Responses of Soybean to Selected Abiotic Stresses—Photoperiod, Temperature and Water
Abstract
:1. Introduction
2. Light Requirements
3. Thermal Stress
4. Water Stress
5. Summary
Author Contributions
Funding
Conflicts of Interest
References
- Sun, J.; Mooney, H.; Wu, W.; Tang, H.; Tong, Y.; Xu, Z.; Huang, B.; Cheng, Y.; Yang, X.; Wei, D.; et al. Importing food damages domestic environment: Evidence from global soybean trade. Proc. Natl. Acad. Sci. USA 2018, 115, 5415–5419. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kotecki, A.; Lewandowska, S. Studia nad Uprawą soi Zwyczajnej (Glycine max (L.) Merrill) w Południowo-Zachodniej Polsce/Studies on the Cultivation of Soybean (Glycine max (L.) Merrill) in South-Western Poland; Wyd. UP Wrocław: Wrocław, Poland, 2020; p. 226. (In Polish) [Google Scholar]
- Święcicki, W.; Chudy, M.; Żuk-Gołaszewska, K. Rośliny strączkowe w projektach badawczych Unii Europejskiej. Zesz. Probl. Post. Nauk Rol. 2007, 522, 55–65. (In Polish) [Google Scholar]
- Bellaloui, N.; Bruns, H.A.; Abbas, H.K.; Mengistu, A.; Fisher, D.K.; Reddy, K.N. Agricultural practices altered soybean seed protein, oil, fatty acids, sugars, and minerals in the Midsouth USA. Front. Plant Sci. 2015, 6, 1–14. [Google Scholar] [CrossRef]
- SoyStats 2021. Available online: http://www.soystats.com (accessed on 15 July 2021).
- Szpunar-Krok, E.; Wondołowska-Grabowska, A.; Bobrecka-Jamro, D.; Jańczak-Pieniążek, M.; Kotecki, A.; Kozak, M. Effect of nitrogen fertilisation and inoculation with Bradyrhizobium japonicum on the fatty acid profile of soybean (Glycine max (L.) Merrill) seeds. Agronomy 2021, 11, 941. [Google Scholar] [CrossRef]
- Szpunar-Krok, E.; Wondołowska-Grabowska, A. Quality evaluation indices for soybean oil in relation to cultivar, application of N fertiliser and seed inoculation with Bradyrhizobium japonicum. Foods 2022, 11, 762. [Google Scholar] [CrossRef] [PubMed]
- Borawska, J.; Darewicz, M.; Iwaniak, A.; Minkiewicz, P. Biologically active peptides from food proteins as factors preventing diet-related diseases/Biologicznie aktywne peptydy pochodzące z białek żywności jako czynniki prewencji wybranych chorób dietozależnych. Bromat. Chem. Toksykol. 2014, 47, 230–236. Available online: https://www.ptfarm.pl/download/?file=File%2FBromatologia%2F2014%2FBR+2-2014+s_+230-236.pdf (accessed on 2 August 2022). (In Polish).
- Martyniuk, S. Naukowe i praktyczne aspekty symbiozy roślin strączkowych z bakteriami brodawkowymi/Scientific and practical aspects of legumes symbiosis with root-nodule bacteria. Pol. J. Agron. 2012, 9, 17–22. Available online: https://iung.pl/PJA/wydane/9/PJA9_3.pdf (accessed on 2 August 2022). (in Polish).
- Kocira, A.; Staniak, M.; Tomaszewska, M.; Kornas, R.; Cymerman, J.; Panasiewicz, K.; Lipińska, H. legume cover crops as one of the elements of strategic weed management and soil quality improvement. A Review. Agriculture 2020, 10, 394. [Google Scholar] [CrossRef]
- Bethlenfalvay, J.G.; Franson, L.R.; Brown, S.M. Nutrition of mycorrhizal soybean evaluated by the diagnosis and recommendation integrated system (DRIS). Agron. J. 1990, 82, 302–304. [Google Scholar] [CrossRef]
- Śliwa, J.; Zając, T.; Oleksy, A.; Klimek-Kopyra, A.; Lorenc-Kozik, A.; Kulig, B. Comparison of the development and productivity of soybean (Glycine max (L.) Merr.) cultivated in western Poland. Acta Sci. Pol. Sec. Agricultura 2015, 14, 81–95. [Google Scholar]
- Badaruddin, M.; Meyer, D.W. Grain legume effects on soil nitrogen, grain yield, and nitrogen nutrition of wheat. Crop Sci. 1994, 34, 1304–1309. [Google Scholar] [CrossRef]
- FAOSTAT. 2022. Available online: http://www.fao.org/faostat/en/#data/QC (accessed on 15 July 2022).
- STATISTA. 2022. Available online: https://www.statista.com/search/?q=soybean (accessed on 15 July 2022).
- Ritchie, H.; Roser, M. Soy. Published Online at OurWorldInData.org. 2021. Available online: https://ourworldindata.org/soy (accessed on 21 December 2022).
- EUROStat. 2022. Available online: http://ec.europa.eu/eurostat/de (accessed on 20 July 2022).
- Kuepper, B.; Stravens, M. Mapping the European Soy Supply Chain—Embedded Soy in Animal Products Consumed in the EU27+UK; Profundo: Amsterdam, The Netherlands, 2022; Available online: https://wwfeu.awsassets.panda.org/downloads/2021_106_european_soy_supply_wnf_2201_final.pdf (accessed on 23 December 2022).
- Gaynor, L.G.; Lawn, R.J.; James, A.T. Agronomic studies on irrigated soybean in southern New South Wales. I. Phenological adaptation of genotypes to sowing date. Crop Pasture Sci. 2011, 62, 1056–1066. [Google Scholar] [CrossRef]
- Câmara, G.M.S.; Sediyama, T.; Dourado-Neto, D.; Bernardes, M.S. Influence of fotoperiod and air temperature on the growth, flowering and maturation of the soybean (Glycine max. L. Merrill). Sci. Agric. 1997, 54, 149–154. Available online: https://www.scienceopen.com/document?vid=f604f4c0-fa07-4018-afaa-ff18d7c9b5aa (accessed on 2 August 2022). [CrossRef]
- Gass, T.; Schori, A.; Fossati, A.; Soldati, A.; Stamp, P. Cold tolerance of soybean (Glycine max. L. Merrill) during the reproductive phase. Eur. J. Agron. 1996, 5, 71–88. [Google Scholar] [CrossRef]
- Gawęda, D.; Nowak, A.; Haliniarz, M.; Woźniak, A. Yield and economic effectiveness of soybean grown under different cropping systems. Int. J. Plant Prod. 2020, 14, 475–485. [Google Scholar] [CrossRef] [Green Version]
- Kühling, I.; Hüsing, B.; Bome, N.; Trautz, D. Soybeans in high latitudes: Effects of Bradyrhizobium inoculation in Northwest Germany and southern West Siberia. Org. Agric. 2018, 8, 159–171. [Google Scholar] [CrossRef]
- Żarski, J.; Kuśmierek-Tomaszewska, R.; Dudek, S.; Krokowski, M.; Kledzik, R. Identifying climatic risk to soybean cultivation in the transitional type of moderate climate in Central Poland. J. Cent. Eur. Agric. 2019, 20, 143–156. [Google Scholar] [CrossRef]
- Karges, K.; Bellingrath-Kimura, S.D.; Watson, C.A.; Stoddard, F.L.; Halwani, M.; Reckling, M. Agro-economic prospects for expanding soybean production beyond its current northerly limit in Europe. Eur. J. Agron. 2022, 133, 126415. [Google Scholar] [CrossRef]
- Chaves, M.M.; Oliveira, M.M. Mechanisms underlying plant resilience to water deficits: Prospects for water-saving agriculture. J. Exp. Bot. 2004, 407, 2365–2379. [Google Scholar] [CrossRef] [Green Version]
- Khan, M.; Jannat, A.; Munir, F.; Fatima, N.; Amir, R. Biochemical and molecular mechanisms of abiotic stress tolerance. In Plant Ecophysiology and Adaptation under Climate Change: Mechanisms and Perspectives II; Hasanuzzaman, M., Ed.; Springer: Singapore, 2020; ISBN 978-981-15-2171-3. Available online: https://link.springer.com/book/10.1007/978-981-15-2172-0 (accessed on 23 December 2022).
