Mycobiome and Pathogenic Fusarium Fungi in the Rhizosphere of Durum Wheat After Seed Dressing with Debaryomyces hansenii
Abstract
:1. Introduction
2. Materials and Methods
2.1. Site Description, Agricultural Practices, Weather Conditions, and Sampling
2.2. Preparation of the Debaryomyces hansenii Suspension for the Biological Treatment
2.3. Soil Characteristics
2.4. DNA Isolation from Soil
2.5. Amplification of DNA Fragments by PCR
2.6. Illumina MiSeq Sequencing
2.7. Analysis of Selected Biometric Parameters and the Yield Potential of Durum Wheat
2.8. Analysis of Stem Base and Grain Health
2.9. Mycological Analysis of the Stem Base and Grain
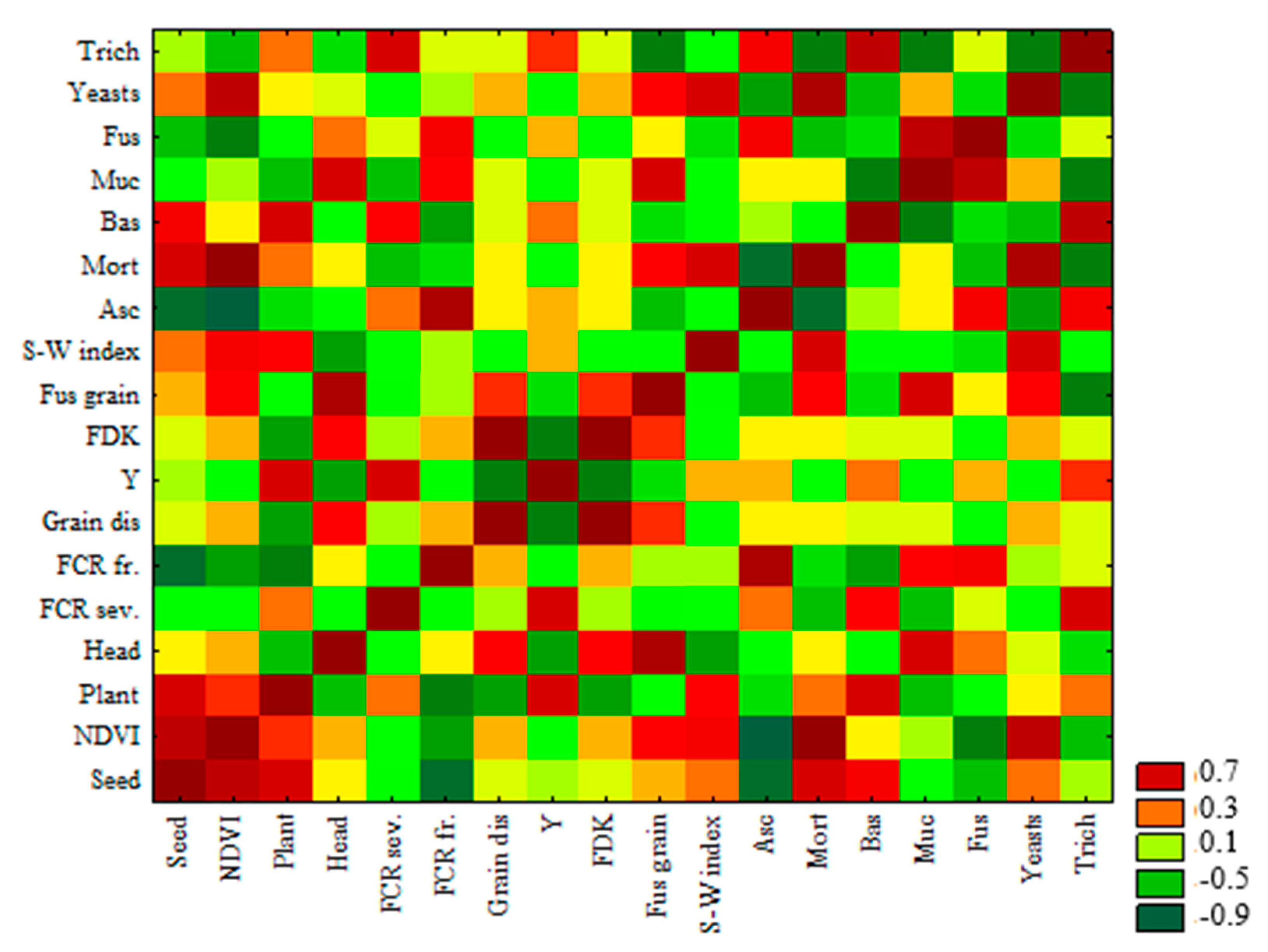
2.10. Statistical Analysis
3. Results
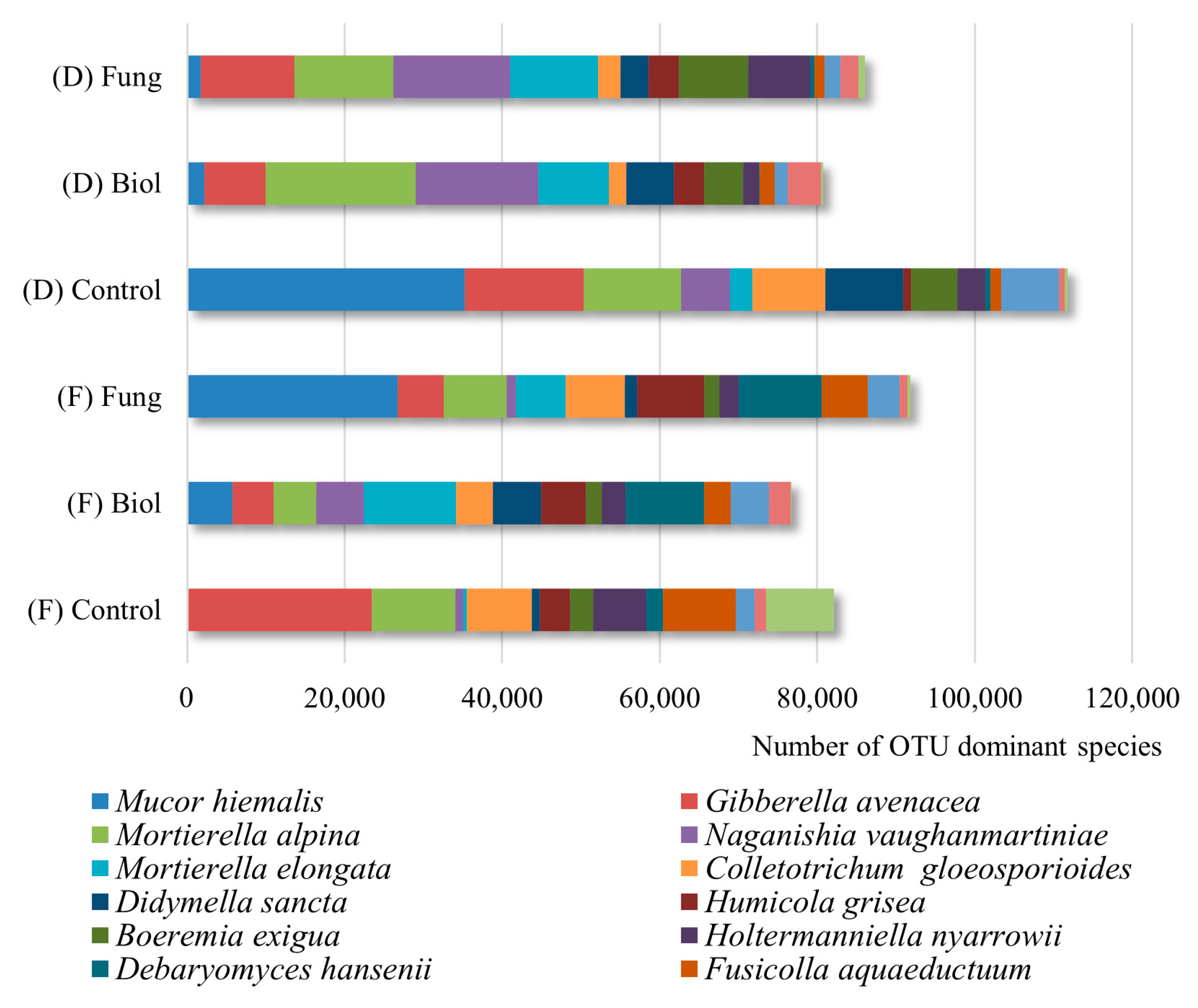
3.1. Fungal Community Profiles
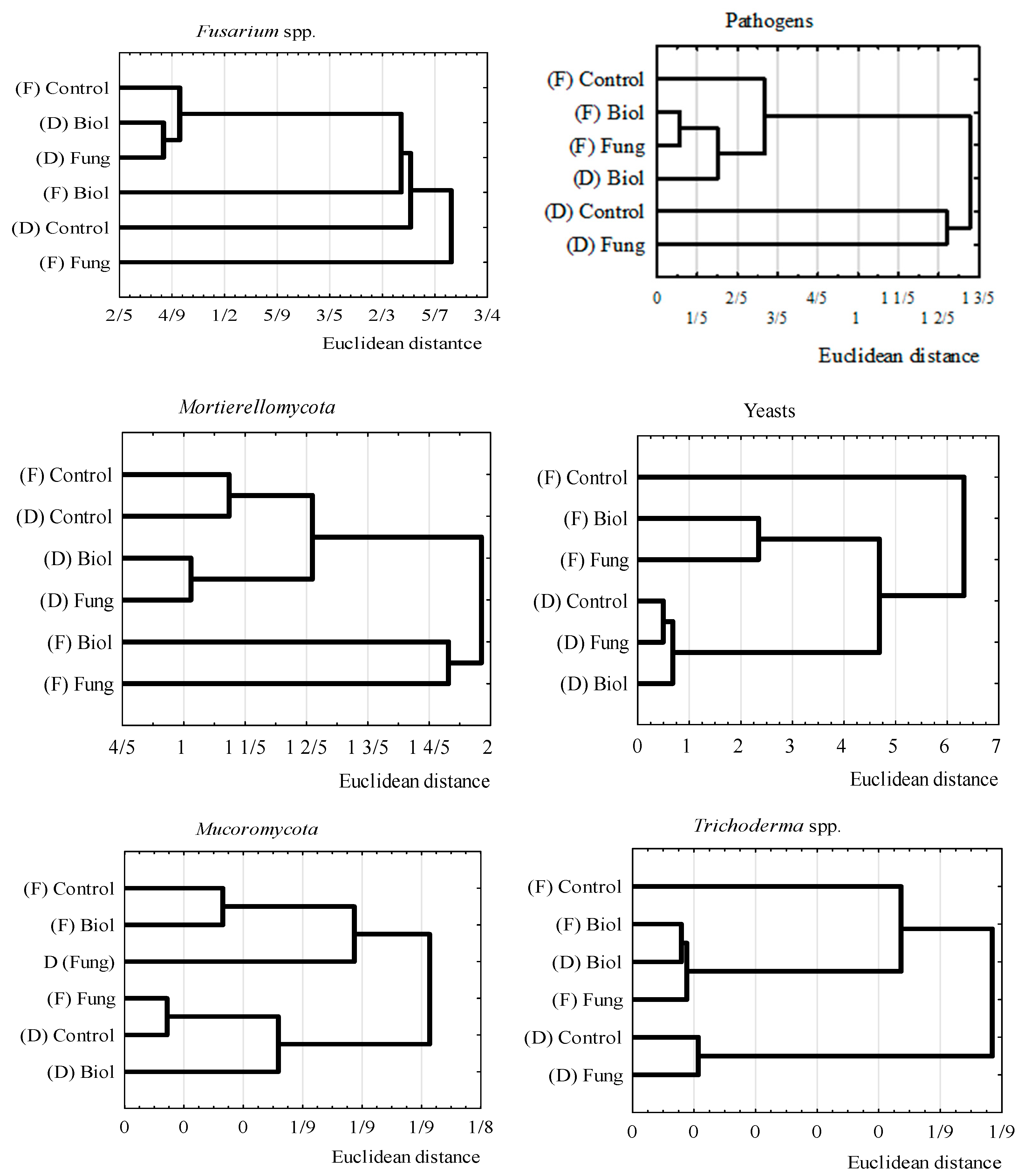
3.2. Differences in Fungal Communities Between Durum Wheat Cultivars and Seed Treatments
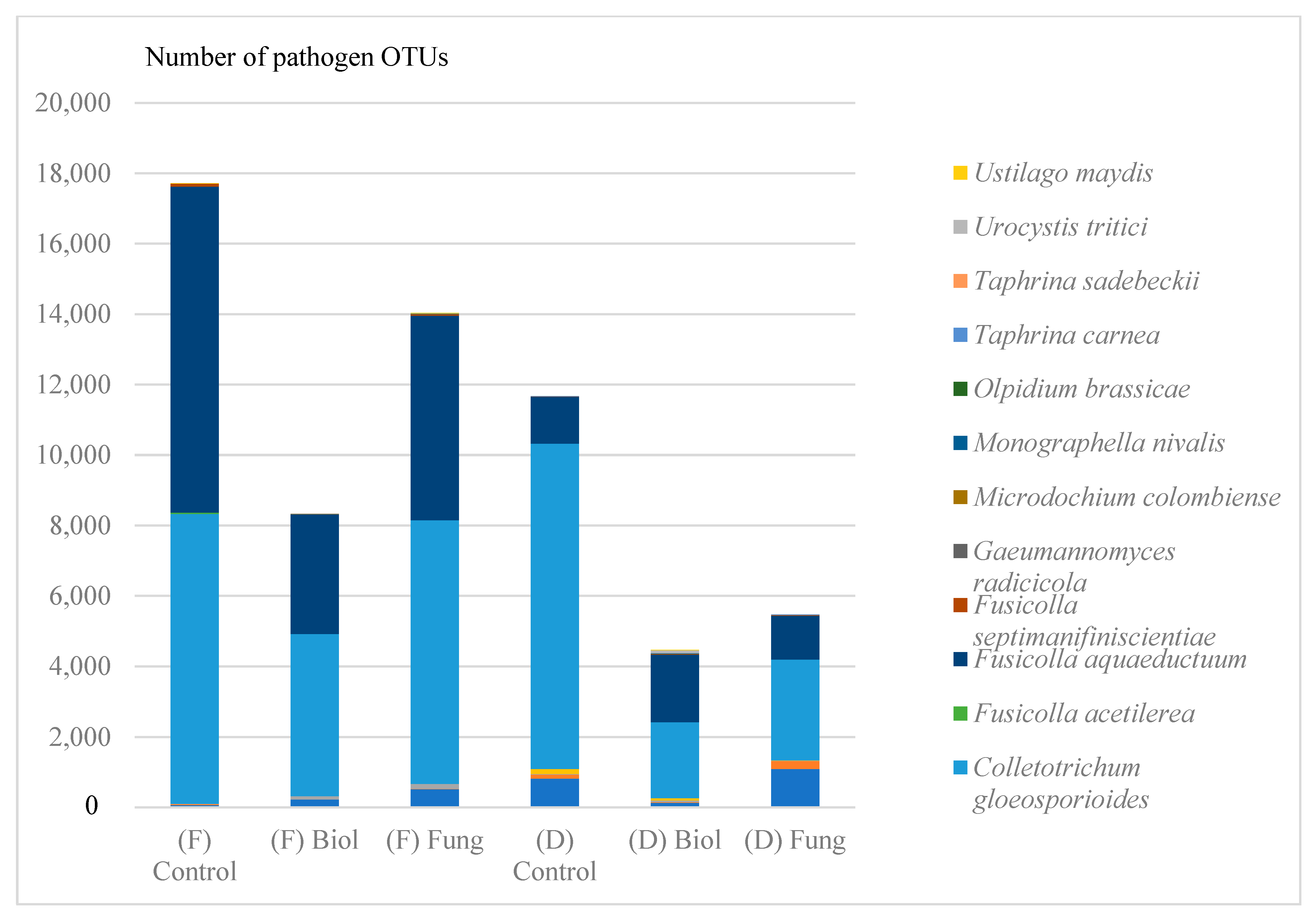
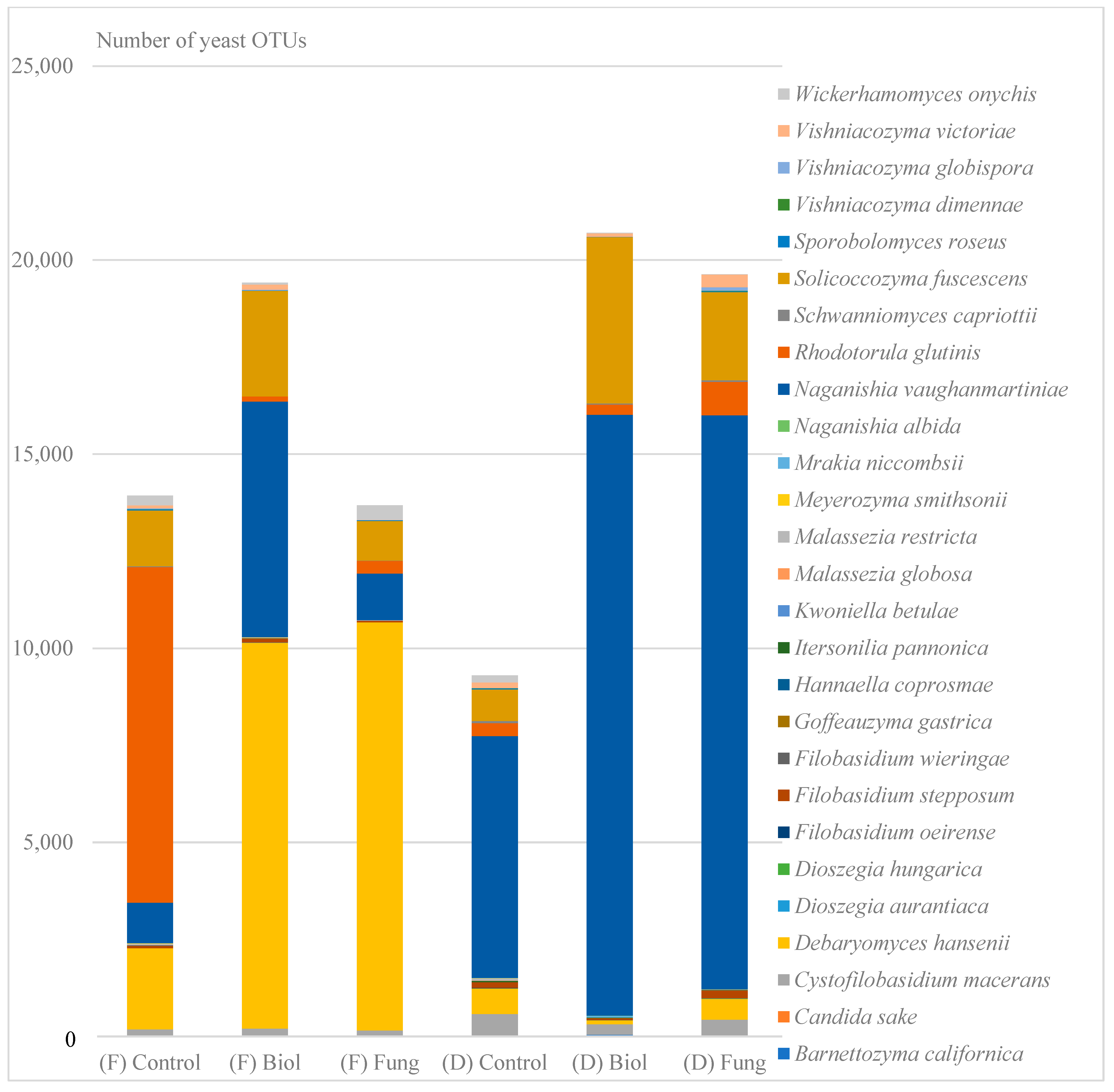
3.3. The Influence of Seed Treatment on the Number of Fungal Taxonomic and Functional Groups
3.4. The Effect of Seed Dressing on Plant Health, Biometric Parameters, and Yield-Related Traits
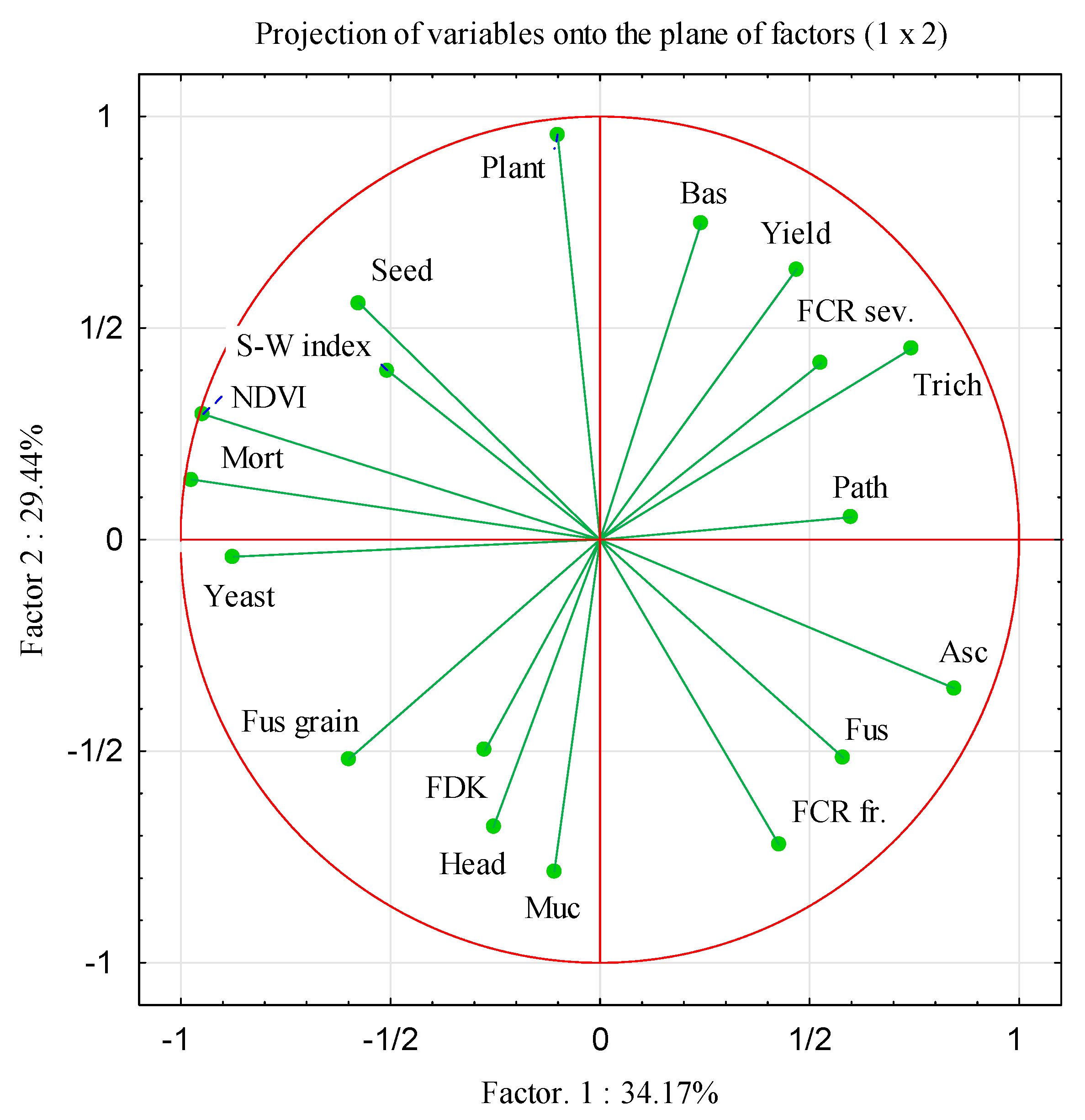
3.5. Principal Component Analysis
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Bobryk-Mamczarz, A.; Kiełtyka-Dadasiewicz, A.; Rachoń, L. Usefulness of hulled wheats grown in polish environment for wholegrain pasta-making. Foods 2021, 10, 458. [Google Scholar] [CrossRef] [PubMed]
- Spychaj, R.; Gil, Z.; Chrzanowska-Drożdż, B. Technological value of winter durum wheat cv. Komnata as dependent on chemical plant protection. Biul. Inst. Hod. Aklim. Rośl. 2011, 262, 25–38. [Google Scholar]
- Kuzdraliński, A.; Szczerba, H.; Tofil, K.; Filipiak, A.; Garbarczyk, E.; Dziadko, P.; Solarska, E. Early PCR-based detection of Fusarium culmorum, F. graminearum, F. sporotrichioides and F. poae on stem bases of winter wheat throughout Poland. Eur. J. Plant Pathol. 2014, 140, 491–502. [Google Scholar] [CrossRef]
