The Effect of Protein Derivatives and Starch Addition on Some Quality Characteristics of Beef Emulsions and Gels
Abstract
:1. Introduction
2. Materials and Methods
2.1. Raw Materials
2.2. Rheological Properties Determinations
2.3. Textural Analysis
2.4. Cooking Losses
2.5. Microscopic Investigations
2.6. Statistical Analysis
3. Results and Discussion
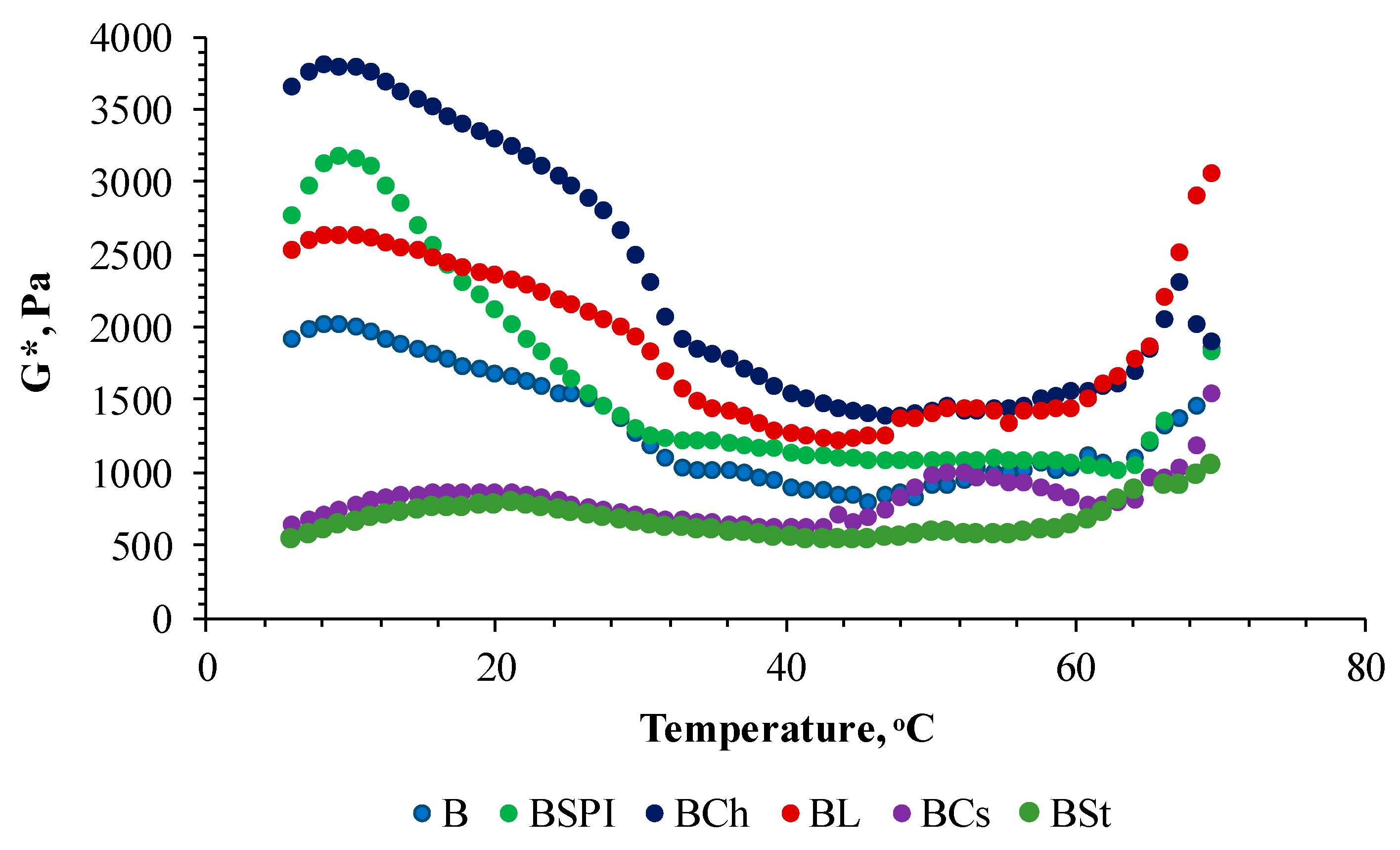
3.1. Rheological Properties
3.2. The Effect of Protein and Starch Addition on the Cutting Force
3.3. The Influence of Amylaceous and Protein Additives on Cooking Losses
3.4. Investigation of the Microscopic Structure of Beef Compositions with Different Additions, Subjected to Heat Treatment
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Rakosky, J. Soy products for the meat industry. J. Agric. Food Chem. 1970, 18, 1005–1009. [Google Scholar] [CrossRef]
- Foegeding, E.A. Gelation in meat batters. Proc. Recip. Meat Conf. 1988, 41, 44–47. Available online: https://earthwormexpress.com/wp-content/uploads/2020/04/gelation-in-meat-batters.pdf (accessed on 4 July 2022).
