Anti-Inflammatory and T-Cell Immunomodulatory Effects of Banana Peel Extracts and Selected Bioactive Components in LPS-Challenged In Vitro and In Vivo Models
Abstract
:1. Introduction
2. Materials and Methods
2.1. Reagents
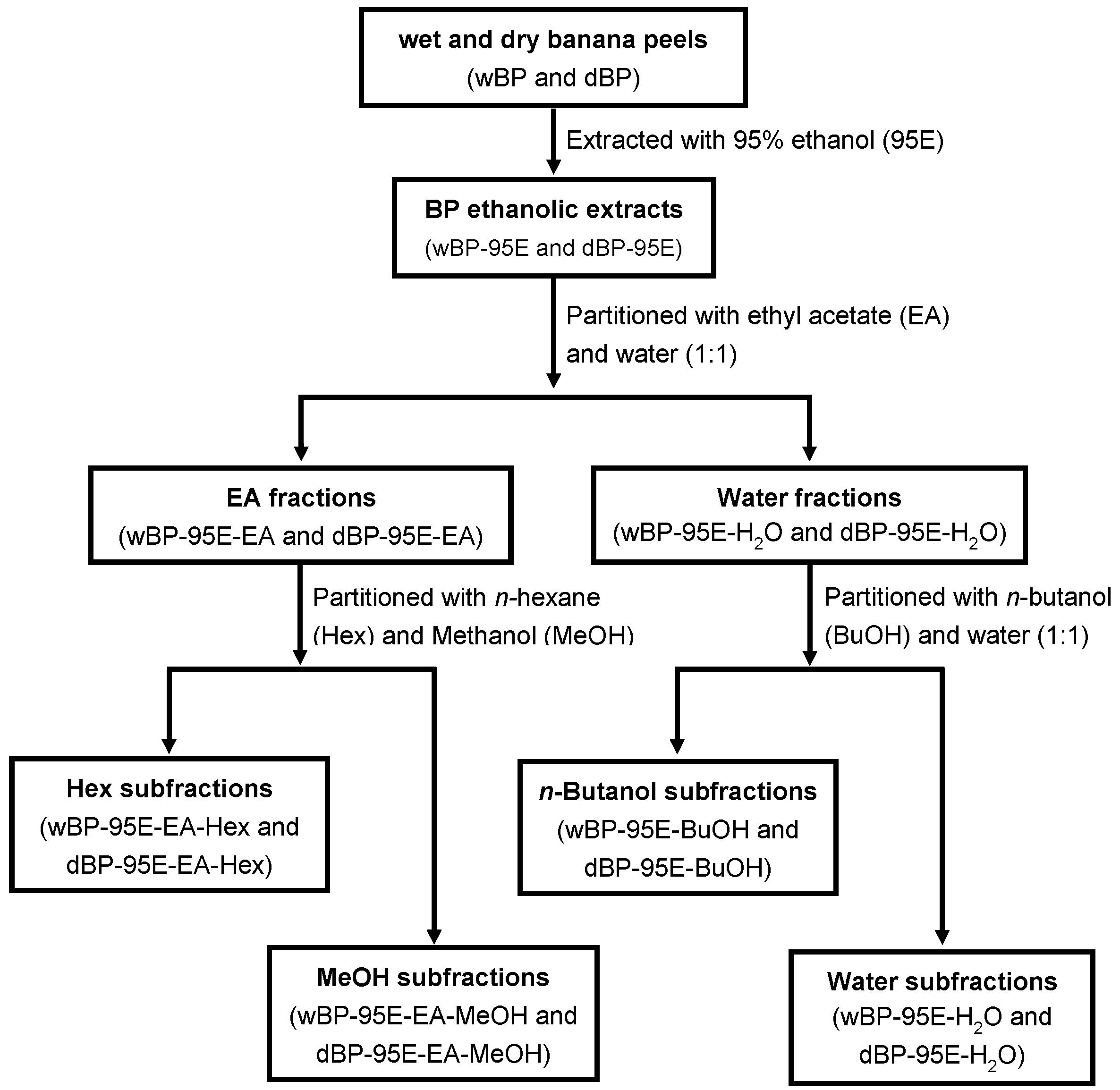
2.2. Plant Materials and Preparation of Banana Peel Extracts
2.3. Cell Culture and Treatment
2.4. Western Blotting
2.5. LPS-Induced Systemic Inflammation in Mice
2.6. Murine Primary Spleen and PEC Cell Culture
2.7. Nitric Oxide Determination
2.8. Pro-Inflammatory and Anti-Inflammatory Cytokines Assay
2.9. Flow Cytometry
2.10. Statistical Analysis
3. Results and Discussions
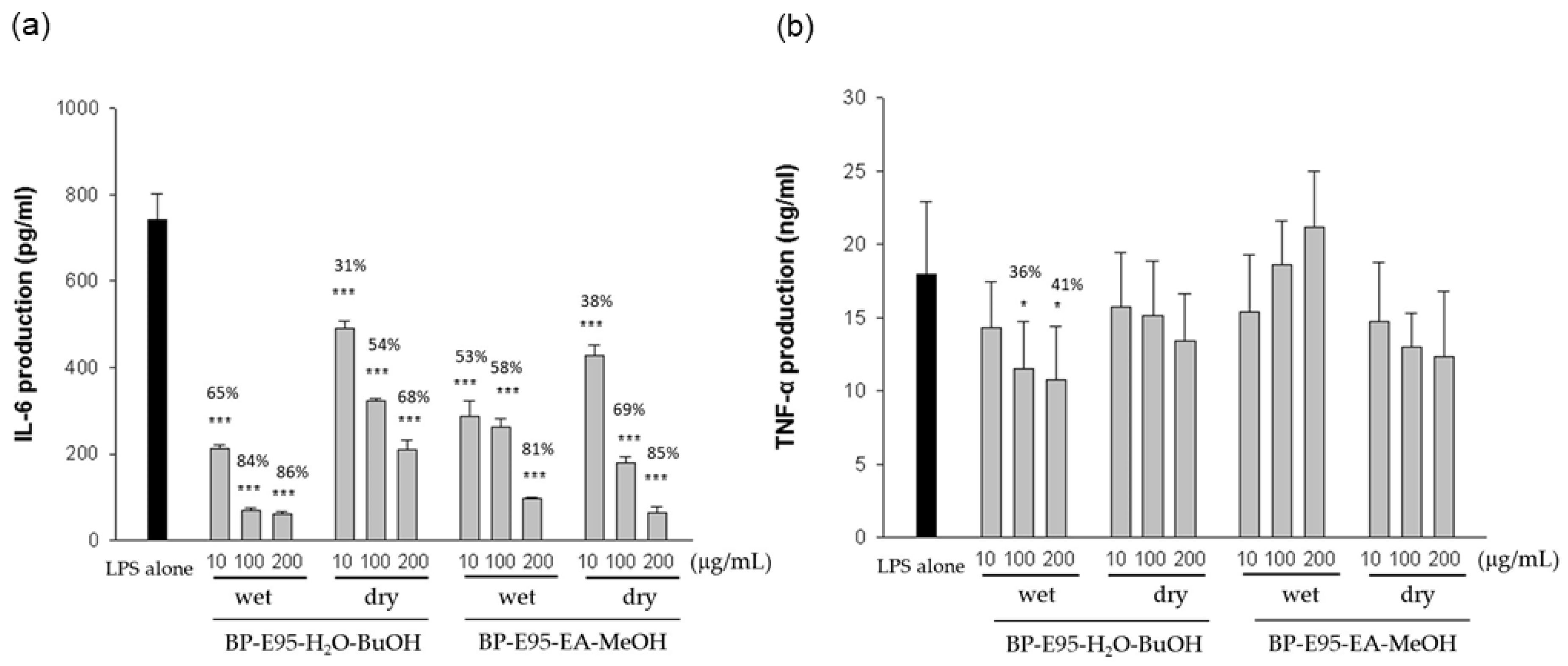
3.1. Anti-Inflammatory Effect of BP Extracts in LPS-Stimulated Macrophages
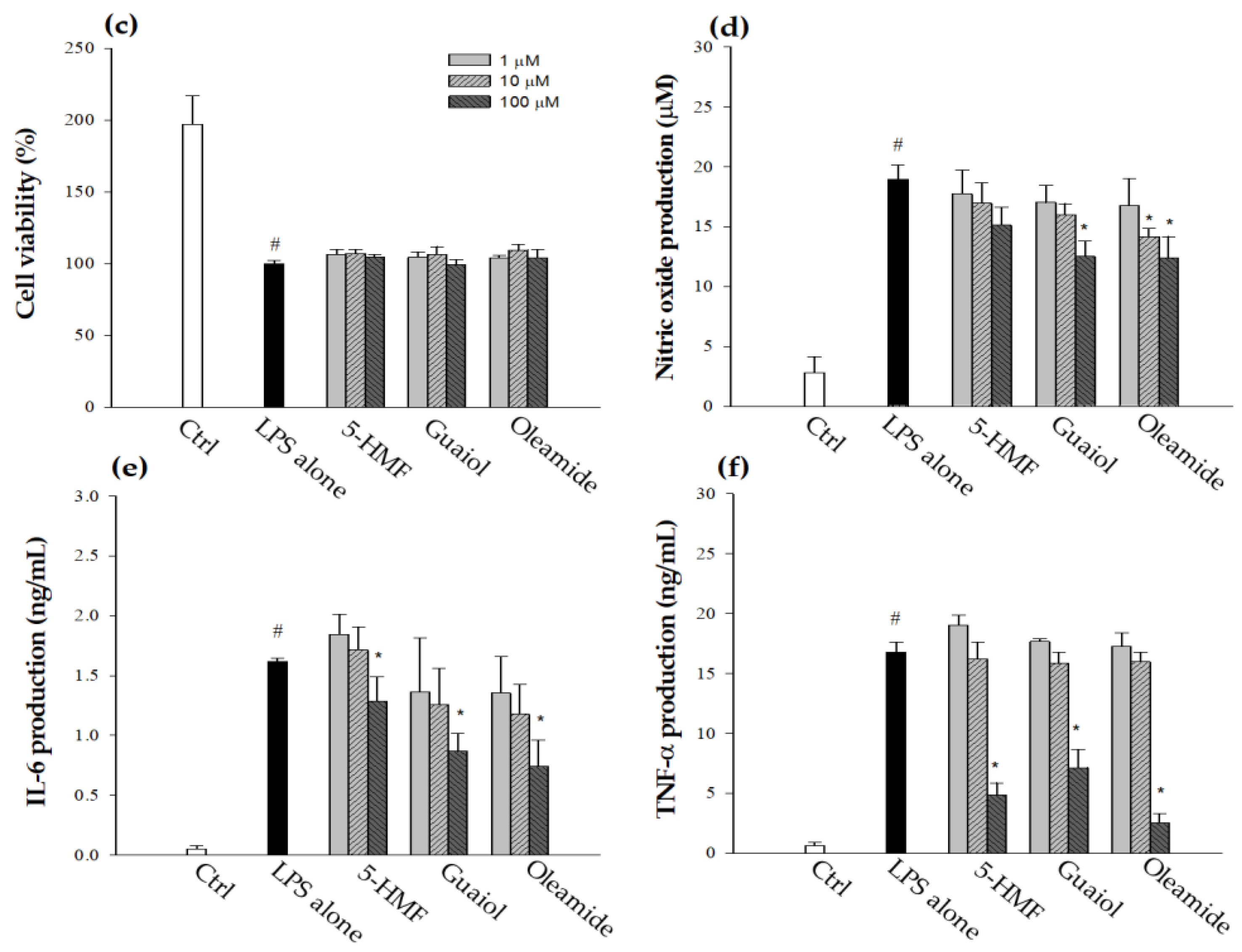
3.2. Identification of Bioactive Compounds in BP-95E-H2O-BuOH Subfractions
3.3. In Vivo Anti-Ianflammatory Effects of BP-95E-H2O-BuOH Subfractions
3.4. Effects of BP-95E-H2O-BuOH extracts on the Regulation of Lymphocytes
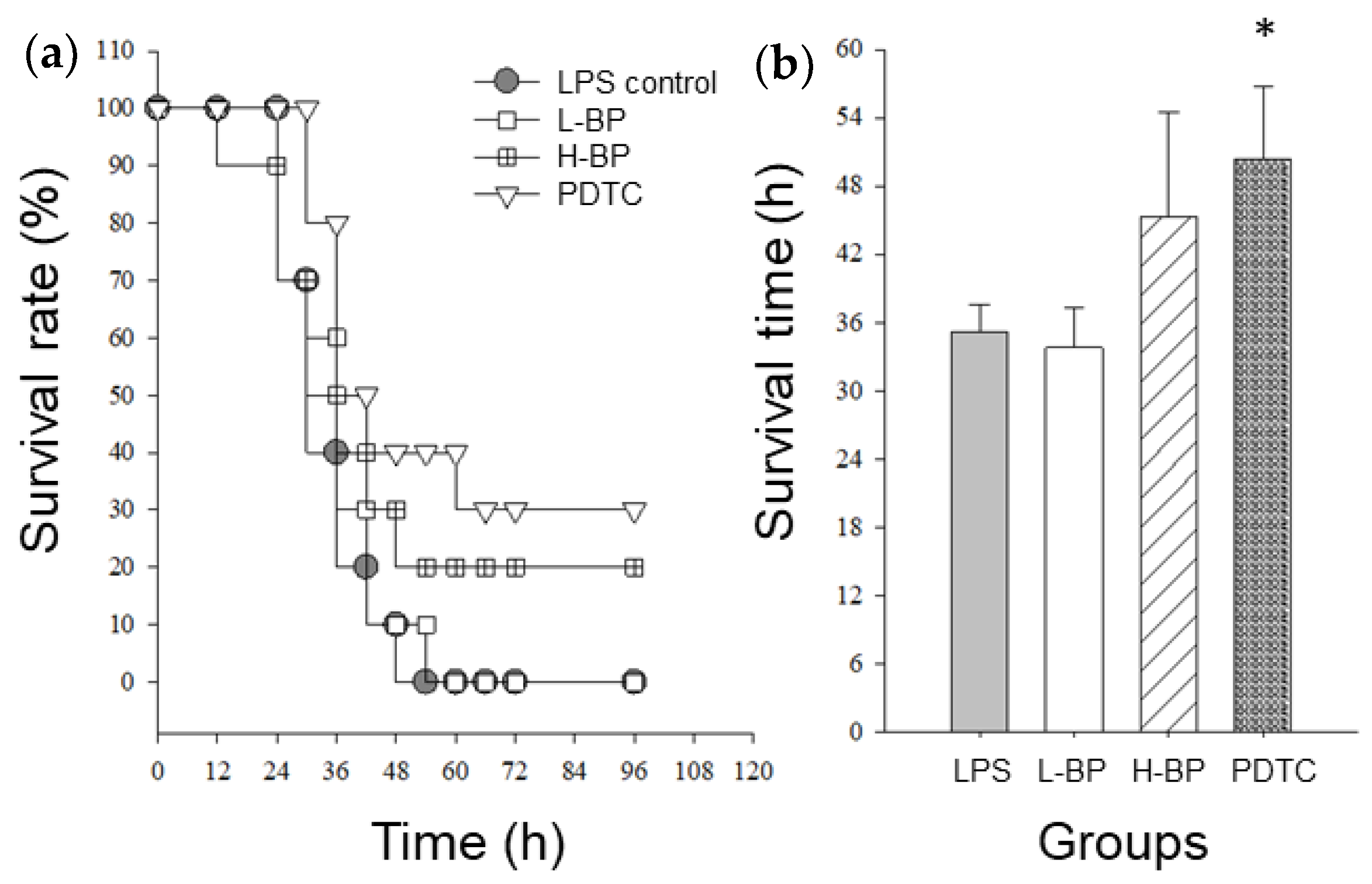
3.5. Effects of BP Extracts on the Survival Rate and Time in LPS-Challenged Mice
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Mezzavilla, M.; Notarangelo, M.; Concas, M.P.; Catamo, E.; Gasparini, P.; Grillotti, M.G.; Robino, A. Investigation of the link between PROP taste perception and vegetables consumption using FAOSTAT data. Int. J. Food Sci. Nutr. 2019, 70, 484–490. [Google Scholar] [CrossRef] [PubMed]
- Hikal, W.M.; Ahl, H.A.H.S.-A.; Bratovcic, A.; Tkachenko, K.G.; Sharifi-Rad, J.; Kačániová, M.; Elhourri, M.; Atanassova, M. Banana Peels: A Waste Treasure for Human Being. Evidence-Based Complement. Altern. Med. 2022, 2022, 7616452. [Google Scholar] [CrossRef] [PubMed]
