1. Introduction
Yellow maize (
Zea mays L.) is one of the most widespread cereals in the world. It is used in various ways as food, feed and biofuel, and in many industrial and commercial products such as flour, cornmeal, grits, starch, snacks, tortillas and breakfast cereals. In domestic animal nutrition, maize is used as a primary source of energy [
1,
2]. In addition to its macronutrient composition, maize contributes to the diet with a variety of phytochemicals, including carotenoids, which provide desirable health benefits for humans and animals due to their antioxidant and provitamin A properties [
3]. For this reason, maize is part of the staple diet of millions of people in Latin America, Asia and Africa [
1]. When used as animal feed, the carotenoids from maize improve the production performance and health of animals [
4], but are also deposited in animal products [
5,
6], which in turn serve as an important source of carotenoids in the human diet.
Yellow maize exhibits considerable natural variation in grain carotenoids (14.48–32.61 g/kg dry matter (DM)), characterized by higher concentrations of xanthophylls (lutein and zeaxanthin) compared to provitamin A carotenoids (α-cryptoxanthin, β-cryptoxanthin and β-carotene) [
7,
8]. Within the maize kernel, approximately 95–97% of the carotenoids are found in the endosperm, while the remaining carotenoids are distributed between the germ (2–4%) and the pericarp (1%) [
9]. Maize hybrids differ in grain vitreousness, which is genetically determined and is expressed as a ratio of the vitreous endosperm, located in the outer part of the kernel, and the floury endosperm, located in the center of the kernel, with more vitreous samples being harder than less vitreous ones [
10,
11]. The hardness distinction between two types of endosperm has been linked to the textural and chemical properties of maize kernels [
10,
12], while more recent findings showed that genotypes with different kernel hardness have the ability to store specific carotenoids [
8,
13,
14].
The carotenoid biosynthetic pathway is divided into two branches (the α- and the β-branch), with lutein and α-cryptoxanthin being the major carotenoids of the α-branch in maize, while the major carotenoids of the β-branch are zeaxanthin, β-cryptoxanthin and β-carotene [
15]. Maize genotypes with harder kernels have been shown to have, on average, a higher total carotenoid content and a higher concentration of β-branch carotenoids. In comparison, higher levels of α-branch carotenoids have been observed in softer kernels [
8,
13]. In addition, the authors of the latter study reported a positive correlation between the total carotenoid content and the kernel vitreousness, and a negative correlation with the flotation index. In agreement, another study showed a higher provitamin A content in a genotype with higher kernel bulk density [
16]. Previous studies have shown that the textural properties associated with kernel hardness are related to the physical and chemical composition [
10,
12]. In this context, measurements of kernel physical properties, such as size and shape, weight, density and milling resistance, can probably serve as predictors of the carotenoid composition of maize kernels.
On the other hand, the differences between endosperm types are the result of the interactions between the main storage products, i.e., proteins, starch and lipids [
17]. The vitreousness reflects the compactness of the starch–protein matrix, with harder kernels generally associated with higher amylose, protein, zein and lipid content, and lower starch content than softer ones [
12,
18,
19]. The carotenoids in maize are predominantly found in the amyloplasts and storage lipids of the endosperm in immature kernels [
14], but to our knowledge, there are few data on their relationship to other kernel chemical properties. It has been reported that harder maize hybrids can store more carotenoids [
8,
13], which may be related to their hydrophobic interaction with protein bodies [
20]. Momany et al. [
21] reported that lutein is located in the core of α-zein segments with a triple helix that stabilizes its configuration. Since a more vitreous endosperm has a greater ability to store zeins [
20], the different protein composition between hard and soft kernels may affect their carotenoid profile [
8,
13]. Nevertheless, differences in chemical composition have been linked to the in vitro carotenoid digestibility of maize kernels [
22], suggesting that the chemical composition may be related to the carotenoid profile of maize hybrids.