- Lenc, L.; Czecholiński, G.; Wyczling, D.; Turów, T.; Kaźmierczak, A. Fusarium head blight (FHB) and Fusarium spp. on grain of spring wheat cultivars grown in Poland. J. Plant Prot. Res. 2015, 55, 266–275. [Google Scholar] [CrossRef]
- Błaszczyk, L.; Salamon, S.; Mikołajczak, K. Fungi inhabiting the wheat endosphere. Pathogens 2021, 10, 1288. [Google Scholar] [CrossRef]
- Wachowska, U.; Sulyok, M.; Wiwart, M.; Suchowilska, E.; Kandler, W.; Krska, R. The application of antagonistic yeasts and bacteria: An assessment of in vivo and under field conditions pattern of Fusarium mycotoxins in winter wheat grain. Food Control 2022, 138, 109039. [Google Scholar] [CrossRef]
- Lyu, D.; Zajonc, J.; Pagé, A.; Tanney, C.A.S.; Shah, A.; Monjezi, N.; Msimbira, L.A.; Antar, M.; Nazari, M.; Backer, R.; et al. Plant holobiont theory: The phytomicrobiome plays a central role in evolution and success. Microorganisms 2021, 24, 675. [Google Scholar] [CrossRef]
- Rosenberg, E.; Zilber-Rosenberg, I. Microbes drive evolution of animals and plants: The hologenome concept. MBio 2016, 7, 11101128. [Google Scholar] [CrossRef]
- Wachowska, U.; Rychcik, B. Plants control the structure of mycorrhizal and pathogenic fungal communities in soil in a 50-year maize monoculture experiment. Plant Soil 2023, 484, 133–153. [Google Scholar] [CrossRef]
- Salamon, S.; Mikołajczak, K.; Błaszczyk, L. Constellation of the endophytic mycobiome in spring and winter wheat cultivars grown under various conditions. Sci. Rep. 2023, 13, 6089. [Google Scholar] [CrossRef]
- Walsh, C.M.; Becker-Uncapher, I.; Carlson, M.; Fierer, N. Variable influences of soil and seed-associated bacterial communities on the assembly of seedling microbiomes. ISME J. 2021, 15, 2748–2762. [Google Scholar] [CrossRef] [PubMed]
- Johnston-Monje, D.; Gutiérrez, J.P.; Lopez-Lavalle, L.A.B. Seed-transmitted bacteria and fungi dominate juvenile plant microbiomes. Front. Microbiol. 2021, 12, 737616. [Google Scholar] [CrossRef] [PubMed]
