, the classification scale was converted into density values using a logarithmic mean. The Latin names were sourced from the Flora Europaea [32], and species are displayed in the ordination graph with EPPO codes [33].
2.2. Crop Management and Environmental Data
The farmers whose fields were surveyed completed a questionnaire about their farm and field management. From this, information was documented about the site, the current cereal species, the crop sequence for the previous 5 years, primary tillage, weed management, and yield. From the farm and field information, five classes of environmental or management data were selected for the research objective: (1) ‘crop’ (if the cereal crop present is sown on its own, intercropped, or undersown), (2) ‘pre-crop’ (if the preceding crop in rotation was a spring cereal, winter cereal, grass clover ley, row crop, or other spring sown crop), (3) ‘crop diversity frequency’ (the number of uses of crop mixtures, undersown crop, or winter catch crop used in the last 5 years of rotation), (4) ‘rotation’ (the number of cultivations of cereals, grass clover ley, or other crops in the last 5 years of rotation, and (5) ‘harrowing’ (the use of physical weed control in the surveyed spring cereal). Primary tillage was not included as a variable, because inversion tillage was the common primary tillage practice on all the farms involved in the survey.
2.3. Trait Data
A trait database was compiled for the observed species. In total, 149 species were included (Table A1, Appendix A). Exceptions were made for species identified at the genus level for reasons of unidentifiable trait variance within the genus, as well as voluntary crops. Both these groups were excluded from the species list. The trait database was based on the database of Bàrberi et al. (2018) [34] and expanded further to include species found during the monitoring, but not previously listed (Table A2, Appendix A; includes sources).
The traits included in the analysis were chosen specifically for our research objectives, which were Raunkiaer life form (RLF), growth form (GTF), Grime’s life strategy (GLS), specific leaf area (cm2·g−1) (SLA), plant height (m) (PLH), seed weight (mg) (SWT), duration of flowering period (months) (DFF), seasonality of germination (SSG), and affinity to soil nutrient conditions (SNC). Detailed traits descriptions can be found in Table A2 (Appendix A).
2.4. Data Analysis
A multivariate analysis of the weed species composition was performed to study the dispersal of species and sites. The weed species records were used as presence/absence data for the multivariate analysis, as the large variation of species density data caused extreme and uninterpretable ordination patterns. A correspondence analysis (CA) [35] was performed on the whole dataset, and the resulting ordination plot displayed sites and species dispersal.
The relationships between region and traits and between crop management and traits were studied by means of a RLQ method, which addresses the fourth-corner problem [36,37]. This analysis was performed on the basis of three datasets. The first was the so-called R-table, which consists of ‘environmental’ or, in our case, management data per field. The second was the L-table, which contains the density data of each species for each field. The third was the Q-table, which contains the trait data for each species. The analysis combines several multivariate techniques in order to relate the species traits to the management or environment data.
The three datasets were first analyzed through the use of ordination methods. This meant a CA for the L table and a Hill-Smith ordination for the Q and R tables [38]. The results from these ordinations were used for the RLQ analysis. The relationships between the management variables and the traits were then tested by means of a fourth-corner analysis [36]. To test the crop management variables more closely, a backward selection was made, excluding the traits which had no significant correlation with management variables. Qualitative traits were tested on all levels but showed no significant relationships. Therefore, the following traits were entered as qualitative, and interpreted from the functional group analysis: RLF, GTF, GLS, SSG, and SNC. Associations between two categorical variables were tested with Pearson chi-square statistics (X2), associations between a categorical variable and a continuous variable were tested using a pseudo-F and Pearson’s r correlation ratio, and associations between two continuous variables were tested using a Pearson correlation coefficient.
Following the fourth-corner analysis, biplots for the management and trait data were compiled, which, after the species distribution on the basis of management variables and traits, were plotted in the ordination space. The functional groups were identified using the hierarchical cluster analysis, using Ward’s method based on the Euclidean distances, for more detailed interpretation of the underlying dynamics. The functional groups were observed to further understand the interactive mechanisms of the management and trait variables.
All the statistical analyses were performed with R software version 3.5.1 [39], utilizing the ade4 package [40].
3. Results
3.1. Region Interactions with Traits in the Weed Community
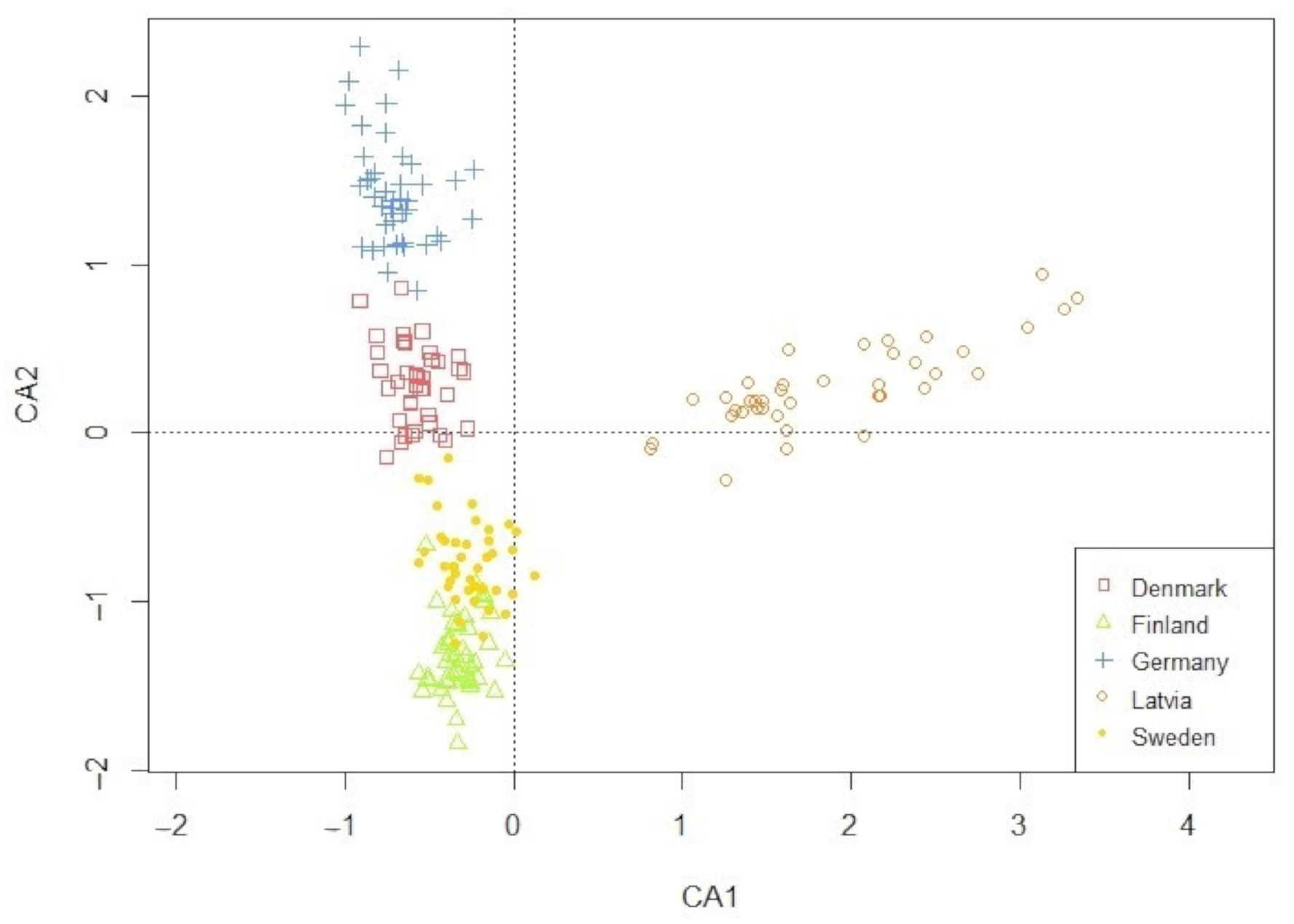
The ordination plot of the CA performed on the weed communities demonstrated a clear geographical clustering (Figure 1). The full species list is available in Table A1 (Appendix A). According to their individual weed communities, fields within a region were visibly similar and clustered in a north-to-south orientation. In contrast, Latvia deviated from this picture by settling into its own cluster. This clustering reflected the regionally diverse environmental factors, which in turn created a weed community that was regionally distinct.
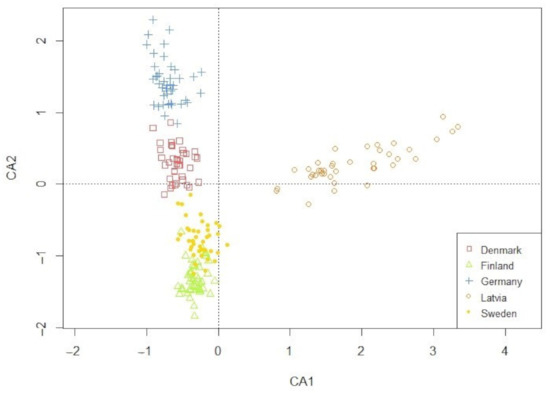
Figure 1.
Projection of the 207 fields on the first two principle axes from the correspondence analysis (CA). Symbols display the five surveyed regions.
