Abstract
Macroalgal wracks can be considered unpleasant for beach users and, consequently, they are usually collected from most touristic beaches and discarded. However, seaweeds are an important source of bioactive lipid compounds, such as phospholipids, glycolipids, and n-3 polyunsaturated fatty acids (n-3 PUFA), displaying multiple health-promoting properties, including antioxidant and antimicrobial activities. The aim of this study is to characterize the lipid composition of twelve marine seaweed species (two green, six red, and four brown species) from macroalgal wracks of Gran Canaria Island, and to evaluate their potential use for several purposes, including human and animal nutrition. Lipid content, lipid classes, and fatty acid profiles of isolated specimens from the macroalgal wracks were determined. Lipid contents ranged between 0.27 and 3.17% of dry weight, with all species showing high phytosterols proportions and balanced omega-6/omega-3 (n-6/n-3) ratios. In addition, Cymopolia barbata, Asparagopsis sp., and Hypnea spinella seem to be an attractive source of both mono- and di-galactosyl-diacylglycerols, while A. stellata, Jania sp., and Lobophora sp. are relatively rich in n-3 LC−PUFA. Finally, both green algae showed the most favorable values for the nutritional indicators of cardiovascular health promotion. Overall, the macroalgal species analyzed could be considered as interesting sources for human and animal nutrition.
1. Introduction
Macroalgae play an important role as components of the marine environment, providing many essential services to the coastal ecosystem engineering [1], including global climate change mitigation [2,3], nutrient supply to the intertidal communities, and coastal protection from marine erosion [1]. Seaweed encompasses the usually benthic, multicellular, and macroscopic algae [4], and is classified into Division Chlorophyta (green macroalgae), Division Rhodophyta (red macroalgae), and Division Ochrophyta (brown macroalgae).
Algae inhabit intertidal, subtidal, and estuarine habitats, being exposed to multiple stressors, such as temperature, light, salinity, wave action, or even predation, competition, and parasitism. As an adaptive response, algae produce a large range of secondary metabolites [4]. Algae are considered an outstanding source of valuable compounds, such as polysaccharides, vitamins, minerals, proteins, peptides and amino acids, pigments (chlorophylls, carotenoids, xanthophylls, or phycobiliproteins), phenolic compounds, and lipids, including the biological active n-3 long-chain (≥C20) polyunsaturated fatty acids (LC−PUFA) [5,6,7,8,9]. Polysaccharides, peptides, carotenoids, and fatty acids (FA) have antiaging, antibiotic, and antioxidant activities [8,10,11], while fucoxanthin, which is mainly present in brown macroalgae and diatoms, has demonstrated anti-obesity and lipolytic properties [12,13]. Among complex lipids, phospholipids and glycolipids, such as mono-galactosyl-diacylglycerol (MGDG), di-galactosyl-diacylglycerol (DGDG), and sulfoquinovosyl-diacylglycerol (SQDG), have been described as anti-inflammatory and anti-thrombotic agents [14,15]. Moreover, phytosterols (PTS) have interesting antioxidative, anti-inflammatory, and antilipidemic properties [16], lowering the total and low-density lipoprotein (LDL) cholesterol levels in humans [17]. Importantly, algae can biosynthesize LC−PUFA, such as eicosapentaenoic acid (EPA; 20:5n-3), docosahexaenoic acid (DHA; 22:6n-3), and arachidonic acid (ARA; 20:4n-6), and their PUFA precursors, alpha-linolenic acid (ALA; 18:3n-3) and linoleic acid (LA; 18:2n-6) [18]. LC−PUFA have been proven to prevent a panel of human pathologies, such as colon and breast cancers, and neurodegenerative or inflammatory disorders [18,19]. As a result, consumers’ demand for algal products is increasing significantly with the rising evidence supporting the nutritional and health benefits of seaweed consumption. Several indices have been established for evaluating the nutritional value of food products based on their FA composition. Among them, the atherogenicity index (AI) and thrombogenicity index (TI), and the ratio between hypocholesterolemic and hypercholesterolemic FAs (hH) are the most commonly used indicators [20].
Additionally, seaweeds are gaining global recognition within the context of transitioning to an environmentally friendly blue bioeconomy. Thus, the use of macroalgae as raw material in food and feed industries, nutraceuticals, natural cosmetics, bio-based materials (biopolymers and bioplastics), and biofuels, or for the extraction of pharmaceutical, biomedical, and biotechnological resources (alginate, agar–agar, and carrageenan) [9,21,22,23,24], is receiving growing attention. Furthermore, algae are acquiring greater acceptance as fertilizers and plant bio-stimulants or as instruments for bioremediation and biomonitoring [25,26,27].
Seaweed farming, practiced in a relatively small number of countries predominantly in Eastern and Southeastern Asia, together with the collection of naturally occurring marine beach-casts and their transformation into a marketable product, are complementary strategies to cope with the global demand for algae. Seaweed aquaculture—which is already the most important source of macroalgae worldwide [28]—offers the opportunity for a more controlled production of target species, resulting in a predictable production outcome. In contrast, the variability in the taxonomic composition and biochemical properties of beach-cast biomass has the potential to offer a broader range of biochemical and bioactive compounds with possible synergistic activity and complementary functions, compared to monospecific biomass. Moreover, the accumulation of large amounts of beach-cast seaweeds on beaches represents an environmental problem, mainly related to the decomposition of the biomass, which can produce anoxic layers and threaten the survival of coastal communities. It can also cause economic damage in touristic areas, where large algal biomass deposition and bad odors are usually unpleasant for beach users [1]. Consequently, beach-cast macroalgae are often removed by local governments, transported, and dumped in landfills, and ultimately wasted, creating the need for developing new strategies to optimize the management and use of this biomass [1,9,21,22,29]. Therefore, the utilization of beach-cast seaweed is especially interesting because it allows the valorization of a waste into a resource, enabling not only to avoid the costs for the generation of biomass ex novo through aquaculture, but also to prevent economical losses related to waste management. In this sense, seaweed wracks could be a potential source of n-3 LC−PUFA in a global scenario of reduced availability of these compounds due to global warming and fish stock reduction [30]. However, the seasonal and geographical variation of the composition and nutritional value of algal biomass, together with the reduced digestibility and bioavailability of many beneficial seaweed compounds to humans or animals [28], slow down the development of this potential industry [25,28,31].
The main objective of this study is to characterize the lipid and FA profiles of 12 seaweed species present in the still understudied macroalgal wracks from Gran Canaria Island coasts. In particular, it is intended to evaluate their potential as sources for both n-3 LC−PUFA and other healthy lipid molecules for human and animal nutrition, encouraging the efficient management of marine resources in coastal communities.
2. Materials and Methods
2.1. Macroalgae Biomass
Over 2000 tons of beach-casts were picked up from Las Canteras beach (28°0824 N, 15°2615 W; Gran Canaria, Spain) as part of the daily routine beach handling carried out by local public administrations during 2018 and 2019. The relative abundance of each collected species is shown in Supplementary Table S1. Samples of at least 20 kg (corresponding to a minimum of 1% of the total biomass harvested in each collection event) were randomly taken and cleaned with seawater to remove sand, encrusted material, and epiphytes, and carefully rinsed with distilled water to remove salts. Subsequently, plastics and other residues were removed manually before the biomass was dried in natural wind in the shade for 24 h. After processing, the average dry vegetable matter was slightly over 10% of the initial fresh biomass weight. Finally, samples were freeze-dried and stored at −20 °C until biochemical analysis in triplicate. After taxonomic identification by morphological observation at light and stereo microscope of both thalli and sections [32,33,34] using samples of wet biomass, twelve seaweeds species were isolated, including two green macroalgae (Chlorophyta, Ulvophyceae): Cymopolia barbata (Linnaeus) J.V. Lamouroux (Order Dasycladales) and Anadyomene stellata (Wulfen) C. Agardh (Order Cladophorales), six species of red macroalgae (Rhodophyta, Florideophyceae): Jania rubens (Linnaeus) J.V. Lamouroux (Order Corallinales), Jania sp. (Order Corallinales), Liagora sp. (Order Nemaliales), Asparagopsis sp. (Order Bonnemaisoniales), Laurencia sp. (Order Ceramiales), and Hypnea spinella (C.Agardh) Kützing (Order Gigartinales), and four species of brown macroalgae (Ochrophyta, Phaeophyceae): Stypocaulon sp. (Order Sphacelariales), Lobophora sp. (Order Dictyotales), Dictyota sp. (Order Dictyotales), and Taonia atomaria (Woodward) J. Agardh (Order Dictyotales) (Supplementary Figure S1).
