Alteration of Carbohydrate Metabolism in Fusarium Infected Wheat Kernels Treated with Fungicides and Its Relation to Baking Technological Parameters and Deoxynivalenol Contamination
Abstract
:1. Introduction
2. Materials and Methods
2.1. Field Experiments
2.2. Determination of Simple Carbohydrates
2.3. Determination of Fructan Content
2.4. Wheat Quality Analysis Methods
2.5. DON Analyses
2.6. Statistical Analysis
3. Results
3.1. Concentrations of Water-Soluble Carbohydrate Components
3.2. Results of Correlation Analyses between Carbohydrate Content, Technological Properties and DON Contamination
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
Appendix A
| WG | FS | HFN | HI | TKW | PRO | ZI | E135 | EXT | MBU | DON | FRU | GLU | SUC | MAL | RAF | NYS | STA | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| FS | 0.010 | |||||||||||||||||
| HFN | −0.296 | −0.071 | ||||||||||||||||
| HI | 0.195 | −0.123 | −0.822 *** | |||||||||||||||
| TKW | 0.214 | −0.410 | 0.628 * | −0.575 | ||||||||||||||
| PRO | 0.869 *** | −0.235 | −0.285 | 0.260 | 0.383 | |||||||||||||
| ZI | 0.582 | −0.277 | −0.006 | 0.168 | 0.448 | 0.826 *** | ||||||||||||
| E135 | 0.181 | −0.002 | 0.179 | −0.206 | 0.634 * | 0.469 | 0.628 * | |||||||||||
| EXT | 0.630 * | 0.103 | −0.676 ** | 0.402 | −0.212 | 0.576 | 0.289 | 0.218 | ||||||||||
| MBU | −0.315 | 0.074 | 0.668 ** | −0.529 | 0.666 * | −0.169 | 0.100 | 0.599 * | −0.576 | |||||||||
| DON | −0.328 | −0.085 | 0.329 | −0.333 | 0.218 | −0.523 | −0.654 * | −0.173 | −0.286 | 0.338 | ||||||||
| FRU | −0.244 | 0.709 ** | −0.106 | −0.193 | −0.298 | −0.460 | −0.495 | −0.088 | −0.160 | 0.247 | 0.190 | |||||||
| GLU | 0.523 | 0.321 | 0.192 | −0.028 | −0.033 | 0.198 | 0.086 | −0.359 | −0.097 | −0.124 | −0.050 | −0.026 | ||||||
| SUC | −0.342 | 0.435 | 0.182 | −0.602 * | 0.071 | −0.287 | −0.226 | 0.339 | −0.014 | 0.296 | −0.033 | 0.568 | −0.480 | |||||
| MAL | 0.443 | 0.388 | 0.159 | −0.113 | −0.214 | 0.055 | 0.052 | −0.384 | 0.106 | −0.317 | −0.123 | 0.023 | 0.830 *** | −0.256 | ||||
| RAF | −0.311 | 0.244 | 0.347 | −0.563 | 0.169 | −0.377 | −0.481 | 0.188 | 0.086 | 0.228 | 0.580 | 0.103 | −0.272 | 0.512 | −0.133 | |||
| NYS | −0.433 | 0.454 | −0.029 | −0.361 | −0.317 | −0.413 | −0.323 | 0.008 | −0.010 | −0.033 | −0.229 | 0.570 | −0.444 | 0.895 *** | −0.137 | 0.294 | 1.000 | |