- Acevedo, S.A.; Carrillo, J.D.; Flórez-López, E.; Grande-Tovar, C.D. Recovery of Banana Waste-Loss from Production and Processing: A Contribution to a Circular Economy. Molecules 2021, 26, 5282. [Google Scholar] [CrossRef] [PubMed]
- Mohapatra, D.; Mishra, S.; Sutar, N. Banana and its by-product utilisation: An overview. J. Sci. Ind. Res. 2010, 69, 323–329. [Google Scholar]
- Annadurai, G.; Juang, R.-S.; Lee, D. Adsorption of heavy metals from water using banana and orange peels. Water Sci. Technol. 2003, 47, 185–190. [Google Scholar] [CrossRef]
- Fries, J.H.; Waldron, R.J. Dehydrated banana in the dietetic management of diarrheas of infancy. J. Pediatr. 1950, 37, 367–372. [Google Scholar] [CrossRef]
- Pereira, A.; Maraschin, M. Banana (Musa spp.) from peel to pulp: Ethnopharmacology, source of bioactive compounds and its relevance for human health. J. Ethnopharmacol. 2015, 160, 149–163. [Google Scholar] [CrossRef]
- Emaga, T.H.; Robert, C.; Ronkart, S.N.; Wathelet, B.; Paquot, M. Dietary fibre components and pectin chemical features of peels during ripening in banana and plantain varieties. Bioresour. Technol. 2008, 99, 4346–4354. [Google Scholar] [CrossRef]
- Vu, H.T.; Scarlett, C.J.; Vuong, Q.V. Phenolic compounds within banana peel and their potential uses: A review. J. Funct. Foods 2018, 40, 238–248. [Google Scholar] [CrossRef]
- Atzingen, D.A.N.C.V.; Gragnani, A.; Veiga, D.F.; Abla, L.E.F.; Cardoso, L.L.F.; Ricardo, T.; dos Anjos Mendonça, A.R.; Ferreira, L.M. Unripe Musa sapientum peel in the healing of surgical wounds in rats. Acta Cir. Bras. 2013, 28, 33–38. [Google Scholar] [CrossRef]
- Vu, H.T.; Scarlett, C.J.; Vuong, Q.V. Effects of drying conditions on physicochemical and antioxidant properties of banana (Musa cavendish) peels. Dry. Technol. 2017, 35, 1141–1151. [Google Scholar] [CrossRef]