2.2. Lipid Extraction
The total lipids (TL) of macroalgae was extracted as described by Folch et al. [35], with small modifications [36]. A Virtis rotor homogenizer (Virtishear, Virtis, Gardiner, NY, USA) was used to homogenize 2 mg of lyophilized samples in 10 mL of chloroform/methanol (2:1, v/v). After adding 0.88% (w/v) potassium chloride (KCl), samples were vigorously shaken and centrifuged at 716× g for 5 min at 4 °C to collect the organic solvent, which was subsequently filtered and evaporated under a stream of nitrogen. The lipid content was determined gravimetrically and kept at −20 °C at a concentration of 10 mg mL−1 in chloroform/methanol (2:1, v/v), containing 0.01% (w/v) butylated hydroxytoluene (Sigma-Aldrich Co., St. Louis, MI, USA) as an antioxidant, until further analysis.
2.3. Lipid Classes’ Composition
Lipid classes were analyzed by the method of Olsen and Henderson [37], with minor adaptations following Reis et al. [38]. A high-performance thin-layer chromatography (HPTLC) in a single-dimension, double-development was carried out to isolate polar and neutral lipids. Polar lipids were separated using 1-propanol/chloroform/methyl acetate/methanol/0.25% KCl (5:5:5:2:1.8, v/v), while a mixture of hexane/diethyl ether/acetic acid (20:5:0.5, v/v) was used for the neutral lipids. Polar and neutral lipids were quantified by calibrated densitometry using a dual-wavelength flying spot scanner CAMAG TLC Visualizer (Camag, Muttenz, Switzerland) [38]. Lipid classes were identified by comparison to a mixture of cod roe lipid extract, SQDG, DGDG, and MGDG (Avanti Polar Lipids, Inc., Alabaster, AL, USA).
2.4. Fatty Acid Composition
Acid-catalyzed transmethylation with toluene and 1% sulfuric acid in methanol (v/v) of 1 mg of TL extract was developed to obtain fatty acid methyl esters (FAME) [36]. FAME were purified by thin-layer chromatography with hexane/diethyl ether/acetic acid (90:10:1, v/v), in 20 × 20 cm plates, separated and quantified by means of a TRACE-GC Ultra Gas Chromatograph (Thermo Scientific, Milan, Italy) equipped with an on-column injection, a flame ionization detector (FID), and a fused silica capillary column Supelcowax® 10 (30 m × 0.32 mm ID, df 0.25 μm; Supelco, Inc., Merck KGaA, Darmstadt, Germany) [5]. The carrier gas was He at a flow rate of 1.5 mL min−1. The oven temperature was set to rise from 50 to 150 °C at a rate of 40 °C min−1, then from 150 °C to 200 °C at 2 °C min−1, to 214 °C at 1 °C min−1 and, finally, to 230 °C at 40 °C min−1 (hold time 3 min). Individual FAME were identified by comparing their retention times with those of a mixture of commercial standards (Mix C4-C24 and PUFA No. 3 from menhaden oil (Supelco Inc.)), and to a well-characterized cod roe oil. When necessary, the identity of FAME was confirmed by GC-MS (DSQ II, Thermo Scientific).
2.5. Nutritional Indices
The atherogenicity index (AI), thrombogenicity index (TI) [39], and the ratio between hypocholesterolemic and hypercholesterolemic FAs (hH) [40] were calculated as follows (1)–(3):
AI = [12:0 + (4 × 14:0) + 16:0]/(∑MUFA + ∑n-6 PUFA + ∑n-3 PUFA)
TI = (14:0 + 16:0 + 18:0)/(0.5 × ∑MUFA + 0.5 × ∑n-6 PUFA + 3 × ∑n-3 PUFA + n-3/n-6 ratio)
hH = (18:1n-9 + 18:2n-6 + 20:4n-6 + 18:3n-3 + 20:5n-3 + 22:5n-3 + 22:6n-3)/(14:0 + 16:0)
2.6. Statistical Analysis
Prior to analysis, the assumptions of normality and homoscedasticity were checked, and appropriate variance-stabilizing transformations were applied when needed. Significant differences in TL and nutritional indices among all seaweeds and lipid compositions (lipid classes and FA profile) from red and brown macroalgae were tested by one-way ANOVA, followed by the Tukey HSD post-hoc test, or by the Welch’s t-test, followed by the Dunnett T3 test when homoscedasticity was rejected. Student’s t-test or Mann–Whitney tests for normal or non-normal distribution of data, respectively, were used to evaluate significant differences between green macroalgae.
Similarities and differences regarding lipid composition (lipid class and fatty acid profiles) between seaweeds were assessed by principal component analyses (PCA). Two hierarchical cluster analyses were performed on the factor scores, using the Ward linkage method and the squared Euclidean distances, to categorize seaweeds into similar lipid patterns. All statistical analyses were carried out using the IBM® SPSS Statistics 26.0 software package (IBM Corp., New York, NY, USA) for Windows, setting the statistical significance at p < 0.05.
3. Results
3.1. Total Lipid Content
The TL of the studied seaweeds ranged greatly between species, from 0.27 ± 0.06% dry weight (DW) (J. rubens, Rhodophyta) to 3.17 ± 0.03% DW (T. atomaria, Ochrophyta; Table 1).

Table 1.
Total lipid content (% dry weight) of macroalgae.
3.2. Lipid Classes Profile
All macroalgae species showed a higher total neutral lipids (TNL) fraction (39–64% of TL) than total polar lipids (TPL; 13–27% of TL; Table 2, Table 3 and Table 4). Independently of macroalgae species, pigments (P) were the most prominent liposoluble fraction, varying from 20 to 40% of TL (Table 2, Table 3 and Table 4).

Table 2.
Main lipid class composition (% of total lipids) of green macroalgae.

Table 3.
Main lipid class composition (% of total lipids) of red macroalgae.

Table 4.
Main lipid class composition (% of total lipids) of brown macroalgae.
Among Chlorophyta, C. barbata presented greater TPL proportions than A. stellata; in particular, SQDG + phosphatidylethanolamine (PE; 11.81%) and DGDG (8.13%). On the contrary, PTS (19.53%), triacylglycerols (TAG; 9.93%), and P (32.38%) were higher in A. stellata (Table 2).
As displayed in Table 3, free fatty acids (FFA) were more abundant in Laurencia sp. (31.80%), while SQDG + PE was especially prominent in Asparagopsis sp. (9.06%) and H. spinella (7.92%).
Within brown macroalgae, T. atomaria stood out with the greatest proportion of diacylglycerols (DAG; 14.55%), and Lobophora sp. with that of PTS (21.84%). Finally, phosphatidylcholine (PC) was highest in Stypocaulon sp. (3.64%; Table 4).