| STA | −0.381 | 0.606 * | 0.018 | −0.383 | −0.087 | −0.383 | −0.329 | 0.271 | −0.106 | 0.392 | 0.045 | 0.846 *** | −0.397 | 0.889 *** | −0.284 | 0.330 | 0.807 *** | |
| FRN | −0.843 *** | 0.295 | 0.304 | −0.327 | −0.362 | −0.832 *** | −0.587 * | −0.359 | −0.650 * | 0.169 | 0.058 | 0.460 | −0.224 | 0.501 | −0.115 | 0.169 | 0.680 ** | 0.529 |
| WG | FS | HFN | HI | TKW | PRO | ZI | E135 | EXT | MBU | DON | FRU | GLU | SUC | MAL | RAF | NYS | STA | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| FS | 0.650 * | |||||||||||||||||
| HFN | 0.638 * | 0.814 *** | ||||||||||||||||
| HI | 0.796 ** | 0.862 *** | 0.899 *** | |||||||||||||||
| TKW | 0.229 | 0.604 * | 0.652 * | 0.450 | ||||||||||||||
| PRO | 0.599 * | 0.119 | −0.027 | 0.320 | −0.588 * | |||||||||||||
| ZI | 0.962 *** | 0.746 ** | 0.640 * | 0.811 *** | 0.333 | 0.536 | ||||||||||||
| E135 | 0.684 ** | 0.796 ** | 0.838 *** | 0.752 ** | 0.826 *** | −0.137 | 0.755 ** | |||||||||||
| EXT | 0.416 | 0.705 ** | 0.630 * | 0.609 * | 0.723 ** | −0.266 | 0.536 | 0.841 *** | ||||||||||
| MBU | 0.639 * | 0.781 ** | 0.850 *** | 0.734 ** | 0.858 *** | −0.200 | 0.709 ** | 0.996 *** | 0.825 *** | |||||||||
| DON | −0.406 | −0.699 ** | −0.696 ** | −0.711 ** | −0.687 ** | 0.203 | −0.526 | −0.792 ** | −0.949 *** | −0.781 ** | ||||||||
| FRU | 0.366 | 0.814 *** | 0.804 *** | 0.655 * | 0.841 *** | −0.340 | 0.502 | 0.867 *** | 0.829 *** | 0.881 *** | −0.821 *** | |||||||
| GLU | −0.820 *** | −0.380 | −0.623 * | −0.640 * | −0.079 | −0.528 | −0.676 ** | −0.444 | −0.006 | −0.433 | 0.048 | −0.153 | ||||||
| SUC | 0.589 * | 0.162 | 0.349 | 0.263 | 0.398 | 0.007 | 0.578 | 0.664 * | 0.447 | 0.649 * | −0.332 | 0.310 | −0.460 | |||||
| MAL | −0.391 | −0.747 ** | −0.784 ** | −0.672 ** | −0.787 ** | 0.300 | −0.535 | −0.876 *** | −0.890 *** | −0.884 *** | 0.905 *** | −0.957 *** | 0.130 | −0.428 | ||||
| RAF | 0.736 ** | 0.819 *** | 0.916 *** | 0.815 *** | 0.680 ** | 0.048 | 0.771 ** | 0.900 *** | 0.611 * | 0.904 *** | −0.613 * | 0.836 *** | −0.661 | 0.513 | −0.783 ** | |||
| NYS | 0.549 | 0.795 ** | 0.859 *** | 0.659 * | 0.742 ** | −0.187 | 0.579 | 0.855 *** | 0.574 | 0.875 *** | −0.498 | 0.821 *** | −0.524 | 0.441 | −0.726 ** | 0.913 *** | ||
| STA | 0.693 ** | 0.057 | −0.024 | 0.255 | −0.345 | 0.829 *** | 0.671 ** | 0.119 | −0.069 | 0.058 | 0.075 | −0.231 | −0.527 | 0.502 | 0.110 | 0.155 | −0.085 | |
| FRN | 0.489 | 0.747 ** | 0.903 *** | 0.714 ** | 0.785 ** | −0.225 | 0.569 | 0.873 *** | 0.643 * | 0.898 *** | −0.672 ** | 0.914 *** | −0.451 | 0.447 | −0.886 *** | 0.935 *** | 0.898 *** | −0.079 |