- Vu, H.T.; Scarlett, C.J.; Vuong, Q.V. Changes of phytochemicals and antioxidant capacity of banana peel during the ripening process; with and without ethylene treatment. Sci. Hortic. 2019, 253, 255–262. [Google Scholar] [CrossRef]
- Someya, S.; Yoshiki, Y.; Okubo, K. Antioxidant compounds from bananas (Musa Cavendish). Food Chem. 2002, 79, 351–354. [Google Scholar] [CrossRef]
- MPharm, S.R. Anti-inflammatory and antioxidant activities of extracts from Musa sapientum peel. J. Med. Assoc. Thai. 2012, 95, S142–S146. [Google Scholar]
- Chabuck, Z.A.G.; Al-Charrakh, A.H.; Hindi, N.K.K.; Hindi, S.K.K. Antimicrobial effect of aqueous banana peel extract, Iraq. Res. Gate. Pharm. Sci. 2013, 1, 73–75. [Google Scholar]
- Kapadia, S.P.; Pudakalkatti, P.S.; Shivanaikar, S. Detection of antimicrobial activity of banana peel (Musa paradisiaca L.) on Porphyromonas gingivalis and Aggregatibacter actinomycetemcomitans: An in vitro study. Contemp. Clin. Dent. 2015, 6, 496–499. [Google Scholar] [CrossRef] [PubMed]
- Singh, C.R.; Kathiresan, K.; Boopathy, N.S.; Anandhan, S.; Govindan, T. Evaluation of microbial potential of different coloured banana peels. Int. J. Preclin. Pharm. Res. 2013, 4, 62–64. [Google Scholar]
- Wang, R.; Feng, X.; Zhu, K.; Zhao, X.; Suo, H. Preventive activity of banana peel polyphenols on CCl4-induced experimental hepatic injury in Kunming mice. Exp. Ther. Med. 2016, 11, 1947–1954. [Google Scholar] [CrossRef]
- Propper, D.J.; Balkwill, F.R. Harnessing cytokines and chemokines for cancer therapy. Nat. Rev. Clin. Oncol. 2022, 19, 237–253. [Google Scholar] [CrossRef]
- Al-Mansoori, L.; Al-Jaber, H.; Prince, M.S.; Elrayess, M.A. Role of Inflammatory Cytokines, Growth Factors and Adipokines in Adipogenesis and Insulin Resistance. Inflammation 2022, 45, 31–44. [Google Scholar] [CrossRef]