- Choudhury, F.K.; Rivero, R.M.; Blumwald, E.; Mittler, R. Reactive oxygen species, abiotic stress and stress combination. Plant J. 2017, 90, 856–867. [Google Scholar] [CrossRef]
- de Melo, B.P.; Carpinetti, P.d.A.; Fraga, O.T.; Rodrigues-Silva, P.L.; Fioresi, V.S.; de Camargos, L.F.; Ferreira, M.F.d.S. Abiotic stresses in plants and their markers: A practice view of plant stress responses and programmed cell death mechanisms. Plants 2022, 11, 1100. [Google Scholar] [CrossRef]
- He, M.; He, C.-Q.; Ding, N.-Z. Abiotic Stresses: General defenses of land plants and chances for engineering multistress tolerance. Front. Plant Sci. 2018, 9, 1771. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hou, G.; Ablett, G.R.; Pauls, K.P.; Rajcan, I. Environmental Effects on Fatty Acid Levels in Soybean Seed Oil. J. Am. Oil Chem. Soc. 2006, 83, 759–763. [Google Scholar] [CrossRef]
- Cramer, G.R.; Urano, K.; Delrot, S.; Pezzotti, M.; Shinozaki, K. Effects of abiotic stress on plants: A systems biology perspective. BMC Plant Biol. 2011, 11, 163. [Google Scholar] [CrossRef] [PubMed]
- Gupta, P.; Sharma, R.; Sharma, M.K.; Sharma, M.P.; Satpute, G.K.; Garg, S.; Singla-Pareek, S.L.; Pareek, A. Signaling cross talk between biotic and abiotic stress responses in soybean. In Abiotic and Biotic Stresses in Soybean Production; Miransari, M., Ed.; Academic Press: Oxford, UK, 2016; pp. 27–52. [Google Scholar] [CrossRef]
- Wang, L.; Liu, L.; Ma, Y.; Li, S.; Dong, S.; Zu, W. Transcriptome profiling analysis characterized the gene expression patterns responded to combined drought and heat stresses in soybean. Comput. Biol. Chem. 2018, 77, 413–429. [Google Scholar] [CrossRef]
- Jumrani, K.; Bhatia, V.S. Interactive effect of temperature and water stress on physiological and biochemical processes in soybean. Physiol. Mol. Biol. Plants. 2019, 25, 667–681. [Google Scholar] [CrossRef] [PubMed]
- Haak, D.C.; Fukao, T.; Grene, R.; Hua, Z.; Ivanov, R.; Perrella, G.; Li, S. Multilevel regulation of abiotic stress responses in plants. Front. Plant Sci. 2017, 8, 1564. [Google Scholar] [CrossRef]
- Cooper, R.L. A delayed flowering barrier to higher soybean yields. Field Crop Res. 2003, 82, 27–35. [Google Scholar] [CrossRef]
- Casal, J.J. Photoreceptor signaling networks in plant responses to shade. Annu. Rev. Plant Biol. 2013, 64, 403–427. Available online: http://ri.agro.uba.ar/files/download/articulo/2013casal1.pdf (accessed on 10 December 2022). [CrossRef] [PubMed]
- Galvão, V.C.; Fankhauser, C. Sensing the light environment in plants: Photoreceptors and early signaling steps. Curr. Opin. Neurobiol. 2015, 34, 46–53. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ji, H.; Xiao, R.; Lyu, X.; Chen, J.; Zhang, X.; Wang, Z.; Deng, Z.; Wang, Y.; Wang, H.; Li, R.; et al. Differential light-dependent regulation of soybean nodulation by papilionoid—Specific HY5 homologs. Curr. Biol. 2022, 32, 783–795.e5. [Google Scholar] [CrossRef]
- Osterlund, C.M.T.; Hardtke, S.; Wei, N.; Deng, X.W. Targeted destabilization of HY5 during light-regulated development of Arabidopsis. Nature 2000, 405, 462–466. [Google Scholar] [CrossRef]
- Heng, Y.; Jiang, Y.; Zhao, X.; Zhou, H.; Wang, X.; Deng, X.W.; Xu, D. BBX4, a phyB-interacting and modulated regulator, directly interacts with PIF3 to fine tune red light-mediated photomorphogenesis. Proc. Natl. Acad. Sci. USA 2020, 117, 4429–4430. [Google Scholar] [CrossRef] [PubMed]
- Iqbalm, Z.; Iqbal, M.S.; Hashemmm, A.; Abd_Allah, E.F.; Mi, A. Plant defense responses to biotic stress and its interplay with fluctuating dark/light conditions. Front. Plant Sci. 2021, 12, 631810. [Google Scholar] [CrossRef]
- Malik, N.S.A.; Pence, M.K.; Calvert, H.E.; Bauer, W.D.; Kettering, C.F. Rhizobium infection and nodule development in soybean are affected by exposure of the cotyledons to light. Plant Physiol. 1984, 75, 90–94. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, T.; Guo, J.; Peng, Y.; Lyu, X.; Liu, B.; Sun, S.; Wang, X. Light-induced mobile factors from shoots regulate rhizobium-triggered soybean root nodulation. Science 2021, 374, 65–71. [Google Scholar] [CrossRef]
- Chattopadhyay, S.; Ang, L.H.; Puente, P.; Deng, X.-W.; Wei, N. Arabidopsis bZIP protein HY5 directly interacts with light-responsive promoters in mediating light control of gene expression. Plant Cell 1998, 10, 673–683. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, X.; Yao, Q.; Gao, X.; Jiang, C.; Harberd, N.P.; Fu, X. Shoot-to-root mobile transcription factor HY5 coordinates plant carbon and nitrogen acquisition. Curr. Biol. 2016, 26, 640–646. [Google Scholar] [CrossRef] [Green Version]
- van Gelderen, K.; Kang, C.; Paalman, R.; Keuskamp, D.; Hayes, S.; Pierik, R. Far-red light detection in the shoot regulates lateral root development through the HY5 transcription factor. Plant Cell 2018, 30, 101–116. [Google Scholar] [CrossRef] [Green Version]
- Garner, W.W.; Allard, H.A. Effect of the relative length of day and night and other factors of the environment on growth and reproduction in plants. J. Agric. Res. 1920, 48, 553–606. [Google Scholar] [CrossRef]
- Garner, W.W.; Allard, H.A. Further studies in photoperiodism. The response of the plant to relative length of day and night. J. Agric. Res. 1923, 23, 871–920. [Google Scholar]
- Zhang, L.; Wang, R.; Hesketh, J.D. Effects of photoperiod on growth and development of soybean floral bud in different maturity. Agron J. 2001, 93, 944–948. [Google Scholar] [CrossRef]
- Wu, T.; Li, J.; Wu, C.; Sun, S.; Mao, T.; Jiang, B.; Hou, W.; Han, T. Analysis of the independent and interactive photo thermal effects on soybean flowering. J. Integr. Agric. 2015, 14, 622–632. [Google Scholar] [CrossRef] [Green Version]
- Kantolic, A.G.; Slafer, G.A. Development and seed number in indeterminate soybean as affected by timing and duration of exposure to long photoperiods after flowering. Ann. Bot. 2007, 99, 925–933. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, S.; Harada, K.; Abe, J. Genetic and molecular bases of photoperiod responses of flowering in soybean. Breed Sci. 2012, 61, 531–543. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xu, M.; Yamagishi, N.; Zhao, C.; Takeshima, R.; Kasai, M.; Watanabe, S.; Kanazawa, A.; Yoshikawa, N.; Liu, B.; Yamada, T.; et al. The soybean-specific maturity gene E1 family of floral repressors controls night-break responses through down-regulation of FLOWERING LOCUS T orthologs. Plant Physiol. 2015, 168, 1735–1746. [Google Scholar] [CrossRef] [Green Version]
- Mao, T.; Li, J.; Wen, Z.; Wu, T.; Wu, C.; Shi, S.; Jiang, B.; Hou, W.; Li, W.; Song, Q.; et al. Association mapping of loci controlling genetic and environmental interaction of soybean flowering time under various photo-thermal conditions. BMC Genom. 2017, 18, 415. [Google Scholar] [CrossRef] [PubMed]
- Mathew, J.P.; Herbert, S.J.; Zhang, S.; Rautenkranz, A.A.F.; Litchfield, G.V. Differential response of soybean yield components to the timing of light enrichment. Agron. J. 2000, 92, 1156–1161. [Google Scholar] [CrossRef] [Green Version]
- Polson, D.E. Day-neutrality in soybeans. Crop. Sci. 1972, 7945. [Google Scholar] [CrossRef]
- Yang, W.Y.; Wu, T.T.; Zhang, X.Y.; Song, W.W.; Xu, C.L.; Sun, S.; Hou, W.S.; Jiang, B.J.; Han, T.F.; Wu, C.X. Critical photoperiod measurement of soybean genotype in different maturity groups. Crop. Sci. 2019, 59, 2055–2061. [Google Scholar] [CrossRef]
- Lin, X.; Liu, B.; Weller, J.L.; Abe, J.; Kong, F. Molecular mechanisms for the photoperiodic regulation of flowering in soybean. J. Integr. Plant Biol. 2020, 63, 981–994. [Google Scholar] [CrossRef]
- Chen, X.Z.; Xie, H.; Li, X. Studies on correlation ship of development stages and agronomic traits of summer sowing soybean. Mol. Plant Breed. 2004, 2, 247–252. [Google Scholar] [CrossRef] [Green Version]
- Cober, E.R.; Morrison, M.J. Regulation of seed yield and agronomic characters by photoperiod sensitivity and growth habit genes in soybean. Theor. Appl. Genet. 2010, 120, 1005–1012. [Google Scholar] [CrossRef]
- Zhang, L.; Liu, W.; Tsegaw, M.; Xu, X.; Qi, Y.; Sapey, E.; Liu, L.; Wu, T.; Sun, S.; Han, T. Principles and practices of the photo-thermal adaptability improvement in soybean. J. Integr. Agric. 2020, 19, 295–310. [Google Scholar] [CrossRef]
- Luo, X.; Yin, M.; He, Y. molecular genetic understanding of photoperiodic regulation of flowering time in Arabidopsis and soybean. Int. J. Mol. Sci. 2022, 23, 466. [Google Scholar] [CrossRef] [PubMed]
- Gai, J.Y.; Wang, Y.S. A study on the varietal eco-regions of soybeans in China. Sci. Agric. Sin. 2001, 34, 139–145. [Google Scholar]
- Seferova, I.V. Soybean in the North-West of the Russian Federation/Macличhьie Kyльtypьi. Hayчho—Texhичeckий бюллetehь Bcepoccийckoгo Hayчhoиccлeдobateльckoгo Иhctиtyta Macличhьix Kyльtyp. Bьin 2016, 3, 101–105. Available online: https://cyberleninka.ru/article/n/soya-v-usloviyah-severo-zapada-rossiyskoy-federatsii (accessed on 10 November 2022).
