Abstract
The aim of this study was to evaluate the applicability of three forms of titanium dioxide (TiO2) and their effect on the yield, chemical and microbiological quality of perennial ryegrass herbage and silage. Two types of titanium dioxide nanoparticles (TiO2NPs) and a commercial product, labeled here as TiO2Com, were selected for the studies. The yield and chemical parameters of herbage did not improve significantly in response to the TiO2 treatment, in comparison to the control group (CONT). The crude protein content of silage was significantly lower in the TiO2Com-treated group than in the TiO2NPs2 group (117 vs. 129 g kg−1 dry matter (DM)). The use of water-soluble carbohydrates during fermentation was limited in the TiO2NPs2 and TiO2Com groups. The fermentation pattern was similar for each investigated group, and a significant difference in pH values was noted between the TiO2NPs2 (pH = 4.49) and CONT (pH = 4.68) group. Lactic acid concentration was the highest in the TiO2NPs1 group (94.8 g kg−1 DM), and the difference relative to the CONT group (83.2 g kg−1 DM) was statistically significant. It was concluded that TiO2 and its nanoparticles have the potential to improve the physicochemical and microbiological quality of herbage and silage.
1. Introduction
Intensive crop production is accompanied by the widespread use of agrochemicals, which maximizes crop yield and quality while exerting negative environmental impacts. Major advances have been made in the cultivation, fertilization and protection technologies of food crops, but not of fodder crops. The production methods and fertilization strategies of fodder crops, including forage grasses, are still insufficiently developed. Moreover, conventional disease, pest and weed control is nearly impossible. Therefore, alternative management options are being sought to optimize fodder crop yields and improve crop quality.
On the global scale, grasslands providing the feed base for livestock (pasture, silage, hay) account for approximately 40% of terrestrial land [1]. In Europe, grasslands cover 35% of the agricultural area [2]. In temperate climates, milk and meat production is largely based on dominant forage grasses, ryegrass (Lolium spp.) and fescue (Festuca spp.). An important role is played by perennial ryegrass (Lolium perenne L.), which is characterized by high yields, a long growing season, adaptation for grazing, high palatability and digestibility [3]. The chemical composition and yields of forage grasses are determined by various factors such as species, variety, growth stage, fertilization and disease susceptibility [4].
Nanotechnology could support forage grass production. Novel nanomaterials applied as biostimulants contribute to reducing the use of fertilizers and crop protection products without compromising their efficiency [5]. The popularity of titanium dioxide nanoparticles (TiO2NPs) has increased recently [6,7]. TiO2NPs have numerous applications in medicine [8], pharmacy, environmental protection [9], electronics industry, food processing industry and agriculture [10].
Nanomaterials used in agricultural crops can affect the microflora of plants. The composition of the microbiota affects the health, yield and quality of herbage, which is then fermented [11,12]. However, the composition of the microbiota may also be unfavorable, including phytopathogens that reduce the quality and yield, i.e., mycotoxin-producing fungi, and bacteria that directly threaten the fermentation process. It is expected that the nanoparticles are tested mainly on the non-beneficial microbiota and the effect is satisfactory, but the impact on the beneficial microbiota is not fully known. However, a large number of studies show that they have a positive or no effect on the beneficial microbiota inhabiting the agrobiocenosis [13].
In the 1990s, Dumon and Ernst [14] found no evidence for the positive or negative (toxic) effects of titanium on the growth of higher plants. However, a beneficial influence of titanium on tobacco plants was observed more than 10 years ago [15]. The neutral attitude to TiO2NPs has changed over the years, and at present they are believed to positively affect plants as biostimulants [10].
Diverse types of nanoparticles enter into interactions with edible plants through accumulation, physiological and biochemical effects [6]. Numerous researchers have investigated the effect of nanoparticles, including TiO2NPs, on the productivity of edible plants [16,17,18]. TiO2NPs have been found to stimulate seed germination and seedling growth, increase root length (vegetables, cereals, soybeans, rapeseed, red clover), increase tolerance to abiotic and biotic stresses (vegetables, wheat, soybean, flax), and increase chlorophyl concentration (rapeseed, tomatoes). The application of TiO2NPs improved the yields of vegetables and other crop plants (barley, wheat, maize, alfalfa), and the noted increase was proportional to the concentration of nanoparticles [19].
However, the extensive use of TiO2NPs has increased their quantities in the environment, which may pose a risk to human and animal health [8]. According to Larue et al. [20], TiO2NPs can be toxic to plants since they induce lipid peroxidation. Foltête et al. [21] demonstrated that TiO2NPs attached to the roots of Vicia faba and suppressed further plant growth. The toxicity of TiO2NPs may vary depending on plant species, time of exposure and nanoparticle properties, e.g., smaller nanoparticles easily penetrate plant tissues and may exert greater effects [8]. The impact of the duration of soil aging on TiO2NPs toxicity is also an important consideration. Wang et al. [18] reported that the aging process alleviated the phytotoxicity of unweathered nanoparticles.
TiO2NPs may exert both positive and negative effects, depending on their physiochemical properties. Their potential toxicity is affected by their morphology, size, crystal form, surface coating and surface charge [22]. Research has shown that TiO2NPs exert multi-level effects by generating oxidative stress, causing cytotoxicity and genotoxicity, affecting seed germination and root elongation. TiO2NPs enter into harmful interactions with DNA, cause damage to meristem cells (onions, tobacco plants), lead to mitotic abnormalities (maize, vetch), and inhibit seed germination and root growth (vegetables, maize, barley) [23,24].
The aim of this study was to evaluate the effect of three forms of TiO2 on the yield, chemical and microbiological quality of perennial ryegrass herbage and silage. The practical applications of TiO2 were also analyzed.
2. Materials and Methods
2.1. Materials
Titanium (IV) oxide anatase (TiO2NPs1) and titanium (IV) oxide nanopowder (TiO2NPs2) were purchased from Sigma Aldrich (Saint Louis, MI, USA). These compounds were used without further purification. Aqueous suspensions of TiO2NPs1 and TiO2NPs2 were prepared by dispersing a proper amount of powders in ultrapure water. The concentration of both prepared TiO2NPs suspensions was equal to 100 mg L−1. A commercially available TiO2 product, hereafter labeled as TiO2Com, was diluted in ultrapure water (1:10 (v/v)) and this suspension was used in the further experiments.
2.2. Physicochemical Characteristics of TiO2NPs
The morphology, average size and size distribution of TiO2NPs were evaluated using micrographs recorded on a JEOL JSM-7500F (JEOL Ltd., Peabody, MA, USA) scanning electron microscope (SEM) equipped with a transmission electron detector (TED). The micrographs were analyzed using a MultiScan software 45. Histograms were generated of the surface area and diameter of 1000 PtNPs.
The stability of TiO2NPs dispersed in the aqueous solutions was evaluated measuring their diffusion coefficient (D) and electrophoretic mobility (μe). The TiO2NPs hydrodynamic diameters were calculated from the Stokes–Einstein relationship, based on the diffusion coefficient (D) measurements carried out using a Zetasizer Nano ZS (Malvern Panalytical, Malvern, UK). The zeta potentials (ζ), were determined based on electrophoretic light scattering (ELS) technique and using a Zetasizer Nano ZS (Malvern Panalytical, Malvern, UK). The values of TiO2NPs zeta potential were calculated using Henry’s model.