- Soysal, P.; Arik, F.; Smith, L.; Jackson, S.E.; Isik, A.T. Inflammation, Frailty and Cardiovascular Disease. Adv. Exp. Med. Biol. 2020, 1216, 55–64. [Google Scholar] [CrossRef]
- Mohammad, S.; Thiemermann, C. Role of Metabolic Endotoxemia in Systemic Inflammation and Potential Interventions. Front. Immunol. 2020, 11, 594150. [Google Scholar] [CrossRef]
- Yu, Z.; Li, X.; Zhao, J.; Sun, S. Identification of hospitalized mortality of patients with COVID-19 by machine learning models based on blood inflammatory cytokines. Front. Public Health 2022, 10, 1001340. [Google Scholar] [CrossRef]
- Okeke, E.B.; Okwor, I.; Uzonna, J.E. Regulatory T Cells Restrain CD4+ T Cells from Causing Unregulated Immune Activation and Hypersensitivity to Lipopolysaccharide Challenge. J. Immunol. 2014, 193, 655–662. [Google Scholar] [CrossRef]
- Chao, W.W.; Hong, Y.H.; Chen, M.L.; Lin, B.F. Inhibitory effects of Angelica sinensis ethyl acetate extract and major compounds on NF-κB trans-activation activity and LPS-induced inflammation. J. Ethnopharmacol. 2010, 129, 244–249. [Google Scholar] [CrossRef]
- Vu, H.T.; Scarlett, C.J.; Vuong, Q.V. Maximising recovery of phenolic compounds and antioxidant properties from banana peel using microwave assisted extraction and water. J. Food Sci. Technol. 2019, 56, 1360–1370. [Google Scholar] [CrossRef]
- Geng, Y.; Zhu, S.; Cheng, P.; Lu, Z.-M.; Xu, H.-Y.; Shi, J.-S.; Xu, Z.-H. Bioassay-guided fractionation of ethyl acetate extract from Armillaria mellea attenuates inflammatory response in lipopolysaccharide (LPS) stimulated BV-2 microglia. Phytomedicine 2017, 26, 55–61. [Google Scholar] [CrossRef] [PubMed]
- Oh, Y.T.; Lee, J.Y.; Lee, J.; Lee, J.H.; Kim, J.E.; Ha, J.; Kang, I. Oleamide suppresses lipopolysaccharide-induced expression of iNOS and COX-2 through inhibition of NF-κB activation in BV2 murine microglial cells. Neurosci. Lett. 2010, 474, 148–153. [Google Scholar] [CrossRef]