Previous studies have shown that different maize hybrids exhibit wide genetic variation and diversity in terms of carotenoid profile and kernel physicochemical properties [
7,
8,
10,
12,
19]. Although studies have shown a relationship between the kernel hardness and the specific carotenoid profile [
8,
13,
14], there are few data on the relationship with most physical and almost all chemical properties. In this way, it would be possible to gain insight into the carotenoid profile of maize hybrids with simpler analyses. Nevertheless, hardness and hardness-related physical properties are affected by the kernel vitreousness, which is related to the carotenoid content and the chemical composition of the maize kernels [
8,
17,
19]. Investigating the relationship between the carotenoid profile and the physical and chemical properties of the grain could, therefore, provide valuable insights into the relationship with specific properties, thus revealing the possible function of carotenoids in the kernel structure. Therefore, the aim of the present study was to investigate the potential relationship between the physicochemical properties and the carotenoid profile in the kernels of 15 commercial maize hybrids. The physical properties determined in the present study were kernel size and shape, 1000 kernel weight and volume, bulk density, Stenvert hardness, breakage susceptibility, kernel density and flotation index, whereas chemical properties consisted of the contents of ash, crude protein, crude fat, starch, amylose, amylopectin and zein.
2. Materials and Methods
2.1. Maize Hybrids
Fifteen high-yielding yellow maize hybrids (
Zea mays L.;
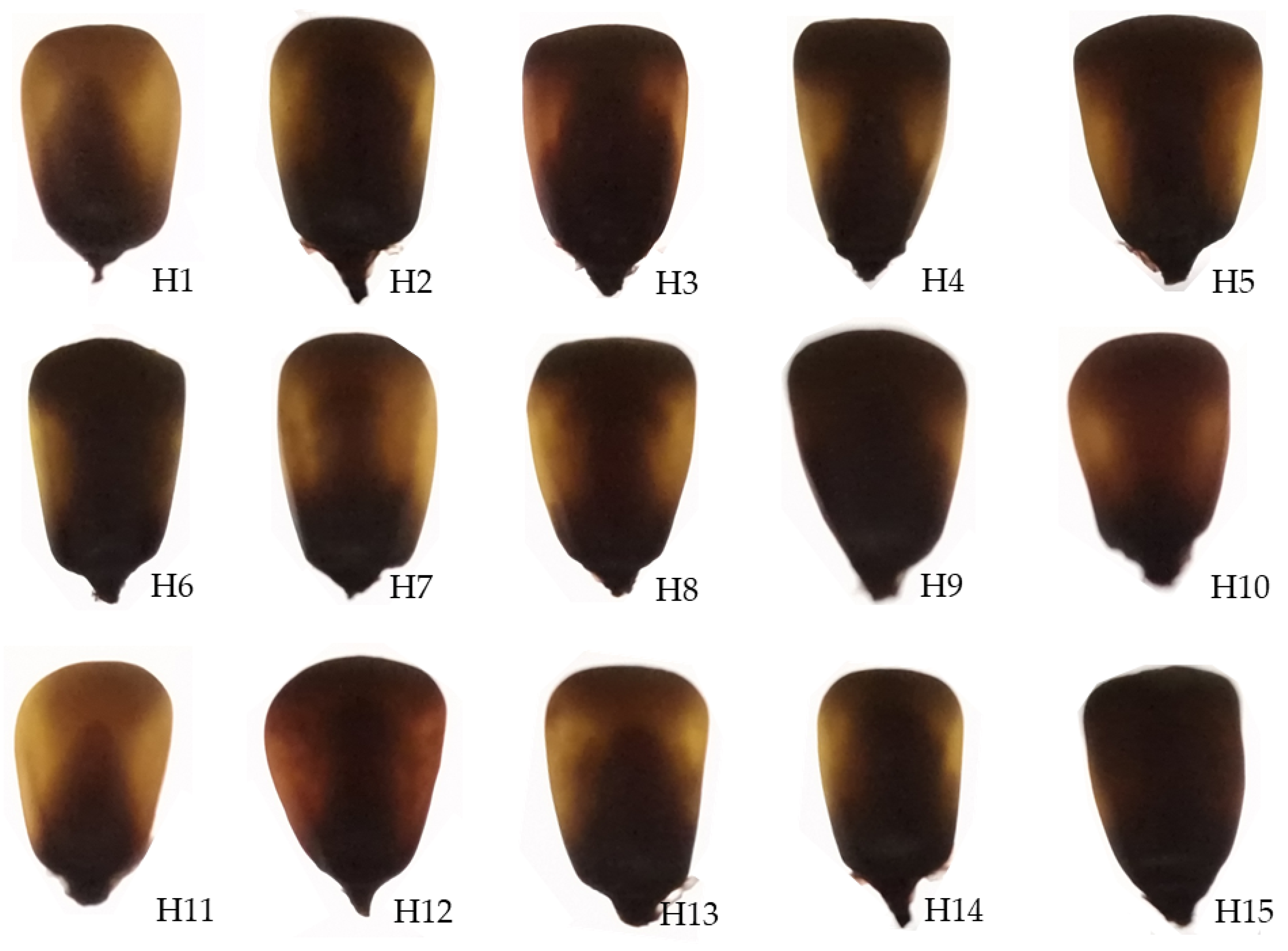
Table 1) belonging to various maturity groups were used in this study. Their visual appearances are shown in
Figure 1, whereas images of kernels on a light box are shown in
Figure 2. In addition, the color parameters according to CIE L*a*b* [
23] are listed in
Table 2. The tested hybrids were selected based on physicochemical properties, carotenoid profile and in vitro carotenoid bioaccessibility from 103 commercial maize hybrids from 9 seed companies (
Table 1) available on the market [
22]. Selection was conducted to obtain a wide range of these properties to ensure the variability of commercial maize hybrids regardless of the seed company.