The detailed characteristics of the TiO2NPs was presented in the previous work [25]. For the sake of convenience, the crucial parameters characterizing the TiO2NPs and TiO2Com are collected in Table 1.

Table 1.
Selected physicochemical properties of used titanium forms.
2.3. Experimental Design
Perennial ryegrass was cultivated in experimental plots (2 m × 5 m) at the Agricultural Experiment Station in Tomaszkowo (53°43′04″ N 20°24′32″ E). The distance among the experimental plots was 1 m. The seeding rate was 35 kg ha−1. Standard fertilizers were applied: N/P/K at 60/30/60 kg ha−1. The experiment was carried out using a randomized complete block design with three biological replications.
The experiment was established in 2019 and lasted for two years. In the first year, the grass cover was stabilized and weed growth was controlled. In the second year, the actual experiment was conducted. The experiment was established on soil characterized by low nitrogen abundance and the following chemical parameters: total nitrogen 0.09%; P2O5, 21.1 mg 100 g−1 of soil; K2O, 19.5 mg 100 g−1 of soil; Mg, 4.3 mg 100 g−1 of soil, and pH 5.9. During the experiment, each form of TiO2 was applied after planting (in the second year, at the beginning of the growing season—first half of May) and after the first cut (second half of June). All forms of TiO2 were applied for each plot with a pressure-operated hand sprayer at dose 8.5 g ha−1 disolved in 100 L H2O ha−1.
Second-cut herbage was harvested. Herbage yield was determined in the first year, and herbage yield and chemical composition were determined in the second year. Second-cut herbage, harvested in the second year of cultivation, after 30 days of regrowth, was used for ensiling. Once cut, the plant material was weighed, and herbage samples were collected from each plot. Each herbage harvested from each plot was divided into two batches. The first one was intended for further physicochemical and microbiological analyses, while the second batch was ensiled. For ensiling, crushed herbage (550 g of fresh matter (FM)) was vacuum-packaged in polyethylene bags (38.6 cm × 27.9 cm) using a vacuum-packaging machine (Vacutronic 2000, PP 5.4, ZTP TEPRO, Koszalin, Poland). Samples was ensiled in triplicates. Silage samples were incubated in a constant temperature room at 18 °C. After 90 days, the silage samples was opened and collected. A portion of the samples was dried at 60 °C for 48 h in the Binder FED 115 dryer (GmbH, Tuttlingen, Germany) and ground in the Retsch SK 100 mill (ZM 200, Retsch, Haan, Germany) to a 1 mm particle size. The herbage and silage samples were assayed for proximate chemical composition and carbohydrate fractions. The silage samples also were analyzed for pH, ammonia nitrogen (N-NH3) and concentrations of lactic acid (LA), volatile fatty acids (VFA) and ethanol (Et).
2.4. Chemical Analysis
The samples of herbage and silage were analyzed to determine their chemical composition: DM, crude protein (CP) and crude ash, according to AOAC [26]; WSC—by the anthrone method [27]; neutral-detergent fiber (NDF)—with heat-stable amylase, expressed exclusive of residual ash; and acid detergent fiber (ADF)—expressed exclusive of residual ash, according to Van Soest et al. [28], using the ANKOM220 fiber analyzer (ANKOM Technology Corp., Macedon, NY, USA). The content of N-NH3 was determined by direct distillation using the 2100 Kjeltec Distillation unit (FOSS Analytical A/S, Hilleröd, Denmark) after the pH of the samples had been increased by adding MgO; acidity in fresh samples after opening silage was measured with the HI 8314 pH meter (Hanna Instruments, Woonsocket, RI, USA). The concentrations of lactic acid (LA) and VFAs (acetic (AA), propionic (PA), butyric (BA) and valeric (VA) acids) were determined as described by Kostulak-Zielińska and Potkański [29]. VFAs and Et were separated and determined by gas chromatography (GC) on the Varian 450 GC with the Varian CP-8410 autosampler, flame-ionization detector (FID), CP-FFAP capillary column (length—25 m, inner diameter—0.53 mm, film thickness—1.0 μm), sample size—1 μL, detector temperature—260 °C, injector temperature—200 °C, column temperature—90 °C to 200 °C, carrier gas—helium (flow rate 5.0 mL min−1). LA content was determined by high-performance liquid chromatography (HPLC, SHIMADZU, Kyoto, Japan) with isocratic flow. Separation was carried out using the Varian, Palo Alto, CA, USA METACARB 67H column (ORGANIC ACIDS COLUMN), mobile phase: 0.002 M solution of sulfuric acid in deionized water, flow rate of 1 cm3 min−1, UV detector, 210 nm. External fatty acid standards were supplied by SUPELCO, and the LA standard—by FLUKA (FlukaChemie GmbH, Buchs, Switzerland).
2.5. Quantitative PCR
The bags containing silage samples were opened after 90 days. A detailed description of laboratory chemical and qPCR analyses is presented in Table 2. qPCR results were expressed as the gene copy number of each of the analyzed microbial groups (g−1 DM).

Table 2.
Quantitative polymerase chain reaction setup for the load of selected microbial groups.
2.6. Statistical Analysis
The statistical analysis of results was preceded by an evaluation of normal distribution (Shapiro–Wilk test) and variance homogeneity (Levene test). Then, analysis of variance (ANOVA) was performed with Tukey’s test or Kruskal–Wallis test. Principal Correlation Analysis (PCA) was performed based on the correlation matrix (Pearson’s correlation coefficients). Agglomerative Hierarchical Clustering (AHC) was performed by Euclidean distances with Ward’s agglomerative method. A scatter plot was constructed and correlations were calculated based on untransformed data. All calculations were performed in XLSTAT [37].
3. Results
The effects of selected forms of TiO2 on the yield and chemical parameters of herbage are presented in Table 3. The highest yield was noted for the TiO2NPs1 group, with a significant (p = 0.026) difference, relative to the TiO2Com group (increase of 1.7 Mg ha−1). Applied TiO2 forms affected the proportion of ADF in total structural carbohydrates, which was lowest in the TiO2NPs2 group (significant differences relative to groups TiO2NPs1 and TiO2Com, p = 0.025). The content of the desirable, easily digestible fraction of structural carbohydrates—hemicellulose—was the highest in the TiO2Com group.

Table 3.
The yield, chemical composition and the selected microbial groups of perennial ryegrass herbage.
