Growth, Solute Accumulation, and Ion Distribution in Sweet Sorghum under Salt and Drought Stresses in a Brazilian Potiguar Semiarid Area
Abstract
1. Introduction
2. Materials and Methods
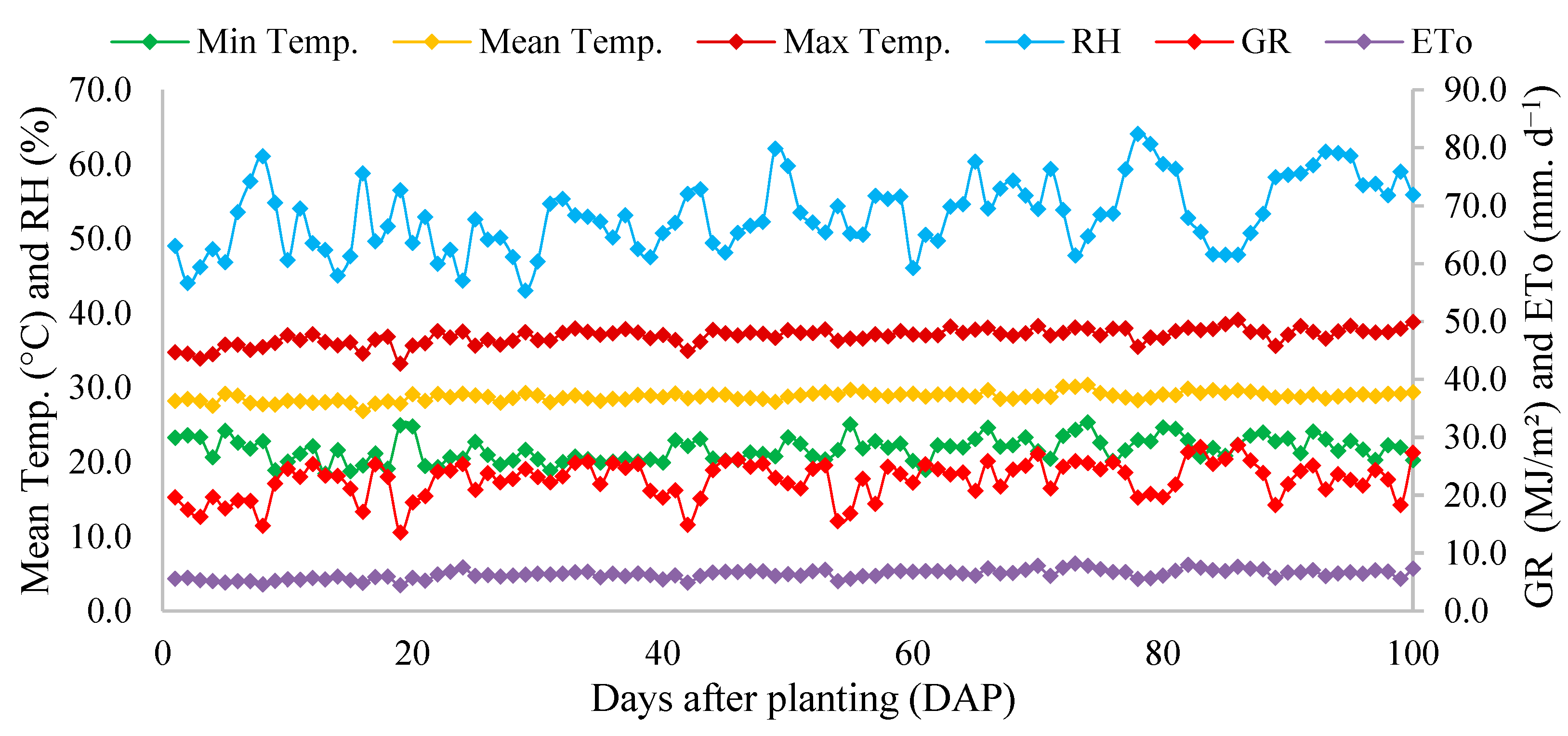
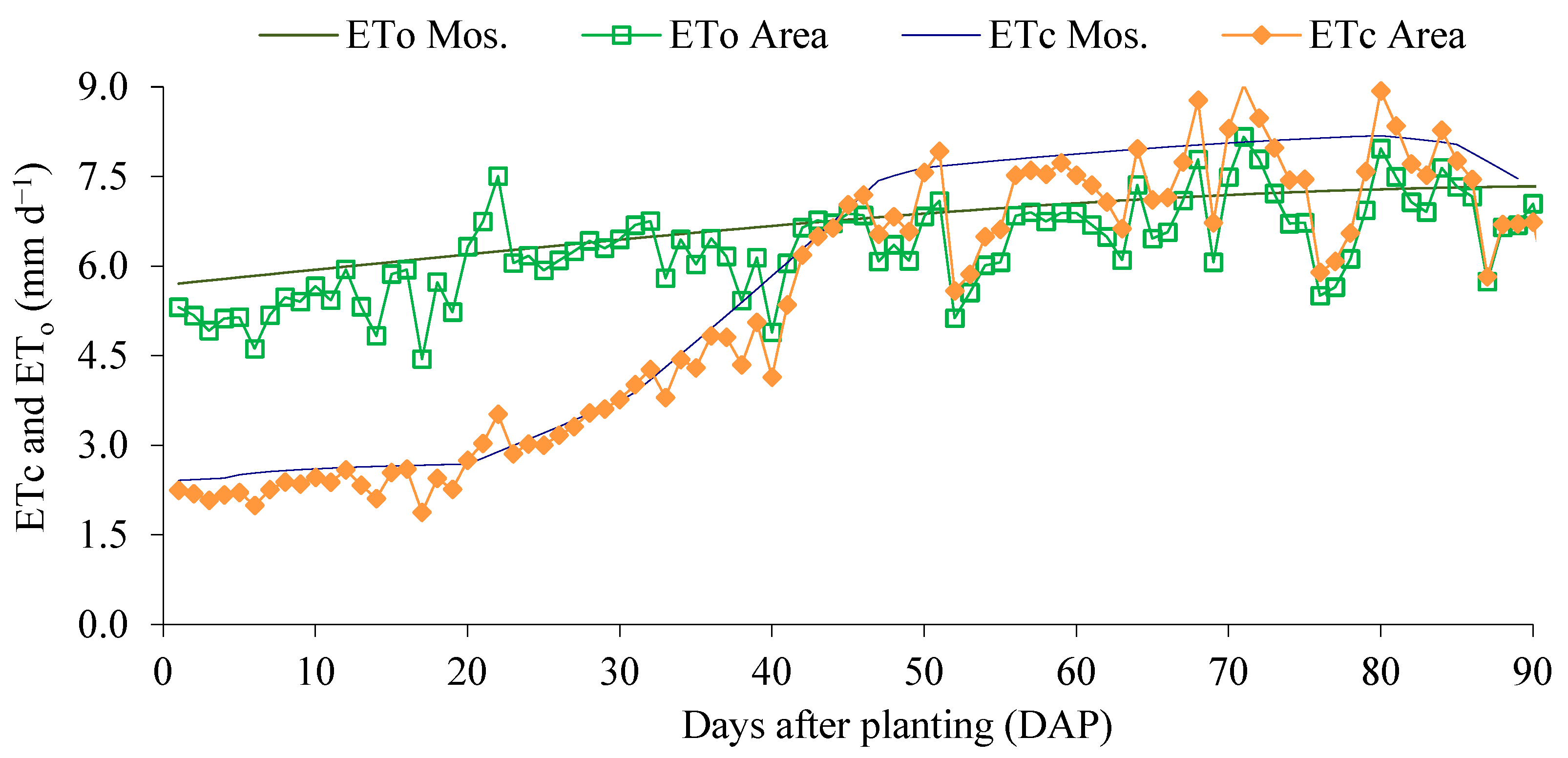
2.1. Location and Characterization of the Experimental Area
2.2. Plant Material, Experimental Design, and Treatments
2.3. Growth Parameters
2.4. Electrolyte Leakage and Relative Water Content
2.5. Proline, Total Amino Acids, and Total Soluble Sugars
2.6. Concentration and Content of Na+, K+, and Cl−
2.7. Statistical Analysis
3. Results
3.1. Growth Parameters
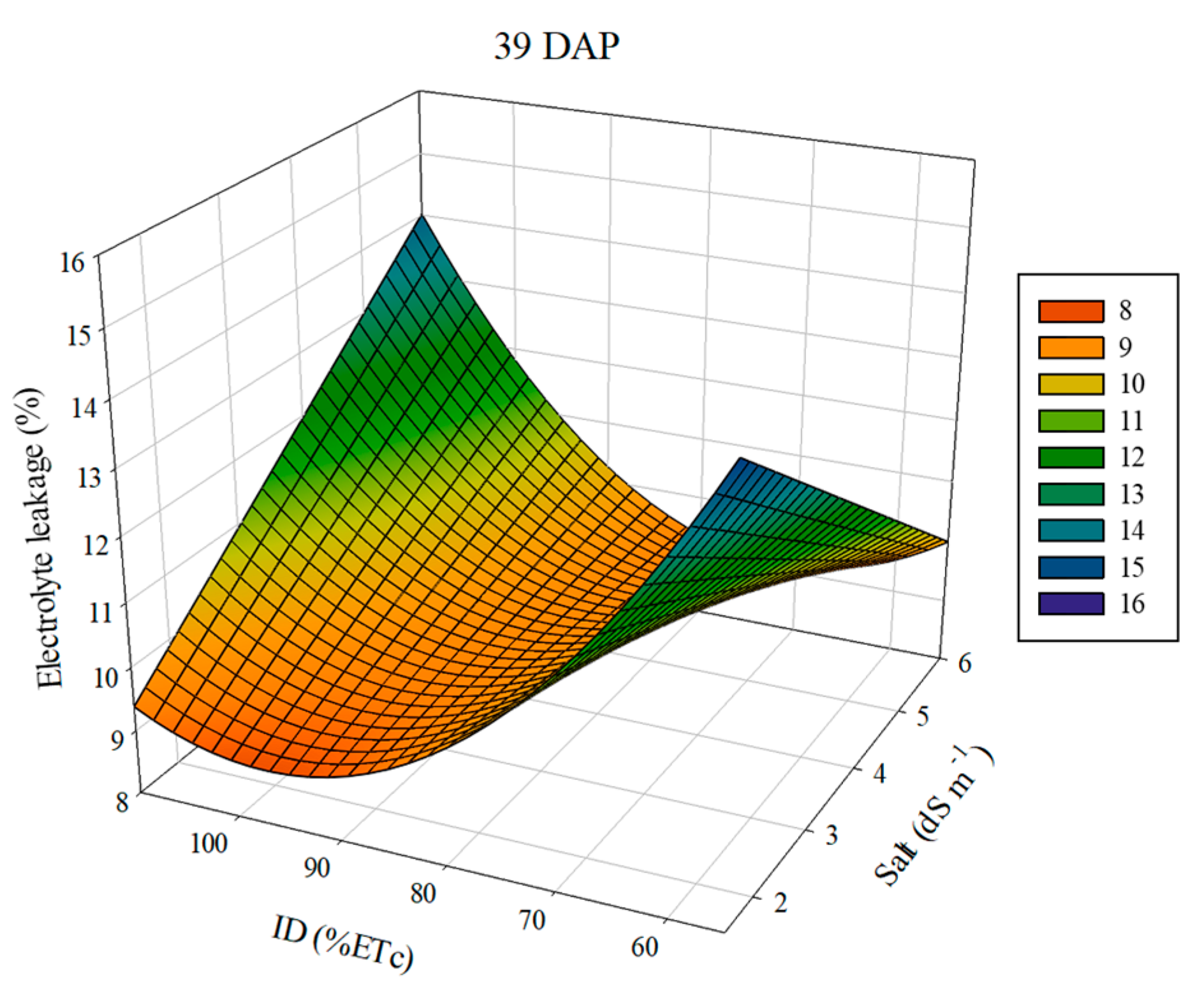
3.2. Electrolyte Leakage and Relative Water Content
3.3. Proline, Total Amino Acids, and Total Soluble Sugars
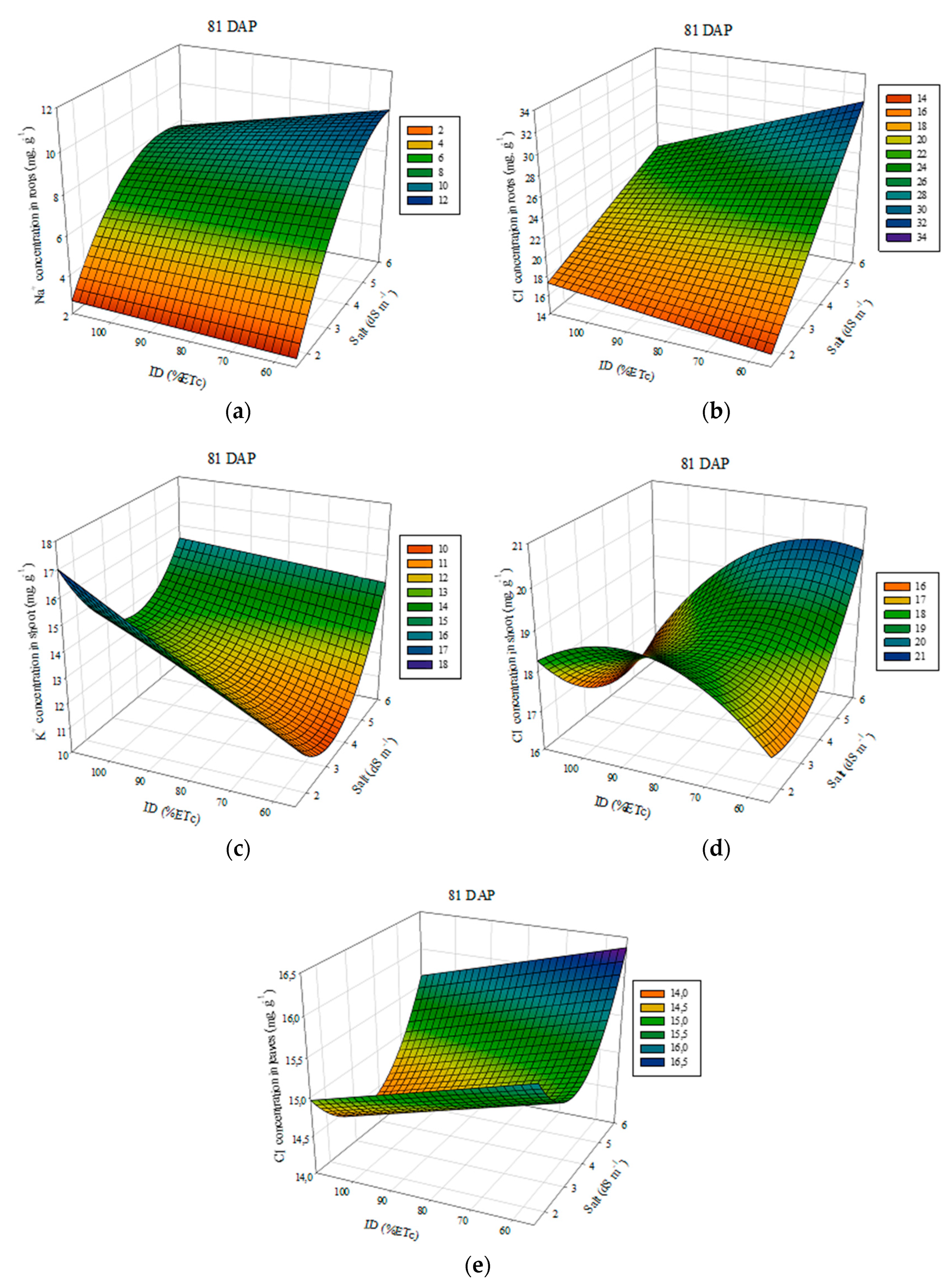
3.4. Concentration and Content of Na+, K+, and Cl− at Plant Tissue
3.5. Pearson Correlation Matrix
3.6. Soil Parameters
3.6.1. Soil Salinity
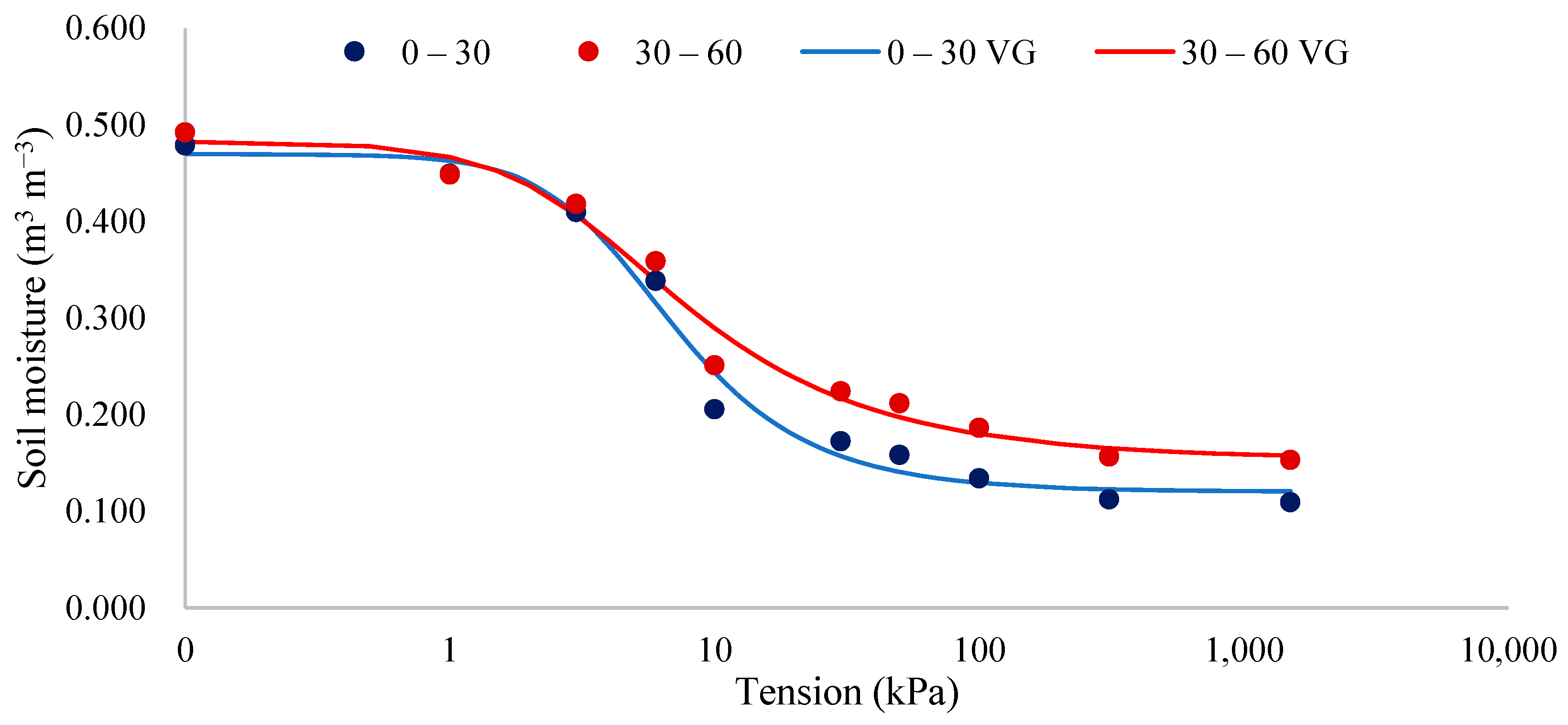
3.6.2. Soil Water Retention
4. Discussion
4.1. Concentration and Content of Na+, K+, and Cl−
4.2. Growth Parameters
4.3. Electrolyte Leakage and Relative Water Content
4.4. Proline, Total Amino Acids, and Total Soluble Sugars
4.5. Pearson Correlation Matrix
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Boretti, A.; Rosa, L. Reassessing the projections of the World Water Development Report. Npj Clean. Water 2019, 2, 15. [Google Scholar] [CrossRef]
- UNESCO World Water Assessment. The United Nations World Water Development Report 2022—Groundwater: Making the Invisible Visible, 1st ed.; UNESCO: Paris, France, 2022; 225p. [Google Scholar]
- FAO. The State of the World’s Land and Water Resources for Food and Agriculture 2021—Systems at Breaking Point, 1st ed.; FAO: Rome, Italy, 2022; 393p. [Google Scholar]
- Dar, R.A.; Dar, E.A.; Kaur, A.; Phutela, U.G. Sweet sorghum-a promising alternative feedstock for biofuel production. Renew. Sustain. Energy Rev. 2018, 82, 4070–4090. [Google Scholar] [CrossRef]
- Ayer, R.S.; Westcot, D.W. Salinity Problems. In Water Quality for Agriculture, 1st ed.; FAO: Rome, Italy, 1985; pp. 13–58. [Google Scholar]
- Motsi, H.; Molapo, M.; Phiri, E.E. A review of the adaptive capacity of sweet sorghum to improve food security and poverty alleviation in sub-Saharan Africa. S. Afr. J. Bot. 2022, 150, 323–329. [Google Scholar] [CrossRef]
- Nxele, N.; Klein, A.; Ndimba, B.K. Drought and salinity stress alters ROS accumulation, water retention, and osmolyte content in sorghum plants. S. Afr. J. Bot. 2017, 108, 261–266. [Google Scholar] [CrossRef]
- Silva, M.L.S.; Sousa, H.G.; Silva, M.L.S.; Lacerda, C.F.; Gomes-Filho, E. Growth and photosynthetic parameters of saccharine sorghum plants subjected to salinity. Acta Sci. Agron. 2019, 41, 42607. [Google Scholar] [CrossRef]