- Totosaus, A.; Montejano, J.G.; Salazar, J.A.; Guerrero, I. A review of physical and chemical protein-gel induction. Int. J. Food Sci. Technol. 2002, 37, 589–601. [Google Scholar] [CrossRef]
- Hall, G.M. Methods of Testing Protein Functionality; Blackie Academic & Professional: London, UK, 1996. [Google Scholar] [CrossRef]
- Bohrer, B.M. An investigation of the formulation and nutritional composition of modern meat analogue products. Food Sci. Hum. Wellness 2019, 8, 320–329. [Google Scholar] [CrossRef]
- Akesowan, A. Quality characteristics of light pork burgers fortified with soy protein isolate. Food Sci. Biotechnol. 2010, 19, 1143–1149. [Google Scholar] [CrossRef]
- Serdaroğlu, M.; Özsümer, M.S. Effects of soy protein, whey powder and wheat gluten on quality characteristics of cooked beef sausages formulated with 5, 10 and 20% fat. Electron. J. Pol. Agric. Univ. 2003, 6, 3. Available online: http://www.ejpau.media.pl/volume6/issue2/food/art-03.html (accessed on 17 May 2022).
- Jalal, H.; Mir, S.; Wani, S.A.; Sofi, A.H.; Pal, M.A.; Rather, F. Development of low fat meat products. Int. J. Food Nutr. Saf. 2013, 4, 98–107. Available online: https://www.researchgate.net/publication/271208161 (accessed on 6 August 2022).
- Dexter, D.; Sofos, J.; Schmidt, G. Quality Characteristics of Turkey Bologna Formulated with Carrageenan, Starch, Milk and Soy Protein. J. Muscle Foods 1993, 4, 207–223. [Google Scholar] [CrossRef]
- Urbonaite, V.; de Jongh, H.; van der Linden, E.; Pouvreau, L. Water holding of soy protein gels is set by coarseness, modulated by calcium binding, rather than gel stiffness. Food Hydrocoll. 2015, 46, 103–111. [Google Scholar] [CrossRef]
- Raj, J.D.; Nelson, J.A.; Rao, K.S.P. A Study on the Effects of Some Reinforcers to Improve Performance of Employees in a Retail Industry. Behav. Modif. 2006, 6, 848–866. [Google Scholar] [CrossRef]
- Gao, X.-Q.; Kang, Z.-L.; Zhang, W.-G.; Li, Y.-P.; Zhou, G.-H. Combination of κ-Carrageenan and Soy Protein Isolate Effects on Functional Properties of Chopped Low-Fat Pork Batters During Heat-Induced Gelation. Food Bioprocess Technol. 2015, 8, 1524–1531. [Google Scholar] [CrossRef]
- Khalafalla, F.A.; Ali, F.H.M.; Abdel-Azeem, A.H.; Kassem, G.M.A.; Emara, M.M.T. Quality and acceptability of value-added beef burger. J. Vet. Med. Res. 2010, 20, 181–187. [Google Scholar] [CrossRef]
- Das, A.K.; Anjaneyulu, A.S.R.; Verma, A.K.; Kondaiah, N. Effect of levels of full-fat soy paste on quality of goat meat patties. J. Food Sci. Technol. 2007, 44, 323–326. Available online: https://www.researchgate.net/publication/224968566_Effect_of_levels_of_full-fat_soy_paste_on_quality_of_goat_meat_patties (accessed on 7 July 2022).