Highly significant differences in the total bacterial load were observed between all treatments. The highest total bacterial load was noted in the TiO2Com treatment (approx. 5.5 × 107 g−1 herbage), followed by the TiO2NPs1 and TiO2NP2 treatments (approx. 2.1 × 107 and 3.5 × 107 g−1 herbage), respectively. The total bacterial load was one order of magnitude lower in the CONT group (2.2 × 106 g−1 herbage) than in the experimental treatments. The only significant difference in Lactobacillus spp. load was between the groups CONT and TiO2NPs1, and the load was the highest in the TiO2NPs2 treatment (approx. 23-fold increase relative to the remaining treatments). The Clostridium spp. load decrease in response to all forms of TiO2 was highly significant, with the highest (over 7-fold) decrease in the TiO2NPs2 treatment. The Bacillus spp. load was highest in the TiO2Com group, whereas the TiO2NPs1 and TiO2NPs2 treatments contributed to a significant decrease in the counts of these spore-forming bacteria, compared to the CONT group.
Unlike the fungal load, the yeast load decreased (by approx. 15%) after the application of TiO2NPs1, and the change was highly significant. Fungal and yeast loads were similar and significantly higher with the TiO2NPs2 treatment, compared to other treatments. An analysis of the gene copy number of toxigenic fungi revealed that the stimulated growth (by over 40%) of Penicillium spp. was highly significant with the application of TiO2NPs1 and TiO2Com. As regards Fusarium spp., a low copy number of TRI5 genes was found in each sample in the CONT group; however, this plant pathogen was completely eliminated by all forms of TiO2 (Table 3).
The effect of different TiO2 forms on the proximate composition and fermentation pattern of silage is presented in Table 4. An analysis of the proximate composition of silage revealed significant differences in CP content, which was higher in the TiO2NPs2 group than in the TiO2Com group (p = 0.046). Differences were also observed in terms of WSC content, which was higher in the groups of TiO2NPs2 and TiO2Com than in the CONT and TiO2NPs1 groups (p = 0.005). Hence, the application of TiO2NPs2 and TiO2Com reduced the use of WSC by microorganisms during fermentation. The fermentation pattern of silage was comparable in all treatments, except for LA production by microorganisms, which was the highest in the TiO2NPs1 group, with significant differences relative to the CONT and TiO2Com (p = 0.02) groups. However, the percentage of LA in total acids was highly similar across all treatments (92–95% on average). The differences in LA levels were not reflected in the differences in pH values. The lowest pH was determined in the case of TiO2NPs2 treatment, with significant differences relative to the CONT and TiO2NPs1 (p = 0.017) groups.

Table 4.
Chemical composition, fermentation pattern and the selected microbial groups of perennial ryegrass silage.
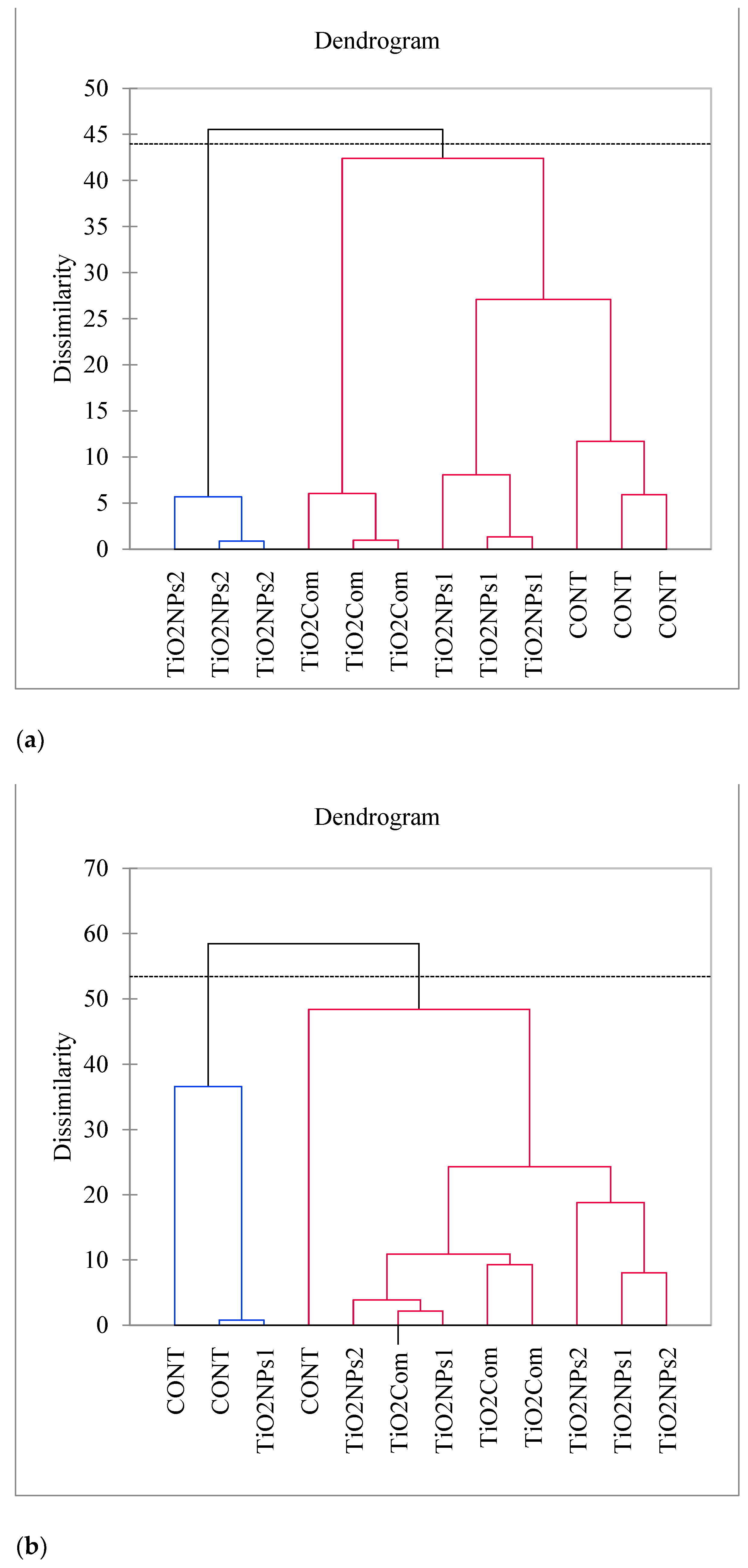
An analysis of the microbiological parameters of silage after 90 days of ensiling revealed fewer significant differences between treatments, in comparison with the herbage. The results of bacteriological analyses were significant only for the Clostridium spp. load, which was nearly completely eliminated in the TiO2NPs2 group and significantly reduced in the TiO2Com group, relative to the CONT group. The total bacterial load and the Lactobacillus spp. load did not differ significantly, but the ratio of Lactobacillus spp. to total bacteria was most favorable in the TiO2NPs2 treatment (i.e., the highest proportion of Lactobacillus spp. in total bacterial counts). In the CONT group, the fungal load was significant only with respect to toxigenic Penicillium spp. The load of this fungal group was significantly decreased by all forms of TiO2, and the noted decrease was the highest in the case of TiO2NPs1. In the CONT group, the total fungal load (yeast and Fusarium spp.) was relatively high in at least one sample. In the experimental treatments, these fungal groups were absent or present only sporadically (Figure 1).
Figure 1.
