Long-Term Stability and Osteogenic Activity of Recycled Polysulfone-Calcium Silicate Bone Implants In Vitro
Abstract
:1. Introduction
2. Materials and Methods
2.1. Preparation of PSF
2.2. Preparation of CaSi
2.3. Preparation of PSF-Based Composites
2.4. In Vitro Stability
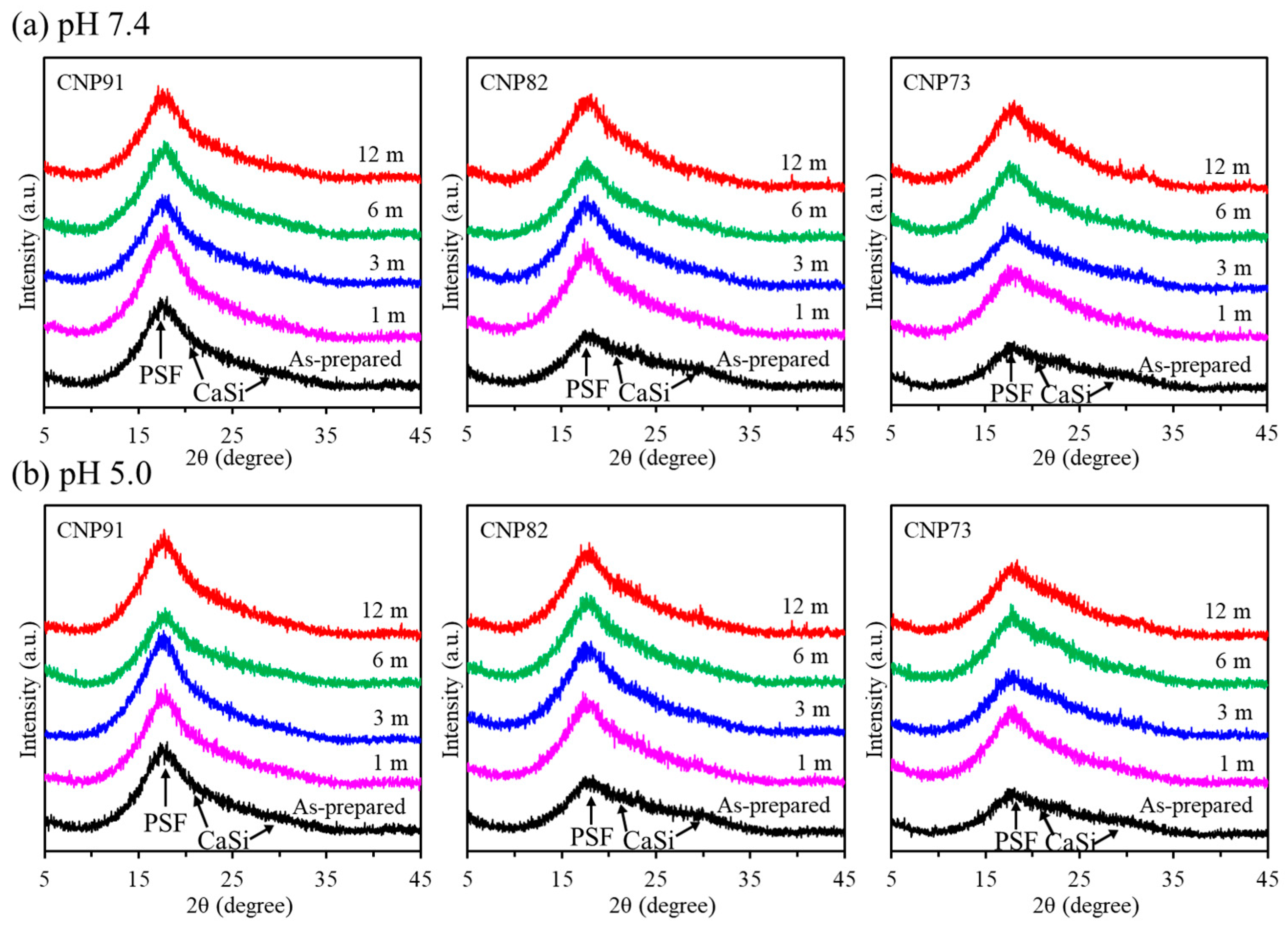
2.5. Phase Composition and Surface Morphology
2.6. Measurement of Weight Loss
2.7. Measurement of Three-Point Bending Strength
2.8. In Vitro Osteogenic Activity
2.9. Cell Attachment and Proliferation
2.10. ALP Activity and Mineralization
2.11. Statistical Analysis
3. Results
3.1. Phase Composition
3.2. Morphology
3.3. Three-Point Bending Strength
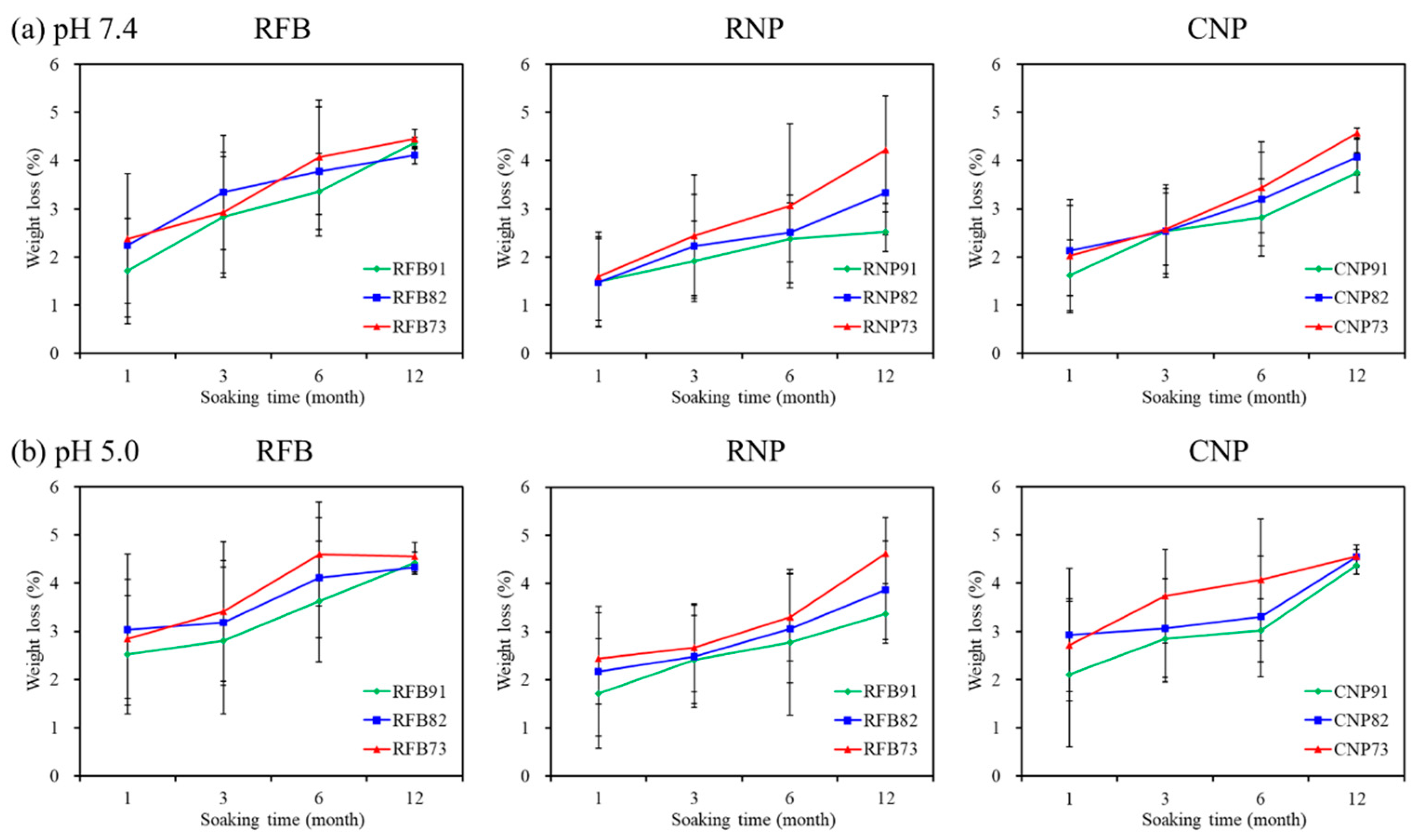
3.4. Weight Loss
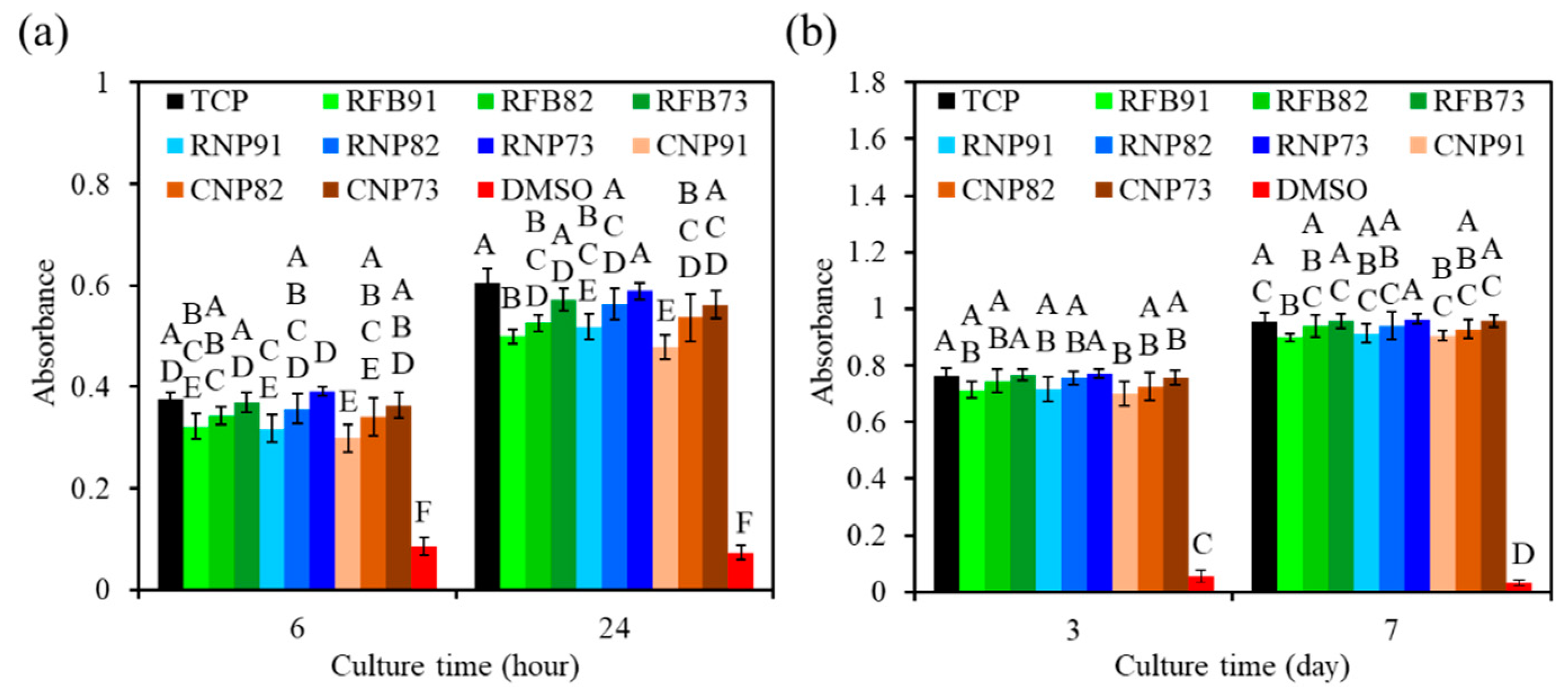
3.5. Cell Attachment and Proliferation
3.6. ALP Activity and Mineralization
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Yu, L.; Zhao, D.; Wang, W. Mechanical properties and long-term durability of recycled polysulfone plastic. Waste Manag. 2019, 84, 402–412. [Google Scholar] [CrossRef] [PubMed]