- Nasrollahzadeh, M. Biopolymer-based metal nanoparticle chemistry for sustainable applications. Elsevier Sci. 2021, 1, 56–58. [Google Scholar]
- Gençcelep, H.; Anil, M.; Saricaoğlu, F.T.; Ağar, B. The Effects of Different Modified Starches on Some Physical and Texture Properties of Meat Emulsion. GIDA/J. Food 2017, 42, 773–786. [Google Scholar] [CrossRef]
- Jiménez-Colmenero, F.; Herrero, A.; Pintado, T.; Solas, M.; Ruiz-Capillas, C. Influence of emulsified olive oil stabilizing system used for pork backfat replacement in frankfurters. Food Res. Int. 2010, 43, 2068–2076. [Google Scholar] [CrossRef]
- Muguruma, M.; Tsuruoka, K.; Katayama, K.; Erwanto, Y.; Kawahara, S.; Yamauchi, K.; Sathe, S.; Soeda, T. Soybean and milk proteins modified by transglutaminase improves chicken sausage texture even at reduced levels of phosphate. Meat Sci. 2002, 63, 191–197. [Google Scholar] [CrossRef]
- Salcedo-Sandoval, L.; Cofrades, S.; Ruiz-Capillas, C.; Matalanis, A.; McClements, D.J.; Decker, E.A.; Jiménez-Colmenero, F. Oxidative stability of n-3 fatty acids encapsulated in filled hydrogel particles and of pork meat systems containing them. Food Chem. 2015, 184, 207–213. [Google Scholar] [CrossRef] [Green Version]
- Nascimento, L.G.L.; Casanova, F.; Silva, N.F.N.; de Carvalho Teixeira, A.V.; de Carvalho, A.F. Casein-based hydrogels: A mini-review. Food Chem. 2020, 314, 126063. [Google Scholar] [CrossRef]
- Kumar, D.; Chatli, M.K.; Singh, R.; Mehta, N.; Kumar, P. Effects of incorporation of camel milk casein hydrolysate on quality, oxidative and microbial stability of goat meat emulsion during refrigerated (4 ± 1 °C) storage. Small Rumin. Res. 2016, 144, 149–157. [Google Scholar] [CrossRef]
- Choudhury, D.; Singh, S.; Seah, J.S.H.; Yeo, D.C.L.; Tan, L.P. Commercialization of Plant-Based Meat Alternatives. Trends Plant Sci. 2020, 25, 1055–1058. [Google Scholar] [CrossRef] [PubMed]
- Duranti, M. Grain legume proteins and nutraceutical properties. Fitoterapia 2006, 77, 67–82. [Google Scholar] [CrossRef] [PubMed]
- Karaca, A.C.; Low, N.; Nickerson, M. Emulsifying properties of chickpea, faba bean, lentil and pea proteins produced by isoelectric precipitation and salt extraction. Food Res. Int. 2011, 44, 2742–2750. [Google Scholar] [CrossRef]
- van de Noort, M. Lupin: An Important Protein and Nutrient. In Sustainable Protein Sources; Academic Press: Cambridge, MA, USA, 2017; pp. 165–183. [Google Scholar] [CrossRef]
- Lara-Rivera, A.H.; García-Alamilla, P.; Lagunes-Gálvez, L.M.; Macias, R.R.; López, P.M.G.; Natera, J.F.Z. Functional Properties of Lupinus angustifolius Seed Protein Isolates. J. Food Qual. 2017, 2017, 8675814. [Google Scholar] [CrossRef] [Green Version]
- Archer, B.J.; Johnson, S.K.; Devereux, H.M.; Baxter, A.L. Effect of fat replacement by inulin or lupin-kernel fibre on sausage patty acceptability, post-meal perceptions of satiety and food intake in men. Br. J. Nutr. 2004, 91, 591–599. [Google Scholar] [CrossRef] [Green Version]
- Keeton, J.T. Fat substitutes introduction and fat modification in processing. Annu. Recipr. Meat Conf. Proc. 1991, 44, 79–91. Available online: https://meatscience.org/docs/default-source/publications-resources/rmc/1991/fat-substitutes-and-fat-modification-in-processing.pdf?sfvrsn=2 (accessed on 18 September 2022).