References
- D’Apollonia, B.L.; Rayas-Duarte, P. Wheat carbohydrates: Structure and functionality. In Wheat Production, Properties and Quality, 1st ed.; Bushuk, W., Rasper, V.F., Eds.; Springer: Boston, MA, USA, 1994; pp. 109–130. [Google Scholar] [CrossRef]
- Shelton, D.R.; Lee, W.J. Cereal Carbohydrates. In Handbook of Cereal Science and Technology, 2nd ed.; Kulp, K., Ponte, J.G., Eds.; CRC Press: Boca Raton, FL, USA, 2000; pp. 385–416. [Google Scholar] [CrossRef]
- Ruuska, S.A.; Rebetzke, G.J.; Herwaarden, A.F.; Richards, R.A.; Fettel, N.A.; Tabe, L.; Jenkins, C.L.D. Genotypic variation in water-soluble carbohydrate accumulation in wheat. Funct. Plant Biol. 2006, 33, 799–809. [Google Scholar] [CrossRef]
- Macarthur, L.A.; Dappolonia, B.L. Comparison of oat and wheat carbohydrates I. Sugars. Cereal Chem. 1979, 56, 455–457. [Google Scholar]
- Fretzdorff, B.; Welge, N. Fructan and the raffinose contents in cereals and pseudocereal grains. Getreide Mehl. Brot. 2003, 57, 3–8. (In German) [Google Scholar]
- Sami, F.; Yusuf, M.; Faizan, M.; Faraz, A.; Hayat, S. Role of sugars under abiotic stress. Plant Physiol. Biochem. 2016, 109, 54–61. [Google Scholar] [CrossRef] [PubMed]
- Verspreet, J.; Hemdane, S.; Dornez, E.; Cuyvers, S.; Pollet, A.; Delcour, J.A. Analysis of storage and structural carbohydrates in developing wheat (Triticum aestivum L.) grains using quantitative analysis and microscopy. J. Agric. Food Chem. 2013, 61, 9251–9259. [Google Scholar] [CrossRef]
- Call, L.; Haider, E.; D’Amico, S.; Reiter, E.; Grausguber, H. Synthesis and accumulation of amylase-trypsin inhibitors and changes in carbohydrate profile during grain development of bread wheat (Triticum aestivum L.). BMC Plant Biol. 2021, 21, 113. [Google Scholar] [CrossRef] [PubMed]
- Halford, N.G.; Curtis, T.Y.; Chen, Z.; Huang, J. Effects of abiotic stress and crop management on cereal grain composition: Implication for food quality and safety. J. Exp. Bot. 2014, 66, 1145–1156. [Google Scholar] [CrossRef] [Green Version]
- Halford, N.G.; Curtis, T.Y.; Muttucumaru, N.; Postles, J.; Mottram, D.S. Sugars in crop plants. Ann. Appl. Biol. 2011, 158, 1–25. [Google Scholar] [CrossRef]
- Jenner, C.F. Effects of exposure of wheat ears to high-temperature on dry matter accumulation and carbohydrate-metabolism in the grain of 2 cultivars. II. Carry-over effects. Aust. J. Plant Physiol. 1991, 18, 179–190. [Google Scholar] [CrossRef]