The only weed trait regions selected for was Raunkiaer life form (RLF), and it specifically discriminated Latvia (Table 1). Latvia as a region selects for perennial species, specifically for geophytes (plants with underground storage organs) and chamaephytes (dwarf shrubs). When the data from Latvia were tested on their own (data not shown), we found tendencies that perennial species positively correlated with crop and pre-crop (X2 = 0.366 and X2 = 0.322, respectively) and negatively correlated with the use of winter catch crops and other crops (other than cereals, grass, or clover ley) in the rotation (X2 = 0.242 and X2 = 0.245, respectively). The other traits and regions had no relationships.

Table 1.
Relationships between weed traits and region variables represented by the adjusted p-values from the fourth corner analysis.
3.2. Crop Management Interactions with Traits in the Weed Community
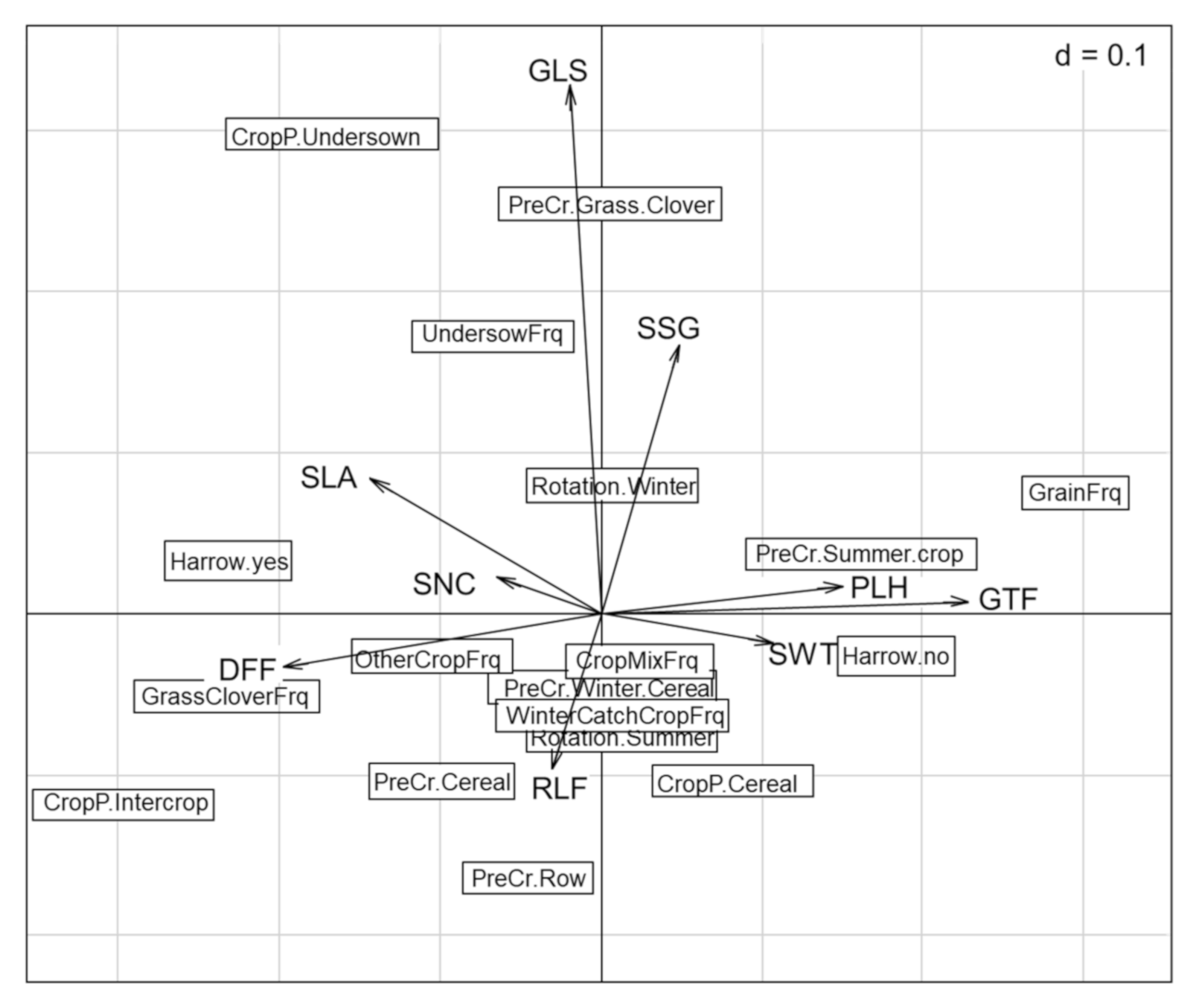
The subsequent analysis excluded the data from Latvia, as this region expressed a distinct trait selection on local level. The first two axes of the RLQ accounted for 46.8% and 21% of the total inertia (Figure 2). The Monte Carlo test indicated the relationships between traits and environment which were generally significant (p < 0.05, based on 9999 permutations). The first two axes of the RLQ showed relatively low correlations (23% and 20%, respectively), but the variance of the environmental scores was well preserved on the first two axes with 76%. The variance of the traits scores on the first two axes was up to 84%.
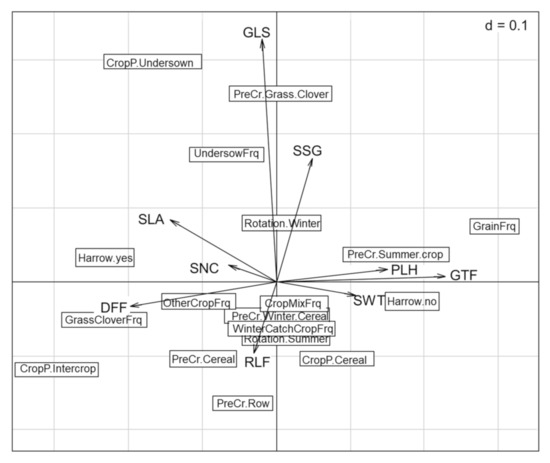
Figure 2.
Biplot representing traits (arrows) and management (boxes) data. Traits: Raunkiaer life form (RLF), growth form (GTF), Grime’s life strategy (GLS), specific leaf area (SLA), plant height (PLH), seed weight (SWT), duration of flowering period (DFF), seasonality of germination (SSG), and affinity to soil nutrient conditions (SNC). Management: harrowing (yes, no), crop present (cereal, intercrop, undersown), previous crop (cereal, grass clover, row crop, summer crop, winter crop), dominant seasonality of the crop rotation (summer, winter), crop diversity frequency in last 5 years (crop mixture, undersowing, winter catch crop), and rotation frequency in last 5 years (grain, grass clover, other crops).
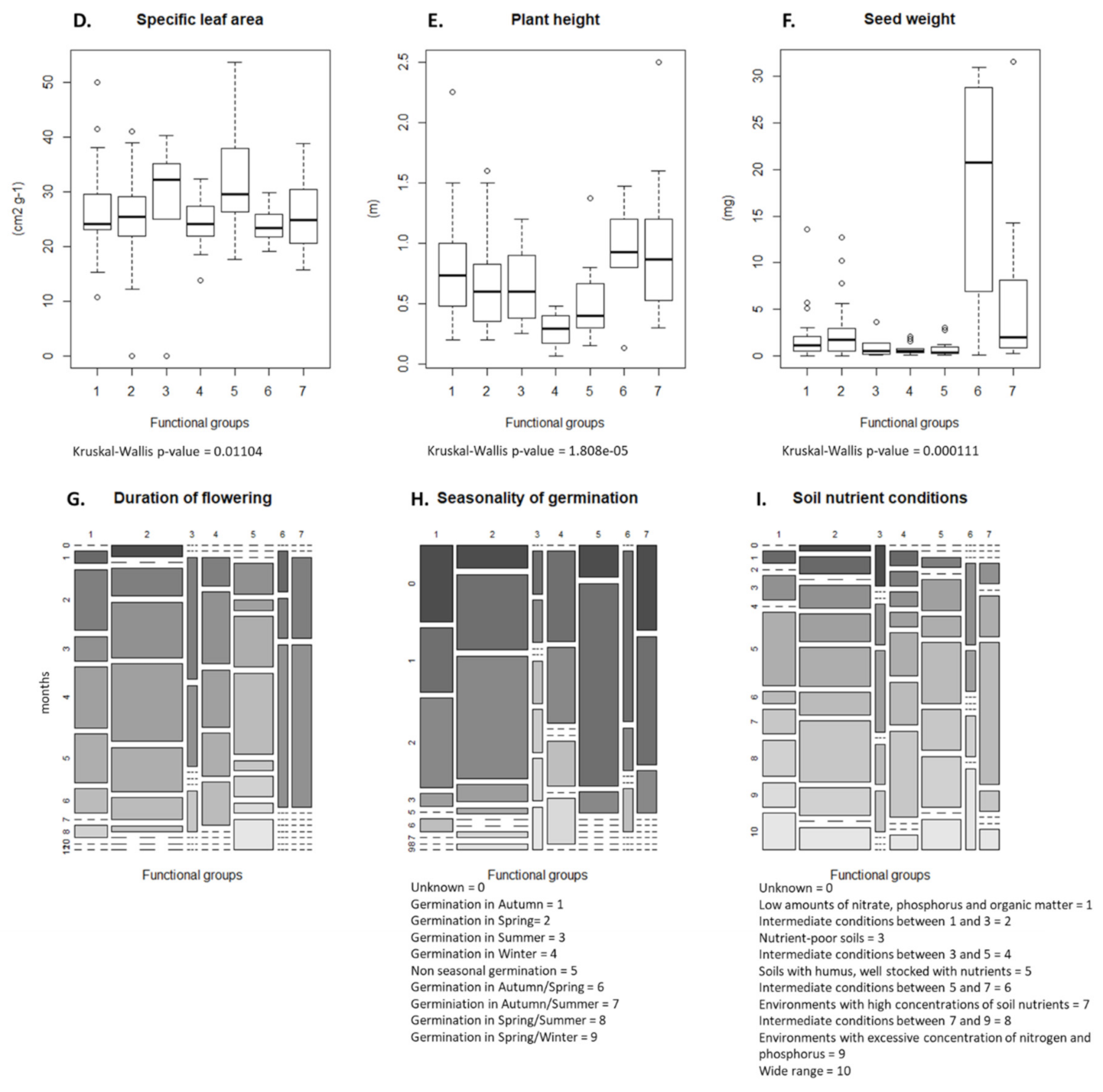
According to the fourth-corner analysis (Table 2 and Figure 2), elements of the rotation in which spring cereals were cropped had the strongest interactions with weed traits. Undersowing was positively associated with Grime’s life strategy, while the years of cereal in the rotation correlated negatively with duration of flowering. These significant results were followed by visible trends in the results (Table 2). Harrowing negatively impacted the plant height and selected for rosette forming species, reducing creeping or ascending species. The years of cereals or grass clover ley selected differently for growth form, with cereals selecting for ascending and creeping species, while grass clover ley selected for rosette species. Grass clover ley was also associated with longer flowering periods. A pre-crop of spring cereals was positively associated with nitrophile species. When the present crop was pure cereals, these tended to select for species with a competitive life strategy. A present crop with undersown crops was associated positively with species with a more stress tolerant or diverse life strategy. Specific leaf area was not affected by any of the tested crop management variables; this is echoed in Figure 2, where this trait is positioned unattached of the other interactions. The number of years a field was under organic cropping was tested, but no trait selection was observed.

