The Evaluation of the Trueness of Dental Mastercasts Obtained through Different 3D Printing Technologies
Abstract
:1. Introduction
2. Materials and Methods
2.1. Test Object Creation
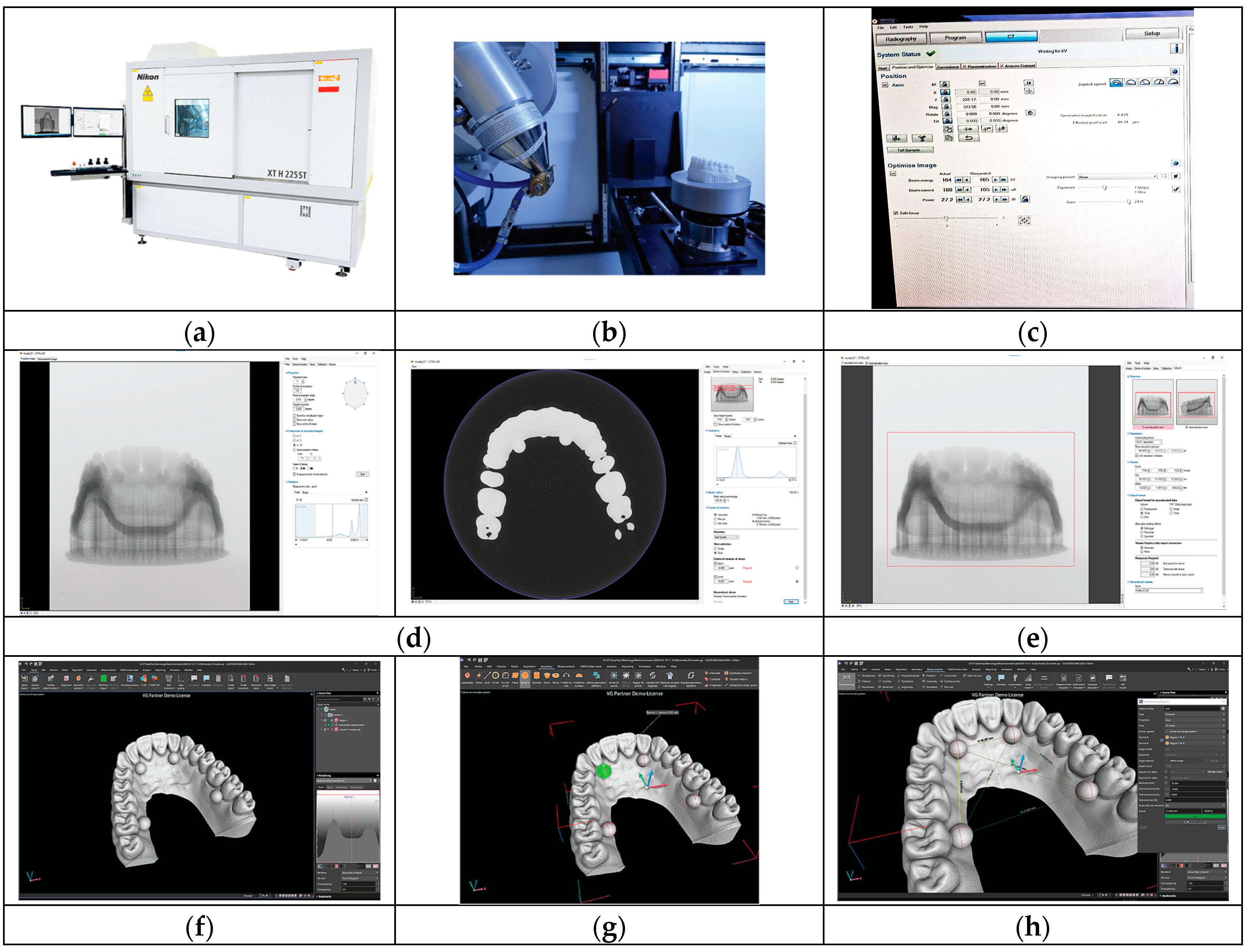
2.2. Three-Dimensional Printing of the Test Object
2.3. Analysis
3. Results
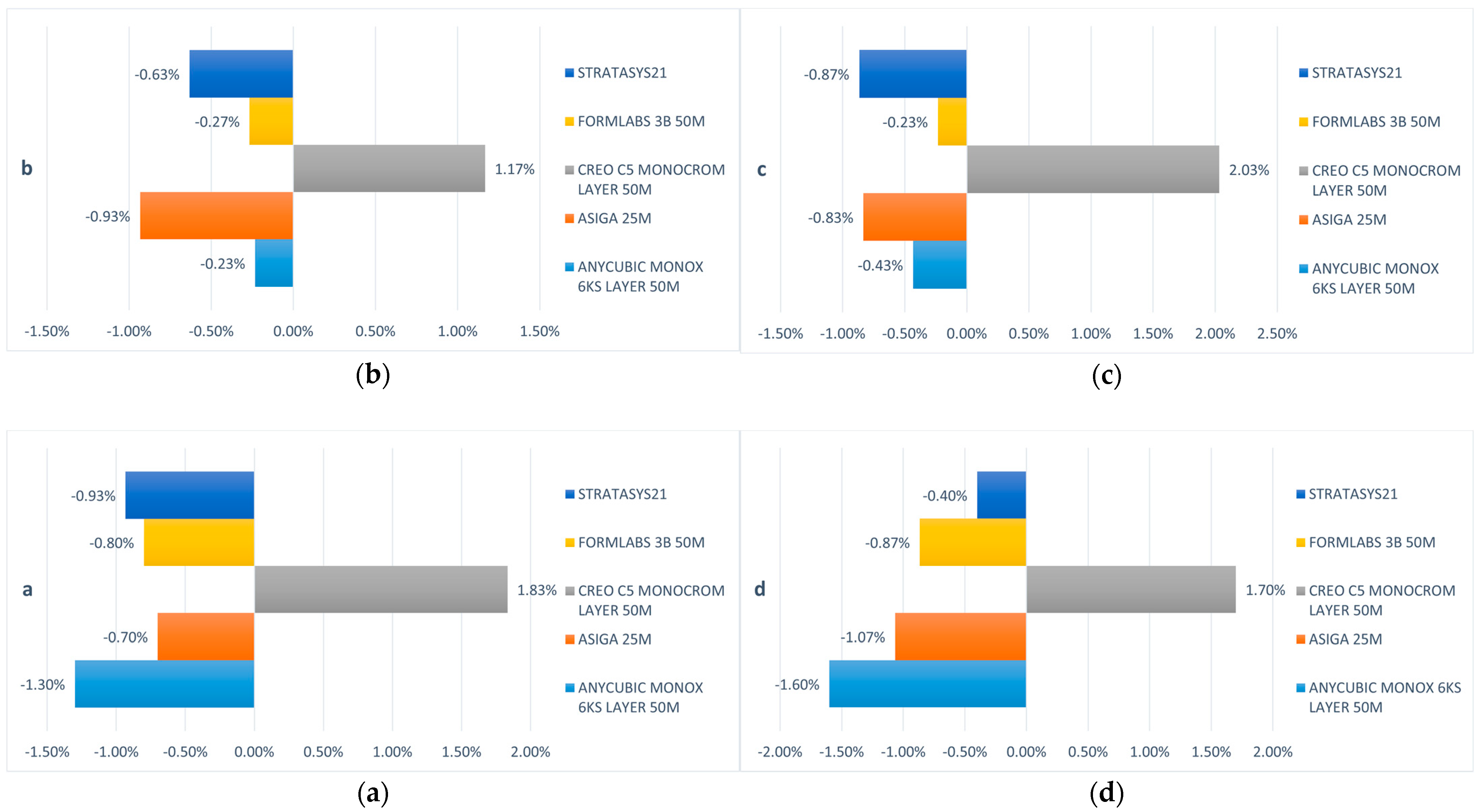
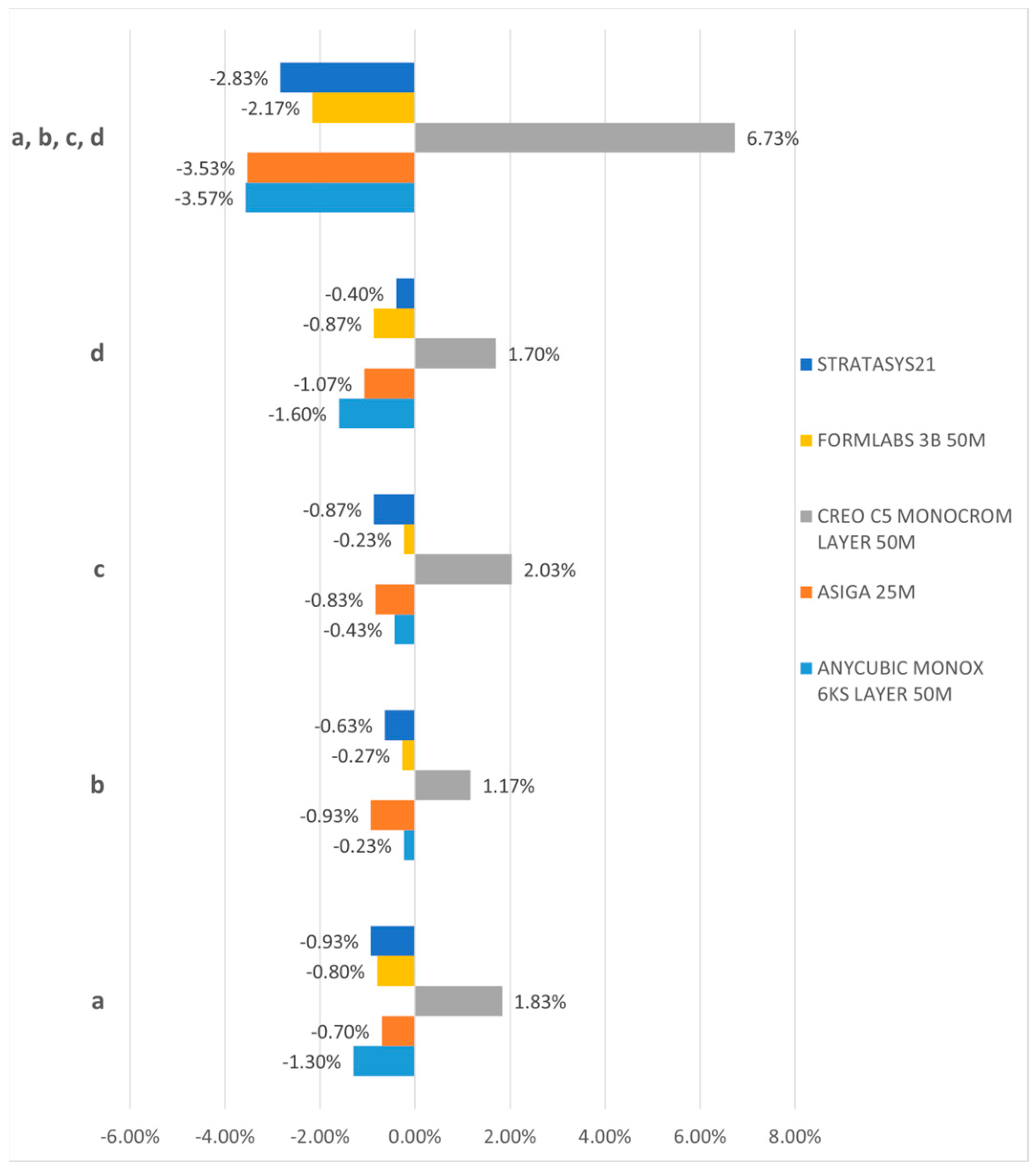
3.1. Three-Dimensional Dimension Trueness
3.2. Linear Trueness and Transversal Distortion