- Shah, N.H.; Paulsen, G.M. Interaction of drought and high temperature on photosynthesis and grain-filling of wheat. Plant Soil 2003, 257, 219–226. [Google Scholar] [CrossRef]
- Ács, K.; Lehoczki-Krsjak, S.; Varga, M.; Kótai, C.; Ács, E.; Salgó, A.; Mesterházy, Á. Reduction of deoxynivalenol (DON) contamination by improved fungicide use in wheat. Part 3. Reduction of Fusarium head blight and influence on quality traits in cultivars with different resistance levels. Eur. J. Plant Pathol. 2018, 151, 21–38. [Google Scholar] [CrossRef]
- Bishop, D.L.; Chatterton, N.J.; Harrison, P.A.; Hatfield, R.D. Changes in carbohydrate coordinated partitioning and cell wall remodeling with stress-induced pathogenesis in wheat sheaths. Physiol. Mol. Plant P. 2002, 61, 53–63. [Google Scholar] [CrossRef]
- Boyacioglu, D.; Hettiarachchy, N.S. Changes in some biochemical components of wheat grain that was infected with Fusarium graminearum. J. Cereal Sci. 1995, 21, 57–62. [Google Scholar] [CrossRef]
- Gherbawy, Y.A.; El-tayeb, M.A.; Maghraby, T.A.; Shebany, Y.M.; El-deeb, B.A. Response of antioxidant enzymes and some metabolic activities in wheat to Fusarium spp. infections. Acta Agron. Hung. 2012, 60, 319–333. [Google Scholar] [CrossRef]
- Bewley, J.D.; Bradford, K.; Hilhorst, H.; Nonogaki, H. Seeds: Physiology of Development, Germination and Dormancy, 3rd ed.; Springer: New York, NY, USA, 2013; pp. 183–246. [Google Scholar]
- Perten, H. Application of the falling number method for evaluating alpha-amylase activity. Cereal Chem. 1964, 41, 127–140. [Google Scholar]
- Nursten, H.E. The Maillard Reaction: Chemistry, Biochemistry and Implications, 1st ed.; Royal Society of Chemistry: London, UK, 2005. [Google Scholar]
- Mesterházy, Á.; Varga, M.; Tóth, B.; Kótai, C.; Bartók, T.; Véha, A.; Ács, K.; Vágvölgyi, C.; Lehoczki-Krsjak, S. Reduction of deoxynivalenol (DON) contamination by improved fungicide use in wheat. Part 2. Farm scale tests with different nozzle types and updating the integrated approach. Eur. J. Plant Pathol. 2018, 151, 1–20. [Google Scholar] [CrossRef]
- Li, B.W.; Schuhmann, P.J. Gas–liquid chromatographic analysis of sugars in ready-to-eat breakfast cereals. J. Food Sci. 1980, 45, 138–141. [Google Scholar] [CrossRef]