- Gupta, A.; Bano, A.; Rai, S.; Mishra, R.; Singh, M.; Sharma, S.; Pathak, N. Mechanistic insights of plant-microbe interaction towards drought and salinity stress in plants for enhancing the agriculture productivity. Plant Stress 2022, 4, 100073. [Google Scholar] [CrossRef]
- Yang, Z.; Li, J.L.; Liu, L.N.; Xie, Q.; Sui, N. Photosynthetic Regulation Under Salt Stress and Salt-Tolerance Mechanism of Sweet Sorghum. Front. Plant Sci. 2020, 10, 1722. [Google Scholar] [CrossRef]
- Martins, A.C.; Larré, C.F.; Bortolini, F.; Borella, J.; Eichholz, R.; Delias, D.; Amarante, L. Tolerância ao déficit hídrico: Adaptação diferencial entre espécies forrageiras. Iheringia Série Botânica 2018, 73, 228–239. [Google Scholar] [CrossRef]
- Wang, Z.; Wei, Y.; Zhao, Y.; Wang, Y.; Zou, F.; Huang, S.; Yang, X.; Xu, Z.; Hu, H. Physiological and transcriptional evaluation of sweet sorghum seedlings in response to single and combined drought and salinity stress. S. Afr. J. Bot. 2022, 146, 459–471. [Google Scholar] [CrossRef]
- Ma, Y.; Dias, M.C.; Freitas, H. Drought and Salinity Stress Responses and Microbe-Induced Tolerance in Plants. Front. Plant Sci. 2020, 11, 1750. [Google Scholar] [CrossRef]
- Ogbaga, C.C.; Stepien, P.; Dyson, B.C.; Rattray, N.J.W.; Ellis, D.I.; Goodacre, R.; Johnson, G.N. Biochemical Analyses of Sorghum Varieties Reveal Differential Responses to Drought. PLoS ONE 2016, 11, 0154423. [Google Scholar] [CrossRef] [PubMed]
- Dinkeloo, K.; Boyd, S.; Pilot, G. Update on amino acid transporter functions and on possible amino acid sensing mechanisms in plants. Semin. Cell Dev. Biol. 2018, 74, 105–113. [Google Scholar] [CrossRef] [PubMed]
- Punia, H.; Tokas, J.; Mor, V.S.; Bhuker, A.; Malik, A.; Singh, N.; Satpal; Alsahli, A.A.; Hefft, D.I. Deciphering reserve mobilization, antioxidant potential, and expression analysis of starch synthesis in sorghum seedlings under salt stress. Plants 2021, 10, 2463. [Google Scholar] [CrossRef] [PubMed]
- Ukwatta, J.; Pabuayon, I.C.M.; Park, J.; Chen, J.; Chai, X.; Zhang, H.; Zhu, J.; Xin, Z.; Shi, H. Comparative physiological and transcriptomic analysis reveals salinity tolerance mechanisms in Sorghum bicolor (L.) Moench. Planta 2021, 254, 98. [Google Scholar] [CrossRef] [PubMed]
- Almodares, A.; Hadi, M.R.; Kholdebarin, B.; Samedani, B.; Kharazian, Z. The response of sweet sorghum cultivars to salt stress and accumulation of Na+, Cl− and K+ ions in relation to salinity. J. Environ. Biol. 2014, 35, 733–739. [Google Scholar] [PubMed]
- Marta, A.; Mancini, M.; Orlando, F.; Natali, F.; Capecchi, L.; Orlandini, S. Sweet sorghum for bioethanol production: Crop responses to different water stress levels. Biomass Bioenergy 2014, 64, 211–219. [Google Scholar] [CrossRef]