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kim, J.; Pinhata-Baptista, O.; Ayres, A.; da Silva, R.; Lima, J.; Urbano, G.; No-Cortes, J.; Vasques, M.; Cortes, A. Accuracy Comparison among 3D-Printing Technologies to Produce Dental Models. Appl. Sci. 2022, 12, 8425. [Google Scholar] [CrossRef]
- ISO 5725-1:2023(en); Accuracy (Trueness and Precision) of Measurement Methods and Results. Part 1: General Principles and Definitions. International Organization for Standardization: Geneva, Switzerland, 2023.
- Masri, R.; Driscoll, C.F. Clinical Applications of Digital Dental Technology, 2nd ed.; Wiley: Hoboken, NJ, USA, 2023; p. 400. [Google Scholar]
- Mehl, A.; Reich, S.; Beuer, F.; Guth, J.F. Accuracy, trueness, and precision—A guideline for the evaluation of these basic values in digital dentistry. Int. J. Comput. Dent. 2021, 24, 341–352. [Google Scholar] [PubMed]
- Al-Hassiny, A. iDD Compares: Medit i700 W vs. 3Shape TRIOS 5. Available online: https://instituteofdigitaldentistry.com/idd-compares/idd-compares-medit-i700-w-vs-3shape-trios-5/ (accessed on 26 February 2024).