Table 2.
Relationships between weed traits and crop management variables represented by the adjusted p-values from the fourth-corner analysis.
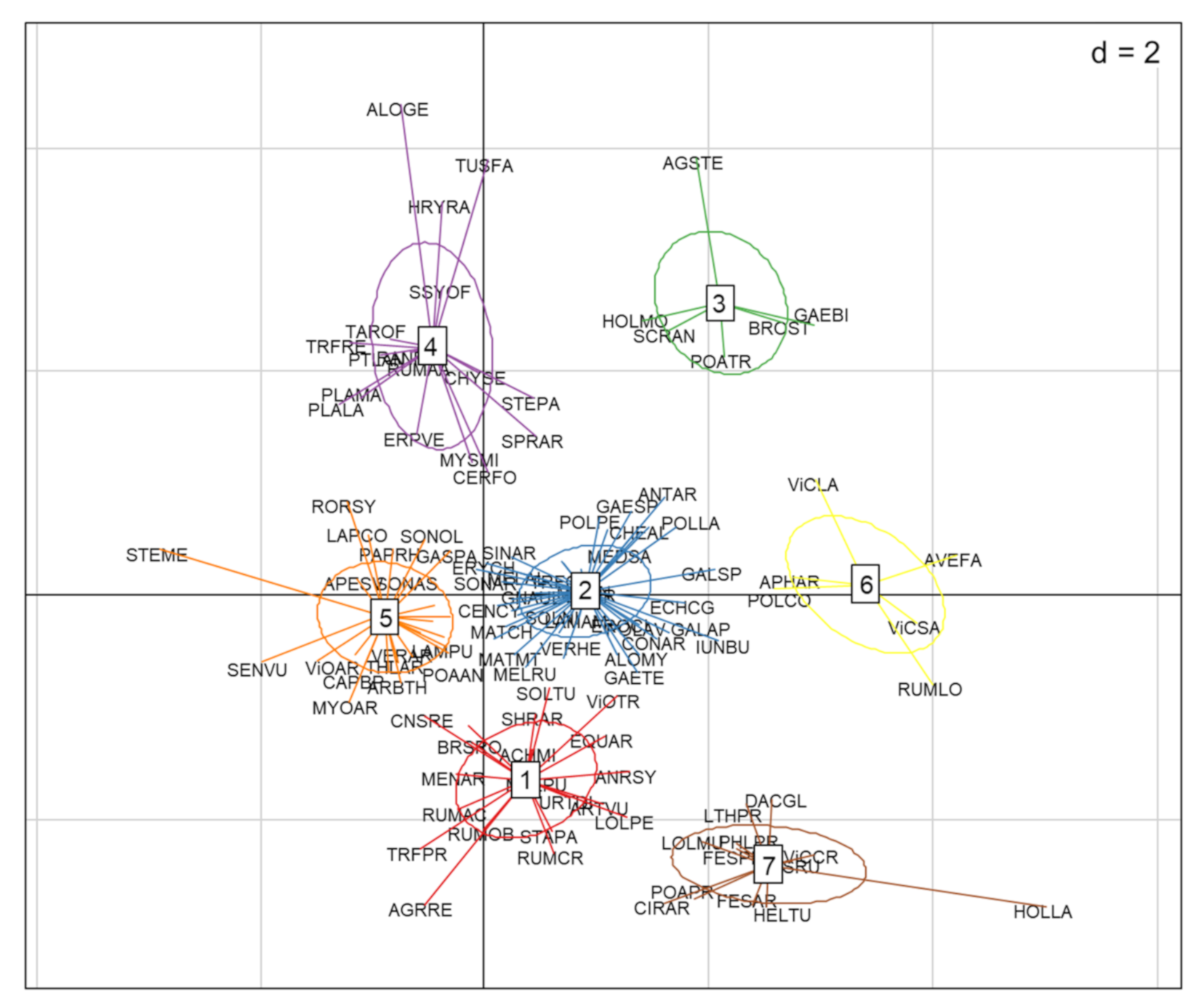
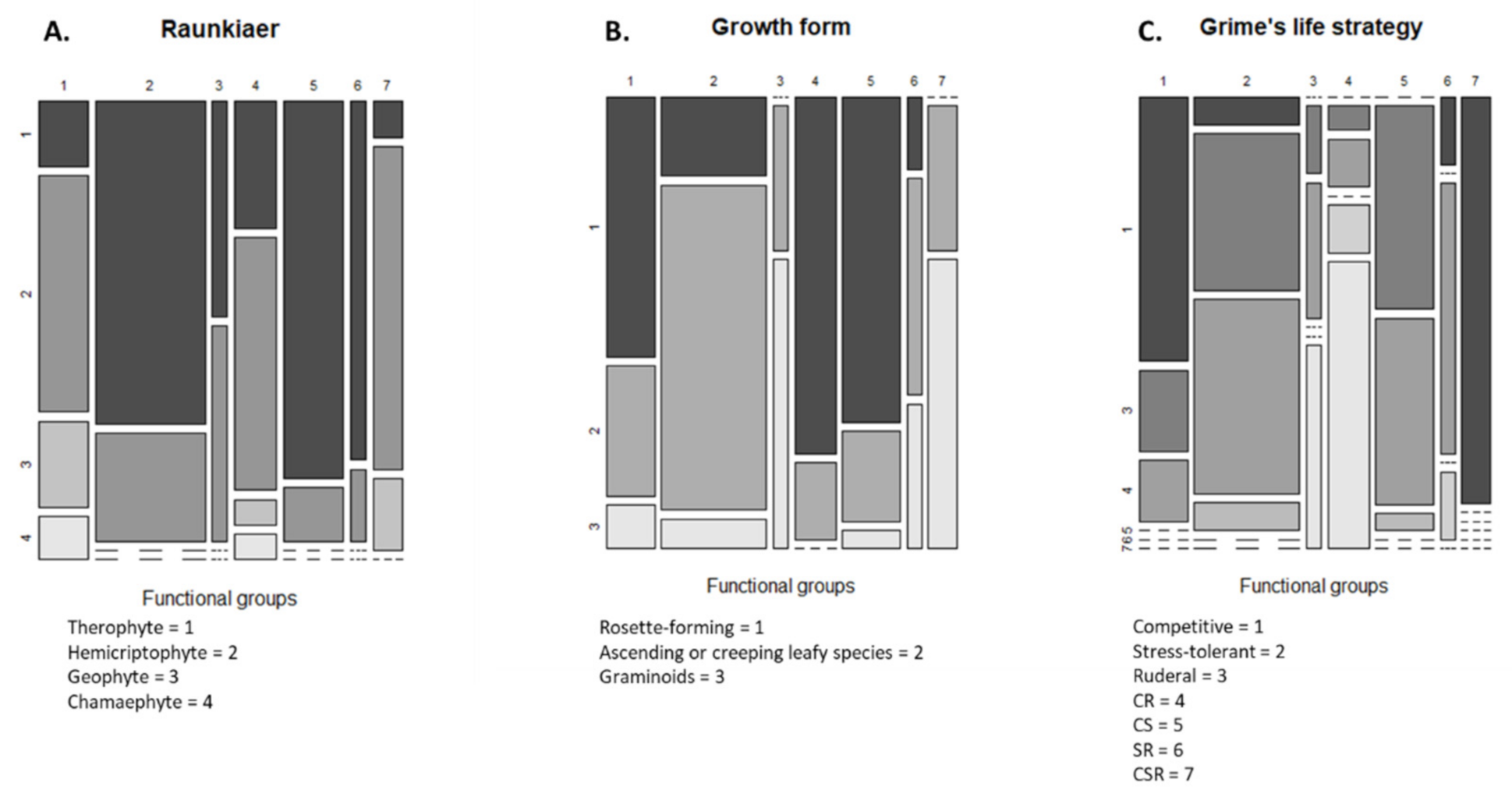
The cluster analysis identified seven functional groups (Figure 3). The distribution values of the nine traits of each of these groups are presented in Figure 4A–I. Group 1 was characterized by competitive perennials, group 2 was characterized by large annuals, group 3 consisted mostly of annual grasses, group 4 was composed of rosette forming perennials, which could be characterized as grassland species, group 5 included rosette-forming autumnal annuals, group 6 specifically involved big-seeded annuals, and group 7 consisted of perennial grasses. When studying Figure 3 and Figure 4 while observing Table 2, patterns emerge. Group 4, consisting of grassland species and characterized by a diversified life strategy, was correlated with the use of grass clover ley and undersown crops. Groups 2 (larger annuals) and 6 (big seeded annuals) were positioned in the direction of growth form, seed weight, and plant height; however, it is less clear how these groups load in the absence of harrowing. Both groups had the same growth form composition and could both be influenced by the frequency of cereal and grass clover ley in the rotation. The duration of flowering positively correlated with the use of grass clover ley, and the group occurring on basis of this trait was group 5 (rosette-forming annuals with long flowering periods). Group 2 with large annuals occurred opposite and toward the cultivation of cereals in the rotation and displayed shorter flowering periods. Group 3, broadly consisting of annual grasses with indifferent or complex seasonal germination, positioned itself along the SSG axes and opposite the use of winter catch crops. Winter catch crops posed a selection pressure on species more clearly autumn- or spring-germinating. Group 1 and 7, large perennials and perennial grasses, showed no clear association according to their positioning. From the lack of interaction of crop management and the Raunkiaer trait, it is clear that annuals and perennials are not directly affected as traits by the crop management.