- Xia, Z.; Watanabe, S.; Yamada, T.; Tsubokura, Y.; Nakashima, H.; Zhai, H.; Anai, T.; Sato, S.; Yamazaki, T.; Lü, S.; et al. Positional cloning and characterization reveal the molecular basis for soybean maturity locus E1 that regulates photoperiodic flowering. Proc. Natl. Acad. Sci. USA 2012, 109, E2155–E2164. [Google Scholar] [CrossRef] [Green Version]
- Watanabe, S.; Xia, Z.; Hideshima, R.; Tsubokura, Y.; Sato, S.; Yamanaka, N.; Takahashi, R.; Anai, T.; Tabata, S.; Kitamura, K.; et al. A map-based cloning strategy employing a residual heterozygous line reveals that the GIGANTEA gene is involved in soybean maturity and flowering. Genetics 2011, 188, 395–407. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bernard, R.L. Two major genes for time of flowering and maturity in soybeans. Crop Sci. 1971, 11, 242–244. [Google Scholar] [CrossRef]
- Buzzell, R.I. Inheritance of a soybean flowering response to fluorescent-daylength conditions. Can. J. Genet. Cytol. 1971, 13, 703–707. [Google Scholar] [CrossRef]
- Watanabe, S.; Hideshima, R.; Xia, Z.; Tsubokura, Y.; Sato, S.; Nakamoto, Y.; Yamanaka, N.; Takahashi, R.; Ishimoto, M.; Anai, T.; et al. Map-based cloning of the gene associated with the soybean maturity locus E3. Genetics 2009, 182, 1251–1262. [Google Scholar] [CrossRef] [Green Version]
- Buzzell, R.I.; Voldeng, H.D. Research Notes: Inheritance of insensitivity to long daylength. Soybean Genet. Newsl. 1980, 7, 13. [Google Scholar]
- Liu, B.; Kanazawa, A.; Matsumura, H.; Takahashi, R.; Harada, K.; Abe, J. Genetic redundancy in soybean photoresponses associated with duplication of the phytochrome A gene. Genetics 2008, 180, 995–1007. [Google Scholar] [CrossRef] [Green Version]
- Cober, E.R.; Voldeng, H.D. Low R:FR light quality delays flowering of E7E7 soybean lines. Crop. Sci. 2001, 41, 1823–1826. [Google Scholar] [CrossRef]
- Zhai, H.; Lü, S.; Liang, S.; Wu, H.; Zhang, X.; Liu, B.; Kong, F.; Yuan, X.; Li, J.; Xia, Z. GmFT4, a homolog of FLOWERING LOCUS T, is positively regulated by E1 and functions as a flowering repressor in soybean. PLoS ONE 2014, 9, e89030. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Samanfar, B.; Molnar, S.J.; Charette, M.; Schoenrock, A.; Dehne, F.; Golshani, A.; Belzile, F.; Cober, E.R. Mapping and identification of a potential candidate gene for a novel maturity locus, E10, in soybean. Theor. Appl. Genet. 2017, 130, 377–390. [Google Scholar] [CrossRef] [PubMed]
- Bonato, E.R.; Vello, N.A. E6, a dominant gene conditioning early flowering and maturity in soybeans. Genet. Mol. Biol. 1999, 22, 229–232. [Google Scholar] [CrossRef] [Green Version]
- Cober, E.R. Long juvenile soybean flowering responses under very short photoperiods. Crop Sci. 2011, 51, 140–145. [Google Scholar] [CrossRef]
- Kong, F.; Liu, B.; Xia, Z.; Sato, S.; Kim, B.M.; Watanabe, S.; Yamada, T.; Tabata, S.; Kanazawa, A.; Harada, K.; et al. Two coordinately regulated homologs of FLOWERING LOCUS T are involved in the control of photoperiodic flowering in soybean. Plant Physiol. 2010, 154, 1220–1231. [Google Scholar] [CrossRef] [Green Version]
- Wang, F.; Nan, H.; Chen, L.; Fang, C.; Zhang, H.; Su, T.; Li, S.; Cheng, Q.; Dong, L.; Liu, B.; et al. A new dominant locus, E11, controls early flowering time and maturity in soybean. Mol. Breed. 2019, 39, 70. [Google Scholar] [CrossRef]
- Ray, J.D.; Hinson, K.; Mankono, J.E.B.; Malo, M.F. Genetic Control of a Long-Juvenile Trait in Soybean. Crop Sci. 1995, 35, 1001. [Google Scholar] [CrossRef]
- Lu, S.; Dong, L.; Fang, C.; Liu, S.; Kong, L.; Cheng, Q.; Chen, L.; Su, T.; Nan, H.; Zhang, D.; et al. Stepwise selection on homeologous PRR genes controlling flowering and maturity during soybean domestication. Nat. Genet. 2020, 52, 428–436. [Google Scholar] [CrossRef]
- Fan, C.; Hu, R.; Zhang, X.; Wang, X.; Zhang, W.; Zhang, Q.; Ma, J.; Fu, Y.F. Conserved CO-FT regulons contribute to the photoperiod flowering control in soybean. BMC Plant Biol. 2014, 14, 9. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Guo, G.; Xu, K.; Zhang, X.; Zhu, J.; Lu, M.; Chen, F.; Liu, L.; Xi, Z.-Y.; Bachmair, A.; Chen, Q.; et al. Extensive analysis of GmFTL and GmCOL expression in northern soybean cultivars in field conditions. PLoS ONE 2015, 10, e0136601. [Google Scholar] [CrossRef]
- Dong, L.; Fang, C.; Cheng, Q.; Su, T.; Kou, K.; Kong, L.; Zhang, C.; Li, H.; Hou, Z.; Zhang, Y.; et al. Genetic basis and adaptation trajectory of soybean from its temperate origin to tropics. Nat. Commun. 2021, 12, 5445. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Fang, C.; Yang, Y.; Lv, T.; Su, T.; Chen, L.; Nan, H.; Li, S.; Zhao, X.; Lu, S.; et al. Overcoming the genetic compensation response of soybean florigens to improve adaptation and yield at low latitudes. Curr. Biol. 2021, 31, 3755–3767.e4. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.; Li, M.; Xu, C.; Yang, X.; Li, D.; Zhao, X.; Wang, K.; Li, Y.; Zhang, X.; Liu, L.; et al. Natural variation in GmGBP1 promoter affects photoperiod control of flowering time and maturity in soybean. Plant J. 2018, 6, 147–162. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, L.; Song, W.; Wang, L.; Sun, X.; Qi, Y.; Wu, T.; Sun, S.; Jiang, B.; Wu, C.; Hou, W.; et al. Allele combinations of maturity genes E1-E4 affect adaptation of soybean to diverse geographic regions and farming systems in China. PLoS ONE 2020, 15, e0235397. [Google Scholar] [CrossRef]
- Song, Y.H.; Shim, J.S.; Kinmonth-Schultz, H.A.; Imaizumi, T. Photoperiodic flowering: Time measurement mechanisms in leaves. Annu. Rev. Plant Biol. 2015, 66, 441. [Google Scholar] [CrossRef] [Green Version]
- Xu, X.; Zhang, L.; Cao, X.; Liu, L.; Jiang, B.; Zhang, C.; Jia, H.; Lyu, X.; Su, Y.; Cai, Y.; et al. Cotyledons facilitate the adaptation of early-maturing soybean varieties to high-latitude long-day environments. Plant Cell Environ. 2021, 44, 2551–2564. [Google Scholar] [CrossRef]
- Huber, S.C.; Rufty, T.W.; Kerr, P.S. Effect of photoperiod on photosynthate partitioning and diurnal rhythms in sucrose phosphate synthase activity in leaves of soybean (Glycine max L. [Merr.]) and tobacco (Nicotiana tabacum L.). Plant Physiol. 1984, 75, 1080–1084. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wallace, D.H.; Yourstone, K.S.; Masaya, P.N.; Zobel, R.W. Photoperiod gene control over partitioning between reproductive and vegetative growth. Theor. Appl. Genet. 1993, 86, 6–16. [Google Scholar] [CrossRef] [PubMed]
- Jarecki, W.; Bobrecka-Jamro, D. Effect of sowing date on the yield and seed quality of soybean (Glycine max (L.) Merr.). J. Elem. 2021, 26, 7–18. [Google Scholar] [CrossRef]
- Morandi, E.N.; Casano, L.M.; Reggiardo, L.M. Post-flowering photoperiodic effect on reproductive efficiency and seed growth in soybean. Field Crop Res. 1988, 18, 227–241. [Google Scholar] [CrossRef]
- Summerfield, R.J.; Roberts, E.H.; Ellis, R.H.; Lawn, R.J. Towards the reliable prediction of time to flowering in six annual crops. I. The development of simple models for fluctuating field environments. Exp. Agric. 1991, 27, 11–31. [Google Scholar] [CrossRef]
- Song, W.; Sun, S.; Ibrahim, S.E.; Xu, Z.; Wu, H.; Hu, X.; Wu, H.; Hu, X.; Jia, H.; Cheng, Y.; et al. Standard cultivar selection and digital quantification for precise classification of maturity groups in soybean. Crop Sci. 2019, 59, 1997–2006. [Google Scholar] [CrossRef]
- Jańczak-Pieniążek, M.; Buczek, J.; Bobrecka-Jamro, D.; Szpunar-Krok, E.; Tobiasz-Salach, R.; Jarecki, W. Morphophysiology, productivity and quality of soybean (Glycine max (L.) Merr.) cv. Merlin in response to row spacing and seeding systems. Agronomy 2021, 11, 403. [Google Scholar] [CrossRef]
- Abdel Latef, A.A.H.; Jan, S.; Abd-Allah, E.F.; Rashid, B.; John, R.; Ahmad, P. Soybean under abiotic stress: Proteomic approach. In Plant-Environment Interaction: Resposes and Approaches to Mitigate Stress; Azooz, M.M., Ahmad, P., Eds.; Wiley-Blackwell: New York, NY, USA, 2016; pp. 28–42. [Google Scholar] [CrossRef]
- Nouri, M.Z.; Toorchi, M.; Komatsu, S. Proteomics approach for identifying abiotic stress responsive proteins in soybean. Soybean Mol. Asp. Breed. 2011, 9, 187–214. [Google Scholar] [CrossRef] [Green Version]
- Chinnusamy, V.; Zhu, J.; Zhu, J.K. Cold stress regulation of gene expression in plants. Trends Plant Sci. 2007, 12, 444–451. [Google Scholar] [CrossRef]
- Grimm, S.S.; Jones, J.W.; Boote, K.J.; Hesketh, J.D. Parameter estimation for predicting flowering date of soybean cultivars. Crop Sci. 1993, 33, 137–144. [Google Scholar] [CrossRef]
- Setiyono, T.D.; Weiss, A.; Specht, J.; Bastidas, A.M.; Cassman, K.G.; Dobermann, A. Understanding and modeling the effect of temperature and daylength on soybean phenology under high-yield conditions. Field Crop Res. 2007, 100, 257–271. [Google Scholar] [CrossRef] [Green Version]
- Boote, K.J. Improving soybean cultivars for adaptation to climate change and climate variability. In Crop Adaptation to Climate Change; Wiley-Blackwell: Oxford, UK, 2011; pp. 370–395. [Google Scholar] [CrossRef]
- Parent, B.; Tardieu, F. Temperature responses of developmental processes have not been affected by breeding in different ecological areas for 17 crop species. New Phytol. 2012, 194, 760–774. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.; Pandey, V.; Shekh, A.; Kumar, M. Growth and yield response of soybean (Glycine max L.) in relation to temperature, photoperiod and sunshine duration at Anand, Gujarat, India. Am. Eur. J. Agron. 2008, 1, 45–50. Available online: http://www.idosi.org/aeja/1(2)08/6.pdf?q=birla-institute-of-technology-mesra-ranchi-835215-india (accessed on 2 August 2022).