- Banjar, A.; Chen, Y.W.; Kostagianni, A.; Finkelman, M.; Papathanasiou, A.; Chochlidakis, K.; Papaspyridakos, P. Accuracy of 3D Printed Implant Casts Versus Stone Casts: A Comparative Study in the Anterior Maxilla. J. Prosthodont. 2021, 30, 783–788. [Google Scholar] [CrossRef] [PubMed]
- Etemad-Shahidi, Y.; Qallandar, O.B.; Evenden, J.; Alifui-Segbaya, F.; Ahmed, K.E. Accuracy of 3-Dimensionally Printed Full-Arch Dental Models: A Systematic Review. J. Clin. Med. 2020, 9, 3357. [Google Scholar] [CrossRef] [PubMed]
- Groth, C.; Kravitz, N.D.; Jones, P.E.; Graham, J.W.; Redmond, W.R. Three-dimensional printing technology. J. Clin. Orthod. 2014, 48, 475–485. [Google Scholar] [PubMed]
- Rouze l’Alzit, F.; Cade, R.; Naveau, A.; Babilotte, J.; Meglioli, M.; Catros, S. Accuracy of commercial 3D printers for the fabrication of surgical guides in dental implantology. J. Dent. 2022, 117, 103909. [Google Scholar] [CrossRef] [PubMed]
- Emir, F.; Ceylan, G.; Ayyildiz, S. In vitro accuracies of 3D printed models manufactured by two different printing technologies. Eur. Oral. Res. 2021, 55, 80–85. [Google Scholar] [CrossRef] [PubMed]
- Moon, W.; Kim, S.; Lim, B.S.; Park, Y.S.; Kim, R.J.; Chung, S.H. Dimensional Accuracy Evaluation of Temporary Dental Restorations with Different 3D Printing Systems. Materials 2021, 14, 1487. [Google Scholar] [CrossRef]
- Czajkowska, M.; Walejewska, E.; Zadrozny, L.; Wieczorek, M.; Swieszkowski, W.; Wagner, L.; Mijiritsky, E.; Markowski, J. Comparison of Dental Stone Models and Their 3D Printed Acrylic Replicas for the Accuracy and Mechanical Properties. Materials 2020, 13, 4066. [Google Scholar] [CrossRef]
- Moron-Conejo, B.; Lopez-Vilagran, J.; Caceres, D.; Berrendero, S.; Pradies, G. Accuracy of five different 3D printing workflows for dental models comparing industrial and dental desktop printers. Clin. Oral. Investig. 2023, 27, 2521–2532. [Google Scholar] [CrossRef]
- Nedelcu, R.; Olsson, P.; Thulin, M.; Nystrom, I.; Thor, A. In vivo trueness and precision of full-arch implant scans using intraoral scanners with three different acquisition protocols. J. Dent. 2023, 128, 104308. [Google Scholar] [CrossRef] [PubMed]
- Onbasi, Y.; Abu-Hossin, S.; Paulig, M.; Berger, L.; Wichmann, M.; Matta, R.E. Trueness of full-arch dental models obtained by digital and conventional impression techniques: An in vivo study. Sci. Rep. 2022, 12, 22509. [Google Scholar] [CrossRef]
- Sushil, C.; Udai, R.; Rajeev, S.; Pragati, S.; Ayushi. Development of 3D Model and Display on Web Using Open-Source Technology. Int. J. Sci. Res. Sci. Technol. 2022, 9, 346–352. [Google Scholar] [CrossRef]
- ISO 20896-1:2019; Dentistry, Digital Impression Devices, Part 1: Methods for Assessing Accuracy. International Organization for Standardization: Geneva, Switzerland. Available online: https://www.iso.org/standard/69402.html (accessed on 6 May 2023).
- Hongkong Anycubic Technology Co. Anycubic Photon Mono X 6Ks. Available online: https://store.anycubic.com/products/photon-mono-x-6ks (accessed on 28 February 2024).
- Formlabs Inc. User Manual Form 3B. 2023. Available online: https://media.formlabs.com/m/1aa00d88fe52d5bc/original/-ENUS-Form-3B-Manual.pdf (accessed on 28 February 2024).
- Asiga Pty Ltd. Max User Guide. 2017. Available online: https://myaccount.asiga.com/accounts/resources/MAX/1%20-%20Quick%20Start%20Guide/MAX%20User%20Guide%2020220211.pdf (accessed on 28 February 2024).
- Planmeca Oy. Planmeca Creo C5—The ultimate dental 3D printer. 2024. Available online: https://www.planmeca.com/cadcam/3d-printing/3d-printer/ (accessed on 26 July 2023).
- Stratasys. J5 DentaJet. Available online: https://www.stratasys.com/en/3d-printers/printer-catalog/polyjet/j5-dentajet/ (accessed on 28 February 2024).
- Asiga Pty Ltd. Instructions For Use—DentaMODEL, 9 ed. 2024. Available online: https://myaccount.asiga.com/media/public/Materials/Asiga-DentaMODEL-IFU-en.pdf (accessed on 28 February 2024).