- Danielewicz, J.; Korbas, M.; Horoszkiewicz-Janka, J.; Jajor, E.; Krzymińska, J. The use of reduced doses of fungicides in integrated plant protection. Zagadnienia Doradz. Rol. 2015, 80, 97–106. [Google Scholar]
- Ayesha, M.S.; Suryanarayanan, T.S.; Nataraja, K.N.; Prasad, S.R.; Shaanker, R.U. Seed treatment with systemic fungicides: Time for review. Front. Plant Sci. 2021, 12, 654512. [Google Scholar] [CrossRef] [PubMed]
- Keyser, C.A.; Jensen, B.; Meyling, N.V. Dual effects of Metarhizium spp. and Clonostachys rosea against an insect and a seed-borne pathogen in wheat. Pest Manag. Sci. 2016, 72, 517–526. [Google Scholar] [CrossRef]
- Del Puerto, O.; Gonçalves, N.P.F.; Medana, C.; Prevot, A.B.; Roslev, P. Attenuation of toxicity and occurrence of degradation products of the fungicide tebuconazole after combined vacuum UV and UVC treatment of drinking water. Environ. Sci. Pollut. Res. 2022, 29, 58312–58325. [Google Scholar] [CrossRef]
- Collinge, D.B.; Jensen, D.F.; Rabiey, M.; Sarrocco, S.; Shaw, M.W.; Shaw, R.H. Biological control of plant diseases—What has been achieved and what is the direction? Plant Pathol. 2022, 71, 1024–1047. [Google Scholar] [CrossRef]
- Ibrahim, E.A.M.; Abo El-Dahab, M.S. Influence of seed dressing by yeast extract and fungicides on seed quality of wheat during storage. J. Plant Prod. Mansoura Univ. 2017, 8, 187–193. [Google Scholar] [CrossRef]
- Paravar, A.; Piri, R.; Balouchi, H.; Ma, Y. Microbial seed coating: An attractive tool for sustainable agriculture. Biot. Rep. 2023, 37, e00781. [Google Scholar] [CrossRef]
- Wallace, M.E. Root-Inhabiting and Rhizosphere Mycobiomes and Crop Yield of Corn and Wheat. Master’s Thesis, The University of Western Ontario, London, ON, Canada, 2022; p. 9028. Available online: https://ir.lib.uwo.ca/etd/9028 (accessed on 14 May 2021).