Dendrogram of AHC results, generated for (a) perennial ryegrass herbage and (b) perennial ryegrass silage, based on the analyzed parameters; the horizontal line and identical color indicate the absence of significant differences within groups of samples.
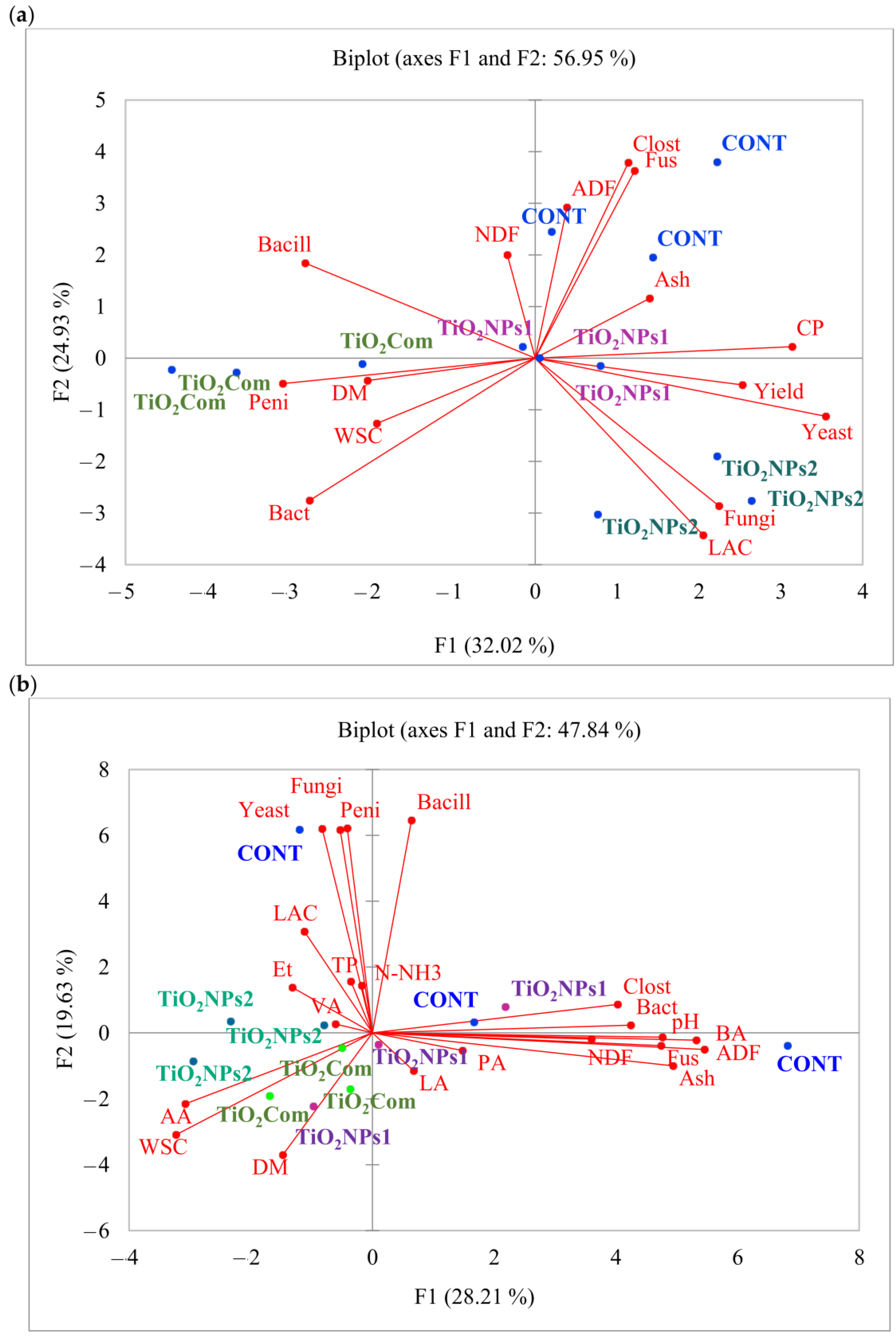
The results of PCA for herbage indicate that high values of ADF, Clostridium spp. and Fusarium spp. were characteristic for the CONT group. The TiO2NPs2 treatment was characterized by high values of Lactobacillus spp., fungal load, yeast load and yield. Some CONT and TiO2NPs2 samples had high CP content. High values of toxigenic Penicillium spp. load, and lower values of total bacterial load, DM content and WSC content were noted in the TiO2Com treatment. No clear relationships were observed in the case of TiO2NPs1 treatment. An analysis of herbage parameters revealed strong correlations between yield, yeast load and CP content, fungal load and Lactobacillus spp. loads, moderate correlations between NDF content, ADF content, Clostridium spp. load and Fusarium spp. load, and moderate correlations between Penicillium spp. load, DM content, WSC content and total bacterial load (Figure 2a).
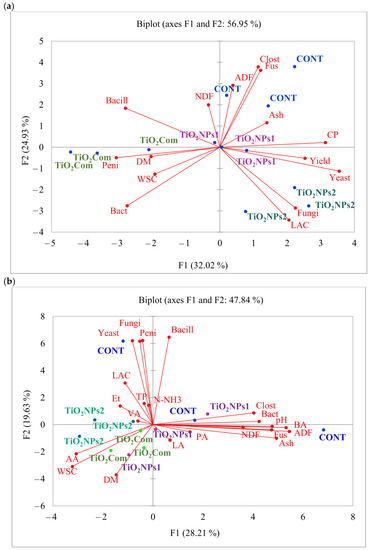
Figure 2.
PCA plot showing correlations between the parameters of (a) perennial ryegrass herbage and (b) perennial ryegrass silage.
Based on the results of the PCA, putative relationships between the factors and the observed features were established. The samples in the CONT group exhibited a low degree of similarity, and there was also some dissimilarity observed between the control and experimental samples. However, one control sample showed significant similarity to the TiO2NPs1 sample, as both were positioned on the positive sides of both vertical lines. The remaining control samples were characterized by higher values of yeast load, Penicillium spp. load, Bacillus spp. load, Clostridium spp. load, Fusarium spp. load, pH and the content of BA, NDF, ADF, and crude ash. In contrast, the TiO2Com treatment exhibited high values of AA, WSC and DM content. On the other hand, the TiO2NPs2 treatment was characterized by high values of AA content, WSC content and to a lesser degree, VA content, Et content and the Lactobacillus spp. load. There was a negative correlation between the contents of AA and WSC and the loads of Clostridium spp. and total bacteria, and a moderate correlation was observed between pH, BA content, ADF content, Fusarium spp. load, crude ash content and NDF content. Additionally, the DM content showed a negative correlation with the Bacillus spp. load and a moderate negative correlation with the Penicillium spp. load, fungal load, and yeast load (Figure 2b).
Regarding the AHC analysis of plants, the results showed well-grouped samples, with three samples forming an individual clade in each treatment. In contrast, silage AHC results were less ordered across treatments and revealed one TiO2NPs1 sample was similar to a control (Figure 1).