- Dubreuil, V.; Fante, K.P.; Planchon, O.; Neto, J.L.S. Os tipos de climas anuais no Brasil: Uma aplicação da classificação de Köppen de 1961 a 2015. Confins 2018. [Google Scholar] [CrossRef]
- EMPARN. Relatório Pluviométrico. 2022. Available online: https://meteorologia.emparn.rn.gov.br/relatorios/relatorios-pluviometricos (accessed on 27 February 2023).
- Mendes, K.R.; Portela, J.C.; Gondim, J.E.F.; Ribeiro, M.A.; Medeiros, J.F.; Queiroz, G.C.M. Physical, chemical and structural attributes of soil in agroecosystems in the Brazilian Semiarid region. Rev. Ciência Agronômica 2022, 53, e20207630. [Google Scholar]
- Teixeira, P.C.; Donagemma, G.K.; Fontana, A.; Teixeira, W.G. Manual de Métodos de Análise de Solo, 3rd ed.; EMBRAPA: Brasília, Brazil, 2017; 574p. [Google Scholar]
- Neves, A.L.A.; Santos, R.D.; Pereira, L.G.R.; Oliveira, G.F.; Scherer, C.B.; Verneque, R.S.; McAllister, T. Agronomic characteristics, silage quality, intake and digestibility of five new Brazilian sorghum cultivars. J. Agric. Sci. 2015, 153, 371–380. [Google Scholar] [CrossRef]
- Medeiros, J.F. Qualidade da Água de Irrigação e Evolução da Salinidade nas Propriedades Assistidas Pelo “GAT” Nos Estados do RN. Ph.D. Thesis, Universidade Federal da Paraíba, Paraíba, Brazil, 1992. [Google Scholar]
- Allen, R.G.; Pereira, L.S.; Raes, D. Crop Evapotranspiration (Guidelines for Computing Crop Water Requirements), 1st ed.; FAO: Rome, Italy, 2004. [Google Scholar]
- Stickler, F.C.; Wearden, S.; Pauli, A.W. Leaf Area determination in Grain Sorghum. Agron. J. 1961, 53, 187–188. [Google Scholar] [CrossRef]
- Weatherley, P.E. Studies in the water relations of the cotton plant. New. Phytol. 1950, 49, 81–97. [Google Scholar] [CrossRef]
- Bates, L.S.; Waldren, R.P.; Teare, I.D. Rapid determination of free proline for water-stress studies. Plant. Soil. 1973, 39, 205–207. [Google Scholar] [CrossRef]
- Yemm, E.W.; Cocking, E.C.; Ricketts, R.E. The determination of amino-acids with ninhydrin. Analyst 1955, 80, 209–214. [Google Scholar] [CrossRef]
- Yemm, E.W.; Willis, A.J. The estimation of carbohydrates in plant extracts by anthrone. Biochem. J. 1954, 57, 508. [Google Scholar] [CrossRef] [PubMed]
- EMBRAPA. Manual de Análises Químicas de Solos, plantas e Fertilizante, 1st ed.; EMBRAPA: Brasília, Brasil, 2009; 370p. [Google Scholar]
- Silva, E.B.; Nogueira, F.D.; Guimarães, P.T.G. BT 31—Análise de Cloreto em Tecido Vegetal, 1st ed.; Editora UFLA: Lavras, Brasil, 2021. [Google Scholar]
- Posit|The Open-Source Data Science Company. Available online: https://posit.co/ (accessed on 27 February 2023).
- Statistica. Available online: https://www.statistica.com/en/ (accessed on 27 February 2023).