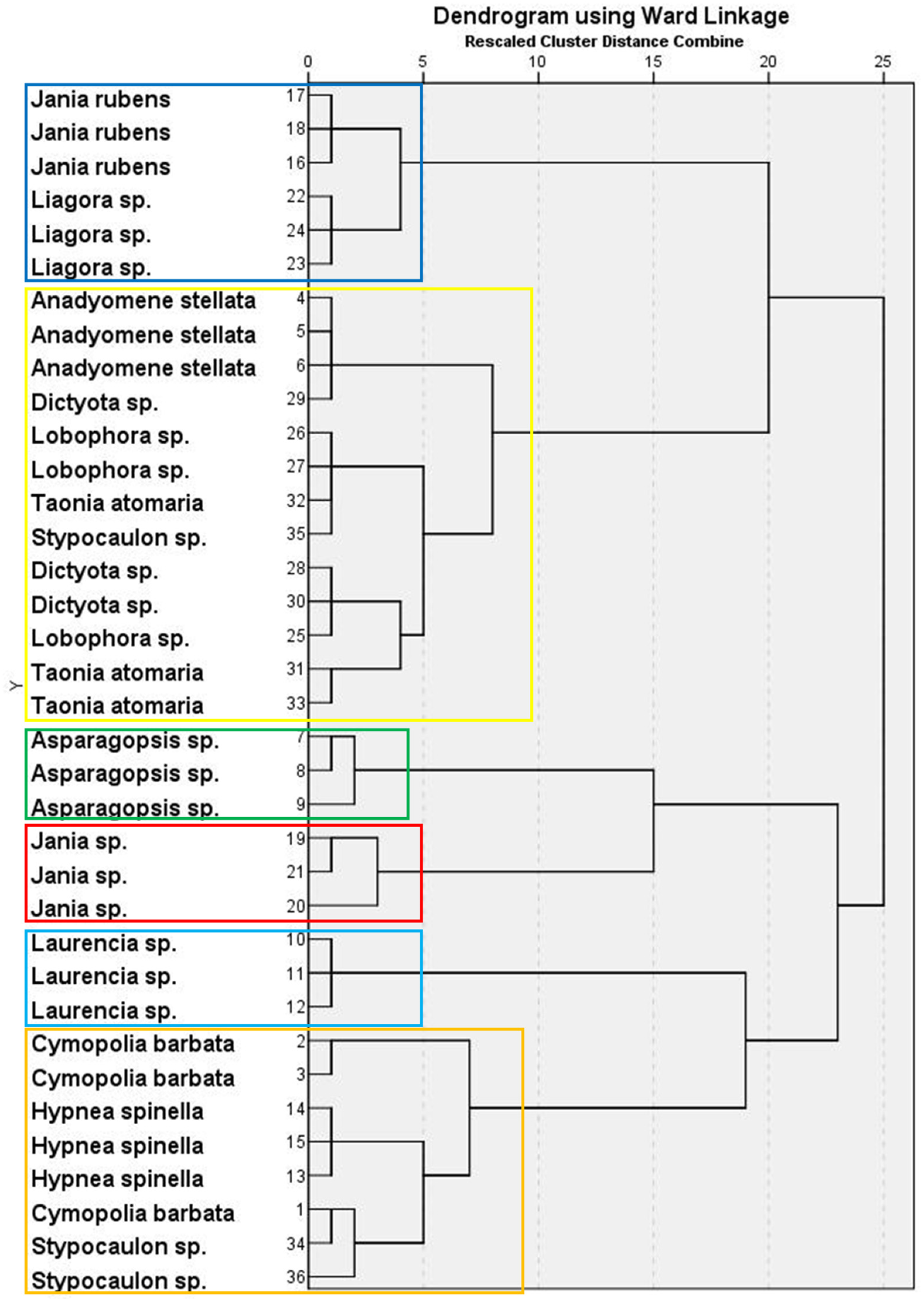
PCA for seaweed lipid classes presented five components with eigenvalues >1, accounting for more than 86% of the total variance. Supplementary Table S2 shows the factor loadings and communalities after applying varimax rotation. The resulting dendrogram exhibited a six-cluster classification (Figure 1). Supplementary Table S3 shows the mean factor scores for each cluster of the dendrogram. Thus, Cluster 1 grouped two of the Rhodophyta species, J. rubens and Liagora sp., both mainly characterized by a high average content of PTS, and low contents of SQDG + PE, DGDG, phosphatidylserine + phosphatidylinositol (PS + PI), DAG, and TAG. A. stellata (green seaweed), together with most species of brown macroalgae studied (Dictyota sp., Lobophora sp., T. atomaria, and one replicate of Stypocaulon sp.), comprised Cluster 2, having high percentages of PS + PI and DAG, but low PC and MGDG. Clusters 3–5 comprised only one red species each: Asparagopsis sp. (Cluster 3) had the highest average proportions of PC and MGDG, high pigment contents, and the lowest FFA, Jania sp. (Cluster 4) was characterized by the highest average percentage of TAG, and Laurencia sp. (Cluster 5) by the highest FFA and SE, and a reduced pigment content. Finally, Cluster 6 comprised species from the three groups, with C. barbata (green), H. spinella (red), and two replicates of Stypocaulon sp. (brown) containing high proportions of SQDG + PE and DGDG, and low percentages of PTS.
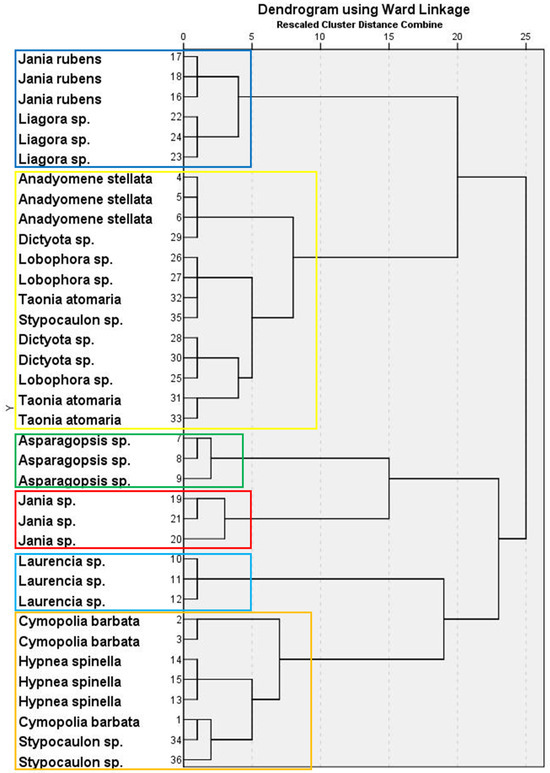
Figure 1.
Hierarchical cluster analysis in a dendrogram format, showing six clusters (each one represented by boxes with different colors) of macroalgal wracks species according to their lipid class composition.
3.3. FA Profile
The FA profiles of green seaweeds greatly varied among species (Table 5). A. stellata contained higher proportions of saturated fatty acids (SFA), mainly the short-chain C14 and C16 compounds, and double n-3 PUFA and n-3 LC−PUFA, while C. barbata was richer in monounsaturated fatty acids (MUFA) and total PUFA content (179.11 ± 13.45 mg 100 g−1 DW). The high amounts of 16:2n-4 and DHA detected in C. barbata were remarkable (7.16 and 2.05% of total FA, respectively). Finally, A. stellata stood out for its abundant EPA (9.26%), ARA (6.90%), and stearidonic acid (SDA, 18:4n-3; 4.28%) contents.

Table 5.
Main fatty acid composition of green macroalgae.
The FA profiles also differed greatly within red macroalgae (Table 6). Thus, C16 SFA was the most prominent FA in all species. Jania sp. had the highest amount of EPA (6.14%), DHA (4.28%), and total n-3 LC−PUFA (10.82%). On the other hand, H. spinella was richer in LA (6.61%) and ARA (5.49%), leading to the highest total PUFA (66 mg 100 g−1 DW), total n-6 PUFA contents (12.90%), and n-6/n-3 ratio (2.22).

Table 6.
Main fatty acid composition of red macroalgae.
Here, the 16:0 represented over 70% of saturates in all brown macroalgae (Table 7). Total MUFA was highest in T. atomaria, mainly due to 16:1n-5, with 18.23 ± 0.30% of total FA. DHA was not detected in either of the Ochrophyta species analyzed. EPA (6.12%) and total n-6 PUFA content, mainly composed of 18:2n-6 (8.89%) and ARA (~6%), were remarkably high in Lobophora sp. Finally, Dictyota sp. also showed relevant amounts of ARA and total PUFA (Table 7).

Table 7.
Main fatty acid composition of brown macroalgae.