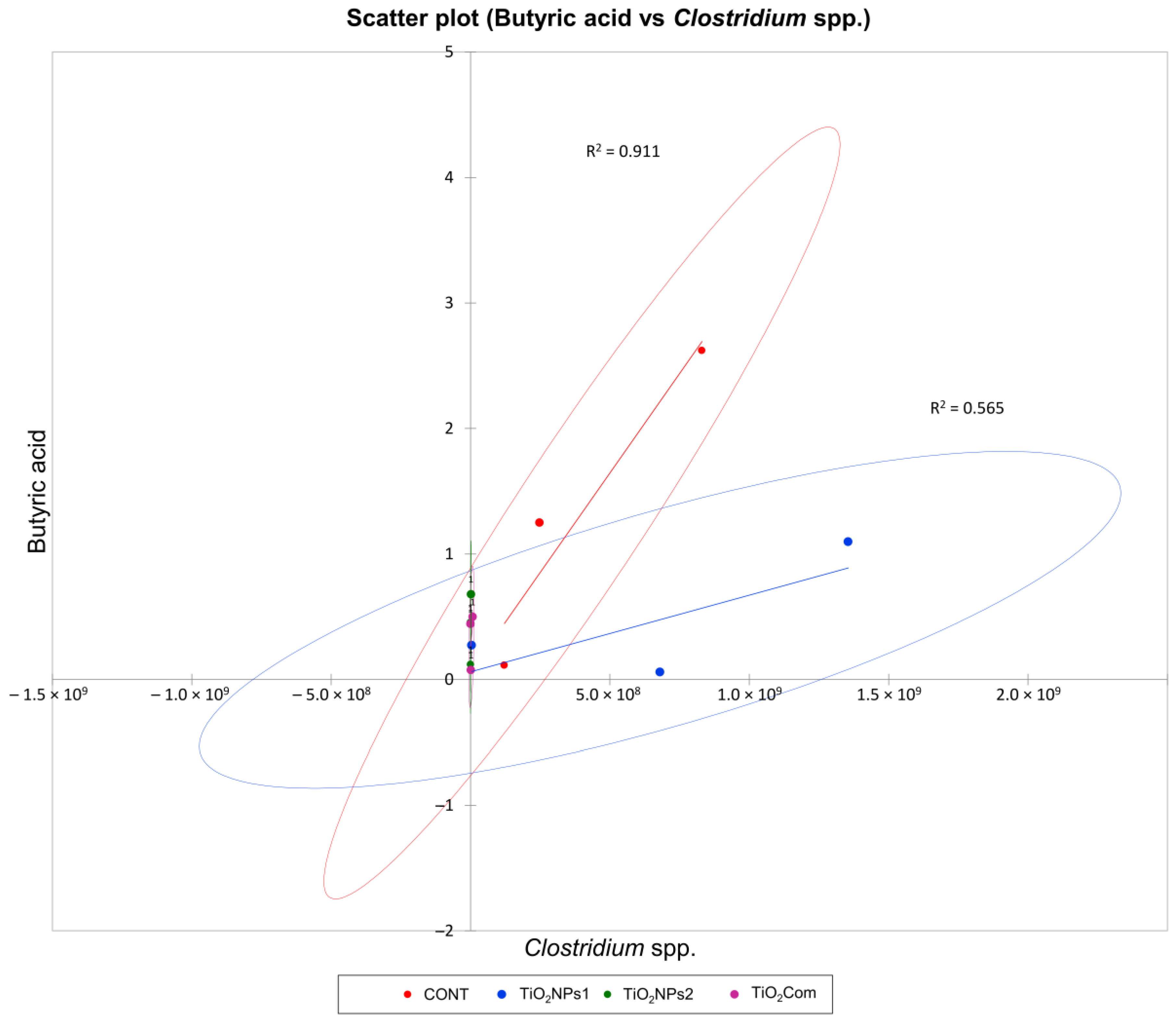
The correlation between BA content and the Clostridium spp. load was very strong (R2 > 0.9) in the CONT group and moderate (R2 = 0.57) in the TiO2NPs1 group. Due to the low Clostridium spp. load and BA concentration, below 1 mg g−1, correlations were not found in the remaining treatments (Figure 3).
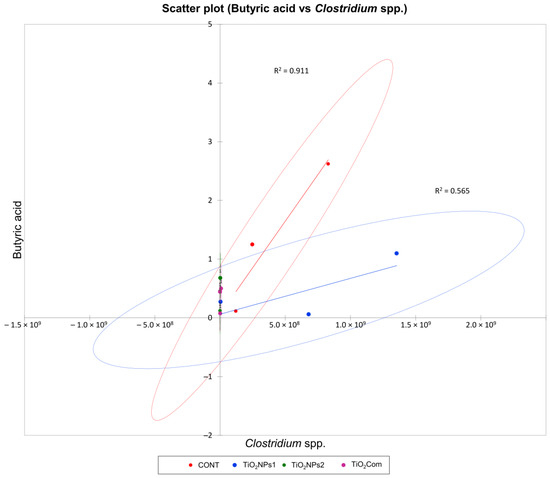
Figure 3.
Scatter plot for perennial ryegrass silage showing a regression curve and a correlation between the Clostridum spp. load (gene copy number g−1 DM silage) and butyric acid content (mg g−1 DM silage).
4. Discussion
The use of nanoparticles in forage production remains insufficiently investigated. The relevant literature data on ensiling and silage are even more limited, which hinders comparative analyses but also testifies to the novelty of this study. However, the effects of metal oxide nanoparticles on plant growth and environmental microbiomes have been extensively researched, which can facilitate the interpretation of the present findings. Nevertheless, it should be remembered that the results presented in this paper are preliminary.
Andersen et al. [38] and Hernández et al. [39] demonstrated that forms of nanoparticles and active TiO2 supported the growth of perennial ryegrass, which was also observed in the case of TiO2NPs1 treatment in the current study, although no significant differences were found (Table 3). The differences in the proportion of structural carbohydrates between treatments could be affected by the mechanisms controlling the biosynthesis of plant cell walls, including hemicellulose biosynthesis [40].
The fermentation pattern of experimental silages was adequate, and the produced feed was characterized by high quality, according to the standards proposed by Kung et al. [41]. The silages had a high proportion of LA in total acids, which points to the high activity of lactic acid bacteria [42]. The efficiency of LA fermentation was the highest in the TiO2Com treatment, as indicated by the highest recovery of WSC in silage and the highest proportion of LA in total acids [42]. In the CONT group, silage was characterized by the highest activity of Clostridium spp., and its BA concentration was higher than the typical suggested values for grass silage with similar DM [41]. In well-fermented silage, BA should be undetectable or present in very low concentrations [43]. These results suggest that the analyzed forms of TiO2 could have a beneficial influence on clostridial fermentation in silage [42]. The rate of CP degradation during ensiling is yet another important indicator of silage quality; in the present experiment, it was not affected by TiO2, as shown by similar N-NH3 levels in the analyzed silages [41,44].