- Bártolo, P.J. Stereolithography; Springer: Berlin/Heidelberg, Germany, 2011. [Google Scholar]
- Taormina, G.; Sciancalepore, C.; Messori, M.; Bondioli, F. 3D printing processes for photocurable polymeric materials: Technologies, materials, and future trends. J. Appl. Biomater. Funct. Mater. 2018, 16, 151–160. [Google Scholar] [CrossRef] [PubMed]
- ISO 17296-2:2015(en); Additive Manufacturing—General Principles. Part 2: Overview of Process Categories and Feedstock. International Organization for Standardization: Geneva, Switzerland, 2015.
- Javaid, M.; Haleem, A. Current status and applications of additive manufacturing in dentistry: A literature-based review. J. Oral Biol. Craniofac. Res. 2019, 9, 179–185. [Google Scholar] [CrossRef] [PubMed]
- Revilla-Leon, M.; Meyers, M.J.; Zandinejad, A.; Ozcan, M. A review on chemical composition, mechanical properties, and manufacturing work flow of additively manufactured current polymers for interim dental restorations. J. Esthet. Restor. Dent. 2019, 31, 51–57. [Google Scholar] [CrossRef] [PubMed]
- Revilla-Leon, M.; Besne-Torre, A.; Sanchez-Rubio, J.L.; Fabrega, J.J.; Ozcan, M. Digital tools and 3D printing technologies integrated into the workflow of restorative treatment: A clinical report. J. Prosthet. Dent. 2019, 121, 3–8. [Google Scholar] [CrossRef]
- Revilla-Leon, M.; Fountain, J.; Piedra Cascon, W.; Ozcan, M.; Zandinejad, A. Workflow description of additively manufactured clear silicone indexes for injected provisional restorations: A novel technique. J. Esthet. Restor. Dent. 2019, 31, 213–221. [Google Scholar] [CrossRef]
- Torabi, K.; Farjood, E.; Hamedani, S. Rapid Prototyping Technologies and their Applications in Prosthodontics, a Review of Literature. J. Dent. 2015, 16, 1–9. [Google Scholar]
- Nulty, A. A comparison of trueness and precision of 12 3D printers used in dentistry. BDJ Open 2022, 8, 14. [Google Scholar] [CrossRef]
- Favero, C.S.; English, J.D.; Cozad, B.E.; Wirthlin, J.O.; Short, M.M.; Kasper, F.K. Effect of print layer height and printer type on the accuracy of 3-dimensional printed orthodontic models. Am. J. Orthod. Dentofacial. Orthop. 2017, 152, 557–565. [Google Scholar] [CrossRef] [PubMed]
- Loflin, W.A.; English, J.D.; Borders, C.; Harris, L.M.; Moon, A.; Holland, J.N.; Kasper, F.K. Effect of print layer height on the assessment of 3D-printed models. Am. J. Orthod. Dentofacial. Orthop. 2019, 156, 283–289. [Google Scholar] [CrossRef] [PubMed]
- Papaspyridakos, P.; Chen, Y.W.; Alshawaf, B.; Kang, K.; Finkelman, M.; Chronopoulos, V.; Weber, H.P. Digital workflow: In vitro accuracy of 3D printed casts generated from complete-arch digital implant scans. J. Prosthet. Dent. 2020, 124, 589–593. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.C.; Li, P.L.; Chu, F.T.; Shen, G. Influence of the three-dimensional printing technique and printing layer thickness on model accuracy. J. Orofac. Orthop. 2019, 80, 194–204. [Google Scholar] [CrossRef] [PubMed]
- Hazeveld, A.; Huddleston Slater, J.J.; Ren, Y. Accuracy and reproducibility of dental replica models reconstructed by different rapid prototyping techniques. Am. J. Orthod. Dentofacial. Orthop. 2014, 145, 108–115. [Google Scholar] [CrossRef] [PubMed]
- Wan Hassan, W.N.; Yusoff, Y.; Mardi, N.A. Comparison of reconstructed rapid prototyping models produced by 3-dimensional printing and conventional stone models with different degrees of crowding. Am. J. Orthod. Dentofacial. Orthop. 2017, 151, 209–218. [Google Scholar] [CrossRef] [PubMed]
- Rungrojwittayakul, O.; Kan, J.Y.; Shiozaki, K.; Swamidass, R.S.; Goodacre, B.J.; Goodacre, C.J.; Lozada, J.L. Accuracy of 3D Printed Models Created by Two Technologies of Printers with Different Designs of Model Base. J. Prosthodont. 2020, 29, 124–128. [Google Scholar] [CrossRef]
- Revilla-Leon, M.; Ozcan, M. Additive Manufacturing Technologies Used for Processing Polymers: Current Status and Potential Application in Prosthetic Dentistry. J. Prosthodont. 2019, 28, 146–158. [Google Scholar] [CrossRef] [PubMed]
- Desimone, J.M.; Er-Moshkin, A.; Samulski, E. Continuous Liquid Interphase Printing. 2014. Available online: https://patents.google.com/patent/WO2014126837A2/en (accessed on 28 February 2024).