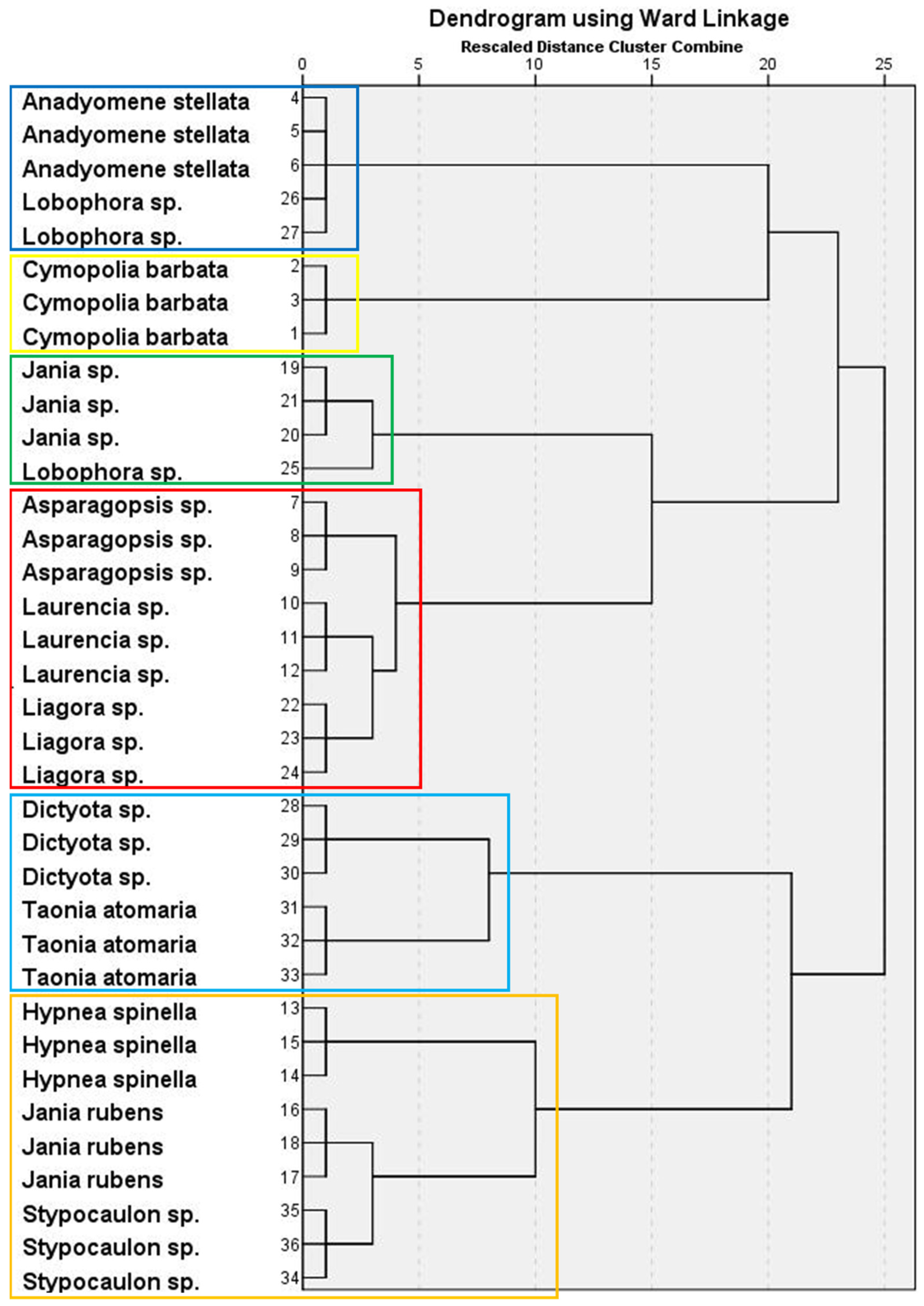
The PCA of macroalgal FA showed that five components had eigenvalues >1, which accounted for more than 89% of the total variance. Factor loadings and communalities are shown in Supplementary Table S4. The hierarchical cluster analysis revealed a dendrogram with six clusters (Figure 2). Factor scores for each cluster are presented in Supplementary Table S5. Thus, Cluster 1 grouped together A. stellata (Chlorophyta) and two replicates of the Ochrophyta Lobophora sp., characterized by the highest contents of 14:0, 18:2n-6, 18:4n-3, ARA, and EPA, and the lowest of 16:0, C16, and C18 MUFA isomers. Cluster 2 comprised one single species, the green seaweed C. barbata, which had the highest average percentages of 16:1n-7 and 18:1n-7, but low contents of 14:0, 18:0, 18:3n-3, 16:1n-5, 18:1n-5, and 18:1n-9 isomers. Jania sp. (Rhodophyta) and one replicate of Lobophora sp. formed cluster 3, characterized by reduced proportions of the 16:1 isomer. Cluster 4 grouped three red macroalgae (Asparagopsis sp., Laurencia sp., and Liagora sp.), having the highest content of 16:0 and low 18:2n-6, ARA, and EPA. Cluster 5 contained the brown seaweeds Dictyota sp. and T. atomaria, which had the highest proportions of 16:1n-5 and 18:1 isomers. Finally, two red macroalgal species (H. spinella and J. rubens) and Stypocaulon sp. (brown seaweed) comprised Cluster 6, characterized by high average contents of 18:0, 18:3n-3, and 16:1 isomers.
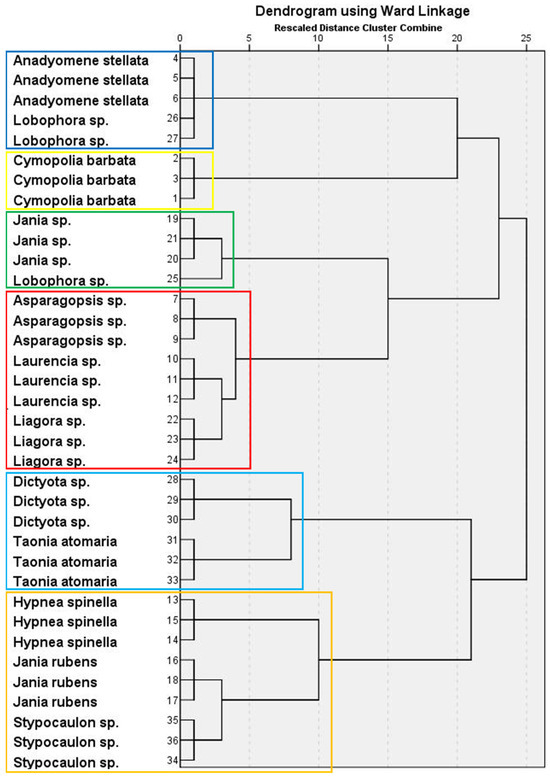
Figure 2.
Hierarchical cluster analysis in a dendrogram format, showing six clusters (each one represented by boxes with different colors) of macroalgal wracks species according to their FA composition.
Lipid classes and FA profiles did not allow grouping Gran Canaria Island beach-cast macroalgae based on their complete lipid profiles, as both parameters showed different patterns. Similarities in their lipid profiles were only evident in the pairs A. stellata–Lobophora sp. and Dictyota sp.–T. atomaria, which were grouped together in both clusters (Figure 1 and Figure 2).
3.4. Nutritional Indices
The lowest and healthiest AI and TI of all analyzed seaweeds were recorded in C. barbata (0.43 ± 0.04 and 0.48 ± 0.05, respectively). The reduced AI in T. atomaria (0.86 ± 0.04) and TI values (<1) in A. stellata, Jania sp., Lobophora sp., Dictyota sp., and T. atomaria were also remarkable. The most health-beneficial hH ratio of around 1 was found in both Chlorophyta species, and in Lobophora sp. and Dictyota sp. (Table 8).

Table 8.
Atherogenicity index (AI), thrombogenicity index (TI), and hypocholesterolemic/hypercholesterolemic fatty acids ratio (hH) of macroalgae.
4. Discussion
The global demand for seafood products, including macroalgae, has been steadily increasing in the last decades, mainly due to the rising awareness of the population about their important nutritional properties [41]. In macroalgae, the lipid levels are generally low (0.2–8% DW) and highly variable intra- and inter-specifically, being influenced by season, environment, or geographic origin, among others [42]. Overall, the set of beach-cast seaweeds analyzed here presented lipid contents in line with values previously reported in the existing literature [5,43]. Among the 12 macroalgae studied, the two Chlorophyta species, together with Dictyota sp. and T. atomaria (Ochrophyta), stood out for their higher lipid proportions (2–3% DW).
Despite the reduced abundance of lipids, macroalgae possess a wide variety of bioactive lipid compounds, exhibiting a broad spectrum of health benefits for humans [41,44]. In particular, antioxidant, antifungal, antiviral, fibrinolytic, and antitumor activities have been described for phospholipids and glycolipids from seaweeds [14,45,46]. The high biological activity of these molecules seems to be closely related to the structural features of the glycosyl and acyl chains [15]. In this sense, the two green macroalgae analyzed here clearly differed in their glycolipid profiles. Interestingly, high SQDG and DGDG levels were found exclusively in C. barbata (Table 2), in agreement with values previously reported for two other Chlorophyta species, Dasycladus vermicularis and Ulva sp. [5]. Also, glycolipid levels were very variable within the red macroalgae of our study [47], emphasizing the highest proportions of SQDG and MGDG in Asparagopsis sp. (9.1 and 5.6% of TL, respectively) and of DGDG in H. spinella (4.9% of TL; Table 3). In contrast, the four Ochrophyta species showed similar contents of glyceroglycolipids (Table 4), preventing them from being considered as a valid taxonomic character within this group of macroalgae [46]. However, the relevant abundance of SQDG in Stypocaulon sp. and T. atomaria (>8% of TL) must be stressed.
PTS are essential components of eukaryotic life, regulating the fluidity and permeability of cellular membranes [48], which cannot be biosynthesized by humans [49]. The Food and Drug Administration (FDA) from the U.S. Department of Health and Human Service (HHS) specifies that the daily dietary intake of PTS may reduce the risk of coronary heart disease (CHD), as their LDL cholesterol-lowering properties have been demonstrated [50]. Thus, the higher the dietary intake of plant sterols, the lower the intestinal absorption of cholesterol, and the lower the serum cholesterol level [49,51]. Remarkably, PTS exceeded 12% of TL in all studied species, particularly A. stellata, J. rubens, Liagora sp., and Asparagopsis sp., with values of 20–23%.