The production of maize grains was described in a previously published paper [
24]. Five locations were selected for each plot, representing 5 replicates for each hybrid, and 10 ears were hand-harvested from each location after physiological maturity. The ears were dried at 40 °C to approximately 120 g/kg moisture, shelled and stored at 4 °C until analysis. Whole grains were used to analyze the physical properties, while the grains for chemical and carotenoid analysis were ground in a laboratory mill (Cyclotec 1093, Foss Tocator, Hoganas, Sweden) with a 1 and 0.3 mm sieve, respectively, immediately before analysis. The moisture content of all samples was determined by drying at 102 °C for 4 (ground grains) or 24 h (whole grains) [
25].
Table 1.
Tested modern maize hybrids belonging to various maturity groups.
Table 1.
Tested modern maize hybrids belonging to various maturity groups.
| Hybrid | Abbreviation | Seed Company | Type 1 | FAO Maturity Group | Estimated Vitreousness [26] 2 |
|---|
| Hybrid 1 | H1 | Bc Institute d.d., Zagreb, Croatia | semiflint | 330 | 70.0 ± 0.28 |
| Hybrid 2 | H2 | Agricultural Institute Osijek, Osijek, Croatia | dent | 350 | 60.5 ± 0.35 |
| Hybrid 3 | H3 | Syngenta Agro d.o.o., Zagreb, Croatia | dent | 350 | 58.1 ± 0.40 |
| Hybrid 4 | H4 | KWS Sjeme d.o.o., Osijek, Croatia | dent | 390 | 57.4 ± 0.28 |
| Hybrid 5 | H5 | Corteva Agriscience Croatia d.o.o., Zagreb, Croatia | hard dent | 380 | 58.7 ± 0.42 |
| Hybrid 6 | H6 | Corteva Agriscience Croatia d.o.o., Zagreb, Croatia | dent | 400 | 56.1 ± 0.62 |
| Hybrid 7 | H7 | Agricultural Institute Osijek, Osijek, Zagreb | dent | 410 | 65.2 ± 0.55 |
| Hybrid 8 | H8 | Bayer d.o.o., Zagreb, Croatia | dent | 450 | 60.3 ± 0.40 |
| Hybrid 9 | H9 | Bc Institute d.d., Zagreb, Croatia | dent | 450 | 56.1 ± 0.31 |
| Hybrid 10 | H10 | Bc Institute d.d., Zagreb, Croatia | hard dent | 460 | 61.6 ± 0.24 |
| Hybrid 11 | H11 | Bc Institute d.d., Zagreb, Croatia | hard dent | 500 | 68.3 ± 0.43 |
| Hybrid 12 | H12 | Bc Institute d.d., Zagreb, Croatia | hard dent | 510 | 63.6 ± 0.32 |
| Hybrid 13 | H13 | Bc Institute d.d., Zagreb, Croatia | hard dent | 510 | 60.1 ± 0.22 |
| Hybrid 14 | H14 | Corteva Agriscience Croatia d.o.o., Zagreb, Croatia | dent | 570 | 58.3 ± 0.75 |
| Hybrid 15 | H15 | Syngenta Agro d.o.o., Zagreb, Croatia | dent | 580 | 53.9 ± 0.17 |
Table 2.
Color parameters of ground kernels of tested maize hybrids (brightness (L*), redness (a*), yellowness (b*)) (n = 5) 1.
Table 2.