- Kaur, R.; Prasad, K. Technological, processing and nutritional aspects of chickpea (Cicer arietinum)—A review. Trends Food Sci. Technol. 2021, 109, 448–463. [Google Scholar] [CrossRef]
- Ghribi, A.M.; Ben Amira, A.; Gafsi, I.M.; Lahiani, M.; Bejar, M.; Triki, M.; Zouari, A.; Attia, H.; Besbes, S. Toward the enhancement of sensory profile of sausage “Merguez” with chickpea protein concentrate. Meat Sci. 2018, 143, 74–80. [Google Scholar] [CrossRef] [PubMed]
- Aydemir, L.Y.; Yemenicioğlu, A. Potential of Turkish Kabuli type chickpea and green and red lentil cultivars as source of soy and animal origin functional protein alternatives. LWT 2013, 50, 686–694. [Google Scholar] [CrossRef] [Green Version]
- Sanjeewa, W.T.; Wanasundara, J.; Pietrasik, Z.; Shand, P.J. Characterization of chickpea (Cicer arietinum L.) flours and application in low-fat pork bologna as a model system. Food Res. Int. 2010, 43, 617–626. [Google Scholar] [CrossRef]
- AOAC. Ch 39 Meat and Meat Products. In Official Methods of Analysis, 18th ed.; Horwitz, W., Latimer, G.W., Eds.; Association of Official Analytical Chemists International: Gaithersburgs, MD, USA, 2006; pp. 1–6. ISBN 0-935584-77-3. [Google Scholar]
- Ding, Y.; Liu, R.; Rong, J.; Liu, Y.; Zhao, S.; Xiong, S. Rheological behavior of heat-induced actomyosin gels from yellowcheek carp and grass carp. Eur. Food Res. Technol. 2012, 235, 245–251. [Google Scholar] [CrossRef]
- Patrascu, L.; Banu, I.; Vasilean, I.; Aprodu, I. Effects of Germination and Fermentation on the Functionality of Whole Soy Flour. Bull. UASVM Anim. Sci. Biotechnol. 2016, 73, 126–134. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ziegler, G.R.; Acton, J.C. Mechanisms of gel formation by proteins of muscle tissue. Food Technol. 1984, 38, 77–81. Available online: https://agris.fao.org/agris-search/search.do?recordID=US19850047665 (accessed on 3 September 2022).