The stimulatory effect of TiO2 and its nanoparticles on plant growth has been well documented, but only a few studies have reported on its positive influence on forage grasses. Latef et al. [45] analyzed the effect of TiO2NPs on the growth of broad bean plants under saline soil conditions. These authors found that due to its antioxidant properties, TiO2NPs significantly decreased plant susceptibility to salinity, already at a low concentration of 0.01%, by enhancing the activity of antioxidant enzymes, which reduced the content of hydrogen peroxide and malondialdehyde. Moreover, elevated levels of proline and other metabolites contributed to osmoprotection, collectively leading to a significant improvement in plant growth under saline conditions. An increase in TiO2 concentrations was not effective, or even suppressed its positive activity. This indicates that TiO2NPs exert a beneficial influence only at low concentrations, which is regulated at the microRNA level in plants. When applied at higher concentrations, TiO2NPs are ineffective, or even toxic to plants, which was reported by Boykov et al. [46]. The above findings suggest that low TiO2NPs concentrations, such as those used in the study, should be recommended and are expected to positively affect the growth and properties of grasses.
An analysis of plant growth revealed that TiO2NPs1 had a potentially beneficial influence of perennial ryegrass yields. However, the wide range of values noted in TiO2NPs1 and CONT groups led to the absence of significant differences and low repeatability of results. The TiO2Com caused a minor decrease in yield, but TiO2NPs1 and TiO2Com tended to increase WSC content relative to CONT group. A previous study [25] investigating TiO2NPs revealed that their positive effect on the growth of bread wheat, a member of the family Poaceae, resulted mostly from improved germination, which was also observed in this field-plot experiment.
In the cited study [25], no significant differences were found in the counts of selected microorganisms, but the study focused on the epiphytic microbiota of wheat and flax roots. In the current experiment, the abundance of microbial biomarkers in perennial ryegrass biomass and roots was considerably different. The microbiological quality of herbage can be improved by pre-treatment, drying, treatment with hydrogen peroxide and ionization [47]. In the present study, the exposure to TiO2 in any form significantly decreased the counts of Clostridium spp., Penicillium spp. and Fusarium spp. in perennial ryegrass biomass. Moreover, the application of TiO2NPs1 and TiO2NPs2 decreased the counts of Bacillus spp., the application of TiO2Com reduced yeast counts, and the proportion of Lactobacillus spp. was higher in the TiO2NPs2 treatment. Despite considerable differences in results and low correlations in the PCA plot, the fact that the abundance of toxigenic fungi and Clostridium spp. (characteristic only of the CONT treatment) was reduced is highly encouraging. Burke et al. [48] investigated the effect of positively and negatively charged iron oxide nanoparticles (Fe3O4NPs) and TiO2NPs on the growth of soybean plants and found that TiO2NPs had a more beneficial influence on the microbiome and nodule formation in the root system than Fe3O4NPs. These results and the findings of Gorczyca et al. [25] suggest that the effect exerted by a given substance on plants and the microbiome is also determined by other factors such as charge, pH, redox potential and particle size. In the current study, three forms of TiO2 exerted varied effects on perennial ryegrass plants and the microbiome.
Silages treated with TiO2Com and TiO2NPs2 had high WSC content and the most desirable pH. The microbiological parameters of silage were partially different than those of herbage. All silages treated with TiO2NPs2 were characterized by a minor decrease in total bacterial counts and Bacillus spp. counts, and almost complete elimination of fungi, yeasts, Penicillium spp. and Fusarium spp. TiO2Com led to a minor decrease in the counts of Lactobacillus spp.; however, this had no influence on LA content. In one sample, TiO2NPs1 did not inhibit the growth of Clostridium spp., which was correlated with increased BA concentration, observed also in the control treatment (Figure 3).
The results of PCA and AHC indicate that silages in groups treated with TiO2NPs2 and TiO2Com were relatively similar, and had a high content of AA, WSC and DM, which was negatively correlated with Clostridium spp. counts, total bacterial counts, high pH, concentrations of BA, ADF and NDF, Fusarium spp. counts and crude ash content. The growth of toxigenic fungi was observed in one sample in group CONT (first quarter in the PCA plot). However, the content of yeasts and filamentous fungi, including Penicillium spp., was negatively correlated with LA concentration. An analysis of AHC results revealed that samples formed clades within treatments. Only the TiO2NPs2 treatment differed significantly from the other treatments. As regards herbage, control and TiO2NPs1 treatments were highly similar. As regards silage, the CONT group and one sample in the TiO2NPs1 treatment differed considerably from the remaining samples. A combined analysis of AHC and PCA results indicates that the application of TiO2NPs2 has a potentially positive effect on the quality of herbage and silage, whereas the effect of TiO2NPs1 was weakest and most similar to that of the CONT treatment. In herbage, the treatments were satisfactorily grouped, pointing to considerable modification of parameters and high stability of experimental treatments relative to CONT group. Such high stability was not noted in silage; nevertheless, the CONT group differed from all TiO2–treated samples, with the exception of TiO2NPs1. It should be remembered that these studies are preliminary and are used to indicate the types of titanium nanoparticles of potential importance for grasses and silages.
5. Conclusions
The present study demonstrated that TiO2NPs have a some potential for agronomic applications, which is consistent with previous findings [49]. TiO2 and its nanoparticles affect the physicochemical and, in particular, microbiological quality of perennial ryegrass herbage and silage. The effects exerted by TiO2 vary significantly depending on its form and properties. Large (207 nm) and negatively charged TiO2NPs2 have a positive effect on the quality of herbage and silage. The analyzed commercial preparation containing non-nano TiO2 exerts an intermediate effect; whereas, small (68 nm) and positively charged TiO2NPs1 may be unsuitable for field applications and ensiling. However, in order to confirm the potential of the tested titanium dioxide nanoparticles for grass cultivation and animal feeding, large-scale field studies and animal studies should be conducted.
Author Contributions
Conceptualization, S.W.P., M.O. and C.P.; methodology, S.W.P., C.P. and M.B.-S.; software, M.O., O.K. and M.B.-S.; validation, S.W.P. and C.P.; formal analysis, S.W.P. and O.K.; investigation, S.W.P. and C.P.; resources, M.B.-S., M.O. and O.K.; data curation, S.W.P., M.O. and M.B.-S.; writing—original draft preparation, S.W.P., O.K., M.O., M.B.-S. and C.P.; writing—review and editing, S.W.P., M.B.-S. and C.P.; visualization, S.W.P. and M.B.-S.; supervision, O.K., C.P. and M.O.; project administration, S.W.P., O.K. and M.B.-S.; funding acquisition, S.W.P. and C.P. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by the University of Warmia and Mazury in Olsztyn, Faculty of Agriculture and Forestry, Department of Entomology, Phytopathology and Molecular Diagnostics, grant No. 30.610.011-110. Project financially supported by Minister of Education and Science in the range of the program entitled “Regional Initiative of Excellence” for the years 2019–2023, Project No. 010/RID/2018/19, amount of funding 12.000.000 PLN.
Institutional Review Board Statement
Not applicable.