Algae are a natural source of PUFA, especially the physiologically important omega-3 FA. The FA profile is characteristic of each macroalga species and may change depending on factors such as season and/or growth conditions [41,42]. Despite this, green macroalgae usually have similar FA patterns to terrestrial plants, with higher proportions of C16 and C18 FA [49,52,53]. Accordingly, both C. barbata and A. stellata were rich in 16:0, 18:1n-9, LA, and ALA, despite the remarkable differences existing between their FA profiles. In addition, C. barbata had notable proportions of 16:2n-4 (~7%) and 18:3n-6 (~10%). Even though 16:2n-4 has been previously cited in some diatoms [54] and green microalgae [55], its proportion in C. barbata is striking and may suggest a taxonomic character that requires further study. Moreover, gamma linolenic acid (18:3n-6), together with its elongation product dihommo−gamma linolenic acid (20:3n-6), have been pointed out as important nutraceutical compounds for preventing the development of atherosclerosis [56].
Green macroalgae usually lack the physiologically important LC−PUFA, especially DHA [57]. Interestingly, C. barbata presented ~2% of DHA, and A. stellata contained ~7% and ~9% of ARA and EPA, respectively, which are valuable compounds for animal health and well-being. The red macroalgae species in our work presented an FA profile consistent with previous studies, where 16:0, 18:1n-9, ARA, and EPA were the major components [5,58,59]. Thus, H. spinella showed the highest abundance of ARA (~5%), while both DHA and EPA were remarkably high in Jania sp. (~4% and ~6%, respectively; Table 6). In contrast, J. rubens contained low amounts of EPA (~1%) and no DHA, thus strengthening the hypothesis of high inter-specific and intra-generic variations in the FA composition of macroalgae. In accordance with our study, brown algae were characterized by moderate–high proportions of C14 and C16 SFA, 18:1n-9, ARA, EPA, and SDA [41,53,60,61]. It is also noticeable that DHA was not detected in any of the four brown species analyzed, but Lobophora sp. and Dictyota sp. were rich in other n-3 PUFAs, such as EPA (6.12 ± 0.97%) and SDA (4.28 ± 0.16%), respectively. SDA is a metabolic intermediate in the n-3 LC−PUFA biosynthetic pathway with similar beneficial physiological effects as EPA [62], enhancing the presence of n-3 PUFA in tissues [63]. SDA has been previously stated as particularly abundant in Dictyota dichotoma [5]. Finally, T. atomaria and Dictyota sp. showed remarkably high contents of 16:1n-5 (~18% and ~6% of total FA, respectively), a quimiotaxonomic FA from the genera Dictyopteris and Dictyota [64]. The high content of this C16 MUFA in T. atomaria points to its broader presence in genera of brown macroalgae other than those already reported in the literature.
Western diets currently have an n-6/n-3 ratio of 10–20/1, which is clearly far from the balanced 1–2/1 ratio of the Paleolithic period. Consequently, current occidental society intake of n-3 PUFA is generally insufficient, whereas that of n-6 PUFA should be reduced [65,66]. Unbalanced n-6/n-3 ratios strengthen systemic inflammation and overweight [67], and at the same time, compromise the n-3 LC−PUFA biosynthesis [68]. It has been proposed that the optimal dietary n-6/n-3 ratio should vary between 1:1 and 5:1 for good health and well-being [69]. In this sense, all macroalgae covered in our study had beneficial n-6/n-3 ratios and could potentially contribute to reducing the dietary n-6/n-3 ratio in the occidental population. Furthermore, particularly interesting macroalgae, such as Jania sp., could likely be considered as an additional natural source of n-3 LC−PUFA.
The dietary FA composition has implications in determining the risk factors of several pathologies, including cardiovascular diseases [19], namely ischemic heart disease and stroke. In this sense, AI, TI, and hH are analytical indicators commonly used to assess the nutritional quality of ingested lipids based on their FA profile, and their potential effects to human health [20,70]. AI and TI are powerful predictors that characterize the atherogenic and thrombogenic potential of FA, respectively [20]. Thus, AI measures the proportion of pro- and anti-atherogenic FAs, evaluating the proportions between FA that favor the adhesion of lipids to cells of the immunological and circulatory systems and FA that inhibit the aggregation of lipid plaques. TI shows the tendency to form clots in the blood vessels [71]. The hH ratio assesses the effect of hypocholesterolemic and hypercholesterolemic fatty acids on cholesterol metabolism [72], as the hypocholesterolemic FAs diminish the LDL cholesterol, while hypercholesterolemic FAs raise it. Low values of AI and TI and high values of hH are considered cardiovascular health promoters, preventing thrombosis and atherosclerosis [70]. Overall, all macroalgae studied here showed AI, TI, and hH values in line with those reported in existing literature [5,20], yet both green algae displayed the most favorable cardiovascular health indicators (Table 8).
The different clusters obtained in both dendrograms (Figure 1 and Figure 2), based on the lipid class and FA composition of macroalgae, evidenced a unique lipid signature for each species. As a result, it is complicated to determine the best macroalgae species from beach-cast seaweeds for practical use in creating valuable biologically active food and feed additives for human consumption and animal husbandry. In spite of this, more than 1000 tons wet weight/year of macroalgal wracks were collected in 2018 and 2019 from Las Canteras beach, in line with previous studies conducted in the same area between 1994 and 2007 [73]. After eliminating water, sand, and other undesirable materials, the average dry vegetal matter represented nearly 10% of the initial weight; that is, 100 tons per year. Based on the PUFA and EPA + DHA contents (Table 5 and Table 7) of the three most abundant macroalgal species collected during 2018 and 2019: Lobophora sp. (41.8% of total biomass), Dictyota sp. (16.4%), and C. barbata (14.8%; Supplementary Table S1), it is estimated that a total of 55 kg of PUFA and 7.6 kg of n-3 LC−PUFA would be obtained per year through the collection of algal wracks from this specific area. In this context, the use of macroalgae as a dietary supplement in several industries, such as cattle raising, in which they have recently been used at low percentages, might be interesting [74,75]. In fact, similar macroalgal wracks collected from the same area have been used as a feed additive for Ctenopharyngodon idella and Sparus aurata, with encouraging results [21,22]. However, seasonal and geographical variations, as well as potential hydrolytic and oxidative rancidity due to degradation processes, which may affect the lipid composition of macroalgal wracks, should also be considered in further studies, as well as by biotechnological companies that might be interested in exploiting this biological resource. Additionally, it should be noticed that beach-cast seaweeds’ compositions in the Northeast Atlantic have very recently and unexpectedly changed due to the proliferation of the invasive Rugolopteryx okamurae, which has caused massive stranding events [76]. It remains to be clarified whether R. okamurae has a similar lipid profile as Dictyota sp., with which it shares the taxonomic (Dictyotales and Ochrophyta) and morphological features, and whether its use for nutritional purposes can be envisaged [77].
5. Conclusions
In conclusion, most stranded macroalgae species analyzed here were characterized by reduced lipid contents but outstanding proportions of bioactive compounds, such as glycolipids and PTS. In particular, C. barbata, Asparagopsis sp., and H. spinella exhibited the best levels of DGDG and MGDG, while J. rubens, Liagora sp., and Lobophora sp. presented relevant PTS proportions (>20% of TL). In addition, all species presented health-benefiting n-6/n-3 FA ratios and balanced nutritional indices (AI, TI, and hH), and A. stellata, Jania sp., and Lobophora sp. contained attractive levels of the nutritionally important n-3 LC−PUFA. The high content of DHA found in C. barbata was also remarkable, as this FA is unusual in green species, as was the high proportion of the nutraceutical SDA in Dictyota sp. Based on all the above, it is suggested that several species of macroalgae analyzed in the present work could be considered as health-promoting dietary ingredients as well as interesting sources of bioactive molecules that can be potentially used as pharmaceutical compounds. However, further studies testing beach-cast macroalgae are needed to confirm the present findings. This work provided insights for the incorporation of usually discarded beach-cast macroalgae biomass into circular economy strategies.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/jmse12060942/s1. Figure S1: Pictures from macroalgae species used in this study. Table S1: Relative abundance of each macroalgal wrack species collected from Las Canteras beach (Gran Canaria, Spain) between 2018 and 2019. Table S2: Rotated component loadings of the first five principal components. Table S3: Mean factor scores for each cluster of the dendrogram based on lipid class data. Table S4: Rotated component loadings of the first five principal components. Table S5: Mean factor scores for each cluster of the dendrogram based on fatty acid data.