- Chen, J.; Sharifi, R.; Khan, M.S.S.; Islam, F.; Bhat, J.A.; Kui, L.; Majeed, A. Wheat microbiome: Structure, dynamics, and role in improving performance under stress environments. Front. Microbiol. 2022, 12, 821546. [Google Scholar] [CrossRef]
- Trivedi, P.; Leach, J.E.; Tringe, S.G.; Sa, T.; Singh, B.K. Plant-microbiome interactions: From community assembly to plant health. Nat. Rev. Microbiol. 2020, 18, 607–621. [Google Scholar] [CrossRef] [PubMed]
- Wachowska, U.; Kucharska, K.; Pluskota, W.; Czaplicki, S.; Stuper-Szablewska, K. Bacteria associated with winter wheat degrade Fusarium mycotoxins and triazole fungicide residues. Agronomy 2020, 10, 1673. [Google Scholar] [CrossRef]
- Rai, A.; Belkacem, M.; Assadi, I.; Bollinger, J.C.; Elfalleh, W.; Assadi, A.A.; Amrane, A.; Mouni, L. Bacteria in soil: Promising bioremediation agents in arid and semi-arid environments for cereal growth enhancement. Appl. Sci. 2022, 12, 11567. [Google Scholar] [CrossRef]
- Fedotov, G.N.; Shoba, S.A.; Fedotova, M.F.; Stepanov, A.L.; Streletsky, R.A. Soil yeasts and their role in seed germination. Eurasian Soil Sci. 2017, 50, 573–579. [Google Scholar] [CrossRef]
- Alfonzo, A.; Sicard, D.; Di Miceli, G.; Guezenec, S.; Settanni, L. Ecology of yeasts associated with kernels of several durum wheat genotypes and their role in co-culture with Saccharomyces cerevisiae during dough leavening. Food Microbiol. 2021, 94, 103666. [Google Scholar] [CrossRef] [PubMed]
- Ming, X.; Wang, Y.; Sui, Y. Pretreatment of the antagonistic yeast, Debaryomyces hansenii, with mannitol and sorbitol improves stress tolerance and biocontrol efficacy. Front. Microbiol. 2020, 11, 601. [Google Scholar] [CrossRef]
- Czarnecka, M.; Żarowska, B.; Połomska, X.; Restuccia, C.; Cirvilleri, G. Role of biocontrol yeasts Debaryomyces hansenii and Wickerhamomyces anomalus in plants’ defence mechanisms against Monilinia fructicola in apple fruits. Food Microbiol. 2019, 83, 1–8. [Google Scholar] [CrossRef]