- Mansour, M.M.F.; Emam, M.M.; Salama, K.H.A.; Morsy, A.A. Sorghum under saline conditions: Responses, tolerance mechanisms, and management strategies. Planta 2021, 254, 24. [Google Scholar] [CrossRef] [PubMed]
- Sayyad-Amin, P.; Borzouei, A.; Jahansooz, M.R.; Parsaeiyan, M. Root biochemical responses of grain and sweet-forage sorghum cultivars under saline conditions at vegetative and reproductive phases. Rev. Bras. De Bot. 2016, 39, 115–122. [Google Scholar] [CrossRef]
- Hussain, S.; Hussain, S.; Ali, B.; Ren, X.; Chen, X.; Li, Q.; Saqib, M.; Ahmad, N. Recent progress in understanding salinity tolerance in plants: Story of Na+/K+ balance and beyond. Plant. Physiol. Biochem. 2021, 160, 239–256. [Google Scholar] [CrossRef]
- Gull, M.; Kausar, A. Screening The Variability in Salt Tolerance of Sorghum Bicolor L. By Nutrients Uptake and Growth Analysis of Four Genotypes. Pharmacophore 2019, 10, 43–50. [Google Scholar]
- Wang, M.; Zheng, Q.; Shen, Q.; Guo, S.; River, Y.; Key, J. The Critical Role of Potassium in Plant Stress Response. Int. J. Mol. Sci. 2013, 14, 7370–7390. [Google Scholar] [CrossRef]
- Kausar, A.; Gull, M. Influence of salinity stress on the uptake of magnesium, phosphorus, and yield of salt susceptible and tolerant sorghum cultivars (Sorghum bicolor L.). J. Appl. Biol. Biotechnol. 2019, 7, 53–58. [Google Scholar] [CrossRef]
- Perrier, L.; Rouan, L.; Jaffuel, S.; Clément-Vidal, A.; Roques, S.; Soutiras, A.; Baptiste, C.; Bastianelli, D.; Fabre, D.; Dubois, C.; et al. Plasticity of Sorghum Stem Biomass Accumulation in Response to Water Deficit: A Multiscale Analysis from Internode Tissue to Plant Level. Front. Plant Sci. 2017, 8, 1516. [Google Scholar] [CrossRef]
- Abreha, K.B.; Enyew, M.; Carlsson, A.S.; Vetukuri, R.R.; Feyissa, T.; Motlhaodi, T.; Ng’uni, D.; Geleta, M. Sorghum in dryland: Morphological, physiological, and molecular responses of sorghum under drought stress. Planta 2021, 255, 20. [Google Scholar] [CrossRef] [PubMed]
- Mansour, M.M.F.; Salama, K.H.A. Cellular basis of salinity tolerance in plants. Environ. Exp. Bot. 2004, 52, 113–122. [Google Scholar] [CrossRef]
- Hniličková, H.; Hnilička, F.; Orsák, M.; Hejnák, V. Effect of salt stress on growth, electrolyte leakage, Na+ and K+ content in selected plant species. Plant Soil Environ. 2019, 65, 90–96. [Google Scholar] [CrossRef]
- Demidchik, V.; Straltsova, D.; Medvedev, S.S.; Pozhvanov, G.A.; Sokolik, A.; Yurin, V. Stress-induced electrolyte leakage: The role of K+-permeable channels and involvement in programmed cell death and metabolic adjustment. J. Exp. Bot. 2014, 65, 1259–1270. [Google Scholar] [CrossRef] [PubMed]
- Hasan, S.A.; Rabei, S.H.; Nada, R.M.; Abogadallah, G.M. Water use efficiency in the drought-stressed sorghum and maize in relation to expression of aquaporin genes. Biol. Plant 2017, 61, 127–137. [Google Scholar] [CrossRef]
- Weimberg, R.; Lerner, H.R.; Poljakoff-Mayber, A. Changes in growth and water-soluble solute concentrations in Sorghum bicolor stressed with sodium and potassium salts. Physiol. Plant 1984, 62, 472–480. [Google Scholar] [CrossRef]
- Uribe, R.A.; Silvério, P.C.; Costa, G.H.; Nogueira, L.C.; Leite, L.A. Chloride levels in biomass sorghum due to fertilization sources. Biomass Bioenergy 2020, 143, 105845. [Google Scholar] [CrossRef]


| Layer | Soil Physics | ||||||
|---|---|---|---|---|---|---|---|
| Coarse Sand | Fine Sand | Silt | Clay | Soil Density | |||
| cm | g g−1 | g cm−3 | |||||
| 0–30 | 0.544 | 0.223 | 0.044 | 0.190 | 1.64 | ||
| 30–60 | 0.480 | 0.218 | 0.052 | 0.250 | 1.62 | ||
| Soil chemistry | |||||||
| ECex | pH | Ca2+ | Mg2+ | Na+ | K+ | P | |
| dS m−1 | cmolc dm−3 | mg dm−3 | |||||
| 0–30 | 0.90 | 7.50 | 4.80 | 1.50 | 0.35 | 0.46 | 15 |
| 30–60 | 0.80 | 7.20 | 5.10 | 1.60 | 0.39 | 0.45 | 8 |
| 39 DAP | 60 DAP | 81 DAP | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| SV | DF | LA | SD | SH | TH | ADM | LA | SD | SH | TH | ADM | SD | SH | TH | ADM |
| Block | 3 | 1.69 | 3.10 * | 1.61 | 1.61 | 3.00 | 0.57 | 0.89 | 1.39 | 1.38 | 0.23 | 1.27 | 0.24 | 0.67 | 2.83 |
| Salt | 2 | 3.41 * | 3.66 * | 15.16 ** | 15.16 ** | 2.47 | 5.00 * | 0.32 | 6.71 ** | 3.15 | 0.85 | 0.23 | 3.46 * | 0.33 | 1.54 |
| ID | 2 | 10.74 ** | 3.12 | 52.86 ** | 52.86 ** | 4.24 * | 0.41 | 0.09 | 37.15 ** | 34.21 ** | 0.08 | 0.69 | 20.62 ** | 3.29 | 0.91 |
| Salt × ID | 4 | 0.44 | 0.05 | 0.73 | 0.73 | 1.99 | 0.09 | 0.54 | 0.71 | 0.68 | 1.44 | 1.09 | 0.48 | 1.22 | 4.31 ** |
| C. V. (%) | 14.2 | 6.1 | 10.1 | 5.2 | 17.0 | 19.4 | 5.9 | 6.6 | 5.8 | 14.3 | 7.2 | 5.0 | 17.2 | 10.0 | |
| Mean values | |||||||||||||||
| cm² | mm | cm | g | cm² | mm | cm | g | mm | cm | g | |||||
| 1.5 dS m−1 | 2309.2 a | 17.87 ab | 68.0 a | 132.1 a | 51.09 a | 2392.0 b | 17.81 a | 224.7 a | 249.3 a | 133.08 a | 16.69 a | 211.6 a | 243.0 a | 530.55 | |
| 3.8 dS m−1 | 2180.2 ab | 18.07 a | 63.2 a | 127.4 a | 45.63 a | 2512.3 ab | 18.04 a | 219.7 a | 246.5 a | 143.53 a | 16.47 a | 207.1 ab | 256.9 a | 569.43 | |
| 6.0 dS m−1 | 1984.6 b | 16.96 b | 54.2 b | 118.0 b | 44.25 a | 3014.1 a | 18.15 a | 204.2 b | 235.6 a | 139.28 a | 16.37 a | 200.6 b | 248.0 a | 556.23 | |
| 55% ETc | 1852.9 b | 17.00 a | 48.1 c | 111.5 c | 42.52 b | 2719.6 a | 18.09 a | 190.0 c | 218.4 c | 140.34 a | 16.67 a | 194.6 b | 237.4 a | 545.14 | |
| 83% ETc | 2190.7 a | 17.99 a | 63.2 b | 127.4 b | 46.47 ab | 2663.9 a | 17.91 a | 218.5 b | 247.3 b | 137.22 a | 16.18 a | 203.7 b | 235.3 a | 569.44 | |
| 110% ETc | 2430.5 a | 17.91 a | 74.1 a | 138.9 a | 51.98 a | 2534.8 a | 18.00 a | 240.1 a | 265.7 a | 138.34 a | 16.67 a | 221.0 a | 275.2 a | 541.63 | |
