Osmolyte Accumulation and Sodium Compartmentation Has a Key Role in Salinity Tolerance of Pistachios Rootstocks
Abstract
:1. Introduction
2. Materials and Methods
3. Results
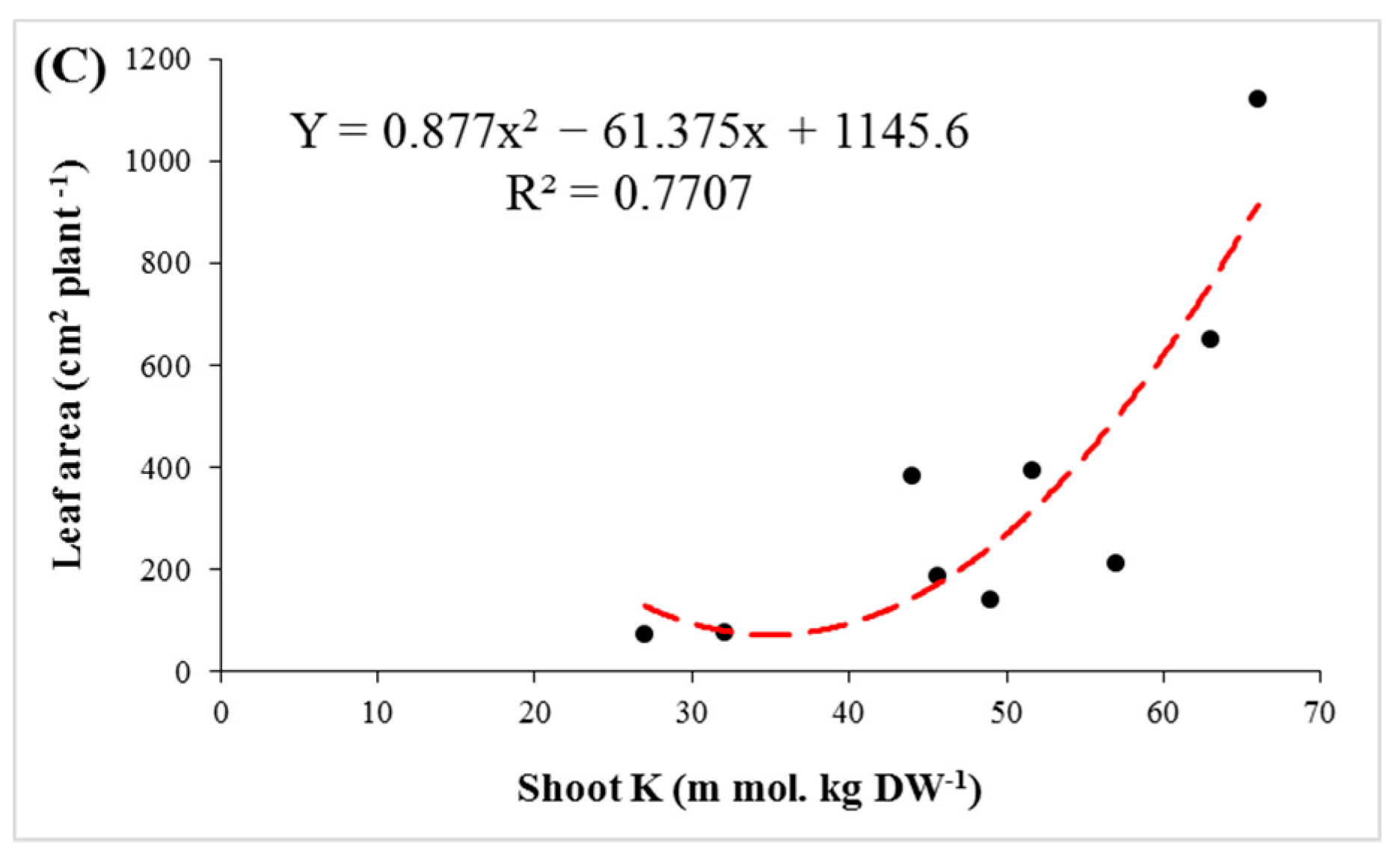
3.1. Plant Growth and Health
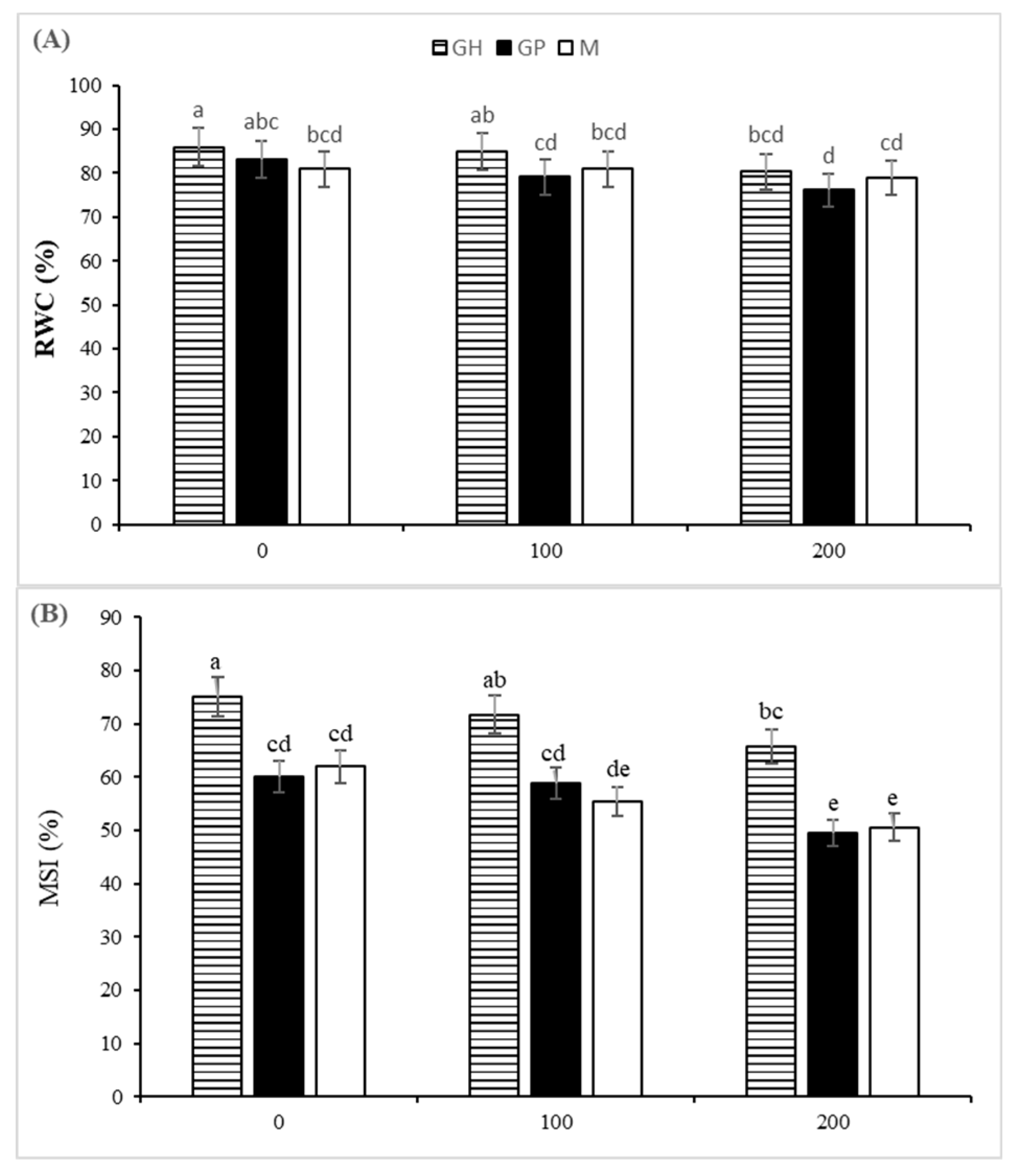
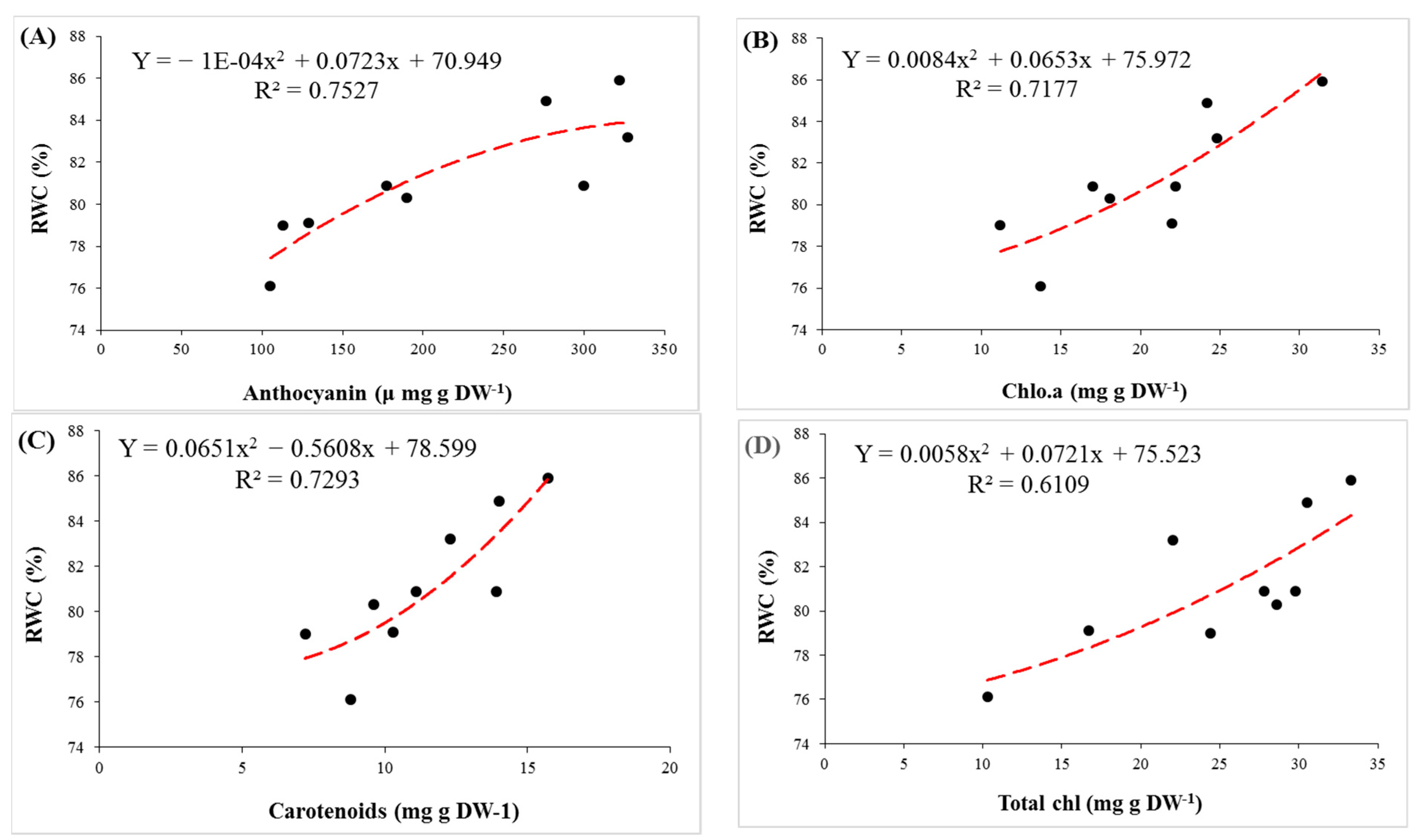
3.2. Physiological Parameters
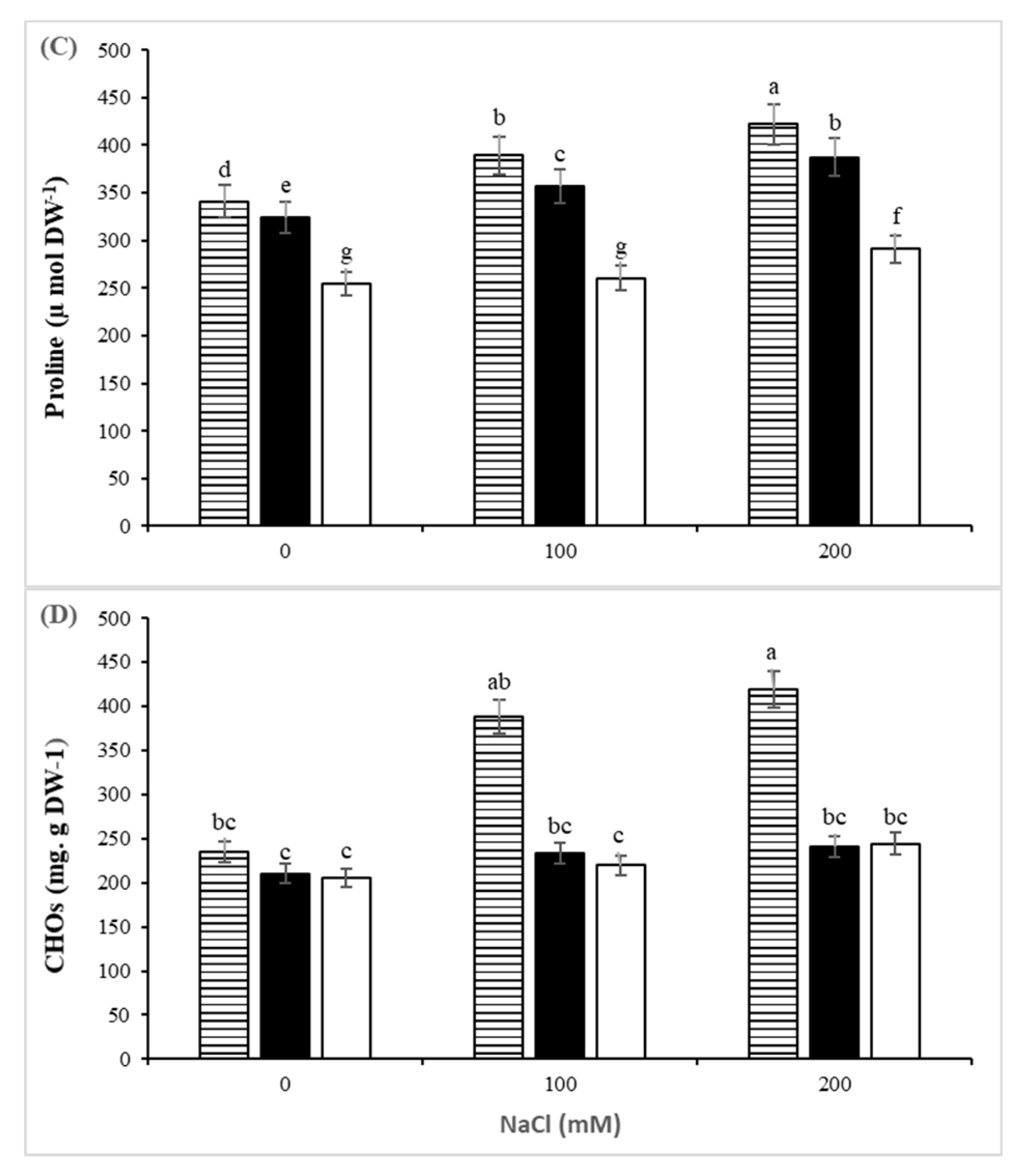
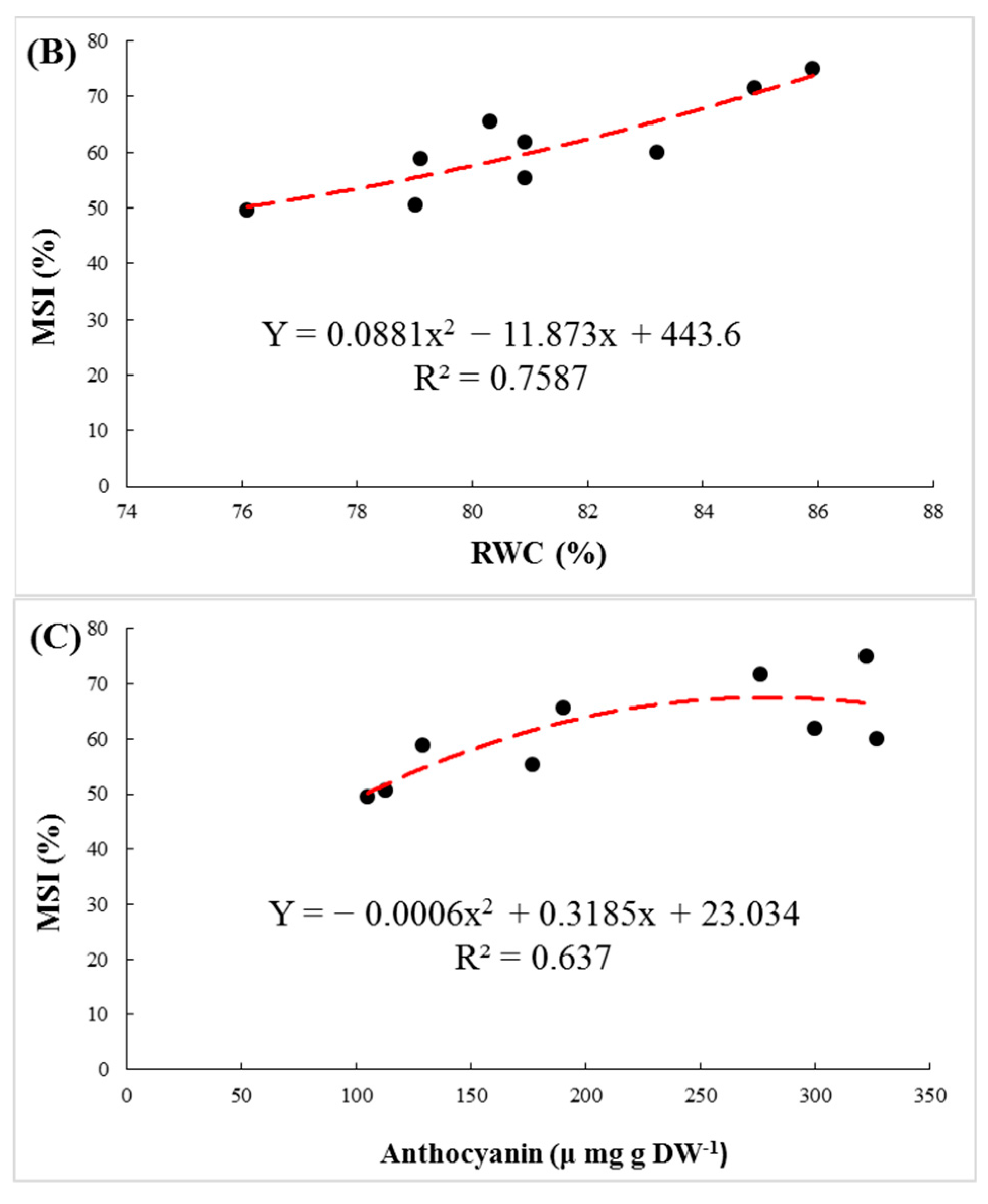
3.3. Biochemical Parameters
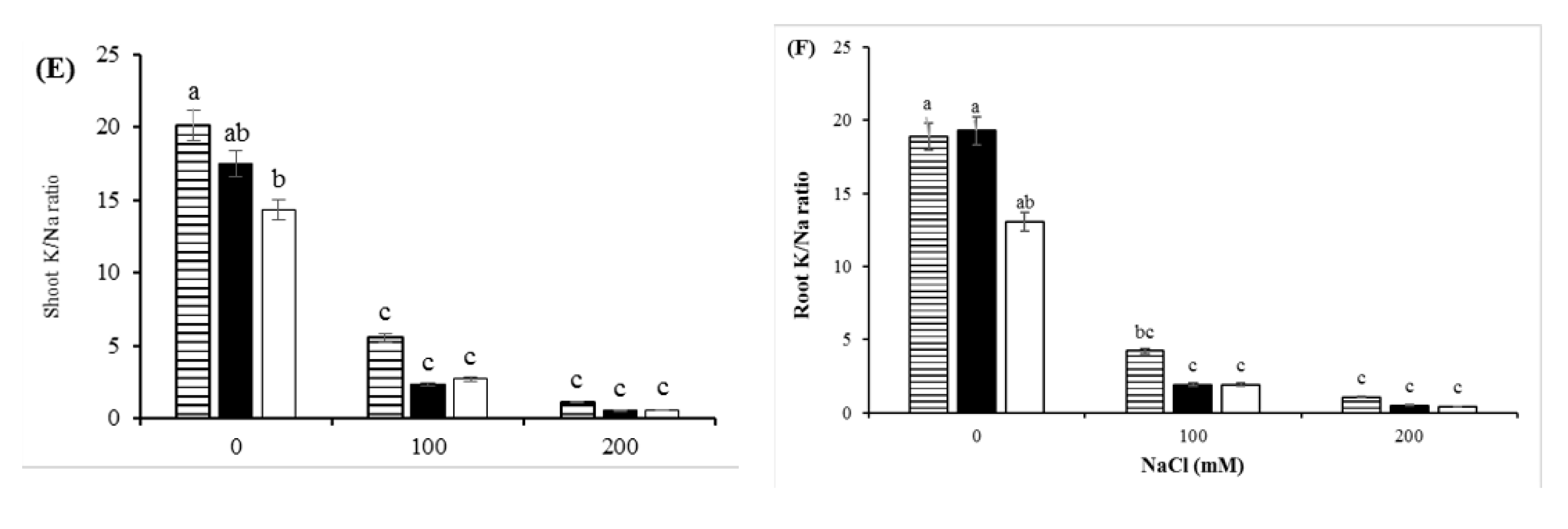
3.4. Ions Distribution
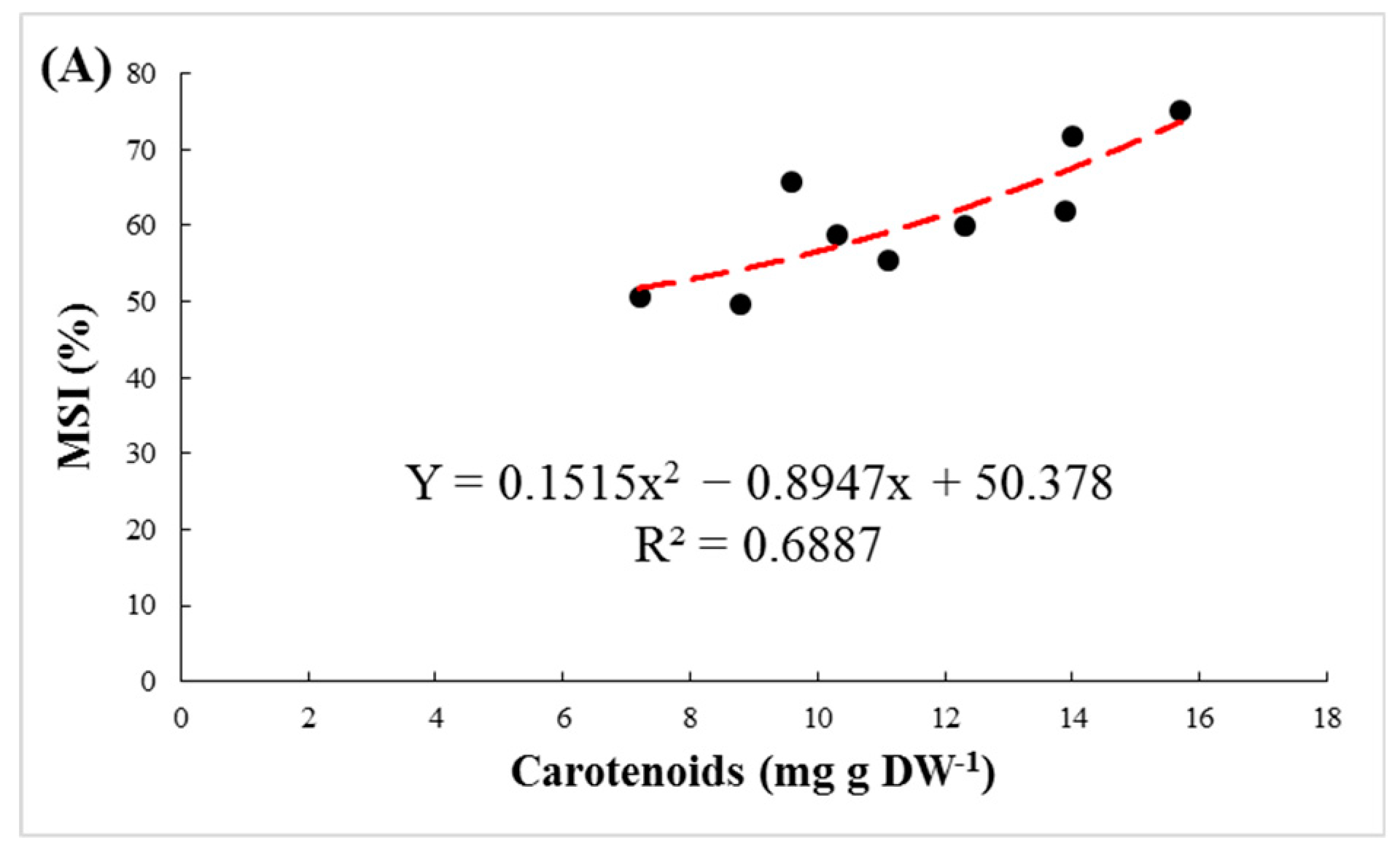
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| Chl | Chlorophyll |
| CHOs | carbohydrates |
| MSI | membrane stability index |
| RWC | relative water content |
| NL | necrotic leaves |
| GP | Pistacia vera ‘Ghermez Pesteh’ |
| GH | P. vera ‘Ghazvini’ |
| M | P. atlantica subsp. mutica |
References
- Singh, A. Soil salinization and waterlogging: A threat to environment and agricultural sustainability. Ecol. Indic. 2015, 57, 128–130. [Google Scholar] [CrossRef]
- Volkov, V.; Beilby, M.J. Salinity Tolerance in Plants: Mechanisms and Regulation of Ion Transport. Front. Plant Sci. 2017, 8, 1795. [Google Scholar]
- Rahneshan, Z.; Nasibi, F.; Moghadam, A.A. Effects of salinity stress on some growth, physiological, biochemical parameters and nutrients in two pistachio (Pistacia vera L.) rootstocks. J. Plant Interact. 2018, 13, 73–82. [Google Scholar] [CrossRef] [Green Version]
- Vahdati, K.; Lotfi, N. Abiotic Stress Tolerance in Plants with Emphasizing on Drought and Salinity Stresses in Walnut. In Abiotic Stress—Plant Responses and Applications in Agriculture; Vahdati, K., Leslie, C., Eds.; InTech: Rijeka, Croatia, 2013; pp. 307–365. [Google Scholar]
- Acosta-Motos, J.R.; Ortuño, M.F.; Bernal-Vicente, A.; Diaz-Vivancos, P.; Sanchez-Blanco, M.J.; Hernandez, J.A. Plant responses to salt stress: Adaptive mechanisms. Agronomy 2017, 7, 18. [Google Scholar] [CrossRef] [Green Version]