Author Contributions
Conceptualization, C.R.; methodology, C.d.M.G. and N.G.A.; formal analysis, A.G., C.d.M.G., and B.A.-J.; resources, M.V.; writing—original draft preparation, A.G.; writing—review and editing, C.R., J.A.P., M.M., and M.V.; supervision, C.R., J.A.P., and N.G.A.; project administration, C.R.; funding acquisition, C.R. and M.V. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by the European Interreg Programme 2014–2020 (MACBIOBLUE; MAC/1.1b/086), the Recovery and Resilience Programme (Next Generation EU), and Gobierno de Canarias (Consejería de Economía, Conocimiento y Empleo; THINKINAZUL project). A. Galindo is sponsored by Cajasiete, Ministerio de Ciencia, Innovación y Universidades (PhD contract) and the Catalina Ruiz Programme, funded by Consejería de Economía, Conocimiento y Empleo, and FSE.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
The authors confirm that the data supporting the findings of this study are available within the article and its Supplementary Materials.
Acknowledgments
Covadonga Rodríguez is a member of Instituto de Tecnologías Biomédicas de Canarias (ITB).
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Harb, T.B.; Chow, F. An Overview of Beach-Cast Seaweeds: Potential and Opportunities for the Valorization of Underused Waste Biomass. Algal Res. 2022, 62, 102643. [Google Scholar] [CrossRef]
- Duarte, C.M.; Wu, J.; Xiao, X.; Bruhn, A.; Krause-Jensen, D. Can Seaweed Farming Play a Role in Climate Change Mitigation and Adaptation? Front. Mar. Sci. 2017, 4, 100. [Google Scholar] [CrossRef]
- Duarte, C.M.; Agusti, S.; Barbier, E.; Britten, G.L.; Castilla, J.C.; Gattuso, J.P.; Fulweiler, R.W.; Hughes, T.P.; Knowlton, N.; Lovelock, C.E.; et al. Rebuilding Marine Life. Nature 2020, 580, 39–51. [Google Scholar] [CrossRef]
- Wan, A.H.L.; Davies, S.J.; Soler-Vila, A.; Fitzgerald, R.; Johnson, M.P. Macroalgae as a Sustainable Aquafeed Ingredient. Rev. Aquac. 2019, 11, 458–492. [Google Scholar] [CrossRef]
- Galindo, A.; Reis, D.B.; Rodríguez, I.; Pérez, J.A.; Abdul-Jalbar, B.; Zárate, R.; Nunes, N.; Pinheiro De Carvalho, M.A.A.; Acosta, N.G.; Rodríguez, C. Lipid Characterization of 14 Macroalgal Species from Madeira Archipelago: Implications for Animal and Human Nutrition. Bot. Mar. 2022, 65, 51–67. [Google Scholar] [CrossRef]
- Gómez-Zorita, S.; González-Arceo, M.; Trepiana, J.; Eseberri, I.; Fernández-Quintela, A.; Milton-Laskibar, I.; Aguirre, L.; González, M.; Portillo, M.P. Anti-Obesity Effects of Macroalgae. Nutrients 2020, 12, 2378. [Google Scholar] [CrossRef]
- Kothari, R.; Singh, H.M.; Azam, R.; Goria, K.; Bharti, A.; Singh, A.; Bajar, S.; Pathak, A.; Pandey, A.K.; Tyagi, V.V. Potential Avenue of Genetic Engineered Algal Derived Bioactive Compounds: Influencing Parameters, Challenges and Future Prospects. Phytochem. Rev. 2023, 22, 935–968. [Google Scholar] [CrossRef]
- Lähteenmäki-Uutela, A.; Rahikainen, M.; Camarena-Gómez, M.T.; Piiparinen, J.; Spilling, K.; Yang, B. European Union Legislation on Macroalgae Products. Aquac. Int. 2021, 29, 487–509. [Google Scholar] [CrossRef]
- Zárate, R.; Portillo, E.; Teixidó, S.; Pinheiro De Carvalho, M.A.A.; Nunes, N.; Ferraz, S.; Seca, A.M.L.; Rosa, G.P.; Barreto, M.C. Pharmacological and Cosmeceutical Potential of Seaweed Beach-Casts of Macaronesia. Appl. Sci. 2020, 10, 5831. [Google Scholar] [CrossRef]
- Lopes, D.; Rey, F.; Leal, M.C.; Lillebø, A.I.; Calado, R.; Domingues, M.R. Bioactivities of Lipid Extracts and Complex Lipids from Seaweeds: Current Knowledge and Future Prospects. Mar. Drugs 2021, 19, 686. [Google Scholar] [CrossRef] [PubMed]
- Alves, E.; Dias, M.; Lopes, D.; Almeida, A.; Domingues, M.D.R.; Rey, F. Antimicrobial Lipids from Plants and Marine Organisms: An Overview of the Current State-of-the-Art and Future Prospects. Antibiotics 2020, 9, 441. [Google Scholar] [CrossRef]
- Fernandes, V.; Mamatha, B.S. Fucoxanthin, a Functional Food Ingredient: Challenges in Bioavailability. Curr. Nutr. Rep. 2023, 12, 567–580. [Google Scholar] [CrossRef] [PubMed]
- Muradian, K.; Vaiserman, A.; Min, K.-J.; Fraifeld, V.E. Fucoxanthin and Lipid Metabolism: A Minireview. Nutr. Metab. Cardiovasc. Dis. 2015, 25, 891–897. [Google Scholar] [CrossRef] [PubMed]
- Guo, S.S.; Wang, Z.G. Glyceroglycolipids in Marine Algae: A Review of Their Pharmacological Activity. Front. Pharmacol. 2022, 13, 1008797. [Google Scholar] [CrossRef] [PubMed]
- Shiels, K.; Tsoupras, A.; Lordan, R.; Zabetakis, I.; Murray, P.; Kumar Saha, S. Anti-Inflammatory and Antithrombotic Properties of Polar Lipid Extracts, Rich in Unsaturated Fatty Acids, from the Irish Marine Cyanobacterium Spirulina subsalsa. J. Funct. Foods 2022, 94, 105124. [Google Scholar] [CrossRef]
- Hannan, M.A.; Sohag, A.A.M.; Dash, R.; Haque, M.N.; Mohibbullah, M.; Oktaviani, D.F.; Hossain, M.T.; Choi, H.J.; Moon, I.S. Phytosterols of Marine Algae: Insights into the Potential Health Benefits and Molecular Pharmacology. Phytomedicine 2020, 69, 153201. [Google Scholar] [CrossRef] [PubMed]
- Ibañez, E.; Cifuentes, A. Benefits of Using Algae as Natural Sources of Functional Ingredients. J. Sci. Food Agric. 2013, 93, 703–709. [Google Scholar] [CrossRef] [PubMed]
- Harwood, J.L. Algae: Critical Sources of Very Long-Chain Polyunsaturated Fatty Acids. Biomolecules 2019, 9, 708. [Google Scholar] [CrossRef] [PubMed]
- Zárate, R.; el Jaber-Vazdekis, N.; Tejera, N.; Pérez, J.A.; Rodríguez, C. Significance of Long Chain Polyunsaturated Fatty Acids in Human Health. Clin. Transl. Med. 2017, 6, 25. [Google Scholar] [CrossRef]
- Chen, J.; Liu, H. Nutritional Indices for Assessing Fatty Acids: A Mini-Review. Int. J. Mol. Sci. 2020, 21, 5695. [Google Scholar] [CrossRef]
- Galindo, A.; Rodríguez, C.; Reis, D.B.; Marrero, M.; Acosta, N.G.; Barreto, M.C.; Jiménez, I.A.; de Urioste, J.; Venuleo, M.; Pérez, J.A. Valorization of Seaweed Wracks: Inclusion as Additive in Diets for Grass Carp (Ctenopharyngodon idella). Aquac. Nutr. 2022, 2022, 6992682. [Google Scholar] [CrossRef] [PubMed]
- Galindo, A.; Pérez, J.A.; Martín, V.; Acosta, N.G.; Reis, D.B.; Jiménez, I.A.; Rosa, G.; Venuleo, M.; Marrero, M.; Rodríguez, C. Seaweed Wracks as Potential Dietary Ingredients for Gilthead Seabream (Sparus aurata). Aquac. Rep. 2023, 31, 101673. [Google Scholar] [CrossRef]
- Kumari, P.; Bijo, A.J.; Mantri, V.A.; Reddy, C.R.K.; Jha, B. Fatty Acid Profiling of Tropical Marine Macroalgae: An Analysis from Chemotaxonomic and Nutritional Perspectives. Phytochemistry 2013, 86, 44–56. [Google Scholar] [CrossRef] [PubMed]