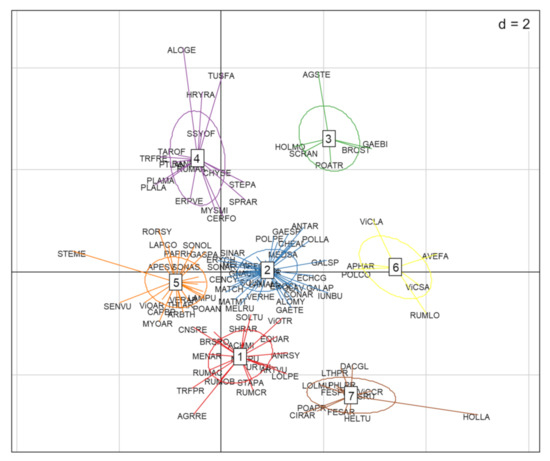
Figure 3.
Ordination plot of species with functional group clusters along the first two RLQ axes. EPPO codes are used to indicate the weed species [33]. Not all species are displayed for readability. Functional groups: (1) competitive perennials, (2) larger annuals, (3) annual grasses, (4) grassland perennials, (5) smaller rosette forming annuals, (6) big seeded annuals, and (7) perennial grasses.
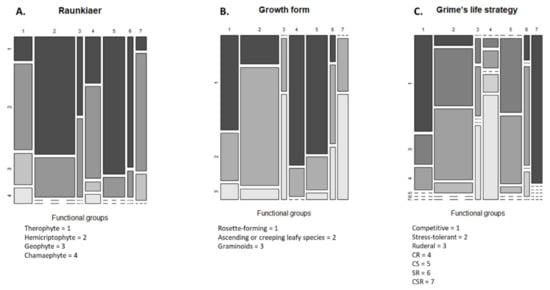
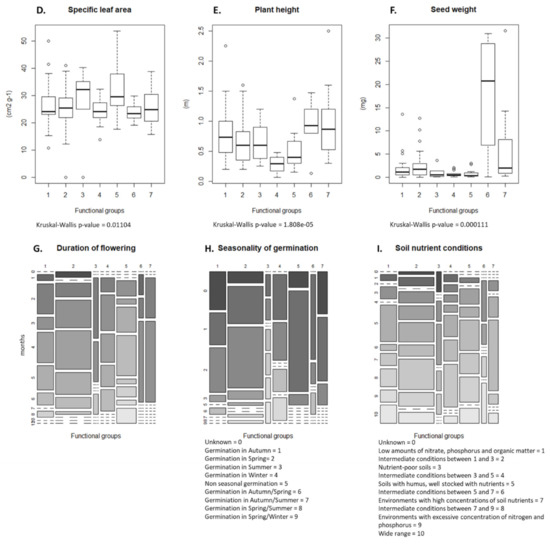
Figure 4.
(A–I) Distribution of trait values within the seven functional groups. Boxplots are used to display continuous traits, while mosaic plots are used for categorical traits; in the case of duration of flowering, a mosaic plot was also chosen. Functional groups: (1) competitive perennials, (2) larg annuals, (3) annual grasses, (4) grassland perennials, (5) rosette forming annuals, (6) big seeded annuals, and (7) perennial grasses.
4. Discussion
Evident from the clear clustering (Figure 1) the weed communities found in this study were regionally specific and influenced by both the local climate and the soil conditions, as well as the local management. The finding that weed species compose locally unique weed communities is coherent with other studies on the effects of local soil type, altitude, and climatic conditions on weed species throughout Europe [23,29,41,42,43]. However, when analyzing, if region selected for certain traits, the results indicate limited influence (Table 1). With the Latvia region as the exception, we found our first hypothesis not supported. An accumulative effect of ‘region’ as a filter for trait selection has not been often investigated, although soil type, temperature, and precipitation have been studied by others [24,42]. Fried et al. (2008) [20] stressed the weak effects of location, as subordinate to the stronger selection of crop choice and sowing date.
Although we conducted the study within spring cereals solely, meaning one crop type and spring sowing, the filter of region was still minor. The Raunkiaer trait selection in Latvia toward hemicryptophytes and geophytes weed species, locally presented in high densities of Elytrigia repens (L.) Nevski, Equisetum arvense L., and Taraxacum officinale F. H. Wigg., could be explained by the relative high use of undersowing, ley farming, and fodder crops. These different choices in management reduce the incidences of (soil) disturbance, favoring perennial species [25,44]. The ordination plot based on the weed species of all sites (Figure 1) revealed that the species composition was very different and specific for each region. Together with the weak selection for traits, this suggests that, although the weed communities were different in species composition, they served the same set of traits. Hence, the regional species pool providing different species with similar traits allows other species in each region to use the opportunities provided by spring cereal fields. Another explanation for the minor differences in traits could be the limited traits geared toward geography, such as latitude and temperature, which were not included in the analysis [41,42].
The crop management variables on undersowing and cereal frequency in the rotation were found to affect weed traits (Table 2), confirming our second hypothesis. The undersowing frequency connected with Grime’s life strategies selected for weed species with a more diverse strategy. As shown in Figure 4, group 4 consists mostly of species which combined all three strategies (competitive, stress-tolerant, and ruderal). The repetitive undersowing of legumes and grasses selected for species which can deal with the additional competition, not through ‘competitiveness’ per se, but by being adaptable. Gunton et al. (2011) [17] found an interaction of the Grime’s life strategy trait with the crop architecture, where single-stemmed or open-rosette crops selected for more competitive species, showing the opposite movement. The dominance of cereals in the previous years of the rotation selected for species with shorter periods of flowering. However, general cereal cropping in the agronomic context of this study was based on spring sowing varieties (4:1 ratio for spring–autumn in our data), thus pushing for weeds with a shorter flowering period by spring tillage [16] and sowing date [17,24].
The functional groups found in Figure 3 and Figure 4 reflected the minor crop management influences and, thus, formed recognizable functional weed groups primarily on the basis of their inherent traits. These groups reflected a rather ordinary organic spring cereal community behavior. The argument has been made before that organic management recreates the arable conditions for this diversity of weed species, allowing them to resettle in the specific niches for the specialist flora selected for by the agronomic constrains [45].
No trait selection was found by the number of years under organic production. This is remarkable as the literature has discussed the selection for perennial species under organic management, especially in the Nordic regions [6,46,47,48]. However, most of these studies were in comparison to conventional management, and it appears from our results that this selection might be initial and does not persist over time. The selection for perennial traits observed by previous studies could have taken place within the first few years of organic cropping. Melander et al. (2016) [49] observed that it took 4–5 years of organic farming to build up a perennial weed problem, hinting at early establishment, although it could take up to 9–10 years in some sites. Hofmeijer et al. (2021) [18] found an increase in species numbers and diversity under organic management over time. Hence, the allocation of weed species reflects the regional species pool but is not trait-related.
The low trait selection in this study might be due to the organic management in general. Needless to say, species numbers in organic arable farming are consistently higher than under conventional management [50,51,52,53]. Richness in the species pool increases the functional redundancy of the weed community [54], i.e., multiple species are able to play equivalent roles, presenting similar functional and physical traits. Multiple species filling functional groups can be assumed to react to agronomical management or other environmental filters. The lack of regional selection for traits supports that even though communities consist of different species, the same traits are fully covered in the agronomical niches.
We see our hypothesis further explored in the recent literature, although for the inverse effect. Studies looking into the intensification of agricultural practices have demonstrated a decrease in weed diversity [55], number of species [56], and, ultimately, functional redundancy [57,58]. Together, these declines could lead to loss of resilience. Organic agriculture is arguably less intensive than conventional, lacking the addition of herbicides and inorganic fertilizers, while also being strong weed species filters. In our study, we observed the reverse effect on the clearly observed increase in species diversity [18]. This could mean a higher level of functional redundancy. Hence, organic management could support a certain agility within the weed community against selection of species and could be additionally considered to buffer rather than filter weed traits.