- Huang, C.; Zhang, L.; Johansen, P.G.; Petersen, M.A.; Arneborg, N.; Jespersen, L. Debaryomyces hansenii strains isolated from danish cheese brines act as biocontrol agents to inhibit germination and growth of contaminating molds. Front. Microbiol. 2021, 12, 662785. [Google Scholar] [CrossRef]
- Rivas-Garcia, T.; Murillo-Amador, B.; Reyes-Pérez, J.J.; Chiquito-Contreras, R.G.; Preciado-Rangel, P.; Ávila-Quezada, G.D.; Lara-Capistran, L.; Hernandez-Montiel, L.G. Debaryomyces hansenii, Stenotrophomonas rhizophila, and ulvan as biocontrol agents of fruit rot disease in muskmelon (Cucumis melo L.). Plants 2022, 11, 184. [Google Scholar] [CrossRef]
- Fröhlich-Wyder, M.T.; Arias-Roth, E.; Jakob, E. Cheese yeasts. Yeast 2019, 36, 129–141. [Google Scholar] [CrossRef]
- Koutsoumanis, K.; Allende, A.; Alvarez-Ordóñez, A.; Bolton, D.; Bover-Cid, S.; Chemaly, M.; Davies, R.; De Cesare, A.; Hilbert, F.; Lindqvist, R.; et al. Update of the list of QPS-recommended biological agents intentionally added to food or feed as notified to EFSA 12: Suitability of taxonomic units notified to EFSA until March 2020. EFSA J. 2020, 18, 6174. [Google Scholar] [CrossRef]
- Wachowska, U.; Sulyok, M.; Wiwart, M.; Suchowilska, E.; Giedrojć, W.; Gontarz, D.; Kandler, W.; Krska, R. Secondary metabolites of pathogenic fungi in Triticum durum grain protected with Debaryomyces hansenii in two different locations in Poland. Agronomy 2023, 13, 721. [Google Scholar] [CrossRef]
- Bijlani, S.; Parker, C.; Singh, N.K.; Sierra, M.A.; Foox, J.; Wang, C.C.C.; Mason, C.E.; Venkateswaran, K. Genomic characterization of the titan-like cell producing Naganishia tulchinskyi, the first novel eukaryote isolated from the international space station. J. Fungi 2022, 8, 165. [Google Scholar] [CrossRef]
- Calvo-Garrido, C.; Viñas, I.; Usall, J.; Rodríguez-Romera, M.; Ramos, M.C.; Teixidó, N. Survival of the biological control agent Candida sake CPA-1 on grapes under the influence of abiotic factors. J. Appl. Microbiol. 2014, 117, 800–811. [Google Scholar] [CrossRef]
- IUNG. 2023. Available online: https://iung.pl (accessed on 29 May 2023).