- Mostafavi, D.; Methani, M.M.; Piedra-Cascon, W.; Zandinejad, A.; Revilla-Leon, M. Influence of the Rinsing Postprocessing Procedures on the Manufacturing Accuracy of Vat-Polymerized Dental Model Material. J. Prosthodont. 2021, 30, 610–616. [Google Scholar] [CrossRef]
- Cooperstein, I.; Layani, M.; Magdassi, S. 3D printing of porous structures by UV-curable O/W emulsion for fabrication of conductive objects. J. Mater. Chem. C 2015, 3, 2040–2044. [Google Scholar] [CrossRef]
- Phrozen Tech Co. Water-Washable Dental Model Resin. Available online: https://dental.phrozen3d.com/pages/dental-resin (accessed on 28 February 2024).
- Gurr, M.; Hofmann, D.; Ehm, M.; Thomann, Y.; Kübler, R.; Mülhaupt, R. Acrylic Nanocomposite Resins for Use in Stereolithography and Structural Light Modulation Based Rapid Prototyping and Rapid Manufacturing Technologies. Adv. Funct. Mater. 2008, 18, 2390–2397. [Google Scholar] [CrossRef]
- Sandoval, J.H.; Soto, K.F.; Murr, L.E.; Wicker, R.B. Nanotailoring photocrosslinkable epoxy resins with multi-walled carbon nanotubes for stereolithography layered manufacturing. J. Mater. Sci. 2006, 42, 156–165. [Google Scholar] [CrossRef]
- Piedra-Cascon, W.; Krishnamurthy, V.R.; Att, W.; Revilla-Leon, M. 3D printing parameters, supporting structures, slicing, and post-processing procedures of vat-polymerization additive manufacturing technologies: A narrative review. J. Dent. 2021, 109, 103630. [Google Scholar] [CrossRef] [PubMed]
- Scherer, M.D.; Barmak, A.B.; Ozcan, M.; Revilla-Leon, M. Influence of postpolymerization methods and artificial aging procedures on the fracture resistance and flexural strength of a vat-polymerized interim dental material. J. Prosthet. Dent. 2022, 128, 1085–1093. [Google Scholar] [CrossRef] [PubMed]
- Reymus, M.; Lumkemann, N.; Stawarczyk, B. 3D-printed material for temporary restorations: Impact of print layer thickness and post-curing method on degree of conversion. Int. J. Comput. Dent. 2019, 22, 231–237. [Google Scholar] [PubMed]
- Arnold, C.; Monsees, D.; Hey, J.; Schweyen, R. Surface Quality of 3D-Printed Models as a Function of Various Printing Parameters. Materials 2019, 12, 1970. [Google Scholar] [CrossRef] [PubMed]
- Park, G.S.; Kim, S.K.; Heo, S.J.; Koak, J.Y.; Seo, D.G. Effects of Printing Parameters on the Fit of Implant-Supported 3D Printing Resin Prosthetics. Materials 2019, 12, 2533. [Google Scholar] [CrossRef]
- Chockalingam, K.; Jawahar, N.; Chandrasekhar, U. Influence of layer thickness on mechanical properties in stereolithography. Rapid Prototyp. J. 2006, 12, 106–113. [Google Scholar] [CrossRef]