- Karimi, S.; Karami, H.; Mokhtassi-Bidgoli, A.; Tavallali, V.; Vahdati, K. Inducing drought tolerance in greenhouse grown Juglans regia by imposing controlled salt stress: The role of osmotic adjustment. Sci. Hortic. 2018, 239, 181–192. [Google Scholar] [CrossRef]
- Akbari, M.; Mahna, N.; Ramesh, K.; Bandehagh, A.; Mazzuca, S. Ion homeostasis, osmoregulation, and physiological changes in the roots and leaves of pistachio rootstocks in response to salinity. Protoplasma 2018, 255, 1349–1362. [Google Scholar] [CrossRef]
- Garriga, M.; Retamales, J.B.; Romero-Bravo, S.; Caligari, P.D.; Lobos, G.A. Chlorophyll, anthocyanin, and gas exchange changes assessed by spectroradiometry in Fragaria chiloensis under salt stress. J. Integr. Plant Biol. 2014, 56, 505–515. [Google Scholar] [CrossRef]
- Nakabayashi, R.; Yonekura-Sakakibara, K.; Urano, K.; Suzuki, M.; Yamada, Y.; Nishizawa, T.; Michael, A.J. Enhancement of oxidative and drought tolerance in Arabidopsis by overaccumulation of antioxidant flavonoids. Plant J. 2014, 77, 367–379. [Google Scholar] [CrossRef] [PubMed]
- Munns, R.; Tester, M. Mechanisms of salinity tolerance. Ann. Rev. Plant Biol. 2008, 59, 651–681. [Google Scholar] [CrossRef] [Green Version]
- Genc, Y.; Oldach, K.; Taylor, J.; Lyons, G.H. Uncoupling of sodium and chloride to assist breeding for salinity tolerance in crops. New Phytol. 2016, 210, 145–156. [Google Scholar] [CrossRef] [Green Version]
- Taiz, L.; Zeiger, E. Plant Physiology, 5th ed.; Sinauer Associates: Sunderland, MA, USA, 2010. [Google Scholar]
- Chen, Z.; Pottosin, I.; Cuin, T.A.; Fuglsang, A.T.; Tester, M.; Jha, D.; Zepeda-Jazo, I.; Zhou, M.; Palmgren, M.G.; Newman, I.A.; et al. Root plasma membrane transporters controlling K+/Na+ homeostasis in salt-stressed barley. Plant Physiol. 2007, 145, 1714–1725. [Google Scholar] [CrossRef] [Green Version]
- Shabala, S.; Cuin, T.A. Potassium transport and plant salt tolerance. Physiol. Plant. 2008, 133, 651–669. [Google Scholar] [CrossRef]
- Hajiboland, R.; Norouzi, F.; Poschenrieder, C. Growth, physiological, biochemical and ionic responses of pistachio seedlings to mild and high salinity. Trees 2014, 28, 1065–1078. [Google Scholar] [CrossRef]