5. Conclusions
The lack of regional trait selection provides insight in the behavior of different weed species within similar agricultural niches, where the exception of Latvia diverts back to the influence of management rather than location. The observed trait selection by crop diversity and the general effects of organic management found in this study are promising for the approach that weed diversity is able to mitigate the dominance of deleterious species [8,9,59]. Generally, smart and diverse implementation of crop management could stimulate a diverse weed flora, which in turn can form a manageable arable weed community. Additionally, the discussed inverse effect of the implementation of less intense or organic management indicates potential for the prevention of species losses, ecosystem service provision, and mitigation of systemic disturbances, such as changes in agricultural management or environmental factors. Hence, this potential buffering effect requires further exploration.
Author Contributions
Conceptualization, M.A.J.H. and B.G.; methodology, M.A.J.H. and B.G.; formal analysis, M.A.J.H. and B.G.; investigation, M.A.J.H., B.G., B.M., J.S., A.L., L.Z.; resources, B.G.; data curation, B.G.; writing—original draft preparation, M.A.J.H.; writing—review and editing, M.A.J.H., B.G., B.M., J.S., A.L., L.Z.; visualization, M.A.J.H.; supervision, B.G.; project administration, B.G.; funding acquisition, M.A.J.H., B.G., B.M., J.S., A.L., L.Z. All authors have read and agreed to the published version of the manuscript.
Funding
This research project was supported within the ERA-net CORE Organic Plus transnational programmes (Project ID 1381). The study was funded in Germany by the Federal Ministry of Food and Agriculture (BMEL) on the basis of a decision by the Parliament of the Federal Republic of Germany via the Federal Office for Agriculture and Food (BLE) under the Program BOELN (organic and other ways of integrated agriculture). We acknowledge the Ministry of Environment and Food of Denmark and the International Center for Research in Organic Food Systems for funding the Danish part of this research. The Rural Support Service of the Ministry of Agriculture funded the research in Latvia. National funding in Finland was received from the MAKERA grant of the Ministry of Agriculture and Forestry.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Data is available on request from the last author.
Acknowledgments
The authors would like to thank all the participating farmers, stakeholders, and students for their facilitating role in this study. The authors also thank Friederike de Mol for her statistical advice.
Conflicts of Interest
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
Appendix A
Figure A1.
Location of the organic farms monitored in Germany, Denmark, Sweden, Finland, and Latvia. Published as Figure 1 in Hofmeijer et al. (2021) [18].
Figure A1.
Location of the organic farms monitored in Germany, Denmark, Sweden, Finland, and Latvia. Published as Figure 1 in Hofmeijer et al. (2021) [18].

Table A1.
Species found in the monitoring and their relative frequency.
Table A1.
Species found in the monitoring and their relative frequency.
| EPPO | Latin Name | Frequency | EPPO | Latin Name | Frequency |
|---|---|---|---|---|---|
| STEME | Stellaria media (L.) Cirillo | 0.903 | GNAUL | Gnaphalium uliginosum L. | 0.043 |
| VIOAR | Viola arvensis Murray | 0.860 | CMPRA | Campanula rapunculoides L. | 0.039 |
| CIRAR | Cirsium arvense (L.) Scop. | 0.850 | GASPA | Galinsoga parviflora Cav. | 0.039 |
| MATIN | Tripleurospermum inodorum (L.) Sch. Bip. | 0.841 | GERMO | Geranium molle L. | 0.039 |
| CHEAL | Chenopodium album L. | 0.778 | HORVX | Hordeum vulgare L. | 0.039 |
| MYOAR | Myosotis arvensis (L.) Hill | 0.734 | AEOPO | Aegopodium podagraria L. | 0.034 |
| AGRRE | Elytrigia repens (L.) Nevski | 0.720 | LOLSS | Lolium L. sp. | 0.034 |
| POLAV | Polygonum aviculare subsp. aviculare L. | 0.662 | LTHSS | Lathyrus L. sp. | 0.034 |
| POLCO | Fallopia convolvulus (L.) Å. Löve | 0.652 | SENVU | Senecio vulgaris L. | 0.034 |
| CAPBP | Capsella bursa-pastoris (L.) Medik. | 0.594 | SSYOF | Sisymbrium officinale (L.) Scop. | 0.034 |
| SPRAR | Spergula arvensis L. | 0.551 | TRFPR | Trifolium pratense L. | 0.034 |
| FUMOF | Fumaria officinalis L. | 0.531 | ANRSY | Anthriscus sylvestris (L.) Hoffm. | 0.029 |
| TAROF | Taraxacum sect. Taraxacum F. H. Wigg. | 0.512 | CHYSE | Glebionis segetum (L.) Fourr. | 0.029 |
| LAMPU | Lamium purpureum L. | 0.507 | DECCA | Deschampsia cespitosa (L.) P. Beauv. | 0.029 |
| EQUAR | Equisetum arvense L. | 0.478 | LOLMU | Lolium multiflorum Lam. | 0.029 |
| LAPCO | Lapsana communis L. | 0.454 | MEDLU | Medicago lupulina L. | 0.029 |
| THLAR | Thlaspi arvense L. | 0.454 | SCRAN | Scleranthus annuus L. | 0.029 |
| SONAR | Sonchus arvensis L. | 0.396 | AGSTE | Agrostis capillaris L. | 0.024 |
| LYCAR | Lycopsis arvensis L. | 0.372 | DAUCA | Daucus carota L. | 0.024 |
| POLPE | Persicaria maculosa x minor | 0.367 | PLALA | Plantago lanceolata L. | 0.024 |
| GAESS | Galeopsis L. | 0.348 | RUMOB | Rumex obtusifolius L. | 0.024 |
| VICSS | Vicia L. sp. | 0.314 | ALCVU | Alchemilla xanthochlora Rothm. | 0.019 |
| ERYCH | Erysimum cheiranthoides L. | 0.300 | ATXPA | Atriplex patula L. | 0.019 |
| TRFRE | Trifolium repens L. | 0.300 | FESSS | Festuca L. sp. | 0.019 |
| VICCR | Vicia cracca L. | 0.295 | HYPSS | Hypericum L. sp. | 0.019 |
| CENCY | Cyanus segetum Hill | 0.285 | LUPSS | Lupinus L. sp. | 0.019 |
| VERAR | Veronica arvensis L. | 0.280 | MEDSS | Medicago L. sp. | 0.019 |
| RUMCR | Rumex crispus L. | 0.266 | RUMAC | Rumex acetosa L. | 0.019 |
| GALAL | Galium album Mill. | 0.261 | SHRAR | Sherardia arvensis L. | 0.019 |
| EROCI | Erodium cicutarium (L.) L’Hér. | 0.256 | ACHPT | Achillea ptarmica L. | 0.014 |
| GALSP | Galium spurium L. | 0.256 | ALOMY | Alopecurus myosuroides Huds. | 0.014 |
| VERPE | Veronica persica Poir. | 0.256 | ARBTH | Arabidopsis thaliana (L.) Heynh. | 0.014 |
| POLLA | Persicaria lapathifolia (L.) Delarbre s. l. | 0.246 | AVEFA | Avena fatua L. | 0.014 |
| ACHMI | Achillea millefolium L. | 0.242 | BRASS | Brachiaria (Trin.) Griseb. sp. | 0.014 |
| POAAN | Ochlopoa annua (L.) H. Scholz | 0.237 | CENJA | Centaurea jacea L. | 0.014 |
| RANRE | Ranunculus repens L. | 0.237 | ERYSS | Erysimum L. sp. | 0.014 |
| VICHI | Vicia hirsuta (L.) Gray | 0.237 | FESPR | Schedonorus pratensis (Huds.) P. Beauv. | 0.014 |
| ARTVU | Artemisia vulgaris L. | 0.222 | GAETE | Galeopsis tetrahit | 0.014 |
| TRFSS | Trifolium L. sp. | 0.222 | GASCI | Galinsoga quadriradiata Ruiz & Pav. | 0.014 |
| CONAR | Convolvulus arvensis L. | 0.217 | MYSMI | Myosurus minimus L. | 0.014 |
| GERPU | Geranium pusillum L. | 0.217 | POAPR | Poa pratensis L. | 0.014 |
| RAPRA | Raphanus raphanistrum L. | 0.217 | VERSS | Veronica L. sp. | 0.014 |
| SINAR | Sinapis arvensis L. | 0.217 | VIOTR | Viola tricolor L. | 0.014 |
| TUSFA | Tussilago farfara L. | 0.217 | ALOGE | Alopecurus geniculatus L. | 0.010 |
| GAESP | Galeopsis speciosa Mill. | 0.213 | APHAR | Aphanes arvensis L. | 0.010 |
| GALAP | Galium aparine L. | 0.203 | BROST | Anisantha sterilis (L.) Nevski | 0.010 |
| PAPRH | Papaver rhoeas L. | 0.184 | CERFO | Cerastium fontanum Baumg. | 0.010 |
| CVPCA | Crepis capillaris (L.) Wallr. | 0.164 | CIRVU | Cirsium vulgare (Savi) Ten. | 0.010 |
| LAMAM | Lamium amplexicaule L. | 0.164 | DACGL | Dactylis glomerata L. | 0.010 |
| MATMT | Matricaria discoidea DC. | 0.159 | FESRU | Festuca rubra L. | 0.010 |
| PLAMA | Plantago major L. | 0.155 | IUNBU | Juncus bufonius L. | 0.010 |
| SONAS | Sonchus asper (L.) Hill | 0.155 | MEDSA | Medicago sativa L. | 0.010 |
| MENAR | Mentha arvensis L. | 0.150 | MELAL | Silene latifolia Poir. | 0.010 |
| LOTSS | Lotus L. sp. | 0.145 | POATR | Poa trivialis L. | 0.010 |
| PHLPR | Phleum pratense L. | 0.145 | RANSS | Ranunculus L. sp. | 0.010 |
| STAPA | Stachys palustris L. | 0.140 | TRFCA | Trifolium campestre Schreb. | 0.010 |
| EPHHE | Euphorbia helioscopia L. | 0.130 | URTDI | Urtica dioica L. | 0.010 |
| RUMSS | Rumex L. sp. | 0.130 | ALOSS | Alopecurus sp | 0.005 |
| CHESS | Chenopodium L. sp. | 0.126 | BARVU | Barbarea vulgaris R. Br. | 0.005 |