- Westphalen, A.D.; Briggs, J.L.; Lonergan, S.M. Influence of muscle type on rheological properties of porcine myofibrillar protein during heat-induced gelation. Meat Sci. 2006, 72, 697–703. [Google Scholar] [CrossRef] [PubMed]
- Tornberg, E. Effects of heat on meat proteins—Implications on structure and quality of meat products. Meat Sci. 2005, 70, 493–508. [Google Scholar] [CrossRef] [PubMed]
- Hamm, R. Possibilities of water binding during cooked sausage production. Fleischwirtschaft 1973, 53, 73–79. [Google Scholar]
- Hidayat, B.; Wea, A.; Andriati, N. Physicochemical, sensory attributes and protein profile by SDS-PAGE of beef sausage substituted with texturized vegetable protein. Food Res. 2017, 2, 20–31. [Google Scholar] [CrossRef]
- Das, A.K.; Anjaneyulu, A.; Gadekar, Y.; Singh, R.; Pragati, H. Effect of full-fat soy paste and textured soy granules on quality and shelf-life of goat meat nuggets in frozen storage. Meat Sci. 2008, 80, 607–614. [Google Scholar] [CrossRef]
- Liu, H.; Xiong, Y.L.; Jiang, L.; Kong, B. Fat reduction in emulsion sausage using an enzyme-modified potato starch. J. Sci. Food Agric. 2008, 88, 1632–1637. [Google Scholar] [CrossRef]
- Paternina, A.L.; Salcedo, J.G.; Romero, P.E.; Paula, C.D. Effect of Addition of Starch and Yam Flour on the Sensory and Instrumental Properties of Low Fat Sausages. Adv. J. Food Sci. Technol. 2018, 15, 166–178. [Google Scholar] [CrossRef]
- Pătraşcu, L.; Ianiţchi, D.; Dobre, I.; Alexe, P. Effect of tumbling time and injection rate on the processing characteristics, ten-derness and color of pork biceps femoris muscle. Ann. Univ. Dunărea Jos Galaţi, Fascicle VI Food Technol. 2011, 35, 9–18. Available online: https://www.researchgate.net/publication/234137180 (accessed on 27 July 2022).
- Cucoaneş, D.I. Research on Chemical and Enzymatic Tenderization of Meat. Doctoral Dissertation, Dunărea de Jos University, Galaţi, Romania, 2009. Available online: http://10.11.10.50/xmlui/handle/123456789/2629 (accessed on 11 August 2022).
- Ianiţchi, D.; Diaconescu, C.; Vlad, I.; Nistor, L.; Hodoşan, C.; Marin, M.; Maftei, M. The influence of auxiliary materials on hardness, heat treatment losses and sensory properties of the meat products. Sci. Pap. Ser. D Anim. Sci. 2019, LXII, 296–301. Available online: http://animalsciencejournal.usamv.ro/pdf/2019/issue_2/Art47.pdf (accessed on 21 September 2022).
- Şükrü, K.; Osman, K. The Effects of Cereal and Legume Flours on the Quality Characteristics of Beef Patties. Kafkas Univ. Vet. Fak. Derg. 2012, 18, 725–730. [Google Scholar] [CrossRef] [Green Version]
- Genccelep, H.; Saricaoglu, F.T.; Anil, M.; Agar, B.; Turhan, S. The effect of starch modification and concentration on steady-state and dynamic rheology of meat emulsions. Food Hydrocoll. 2015, 48, 135–148. [Google Scholar] [CrossRef]
- Bushway, A.A.; Belyea, P.R.; True, R.H.; Work, T.M.; Russell, D.O.; McGann, D.F. Potato Starch and Flour in Frankfurters: Effect on Chemical and Sensory Properties, and Total Plate Counts. J. Food Sci. 1982, 47, 402–404. [Google Scholar] [CrossRef]
- Hachmeister, K.; Herald, T. Thermal and rheological properties and textural attributes of reduced-fat turkey batters. Poult. Sci. 1998, 77, 632–638. [Google Scholar] [CrossRef]