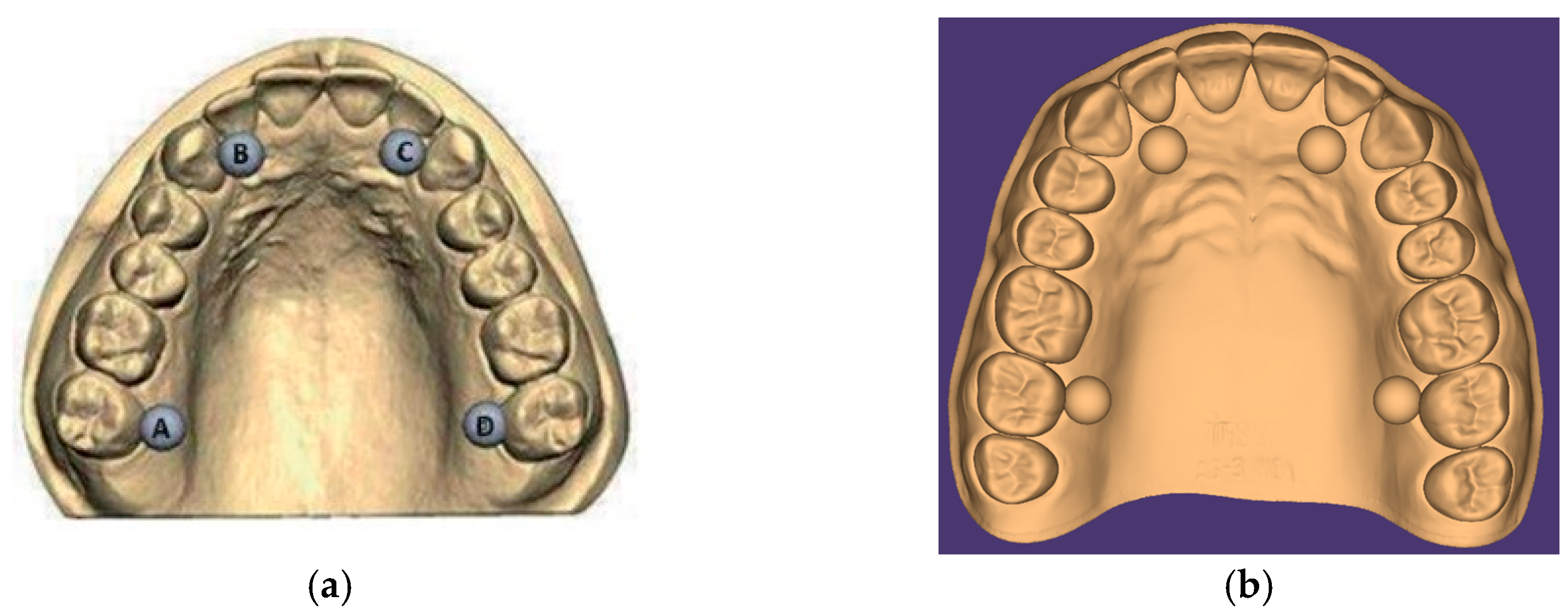


| Distances between the Centers of the Balls | ||
|---|---|---|
| a–b: (40 ± 4) mm | OR | a to b: (35 ± 4) mm |
| b–c: (35 ± 4) mm | b to c: (20 ± 4) mm | |
| c–d: (40 ± 4) mm | c to d: (35 ± 4) mm | |
| a–d: (55 ± 6) mm | a to d: (35 ± 6) mm | |
| a–c: (60 ± 6) mm | a to c: (44 ± 6) mm | |
| b–d: (60 ± 6) mm | b to d: (44 ± 6) mm | |
| Parameter | Value |
|---|---|
| a–b | 32 mm |
| b–c | 19.474 mm |
| c–d | 32 mm |
| a–d | 38 mm |
| a–c | 42 mm |
| b–d | 42 mm |
| Printer | Anycubic Mono X 6Ks | Asiga Max | Creo C5 | Form 3B | J5 DentaJet |
|---|---|---|---|---|---|
| Manufacturer | Hongkong Anycubic Technology Co., China | Asiga, Australia | Planmeca Oy, Finland | Formlabs Inc., USA | Stratasys, USA |
| Technology | mSLA | DLP | DLP | SLA | Polyjet |
| Wavelength | 405 nm | 385/405 nm | 385 nm | 405 nm | NA |
| XY Resolution | 34.4 microns | 62 microns | 50 microns | 25 microns * | 18.75/20.625 microns |
| Layer Height | 50 microns | 25 microns | 50 microns | 50 microns | 18 microns |
| Software | Chitubox Basic | Asiga Composer | Planmeca Creo C5 Studio | PreForm | GrabCAD Print |
| Resin | White-Colored UV Resin Anycubic | DentaMODEL, Asiga | FotoDent model 2, Dreve Dentamid | Model Resin, Formlabs | TrueDent White, Stratasys |
| IPA Wash | 10 min immersion in 99% IPA | 10 min immersion in 99% IPA + 10 min immersion in 99% fresh IPA | 2 cycles of 6 min each in fresh 99% IPA | 10 min immersion in 99% IPA | 30 min immersion in 99% IPA ** |
| Curing | 10 min in the Form Cure, without heating | Asiga Flash for 20 min, no heating | 12 min in the Form Cure, without heating | 5 min at 60 °C in the Form Cure | 2 h while immersed in glycerol at 80 °C ** |
| ISO Values | Nominal Values | Anycubic Monox 6KS 50 m | Asiga 25 m | Asiga 75 m | Creo c5 Monocrom 50 m | Formlabs 3B 50 m | Stratasys 21 |
|---|---|---|---|---|---|---|---|
| a = 6 ± 2 | 6 | 5.922 | 5.958 | 5.880 | 6.110 | 5.952 | 5.944 |
| b = 6 ± 2 | 6 | 5.986 | 5.944 | 5.880 | 6.070 | 5.984 | 5.962 |
| c = 6 ± 2 | 6 | 5.974 | 5.950 | 5.880 | 6.122 | 5.986 | 5.948 |
| d = 6 ± 2 | 6 | 5.904 | 5.936 | 5.856 | 6.102 | 5.948 | 5.976 |
| a–b = 35 ± 4 | 32 | 31.657 | 31.933 | 31.882 | 31.845 | 31.982 | 32.027 |
| b–c = 20 ± 4 | 19.474 | 19.306 | 19.388 | 19.358 | 19.367 | 19.427 | 19.503 |
| c–d = 35 ± 4 | 32 | 31.647 | 31.931 | 31.877 | 31.841 | 31.964 | 31.995 |