- Hokmabadi, H.; Arzani, K.; Grierson, P.F. Growth, chemical composition, and carbon isotope discrimination of pistachio (Pistacia vera L.) rootstock seedlings in response to salinity. Aust. J. Agric. Res. 2005, 56, 135–144. [Google Scholar] [CrossRef]
- Karimi, S.; Rahemi, M.; Maftoun, M.; Tavallali, V. Effects of long-term salinity on growth and performance of two pistachio (Pistacia L.) rootstocks. Aust. J. Basic Appl. Sci. 2009, 3, 1630–1639. [Google Scholar]
- Nadernejad, N.; Ahmadimoghadam, A.; Hossyinifard, J.; Poorseyedi, S. Effect of different rootstocks on PAL activity and phenolic compounds in flowers, leaves, hulls and kernels of three pistachio (Pistacia vera L.) cultivars. Trees 2013, 27, 1681–1689. [Google Scholar] [CrossRef]
- Zeng, L.; Tu, X.L.; Dai, H.; Han, F.M.; Lu, B.S.; Wang, M.S.; Nanaei, H.A.; Tajabadipour, A.; Mansouri, M.; Li, X.L.; et al. Whole genomes and transcriptomes reveal adaptation and domestication of pistachio. Genome Biol. 2019, 20, 79. [Google Scholar] [CrossRef]
- Momenpour, A.; Imani, A. Evaluation of salinity tolerance in fourteen selected pistachio (Pistacia vera L.) cultivars. Adv. Hortic. Sci. 2018, 32, 249–264. [Google Scholar]
- Jamshidi, K.G.; Amirmahani, F.; Salehi, F. Assessment of changes in physiological and biochemical traits in four pistachio rootstocks under drought, salinity and drought+ salinity stresses. Physiol. Plant. 2020, 168, 973–989. [Google Scholar] [CrossRef]
- Akbari, M.; Katam, R.; Husain, R.; Farajpour, M.; Mazzuca, S.; Mahna, N. Sodium Chloride Induced Stress Responses of Antioxidative Activities in Leaves and Roots of Pistachio Rootstock. Biomolecules 2020, 10, 189. [Google Scholar] [CrossRef] [Green Version]
- Mirfattahi, Z.; Karimi, S.; Roozban, M.R. Salinity induced changes in water relations, oxidative damage and morpho-physiological adaptations of pistachio genotypes in soilless culture. Acta Agric. Slov. 2017, 109, 291–302. [Google Scholar] [CrossRef] [Green Version]