Color parameters of ground kernels of tested maize hybrids (brightness (L*), redness (a*), yellowness (b*)) (n = 5) 1.
| Hybrid | L* | a* | b* |
|---|
| H1 | 85.46 ± 0.36 | −0.28 ± 0.04 | 34.82 ± 0.23 |
| H2 | 87.56 ± 0.18 | −0.38 ± 0.11 | 32.53 ± 0.37 |
| H3 | 89.31 ± 0.22 | −0.24 ± 0.06 | 25.21 ± 0.28 |
| H4 | 89.00 ± 0.40 | −1.24 ± 0.06 | 29.91 ± 0.37 |
| H5 | 88.80 ± 0.28 | −0.67 ± 0.13 | 30.88 ± 0.54 |
| H6 | 90.01 ± 0.45 | −1.24 ± 0.11 | 28.37 ± 0.43 |
| H7 | 87.15 ± 0.49 | −0.23 ± 0.08 | 37.70 ± 0.38 |
| H8 | 88.30 ± 0.31 | −0.62 ± 0.07 | 32.89 ± 0.23 |
| H9 | 89.08 ± 0.34 | −1.44 ± 0.08 | 30.78 ± 0.28 |
| H10 | 85.52 ± 0.28 | −0.20 ± 0.07 | 32.59 ± 0.21 |
| H11 | 85.77 ± 0.41 | 1.11 ± 0.16 | 38.73 ± 0.30 |
| H12 | 85.41 ± 0.55 | 0.97 ± 0.15 | 36.38 ± 0.37 |
| H13 | 88.82 ± 0.30 | −1.57 ± 0.13 | 37.10 ± 0.50 |
| H14 | 89.13 ± 0.47 | −1.16 ± 0.08 | 30.08 ± 0.71 |
| H15 | 90.59 ± 0.40 | −2.10 ± 0.07 | 32.55 ± 0.19 |
Figure 1.
The visual appearance of 15 tested maize hybrids. H1–H15—the abbreviated names of the tested maize hybrids as listed in
Table 1.
Figure 1.
The visual appearance of 15 tested maize hybrids. H1–H15—the abbreviated names of the tested maize hybrids as listed in
Table 1.
Figure 2.
Kernels of tested maize hybrids on a light box. H1–H15—the abbreviated names of the tested maize hybrids as listed in
Table 1.
Figure 2.
Kernels of tested maize hybrids on a light box. H1–H15—the abbreviated names of the tested maize hybrids as listed in
Table 1.
2.2. Analyses of Physical Properties
The kernel dimensions (length, width and thickness) were measured using a digital caliper and the data were used to calculate the kernel sphericity [
27]. Weight and volume of 250 g per replicate were recorded, and the test weight was calculated by dividing the weight by the volume. Kernel density was determined based on kernel weight and the volume it occupied using a pycnometer with ethanol as solvent. The flotation index was determined by stirring the maize kernels in a sodium nitrate solution with a relative density of 1.25 [
11]. The breakage susceptibility was determined using the HT-I drop test apparatus, which was constructed according to the scheme described by Kim et al. [
28]. Kernel hardness was determined using the Stenvert hardness test according to the method described by Pomeranz et al. [
27]. The parameters of Stenvert kernel hardness were the time required to grind 17 mL of grits, the height of the grits in the grinding column and the ratio of coarse (>0.7 mm) to fine particles (<0.5 mm; C/F) in the grits; harder kernels have a longer grinding time, greater height of grits and a higher C/F. Each sample was analyzed in at least triplicate.
2.3. Analyses of Chemical Properties
The ash, crude protein (CP), crude fat (CF) and starch content of the maize samples was determined according to the standard methods. To determine ash content, the samples were ashed at 550 °C to a constant weight (ISO 5984:2022) [
29]. The CP content was calculated as Kjeldahl nitrogen multiplied by a factor of 6.25 (ISO 5983-2:2009; [
30]), while the CF content was determined gravimetrically using the Soxhlet extraction method (ISO 6492:1999; [
31]). The total starch content was determined using a commercial enzymatic kit (Total Starch Assay Procedure, amyloglucosidase/α-amylase method, Megazyme, Ireland) according to the AOAC 996.11 method [
32].