- Tabassum, M.R.; Xia, A.; Murphy, J.D. Potential of Seaweed as a Feedstock for Renewable Gaseous Fuel Production in Ireland. Renew. Sustain. Energy Rev. 2017, 68, 136–146. [Google Scholar] [CrossRef]
- Araújo, R.; Vázquez Calderón, F.; Sánchez López, J.; Azevedo, I.C.; Bruhn, A.; Fluch, S.; Garcia Tasende, M.; Ghaderiardakani, F.; Ilmjärv, T.; Laurans, M.; et al. Current Status of the Algae Production Industry in Europe: An Emerging Sector of the Blue Bioeconomy. Front. Mar. Sci. 2021, 7, 626389. [Google Scholar] [CrossRef]
- Khan, W.; Rayirath, U.P.; Subramanian, S.; Jithesh, M.N.; Rayorath, P.; Hodges, D.M.; Critchley, A.T.; Craigie, J.S.; Norrie, J.; Prithiviraj, B. Seaweed Extracts as Biostimulants of Plant Growth and Development. J. Plant Growth Regul. 2009, 28, 386–399. [Google Scholar] [CrossRef]
- Marinho-Soriano, E.; Azevedo, C.A.A.; Trigueiro, T.G.; Pereira, D.C.; Carneiro, M.A.A.; Camara, M.R. Bioremediation of Aquaculture Wastewater Using Macroalgae and Artemia. Int. Biodeterior. Biodegrad. 2011, 65, 253–257. [Google Scholar] [CrossRef]
- Buschmann, A.H.; Camus, C.; Infante, J.; Neori, A.; Israel, Á.; Hernández-González, M.C.; Pereda, S.V.; Gomez-Pinchetti, J.L.; Golberg, A.; Tadmor-Shalev, N.; et al. Seaweed Production: Overview of the Global State of Exploitation, Farming and Emerging Research Activity. Eur. J. Phycol. 2017, 52, 391–406. [Google Scholar] [CrossRef]
- Nunes, N.; Valente, S.; Ferraz, S.; Barreto, M.C.; De Carvalho, M.A.A.P. Biochemical Study of Attached Macroalgae from the Madeira Archipelago and Beach-Cast Macroalgae from the Canary Islands: Multivariate Analysis to Determine Bioresource Potential. Bot. Mar. 2020, 63, 283–298. [Google Scholar] [CrossRef]
- Colombo, S.M.; Rodgers, T.F.M.; Diamond, M.L.; Bazinet, R.P.; Arts, M.T. Projected Declines in Global DHA Availability for Human Consumption as a Result of Global Warming. Ambio 2020, 49, 865–880. [Google Scholar] [CrossRef]
- FAO. The Global Status of Seaweed Production, Trade and Utilization; Globefish Research Programme; FAO: Rome, Italy, 2018; Volume 124. [Google Scholar]
- Braune, W. Seaweeds: A Colour Guide to Common Benthic Green, Brown and Red Algae of the World’s Oceans; Koeltz Scientific Books: Oberreifenberg, Germany, 2011; ISBN 978-3-906166-90-2. [Google Scholar]
- Afonso-Carrillo, J.; Marta, S. Algas, Hongos y Fanérogamas Marinas de Las Islas Canarias. Clave Analítica; Universidad de La Laguna: San Cristóbal de La Laguna, Spain, 1999. [Google Scholar]
- Haroun, R.J.; Gil-Rodrìguez, M.C.; De Castro, J.D.; Van Reine, W.F.P. A Checklist of the Marine Plants from the Canary Islands (Central Eastern Atlantic Ocean). Bot. Mar. 2002, 45, 139–169. [Google Scholar] [CrossRef]
- Folch, J.; Lees, M.; Sloane Stanley, G.H. A Simple Method for the Isolation and Purification of Total Lipides from Animal Tissues. J. Biol. Chem. 1957, 226, 497–509. [Google Scholar] [CrossRef] [PubMed]
- Christie, W.W.; Han, X. Lipid Analysis: Isolation, Separation, Identification and Lipidomic Analysis; Oily Press: Bridgwater, UK, 2010; ISBN 9780955251245. [Google Scholar]
- Olsen, R.E.; Henderson, R.J. The Rapid Analysis of Neutral and Polar Marine Lipids Using Double-Development HPTLC and Scanning Densitometry. J. Exp. Mar. Biol. Ecol. 1989, 129, 189–197. [Google Scholar] [CrossRef]
- Reis, D.B.; Acosta, N.G.; Almansa, E.; Garrido, D.; Andrade, J.P.; Sykes, A.V.; Rodríguez, C. Effect of Artemia Inherent Fatty Acid Metabolism on the Bioavailability of Essential Fatty Acids for Octopus Vulgaris Paralarvae Development. Aquaculture 2019, 500, 264–271. [Google Scholar] [CrossRef]
- Ulbricht, T.L.V.; Southgate, D.A.T. Coronary Heart Disease: Seven Dietary Factors. Lancet 1991, 338, 985–992. [Google Scholar] [CrossRef] [PubMed]
- Santos-Silva, J.; Bessa, R.J.B.; Santos-Silva, F. Effect of Genotype, Feeding System and Slaughter Weight on the Quality of Light Lambs: II. Fatty Acid Composition of Meat. Livest. Prod. Sci. 2002, 77, 187–194. [Google Scholar] [CrossRef]
- Lopes, D.; Melo, T.; Rey, F.; Meneses, J.; Monteiro, F.L.; Helguero, L.A.; Abreu, M.H.; Lillebø, A.I.; Calado, R.; Domingues, M.R. Valuing Bioactive Lipids from Green, Red and Brown Macroalgae from Aquaculture, to Foster Functionality and Biotechnological Applications. Molecules 2020, 25, 3883. [Google Scholar] [CrossRef]
- Pereira, H.; Barreira, L.; Figueiredo, F.; Custódio, L.; Vizetto-Duarte, C.; Polo, C.; Rešek, E.; Aschwin, E.; Varela, J. Polyunsaturated Fatty Acids of Marine Macroalgae: Potential for Nutritional and Pharmaceutical Applications. Mar. Drugs 2012, 10, 1920–1935. [Google Scholar] [CrossRef] [PubMed]
- Salehi, B.; Sharifi-Rad, J.; Seca, A.M.L.; Pinto, D.C.G.A.; Michalak, I.; Trincone, A.; Mishra, A.P.; Nigam, M.; Zam, W.; Martins, N. Current Trends on Seaweeds: Looking at Chemical Composition, Phytopharmacology, and Cosmetic Applications. Molecules 2019, 24, 4182. [Google Scholar] [CrossRef] [PubMed]
- Domingues, R.M.; Calado, R. Lipids of Marine Algae—Biomolecules with High Nutritional Value and Important Bioactive Properties. Biomolecules 2022, 12, 134. [Google Scholar] [CrossRef]
- Gerasimenko, N.; Logvinov, S. Seasonal Composition of Lipids, Fatty Acids Pigments in the Brown Alga Sargassum Pallidum: The Potential for Health. Open J. Mar. Sci. 2016, 06, 498–523. [Google Scholar] [CrossRef]
- Wielgosz-Collin, G.; Kendel, M.; Couzinet-Mossion, A. Lipids, Fatty Acids, Glycolipids, and Phospholipids. In Seaweed in Health and Disease Prevention; Fleurence, J., Levine, I., Eds.; Academic Press: San Diego, CA, USA, 2016; pp. 185–221. ISBN 9780128027936. [Google Scholar]
- Khotimchenko, S.V. Distribution of Glyceroglycolipids in Marine Algae and Grasses. Chem. Nat. Compd. 2002, 38, 223–229. [Google Scholar] [CrossRef]
- Piironen, V.; Lindsay, D.G.; Miettinen, T.A.; Toivo, J.; Lampi, A.M. Plant Sterols: Biosynthesis, Biological Function and Their Importance to Human Nutrition. J. Sci. Food Agric. 2000, 80, 939–966. [Google Scholar] [CrossRef]
- Kendel, M.; Wielgosz-Collin, G.; Bertrand, S.; Roussakis, C.; Bourgougnon, N.B.; Bedoux, G. Lipid Composition, Fatty Acids and Sterols in the Seaweeds Ulva Armoricana, and Solieria Chordalis from Brittany (France): An Analysis from Nutritional, Chemotaxonomic, and Antiproliferative Activity Perspectives. Mar. Drugs 2015, 13, 5606–5628. [Google Scholar] [CrossRef] [PubMed]
- Elke, T.; Mensink, R.P. Simultaneous Intake of β-Glucan and Plant Stanol Esters Affects Lipid Metabolism in Slightly Hypercholesterolemic Subjects. J. Nutr. 2007, 137, 583–588. [Google Scholar] [CrossRef] [PubMed]