- Miransari, M. Soybean Production. In Abiotic and Biotic Stresses in Soybean Production; Miransari, M., Ed.; Academic Press: London, UK, 2015; Available online: https://books.google.pl/books?hl=pl&lr=&id=ILV0BgAAQBAJ&oi=fnd&pg=PP1&dq=Miransari,+M.+Soybean+Production+In:+Miransari,+M.+(Ed.).+Abiotic+and+biotic+stresses+in+soybean+production:+Vol.1,+Vol.+1.+Academic+press,+London,+UK.+2015&ots=2qlhLfAupF&sig=YhEa8nDlLwD-F7pqND9K_EJktPs&redir_esc=y#v=onepage&q=Miransari%2C%20M.%20Soybean%20Production%20In%3A%20Miransari%2C%20M.%20(Ed.).%20Abiotic%20and%20biotic%20stresses%20in%20soybean%20production%3A%20Vol.1%2C%20Vol.%201.%20Academic%20press%2C%20London%2C%20UK.%202015&f=false (accessed on 2 August 2022).
- Yamaguchi, N.; Yamazaki, H.; Ohnishi, S.; Suzuki, C.; Hagihara, S.; Miyoshi, T.; Senda, M. Method for selection of soybeans tolerant to seed cracking under chilling temperatures. Breed. Sci. 2014, 64, 103–108. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Staniak, M.; Stępień-Warda, A.; Czopek, K.; Kocira, A.; Baca, E. Seeds quality and quantity of soybean [Glycine max (L.) Merr.] cultivars in response to cold stress. Agronomy 2021, 11, 520. [Google Scholar] [CrossRef]
- Markowski, A. Influence of initial seed moisture and temperature conditions during germination and emergence on seedling survival and yields of soybean (Glycine max L. Merrill). Acta Agrobot. 1982, 35, 43–59. [Google Scholar] [CrossRef] [Green Version]
- Janas, K.; Cvirkova, M.; Pałągiewicz, A.; Eder, J. Alterations in phenylpropanoid content in soybean roots during low temperature acclimation. Plant Physiol. Biochem. 2000, 38, 587–593. [Google Scholar] [CrossRef]
- Zhang, F.; Smith, D.L. Effects of low root zone temperature on the early stages of symbiosis establishment between soybean (Glycine max (L.) Merr) and Bradyrhizobium japonicum. J. Exp. Bot. 1994, 45, 1467–1473. [Google Scholar] [CrossRef]
- Duzan, H.M.; Zhou, X.; Souleimanov, A.; Smith, D.L. Perception of Bradyrhizobium japonicum nod factor by soybean [Glycine max (L.) Merr.] root hairs under abiotic stress conditions. J. Exp. Bot. 2004, 55, 2641–2646. [Google Scholar] [CrossRef] [Green Version]
- Egli, D.B.; Cornelius, P.L. A regional analysis of the response of soybean yield to planting date. Agron. J. 2009, 101, 330–335. [Google Scholar] [CrossRef]
- De Bruin, J.L.; Pedersen, P. Soybean seed yield response to planting date and seeding rate in the upper Midwest. Agron. J. 2008, 100, 696–703. [Google Scholar] [CrossRef]
- Marburger, D.A.; Smith, D.L.; Conley, S.P. Revisiting planting date and cultivar effects on soybean sudden death syndrome development and yield loss. Plant Dis. 2016, 100, 2152–2157. [Google Scholar] [CrossRef] [Green Version]
- Gaspar, A.P.; Conley, S.P. Responses of canopy reflectance, light interception, and soybean seed yield to replanting suboptimal stands. Crop Sci. 2015, 55, 377–385. [Google Scholar] [CrossRef]
- Księżak, J.; Bojarszczuk, J. The seed yield of soybean cultivars and their quantity depending on sowing term. Agronomy 2022, 12, 1066. [Google Scholar] [CrossRef]
- Egli, D.B.; Bruening, W.P. Potential of early-maturing soybean cultivars in late plantings. Agron. J. 2000, 92, 532–537. [Google Scholar] [CrossRef]
- Pedersen, P.; Lauer, J.G. Response of soybean yield components to management system and planting date. Agron. J. 2004, 96, 1372–1381. [Google Scholar] [CrossRef] [Green Version]
- Lemichhane, J.R.; Constantina, J.; Schovinga, C.; Mauryb, P.; Debaekea, P.; Aubertota, J.-N.; Dürrc, C. Analysis of soybean germination, emergence, and prediction of a possible northward establishment of the crop under climate change. Eur. J. Agron. 2020, 113, 125972. [Google Scholar] [CrossRef]
- Meyer, D.W.; Badaruddin, M. Frost tolerance of ten seedling legume species at four growth stages. Crop Sci. 2001, 41, 1838–1842. [Google Scholar] [CrossRef]
- Mahieu, P.J.; Brinkman, M.A. Double-cropping soybean after harvesting small grains as forage in the north central USA. J. Prod. Agric. 1990, 3, 385–389. [Google Scholar] [CrossRef]
- Shapiro, B.I.; Brorsen, B.W.; Doster, D.H. Adoption of double-cropping soybeans and wheat. South. J. Agric. Econ. 1992, 24, 33–40. [Google Scholar] [CrossRef] [Green Version]
- Staniak, M.; Czopek, K.; Stępień-Warda, A.; Kocira, A.; Przybyś, M. Cold Stress during Flowering Alters Plant Structure, Yield and Seed Quality of Different Soybean Genotypes. Agronomy 2021, 11, 2059. [Google Scholar] [CrossRef]
- Jähne, F.; Balko, C.; Hahn, V.; Würschum, T.; Leiser, W. Cold stress tolerance of soybeans during flowering: QTL mapping and efficient selection strategies under controlled conditions. Plant Breed. 2019, 138, 708–720. [Google Scholar] [CrossRef]
- Nawracała, J. Efektywność kwitnienia zróżnicowanych genotypów soi (Glycine max (L.) Merrill) w warunkach środowiskowych Wielkopolski/Efficiency of flowering of varied soybean (Glycine max (L.) Merrill) genotypes in the environmental conditions of Wielkopolska. Rośliny Oleiste—Oilseed Crops 2001, 26, 27–44. (In Polish) [Google Scholar]
- Ohnishi, S.; Miyoshi, T.; Shirai, S. Low temperature stress at different flower developmental stages affects pollen development, pollination, and pod set in soybean. Environ. Exp. Bot. 2010, 69, 56–62. [Google Scholar] [CrossRef]
- Kurosaki, H.; Yumoto, S. Effects of low temperature and shading during flowering on the yield components in soybeans. Plant Prod. Sci. 2003, 6, 17–23. [Google Scholar] [CrossRef]
- Zheng, H.; Chen, L.; Han, X. Response of soybean yield to daytime temperature change during seed filling: A long-term field study in Northeast China. Plant Prod. Sci. 2009, 12, 526–532. [Google Scholar] [CrossRef]
- Hatfield, J.L.; Boote, K.J.; Kimnball, B.A.; Ziska, L.H.; Izaurralde, R.C.; Ort, D.; Thomson, A.M.; Wolfe, D. Climate impacts on agriculture: Implications for crop production. Agron. J. 2011, 103, 351–370. [Google Scholar] [CrossRef] [Green Version]
- Kurosaki, H.; Yumoto, S.; Matsukawa, I. Pod setting pattern during and after low temperature and the mechanism of cold-weather tolerance at the flowering stage in soybeans. Plant Prod. Sci. 2003, 6, 247–254. [Google Scholar] [CrossRef]
- Hume, D.J.; Jackson, A.K.H. Pod formation in soybeans at low temperatures. Crop Sci. 1981, 21, 933. [Google Scholar] [CrossRef]
- Schor, A.; Fossati, A.; Soldat, A.; Stamp, P. Cold tolerance in soybean (Glycine max L. Merr.) in relation to flowering habit, pod set and compensation for lost reproductive organs. Eur. J. Agron. 1993, 2, 173–178. [Google Scholar] [CrossRef]
- Rahman, M.M.; Hampton, J.G.; Hill, M.J. Soybean development under the cool temperate environment of Canterbury, New Zealand. J. New Seeds 2008, 7, 17–36. [Google Scholar] [CrossRef]
- Rose, G.; Osborne, T.; Greatrex, H.; Wheeler, T. Impact of progressive global warming on the global-scale yield of maize and soybean. Clim. Chang. 2016, 134, 417–428. [Google Scholar] [CrossRef] [Green Version]
- Wamsler, C.; Niven, L.; Beery, T.H.; Bramryd, T.; Ekelund, N.; Jönsson, K.I.; Osmani, A.; Palo, T.; Stålhammar, S. Operationalizing ecosystem-based adaptation: Harnessing ecosystem services to buffer communities against climate change. Ecol. Soc. 2016, 21, 31. [Google Scholar] [CrossRef] [Green Version]
- Poehlman, J.M. Breeding Soybeans. In Breeding Field Crops; Springer: Dordrecht, The Netherlands, 1987. [Google Scholar] [CrossRef]
- Kurasch, A.K.; Hahn, V.; Leiser, W.L.; Vollmann, J.; Schori, A.; Bétrix, C.A.; Mayr, B.; Winkler, J.; Mechtler, K.; Aper, J.; et al. Identification of mega-environments in Europe and effect of allelic variation at maturity E loci on adaptation of European soybean. Plant Cell Environ. 2017, 40, 765–778. [Google Scholar] [CrossRef]
- Zhang, L.X.; Kyei-Boahen, S.; Zhang, J.; Zhang, M.H.; Freeland, T.B.; Watson, C.E.; Liu, X. Modifications of optimum adaptation zones for soybean maturity groups in the USA. Crop Manag. 2007, 6, 1–10. [Google Scholar] [CrossRef]
- Alliprandini, L.F.; Abatti, C.; Bertagnolli, P.F.; Cavassim, J.E.; Gabe, H.L.; Kurek, A.; Matsumoto, M.N.; Rott de Oliveira, M.A.; Pitol, C.; Prado, L.C.; et al. Understanding soybean maturity groups in Brazil: Environment, cultivar classification, and stability. Crop Sci. 2009, 49, 801–808. [Google Scholar] [CrossRef]