- Zaini, H.M.; Roslan, J.; Saallah, S.; Munsu, E.; Sulaiman, N.S.; Pindi, W. Banana peels as a bioactive ingredient and its potential application in the food industry. J. Funct. Foods 2022, 92, 105054. [Google Scholar] [CrossRef]
- Iriondo, A.; Agirre, I.; Viar, N.; Requies, J. Value-Added Bio-Chemicals Commodities from Catalytic Conversion of Biomass Derived Furan-Compounds. Catalysts 2020, 10, 895. [Google Scholar] [CrossRef]
- Leamon, A.A.A.; Venegas, M.P.; Orsat, V.; Auclair, K.; Dumont, M.-J. Semisynthetic transformation of banana peel to enhance the conversion of sugars to 5-hydroxymethylfurfural. Bioresour. Technol. 2022, 362, 127782. [Google Scholar] [CrossRef] [PubMed]
- Okada, Y.; Tsuzuki, Y.; Sato, H.; Narimatsu, K.; Hokari, R.; Kurihara, C.; Watanabe, C.; Tomita, K.; Komoto, S.; Kawaguchi, A.; et al. Trans fatty acids exacerbate dextran sodium sulphate-induced colitis by promoting the up-regulation of macrophage-derived proinflammatory cytokines involved in T helper 17 cell polarization. Clin. Exp. Immunol. 2013, 174, 459–471. [Google Scholar] [CrossRef] [PubMed]
- Apel, M.A.; Lima, M.E.; Sobral, M.; Young MC, M.; Cordeiro, I.; Schapoval, E.E.; Henriques, A.T.; Moreno, P.R.H. Anti-inflammatory activity of essential oil from leaves of Myrciaria tenella and Calycorectes sellowianus. Pharm. Biol. 2010, 48, 433–438. [Google Scholar] [CrossRef] [PubMed]
- Moon, S.-M.; Lee, S.A.; Hong, J.H.; Kim, J.-S.; Kim, D.K.; Kim, C.S. Oleamide suppresses inflammatory responses in LPS-induced RAW264.7 murine macrophages and alleviates paw edema in a carrageenan-induced inflammatory rat model. Int. Immunopharmacol. 2018, 56, 179–185. [Google Scholar] [CrossRef] [PubMed]