- Patch, C.S.; Tapsell, L.C.; Williams, P.G.; Gordon, M. Plant Sterols as Dietary Adjuvants in the Reduction of Cardiovascular Risk: Theory and Evidence. Vasc. Health Risk Manag. 2006, 2, 157–162. [Google Scholar] [CrossRef]
- Nakamura, Y.; Li-Beisson, Y. Lipids in Plant and Algae Development; Springer International Publishing: Berlin, Germany, 2016; ISBN 978-3-319-25979-6. [Google Scholar]
- Santos, J.P.; Guihéneuf, F.; Fleming, G.; Chow, F.; Stengel, D.B. Temporal Stability in Lipid Classes and Fatty Acid Profiles of Three Seaweed Species from the North-Eastern Coast of Brazil. Algal Res. 2019, 41, 101572. [Google Scholar] [CrossRef]
- De Carvalho, C.C.C.R.; Caramujo, M.J. The Various Roles of Fatty Acids. Molecules 2018, 23, 2583. [Google Scholar] [CrossRef]
- Dolganyuk, V.; Andreeva, A.; Budenkova, E.; Sukhikh, S.; Babich, O.; Ivanova, S.; Prosekov, A.; Ulrikh, E. Study of Morphological Features and Determination of the Fatty Acid Composition of the Microalgae Lipid Complex. Biomolecules 2020, 10, 1571. [Google Scholar] [CrossRef]
- Moss, J.W.E.; Ramji, D.P. Nutraceutical Therapies for Atherosclerosis. Nat. Rev. Cardiol. 2016, 13, 513–532. [Google Scholar] [CrossRef]
- McCauley, J.I.; Meyer, B.J.; Winberg, P.C.; Skropeta, D. Parameters Affecting the Analytical Profile of Fatty Acids in the Macroalgal Genus Ulva. Food Chem. 2016, 209, 332–340. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-Machado, D.I.; López-Cervantes, J.; López-Hernández, J.; Paseiro-Losada, P. Fatty Acids, Total Lipid, Protein and Ash Contents of Processed Edible Seaweeds. Food Chem. 2004, 85, 439–444. [Google Scholar] [CrossRef]
- Schmid, M.; Kraft, L.G.K.; van der Loos, L.M.; Kraft, G.T.; Virtue, P.; Nichols, P.D.; Hurd, C.L. Southern Australian Seaweeds: A Promising Resource for Omega-3 Fatty Acids. Food Chem. 2018, 265, 70–77. [Google Scholar] [CrossRef] [PubMed]
- Dellatorre, F.G.; Avaro, M.G.; Commendatore, M.G.; Arce, L.; Díaz de Vivar, M.E. The Macroalgal Ensemble of Golfo Nuevo (Patagonia, Argentina) as a Potential Source of Valuable Fatty Acids for Nutritional and Nutraceutical Purposes. Algal Res. 2020, 45, 101726. [Google Scholar] [CrossRef]
- Kumari, P.; Kumar, M.; Gupta, V.; Reddy, C.R.K.; Jha, B. Tropical Marine Macroalgae as Potential Sources of Nutritionally Important PUFAs. Food Chem. 2010, 120, 749–757. [Google Scholar] [CrossRef]
- Whelan, J. Dietary Stearidonic Acid Is a Long Chain (n-3) Polyunsaturated Fatty Acid with Potential Health Benefits. J. Nutr. 2009, 139, 5–10. [Google Scholar] [CrossRef] [PubMed]
- Guil-Guerrero, J.L. Stearidonic Acid (18:4n-3): Metabolism, Nutritional Importance, Medical Uses and Natural Sources. Eur. J. Lipid Sci. Technol. 2007, 109, 1226–1236. [Google Scholar] [CrossRef]
- Khotimchenko, S. V Uncommon 16:1 (n-5) Acid from Dictyota Dichotoma and Fatty Acids of Some Brown Algae of Dictyotaceae. Phytochemistry 1995, 38, 1411–1415. [Google Scholar] [CrossRef]
- Simopoulos, A.P. An Increase in the Omega-6/Omega-3 Fatty Acid Ratio Increases the Risk for Obesity. Nutrients 2016, 8, 128. [Google Scholar] [CrossRef]
- Simopoulos, A.P. The Importance of the Ratio of Omega-6/Omega-3 Essential Fatty Acids. Biomed. Pharmacother. 2002, 56, 375–379. [Google Scholar] [CrossRef]
- Mariamenatu, A.H.; Abdu, E.M. Overconsumption of Omega-6 Polyunsaturated Fatty Acids (PUFAs) versus Deficiency of Omega-3 PUFAs in Modern-Day Diets: The Disturbing Factor for Their “Balanced Antagonistic Metabolic Functions” in the Human Body. J. Lipids 2021, 2021, 8848161. [Google Scholar] [CrossRef] [PubMed]
- Smink, W.; Gerrits, W.J.J.; Gloaguen, M.; Ruiter, A.; Van Baal, J. Linoleic and α-Linolenic Acid as Precursor and Inhibitor for the Synthesis of Long-Chain Polyunsaturated Fatty Acids in Liver and Brain of Growing Pigs. Animal 2012, 6, 262–270. [Google Scholar] [CrossRef] [PubMed]
- González-Becerra, K.; Barron-Cabrera, E.; Muñoz-Valle, J.F.; Torres-Castillo, N.; Rivera-Valdes, J.J.; Rodriguez-Echevarria, R.; Martinez-Lopez, E. A Balanced Dietary Ratio of N-6:N-3 Polyunsaturated Fatty Acids Exerts an Effect on Total Fatty Acid Profile in RBCs and Inflammatory Markers in Subjects with Obesity. Healthcare 2023, 11, 2333. [Google Scholar] [CrossRef] [PubMed]
- Szpunar-Krok, E.; Wondołowska-Grabowska, A. Quality Evaluation Indices for Soybean Oil in Relation to Cultivar, Application of N Fertiliser and Seed Inoculation with Bradyrhizobium Japonicum. Foods 2022, 11, 762. [Google Scholar] [CrossRef] [PubMed]
- Garaffo, M.A.; Vassallo-Agius, R.; Nengas, Y.; Lembo, E.; Rando, R.; Maisano, R.; Dugo, G.; Giuffrida, D. Fatty Acids Profile, Atherogenic (IA) and Thrombogenic (IT) Health Lipid Indices, of Raw Roe of Blue Fin Tuna (Thunnus thynnus L.) and Their Salted Product “Bottarga”. Food Nutr. Sci. 2011, 2, 736–743. [Google Scholar] [CrossRef]
- Dietschy, J.M. Symposium: Evolution of Ideas about the Nutritional Value of Dietary Fat Dietary Fatty Acids and the Regulation of Plasma Low Density Lipoprotein Cholesterol Concentrations 1,2. J. Nutr. 1998, 128, 444–448. [Google Scholar] [CrossRef] [PubMed]
- Portillo, E. Arribazones de Algas y Plantas Marinas En Gran Canaria: Características, Gestión y Posibles Usos; Instituto Tecnológico de Canarias: Santa Lucía de Tirajana, Spain, 2008; ISBN 978-84-691-5105-I. [Google Scholar]
- Kulshreshtha, G.; Hincke, M.T.; Prithiviraj, B.; Critchley, A. A Review of the Varied Uses of Macroalgae as Dietary Supplements in Selected Poultry with Special Reference to Laying Hen and Broiler Chickens. J. Mar. Sci. Eng. 2020, 8, 536. [Google Scholar] [CrossRef]
- González-Meza, G.M.; Elizondo-Luevano, J.H.; Cuellar-Bermudez, S.P.; Sosa-Hernández, J.E.; Iqbal, H.M.N.; Melchor-Martínez, E.M.; Parra-Saldívar, R. New Perspective for Macroalgae-Based Animal Feeding in the Context of Challenging Sustainable Food Production. Plants 2023, 12, 3609. [Google Scholar] [CrossRef] [PubMed]
- Faria, J.; Cl Prestes, A.; Moreu, I.; Martins, G.M.; Neto, A.I.; Cacabelos, E. Arrival and Proliferation of the Invasive Seaweed Rugulopteryx Okamurae in NE Atlantic Islands. Bot. Mar. 2022, 61, 45–50. [Google Scholar] [CrossRef]
- Barcellos, L.; Pham, C.K.; Menezes, G.; Bettencourt, R.; Rocha, N.; Carvalho, M.; Felgueiras, H.P. A Concise Review on the Potential Applications of Rugulopteryx Okamurae Macroalgae. Mar. Drugs 2023, 21, 40. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).