- Antonio, C.; Larson, T.; Gilday, A.; Graham, I.; Bergström, E.; Thomas-Oates, J. Hydrophilic interaction chromatography/electrospray mass spectrometry analysis of carbohydrate-related metabolites from Arabidopsis thaliana leaf tissue. Rapid Commun. Mass Spectrom. 2008, 22, 1399–1407. [Google Scholar] [CrossRef]
- McCleary, B.V.; Murphy, A.; Mugford, D.C. Determination of oligofructans and fructan polysaccharides in foodstuffs by an enzymatic/spectrophotometric method: Collaborative study. J. AOAC Int. 2000, 83, 356–364. [Google Scholar] [CrossRef] [Green Version]
- Mesterházy, Á.; Bartók, T.; Lamper, C. Influence of wheat cultivar, species of Fusarium, and isolate aggressiveness on the efficacy of fungicides for control of Fusarium Head Blight. Plant Dis. 2003, 87, 1107–1115. [Google Scholar] [CrossRef] [Green Version]
- Wang, J.; Pawelzi, E.; Weinert, J.; Wolf, G. Impact of Fusarium culmorum on the polysaccharides of wheat flour. J. Agric. Food Chem. 2005, 53, 5818–5823. [Google Scholar] [CrossRef]
- Kreuzberger, M.; Limsuwan, S.; Eggert, K.; Karlovsky, P.; Pawelzik, E. Impact of Fusarium spp. infection of bread wheat (Triticum aestivum L.). on composition and quality of flour in association with EU maximum level for deoxynivalenol. J. Appl. Bot. Food Qual. 2015, 88, 177–185. [Google Scholar] [CrossRef]
- Goswami, R.; Kistler, H. Heading for disaster: Fusarium graminearum on cereal crops. Mol. Plant Pathol. 2004, 5, 515–525. [Google Scholar] [CrossRef] [PubMed]
- Ribichich, K.F.; Lopez, S.E.; Vegetti, A.C. Histopathological spikelet changes produced by Fusarium graminearum in susceptible and resistant wheat cultivars. Plant Dis. 2000, 84, 794–802. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Casale, W.L.; Hart, L.P. Inhibition of 3H-leucine incorporation by trichothecene mycotoxins in maize and wheat tissue. Phytopathology 1988, 78, 1673–1677. [Google Scholar] [CrossRef] [Green Version]
- Bechtel, D.B.; Kaleikai, L.A.; Gaines, R.L.; Seitz, L.M. The effect of Fusarium graminearum infection on wheat kernels. Cereal Chem. 1985, 62, 191–197. [Google Scholar]
- Eggert, K.; Rawel, H.M.; Pawelzik, E. In vitro degradation of wheat gluten fractions by Fusarium graminearum proteases. Eur. Food Res. Technol. 2011, 233, 697–705. [Google Scholar] [CrossRef] [Green Version]
- Hongyun, G.; Jishan, N.; Xiwen, Y.; Dexian, H.; Wang, C. Impacts of powdery mildew on wheat grain sugar metabolism and starch accumulation in developing grains. Starch 2014, 66, 947–958. [Google Scholar] [CrossRef]