Data Availability Statement
The datasets utilized in this study’s analysis are available upon reasonable request.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Blackmore, T.; Thomas, I.; McMahon, R.; Powell, W.; Hegarty, M. Genetic–geographic correlation revealed across a broad European ecotypic sample of perennial ryegrass (Lolium perenne) using array-based SNP genotyping. Theor. Appl. Genet. 2015, 128, 1917–1932. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Griffiths, P.; Nendel, C.; Pickert, J.; Hostert, P. Towards national-scale characterization of grassland use intensity from integrated Sentinel-2 and Landsat time series. Remote Sens. Environ. 2020, 238, 111124. [Google Scholar] [CrossRef]
- Humphreys, M.W.; Yadav, R.S.; Cairns, A.J.; Turner, L.B.; Humphreys, J.; Skot, L. A changing climate for grassland research. New Phytol. 2006, 169, 9–26. [Google Scholar] [CrossRef] [PubMed]
- Bumane, S. The influence of NPK fertilization on Lolium perenne L. forage quality. Agron. Res. 2010, 8, 531–536. [Google Scholar]
- He, X.; Deng, H.; Hwang, H. The current application of nanotechnology in food and agriculture. J. Food Drug. Anal. 2019, 27, 1–21. [Google Scholar] [CrossRef] [PubMed]
- Du, W.; Sun, Y.; Ji, R.; Zhu, J.; Wu, J.; Guo, H. TiO2 and ZnO nanoparticles negatively affect wheat growth and soil enzyme activities in agricultural soil. J. Environ. Monit. 2011, 13, 822–828. [Google Scholar] [CrossRef] [PubMed]
- Servin, A.D.; Morales, M.I.; Castillo-Michel, H.; Hernandez-Viezcas, J.A.; Munoz, B.; Zhao, L.; Gardea-Torresdey, J.L. Synchrotron verification of TiO2 accumulation in cucumber fruit: A possible pathway of TiO2 nanoparticle transfer from soil into the food chain. Environ. Sci. Technol. 2013, 47, 11592–11598. [Google Scholar] [CrossRef]
- Kosmala, K.; Szymańska, R. Nanocząstki tlenku tytanu (IV)-otrzymywanie, właściwości i zastosowanie. Kosmos 2016, 65, 235–245. (In Polish) [Google Scholar]
- Fujishima, A.; Zhang, X.; Tryk, D.A. TiO2 photocatalysis and related surface phenomena. Surf. Sci. Rep. 2008, 63, 515–582. [Google Scholar] [CrossRef]
- Radkowski, A.; Radkowska, I.; Lemek, T. Effects of foliar application of titanium on seed yield in timothy (Phleum pratense L.). Ecol. Chem. Eng. S 2015, 22, 691–701. [Google Scholar] [CrossRef]
- Topalović, O.; Hussain, M.; Heuer, H. Plants and associated soil microbiota cooperatively suppress plant-parasitic nematodes. Front. Microbiol. 2020, 11, 313. [Google Scholar] [CrossRef] [PubMed]
- Lin, J.; Li, G.; Sun, L.; Wang, S.; Meng, X.; Sun, L.; Yuan, L.; Xu, L. Varieties and ensiling: Impact on chemical composition, fermentation quality and bacterial community of alfalfa. Front. Microbiol. 2023, 13, 1091491. [Google Scholar] [CrossRef]
- Przemieniecki, S.W.; Oćwieja, M.; Ciesielski, S.; Halecki, W.; Matras, E.; Gorczyca, A. Chemical Structure of Stabilizing Layers of Negatively Charged Silver Nanoparticles as an Effector of Shifts in Soil Bacterial Microbiome under Short-Term Exposure. Int. J. Environ. Res. Public Health 2022, 19, 14438. [Google Scholar] [CrossRef]
- Dumon, J.C.; Ernst, W.H.O. Titanium in plants. J. Plant. Physiol. 1988, 133, 203–209. [Google Scholar] [CrossRef]
- Carvajal, M.; Alcaraz, C.F. Why titanium is a beneficial element for plants. J. Plant Nutr. 1998, 21, 655–664. [Google Scholar] [CrossRef]
- Zheng, L.; Hong, F.; Lu, S.; Liu, C. Effect of nano-TiO2 on strength of naturally aged seeds and growth of spinach. Biol. Trace. Elem. Res. 2005, 104, 83–91. [Google Scholar] [CrossRef]
- Rico, C.M.; Barrios, A.C.; Tan, W.; Rubenecia, R.; Lee, S.C.; Varela-Ramirez, A.; Gardea-Torresdey, J.L. Physiological and biochemical response of soil-grown barley (Hordeum vulgare L.) to cerium oxide nanoparticles. Environ. Sci. Pollut. Res. 2015, 22, 10551–10558. [Google Scholar] [CrossRef]
- Wang, Y.; Deng, C.; Cota-Ruiz, K.; Peralta-Videa, J.R.; Hernandez-Viezcas, J.A.; Gardea-Torresdey, J.L. Soil-aged nano titanium dioxide effects on full-grown carrot: Dose and surface-coating dependent improvements on growth and nutrient quality. Sci. Total. Environ. 2021, 774, 145699. [Google Scholar] [CrossRef]
- Lyu, S.; Wei, X.; Chen, J.; Wang, C.; Wang, X.; Pan, D. Titanium as a beneficial element for crop production. Front. Plant. Sci. 2017, 8, 597. [Google Scholar] [CrossRef]
- Larue, C.; Laurette, J.; Herlin-Boime, N.; Khodja, H.; Fayard, B.; Flank, A.M.; Brisset, F.; Carriere, M. Accumulation, translocation and impact of TiO2 nanoparticles in wheat (Triticum aestivum spp.): Influence of diameter and crystal phase. Sci. Total Environ. 2012, 431, 197–208. [Google Scholar] [CrossRef]
- Foltête, A.S.; Masfaraud, J.F.; Bigorgne, E.; Nahmani, J.; Chaurand, P.; Botta, C.; Labille, J.; Rose, J.; Férard, J.F.; Cotelle, S. Environmental impact of sunscreen nanomaterials: Ecotoxicity and genotoxicity of altered TiO2 nanocomposites on Vicia faba. Environ. Pollut. 2011, 159, 2515–2522. [Google Scholar] [CrossRef] [PubMed]
- Tan, W.; Peralta-Videa, J.R.; Gardea-Torresdey, J.L. Interaction of titanium dioxide nanoparticles with soil components and plants: Current knowledge and future research needs—A critical review. Environ. Sci. Nano 2018, 5, 257–278. [Google Scholar] [CrossRef]
- Cox, A.; Venkatachalam, P.; Sahi, S.; Sharma, N. Review: Reprint of: Silver and titanium dioxide nanoparticle toxicity in plants: A review of current research. Plant. Physiol. Biochem. 2017, 110, 33–49. [Google Scholar] [CrossRef]
- Faraz, A.; Faizan, M.; Fariduddin, Q.; Hayat, S. Response of Titanium Nanoparticles to Plant Growth: Agricultural Perspectives. Agric. Rev. 2020, 41, 101–110. [Google Scholar] [CrossRef]