| ADM_81 | ||||
|---|---|---|---|---|
| g | ||||
| 55% ETc | 83% ETc | 110% ETc | Mean | |
| 1.5 dS m−1 | 176.74 aA | 183.93 aA | 169.88 bA | 530.55 |
| 3.8 dS m−1 | 175.38 aB | 184.14 aAB | 209.91 aA | 569.43 |
| 6.0 dS m−1 | 193.02 aAB | 201.37 aA | 161.84 bB | 556.23 |
| Mean | 545.14 | 569.44 | 541.63 | |
| No tested equations were significant | ||||
| 39 DAP | 60 DAP | ||||
|---|---|---|---|---|---|
| SV | DF | EL | RWC | EL | RWC |
| Block | 3 | 0.79 | 2.95 | 14.21 ** | 0.75 |
| Salt | 2 | 0.48 | 1.89 | 3.91 * | 4.09 * |
| ID | 2 | 5.41 * | 0.85 | 2.67 | 0.31 |
| Salt × ID | 4 | 4.60 ** | 3.72 * | 1.66 | 1.32 |
| C. V. (%) | 19.7 | 3.2 | 11.4 | 2.9 | |
| Mean values | |||||
| % | % | ||||
| 1.5 dS m−1 | 11.45 a | 89.87 a | 22.12 b | 95.87 a | |
| 3.8 dS m−1 | 10.68 a | 90.54 a | 22.93 ab | 93.26 ab | |
| 6.0 dS m−1 | 11.44 a | 92.09 a | 25.06 a | 92.92 b | |
| 55% ETc | 12.36 | 90.21 a | 24.41 a | 94.19 a | |
| 83% ETc | 9.52 | 90.61 a | 23.72 a | 94.35 a | |
| 110% ETc | 11.70 | 91.68 a | 21.98 a | 93.51 a | |
| EL_39 | RWC_39 | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 55% ETc | 83% ETc | 110% ETc | Mean | 55% ETc | 83% ETc | 110% ETc | Mean | ||
| 1.5 dS m−1 | 15.26 aA | 9.26 aB | 9.84 bB | 11.45 | 1.5 dS m−1 | 88.62 bA | 90.18 aA | 90.81 aA | 89.87 |
| 3.8 dS m−1 | 11.60 abA | 9.33 aA | 11.13 abA | 10.68 | 3.8 dS m−1 | 87.27 bB | 92.43 aA | 91.94 aAB | 90.54 |
| 6.0 dS m−1 | 10.21 bB | 9.98 aB | 14.13 aA | 11.44 | 6.0 dS m−1 | 94.75 aA | 89.22 aB | 92.30 aAB | 92.09 |
| Mean | 12.36 | 9.52 | 11.70 | Mean | 90.21 | 90.61 | 91.68 | ||
| EL_39 = 44.72 *** −3.12 EC *** − 6.99.10−1 ID *** + 3.30.10−3 ID² ** + 3.78.10−2 ECID *** (R² = 0.95) | No tested equations were significant | ||||||||
| SV | DF | Proline | Total Amino Acids | Total Soluble Sugars |
|---|---|---|---|---|
| Block | 3 | 2.87 ns | 1.10 ns | 0.93 ns |
| Salt | 2 | 0.72 ns | 0.08 ns | 1.56 ns |
| ID | 2 | 0.07 ns | 0.36 ns | 2.73 ns |
| Salt × ID | 4 | 1.21 ns | 0.26 ns | 0.87 ns |
| C. V. (%) | 2.6 | 11.5 | 18.1 | |
| General mean | ||||
| µmol g FM−1 | µg g FM−1 | |||
| 3.96 | 12.21 | 17.97 | ||
| 39 DAP | 60 DAP | 81 DAP | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| SV | DF | Na+ | K+ | Cl− | Na+ | K+ | Cl− | Na+ | K+ | Cl− |
| Roots | ||||||||||
| mg g−1 | mg g−1 | mg g−1 | ||||||||
| Block | 3 | 0.72 | 0.43 | 0.05 | 1.66 | 0.34 | 0.68 | 0.33 | 0.69 | 2.17 |
| Salt | 2 | 129.43 ** | 10.77 ** | 17.75 ** | 223.17 ** | 1.30 | 97.08 ** | 184.44 ** | 16.47 ** | 42.73 ** |
| ID | 2 | 1.30 | 1.90 | 16.24 ** | 0.68 | 6.80 ** | 1.65 | 4.78 * | 1.69 | 3.46 * |
| Salt × ID | 4 | 12.62 ** | 2.76 | 3.53 * | 0.64 | 11.67 ** | 16.83 ** | 3.49 * | 2.63 | 3.34 * |
| C. V. (%) | 16.4 | 14.9 | 11.9 | 17.7 | 12.0 | 11.7 | 13.5 | 16.9 | 13.5 | |
| 1.5 dS m−1 | 1.64 | 13.19 a | 13.56 | 1.00 c | 10.00 a | 10.42 | 2.55 | 10.71 a | 15.92 | |
| 3.8 dS m−1 | 4.93 | 10.30 b | 16.58 | 6.89 b | 9.48 a | 17.44 | 7.73 | 7.17 c | 22.50 | |
| 6.0 dS m−1 | 5.92 | 13.41 a | 18.11 | 9.90 a | 10.24 a | 21.08 | 8.89 | 8.87 b | 26.92 | |
| 55% ETc | 4.06 a | 13.00 a | 18.64 | 5.94 a | 10.00 | 16.75 a | 6.98 | 9.49 a | 23.00 | |
| 83% ETc | 4.42 a | 12.35 a | 14.67 | 5.68 a | 10.75 | 15.50 a | 6.30 | 8.90 a | 22.33 | |
| 110% ETc | 4.00 a | 11.54 a | 14.94 | 6.18 a | 8.97 | 16.69 a | 5.90 | 8.36 a | 20.00 | |
| Shoot | ||||||||||
| SV | DF | mg g−1 | mg g−1 | mg g−1 | ||||||
| Block | 3 | 1.54 | 6.05 | 0.23 | 0.15 | 2.59 | 3.20* | 0.32 | 0.72 | 1.47 |
| Salt | 2 | 42.52 ** | 1.45 | 0.43 | 78.00 ** | 2.83 | 0.75 | 97.64 ** | 13.05 ** | 1.60 |
| ID | 2 | 0.07 | 3.16 | 0.23 | 17.56 ** | 6.77 ** | 3.07 | 18.03 ** | 10.15 ** | 2.41 |
| Salt × ID | 4 | 1.45 | 2.41 | 0.65 | 5.85 ** | 4.90 ** | 1.74 | 13.63 ** | 4.47 ** | 3.32 * |
| C. V. (%) | 15.5 | 7.3 | 8.8 | 17.0 | 11.4 | 10.3 | 20.6 | 10.6 | 10.5 | |
| 1.5 dS m−1 | 0.55 c | 56.98 a | 46.83 a | 0.27 | 18.74 | 24.00 a | 0.15 | 14.61 | 18.08 a | |
| 3.8 dS m−1 | 0.84 b | 54.20 a | 47.17 a | 0.69 | 20.89 | 25.17 a | 0.42 | 12.26 | 17.33 a | |
| 6.0 dS m−1 | 1.01 a | 55.32 a | 48.33 a | 0.65 | 19.55 | 24.17 a | 0.60 | 15.20 | 18.72 a | |
| 55% ETc | 0.80 a | 55.38 a | 46.83 a | 0.51 | 17.85 | 23.00 a | 0.44 | 12.51 | 18.17 a | |
| 83% ETc | 0.80 a | 53.50 a | 48.00 a | 0.44 | 20.26 | 24.92 a | 0.46 | 14.38 | 18.83 a | |
| 110% ETc | 0.79 a | 57.62 a | 47.50 a | 0.65 | 21.08 | 25.42 a | 0.28 | 15.19 | 17.14 a | |
| Leaves | ||||||||||
| SV | DF | mg g−1 | mg g−1 | mg g−1 | ||||||
| Block | 3 | 0.52 | 3.22 * | 3.96 * | 1.95 | 0.56 | 6.42 ** | 1.50 | 2.70 | 7.37 ** |
| Salt | 2 | 0.90 | 0.41 | 1.57 | 5.53 * | 0.30 | 0.86 | 0.00 | 6.78 ** | 2.66 |
| ID | 2 | 1.40 | 0.32 | 0.17 | 6.79 ** | 3.03 | 2.42 | 2.01 | 0.90 | 1.44 |
| Salt × ID | 4 | 1.39 | 1.14 | 1.84 | 1.85 | 1.47 | 4.02 * | 0.37 | 0.84 | 2.93 * |
| C. V. (%) | 17.2 | 6.8 | 9.1 | 18.2 | 9.7 | 6.5 | 19.4 | 9.0 | 8.6 | |
| 1.5 dS m−1 | 0.41 a | 27.74 a | 16.83 a | 0.14 b | 13.62 a | 14.58 a | 0.12 a | 11.65 ab | 15.33 a | |
| 3.8 dS m−1 | 0.39 a | 27.08 a | 16.00 a | 0.17 ab | 14.04 a | 15.08 a | 0.12 a | 10.86 b | 14.75 a | |
| 6.0 dS m−1 | 0.43 a | 27.27 a | 15.83 a | 0.17 a | 13.82 a | 14.94 a | 0.12 a | 12.45 a | 16.00 a | |
| 55% ETc | 0.43 a | 27.70 a | 16.08 a | 0.18 a | 14.42 a | 15.28 a | 0.12 a | 11.37 a | 15.58 a | |
| 83% ETc | 0.38 a | 27.13 a | 16.17 a | 0.14 b | 13.96 a | 14.92 a | 0.13 a | 11.94 a | 15.67 a | |
| 110% ETc | 0.41 a | 27.26 a | 16.42 a | 0.15 ab | 13.09 a | 14.42 a | 0.11 a | 11.65 a | 14.83 a | |
| 39 | DAP | 60 | DAP | 81 | DAP | ||||
|---|---|---|---|---|---|---|---|---|---|