The amylose content was determined spectrophotometrically by dissolving ground maize samples with a DMSO-iodine solution. The apparent amylose content was corrected for the amylopectin content using the formula of Knutson [
33]. Total zein was extracted using sodium borate buffer containing sodium dodecyl sulfate and 2-mercaptoethanol [
34]. The extractions were performed three times, the supernatants were pooled and the non-zein proteins were precipitated with ethanol. The supernatant, representing the total zein fraction, was dried at 60 °C in Kjeldahl tubes and analyzed for nitrogen using the Kjeldahl method. The zein content was calculated using a conversion factor of 6.25, and the zein was expressed as content in DM and as a percentage of CP.
2.4. Analyses of Chemical Properties
Carotenoids were extracted and quantified from the maize samples according to the method described by Kurilich and Juvik [
35], using β-apo-carotenal as an internal standard. Samples were sonicated (10 min; Sonorex TK 52, Bandelin, Berlin, Germany) and homogenized with ethanol containing 0.1% butylhydroxytoluene (BHT) (1 min per sample; T10 Ultra-Turaxx, IKA, Staufen, Germany). The mixtures were then saponified with 80% KOH (10 min) in a water bath at 85 °C. After cooling, the carotenoids were extracted with hexane until the upper hexane layer was colorless. The collected supernatants were evaporated (RVC 2-25CD plus, Martin Christ, Osterode am Harz, Germany) and dissolved in 200 µL of acetonitrile/methanol/methylene chloride (45:20:35,
v/
v/
v) containing 0.1% BHT. Extractions were carried out under dim light, and extracts were analyzed further using HPLC on the same day.
Carotenoids were separated and quantified using the SpectraSystem HPLC instrument (Thermo Separation Products, Inc., Waltham, MA, USA) equipped with a quaternary gradient pump, an autosampler and a UV-vis detector. Carotenoids were separated on two sequentially connected C18 reversed-phase columns, Vydac 201TP54 column (5 µm, 4.6 × 150 mm; Hichrom, Reading, UK) and Zorbax RX-C18 column (5 µm, 4.6 × 150 mm; Agilent Technologies, Santa Clara, CA, USA). The separation columns were protected by a Supelguard Discovery C18 guard column (5 µm, 4 × 20 mm; Supelco, Bellefonte, PA, USA). The mobile phase consisted of acetonitrile/methanol/dichloromethane (75:25:5, v/v/v) containing 0.1% BHT and 0.05% triethylamine. An aliquot of 30 µL was injected, and the flow rate was 1.8 mL/min. The separations were performed at room temperature. Carotenoids were monitored on a UV-Vis detector at 450 nm.
Separated compounds were identified by comparing their retention times and quantified using external standardization with calibration curves using commercially available carotenoid standards (lutein (purity 99%), zeaxanthin (purity 99%), α- and β-cryptoxanthin (purity of both 99%) and β-carotene (purity 98%) (Extrasynthese, Genay, France); and α-carotene (purity 97%) (Supelco, Merck KGaA, Darmstadt, Germany); R2 ≥ 0.99 for all carotenoids). The total carotenoid content was calculated by summing the contents of the individual carotenoids. Each hybrid was analyzed in triplicate and the mean value was taken as the result.
The content of individual and total carotenoids in the tested hybrids was previously presented [
36], and in the present study, the carotenoid profile of tested hybrids was presented as content of fractions of the following: xanthophylls (by summing the contents of lutein, zeaxanthin, α- and β-cryptoxanthin), carotenes (by summing the contents of α- and β-carotene), α-branch carotenoids (by summing the contents of lutein, α-cryptoxanthin and α-carotene) and β-branch carotenoids (by summing the contents of zeaxanthin, β-cryptoxanthin and β-carotene).