- Raoufi, A.; Rahemi, M.; Salehi, H.; Javanshah, A. Selecting high-performance rootstocks for pistachio cultivars under salinity stress based on their morpho-physiological characteristics. Int. J. Fruit Sci. 2019, 20 (Suppl. 2), S29–S47. [Google Scholar] [CrossRef]
- Raoufi, A.; Rahemi, M.; Salehi, H.; Pessarakli, M. Pistacia vera L. genotypes; a potential rival for UCB-1 rootstock for cultivating under salt stress conditions. Biocatal. Agric. Biotechnol. 2020, 25, 101515. [Google Scholar] [CrossRef]
- Karimi, S.; Tavallali, V.; Rahemi, M.; Rostami, A.A.; Vaezpour, M. Estimation of leaf growth on the basis of measurements of leaf hights and widths, choosing pistachio seedlings as model. Aust. J. Basic Appl. Sci. 2009, 3, 1070–1075. [Google Scholar]
- Whetherley, P.E. Studies in the water relations of cotton plants. I. The field measurement of water deficit in leaves. New Phytol. 1950, 49, 81–87. [Google Scholar] [CrossRef]
- Bates, L.S.; Waldren, R.P.; Teare, I.D. Rapid determination of free proline for water-stress studies. Plant Soil 1973, 39, 205–207. [Google Scholar] [CrossRef]
- Grieve, C.M.; Grattan, S.R. Rapid assay for determination of water soluble quaternary ammonium compounds. Plant Soil 1983, 70, 303–307. [Google Scholar] [CrossRef]
- Vandoorne, B.; Mathieu, A.S.; Van den Ende, W.; Vergauwen, R.; Périlleux, C.; Javaux, M.; Lutts, S. Water stress drastically reduces root growth and inulin yield in Cichorium intybus var. sativum independently of photosynthesis. J. Exp. Bot. 2012, 63, 4359–4373. [Google Scholar] [CrossRef]
- Wagner, G.R. Content and vacuole/extravacuole distribution of neutral sugars, free amino acids, and anthocyanin in protoplasts. Plant physiol. 1979, 64, 88–93. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lichtenthaler, H.K. Chlorophylls and carotenoids: Pigments of photosynthetic biomembranes. Methods Enzymol. 1987, 148, 350–382. [Google Scholar]
- Zhu, J.K.; Liu, J.; Xiong, L. Genetic analysis of salt tolerance in Arabidopsis: Evidence for a critical role of potassium nutrition. Plant Cell. 1998, 10, 1181–1191. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Läuchli, A.; Grattan, S.R. Plant Growth and Development under Salinity Stress. Advances in Molecular Breeding Toward Drought and Salt Tolerant Crops. Springer: Dordrecht, The Netherland, 2007; pp. 1–32. [Google Scholar]