- Kuś, J.; Kopiński, J. The management of soil organic matter in contemporary agriculture. Zagadnienia Doradz. Rol. 2012, 2, 5–27. [Google Scholar]
- Meier, U. Phenological Growth Stages. In Phenology: An Integrative Environmental Science; Schwartz, M.D., Ed.; Tasks for Vegetation Science; Springer: Dordrecht, The Netherlands, 2003; p. 39. [Google Scholar] [CrossRef]
- Pettorelli, N. The Normalized Difference Vegetation Index; Oxford University Press: New York, NY, USA, 2013. [Google Scholar]
- Sultana, S.R.; Ali, A.; Ahmad, A.; Mubeen, M.; Zia-Ul-Haq, M.; Ahmad, S.; Ercisli, S.; Jaafar, H.Z.E. Normalized Difference Vegetation Index as a tool for wheat yield estimation: A case study from Faisalabad, Pakistan. Sci. World J. 2014, 2014, 725326. [Google Scholar] [CrossRef]
- EPPO. European and Mediterranean Plant Protection Organization 1/28 (3). 2019. Available online: https://www.eppo.int/RESOURCES/eppo_standards/pp1_list (accessed on 2 July 2019).
- Gams, W. Cephalosporium-Artige Schimmelpilze (Hyphomycetes); Fischer, G., Ed.; Taylor & Francis, Ltd.: Stuttgart, Germay, 1971. [Google Scholar]
- Leslie, J.F.; Summerell, B.A. The Fusarium Laboratory Manual; Blackwell Publishing Professional: Oxford, UK, 2006; ISBN 9780813819198. [Google Scholar] [CrossRef]
- Özkurt, E.; Hassani, M.A.; Sesiz, U.; Künzel, S.; Dagan, T.; Özkan, H.; Stukenbrock, E.H. Seed-derived microbial colonization of wild emmer and domesticated bread wheat (Triticum dicoccoides and T. aestivum) seedlings shows pronounced differences in overall diversity and composition. mBio 2020, 11, e02637-20. [Google Scholar] [CrossRef]
- Levi, M.; Applebaum, I.; Sherman, C.; Doniger, T.; Steinberger, Y. Soil fungal community of wheat Triticum aestivum rhizosphere at different phenological stages under a rain-fed management. Rhizosphere 2022, 24, 100605. [Google Scholar] [CrossRef]
- Sui, L.; Li, J.; Philp, J.; Yang, K.; Wei, Y.; Li, H.; Li, J.; Li, L.; Ryder, M.; Toh, R.; et al. Trichoderma atroviride seed dressing influenced the fungal community and pathogenic fungi in the wheat rhizosphere. Sci. Rep. 2022, 12, 9677. [Google Scholar] [CrossRef]
- Cardarelli, M.; Woo, S.L.; Rouphael, Y.; Colla, G. Seed treatments with microorganisms can have a biostimulant effect by influencing germination and seedling growth of crops. Plants 2022, 11, 259. [Google Scholar] [CrossRef]
- Kthiri, Z.; Jabeur, M.B.; Chairi, F.; López-Cristoffanini, C.; López-Carbonell, M.; Serret, M.D.; Araus, J.L.; Karmous, C.; Hamada, W. Exploring the potential of Meyerozyma guilliermondii on physiological performances and defense response against Fusarium crown rot on durum wheat. Pathogens 2021, 10, 52. [Google Scholar] [CrossRef] [PubMed]
- Australian Pesticides and Veterinary Medicines Authority. Fluxapyroxad in the Product Systiva Seed Treatment Fungicide. 2015. Available online: https://apvma.gov.au (accessed on 30 July 2021).