| PTLAN | Argentina anserina (L.) Rydb. | 0.126 | BORSS | Borago L. sp. | 0.005 |
| EPHES | Euphorbia esula L. | 0.121 | CNSRE | Consolida regalis Gray | 0.005 |
| GAEBI | Galeopsis bifida Boenn. | 0.121 | CONSS | Convolvulus L. sp. | 0.005 |
| PLAME | Plantago media L. | 0.121 | CRDSS | Arabidopsis Heynh. sp. | 0.005 |
| BARSS | Barbarea W. T. Aiton sp. | 0.116 | CRUCR | Carduus crispus L. | 0.005 |
| BRSRO | Brassica rapa subsp. oleifera (DC.) Metzg. | 0.116 | ECHCG | Echinochloa crus-galli (L.) P. Beauv. | 0.005 |
| LOLPE | Lolium perenne L. | 0.116 | FESAR | Schedonorus arundinaceus (Schreb.) Dumort. | 0.005 |
| RUMAA | Rumex acetosella subsp. acetosella L. | 0.116 | HELTU | Helianthus tuberosus L. | 0.005 |
| SONSS | Sonchus L. sp. | 0.111 | HERMZ | Heracleum mantegazzianum Sommier & Levier | 0.005 |
| CERAR | Cerastium arvense L. | 0.106 | HOLLA | Holcus lanatus L. | 0.005 |
| PRASS | Persicaria Mill. sp. | 0.106 | HOLMO | Holcus mollis L. | 0.005 |
| EPHSS | Euphorbia L. sp. | 0.101 | HRYRA | Hypochaeris radicata L. | 0.005 |
| ANTAR | Anthemis arvensis L. | 0.097 | LITAR | Buglossoides arvensis (L.) I. M. Johnst. | 0.005 |
| MATCH | Matricaria chamomilla L. | 0.097 | LIUUT | Linum usitatissimum L. | 0.005 |
| LAMSS | Lamium L. sp. | 0.087 | MALPU | Malva pusilla Sm. | 0.005 |
| LTHPR | Lathyrus pratensis L. | 0.087 | MELRU | Silene dioica (L.) Clairv. | 0.005 |
| RUMLO | Rumex longifolius DC. | 0.082 | MEUAL | Melilotus albus Medik. | 0.005 |
| BRSNN | Brassica napus L. | 0.077 | PIBSA | Pisum sativum L. subsp. sativum | 0.005 |
| PRUVU | Prunella vulgaris L. | 0.077 | PIEAB | Picea abies (L.) H. Karst. | 0.005 |
| SILSS | Silene L. sp. | 0.072 | PLASS | Plantago L. sp. | 0.005 |
| SONOL | Sonchus oleraceus L. | 0.072 | POASS | Poa L. sp. | 0.005 |
| CHYLE | Leucanthemum vulgare (Vaill.) Lam. | 0.068 | RORSY | Rorippa sylvestris (L.) Besser | 0.005 |
| RUMCO | Rumex conglomeratus Murray | 0.068 | SETSS | Setaria P. Beauv. sp. | 0.005 |
| RANAC | Ranunculus acris L. | 0.063 | SOLNI | Solanum nigrum L. | 0.005 |
| ERPVE | Erophila verna (L.) Chevall. | 0.058 | SOLTU | Solanum tuberosum L. | 0.005 |
| VERHE | Veronica hederifolia L. | 0.058 | SOOCA | Solidago canadensis L. | 0.005 |
| APESV | Apera spica-venti (L.) P. Beauv. | 0.053 | SSYSS | Sisymbrium L. | 0.005 |
| GERSS | Geranium L. | 0.053 | STASS | Stachys L. sp. | 0.005 |
| VICSA | Vicia sativa L. | 0.053 | STEPA | Stellaria palustris Hoffm. | 0.005 |
| ARFTO | Arctium tomentosum Mill. | 0.048 | TRKMO | Medicago sativa L. | 0.005 |
| AMSSS | Amsinckia Lehm. sp. | 0.043 | URTUR | Urtica urens L. | 0.005 |
| ANGAR | Anagallis arvensis L. | 0.043 | VESSS | Verbascum L. sp. | 0.005 |
| BIDTR | Bidens tripartitus L. | 0.043 | VICLA | Vicia lathyroides L. | 0.005 |
| BRSRA | Brassica rapa subsp. campestris (L.) A. R. Clapham | 0.043 | VICVI | Vicia villosa Roth | 0.005 |
Volunteer crops were removed from this list. EPPO codes [33] and Latin names based on the Flora Europaea [32] were used to define species.

Table A2.
The nine functional traits included in the database.
Table A2.
The nine functional traits included in the database.
| Trait Code | Source of Information | Trait Level/Value | Trait Explanation |
|---|---|---|---|
| 1. Raunkiaer life form (RLF) | A, C, H | Qualitative; response and effect trait | The Raunkiær system (1934) is based on the place of the plant’s growth point (bud) during seasons with adverse conditions (cold seasons, dry seasons) Therophytes: Annual plants which survive the unfavorable season in the form of seeds and complete their life cycle during favorable seasons; annual species are therophytes Hemicryptophytes: Buds at or near the soil surface Geophytes: Below ground, with resting buds lying either beneath the surface of the ground as a rhizome (bulb, corm, etc.) Chamaephytes: Buds on persistent shoots near the ground; woody plants with perennating buds borne close to the ground, no more than 25 cm above the soil surface |
| 1.1 1.2 1.3 1.4 | Therophyte = 1 Hemicriptophyte = 2 Geophyte = 3 Chamaephyte = 4 | ||
| 2. Growth form (GTF) | A, C | Qualitative; response and effect trait | Species can be grouped into growth form classes on the basis of their similarities in structure and function. Herbaceous species can be grouped into rosette-forming, ascending, or creeping leafy species and graminoids considering the architecture and occupancy of the space Rosette-forming: Cluster of leaves with very short internodes that are crowded together, normally on the soil surface but sometimes higher on the stem Ascending or creeping leafy stems: Growing uprightly, in an upward direction, heading in the direction of the top or growing along the ground and producing roots at intervals along surface Graminoids: Grass or grass-like plant, including grasses (Poaceae), sedges (Cyperaceae), and rushes (Juncaceae) |
| 2.1 2.2 2.3 | Rosette-forming = 1 Ascending or creeping leafy species = 2 Graminoids = 3 | ||
| 3. Grime’s life strategy (GLS) | C, D, F, I | Qualitative; response and effect trait | Plants are classified according to their life strategy (Grime, 1974) (C as competitive, S as stress-tolerant, R as ruderal, and the combined strategies CR, CS, SR, and CSR). Despite some species being able to vary their strategy according to environmental and agronomic factors, their main life strategy is indicated For CSR strategy: for some species classes were attributed using the information available for similar species |
| 4.1 4.2 4.3 4.4 4.5 4.6 4.7 | Competitive = 1 Stress-tolerant = 2 Ruderal = 3 CR = 4 CS = 5 SR = 6 CSR = 7 | ||
| 4. Specific leaf area (SLA) | H | Quantitative | SLA is a proxy for a plant’s ability to use light efficiently within the classical acquisition/conservation tradeoff |
| 5. Plant height (PLH) (m) | E, H, J | Quantitative | Plant height characterizes species ability to compete for light with neighboring plants and especially with crop individuals |
| 6. Seed weight (SWT) (mg) | D, G, J | Quantitative | Seed weight is related to species ability to disperse, colonize soil, and persist |
| 7. Seasonality of germination (SSG) | D | Qualitative; response and effect trait | Seed germination period determines the match between a weed species life cycle and the growing cycle of a crop and, hence, its ability to escape disturbance posed by farming practices |
| 7.0 7.1 7.2 7.3 7.4 7.5 7.6 7.7 7.8 7.9 | Unknown = 0 Germination in autumn = 1 Germination in spring= 2 Germination in summer = 3 Germination in winter = 4 Non seasonal germination = 5 Germination in autumn/spring = 6 Germination in autumn/summer = 7 Germination in spring/summer = 8 Germination in spring/winter = 9 | ||
| 8. Duration of flowering period (DFF) (months) | C | Quantitative | Duration of the flowering period indicates the length of the reproduction phase. Mechanical removal of weed seeds before shedding is an excellent strategy preventing weed seeds from entering the seed bank. Duration of the flowering period also informs on the provision of floral resources for higher trophic levels. |
| 9. Affinity to soil nutrient conditions (SNC) | B | Semi-quantitative; response and effect trait | Species are classified on the basis of their affinity to soil nutrient conditions (N) following Ellenberg (1979) nutrient indicator values |
| 9.0 9.1 9.2 9.3 9.4 9.5 9.6 9.7 9.8 9.9 9.10 | Unknown = 0 Oligotrophic soils which contain low amounts of nitrate, as well as phosphorus and organic matter = 1 Intermediate conditions between a and c = 2 Nutrient-poor soils = 3 Intermediate conditions between c and e = 4 Soils with humus, well stocked with nutrients = 5 Intermediate conditions between e and g = 6 Environments with high concentrations of soil nutrients = 7 Intermediate conditions between g and i = 8 Environments with excessive concentration of nitrogen and phosphorus = 9 Wide range = 10 |
Sources of information: A: Pignatti (1982) [60]; B: Ellenberg (1979) [61]; C: Klotz et al. (2002) [62]; D: Fitter and Peat (1994) [63]; E: Missouri Botanical Garden (2016) [64]; F: Grime et al. (2007) [65]; G: Kew Royal Botanical Gardens (2016) [66]; H: Kleyer et al. (2008) [67]; I: Hodgson (2016) [68]. J: Westoby (1998) [69]. Based on the original database of Bàrberi et al. (2018) [34] and extended.