- Negrão, S.; Schmöckel, S.M.; Tester, M. Evaluating physiological responses of plants to salinity stress. Ann. Bot. 2017, 119, 1–11. [Google Scholar] [CrossRef] [Green Version]
- Parida, A.K.; Das, A.B. Salt tolerance and salinity effects on plants: A review. Ecotoxicol. Environ. Saf. 2005, 60, 324–349. [Google Scholar] [CrossRef] [PubMed]
- Sairam, R.K.; Rao, K.V.; Srivastava, G.C. Differential response of wheat rootstocks to long term salinity stress in relation to oxidative stress, antioxidant activity and osmolyte concentration. Plant Sci. 2002, 163, 1037–1046. [Google Scholar] [CrossRef]
- Karimi, S.; Mirfattahi, Z.; Ferguson, L.; Tavallali, V. Using controlled salt stress and β-aminobutyric acid signaling to decrease transplant failure. Sci. Hortic. 2017, 225, 156–162. [Google Scholar] [CrossRef]
- Ben-Romdhane, W.; Ben-Saad, R.; Meynard, D.; Zouari, N.; Mahjoub, A.; Fki, L.; Guiderdoni, E.; Al-Doss, A.; Hassairi, A. Overexpression of AlTMP2 gene from the halophyte grass Aeluropus littoralis in transgenic tobacco enhances tolerance to different abiotic stresses by improving membrane stability and deregulating some stress-related genes. Protoplasma 2018, 255, 1161–1177. [Google Scholar] [CrossRef] [PubMed]
- Tufail, A.; Li, H.; Naeem, A.; Li, T.X. Leaf cell membrane stability-based mechanisms of zinc nutrition in mitigating salinity stress in rice. Plant Biol. 2018, 20, 338–345. [Google Scholar] [CrossRef]
- Verbruggen, N.; Hermans, C. Proline accumulation in plants: A review. Amino Acids 2008, 35, 753–759. [Google Scholar] [CrossRef]
- Hayat, S.; Hayat, Q.; Alyemeni, M.N.; Wani, A.S.; Pichtel, J.; Ahmad, A. Role of proline under changing environments: A review. Plant Signal. Behav. 2012, 7, 1456–1466. [Google Scholar] [CrossRef] [Green Version]
- Kamiab, F.; Talaie, A.; Khezri, M.; Javanshah, A. Exogenous application of free polyamines enhance salt tolerance of pistachio (Pistacia vera L.) seedlings. Plant Growth Regul. 2014, 72, 257–268. [Google Scholar] [CrossRef]