- Sawinska, Z.; Krzyzińska, B.; Kazikowski, P.; Głazek, M. Fluxapyroxad-new strategy in reducing leaf disease of spring barley. Fragm. Agron. 2014, 31, 85–91. [Google Scholar]
- Powell, W.A.; Klingeman, W.E.; Ownley, B.H.; Gwinn, K.D. Evidence of endophytic Beauveria bassiana in seed-treated tomato plants acting as a systemic entomopathogen to larval Helicoverpa zea (Lepidoptera: Noctuidae). J. Entomol. Sci. 2009, 44, 391–396. [Google Scholar] [CrossRef]
- Perello, A.E.; Dal Bello, G.M. Suppression of tan spot and plant growth promotion of wheat by synthetic and biological inducersunder field conditions. Ann. Appl. Biol. 2011, 158, 267–274. [Google Scholar] [CrossRef]
- Nandya, S.K.; Srivastavab, R.K. A review on sustainable yeast biotechnological processes and applications. Microbiol. Res. 2018, 20, 83–90. [Google Scholar] [CrossRef]
- Shude, S.P.N.; Mbili, N.C.; Yobo, K.S. Epiphytic yeasts as potential antagonists against Fusarium head blight of wheat (Triticum aestivum L.) caused by Fusarium graminearum sensu strict. J. Saudi Soc. Agric. Sci. 2022, 21, 404–411. [Google Scholar] [CrossRef]
- El-Ghwas, D.E.; Elkhateeb, W.A.; Akram, M.; Daba, G.M. Yeast as biotechnological tool in food industry. J. Pharm. Sci. 2014, 5, 1–6. [Google Scholar] [CrossRef]
- Li, B.; Peng, H.; Tian, S. Attachment capability of antagonistic yeast Rhodotorula glutinis to Botrytis cinerea contributes to biocontrol efficacy. Front. Microbiol. 2016, 7, 601. [Google Scholar] [CrossRef]
- Janisiewicz, W.J.; Petersen, D.L.; Bors, S. Control of storage decay of apples with Sporobolomyces roseus. Plant Dis. 1994, 78, 466. [Google Scholar] [CrossRef]
- Mašínová, T.; Bahnmann, B.D.; Větrovský, T.; Tomšovský, M.; Merunková, K.; Baldrian, P. Drivers of yeast community composition in the litter and soil of a temperate forest. FEMS Microbiol. Ecol. 2017, 93, fiw223. [Google Scholar] [CrossRef]
- Fotedar, R.; Kolecka, A.; Boekhout, T.; Fell, J.W.; Anand, A.; Al Malaki, A.; Zeyara, A.; Al Marri, M. Naganishia qatarensis sp. nov., a novel basidiomycetous yeast species from a hypersaline marine environment in Qatar. Int. J. Syst. Evol. Microbiol. 2018, 68, 2924–2929. [Google Scholar] [CrossRef] [PubMed]
- Turchetti, B.; Selbmann, L.; Blanchette, R.A.; Di Mauro, S.; Marchegiani, E.; Zucconi, L.; Arenz, B.E.; Buzzini, P. Cryptococcus vaughanmartiniae sp. nov. and Cryptococcus onofrii sp. nov.: Two new species isolated from worldwide cold environments. Extremophiles 2015, 19, 149. [Google Scholar] [CrossRef] [PubMed]
- Yuan, X.; Chen, Y.; Qin, W.; Xu, T.; Mao, Y.; Wang, Q.; Chen, K.; Zhu, B. Plant and microbial regulations of soil carbon dynamics under warming in two alpine swamp meadow ecosystems on the Tibetan Plateau. Sci. Total Environ. 2021, 790, 148072. [Google Scholar] [CrossRef]
- Lebeis, S.L.; Paredes, S.H.; Lundberg, D.S.; Breakfield, N.; Gehring, J.; McDonald, M.; Malfatti, S.; Del Rio, G.T.; Jones, C.D.; Tringe, S.G.; et al. Salicylic acid modulates colonization of the root microbiome by specific bacterial taxa. Science 2015, 349, 860–864. [Google Scholar] [CrossRef] [PubMed]
- Yuan, J.; Zhao, J.; Wen, T.; Zhao, M.; Li, R.; Goossens, P.; Huang, Q.; Bai, Y.; Vivanco, J.M.; Kowalchuk, G.A.; et al. Root exudates drive the soil-borne legacy of aboveground pathogen infection. Microbiome 2018, 6, 156. [Google Scholar] [CrossRef]
- Sevillano-Caño, J.; García, M.J.; Córdoba-Galván, C.; Luque-Cruz, C.; Agustí-Brisach, C.; Lucena, C.; Ramos, J.; Pérez-Vicente, R.; Romera, F.J. Exploring the role of Debaryomyces hansenii as biofertilizer in iron-deficient environments to enhance plant nutrition and crop production sustainability. Int. J. Mol. Sci. 2024, 25, 5729. [Google Scholar] [CrossRef]
- Pieterse, C.M.J.; Zamioudis, C.; Berendsen, R.L.; Weller, D.M.; Van Wees, S.C.M.; Bakker, P.A.H.M. Induced systemic resistance by beneficial microbes. Annu. Rev. Phytopathol. 2014, 52, 347–375. [Google Scholar] [CrossRef]
- Ozimek, E.; Hanaka, A. Mortierella Species as the plant growth-promoting fungi present in the agricultural soils. Agriculture 2021, 11, 7. [Google Scholar] [CrossRef]


| Compound/Element | Content (mg/100 g of Soil) | Classification Based on Standard Values for Light Soils * |
|---|---|---|
| P2O5 | 36 | Very high |
| K2O | 41 | Very high |
| Mg | 3.9 | Moderate |
| Mn | 257.2 | Moderate |
| Cu | 2.3 | Moderate |
| Zn | 10.7 | High |
| Fe | 1332 | Low |
| pH in KCl | 6.2 | Slightly acidic |
| Corg (%) ** | 1.26 | Low |
| Humus (%) ** | 2.18 | Moderate |
| Cultivar | Treatment | Number of OTUs | Shannon–Wiener Index | Total Number of Species | Ascomycota | Mortierellomycota | Basidiomycota | Mucoromycota | Chytridiomycota | Olpidiomycota | Zoopagomycota | Aphelidiomycota |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Percentage Total OTUs | ||||||||||||
| Floradur | Control | 121,973 | 1.187 | 172 | 11.54 | 1.93 | 3.4 | 0.03 | 0.012 | 0.0007 | 0 | 0 |
| Biol | 116,422 | 1.309 | 160 | 7.71 | 5.51 | 1.92 | 0.94 | 0.013 | 0 | 0.003 | 0.0003 | |
| Fung | 128,520 | 1.218 | 181 | 8.31 | 4.77 | 0.89 | 3.80 | 0.027 | 0.0006 | 0 | 0.0001 | |