- Dardanelli, J.L.; Balzarini, M.; Martínez, M.J.; Cuniberti, M.; Resnik, S.; Ramunda, S.F.; Herrero, R.; Baigorri, H. Soybean maturity groups, environments, and their interaction define mega-environments for seed composition in Argentina. Crop Sci. 2006, 46, 1939–1947. [Google Scholar] [CrossRef]
- Fageria, N.K.; Baligar, V.C.; Jones, C.A. Growth and Mineral Nutrition of Field Crops; CRC Press: Boca Raton, FL, USA, 2010; p. 590. [Google Scholar]
- Kumagai, E.; Sameshima, R. Genotypic differences in soybean yield responses to increasing temperature in a cool climate are related to maturity group. Agric. Forest Meteorol. 2014, 198–199, 265–272. [Google Scholar] [CrossRef]
- Mourtzinis, S.; Gaspar, A.P.; Naeve, S.L.; Conley, S.P. Planting date, maturity, and temperature effects on soybean seed yield and composition. Agron. J. 2017, 109, 2040–2049. [Google Scholar] [CrossRef] [Green Version]
- Ghosh, D.; Xu, J. Abiotic stress responses in plant roots: A proteomics perspective. Front. Plant Sci. 2014, 5, 6. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sharma, P.; Sharma, N.; Deswal, R. The molecular biology of the low-temperature response in plants. Bioessays 2005, 27, 1048–1059. [Google Scholar] [CrossRef]
- Lee, D.-G.; Ahsan, N.; Lee, S.-H.; Lee, J.J.; Bahk, J.D.; Kang, K.Y.; Lee, B.-H. Chilling stress-induced proteomic changes in rice roots. J. Plant Physiol. 2009, 166, 1–11. [Google Scholar] [CrossRef]
- Dumont, E.; Bahrman, N.; Goulas, E.; Valot, B.; Sellier, H.; Hilbert, J.L.; Vuylsteker, C.; Lejeune-Hénaut, I.; Delbreil, B. A proteomic approach to decipher chilling response from cold acclimation in pea (Pisum sativum L.). Plant Sci. 2011, 180, 86–98. [Google Scholar] [CrossRef]
- Mckhann, H.I.; Gery, C.; Berard, A.; Leveque, S.; Zuther, E.; Hincha, D.K.; De Mita, S.; Brunel, D.; Téoulé, E. Natural variation in CBF gene sequence, gene expression and freezing tolerance in the Versailles core collection of Arabidopsis thaliana. BMC Plant Biol. 2008, 8, 105. [Google Scholar] [CrossRef]
- Lata, C.; Prasad, M. Role of DREBs in regulation of abiotic stress responses in plants. J. Exp. Bot. 2011, 62, 4731–4748. [Google Scholar] [CrossRef]
- Hossain, Z.; Khatoon, A.Z.; Komatsu, S. Soybean proteomics for unraveling abiotic stress response mechanism. J. Proteome Res. 2013, 12, 4670–4684. [Google Scholar] [CrossRef] [PubMed]
- Waraich, E.A.; Ahmad, R.; Halim, A.; Aziz, T. Alleviation of temperature stress by nutrient management in crop plants: A review. J. Soil Sci. Plant Nutr. 2012, 12, 221–244. [Google Scholar] [CrossRef] [Green Version]
- Gibson, L.R.; Mullen, R.E. Influence of day and night temperature on soybean seed yield. Crop Sci. 1996, 36, 98–104. [Google Scholar] [CrossRef]
- Wheeler, T.R.; Craufurd, P.Q.; Ellis, R.H.; Porter, J.R.; Prasad, P.V.V. Temperature variability and the yield of annual crops. Agric. Ecosyst. Environ. 2000, 82, 159–167. [Google Scholar] [CrossRef]
- Miransari, M. Handling soybean (Glycine_max L.) under stress. In Crop Improvement; Hakeem, K., Ahmad, P., Ozturk, M., Eds.; Springer: Boston, MA, USA, 2013; pp. 421–439. [Google Scholar] [CrossRef]
- Schlenker, W.; Roberts, M.J. Estimating the Impact of Climate Change on Crop Yields: The Importance of Nonlinear Temperature Effects; Working Paper; National Bureau of Economic Research: Cambridge, MA, USA, 2008; p. 13799. [Google Scholar] [CrossRef]
- Dornbos, D.L.; Mullen, R.E. Influence of stress during soybean seed fill on seed weight, germination, and seedling growth rate. Can. J. Plant Sci. 1991, 71, 373–383. [Google Scholar] [CrossRef] [Green Version]
- Ren, S.; Bilyeu, K.D.; Beuselinck, P.R. Composition, vigor, and proteome of mature soybean seeds developed under high temperature. Crop Sci. 2009, 49, 1010–1022. [Google Scholar] [CrossRef]
- Mustafa, H.S.B.; Hasan, E.; Hassan, M.; Sarwar, S.; Qayyum, A.; Mahmood, T. Influence of Climatic Conditions on Chemical Configuration of Seeds in Safflower, Soybean, Linseed and Sesame. Nat. Sci. 2016, 14, 125–140. [Google Scholar] [CrossRef]
- Hinson, K.; Hartwig, E.E. Soybean Production in the Tropics; FAO Plant Production and Protection Paper; FAO: Roma, Italy, 1982; Volume 4, pp. 2–12. [Google Scholar]
- Keigley, P.J.; Mullen, R.E. Changes in soybean seed quality from high temperature during seed fill and maturation. Crop Sci. 1986, 26, 1212–1216. [Google Scholar] [CrossRef]
- Spears, J.F.; TeKrony, D.M.; Egli, D.B. Temperature during seed filling and soybean seed germination and vigour. Seed Sci. Technol. 1997, 25, 233–244. [Google Scholar]
- Egli, D.B.; TeKrony, D.M.; Heitholt, J.J.; Rupe, J. Air temperature during seed filling and soybean seed germination and vigor. Crop Sci. 2005, 45, 1329–1335. [Google Scholar] [CrossRef]
- Salem, M.A.; Kakani, V.G.; Koti, S.; Reddy, K.R. Pollen-based screening of soy- bean genotypes for high temperatures. Crop Sci. 2007, 47, 219–231. [Google Scholar] [CrossRef] [Green Version]
- Charng, Y.Y.; Liu, H.C.; Liu, N.Y.; Chi, W.T.; Wang, C.N.; Chang, S.H.; Wang, T.T. A heat-inducible transcription factor, HsfA2, is required for extension of acquired thermotolerance in Arabidopsis. Plant Physiol. 2007, 143, 251–262. [Google Scholar] [CrossRef] [Green Version]
- Yoshida, T.; Ohama, N.; Nakajima, J.; Kidokoro, S.; Mizoi, J.; Nakashima, K.; Maruyama, K.; Kim, J.-M.; Seki, M.; Todaka, D.; et al. Arabidopsis HsfA1 transcription factors function as the main positive regulators in heat shock-responsive gene expression. Mol. Genet. Genom. 2011, 286, 321–332. [Google Scholar] [CrossRef]
- Thomas, J.M.G.; Boote, K.J.; Allen, L.H.; Gallo-Meagher, M.; Davis, J.M. Elevated temperature and carbon dioxide effects on soybean seed composition and transcript abundance. Crop Sci. 2003, 43, 1548–1557. [Google Scholar] [CrossRef]
- Wilson, R.F. Seed composition. In Soybeans: Improvement, Production, and Uses; Boerma, H.R., Specht, J.E., Eds.; The American Society of America, Crop Science Society of America, Soil Science Society of America: Madison, WI, USA, 2004; pp. 621–677. [Google Scholar]
- Yaklich, R.W.; Vinyard, B.T. A method to estimate soybean seed protein and oil concentration before harvest. J. Am. Oil Chem. Soc. 2004, 81, 1021–1027. [Google Scholar] [CrossRef]
- Dornbos, D.L.; Mullen, R.E. Soybean seed protein and oil contents and fatty acid composition adjustments by drought and temperature. J. Am. Oil. Chem. Soc. 1992, 69, 228–231. [Google Scholar] [CrossRef]
- Alsajri, F.A.; Wijewardana, C.; Irby, J.T.; Bellaloui, N.; Krutz, L.J.; Golden, B.; Gao, W.; Reddy, R.K. Developing functional relationships between temperature and soybean yield and seed quality. Agron. J. 2020, 112, 194–204. [Google Scholar] [CrossRef] [Green Version]
- Khan, A.Z.; Shah, P.; Khan, H.; Nigar, S.; Perveen, S.; Shah, M.K.; Amanullah; Khalil, S.K.; Munir, S.; Zubair, M. Seed quality and vigor of soybean cultivars as influenced by canopy temperature. Pak. J. Bot. 2011, 43, 643–648. Available online: http://www.pakbs.org/pjbot/PDFs/43(1)/PJB43(1)643.pdf (accessed on 2 August 2022).
- Wolf, R.B.; Cavins, J.F.; Kleiman, R.; Black, L.T. Effect of temperature on soybean seed constituents: Oil, protein, moisture, fatty acids, amino acid, and sugars. J. Am. Oil Chem. Soc. 1982, 59, 230–232. [Google Scholar] [CrossRef]
- Nakagawa, A.C.S.; Ario, N.; Tomita, Y.; Tanaka, S.; Murayama, N.; Mizuta, C.; Iwaya-Inoue, M.; Ishibashi, Y. High temperature during soybean seed development differentially alters lipid and protein metabolism. Plant Prod. Sci. 2020, 23, 504–512. [Google Scholar] [CrossRef]
- Rotundo, J.L.; Miller-Garvin, J.E.; Naeve, S.L. Regional and temporal variation in soybean seed protein and oil across the United States. Crop Sci. 2016, 56, 797–808. [Google Scholar] [CrossRef]
- Kołodziej, J.; Pisulewska, E. Wpływ czynników meteorologicznych na plon nasion i tłuszczu oraz zawartość tłuszczu w nasionach dwóch odmian soi. Rośliny Oleiste-Oilseed Crops 2000, 21, 759–773. Available online: https://yadda.icm.edu.pl/agro/element/bwmeta1.element.agro-article-f4b4538a-b39c-455e-a468-52ca8b44ea05 (accessed on 2 August 2022). (In Polish).