- Abbaspour, H.; Afshari, H.; Abdel-Wahhab, M.A. Influence of salt stress on growth, pigments, soluble sugars and ion accumulation in three pistachio cultivars. J. Med. Plant Res. 2012, 6, 2468–2473. [Google Scholar] [CrossRef]
- Ashraf, M.H.P.J.C.; Harris, P.J.C. Photosynthesis under stressful environments: An overview. Photosynthetica 2013, 51, 163–190. [Google Scholar] [CrossRef]
- Rasool, S.; Ahmad, A.; Siddiqi, T.O.; Ahmad, P. Changes in growth, lipid peroxidation and some key antioxidant enzymes in chickpea rootstocks under salt stress. Acta Physiol. Plant. 2013, 35, 1039–1050. [Google Scholar] [CrossRef]
- Abdallah, S.B.; Aung, B.; Amyot, L.; Lalin, I.; Lachâal, M.; Karray-Bouraoui, N.; Hannoufa, A. Salt stress (NaCl) affects plant growth and branch pathways of carotenoid and flavonoid biosyntheses in Solanum nigrum. Acta Physiol. Plant. 2016, 38, 72. [Google Scholar] [CrossRef]
- Havaux, M. Carotenoid oxidation products as stress signals in plants. Plant J. 2014, 79, 597–606. [Google Scholar] [CrossRef]
- Gould, K.S.; McKelvie, J.; Markham, K.R. Do anthocyanins function as antioxidants in leaves? Imaging of H2O2 in red and green leaves after mechanical injury. Plant Cell Environ. 2002, 25, 1261–1269. [Google Scholar] [CrossRef]
- Jiang, Y.; Deyholos, M.K. Comprehensive transcriptional profiling of NaCl-stressed Arabidopsis roots reveals novel classes of responsive genes. BMC Plant Biol. 2006, 6, 25. [Google Scholar] [CrossRef] [Green Version]
- Cassaniti, C.; Leonardi, C.; Flowers, T.J. The effects of sodium chloride on ornamental shrubs. Sci. Hortic. 2009, 122, 586–593. [Google Scholar] [CrossRef]
- Olivier, S.; Zhang, W.Q.; Zwiazek, J.J. Tissue sodium and chloride concentrations in relation to needle injury in boreal conifer seedlings subjected to salt stress. Trees 2020, 34, 521–529. [Google Scholar] [CrossRef]
- Flowers, T.J.; Munns, R.; Colmer, T.D. Sodium chloride toxicity and the cellular basis of salt tolerance in halophytes. Ann. Bot. 2014, 115, 419–431. [Google Scholar] [CrossRef] [Green Version]