| Trat | CNa+ | CK+ | CCl− | CNa+ | CK+ | CCl− | CNa+ | CK+ | CCl− |
| Roots | |||||||||
| mg | mg | mg | |||||||
| S1ID1 | 17.2 ± 1.9 | 77.0 ± 6.1 | 110.7 ± 8.4 | 12.2 ± 1.1 | 161.6 ± 11.9 | 174.6 ± 18.5 | 45.8 ± 6.1 | 223.6 ± 33.3 | 233.4 ± 33.7 |
| S1ID2 | 10.4 ± 1.7 | 113.2 ± 11.1 | 93.9 ± 4.9 | 16.8 ± 1.7 | 210.0 ± 15.1 | 169.1 ± 20.3 | 76.9 ± 5.8 | 273.6 ± 17.3 | 327.6 ± 25.7 |
| S1ID3 | 7.2 ± 0.7 | 116.1 ± 13.3 | 96.2 ± 11.5 | 24.3 ± 2.3 | 162.4 ± 22.6 | 214.6 ± 28.2 | 60.0 ± 7.8 | 260.4 ± 22.7 | 284.5 ± 15.6 |
| S2ID1 | 23.4 ± 1.4 | 61.2 ± 3.9 | 105.1 ± 4.8 | 121.3 ± 11.0 | 153.1 ± 16.6 | 316.7 ± 24.7 | 153.6 ± 6.0 | 149.0 ± 16.1 | 429.1 ± 27.8 |
| S2ID2 | 36.1 ± 3.8 | 82.3 ± 4.7 | 112.9 ± 14.9 | 102.3 ± 13.6 | 146.2 ± 5.9 | 212.2 ± 18.9 | 155.8 ± 13.3 | 171.6 ± 18.9 | 362.7 ± 13.6 |
| S2ID3 | 60.7 ± 2.5 | 91.1 ± 5.8 | 171.2 ± 10.2 | 129.4 ± 16.2 | 186.9 ± 14.9 | 368.1 ± 37.7 | 159.8 ± 22.7 | 114.1 ± 14.7 | 360.1 ± 33.8 |
| S3ID1 | 27.1 ± 2.9 | 80.9 ± 7.9 | 100.4 ± 10.0 | 174.0 ± 5.8 | 201.6 ± 16.5 | 377.3 ± 15.5 | 212.1 ± 18.9 | 190.0 ± 14.9 | 532.7 ± 43.8 |
| S3ID2 | 38.1 ± 3.7 | 62.5 ± 6.0 | 91.0 ± 3.0 | 152.8 ± 7.0 | 185.2 ± 10.1 | 377.1 ± 21.5 | 165.9 ± 11.2 | 172.2 ± 22.2 | 416.0 ± 24.5 |
| S3ID3 | 42.0 ± 4.7 | 100.4 ± 7.8 | 138.3 ± 15.1 | 136.3 ± 12.3 | 100.8 ± 10.1 | 243.8 ± 25.6 | 141.8 ± 12.5 | 154.4 ± 17.7 | 320.5 ± 15.7 |
| Shoot | |||||||||
| mg | mg | mg | |||||||
| S1ID1 | 12.6 ± 1.8 | 1175.5 ± 38.5 | 1045.7 ± 45.4 | 21.3 ± 1.9 | 1578.0 ±123.3 | 2103.7 ± 48.9 | 19.2 ± 1.6 | 1668.9 ± 151.2 | 1582.9 ± 21.7 |
| S1ID2 | 18.5 ± 2.2 | 1713.6 ± 158.5 | 1445.7 ± 164.1 | 25.2 ± 2.2 | 1664.3 ± 245.3 | 2267.2 ± 206.4 | 19.3 ± 1.9 | 2097.7 ± 252.6 | 1843.1 ± 173.3 |
| S1ID3 | 14.4 ± 2.3 | 1835.3 ± 199.3 | 1397.3 ± 169.1 | 29.8 ± 3.7 | 2110.4 ± 242.1 | 2458.4 ± 256.3 | 22.5 ± 2.2 | 2291.6 ± 160.7 | 1751.8 ± 307.8 |
| S2ID1 | 15.3 ± 1.1 | 1064.2 ± 139.2 | 876.7 ± 115.7 | 70.6 ± 3.5 | 1809.8 ± 144.5 | 2358.4 ± 198.2 | 63.1 ± 8.0 | 1410.7 ± 151.6 | 1875.9 ± 160.5 |
| S2ID2 | 16.5 ± 1.6 | 1126.6 ± 145.6 | 1087.2 ± 123.2 | 64.3 ± 7.8 | 2599.5 ± 289.4 | 2893.0 ± 166.4 | 60.7 ± 5.1 | 1975.0 ± 121.4 | 1936.5 ± 213.2 |
| S2ID3 | 24.5 ± 2.4 | 1704.8 ± 264.7 | 1402.9 ± 205.8 | 79.4 ± 9.2 | 2130.8 ± 127.0 | 2553.9 ± 67.2 | 61.0 ± 8.6 | 2098.4 ± 90.1 | 1614.5 ± 174.2 |
| S3ID1 | 22.7 ± 1.1 | 1287.5 ± 122.1 | 1075.3 ± 122.3 | 64.9 ± 7.8 | 2073.6 ± 163.9 | 2584.6 ± 278.5 | 109.1 ± 11.5 | 2291.7 ± 86.5 | 2103.5 ± 148.1 |
| S3ID2 | 21.3 ± 1.3 | 1235.7 ± 108.4 | 1102.7 ± 105.4 | 41.7 ± 1.9 | 1789.6 ± 115.0 | 2145.8 ± 82.2 | 128.2 ± 15.3 | 2318.8 ± 145.9 | 1761.7 ± 131.6 |
| S3ID3 | 26.6 ± 3.7 | 1345.8 ± 93.3 | 1235.5 ± 157.7 | 96.2 ± 8.1 | 2200.9 ± 301.0 | 2733.6 ± 252.0 | 35.5 ± 4.5 | 1878.7 ± 204.2 | 1927.4 ± 222.5 |
| Leaves | |||||||||
| mg | mg | mg | |||||||
| S1ID1 | 9.2 ± 0.6 | 589.0 ± 51.4 | 357.5 ± 24.9 | 6.7 ± 0.7 | 534.7 ± 25.2 | 586.9 ± 54.2 | 4.9 ± 0.5 | 439.8 ± 34.8 | 586.0 ± 33.8 |
| S1ID2 | 9.6 ± 1.7 | 650.3 ± 33.6 | 374.5 ± 37.5 | 4.6 ± 1.6 | 519.0 ± 61.4 | 544.7 ± 47.0 | 4.9 ± 0.4 | 481.8 ± 46.9 | 623.4 ± 52.3 |
| S1ID3 | 9.9 ± 1.3 | 720.2 ± 103.2 | 454.2 ± 53.9 | 4.3 ± 0.6 | 483.0 ± 25.2 | 515.0 ± 43.8 | 3.9 ± 0.2 | 442.7 ± 33.7 | 582.5 ± 18.2 |
| S2ID1 | 7.9 ± 1.0 | 515.8 ± 54.7 | 279.1 ± 20.4 | 6.1 ± 0.7 | 506.0 ± 52.8 | 522.6 ± 46.9 | 4.0 ± 0.4 | 378.0 ± 7.7 | 544.1 ± 34.0 |
| S2ID2 | 8.2 ± 0.6 | 574.9 ± 36.2 | 350.4 ± 33.0 | 7.1 ± 0.6 | 676.3 ± 81.0 | 740.9 ± 84.0 | 5.8 ± 0.4 | 493.6 ± 33.0 | 669.0 ± 33.5 |
| S2ID3 | 9.4 ± 0.6 | 675.6 ± 72.8 | 418.5 ± 40.0 | 6.3 ± 0.7 | 490.2 ± 19.5 | 533.0 ± 25.4 | 4.2 ± 0.4 | 433.4 ± 17.4 | 552.6 ± 56.4 |
| S3ID1 | 10.1 ± 0.8 | 662.0 ± 83.1 | 383.0 ± 45.4 | 8.4 ± 0.4 | 636.0 ± 73.3 | 666.7 ± 61.8 | 4.7 ± 0.5 | 499.3 ± 18.7 | 679.9 ± 63.5 |
| S3ID2 | 6.8 ± 0.7 | 495.2 ± 51.4 | 295.7 ± 12.1 | 4.5 ± 0.8 | 425.0 ± 36.1 | 444.0 ± 19.0 | 5.2 ± 0.6 | 530.7 ± 34.7 | 679.3 ± 48.5 |
| S3ID3 | 9.5 ± 0.9 | 544.2 ± 43.8 | 299.7 ± 30.6 | 6.3 ± 0.8 | 451.1 ± 25.4 | 519.7 ± 24.2 | 4.9 ± 0.7 | 550.0 ± 26.5 | 677.3 ± 27.8 |
| Na_R39 | Cl_R39 | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 55% ETc | 83% Etc | 110% Etc | Mean | 55% Etc | 83% Etc | 110% Etc | Mean | ||
| 1.5 dS m−1 | 2.83 bA | 1.28 cB | 0.80 bB | 1.64 | 1.5 dS m−1 | 18.33 aA | 11.67 bB | 10.67 bB | 13.56 |
| 3.8 dS m−1 | 4.08 aB | 4.81 bAB | 5.88 aA | 4.93 | 3.8 dS m−1 | 18.25 aA | 15.00 abA | 16.50 aA | 16.58 |
| 6.0 dS m−1 | 5.27 aB | 7.16 aA | 5.33 aB | 5.92 | 6.0 dS m−1 | 19.33 aA | 17.33 aA | 17.67 aA | 18.11 |
| Mean | 4.06 | 4.42 | 4.00 | Mean | 18.64 | 14.67 | 14.94 | ||
| No tested equations were significant | Cl_R39 = 42.81 *** − 9.98.10−1 EC – 6.15.10−1 ID *** + 2.77.10−3 ID² ** + 2.43.10−2 ECID ** (R² = 0.93) | ||||||||
| K_R60 | Cl_R60 | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| mg g−1 | mg g−1 | ||||||||
| 55% ETc | 83% ETc | 110% ETc | Mean | 55% ETc | 83% ETc | 110% ETc | Mean | ||
| 1.5 dS m−1 | 9.73 abAB | 11.31 aA | 8.97 abB | 10.00 | 1.5 dS m−1 | 10.50 cA | 9.00 cA | 11.75 cA | 10.42 |
| 3.8 dS m−1 | 8.52 bB | 9.02 bAB | 10.88 aA | 9.48 | 3.8 dS m−1 | 17.75 bB | 13.25 bC | 21.33 aA | 17.44 |
| 6.0 dS m−1 | 11.75 aA | 11.91 aA | 7.05 bB | 10.24 | 6.0 dS m−1 | 22.00 aA | 24.25 aA | 17.00 bB | 21.08 |
| Mean | 10.00 | 10.75 | 8.97 | Mean | 16.75 | 15.50 | 16.69 | ||
| No tested equations were significant | No tested equations were significant | ||||||||
| Na_S60 | K_S60 | ||||||||
| mg g−1 | mg g−1 | ||||||||
| 55% ETc | 83% ETc | 110% ETc | Mean | 55% ETc | 83% ETc | 110% ETc | Mean | ||
| 1.5 dS m−1 | 0.22 bA | 0.27 bA | 0.32 bA | 0.27 | 1.5 dS m−1 | 15.94 aB | 17.50 bB | 22.78 aA | 18.74 |