- Gorczyca, A.; Przemieniecki, S.W.; Kurowski, T.; Oćwieja, M. Early plant growth and bacterial community in rhizoplane of wheat and flax exposed to silver and titanium dioxide nanoparticles. Environ. Sci. Pollut. Res. 2018, 25, 33820–33826. [Google Scholar] [CrossRef]
- AOAC. Official Methods of Analysis, 15th ed.; Association of Official Analytical Chemists: Washington, DC, USA, 2005. [Google Scholar]
- Thomas, T.A. An automated procedure for the determination of soluble carbohydrates in herbage. J. Sci. Food Agric. 1977, 28, 639–642. [Google Scholar] [CrossRef]
- Van Soest, P.J.; Robertson, J.B.; Lewis, B.A. Symposium: Carbohydrate methodology, metabolism and nutritional implications in dairy cattle. J. Dairy Sci. 1991, 74, 3583–3597. [Google Scholar]
- Kostulak-Zielińska, M.; Potkański, A. Quality of baled grass-clover silages ensiled with chemical additives. Chemical composition. Ann. Anim. Sci. 2011, 1, 153–165. [Google Scholar]
- Haarman, M.; Knol, J. Quantitative real-time PCR analysis of fecal Lactobacillus species ininfants receiving a prebiotic infant formula. App. Environ. Microb. 2006, 72, 2359–2365. [Google Scholar] [CrossRef]
- Song, Y.; Liu, C.; Finegold, S.M. Real-time PCR quantitation of clostridia in feces of autistic children. Appl. Environ. Microbiol. 2004, 70, 6459–6465. [Google Scholar] [CrossRef]
- Hierro, N.; Esteve-Zarzoso, B.; Gonzalez, A.; Mas, A.; Guillamon, J.M. Real-time quantitative PCR (QPCR) and reverse transcription-QPCR for detection and enumeration of total yeasts in wine. Appl. Environ. Microbiol. 2006, 72, 7148–7155. [Google Scholar] [CrossRef] [PubMed]
- Martin, K.J.; Rygiewicz, P.T. Fungal-specific PCR primers developed for analysis of the ITS region of environmental DNA extracts. BMC Microbiol. 2005, 18, 28. [Google Scholar] [CrossRef]
- Hemkemeyer, M.; Christensen, B.T.; Martens, R.; Tebbe, C.C. Soil particle size fractions harbour distinct microbial communities and differ in potential for microbial mineralisation of organic pollutants. Soil Biol. Biochem. 2015, 90, 255–265. [Google Scholar] [CrossRef]
- Mora, I.; Cabrefiga, J.; Montesinos, E. Antimicrobial peptide genes in Bacillus strains from plant environments. Int. Microbiol. 2011, 14, 213–223. [Google Scholar] [CrossRef]
- Schnerr, H.; Niessen, L.; Vogel, R.F. Real time detection of the tri5 gene in Fusarium species by LightCycler-PCR using SYBR Green I for continuous fluorescence monitoring. Int. J. Food Microbiol. 2001, 71, 53–61. [Google Scholar] [CrossRef]
- Lumivero. XLSTAT statistical and data analysis solution. New York, USA, 2023. Available online: https://www.xlstat.com/en (accessed on 5 January 2023).
- Andersen, C.P.; King, G.; Plocher, M.; Storm, M.; Pokhrel, L.R.; Johnson, M.G.; Rygiewicz, P.T. Germination and early plant development of ten plant species exposed to titanium dioxide and cerium oxide nanoparticles. Environ. Toxicol. Chem. 2016, 35, 2223–2229. [Google Scholar] [CrossRef]
- Hernández, A.; Loera, N.; Pérez, G.; Blockstrand, F. Vinylic and Waterproofing Paint with TiO2 as Photocatalytic Active Effects in Lolium perenne Germination. In Energy Technology; Wang, T., Chen, X., Guillen, D.P., Zhang, L., Sun, Z., Wang, C., Haque, N., Howarter, J.A., Neelameggham, N.R., Ikhmayies, S., et al., Eds.; The Minerals, Metals & Materials Series; Springer: Cham, Germany, 2019. [Google Scholar]
- Pauly, M.; Gille, S.; Liu, L.; Mansoori, N.; de Souza, A.; Achultink, A.; Xiong, G. Hemicellulose biosynthesis. Planta 2013, 238, 627–642. [Google Scholar] [CrossRef]
- Kung, L., Jr.; Shaver, R.D.; Grant, R.J.; Schmidt, R.J. Silage review: Interpretation of chemical, microbial, and organoleptic components of silages. J. Dairy Sci. 2018, 101, 4020–4033. [Google Scholar] [CrossRef]
- McDonald, P.; Henderson, A.R.; Heron, S.J.E. The Biochemistry of Silage, 2nd ed.; Chalcombe Publications: Marlow Bucks, UK, 1991. [Google Scholar]
- Pahlow, G.; Muck, R.E.; Driehuis, F.; Oude Elferink, S.J.W.H.; Spoelstra, S.F. Microbiology of Ensiling; Buxton, D.R., Muck, R.E., Harrison, J.H., Eds.; Silage Science and Technology, American Society of Agronomy: Madison, WI, USA, 2003; pp. 31–93. [Google Scholar]
- Davies, D.R.; Merry, R.J.; Williams, A.P.; Bakewell, E.L.; Leemans, D.K.; Tweed, J.K. Proteolysis during ensilage of forages varying in soluble sugar content. J. Dairy Sci. 1998, 81, 444–453. [Google Scholar] [CrossRef]
- Latef, A.A.H.A.; Srivastava, A.K.; Abd El-Sadek, M.S.; Kordrostami, M.; Tran, L.-S.P. Titanium Dioxide Nanoparticles Improve Growth and Enhance Tolerance of Broad Bean Plants under Saline Soil Conditions. Land Degrad. Dev. 2018, 29, 1065–1073. [Google Scholar] [CrossRef]
- Boykov, I.N.; Shuford, E.; Zhang, B. Nanoparticle titanium dioxide affects the growth and microRNA expression of switchgrass (Panicum virgatum). Genomics 2019, 111, 450–456. [Google Scholar] [CrossRef] [PubMed]
- Kukier, E.; Kwiatek, K.; Grenda, T.; Goldsztejn, M. Microflora of silages. Vet. Life 2014, 89, 1031–1036. [Google Scholar]
- Burke, D.J.; Pietrasiak, N.; Situ, S.F.; Abenojat, E.C.; Porche, M.; Kraj, P.; Lakliang, Y.; Samia, A.C. Iron Oxide and Titanium Dioxide Nanoparticle Effects on Plant Performance and Root Associated Microbes. Int. J. Mol. Sci. 2015, 16, 23630–23650. [Google Scholar] [CrossRef] [PubMed]
- Šebesta, M.; Kolenčík, M.; Sunil, B.R.; Illa, R.; Mosnáček, J.; Ingle, A.P.; Urík, M. Field Application of ZnO and TiO2 Nanoparticles on Agricultural Plants. Agronomy 2021, 11, 2281. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).