| a–d = 35 ± 6 | 38 | 37.591 | 37.876 | 37.845 | 37.852 | 37.888 | 38.086 |
| a–c = 44 ± 6 | 42 | 41.566 | 41.880 | 41.818 | 41.772 | 41.958 | 42.039 |
| b–d = 44 ± 6 | 42 | 41.563 | 41.882 | 41.822 | 41.811 | 41.907 | 42.045 |
| Parameter | Anycubic Monox 6KS 50 m | Asiga 25 m | Creo c5 Monocrom 50 m | Formlabs 3B 50 m | Stratasys 21 | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| Trueness | Trueness Deviation | Trueness | Trueness Deviation | Trueness | Trueness Deviation | Trueness | Trueness Deviation | Trueness | Trueness Deviation | |
| a | 98.70% | −1.30% | 99.30% | −0.70% | 101.83% | 1.83% | 99.20% | –0.80% | 99.07% | –0.93% |
| b | 99.77% | −0.23% | 99.07% | −0.93% | 101.17% | 1.17% | 99.73% | −0.27% | 99.37% | −0.63% |
| c | 99.57% | −0.43% | 99.17% | −0.83% | 102.03% | 2.03% | 99.77% | −0.23% | 99.13% | −0.87% |
| d | 98.40% | −1.60% | 98.93% | −1.07% | 101.70% | 1.70% | 99.13% | −0.87% | 99.60% | −0.40% |
| a–b | 98.93% | −1.07% | 99.79% | −0.21% | 99.52% | −0.48% | 99.94% | −0.06% | 100.08% | 0.08% |
| b–c | 99.14% | −0.86% | 99.56% | −0.44% | 99.45% | −0.55% | 99.76% | −0.24% | 100.15% | 0.15% |
| c–d | 98.90% | −1.10% | 99.78% | −0.22% | 99.50% | −0.50% | 99.89% | −0.11% | 99.98% | −0.02% |
| a–d | 98.92% | −1.08% | 99.67% | −0.33% | 99.61% | −0.39% | 99.71% | −0.29% | 100.23% | 0.23% |
| a–c | 98.97% | −1.03% | 99.71% | −0.29% | 99.46% | −0.54% | 99.90% | −0.10% | 100.09% | 0.09% |
| b–d | 98.96% | −1.04% | 99.72% | −0.28% | 99.55% | −0.45% | 99.78% | −0.22% | 100.11% | 0.11% |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ciocan, L.T.; Vasilescu, V.G.; Pantea, M.; Pițuru, S.M.; Imre, M.; Ripszky Totan, A.; Froimovici, F.O. The Evaluation of the Trueness of Dental Mastercasts Obtained through Different 3D Printing Technologies. J. Funct. Biomater. 2024, 15, 210. https://doi.org/10.3390/jfb15080210
Ciocan LT, Vasilescu VG, Pantea M, Pițuru SM, Imre M, Ripszky Totan A, Froimovici FO. The Evaluation of the Trueness of Dental Mastercasts Obtained through Different 3D Printing Technologies. Journal of Functional Biomaterials. 2024; 15(8):210. https://doi.org/10.3390/jfb15080210
Chicago/Turabian StyleCiocan, Lucian Toma, Vlad Gabriel Vasilescu, Mihaela Pantea, Silviu Mirel Pițuru, Marina Imre, Alexandra Ripszky Totan, and Florin Octavian Froimovici. 2024. "The Evaluation of the Trueness of Dental Mastercasts Obtained through Different 3D Printing Technologies" Journal of Functional Biomaterials 15, no. 8: 210. https://doi.org/10.3390/jfb15080210