| 3.8 dS m−1 | 0.72 aAB | 0.59 aB | 0.75 aA | 0.69 | 3.8 dS m−1 | 18.43 aB | 23.90 aA | 20.35 aAB | 20.89 |
| 6.0 dS m−1 | 0.60 aB | 0.45 aB | 0.89 aA | 0.65 | 6.0 dS m−1 | 19.16 aA | 19.38 bA | 20.12 aA | 19.55 |
| Mean | 0.51 | 0.44 | 0.65 | Mean | 17.85 | 20.26 | 21.08 | ||
| No tested equations were significant | No tested equations were significant | ||||||||
| Cl_L60 | |||||||||
| mg g−1 | |||||||||
| 55% ETc | 83% ETc | 110% ETc | Mean | ||||||
| 1.5 dS m−1 | 15.00 abA | 14.75 abA | 14.00 aA | 14.58 | |||||
| 3.8 dS m−1 | 14.50 bA | 16.00 aA | 14.75 aA | 15.08 | |||||
| 6.0 dS m−1 | 16.33 aA | 14.00 bB | 14.50 aB | 14.94 | |||||
| Mean | 15.28 | 14.92 | 14.42 | ||||||
| No tested equations were significant | |||||||||
| Na_R81 | Cl_R81 | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| mg g−1 | mg g−1 | ||||||||
| 55% ETc | 83% ETc | 110% ETc | Mean | 55% ETc | 83% ETc | 110% ETc | Mean | ||
| 1.5 dS m−1 | 2.32 bA | 2.71 cA | 2.63 bA | 2.55 | 1.5 dS m−1 | 14.00 cA | 17.75 bA | 16.00 bA | 15.92 |
| 3.8 dS m−1 | 8.67 aA | 7.04 bB | 7.48 aAB | 7.73 | 3.8 dS m−1 | 24.00 bA | 22.50 abA | 21.00 abA | 22.50 |
| 6.0 dS m−1 | 9.94 aA | 9.15 aA | 7.60 aB | 8.89 | 6.0 dS m−1 | 31.00 aA | 26.75 aAB | 23.00 aB | 26.92 |
| Mean | 6.98 | 6.30 | 5.90 | Mean | 23.00 | 22.23 | 20.00 | ||
| Na_R81 = 4.84 ** + 5.27 EC *** − 3.96.10−1 EC² *** + 2.07.10−2 ID − 1.07.10−2 ECID ** (R² = 0.99) | Cl_R81 = 4.54 + 5.80 EC *** + 9.77.10−2 ID * − 4.0.10−2 ECID ** (R² = 0.97) | ||||||||
| Na_S81 | K_S81 | ||||||||
| mg g−1 | mg g−1 | ||||||||
| 55% ETc | 83% ETc | 110% ETc | Mean | 55% ETc | 83% ETc | 110% ETc | Mean | ||
| 1.5 dS m−1 | 0.14 cA | 0.13 cA | 0.17 bA | 0.15 | 1.5 dS m−1 | 12.21 bB | 14.50 aB | 17.14 aA | 14.61 |
| 3.8 dS m−1 | 0.45 bA | 0.44 bA | 0.36 aA | 0.42 | 3.8 dS m−1 | 10.12 bB | 14.12 aA | 12.55 bAB | 12.26 |
| 6.0 dS m−1 | 0.72 aA | 0.80 aA | 0.30 abB | 0.60 | 6.0 dS m−1 | 15.22 aA | 14.52 aA | 15.87 aA | 15.20 |
| Mean | 0.44 | 0.46 | 0.28 | Mean | 12.51 | 14.38 | 15.19 | ||
| No tested equations were significant | K_C81 = 9.72 *** − 2.36 EC ** + 5.22.10−1 EC² *** + 1.14.10−1 ID *** − 1.73.10−2 ECID ** (R² = 0.84) | ||||||||
| Cl_S81 | Cl_L81 | ||||||||
| mg g−1 | mg g−1 | ||||||||
| 55% ETc | 83% ETc | 110% ETc | Mean | 55% ETc | 83% ETc | 110% ETc | Mean | ||
| 1.5 dS m−1 | 16.00 bB | 19.75 aA | 18.50 aAB | 18.08 | 1.5 dS m−1 | 14.50 aA | 15.50 aA | 16.00 aA | 15.33 |
| 3.8 dS m−1 | 19.00 abA | 17.75 aAB | 15.25 aB | 17.33 | 3.8 dS m−1 | 16.00 aA | 15.25 aAB | 13.00 bB | 14.75 |
| 6.0 dS m−1 | 19.50 aA | 19.00 aA | 17.67 aA | 18.72 | 6.0 dS m−1 | 16.25 aA | 16.25 aA | 15.50 aA | 16.00 |
| Mean | 18.17 | 18.83 | 17.14 | Mean | 15.58 | 15.67 | 14.83 | ||
| Cl_C81 = 5.94 + 1.28.10−2 EC + 2.11.10−1 EC² + 3.07.10−1 ID * − 1.57.10−3 ID²* − 1.76.10−2 ECID * (R² = 0.62) | Cl_F81 = 17.86 ** − 1.21 EC * + 1.81.10−1 EC² − 1.35.10−2 ID (R² = 0.36) | ||||||||
| EL39 | RWC39 | ADM_39 | Na_R39 | K_R39 | Cl_R39 | Na_S39 | K_S39 | Cl_S39 | Na_L39 | K_L39 | Cl_L39 | LA39 | SD39 | SH39 | TH39 | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| EL39 | 1.00 | |||||||||||||||
| RWC39 | −0.07 | 1.00 | ||||||||||||||
| ADM_39 | −0.31 | 0.09 | 1.00 | |||||||||||||
| Na_R39 | 0.07 | 0.14 | −0.30 | 1.00 | ||||||||||||
| K_R39 | 0.07 | 0.18 | −0.12 | −0.21 | 1.00 | |||||||||||
| Cl_R39 | 0.37 | −0.00 | −0.38 | 0.64 | 0.04 | 1.00 | ||||||||||
| Na_S39 | 0.02 | 0.19 | −0.37 | 0.72 | −0.03 | 0.50 | 1.00 | |||||||||
| K_S39 | −0.08 | 0.11 | 0.04 | −0.31 | 0.05 | −0.22 | −0.16 | 1.00 | ||||||||
| Cl_S39 | −0.19 | 0.23 | −0.10 | 0.09 | 0.06 | 0.12 | 0.26 | 0.29 | 1.00 | |||||||
| Na_L39 | 0.37 | −0.04 | −0.16 | 0.01 | 0.15 | 0.32 | 0.33 | 0.03 | 0.14 | 1.00 | ||||||
| K_L39 | 0.09 | −0.00 | 0.24 | −0.22 | 0.21 | −0.09 | −0.18 | 0.28 | 0.18 | 0.08 | 1.00 | |||||
| Cl_L39 | −0.26 | −0.13 | 0.19 | −0.21 | 0.04 | −0.13 | −0.09 | −0.12 | 0.19 | 0.15 | 0.00 | 1.00 | ||||
| LA39 | 0.17 | 0.11 | 0.29 | −0.16 | −0.16 | −0.49 | −0.38 | 0.13 | 0.03 | −0.26 | 0.21 | −0.10 | 1.00 | |||
| SD39 | −0.09 | 0.15 | 0.23 | −0.17 | −0.14 | −0.26 | −0.33 | 0.00 | −0.03 | −0.32 | 0.06 | −0.06 | 0.52 | 1.00 | ||
| SH39 | −0.13 | 0.13 | 0.56 | −0.36 | −0.14 | −0.68 | −0.39 | 0.14 | −0.07 | −0.23 | 0.12 | 0.04 | 0.76 | 0.52 | 1.00 | |
| TH39 | −0.13 | 0.13 | 0.56 | −0.36 | −0.14 | −0.68 | −0.39 | 0.14 | −0.07 | −0.23 | 0.12 | 0.04 | 0.76 | 0.52 | 1.00 | 1.00 |
| EL60 | RWC60 | ADM_60 | Na_R60 | K_R60 | Cl_R60 | Na_S60 | K_S60 | Cl_S60 | Na_L60 | K_L60 | Cl_L60 | LA60 | SD60 | SH60 | TH60 | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| EL60 | 1.00 | |||||||||||||||
| RWC60 | −0.16 | 1.00 | ||||||||||||||
| ADM_60 | 0.03 | −0.09 | 1.00 | |||||||||||||
| Na_R60 | 0.17 | −0.32 | 0.16 | 1.00 | ||||||||||||
| K_R60 | −0.14 | 0.18 | −0.03 | 0.06 | 1.00 | |||||||||||
| Cl_R60 | 0.13 | −0.18 | 0.03 | 0.83 | 0.36 | 1.00 | ||||||||||
| Na_S60 | 0.08 | −0.46 | 0.21 | 0.74 | −0.34 | 0.52 | 1.00 | |||||||||
| K_S60 | 0.12 | −0.12 | 0.18 | 0.13 | −0.18 | 0.05 | 0.26 | 1.00 | ||||||||
| Cl_S60 | −0.09 | 0.05 | −0.18 | 0.08 | −0.24 | −0.10 | 0.17 | 0.63 | 1.00 | |||||||
| Na_L60 | 0.15 | −0.16 | 0.16 | 0.40 | 0.02 | 0.34 | 0.35 | −0.06 | −0.04 | 1.00 | ||||||
| K_L60 | −0.07 | 0.15 | 0.25 | 0.06 | 0.37 | 0.10 | −0.04 | 0.10 | 0.19 | 0.24 | 1.00 | |||||
| Cl_L60 | 0.26 | −0.12 | 0.42 | 0.10 | 0.07 | 0.06 | 0.07 | 0.16 | 0.11 | 0.09 | 0.45 | 1.00 | ||||
| LA60 | 0.27 | −0.15 | 0.07 | 0.42 | 0.11 | 0.42 | 0.29 | −0.06 | −0.18 | 0.24 | 0.25 | 0.15 | 1.00 | |||