- Hong, Y.H.; Weng, L.W.; Chang, C.C.; Hsu, H.F.; Wang, C.P.; Wang, S.W.; Houng, J.Y. Anti-inflammatory effects of Siegesbeckia orientalis ethanol extract in in vitro and in vivo models. Biomed. Res. Int. 2014, 2014, 329712. [Google Scholar] [CrossRef]
- Gomez, C.R.; Karavitis, J.; Palmer, J.L.; Faunce, D.E.; Ramirez, L.; Nomellini, V.; Kovacs, E.J. Interleukin-6 Contributes to Age-Related Alteration of Cytokine Production by Macrophages. Mediat. Inflamm. 2010, 2010, 475139. [Google Scholar] [CrossRef]
- Burke, J.D.; Young, H.A. IFN-gamma: A cytokine at the right time, is in the right place. Semin. Immunol. 2019, 43, 101280. [Google Scholar] [CrossRef]
- Liu, C.-J.; Lin, J.-Y. Anti-inflammatory effects of phenolic extracts from strawberry and mulberry fruits on cytokine secretion profiles using mouse primary splenocytes and peritoneal macrophages. Int. Immunopharmacol. 2013, 16, 165–170. [Google Scholar] [CrossRef]
- Na, H.; Cho, M.; Chung, Y. Regulation of Th2 Cell Immunity by Dendritic Cells. Immune Netw. 2016, 16, 1–12. [Google Scholar] [CrossRef]
- Eisenbarth, S.C.; Piggott, D.A.; Huleatt, J.W.; Visintin, I.; Herrick, C.A.; Bottomly, K. Lipopolysaccharide-enhanced, toll-like receptor 4-dependent T helper cell type 2 responses to inhaled antigen. J. Exp. Med. 2002, 196, 1645–1651. [Google Scholar] [CrossRef]
- Varma, T.K.; Lin, C.Y.; Toliver-Kinsky, T.E.; Sherwood, E.R. Endotoxin-Induced Gamma Interferon Production: Contributing Cell Types and Key Regulatory Factors. Clin. Vaccine Immunol. 2002, 9, 530–543. [Google Scholar] [CrossRef] [PubMed]
- Yu, D.; Wang, Y.; Yu, C.; Song, M.; Zhou, Q.; Liu, S. High-throughput serum metabolomics analysis of gouty arthritis rat treated by total saponins of Rhizoma Dioscoreae Makino by UPLC–Q/TOF–MS. Biomed. Chromatogr. 2020, 34, e4867. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.-J.; Wang, X.-H.; Dai, Y.-Y.; Ma, M.-H.; Rahman, K.; Nian, H.; Zhang, H. Prunella vulgaris: A Comprehensive Review of Chemical Constituents, Pharmacological Effects and Clinical Applications. Curr. Pharm. Des. 2019, 25, 359–369. [Google Scholar] [CrossRef] [PubMed]