- Khanzada, N.K.; Al-Juboori, R.A.; Khatri, M.; Ahmed, F.E.; Ibrahim, Y.; Hilal, N. Sustainability in membrane technology: Membrane recycling and fabrication using recycled waste. Membranes 2024, 14, 52. [Google Scholar] [CrossRef] [PubMed]
- Khaliha, S.; Tunioli, F.; Foti, L.; Bianchi, A.; Kovtun, A.; Marforio, T.D.; Zambianchi, M.; Bettini, C.; Briñas, E.; Vázquez, E.; et al. Upcycling of plastic membrane industrial scraps and reuse as sorbent for emerging contaminants in water. Environ. Sci. Water Res. Technol. 2024, 10, 1097–1107. [Google Scholar] [CrossRef]
- Chang, C.N.; Chung, J.J.; Jiang, H.Y.; Ding, S.J. Calcium silicate promoting the upcycling potential of polysulfone medical waste in load-bearing applications. J. Funct. Biomater. 2024, 15, 323. [Google Scholar] [CrossRef]
- Dickinson, B.L. UDEL® Polysulfone for medical applications. J. Biomater. Appl. 1988, 3, 605–634. [Google Scholar] [CrossRef]
- Nakashima, A.; Ogata, S.; Doi, S.; Yamahira, M.; Naraki, S.; Takasugi, N.; Ohmoto, T.; Ito, T.; Masaki, T.; Yorioka, N. Performance of polysulfone membrane dialyzers and dialysate flow pattern. Clin. Exp. Nephrol. 2006, 10, 210–215. [Google Scholar] [CrossRef]