- Mahajan, S.; Tuteja, N. Cold, salinity and drought stresses: An overview. Arch. Biochem. Biophys. 2005, 444, 139–158. [Google Scholar] [CrossRef] [PubMed]
- Tuteja, N. Mechanisms of high salinity tolerance in plants. Methods Enzymol. 2007, 428, 419–438. [Google Scholar] [PubMed]
- Isayenkov, S.V.; Maathuis, F.J. Plant salinity stress: Many unanswered questions remain. Front. Plant Sci. 2019, 10, 80. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, M.; Zheng, Q.; Shen, Q.; Guo, S. The critical role of potassium in plant stress response. Int. J. Mol. Sci. 2013, 14, 7370–7390. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cakmak, I. The role of potassium in alleviating detrimental effects of abiotic stresses in plants. J. Plant Nutr. Soil Sci. 2005, 168, 521–530. [Google Scholar] [CrossRef]
- Wu, H.; Shabala, L.; Shabala, S.; Giraldo, J.P. Hydroxyl radical scavenging by cerium oxide nanoparticles improves Arabidopsis salinity tolerance by enhancing leaf mesophyll potassium retention. Environ. Sci. Nano. 2018, 5, 1567–1583. [Google Scholar] [CrossRef]
- Forouzi, A.; Ghasemnezhad, A.; Ghorbani Nasrabad, R. Effects of growth stimulator microbes on growth and ions concentration of stevia under salinity stress conditions. Int. J. Hortic. Sci. Technol. 2019, 6, 217–236. [Google Scholar]
- Aboutalebi Jahromi, A.; Hosseini Farahi, M. Seed germination, vegetative growth and concentration of some elements in French marigold (Tageta patula) as influenced by salinity and ammonium nitrate. Int. J. Hortic. Sci. Technol. 2016, 3, 199–209. [Google Scholar]



| Rootstock | NaCl (mM) | Shoot Height (cm) | Green Leaves (Number) | Necrotic Leaves (%) | Fallen Leaves (Number) | Plant Leaf Area (cm2) |
|---|---|---|---|---|---|---|
| 0 | 28.0 a | 47.0 a | 10.8 h | 12.3 c | 1123.4 a | |
| GH | 100 | 25.0 ab | 38.3 ab | 16.5 f | 15.3 bc | 652.2 b |
| 200 | 22.3 abc | 30.6 bc | 17.5 e | 20.3 abc | 384.0 c | |
| 0 | 20.6 bcd | 18.6 de | 14.1 g | 26.6 abc | 213.9 d | |