- Luo, J.; Zhang, L.; Zhang, X.; Long, Y.; Zou, F.; Yan, C.; Zou, W. Protective effects and active ingredients of Salvia miltiorrhiza Bunge extracts on airway responsiveness, inflammation and remodeling in mice with ovalbumin-induced allergic asthma. Phytomedicine 2019, 52, 168–177. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.F.; Ye, X.; Malik, A.B. Inhibition of NF-κB activation by pyrrolidine dithiocarbamate prevents in vivo expression of proinflammatory genes. Circulation 1999, 100, 1330–1337. [Google Scholar] [CrossRef]


| Groups | Control a | L-BP | H-BP | PDTC |
|---|---|---|---|---|
| Serum b | ||||
| IL-6 | 755 ± 88 | 661 ± 78 | 556 ± 75 * | 562 ± 43 * |
| TNF-α | 0.99 ± 0.11 | 0.66 ± 0.10 * | 0.69 ± 0.13 * | 0.14 ± 0.04 ** |
| PECs (spontaneous, without LPS) c | ||||
| NO | 10.30 ± 1.74 | 11.50 ± 2.35 | 9.83 ± 2.19 | 10.20 ± 1.83 |
| IL-6 | 8.10 ± 0.52 | 5.18 ± 0.69 ** | 5.14 ± 0.69 ** | 4.55 ± 0.73 ** |
| TNF-α | 0.18 ± 0.02 | 0.15 ± 0.02 | 0.18 ± 0.02 | 0.10 ± 0.02 * |
| PECs (LPS stimulation) | ||||
| NO | 18.50 ± 3.87 | 17.40 ± 3.85 | 16.30 ± 3.61 | 16.90 ± 3.44 |
| IL-6 | 23.50 ± 3.45 | 11.50 ± 2.61 * | 11.30 ± 1.99 ** | 12.80 ± 3.69 * |
| TNF-α | 0.69 ± 0.11 | 0.56 ± 0.14 | 0.77 ± 0.14 | 0.22 ± 0.07 ** |
| Groups | Control a | L-BP | H-BP | PDTC |
|---|---|---|---|---|
| Spontaneous (without Con A) b | ||||
| IFN-γ | 0.28 ± 0.05 | 0.11 ± 0.01 ** | 0.09 ± 0.02 ** | 0.12 ± 0.02 * |
| IL-4 | 6.52 ± 1.06 | 6.38 ± 2.07 | 5.19 ± 1.38 | 10.4 ± 2.49 |
| IL-6 | 0.09 ± 0.01 | 0.08 ± 0.01 | 0.08 ± 0.02 | 0.15 ± 0.04 |
| IL-10 | 0.21 ± 0.04 | 0.23 ± 0.06 | 0.21 ± 0.09 | 0.32 ± 0.18 |
| Con A stimulation c | ||||