- Rotundo, J.L.; Westgate, M.E. Meta-analysis of environmental effects on soybean seed composition. Field Crops Res. 2009, 110, 147–156. [Google Scholar] [CrossRef]
- Rinker, K.; Nelson, R.; Specht, J.; Sleper, D.; Cary, R.; Cianzio, S.; Casteel, S.; Conley, S.; Chen, P.; Davis, V.; et al. Genetic improvement of soybean in maturity groups II, III, and IV. Crop Sci. 2014, 54, 1–14. [Google Scholar] [CrossRef]
- Patil, G.; Mian, R.; Vuong, T.; Pantalone, V.; Song, Q.; Chen, P.; Shannon, J.; Carter, T.C.; Nguyen, H.T. Molecular mapping and genomics of soybean seed protein: A review and perspective for the future. Theor. Appl. Genet. 2017, 130, 1975–1991. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Starck, Z. Wpływ warunków stresowych na kondycję wytwarzania i dystrybucji fotoasymilatów/Effect of stress conditions on coordination of photosynthetic production and resources allocation. Zesz. Probl. Post. Nauk Rol. 2010, 62, 9–26. [Google Scholar]
- Górski, T.; Kozyra, J.; Doroszewski, A. Field crop losses in Poland due to extreme weather conditions: Case studies. In The Influence of Extreme Phenomena on the Natural Environment and Human Living Conditions; Liszewski, S., Ed.; ŁTN: Łodź, Poland, 2008; pp. 35–49. [Google Scholar]
- Gerten, D.; Rost, S. Climate Change Impacts on Agricultural Water Stress and Impact Mitigation Potential; Background note to the World Development Report 2010; World Bank: Washington, DC, USA, 2009; Available online: https://publications.pik-potsdam.de/pubman/item/item_16181 (accessed on 10 December 2022).
- Karaczun, Z.; Kozyra, J. Wpływ Zmiany Klimatu na Bezpieczeństwo Żywnościowe Polski; Wydawnictwo SGGW: Warszawa, Poland, 2020; p. 120. Available online: http://zgpke.pl/wp-content/uploads/2020/11/Raport_Klimat_bezpieczenstwo_zywienowe_Karaczun_20.03.pdf (accessed on 2 August 2022). (In Polish)
- Souza, G.M.; Catuchi, T.A.; Bertolli, S.C.; Soratto, R.P. Soybean under water deficit: Physiological and yield responses. In A Comprehensive Survey of International Soybean Research—Genetics, Physiology, Agronomy and Nitrogen Relationships; INTECH: London, UK, 2013; pp. 273–298. [Google Scholar] [CrossRef]
- Ohashi, Y.; Nakayama, N.; Saneoka, H.; Mohapatra, P.K.; Fujita, K. Differences in the responses of stem diameter and pod thickness to drought stress during the grain filling stage in soybean plants. Acta Physiol Plant. 2009, 31, 271–277. [Google Scholar] [CrossRef]
- Catuchi, T.A.; Guidorizzi, F.V.C.; Guidorizi, K.A.; Barbosa, A.M.; Souza, G.M. Respostas fisiológicas de cultivares de soja à adubação potássica sob diferentes regimes hídricos. Pesq. Agropec. Bras. 2012, 47, 519–527. [Google Scholar] [CrossRef] [Green Version]
- Da Mota, F.S. Soya bean and weather. World Meteorological Organization (WMO). Tech. Not. 1978, 498, 64. [Google Scholar]
- Michałek, S.; Borowski, E. Seed Germination and Seedling Growth of the Polish Soybean (Glycine max (L.) Merr.) Cultivars in Drought Conditions/Kiełkowanie Nasion i Wzrost Siewek Krajowych Odmian soi (Glycine max (L.) Merr.) w Warunkach suszy. 2002, 223/224, 195–201. Available online: https://www.researchgate.net/publication/303043373_Seed_germination_and_seedling_growth_of_the_soybean_Glycine_max_L_Merr_cultivars_in_drought_conditions (accessed on 2 August 2022). (In Polish).
- Hafeez, Y.; Iqbal, S.; Jabeen, K.; Shahzad, S.; Jahan, S.; Rasul, F. Effect of biochar application on seed germination and seedling growth of Glycine max (L.) Merr. under drought stress. Pak. J. Bot. 2017, 49, 7–13. Available online: https://www.cabdirect.org/cabdirect/abstract/20173269117 (accessed on 2 August 2022).
- Desclaux, D.; Huynh, T.T.; Roumet, P. Identification of soybean plant characteristics that indicate the timing of drought stress. Crop Sci. 2000, 40, 716–722. [Google Scholar] [CrossRef]
- Staniak, M.; Stępień, A.; Czopek, K. Reakcja soi zwyczajnej (Glycine max (L.) Merr.) na wybrane stresy abiotyczne. Studia i Raporty IUNG-PIB 2018, 57, 51–62. Available online: https://iung.pl/sir/zeszyt57_5.pdf (accessed on 2 August 2022). (In Polish).
- Akýnci, S.; Lösel, D.M. Plant water-stress response mechanisms. In Water Stress; Rahman, I.M.M., Hasegawa, H., Eds.; InTech: London, UK, 2012; pp. 15–42. Available online: https://studylib.net/doc/18748898/plant-water-stress-response-mechanisms (accessed on 2 August 2022).
- Borowska, M.; Prusiński, J. Effect of soybean cultivars sowing dates on seed yield and its correlation with yield parameters. Plant Soil Environ. 2021, 67, 360–366. [Google Scholar] [CrossRef]
- Mandić, V.; Krnjaja, V.; Tomić, Z.; Bijelić, Z.; Simić, A.; Đorđević, S.; Stanojković, A.; Gogić, M. Effect of water stress on soybean production. In Proceedings of the 4th International Congress New Perspectives an Challenges of Sustainable Livestock Production, Belgrade, Serbia, 7–9 October 2015; Available online: http://r.istocar.bg.ac.rs/handle/123456789/602 (accessed on 2 August 2022).
- Sadeghipour, O.; Abbasi, S. Soybean response to drought and seed inoculation. World Appl. Sci. J. 2012, 17, 55–56. [Google Scholar]
- Sionit, N.; Kramer, P.J. Effect of water stress during different stages of growth of soybean. Agron. J. 1977, 69, 274–278. [Google Scholar] [CrossRef]
- de Souza, P.I.; Egli, D.B.; Bruening, W.P. Water stress during seed filling and leaf senescence in soybean. Agron. J. 1997, 89, 807–812. [Google Scholar] [CrossRef]
- Michałek, S.; Borowski, E. Plonowanie oraz zawartość tłuszczu, kwasów tłuszczowych i białka w nasionach krajowych odmian soi w warunkach suszy/Yielding, oil, fatty acids and protein content in the seeds of Polish soybean cultivars under drought conditions. Acta Agroph. 2006, 8, 459–471. (In Polish) [Google Scholar]
- Eck, H.V.; Mathers, A.C.; Musick, J.Y. Plant water stress at various growth stages and growth and yield of soybeans. Field Crops Res. 1987, 17, 716–722. [Google Scholar] [CrossRef]
- Frieler, K.; Schauberger, B.; Arneth, A.; Balkovič, J.; Chryssanthacopoulos, J.; Deryng, D.; Elliott, J.; Folberth, C.; Khabarov, N.; Müller, C.; et al. Understanding the weather signal in national crop-yield variability. Earth’s Future 2017, 5, 605–616. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- di Mauro, G.; Borrás, L.; Rugeroni, P.; Rotundo, J.L. Exploring soybean management options for environments with contrasting water availability. J. Agron. Crop Sci. 2019, 205, 274–282. [Google Scholar] [CrossRef]
- Kobraee, S.; Shamsi, K. Effect of drought stress on dry matter accumulation and morphological traits in soybean. Int. J. Biosci. 2012, 10, 73–79. [Google Scholar]
- Ergo, V.V.; Lascano, R.; Vega, C.R.C.; Parola, R.; Carrera, C.S. Heat and water stressed field-grown soybean: A multivariate study on the relationship between physiological-biochemical traits and yield. Environ. Exp. Bot. 2017, 148, 1–11. [Google Scholar] [CrossRef]