| Durasol | Control | 135,663 | 1.112 | 153 | 8.03 | 3.81 | 1.91 | 4.99 | 0.006 | 0 | 0 | 0 |
| Biol | 103,216 | 1.670 | 159 | 5.25 | 5.17 | 3.39 | 0.39 | 0.032 | 0.0003 | 0 | 0 | |
| Fung | 112,516 | 1.204 | 168 | 6.26 | 4.75 | 4.22 | 0.28 | 0.008 | 0 | 0 | 0.0004 | |
| Total | 718,310 | 270 | 47.12 | 25.94 | 15.73 | 10.44 | 0.098 | 0.002 | 0.003 | 0.0008 | ||
| Species | cv. Floradur | cv. Durasol | ||||
|---|---|---|---|---|---|---|
| Control | Biol | Fung | Control | Biol | Fung | |
| Mortierellomycota | ||||||
| Mortierella alpina | 10,652 | 5415 | 8002 | 12,411 | 19,087 | 12,607 |
| Mortierella elongata | 456 | 11,730 | 6301 | 2804 | 9055 | 11,172 |
| Mortierella exigua | 6 | 387 | 1359 | 0 | 48 | 36 |
| Mortierella fatshederae | 0 | 0 | 5 | 0 | 5 | 4 |
| Mortierella hyalina | 83 | 6744 | 1291 | 933 | 261 | 528 |
| Mortierella sarnyensis | 2409 | 4829 | 4047 | 7290 | 1646 | 2067 |
| Mortierella sclerotiella | 1 | 15 | 59 | 0 | 12 | 0 |
| Mortierella zonata | 0 | 2 | 0 | 7 | 14 | 20 |
| Mortierella spp. | 239 | 10,232 | 13,030 | 3524 | 4745 | 6549 |
| Mucoromycota | ||||||
| Mucor circinelloides | 15 | 482 | 281 | 574 | 75 | 122 |
| Mucor fragilis | 0 | 0 | 11 | 17 | 0 | 46 |
| Mucor hiemalis | 183 | 5771 | 26,744 | 35,222 | 2182 | 1773 |
| Mucor moelleri | 0 | 70 | 16 | 36 | 35 | 15 |
| Mucor mucedo | 0 | 0 | 1 | 0 | 0 | 0 |
| Mucor racemosus | 0 | 2 | 11 | 1 | 0 | 0 |
| Rhizopus arrhizus | 0 | 58 | 11 | 0 | 0 | 0 |
| Trichoderma spp. | ||||||
| Trichoderma hamatum | 172 | 259 | 383 | 618 | 233 | 125 |
| Trichoderma harzianum | 2 | 0 | 0 | 0 | 0 | 1 |
| Trichoderma lanuginosum | 0 | 0 | 4 | 0 | 0 | 0 |
| Trichoderma spirale | 14 | 12 | 18 | 2 | 13 | 2 |
| Trichoderma virens | 0 | 0 | 2 | 0 | 1 | 1 |
| Cultivar | Seed Treatment | Fusarium Crown Rot (FCR) | Fusarium Species Isolated from Stems with Symptoms of FCR (%) | Grain Health | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Severity | Prevalence | Fp | Fg | Fspo | Fa | Fs | Ft | Fc | Fo | FDK | Fusarium spp. (%) | ||
| Durasol | Control | 1.75 a | 80 a | 37.50 | 0 | 0 | 0 | 0 | 0 | 0 | 4.17 | 30.00 | 7.00 |
| Fung | 1.21 b | 70 ab | 37.50 | 0 | 4.17 | 0 | 4.17 | 0 | 0 | 4.17 | 22.50 | 9.00 | |
| Biol | 1.24 b | 85 a | 33.33 | 12.50 | 0 | 0 | 0 | 4.17 | 0 | 0 | 47.50 | 19.00 | |
| Floradur | Control | 1.29 ab | 70 ab | 25.00 | 0 | 8.33 | 4.17 | 4.17 | 0 | 0 | 0 | 17.50 | 16.00 |
| Fung | 1.71 ab | 30 c | 5.56 | 33.33 | 0 | 0 | 0 | 0 | 0 | 0 | 27.50 | 16.54 | |
| Biol | 1.25 b | 45 bc | 4.17 | 4.17 | 0 | 8.33 | 8.33 | 0 | 4.17 | 0 | 37.50 | 12.8 | |
| Mean for cultivar | Durasol | 1.40 | 78.33 A | 36.11 A | 4.17 | 1.39 | 0 | 1.39 | 1.39 | 0 | 2.78 | 33.33 | 11.67 |
| Floradur | 1.38 | 48.33 B | 12.12 B | 10.61 | 3.03 | 4.55 | 4.55 | 0 | 1.52 | 0 | 27.50 | 15.11 | |
| Mean for seed treatment | Control | 1.53 | 75 X | 31.25 | 0 | 4.17 | 2.08 | 2.08 | 2.08 | 0 | 2.08 | 23.75 | 11.50 |
| Fung | 1.38 | 50 Y | 23.81 | 14.29 | 2.38 | 0 | 2.38 | 2.38 | 0 | 2.38 | 25.00 | 12.77 | |
| Biol | 1.24 | 65 XY | 18.75 | 8.33 | 0 | 4.17 | 4.17 | 4.17 | 2.08 | 0 | 42.50 | 15.90 | |
| Cultivar | Seed Treatment | Number of Seedlings per m2 | NDVI of Leaves | Number of Spikes per m² | Plant Height [cm] | Spike Density | Yield Per m2 [g] |
|---|---|---|---|---|---|---|---|
| Durasol | Control | 43.21 b | 0.4895 | 178.2 bc | 57.57 ab | 2.38 b | 242.15 |
| Fung | 50.93 ab | 0.5523 | 273.9 a | 62.31 a | 2.32 c | 232.32 | |
| Biol | 45.37 ab | 0.5433 | 108.9 d | 48.87 b | 2.56 a | 191.75 | |
| Floradur | Control | 49.69 ab | 0.5197 | 138.6 cd | 58.53 ab | 2.52 ab | 229.99 |
| Fung | 54.01 ab | 0.5698 | 148.5 cd | 64.69 a | 2.44 ab | 242.26 | |
| Biol | 58.95 a | 0.5565 | 214.5 b | 61.91 a | 2.50 ab | 201.85 | |
| Mean for cultivar | Durasol | 46.50 B | 0.5319 | 187.0 | 56.25 B | 2.42 | 222.07 |
| Floradur | 54.22 A | 0.5487 | 167.2 | 61.71 A | 2.48 | 224.70 | |
| Mean for seed treatment | Control | 46.45 | 0.5068 | 158.4 Y | 58.05 XY | 2.45 XY | 236.07 |
| Fung | 52.47 | 0.5610 | 211.2 X | 63.51 X | 2.38 Y | 237.29 | |
| Biol | 52.16 | 0.5499 | 161.7 Y | 55.39 Y | 2.53 X | 196.80 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Giedrojć, W.; Wachowska, U. Mycobiome and Pathogenic Fusarium Fungi in the Rhizosphere of Durum Wheat After Seed Dressing with Debaryomyces hansenii. Agriculture 2025, 15, 639. https://doi.org/10.3390/agriculture15060639
Giedrojć W, Wachowska U. Mycobiome and Pathogenic Fusarium Fungi in the Rhizosphere of Durum Wheat After Seed Dressing with Debaryomyces hansenii. Agriculture. 2025; 15(6):639. https://doi.org/10.3390/agriculture15060639
Chicago/Turabian StyleGiedrojć, Weronika, and Urszula Wachowska. 2025. "Mycobiome and Pathogenic Fusarium Fungi in the Rhizosphere of Durum Wheat After Seed Dressing with Debaryomyces hansenii" Agriculture 15, no. 6: 639. https://doi.org/10.3390/agriculture15060639
APA StyleGiedrojć, W., & Wachowska, U. (2025). Mycobiome and Pathogenic Fusarium Fungi in the Rhizosphere of Durum Wheat After Seed Dressing with Debaryomyces hansenii. Agriculture, 15(6), 639. https://doi.org/10.3390/agriculture15060639