- Khatun, S.; Ahmed, J.U. Response of elevated temperature on carbohydrate accumulation and grain yield in different wheat cultivars. Bangladesh J. Agric. Res. 2015, 40, 205–215. [Google Scholar] [CrossRef] [Green Version]
- Singletary, G.W. Starch synthesis and grain filling in wheat. In Carbohydrate Reserves in Plants–Synthesis and Regulation, 1st ed.; Gupta, A.K., Kaur, N., Eds.; Elsevier Science: Amsterdam, The Netherlands, 2000; pp. 79–105. [Google Scholar] [CrossRef]

| Commercial Name and Application Rate (L/ha) | Active Ingredient (g/L) | Abbreviation of Active Ingredient |
|---|---|---|
| Alert S 1.0 | fluzilazole 125 + carbendazim 250 | FC |
| Cherokee 2.0 | ciproconazole 50 + propioconazole 62.5 + chloronitrile 375 | CPC |
| Prosaro 1.0 | prothioconazole 125, tebuconazole 125 | PT |
| Caramba 1.2 | metconazole 90 | M90 |
| Eminent 1.0 | tetraconazole 125 | TET |
| Falcon 0.8 | tebuconazole 167 + spiroxamine 250 + triadimenol 43 | TST |
| Folicur Solo 1.0 | tebuconazole 250 | T250 |
| Juwel TT 1.0 | epoxyconazole 83 + kresoxym-methyl 83 + fenpropimorf 317 | EKF |
| Fructose | Glucose | Sucrose | Maltose | Nystose | Raffinose | Stachyose | Fructan | Total | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Fungicide | (mg/kg) | (mg/kg) | (mg/kg) | (mg/kg) | (mg/kg) | (mg/kg) | (mg/kg) | (mg/kg) | (mg/kg) | |||||||||
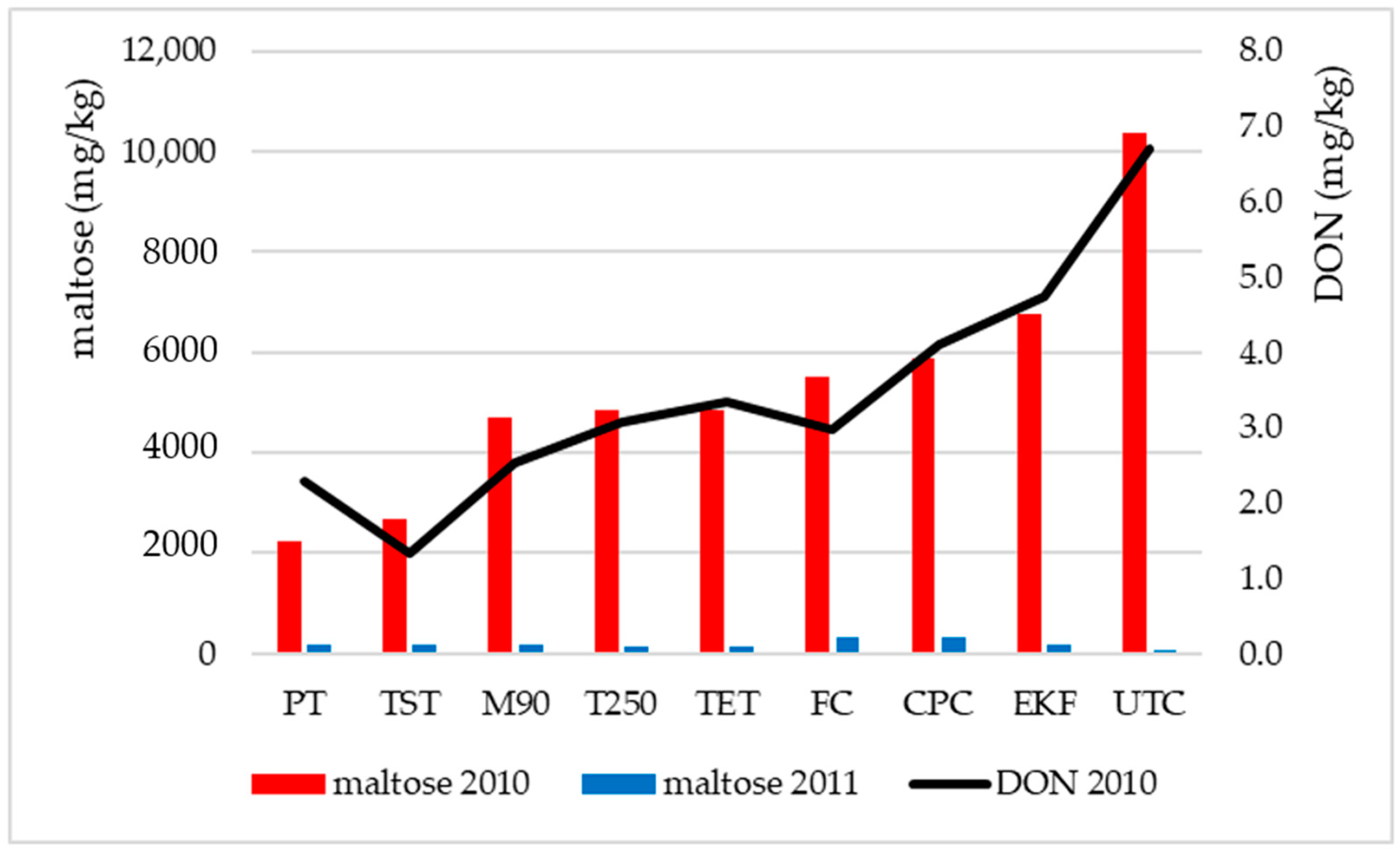
| TST * | 143 | d | 262 | a | 2101 | a,b | 2670 | a | 42 | d | 577 | d | 4 | a | 8933 | f | 14,732 | a |
| PT | 152 | e | 271 | a,b | 2340 | b | 2235 | a | 50 | e | 655 | d | 4 | a | 9433 | g | 15,139 | a |
| M90 | 125 | b,c | 362 | b,c | 2225 | a,b | 4718 | b | 35 | a–c | 348 | b,c | 5 | a | 7817 | c | 15,634 | a,b |
| TET | 123 | b,c | 384 | c | 2007 | a,b | 4858 | b,c | 33 | a,b | 281 | a–c | 3 | a | 8200 | e | 15,888 | a,b |
| T250 | 131 | c,d | 368 | b,c | 2242 | a,b | 4837 | b,c | 37 | a–d | 366 | c | 3 | a | 8167 | e | 16,152 | a,b |
| CPC | 128 | b,c | 396 | c | 1708 | a | 5887 | b,c | 39 | c,d | 297 | a–c | 3 | a | 7967 | d | 16,425 | a,b |
| FC | 117 | b | 332 | a–c | 2275 | a,b | 5497 | b,c | 38 | b–d | 305 | a–c | 4 | a | 7933 | d | 16,500 | a,b |
| EKF | 118 | b,c | 362 | b,c | 1830 | a,b | 6746 | c | 33 | a,b | 270 | a,b | 3 | a | 7583 | b | 16,944 | a,b |
| UTC | 92 | a | 279 | a,b | 2039 | a,b | 10,377 | d | 32 | a | 257 | a | 4 | a | 7283 | a | 20,363 | b |