References
- Fried, G.; Petit, S.; Dessaint, F.; Reboud, X. Arable Weed Decline in Northern France: Crop Edges as Refugia for Weed Conservation? Biol. Conserv. 2009, 142, 238–243. [Google Scholar] [CrossRef]
- Storkey, J.; Meyer, S.; Still, K.S.; Leuschner, C. The Impact of Agricultural Intensification and Land-Use Change on the European Arable Flora. Proc. R. Soc. B Biol. Sci. 2012, 279, 1421–1429. [Google Scholar] [CrossRef]
- Marshall, E.J.P.; Brown, V.K.; Boatman, N.D.; Lutman, P.J.W.; Squire, G.R.; Ward, L.K. The Role of Weeds in Supporting Biological Diversity within Crop Fields. Weed Res. 2003, 43, 77–89. [Google Scholar] [CrossRef]
- Petit, S.; Munier-Jolain, N.; Bretagnolle, V.; Bockstaller, C.; Gaba, S.; Cordeau, S.; Lechenet, M.; Mézière, D.; Colbach, N. Ecological Intensification Through Pesticide Reduction: Weed Control, Weed Biodiversity and Sustainability in Arable Farming. Environ. Manag. 2015, 56, 1078–1090. [Google Scholar] [CrossRef]
- Alrøe, H.F.; Halberg, N. Udvikling, Vækst og Integritet i Den Danske Økologisektor. Vidensyntese om Muligheder og Barrierer for Fortsat Udvikling og Markedsbaseret Vækst i Produktion, Forarbejdning og Omsætning af Økologiske Produkter; Alrøe, H.F., Halberg, N., Eds.; Internationalt Center for Forskning i Økologisk Jordbrug og Fødevaresystemer (ICROFS): Tjele, Denmark, 2008. [Google Scholar]
- Turner, R.J.; Davies, G.; Moore, H.; Grundy, A.C.; Mead, A. Organic Weed Management: A Review of the Current UK Farmer Perspective. Crop. Prot. 2007, 26, 377–382. [Google Scholar] [CrossRef]
- Penfold, C.M.; Miyan, M.S.; Reeves, T.G.; Grierson, I.T. Biological Farming for Sustainable Agricultural Production. Aust. J. Exp. Agric. 1995, 35, 849–856. [Google Scholar] [CrossRef]
- Melander, B.; Rasmussen, I.A.; Bàrberi, P. Integrating Physical and Cultural Methods of Weed Control—Examples from European Research. Weed Sci. 2005, 53, 369–381. [Google Scholar] [CrossRef]
- Storkey, J.; Neve, P. What Good Is Weed Diversity? Weed Res. 2018, 58, 239–243. [Google Scholar] [CrossRef]
- Adeux, G.; Vieren, E.; Carlesi, S.; Bàrberi, P.; Munier-Jolain, N.; Cordeau, S. Mitigating Crop Yield Losses through Weed Diversity. Nat. Sustain. 2019, 2, 1018–1026. [Google Scholar] [CrossRef]
- Navas, M.-L. Trait-Based Approaches to Unravelling the Assembly of Weed Communities and Their Impact on Agro-Ecosystem Functioning. Weed Res. 2012, 52, 479–488. [Google Scholar] [CrossRef]
- Macarthur, R.; Levins, R. The Limiting Similarity, Convergence, and Divergence of Coexisting Species. Am. Nat. 1967, 101, 377–385. [Google Scholar] [CrossRef]
- Booth, B.D.; Swanton, C.J. Assembly Theory Applied to Weed Communities. Weed Sci. 2002, 50, 2–13. [Google Scholar] [CrossRef]
- Storkey, J.; Moss, S.R.; Cussans, J.W. Using Assembly Theory to Explain Changes in a Weed Flora in Response to Agricultural Intensification. Weed Sci. 2010, 58, 39–46. [Google Scholar] [CrossRef]
- Gaba, S.; Fried, G.; Kazakou, E.; Chauvel, B.; Navas, M.-L. Agroecological Weed Control Using a Functional Approach: A Review of Cropping Systems Diversity. Agron. Sustain. Dev. 2014, 34, 103–119. [Google Scholar] [CrossRef]
- Smith, R.G. Timing of Tillage Is an Important Filter on the Assembly of Weed Communities. Weed Sci. 2006, 54, 705–712. [Google Scholar] [CrossRef]
- Gunton, R.M.; Petit, S.; Gaba, S. Functional Traits Relating Arable Weed Communities to Crop Characteristics. J. Veg. Sci. 2011, 22, 541–550. [Google Scholar] [CrossRef]
- Hofmeijer, M.A.J.; Melander, B.; Salonen, J.; Lundkvist, A.; Zarina, L.; Gerowitt, B. Crop Diversification Affects Weed Communities and Densities in Organic Spring Cereal Fields in Northern Europe. Agric. Ecosyst. Environ. 2021, 308, 107251. [Google Scholar] [CrossRef]
- Salonen, J. Weed Infestation and Factors Affecting Weed Incidence in Spring Cereals in Finland—A Multivariate Approach. Agric. Food Sci. 1993, 2, 525–536. [Google Scholar] [CrossRef]
- Fried, G.; Norton, L.R.; Reboud, X. Environmental and Management Factors Determining Weed Species Composition and Diversity in France. Agric. Ecosyst. Environ. 2008, 128, 68–76. [Google Scholar] [CrossRef]
- Fried, G.; Cordeau, S.; Metay, A.; Kazakou, E. Relative Importance of Environmental Factors and Farming Practices in Shaping Weed Communities Structure and Composition in French Vineyards. Agric. Ecosyst. Environ. 2019, 275, 1–13. [Google Scholar] [CrossRef]
- Andreasen, C.; Skovgaard, I.M. Crop and Soil Factors of Importance for the Distribution of Plant Species on Arable Fields in Denmark. Agric. Ecosyst. Environ. 2009, 133, 61–67. [Google Scholar] [CrossRef]
- Hanzlik, K.; Gerowitt, B. The Importance of Climate, Site and Management on Weed Vegetation in Oilseed Rape in Germany. Agric. Ecosyst. Environ. 2011, 141, 323–331. [Google Scholar] [CrossRef]
- Fried, G.; Kazakou, E.; Gaba, S. Trajectories of Weed Communities Explained by Traits Associated with Species’ Response to Management Practices. Agric. Ecosyst. Environ. 2012, 158, 147–155. [Google Scholar] [CrossRef]
- Armengot, L.; Blanco-Moreno, J.M.; Bàrberi, P.; Bocci, G.; Carlesi, S.; Aendekerk, R.; Berner, A.; Celette, F.; Grosse, M.; Huiting, H.; et al. Tillage as a Driver of Change in Weed Communities: A Functional Perspective. Agric. Ecosyst. Environ. 2016, 222, 276–285. [Google Scholar] [CrossRef]
- Smith, R.G.; Gross, K.L. Assembly of Weed Communities along a Crop Diversity Gradient. J. Appl. Ecol. 2007, 44, 1046–1056. [Google Scholar] [CrossRef]
- Meiss, H.; Médiène, S.; Waldhardt, R.; Caneill, J.; Munier-Jolain, N. Contrasting Weed Species Composition in Perennial Alfalfas and Six Annual Crops: Implications for Integrated Weed Management. Agron. Sustain. Dev. 2010, 30, 657–666. [Google Scholar] [CrossRef]
- Hallgren, E.; Palmer, M.W.; Milberg, P. Data Diving with Cross-Validation: An Investigation of Broad-Scale Gradients in Swedish Weed Communities. J. Ecol. 1999, 87, 1037–1051. [Google Scholar] [CrossRef]
- Andersson, T.N.; Milberg, P. Weed Flora and the Relative Importance of Site, Crop, Crop Rotation, and Nitrogen. Weed Sci. 1998, 46, 30–38. [Google Scholar] [CrossRef]
- Pinke, G.; Pál, R.; Botta-Dukát, Z. Effects of Environmental Factors on Weed Species Composition of Cereal and Stubble Fields in Western Hungary. Cent. Eur. J. Biol. 2010, 5, 283–292. [Google Scholar] [CrossRef]
- Meier, U. Phenological Growth Stages. In Phenology: An Integrative Environmental Science; Schwartz, M.D., Ed.; Tasks for Vegetation Science; Springer Netherlands: Dordrecht, The Netherlands, 2003; pp. 269–283. ISBN 978-94-007-0632-3. [Google Scholar]
- The Euro + Med Plantbase Project. Available online: https://ww2.bgbm.org/EuroPlusMed/query.asp (accessed on 7 April 2021).