| SD60 | 0.05 | −0.17 | −0.05 | 0.01 | −0.04 | 0.03 | −0.07 | 0.06 | 0.08 | −0.01 | 0.29 | 0.05 | 0.35 | 1.00 | ||
| SH60 | −0.18 | −0.00 | −0.02 | −0.29 | −0.26 | −0.33 | −0.03 | 0.43 | 0.40 | −0.34 | −0.38 | −0.18 | −0.38 | 0.09 | 1.00 | |
| TH60 | −0.15 | −0.03 | −0.03 | −0.25 | −0.22 | −0.28 | −0.03 | 0.42 | 0.39 | −0.34 | −0.37 | −0.15 | −0.29 | 0.14 | 0.99 | 1.00 |
| Pro | TAA | TSS | ADM_81 | Na_R81 | K_R81 | Cl_R81 | Na_S81 | K_S81 | Cl_S81 | Na_L81 | K_L81 | Cl_L81 | SD81 | SH81 | TH81 | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Pro | 1.00 | |||||||||||||||
| TAA | 0.25 | 1.00 | ||||||||||||||
| TSS | −0.02 | −0.12 | 1.00 | |||||||||||||
| ADM_81 | −0.13 | 0.41 | −0.14 | 1.00 | ||||||||||||
| Na_R81 | 0.15 | −0.07 | 0.27 | 0.13 | 1.00 | |||||||||||
| K_R81 | 0.14 | 0.23 | −0.11 | −0.19 | −0.48 | 1.00 | ||||||||||
| Cl_R81 | 0.13 | 0.08 | 0.23 | 0.10 | 0.82 | −0.17 | 1.00 | |||||||||
| Na_S81 | 0.06 | −0.01 | 0.16 | 0.31 | 0.83 | −0.25 | 0.74 | 1.00 | ||||||||
| K_S81 | 0.25 | −0.06 | −0.30 | −0.10 | −0.12 | 0.24 | −0.01 | 0.01 | 1.00 | |||||||
| Cl_S81 | −0.00 | −0.35 | 0.16 | −0.17 | 0.12 | 0.22 | 0.22 | 0.20 | 0.33 | 1.00 | ||||||
| Na_L81 | −0.12 | −0.23 | 0.13 | −0.42 | 0.08 | −0.06 | 0.23 | 0.11 | −0.11 | −0.01 | 1.00 | |||||
| K_L81 | 0.10 | −0.22 | 0.26 | −0.04 | 0.10 | −0.07 | 0.01 | 0.21 | 0.40 | 0.38 | 0.06 | 1.00 | ||||
| Cl_L81 | −0.06 | −0.25 | 0.02 | −0.43 | 0.14 | 0.29 | 0.33 | 0.21 | 0.18 | 0.47 | 0.31 | 0.06 | 1.00 | |||
| SD81 | −0.15 | 0.17 | 0.03 | 0.03 | −0.10 | −0.03 | −0.14 | −0.05 | −0.13 | −0.25 | 0.04 | −0.06 | 0.07 | 1.00 | ||
| SH81 | 0.13 | 0.12 | −0.49 | −0.13 | −0.31 | −0.02 | −0.39 | −0.45 | 0.37 | −0.21 | −0.30 | −0.14 | −0.12 | 0.04 | 1.00 | |
| TH81 | −0.04 | 0.05 | −0.55 | −0.15 | −0.32 | 0.05 | −0.32 | −0.40 | 0.47 | −0.07 | −0.14 | −0.12 | −0.04 | 0.18 | 0.84 | 1.00 |
| 0–20 cm | 20–40 cm | ||||
|---|---|---|---|---|---|
| SV | DF | EC | ESP | EC | ESP |
| Block | 3 | 1.75 | 0.22 | 0.52 | 0.57 |
| Salt | 2 | 7.93 ** | 71.26 ** | 20.94 ** | 46.21 ** |
| ID | 2 | 0.90 | 1.63 | 12.57 ** | 0.07 |
| Salt × ID | 4 | 0.52 | 0.39 | 1.81 | 0.40 |
| C. V. (%) | 51.4 | 24.4 | 34.5 | 36.7 | |
| 1.5 dS m−1 | 1.58 | 2.85 | 1.72 | 1.99 | |
| 3.8 dS m−1 | 3.84 | 9.76 | 4.77 | 8.46 | |
| 6.0 dS m−1 | 2.71 | 12.50 | 4.20 | 13.38 | |
| 55% ETc | 2.98 | 9.24 | 4.71 | 7.78 | |
| 83% ETc | 2.87 | 7.93 | 3.76 | 7.85 | |
| 110% ETc | 2.27 | 7.94 | 2.22 | 8.20 | |
| Treatment | Period from Sowing (Days) | Period from Sowing (Days) | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 15–21 | 22–35 | 36–49 | 50–63 | 64–77 | 78–91 | 15–21 | 22–35 | 36–49 | 50–63 | 64–77 | 78–91 | |
| Moisture at 0–30 cm Layer (m³ m−3) | Moisture at 30–60 cm Layer (m³ m−3) | |||||||||||
| S1ID1 | 0.125 | 0.121 | 0.121 | 0.121 | 0.122 | 0.121 | 0.178 | 0.161 | 0.160 | 0.159 | 0.160 | 0.160 |
| S1ID2 | 0.138 | 0.121 | 0.121 | 0.121 | 0.122 | 0.121 | 0.189 | 0.162 | 0.160 | 0.160 | 0.160 | 0.160 |
| S1ID3 | 0.136 | 0.130 | 0.127 | 0.133 | 0.133 | 0.129 | 0.200 | 0.165 | 0.177 | 0.192 | 0.186 | 0.182 |
| S2ID1 | 0.128 | 0.123 | 0.123 | 0.124 | 0.124 | 0.122 | 0.178 | 0.161 | 0.160 | 0.159 | 0.159 | 0.159 |
| S2ID2 | 0.137 | 0.126 | 0.125 | 0.129 | 0.137 | 0.125 | 0.191 | 0.166 | 0.169 | 0.177 | 0.188 | 0.170 |
| S2ID3 | 0.137 | 0.132 | 0.125 | 0.131 | 0.136 | 0.128 | 0.197 | 0.188 | 0.171 | 0.191 | 0.196 | 0.185 |
| S3ID1 | 0.133 | 0.130 | 0.128 | 0.129 | 0.126 | 0.124 | 0.184 | 0.161 | 0.177 | 0.170 | 0.178 | 0.171 |
| S3ID2 | 0.132 | 0.127 | 0.128 | 0.131 | 0.125 | 0.127 | 0.181 | 0.163 | 0.173 | 0.177 | 0.173 | 0.167 |
| S3ID3 | 0.135 | 0.134 | 0.125 | 0.132 | 0.134 | 0.131 | 0.199 | 0.197 | 0.188 | 0.197 | 0.192 | 0.187 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
de Queiroz, G.C.M.; de Medeiros, J.F.; da Silva, R.R.; da Silva Morais, F.M.; de Sousa, L.V.; de Souza, M.V.P.; da Nóbrega Santos, E.; Ferreira, F.N.; da Silva, J.M.C.; Clemente, M.I.B.; et al. Growth, Solute Accumulation, and Ion Distribution in Sweet Sorghum under Salt and Drought Stresses in a Brazilian Potiguar Semiarid Area. Agriculture 2023, 13, 803. https://doi.org/10.3390/agriculture13040803
de Queiroz GCM, de Medeiros JF, da Silva RR, da Silva Morais FM, de Sousa LV, de Souza MVP, da Nóbrega Santos E, Ferreira FN, da Silva JMC, Clemente MIB, et al. Growth, Solute Accumulation, and Ion Distribution in Sweet Sorghum under Salt and Drought Stresses in a Brazilian Potiguar Semiarid Area. Agriculture. 2023; 13(4):803. https://doi.org/10.3390/agriculture13040803
Chicago/Turabian Stylede Queiroz, Gabriela Carvalho Maia, José Francismar de Medeiros, Rodrigo Rafael da Silva, Francimar Maik da Silva Morais, Leonardo Vieira de Sousa, Maria Vanessa Pires de Souza, Elidayane da Nóbrega Santos, Fagner Nogueira Ferreira, Juliana Maria Costa da Silva, Maria Isabela Batista Clemente, and et al. 2023. "Growth, Solute Accumulation, and Ion Distribution in Sweet Sorghum under Salt and Drought Stresses in a Brazilian Potiguar Semiarid Area" Agriculture 13, no. 4: 803. https://doi.org/10.3390/agriculture13040803
APA Stylede Queiroz, G. C. M., de Medeiros, J. F., da Silva, R. R., da Silva Morais, F. M., de Sousa, L. V., de Souza, M. V. P., da Nóbrega Santos, E., Ferreira, F. N., da Silva, J. M. C., Clemente, M. I. B., de Castro Granjeiro, J. C., de Araújo Sales, M. N., Constante, D. C., Nobre, R. G., & da Silva Sá, F. V. (2023). Growth, Solute Accumulation, and Ion Distribution in Sweet Sorghum under Salt and Drought Stresses in a Brazilian Potiguar Semiarid Area. Agriculture, 13(4), 803. https://doi.org/10.3390/agriculture13040803