- Napoles, M.C.; Guevara, E.; Montero, F.; Rossi, A.; Ferreira, A. Role of Bradyrhizobium japonicum induced by genistein on soybean stressed by water deficit. Span. J. Agric. Res. 2009, 7, 665–671. [Google Scholar] [CrossRef]
- Kunert, K.J.; Vorster, B.J.; Fenta, B.A.; Kibido, T.; Dionisio, G.; Foyer, C.H. Drought stress responses in soybean roots and nodules. Front. Plant Sci. 2016, 7, 1015. [Google Scholar] [CrossRef] [Green Version]
- Korsak-Adamowicz, M.; Starczewski, J.; Dopka, D. Oddziaływanie niektórych zabiegów agrotechnicznych na brodawkowanie soi. Fragm. Agron. 2007, 3, 232–237. (In Polish) [Google Scholar]
- Arrese-Igor, C.; González, E.; Marino, D.; Ladrera, R.; Larrainzar, E.; Gil-Quintana, E. Physiological response of legume nodules to drought. Plant Stress 2011, 5, 24–31. [Google Scholar]
- Collier, R.; Tegeder, M. Soybean ureide transporters play a critical role in nodule development, function and nitrogen export. Plant J. 2012, 72, 355–367. [Google Scholar] [CrossRef] [PubMed]
- Purcell, L.C.; Serraj, R.; Sinclair, T.R.; De, A. Soybean N2 fixation estimates, ureide concentration, and yield responses to drought. Crop Sci. 2004, 44, 484–492. [Google Scholar] [CrossRef] [Green Version]
- Oya, T.; Nepomuceno, A.L.; Neumaier, N.; Farias, J.R.B.; Tobita, S.; Ito, O. Drought tolerance characteristics of Brazilian soybean cultivars—Evaluation and characterization of drought tolerance of various Brazilian soybean cultivars in the field. Plant. Prod. Sci. 2004, 7, 129–137. [Google Scholar] [CrossRef]
- Maleki, A.; Naderi, A.; Naseri, R.; Fathi, A.; Bahamin, S.; Maleki, R. Physiological performance of soybean cultivars under drought stress. Bull. Environ. Pharmacol. Life Sci. 2013, 2, 38–44. [Google Scholar]
- Du, Y.; Zhao, Q.; Chen, L.; Yao, X.; Xie, F. Effect of drought stress at reproductive stages on growth and nitrogen metabolism in soybean. Agronomy 2020, 10, 302. [Google Scholar] [CrossRef] [Green Version]
- Dayoub, E.; Lamichhane, J.R.; Schoving, C.; Debaeke, P.; and Maury, P. Early-stage phenotyping of root traits provides insights into the drought tolerance level of soybean cultivars. Agronomy 2021, 11, 188. [Google Scholar] [CrossRef]
- Buezo, J.; Sanz-Saez, A.; Moran, J.F.; Soba, D.; Aranjuelo, I.; Esteban, R. Drought tolerance response of high-yielding soybean varieties to mild drought: Physiological and photochemical adjustments. Physiol. Plant. 2019, 166, 88–104. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ku, Y.S.; Au-Yeung, W.K.; Yung, Y.L.; Li, M.W.; Wen, C.Q.; Liu, X.; Lam, H.M. Drought stress and tolerance in soybean. In A Comprehensive Survey of International Soybean Research—Genetics, Physiology, Agronomy and Nitrogen Relationships; Board, J.E., Ed.; IntechOpen: London, UK, 2013; pp. 209–237. [Google Scholar] [CrossRef] [Green Version]
- Wijewardana, C.; Reddy, K.R.; Bellaloui, N. Soybean seed physiology, quality, and chemical composition under soil moisture stress. Food Chem. 2019, 278, 92–100. [Google Scholar] [CrossRef]
- Ghassemi-Golezani, K.; Lotfi, R. Influence of Water Stress and Pod Position on Oil and Protein Accumulation in Soybean Grains. Inter. J. Agron. Plant Prod. 2013, 4, 2341–2345. [Google Scholar]
- Fecak, P.; Sarikova, D.; Cerny, I. Influence of tillage system and starting fertilization on seed yield and quality of soybean Glycine max (L.) Merrill. Plant Soil Environ. 2010, 56, 105–110. [Google Scholar] [CrossRef] [Green Version]
- Lorenc-Kozik, A.M.; Pisulewska, E.; Gondek, K. Wpływ warunków pogodowych na skład chemiczny trzech odmian soi. Ecol. Chem. Eng. 2011, 18, 1079–1085. (In Polish) [Google Scholar]
- Vollmann, J.; Fritz, C.N.; Wagentristl, H.; Ruckenbauer, P. Environmental and genetic variation of soybean seed protein content under Central European growing conditions. J. Sci. Food Agric. 2000, 80, 1300–1306. [Google Scholar] [CrossRef]
- Kumar, V.; Rani, A.; Solanki, S.; Hussain, S.M. Influence of growing environment on the biochemicall composition and physical characteristics of soybean seeds. J. Food Comp. Anal. 2006, 19, 188–195. [Google Scholar] [CrossRef]
- Toorchi, M.; Yukawa, K.; Nouri, M.Z.; Komatsu, S. Proteomics approach for identifying osmotic-stress-related proteins in soybean roots. Peptides 2009, 30, 2108–2117. [Google Scholar] [CrossRef]
- Alam, I.; Sharmin, S.A.; Kim, K.H.; Yang, J.K.; Choi, M.S.; Lee, B.H. Proteome analysis of soybean roots subjected to short-term drought stress. Plant Soil 2010, 333, 491–505. [Google Scholar] [CrossRef]
- Mohammadi, P.P.; Moieni, A.; Hiraga, S.; Komatsu, S. Organ-specific proteomic analysis of drought-stressed soybean seedlings. J. Proteom. 2012, 75, 1906–1923. [Google Scholar] [CrossRef]
- Mickelbart, M.V.; Hasegawa, P.M.; Bailey-Serres, J. Genetic mechanisms of abiotic stress tolerance that translate to crop yield stability. Nat. Rev. Genet. 2015, 16, 237–251. [Google Scholar] [CrossRef]
- Zhang, S.; Yang, W.; Zhao, Q.; Zhou, X.; Jiang, L.; Ma, S.; Liu, X.; Li, Y.; Zhang, C.; Fan, Y.; et al. Analysis of weighted co-regulatory networks in maize provides insights into new genes and regulatory mechanisms related to inositol phosphate metabolism. BMC Genom. 2016, 17, 129. [Google Scholar] [CrossRef] [Green Version]
- Muthamilarasan, M.; Bonthala, V.S.; Khandelwal, R.; Jaishankar, J.; Shweta, S.; Nawaz, K.; Prasad, M. Global analysis of WRKY transcription factor superfamily in Setaria identifies potential candidates involved in abiotic stress signaling. Front. Plant Sci. 2015, 6, 910. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Luo, X.; Bai, X.; Zhu, D.; Li, Y.; Ji, W.; Cai, H.; Wu, J.; Liu, B.; Zhu, Y. GsZFP1, a new Cys2/His2-type zinc-finger protein, is a positive regulator of plant tolerance to cold and drought stress. Planta 2011, 235, 1141–1155. [Google Scholar] [CrossRef] [PubMed]
- Striker, G.G. Flooding stress on plants: Anatomical, morphological and physiological responses. In Flooding Stress on Plants: Anatomical, Morphological and Responses; Mworia, J.K., Ed.; InTechOpen: London, UK, 2012; pp. 3–28. [Google Scholar] [CrossRef]
- Tian, L.; Zhang, Y.; Chen, P.; Zhang, F.; Li, J.; Yan, F.; Dong, Y.; Feng, B. How does the waterlogging regime affect crop yield? A global meta-analysis. Front. Plant Sci. 2021, 12, 634898. [Google Scholar] [CrossRef] [PubMed]
- Ploschuk, R.A.; Miralles, D.J.; Colmer, T.D.; Ploschuk, E.L.; Striker, G.G. Waterlogging of winter crops at early and late stages: Impacts on leaf physiology, growth and yield. Front. Plant Sci. 2018, 9, 1863. [Google Scholar] [CrossRef] [Green Version]
- Sairam, R.K.; Dharmar, K.; Chinnusamy, V.; Meena, R.C. Waterlogging-induced increase in sugar mobilization, fermentation, and related gene expression in the roots of mung bean (Vigna radiata). J. Plant Physiol. 2009, 166, 602–616. [Google Scholar] [CrossRef] [PubMed]
- Voesenek, L.; Bailey-Serres, J. Flooding tolerance: O2 sensing and survival strategies. Curr. Opin. Plant Biol. 2013, 16, 647–653. [Google Scholar] [CrossRef] [PubMed]