| IFN-γ | 5.35 ± 1.06 | 3.33 ± 1.10 | 2.38 ± 0.47 * | 2.94 ± 0.95 * |
| IL-4 | 24.8 ± 9.25 | 34.1 ± 11.1 | 64.2 ± 16.7 | 90.10 ± 32.90 |
| IL-6 | 0.63 ± 0.10 | 0.70 ± 0.17 | 0.25 ± 0.06 * | 0.83 ± 0.18 * |
| IL-10 | 0.31 ± 0.13 | 0.61 ± 0.20 | 0.81 ± 0.24 | 1.98 ± 0.37 * |
| T-cell populations d | ||||
| CD3CD69 | 23.8 ± 4.56 | 19.3 ± 5.24 | 15.9 ± 4.17 * | 18.3 ± 3.41 * |
| CD3CD4CD25 | 65.3 ± 4.89 | 67.4 ± 5.89 | 65.6 ± 1.62 | 63.4 ± 2.90 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hong, Y.-H.; Kao, C.; Chang, C.-C.; Chang, F.-K.; Song, T.-Y.; Houng, J.-Y.; Wu, C.-H. Anti-Inflammatory and T-Cell Immunomodulatory Effects of Banana Peel Extracts and Selected Bioactive Components in LPS-Challenged In Vitro and In Vivo Models. Agriculture 2023, 13, 451. https://doi.org/10.3390/agriculture13020451
Hong Y-H, Kao C, Chang C-C, Chang F-K, Song T-Y, Houng J-Y, Wu C-H. Anti-Inflammatory and T-Cell Immunomodulatory Effects of Banana Peel Extracts and Selected Bioactive Components in LPS-Challenged In Vitro and In Vivo Models. Agriculture. 2023; 13(2):451. https://doi.org/10.3390/agriculture13020451
Chicago/Turabian StyleHong, Yong-Han, Chieh Kao, Chi-Chang Chang, Fu-Kuei Chang, Tuzz-Ying Song, Jer-Yiing Houng, and Chi-Hao Wu. 2023. "Anti-Inflammatory and T-Cell Immunomodulatory Effects of Banana Peel Extracts and Selected Bioactive Components in LPS-Challenged In Vitro and In Vivo Models" Agriculture 13, no. 2: 451. https://doi.org/10.3390/agriculture13020451
APA StyleHong, Y.-H., Kao, C., Chang, C.-C., Chang, F.-K., Song, T.-Y., Houng, J.-Y., & Wu, C.-H. (2023). Anti-Inflammatory and T-Cell Immunomodulatory Effects of Banana Peel Extracts and Selected Bioactive Components in LPS-Challenged In Vitro and In Vivo Models. Agriculture, 13(2), 451. https://doi.org/10.3390/agriculture13020451