| Mean | 125 | 335 | 2085 | 5314 | 37 | 373 | 4 | 8146 | 16,420 | |||||||||
| LSD 5% ** | 13 | 98 | 577 | 1996 | 5 | 88 | 2 | 102 | 4784 | |||||||||
| Fructose | Glucose | Sucrose | Maltose | Nystose | Raffinose | Stachyose | Fructan | Total | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Fungicide | (mg/kg) | (mg/kg) | (mg/kg) | (mg/kg) | (mg/kg) | (mg/kg) | (mg/kg) | (mg/kg) | (mg/kg) | |||||||||
| TST * | 101 | a | 280 | a | 681 | a | 135 | a | 74 | a | 37 | a | 1 | a | 5217 | b | 6526 | a |
| PT | 108 | a,b | 291 | a | 1035 | a–c | 168 | a,b | 76 | a | 65 | c | 2 | a | 5233 | b | 6979 | a |
| M90 | 117 | a–c | 274 | a | 1304 | c | 153 | a | 110 | a | 59 | b,c | 3 | a | 5033 | a | 7053 | a |
| TET | 110 | a,b | 373 | b | 760 | a,b | 326 | c | 72 | a | 41 | a,b | 1 | a | 5483 | d | 7166 | a |
| T250 | 128 | c,d | 319 | a | 955 | a–c | 315 | b,c | 90 | a | 47 | a–c | 2 | a | 5367 | c,d | 7223 | a |
| CPC | 142 | d,e | 296 | a | 1224 | c | 166 | a,b | 97 | a | 41 | a,b | 4 | a | 5383 | c,d | 7352 | a |
| FC | 150 | e | 300 | a | 1202 | b,c | 178 | a–c | 108 | a | 57 | b,c | 4 | a | 5717 | e | 7715 | a |
| EKF | 119 | b,c | 275 | a | 1281 | c | 165 | a,b | 115 | a | 46 | a,b | 3 | a | 5733 | e | 7737 | a |
| UTC | 119 | b,c | 289 | a | 913 | a–c | 114 | a | 76 | a | 45 | a,b | 2 | a | 5317 | b,c | 6875 | a |
| Mean | 122 | 300 | 1039 | 191 | 91 | 49 | 3 | 5387 | 7177 | |||||||||
| LSD 5% ** | 17 | 48 | 456 | 151 | 45 | 18 | 3 | 130 | 1413 | |||||||||
| Fungicide | Maltose Content (mg/kg) 2010 | Maltose Content (mg/kg) 2011 | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| S | MS | MR | Mean | S | MS | MR | Mean | |||||||
| TST * | 4538 | a | 2457 | a | 1016 | a | 2670 | 184 | a | 172 | a,b | 178 | a,b | 178 |
| M90 | 9230 | b,c | 2436 | a | 2489 | a | 4718 | 177 | a | 166 | a,b | 156 | a,b | 166 |
| TET | 7107 | a,b | 3316 | a | 4153 | a,b | 4858 | 158 | a | 142 | a | 158 | a,b | 153 |
| EKF | 13,702 | d,e | 3398 | a | 3139 | a,b | 6746 | 125 | a | 211 | a,b | 168 | a,b | 168 |
| PT | 3893 | a | 1953 | a | 860 | a | 2235 | 156 | a | 172 | a,b | 168 | a,b | 165 |
| T250 | 9967 | b,c | 2893 | a | 1653 | a | 4837 | 139 | a | 131 | a | 136 | a,b | 135 |
| CPC | 10,817 | c,d | 4188 | a | 2657 | a | 5887 | 365 | b | 302 | b | 278 | a,b | 315 |
| FC | 9465 | b,c | 4695 | a | 2332 | a | 5497 | 237 | a | 163 | a,b | 578 | c | 326 |