- EPPO Global Database. Available online: https://gd.eppo.int/ (accessed on 6 April 2021).
- Bàrberi, P.; Bocci, G.; Carlesi, S.; Armengot, L.; Blanco-Moreno, J.M.; Sans, F.X. Linking Species Traits to Agroecosystem Services: A Functional Analysis of Weed Communities. Weed Res. 2018, 58, 76–88. [Google Scholar] [CrossRef]
- Canonical Correspondence Analysis: A New Eigenvector Technique for Multivariate Direct Gradient Analysis-Ter Braak-1986-Ecology-Wiley Online Library. Available online: https://esajournals.onlinelibrary.wiley.com/doi/abs/10.2307/1938672 (accessed on 6 April 2021).
- Dray, S.; Legendre, P. Testing the Species Traits–Environment Relationships: The Fourth-Corner Problem Revisited. Ecology 2008, 89, 3400–3412. [Google Scholar] [CrossRef] [PubMed]
- Dolédec, S.; Chessel, D.; ter Braak, C.J.F.; Champely, S. Matching Species Traits to Environmental Variables: A New Three-Table Ordination Method. Environ. Ecol. Stat. 1996, 3, 143–166. [Google Scholar] [CrossRef]
- Hill, M.O.; Smith, A.J.E. Principal Component Analysis of Taxonomic Data with Multi-State Discrete Characters. Taxon 1976, 25, 249–255. [Google Scholar] [CrossRef]
- R Core Team; R Foundation for Statistical Computing: Vienna, Austria, 2017.
- Dray, S.; Dufour, A.-B. The Ade4 Package: Implementing the Duality Diagram for Ecologists. J. Stat. Softw. 2007, 22. [Google Scholar] [CrossRef]
- Glemnitz, M.; Czimber, G.; Radics, L.; Hoffmann, J. Weed Flora Composition along a North-South Climate Gradient in Europe. Acta Agron. Óvár. 2000, 42, 155–169. [Google Scholar]
- Lososová, Z.; Chytrý, M.; Cimalová, S.; Kropáč, Z.; Otýpková, Z.; Pyšek, P.; Tichý, L. Weed Vegetation of Arable Land in Central Europe: Gradients of Diversity and Species Composition. J. Veg. Sci. 2004, 15, 415–422. [Google Scholar] [CrossRef]
- Šilc, U.; Vrbničanin, S.; Božić, D.; Čarni, A.; Stevanović, Z.D. Weed Vegetation in the North-Western Balkans: Diversity and Species Composition. Weed Res. 2009, 49, 602–612. [Google Scholar] [CrossRef]
- Andreasen, C.; Stryhn, H. Increasing Weed Flora in Danish Arable Fields and Its Importance for Biodiversity. Weed Res. 2008, 48, 1–9. [Google Scholar] [CrossRef]
- Vigueira, C.C.; Olsen, K.M.; Caicedo, A.L. The Red Queen in the Corn: Agricultural Weeds as Models of Rapid Adaptive Evolution. Heredity 2013, 110, 303–311. [Google Scholar] [CrossRef]
- Albrecht, H. Development of Arable Weed Seedbanks during the 6 Years after the Change from Conventional to Organic Farming. Weed Res. 2005, 45, 339–350. [Google Scholar] [CrossRef]
- Rydberg, N.T.; Milberg, P. A Survey of Weeds in Organic Farming in Sweden. Biol. Agric. Hortic. 2000, 18, 175–185. [Google Scholar] [CrossRef]
- Salonen, J.; Hyvönen, T.; Jalli, H. Composition of Weed Flora in Spring Cereals in Finland—A Fourth Survey. Agric. Food Sci. 2011, 20, 245–261. [Google Scholar] [CrossRef]
- Melander, B.; Rasmussen, I.A.; Olesen, J.E. Incompatibility between Fertility Building Measures and the Management of Perennial Weeds in Organic Cropping Systems. Agric. Ecosyst. Environ. 2016, 220, 184–192. [Google Scholar] [CrossRef]
- Hald, A.B. Weed Vegetation (Wild Flora) of Long Established Organic versus Conventional Cereal Fields in Denmark. Ann. Appl. Biol. 1999, 134, 307–314. [Google Scholar] [CrossRef]
- Roschewitz, I.; Gabriel, D.; Tscharntke, T.; Thies, C. The Effects of Landscape Complexity on Arable Weed Species Diversity in Organic and Conventional Farming. J. Appl. Ecol. 2005, 42, 873–882. [Google Scholar] [CrossRef]
- Hyvönen, T.; Ketoja, E.; Salonen, J.; Jalli, H.; Tiainen, J. Weed Species Diversity and Community Composition in Organic and Conventional Cropping of Spring Cereals. Agric. Ecosyst. Environ. 2003, 97, 131–149. [Google Scholar] [CrossRef]
- Armengot, L.; Sans, F.X.; Fischer, C.; Flohre, A.; José-María, L.; Tscharntke, T.; Thies, C. The β-Diversity of Arable Weed Communities on Organic and Conventional Cereal Farms in Two Contrasting Regions. Appl. Veg. Sci. 2012, 15, 571–579. [Google Scholar] [CrossRef]
- Fonseca, C.R.; Ganade, G. Species Functional Redundancy, Random Extinctions and the Stability of Ecosystems. J. Ecol. 2001, 89, 118–125. [Google Scholar] [CrossRef]
- Henckel, L.; Börger, L.; Meiss, H.; Gaba, S.; Bretagnolle, V. Organic Fields Sustain Weed Metacommunity Dynamics in Farmland Landscapes. Proc. R. Soc. B Biol. Sci. 2015, 282, 20150002. [Google Scholar] [CrossRef]
- Munoz, F.; Fried, G.; Armengot, L.; Bourgeois, B.; Bretagnolle, V.; Chadoeuf, J.; Mahaut, L.; Plumejeaud, C.; Storkey, J.; Violle, C.; et al. Ecological Specialization and Rarity of Arable Weeds: Insights from a Comprehensive Survey in France. Plants 2020, 9, 824. [Google Scholar] [CrossRef]
- Laliberté, E.; Wells, J.A.; DeClerck, F.; Metcalfe, D.J.; Catterall, C.P.; Queiroz, C.; Aubin, I.; Bonser, S.P.; Ding, Y.; Fraterrigo, J.M.; et al. Land-Use Intensification Reduces Functional Redundancy and Response Diversity in Plant Communities. Ecol. Lett. 2010, 13, 76–86. [Google Scholar] [CrossRef] [PubMed]
- Carmona, C.P.; Guerrero, I.; Peco, B.; Morales, M.B.; Oñate, J.J.; Pärt, T.; Tscharntke, T.; Liira, J.; Aavik, T.; Emmerson, M.; et al. Agriculture Intensification Reduces Plant Taxonomic and Functional Diversity across European Arable Systems. Funct. Ecol. 2020, 34, 1448–1460. [Google Scholar] [CrossRef]
- Melander, B.; Rasmussen, I.A.; Olesen, J.E. Legacy Effects of Leguminous Green Manure Crops on the Weed Seed Bank in Organic Crop Rotations. Agric. Ecosyst. Environ. 2020, 302, 107078. [Google Scholar] [CrossRef]
- Pignatti, S. Flora d’Italia; Edagricole: Bologna, Italy, 1982. [Google Scholar]
- Ellenberg, H. Zeigerwerte Der Pflanzen in Mitteleuropa, 2nd ed.; Erich Goltze KG: Goettingen, Germany, 1979. [Google Scholar]
- Klotz, S.; Kühn, I.; Durka, W.; Briemle, G. Bundesamt für Naturschutz BIOLFLOR: Eine Datenbank Mit Biologisch-Ökologischen Merkmalen zur Flora von Deutschland; BfN-Schriftenvertrieb im Landwirtschaftsverlag: Münster, Germany, 2002; ISBN 978-3-7843-3508-7. [Google Scholar]
- Fitter, A.H.; Peat, H.J. The Ecological Flora Database. J. Ecol. 1994, 82, 415–425. [Google Scholar] [CrossRef]
- Missouri Botanical Garden. Available online: http://www.efloras.org (accessed on 11 May 2016).
- Grime, J.P.; Hodgson, J.C.; Hunt, R. Comparative Plant. Ecology: A Functional Approach to Common British Species, 2nd ed.; Castlepoint Press: Colvend, UK, 2007; ISBN 978-1-897604-30-4. [Google Scholar]
- Seed Information Database: Royal Botanic Gardens, Kew. Available online: http://data.kew.org/sid/ (accessed on 10 April 2021).
- Kleyer, M.; Bekker, R.M.; Knevel, I.C.; Bakker, J.P.; Thompson, K.; Sonnenschein, M.; Poschlod, P.; Groenendael, J.M.V.; Klimeš, L.; Klimešová, J.; et al. The LEDA Traitbase: A Database of Life-History Traits of the Northwest European Flora. J. Ecol. 2008, 96, 1266–1274. [Google Scholar] [CrossRef]
- Hodgson, J. Allocating C-S-R Types to Plant Species. Available online: http://people.exeter.ac.uk/rh203/csr_lookup_table.xls (accessed on 11 May 2016).
- Westoby, M. A Leaf-Height-Seed (LHS) Plant Ecology Strategy Scheme. Plant. Soil 1998, 199, 213–227. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).