| GP | 100 | 17.3 cd | 12.6 ef | 21.9 d | 30.3 ab | 142.6 f |
| 200 | 15.3 d | 7.6 f | 24.5 c | 36.6 a | 76.0 g | |
| 0 | 16.3 cd | 32.0 bc | 14.8 g | 16.6 bc | 396.2 c | |
| M | 100 | 15.0 d | 24.3 de | 26.8 b | 24.3 abc | 189.2 e |
| 200 | 14.6 d | 14.3 ef | 28.6 a | 36.0 a | 72.5 g |
| Rootstock | NaCl (mM) | Chl. a (mg g FW−1) | Chl. a/b Ratio | Total Chl. (mg g FW−1) | Carotenoids (mg g FW−1) | Anthocyanin (µmol g FW−1) |
|---|---|---|---|---|---|---|
| 0 | 31.4 a | 22.8 ab | 33.3 a | 15.7 a | 322 a | |
| GH | 100 | 24.2 b | 19.4 abc | 30.5 ab | 14.1 ab | 276 c |
| 200 | 13.7 ef | 15.6 cd | 28.6 ab | 9.6 ce | 189.6 d | |
| 0 | 24.8 b | 24.6 a | 22 bc | 13.3 abc | 327.8 a | |
| GP | 100 | 22 bc | 17.5 bcd | 16.7 cd | 10.3 bcde | 129 f |
| 200 | 18.1 cd | 9.4 e | 10.3 d | 8.8 de | 105.3 g | |
| 0 | 22.2 bc | 21.4 abc | 29.8 ab | 13.9 ab | 300 b | |
| M | 100 | 17 de | 17.5 bc | 27.8 ab | 12.1 abcd | 177 e |
| 200 | 11.2 f | 12 de | 24.4 ab | 7.2 e | 113 g |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Behzadi Rad, P.; Roozban, M.R.; Karimi, S.; Ghahremani, R.; Vahdati, K. Osmolyte Accumulation and Sodium Compartmentation Has a Key Role in Salinity Tolerance of Pistachios Rootstocks. Agriculture 2021, 11, 708. https://doi.org/10.3390/agriculture11080708
Behzadi Rad P, Roozban MR, Karimi S, Ghahremani R, Vahdati K. Osmolyte Accumulation and Sodium Compartmentation Has a Key Role in Salinity Tolerance of Pistachios Rootstocks. Agriculture. 2021; 11(8):708. https://doi.org/10.3390/agriculture11080708
Chicago/Turabian StyleBehzadi Rad, Pegah, Mahmoud Reza Roozban, Soheil Karimi, Reza Ghahremani, and Kourosh Vahdati. 2021. "Osmolyte Accumulation and Sodium Compartmentation Has a Key Role in Salinity Tolerance of Pistachios Rootstocks" Agriculture 11, no. 8: 708. https://doi.org/10.3390/agriculture11080708
APA StyleBehzadi Rad, P., Roozban, M. R., Karimi, S., Ghahremani, R., & Vahdati, K. (2021). Osmolyte Accumulation and Sodium Compartmentation Has a Key Role in Salinity Tolerance of Pistachios Rootstocks. Agriculture, 11(8), 708. https://doi.org/10.3390/agriculture11080708