- Ben Akacha, B.; Švarc-Gajić, J.; Elhadef, K.; Ben Saad, R.; Brini, F.; Mnif, W.; Smaoui, S.; Ben Hsouna, A. The Essential Oil of Tunisian Halophyte Lobularia maritima: A Natural Food Preservative Agent of Ground Beef Meat. Life 2022, 12, 1571. [Google Scholar] [CrossRef]
- Ben Hsouna, A.; Michalak, M.; Ben Akacha, B.; Dhifi, W.; Ben Saad, R.; Brini, F.; Mnif, W. Assessment of the phytochemical composition, antimicrobial activity and anti-inflammatory effects of Lobularia maritima extracts on lipopolysaccharide-stimulated RAW 264.7 cells and their capacity to extend the shelf life of raw minced beef. J. Funct. Foods 2022, 99, 105327. [Google Scholar] [CrossRef]

| Ingredients, g | Samples | |||||
|---|---|---|---|---|---|---|
| B | BSPI | BCh | BL | BCs | BSt | |
| Beef | 70 | 70 | 70 | 70 | 70 | 70 |
| Fat | 30 | 30 | 30 | 30 | 30 | 30 |
| Water | 20 | 28 | 28 | 28 | 28 | 28 |
| NaCl | 2 | 2 | 2 | 2 | 2 | 2 |
| Polyphosphates | 0.3 | 0.3 | 0.3 | 0.3 | 0.3 | 0.3 |
| NaNO2 | 0.015 | 0.015 | 0.015 | 0.015 | 0.015 | 0.015 |
| Chickpea flour | - | - | 2 | - | - | - |
| Lupine protein concentrate | - | - | - | 2 | - | - |
| Soy protein isolate | - | 2 | - | - | - | - |
| Sodium caseinate | - | - | - | - | 2 | - |
| Starch | - | - | - | - | - | 2 |
| Sample | Denaturation Temperature (D.T.) | Delta Value, ° | G’ Value, Pa | ||
|---|---|---|---|---|---|
| 5 °C—D.T. | D.T.—70 °C | 5 °C—D.T. | D.T.—70 °C | ||
| B | 47.9 ± 0.23 b | 16.39 ± 0.21 a | 18.22 ± 0.10 a | 1353.4 ± 9.8 a | 1057.8 ± 8.6 c |
| BSPI | 46.7 ± 0.26 b | 18.18 ± 0.25 b | 16.12 ± 0.47 b | 1732.8 ± 2.7 b | 1068.0 ± 3.7 c |
| BCh | 50.3 ± 0.34 b | 16.74 ± 0.30 a | 15.50 ± 0.20 b | 2496.4 ± 40.5 c | 1606.2 ± 20.2 d |
| BL | 47.4 ± 0.75 b | 16.75 ± 0.19 a | 15.86 ± 0.11 b | 1883.1 ± 23.7 d | 1693.4 ± 42.1 d |
| BCs | 42.6 ± 0.16 a | 30.13 ± 0.37 c | 17.78 ± 0.34 a | 646.4 ± 6.4 e | 864.0 ± 24.1 b |
| BSt | 42.6 ± 0.88 a | 31.63 ± 0.03 d | 17.98 ± 0.35 a | 571.4 ± 6.8 e | 636.6 ± 16.1 a |
| Parameter | Composition | |||||
|---|---|---|---|---|---|---|
| B | BSPI | BCh | BL | BCs | BSt | |
| Cutting force, kgf | 2.83 ± 0.038 e | 2.65 ± 0.023 c | 2.13 ± 0.043 d | 2.52 ± 0.013 d | 1.65 ± 0.006 b | 1.08 ± 0.002 a |
| Cooking losses, % | 11.89 ± 1.138 d | 9.07 ± 0.115 c | 7.69 ± 0.163 b | 8.64 ± 0.143 c | 5.31 ± 0.152 a | 4.54 ± 0.188 a |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ianiţchi, D.; Pătraşcu, L.; Cercel, F.; Dragomir, N.; Vlad, I.; Maftei, M. The Effect of Protein Derivatives and Starch Addition on Some Quality Characteristics of Beef Emulsions and Gels. Agriculture 2023, 13, 772. https://doi.org/10.3390/agriculture13040772
Ianiţchi D, Pătraşcu L, Cercel F, Dragomir N, Vlad I, Maftei M. The Effect of Protein Derivatives and Starch Addition on Some Quality Characteristics of Beef Emulsions and Gels. Agriculture. 2023; 13(4):772. https://doi.org/10.3390/agriculture13040772
Chicago/Turabian StyleIaniţchi, Daniela, Livia Pătraşcu, Floricel Cercel, Nela Dragomir, Iulian Vlad, and Marius Maftei. 2023. "The Effect of Protein Derivatives and Starch Addition on Some Quality Characteristics of Beef Emulsions and Gels" Agriculture 13, no. 4: 772. https://doi.org/10.3390/agriculture13040772