2.5. Statistical Analysis
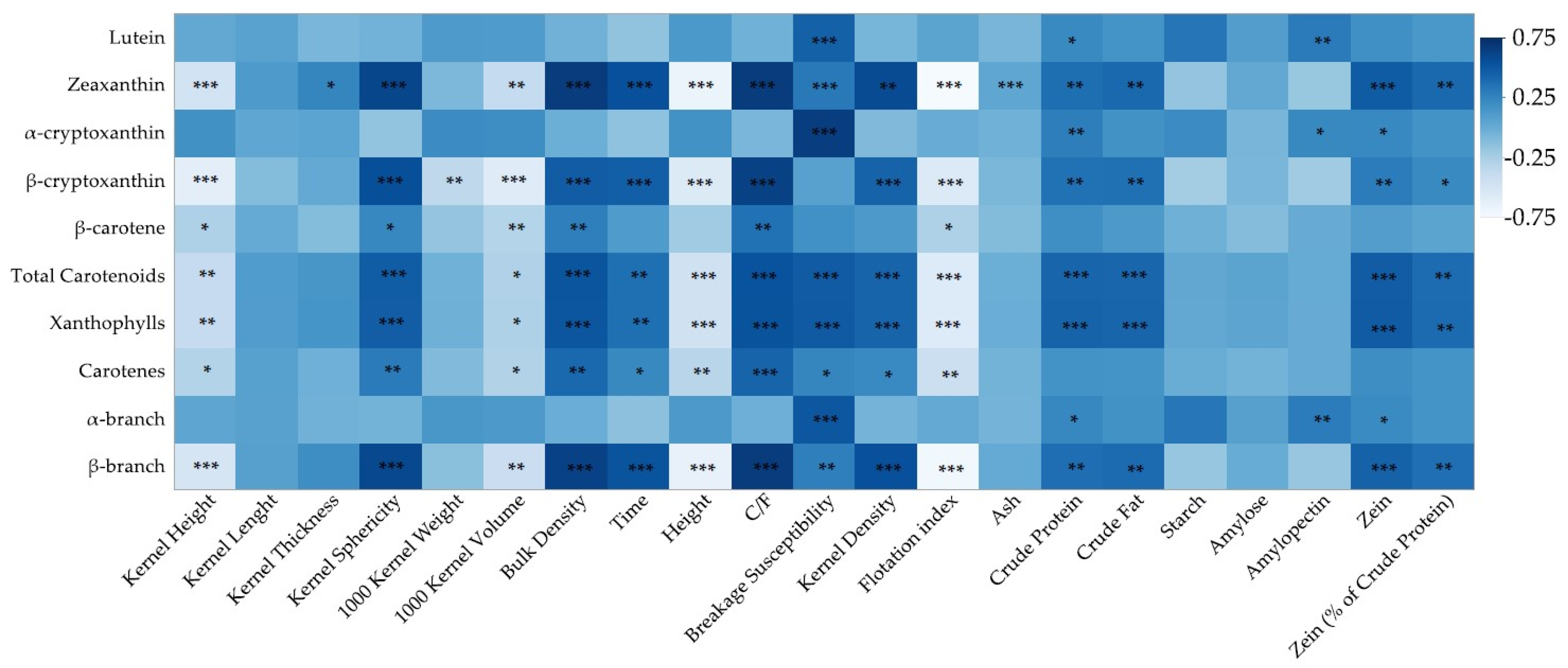
The obtained results were analyzed using SAS statistical software (version 9.4; SAS Institute Inc., Cary, NC, USA). The experiment was conducted in a completely randomized design with five replicates. Differences between the hybrids in analyzed properties were determined using analysis of variance with hybrid as a fixed effect using the MIXED procedure. Mean values were defined using the least squares means statement and compared using the PDIFF option; letter groups were determined using the PDMIX macro procedure. The relationship between determined properties, including contents of individual and total carotenoids, was analyzed using the CORR procedure. The threshold for statistical significance was defined as p < 0.05.
4. Conclusions
The tested hybrids differed in their physical properties, macronutrient composition and contents of xanthophylls, carotenes, and α- and β-branch carotenoids. Hybrids with a higher kernel hardness, crude protein, crude fat and zein content had higher contents of zeaxanthin, β-cryptoxanthin, total carotenoids, xanthophylls and β-branch carotenoids. However, regardless of the positive correlation between xanthophylls, total carotenoids, physical properties and nutrient composition, the contents of lutein, α-cryptoxanthin and α-branch carotenoids were only positively correlated with the crude protein content. Surprisingly, they were also correlated positively with amylopectin. The results obtained support previous findings that maize genotypes of higher hardness have a higher content of β-branch carotenoids, and it was confirmed that this relationship exists within genotypes of the same type since 14 of the tested hybrids were dent type. In addition, the results gave an insight into the relationship between the carotenoid profile and grain hardness-related properties, such as the kernel size, density, bulk density, and zein and amylose content. Specific carotenoids were associated with specific traits, leading to a separate role of α- and β-branch carotenoids and implying a different role of β-carotenoids in the hardness-associated properties of commercial maize hybrids. These hardness-associated properties also imply a higher quality of maize grain, and the production of maize hybrids with higher hardness, which can be easily determined using simple methods, implies an enhanced nutritional value for humans and animals due to the higher carotenoid content.