- Sullivan, M.; VanToai, T.; Fausey, N.; Beuerlein, J.; Parkinson, R.; Soboyejo, A. Evaluating On-Farm Flooding Impacts on Soybean. Crop Sci. 2001, 41, 93–100. [Google Scholar] [CrossRef] [Green Version]
- Linkemer, G.; Board, J.E.; Musgrave, M.E. Waterlogging effect on growth and yield components of late-planted soybean. Crop Sci. 1998, 38, 1576–1584. [Google Scholar] [CrossRef] [PubMed]
- Scott, H.D.; DeAngulo, J.; Daniels, M.B.; Wood, L.S. Flood duration effects on soybean growth and yield. Agron. J. 1989, 81, 631–636. [Google Scholar] [CrossRef]
- Oosterhuis, D.M.; Scott, H.D.; Hampton, R.E.; Wullschleger, S.D. Physiological responses of two soybean (Glycine max (L.) Merr.) cultivars to short-term flooding. Env. Exp. Bot. 1990, 30, 85–92. [Google Scholar] [CrossRef]
- Board, J.E. Waterlogging effects on plant nutrient concentrations in soybean. J. Plant Nutr. 2008, 31, 828–838. [Google Scholar] [CrossRef]
- Ara, R.; Mannan, M.A.; Khaliq, Q.A.; Uddin Miah, M.M. Waterlogging tolerance of soybean. Bangladesh Agron. J. 2015, 18, 105–109. [Google Scholar] [CrossRef] [Green Version]
- Jackson, M.B.; Ram, P.C. Physiological and molecular basis of susceptibility and tolerance of rice plants to complete submergence. Ann. Bot. 2003, 91, 227–241. [Google Scholar] [CrossRef] [Green Version]
- Nanjo, Y.; Skultety, L.; Ashraf, Y.; Komatsu, S. Comparative proteomic analysis of early-stage soybean seedlings responses to flooding by using gel and gel-free techniques. J. Proteome Res. 2010, 9, 3989–4002. [Google Scholar] [CrossRef] [PubMed]
- Kong, F.J.; Oyanagi, A.; Komatsu, S. Cell wall proteome of wheat roots under flooding stress using gel-based and LCMS/MS-based proteomics approaches. Biochim. Biophys. Acta 2010, 1804, 124–136. [Google Scholar] [CrossRef]
- Shi, F.; Yamamoto, R.; Shimamura, S.; Hiraga, S.; Nakayama, N.; Nakamura, T.; Yukawa, K.; Hachinohe, M.; Matsumoto, H.; Komatsu, S. Cytosolic ascorbate peroxidase 2 (cAPX 2) is involved in the soybean response to flooding. Phytochemistry 2008, 69, 1295–1303. [Google Scholar] [CrossRef] [PubMed]
- Komatsu, S.; Hiraga, S.; Yanagawa, Y. Proteomics techniques for the development of flood tolerant crops. J. Proteome Res. 2012, 11, 68–78. [Google Scholar] [CrossRef] [PubMed]
- Komatsu, S.; Sugimoto, T.; Hoshino, T.; Nanjo, Y.; Furukawa, K. Identification of flooding stress responsible cascades in root and hypocotyl of soybean using proteome analysis. Amino Acids 2010, 38, 729–738. [Google Scholar] [CrossRef] [PubMed]
- Fukao, T.; Xiong, L. Genetic mechanisms conferring adaptation to submergence and drought in rice: Simple or complex? Curr. Opin. Plant Biol. 2013, 16, 196–204. [Google Scholar] [CrossRef]
- Xu, K.; Xu, X.; Fukao, T.; Canlas, P.; Maghirang-Rodriguez, R.; Heuer, S.; Ismail, A.M.; Bailey-Serres, J.; Roland, P.C.; Mackill, D.J. Sub1A is an ethylene-response-factor-like gene that confers submergence tolerance to rice. Nature 2006, 442, 705–708. [Google Scholar] [CrossRef] [Green Version]
- Tamang, B.G.; Fukao, T. Plant adaptation to multiple stresses during submergence and following desubmergence. Int. J. Mol. Sci. 2015, 16, 30164–30180. [Google Scholar] [CrossRef] [Green Version]
- Pucciariello, C.; Perata, P. New insights into reactive oxygen species and nitric oxide signalling under low oxygen in plants. Plant Cell Environ. 2017, 40, 473–482. [Google Scholar] [CrossRef]
- Holman, T.J.; Jones, P.D.; Russell, L.; Medhurst, A.; Ubeda Tomás, S.; Talloji, P.; Marquez, J.; Schmuths, H.; Tung, S.-A.; Taylor, I.; et al. The N-end rule pathway promotes seed germination and establishment through removal of ABA sensitivity in Arabidopsis. Proc. Natl. Acad. Sci. USA 2009, 106, 4549–4554. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gibbs, D.J.; Md Isa, N.; Movahedi, M.; Lozano-Juste, J.; Mendiondo, G.M.; Berckhan, S.; Marín-de la Rosa, N.; Vicente Conde, J.; Sousa Correia, C.; Pearce, S.P.; et al. Nitric oxide sensing in plants is mediated by proteolytic control of group VII ERF transcription factors. Mol. Cell 2014, 53, 369–379. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Graciet, E.; Walter, F.; Maoileidigh, D.O.; Pollmann, S.; Meyerowitz, E.M.; Varshavsky, A.; Wellmer, F. The N-end rule pathway controls multiple functions during Arabidopsis shoot and leaf development. Proc. Natl. Acad. Sci. USA 2009, 106, 13618–13623. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Abbas, M.; Berckhan, S.; Rooney, D.J.; Gibbs, D.J.; Vicente Conde, J.; Sousa Correia, C.; Bassel, G.W.; Marín-de la Rosa, N.; León, J.; Alabadí, D.; et al. Oxygen sensing coordinates photomorphogenesis to facilitate seedling survival. Curr. Biol. 2015, 25, 1483–1488. [Google Scholar] [CrossRef] [PubMed]
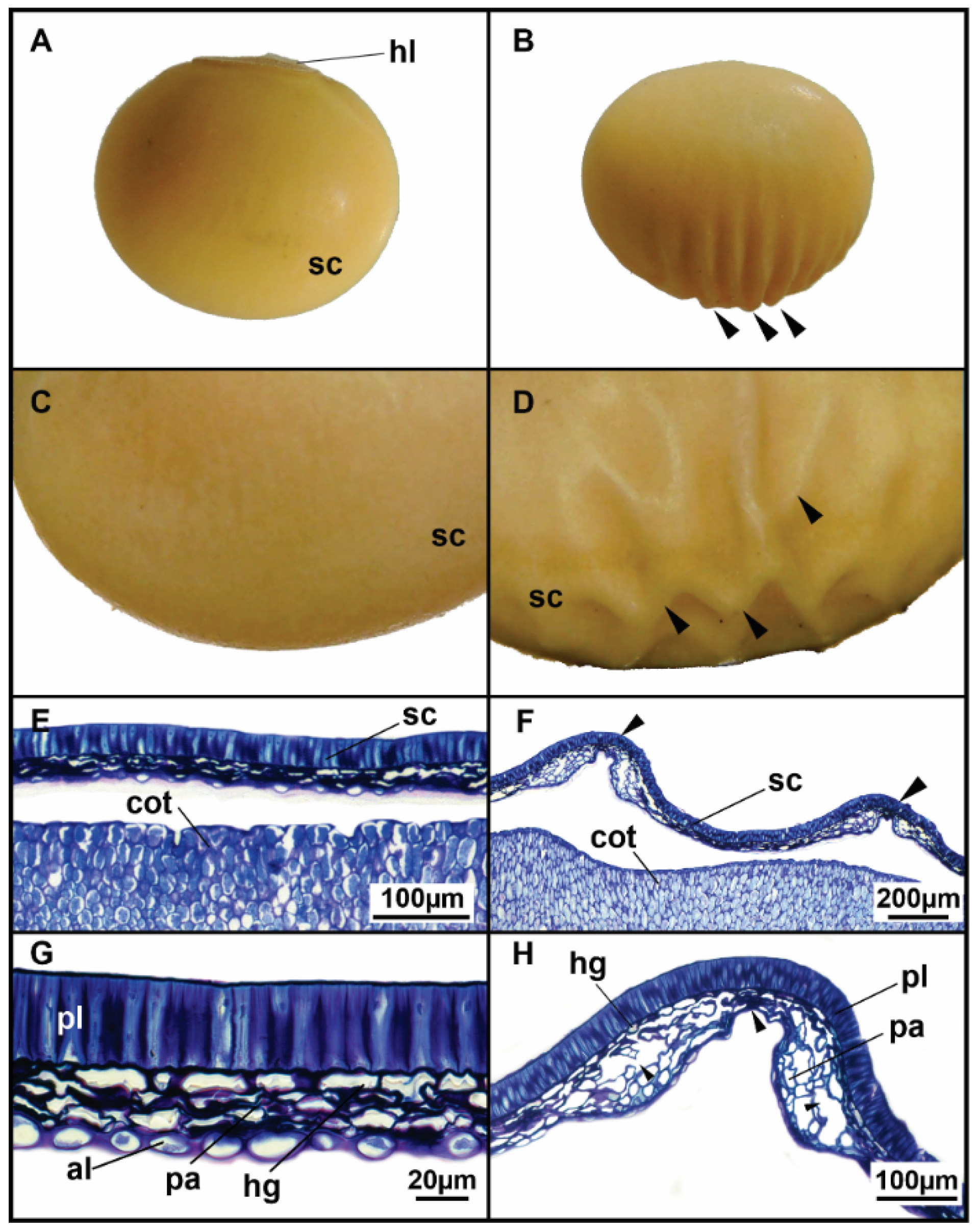
- Pinheiro, D.T.; Dias, D.C.F.; Medeiros, A.D.; Ribeiro, J.P.O.; Silva, F.L.; Silva, L.J. Weathering deterioration in pre-harvest of soybean seeds: Physiological, physical, and morpho-anatomical changes. Sci. Agric. 2021, 78, e20200166. [Google Scholar] [CrossRef]


| Number of Days with Chilling Stress | Root Lenght (cm) | Dry Mass (mg·g−1) | ||
|---|---|---|---|---|
| 25 °C | 10 °C | 25 °C | 10 °C | |
| 0 | 3.7 ± 0.4 | 3.7 ± 0.4 | 78.0 ± 7.0 | 78.0 ± 7.0 |
| 1 | 5.0 ± 0.4 | 3.9 ± 0.7 | 79.0 ± 6.0 | 76.5 ± 10.5 |
| 2 | 6.6 ± 3.3 | 4.0 ± 0.5 | 81.0 ± 8.0 | 79.5 ± 6.5 |
| 3 | 7.4 ± 0.4 | 4.1 ± 0.9 | 80.0 ± 10.2 | 79.0 ± 7.8 |
| 4 | 8.4 ± 1.8 | 4.1 ± 0.4 | 76.0 ± 5.6 | 79.5 ± 10.1 |
| Freezing Temperature (°C) | Development Stage | |||
|---|---|---|---|---|
| Hypocotyl Arch (1 Week after Planting) | Fully Expanded Cotyledons (2 Weeks after Planting) | First Trifoliolate Emerged (3 Weeks after Planting) | Second Trifoliolate Emerged (4 Weeks after Planting) | |
| −2 | 100 | 94 | 94 | 87 |
| −4 | 81 | 56 | 63 | 44 |
| −6 | 75 | 19 | 0 | 13 |
| −8 | 0 | 0 | 0 | 0 |
| Features | Natural Conditions (Average 24/17 °C) | 7-Day Cold Stress (17/13 °C) | NIR (p = 0.05) Tukey Test |
|---|---|---|---|
| Plant height (cm) | 79.6 | 74.8 | 2.57 |
| Number of nodes | 11.0 | 10.2 | 0.34 |
| Stem dry mass (g) | 5.35 | 4.56 | 0.51 |
| Number of pods per plant | 18.0 | 14.9 | 1.59 |
| Weight of pods per plant (g) | 12.0 | 9.3 | 0.96 |
| Number of seeds per pod | 2.36 | 2.27 | 0.055 |
| Number of seeds per plant | 41.7 | 32.5 | 3.13 |
| Weight of seeds per plant (g) | 8.17 | 6.61 | 0.66 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Staniak, M.; Szpunar-Krok, E.; Kocira, A. Responses of Soybean to Selected Abiotic Stresses—Photoperiod, Temperature and Water. Agriculture 2023, 13, 146. https://doi.org/10.3390/agriculture13010146
Staniak M, Szpunar-Krok E, Kocira A. Responses of Soybean to Selected Abiotic Stresses—Photoperiod, Temperature and Water. Agriculture. 2023; 13(1):146. https://doi.org/10.3390/agriculture13010146
Chicago/Turabian StyleStaniak, Mariola, Ewa Szpunar-Krok, and Anna Kocira. 2023. "Responses of Soybean to Selected Abiotic Stresses—Photoperiod, Temperature and Water" Agriculture 13, no. 1: 146. https://doi.org/10.3390/agriculture13010146