| UTC | 14,898 | e | 9822 | b | 6411 | b | 10,377 | 113 | a | 129 | a | 101 | a | 114 |
| Mean | 9290 | 3906 | 2745 | 5314 | 181 | 176 | 213 | 191 | ||||||
| LSD 5% variety | 1153 | 50 | ||||||||||||
| LSD 5% fungicide | 1996 | 87 | ||||||||||||
| Traits | PC1 | PC2 | PC3 | PC4 |
|---|---|---|---|---|
| WG | 0.7147 | 0.6805 | 0.0454 | −0.0329 |
| FS | 0.8694 | 0.0325 | −0.3228 | −0.2420 |
| HFN | 0.9173 | 0.0074 | −0.2980 | 0.1058 |
| HI | 0.8521 | 0.2814 | −0.3088 | −0.2196 |
| TKW | 0.7709 | −0.5076 | 0.1219 | 0.1631 |
| PRO | −0.0448 | 0.9386 | −0.1724 | −0.2755 |
| ZI | 0.7829 | 0.5708 | 0.0924 | −0.1615 |
| E135 | 0.9770 | −0.0478 | 0.1815 | 0.0453 |
| EXT | 0.8082 | −0.3008 | 0.2666 | −0.3866 |
| RMAX | 0.9738 | −0.1027 | 0.1586 | 0.0979 |
| DON | −0.8021 | 0.2760 | −0.1298 | 0.4605 |
| FRU | 0.8950 | −0.3778 | −0.0746 | −0.0697 |
| GLU | −0.5093 | −0.6887 | 0.2037 | −0.4174 |
| SUC | 0.5498 | 0.2623 | 0.7179 | 0.3147 |
| MAL | −0.8929 | 0.3271 | −0.0931 | 0.1715 |
| RAF | 0.9502 | 0.1143 | −0.1333 | 0.1996 |
| NYS | 0.8775 | −0.0944 | −0.1756 | 0.3503 |
| STA | 0.1153 | 0.9026 | 0.3473 | −0.1227 |
| FRN | 0.9180 | −0.1720 | −0.1169 | 0.2197 |
| Eigenvalue | 11.9316 | 3.8463 | 1.2436 | 1.1543 |
| % of total variance | 62.80 | 20.24 | 6.55 | 6.08 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Acs, K.; Varga, M.; Szekeres, A.; Salgo, A.; Lantos, C.; Bekes, F.; Pauk, J.; Mesterhazy, A. Alteration of Carbohydrate Metabolism in Fusarium Infected Wheat Kernels Treated with Fungicides and Its Relation to Baking Technological Parameters and Deoxynivalenol Contamination. Agriculture 2023, 13, 868. https://doi.org/10.3390/agriculture13040868
Acs K, Varga M, Szekeres A, Salgo A, Lantos C, Bekes F, Pauk J, Mesterhazy A. Alteration of Carbohydrate Metabolism in Fusarium Infected Wheat Kernels Treated with Fungicides and Its Relation to Baking Technological Parameters and Deoxynivalenol Contamination. Agriculture. 2023; 13(4):868. https://doi.org/10.3390/agriculture13040868
Chicago/Turabian StyleAcs, Katalin, Monika Varga, Andras Szekeres, Andras Salgo, Csaba Lantos, Ferenc Bekes, Janos Pauk, and Akos Mesterhazy. 2023. "Alteration of Carbohydrate Metabolism in Fusarium Infected Wheat Kernels Treated with Fungicides and Its Relation to Baking Technological Parameters and Deoxynivalenol Contamination" Agriculture 13, no. 4: 868. https://doi.org/10.3390/agriculture13040868