- Wang, C.; Lin, B.; Qiu, Y. Enhanced hydrophilicity and anticoagulation of polysulfone materials modified via dihydroxypropyl, sulfonic groups and chitosan. Colloids Surf. B 2022, 210, 112243. [Google Scholar] [CrossRef]
- Ramakrishna, S.; Mayer, J.; Wintermantel, E.; Leong, K.W. Biomedical applications of polymer-composite materials: A review. Compos. Sci. Technol. 2001, 61, 1189–1224. [Google Scholar] [CrossRef]
- Oréfice, R.; West, J.; LaTorre, G.; Hench, L.; Brennan, A. Effect of long-term in vitro testing on the properties of bioactive glass-polysulfone composites. Biomacromolecules 2010, 11, 657–665. [Google Scholar] [CrossRef]
- Michael, F.M.; Khalid, M.; Walvekar, R.; Ratnam, C.T.; Ramarad, S.; Siddiqui, H.; Hoque, M.E. Effect of nanofillers on the physico-mechanical properties of load bearing bone implants. Mater. Sci. Eng. C 2016, 67, 792–806. [Google Scholar] [CrossRef]
- Alcántara-Arreola, E.A.; Silva-Garcés, K.N.; Mendoza-Martínez, J.; Cardoso-Palomares, M.A.; Torres-SanMiguel, C.R. Experimental analysis of stress shielding effects in screw spacers placed in porcine spinal tissue. J. Funct. Biomater. 2024, 15, 238. [Google Scholar] [CrossRef] [PubMed]
- Kettunen, J.; Makela, E.A.; Miettinen, H.; Nevalainen, T.; Heikkilä, M.; Pohjonen, T.; Törmälä, P.; Rokkanen, P. Mechanical properties and strength retention of carbon fibre-reinforced liquid crystalline polymer (LCP/CF) composite: An experimental study on rabbits. Biomaterials 1998, 19, 1219–1228. [Google Scholar] [CrossRef] [PubMed]
- Isaksson, H. Recent advances in mechanobiological modeling of bone regeneration. Mech. Res. Commun. 2012, 42, 22–31. [Google Scholar] [CrossRef]
- Mehboob, H.; Chang, S.H. Application of composites to orthopedic prostheses for effective bone healing: A review. Compos. Struct. 2014, 118, 328–341. [Google Scholar] [CrossRef]
- Bhat, K.A.; Prakash, P.L.; Manoharan, N.; Lakshmibai, A.; Sangeetha, D. Fabrication of polymethyl methacrylate/polysulfone/nanoceramic composites for orthopedic applications. J. Appl. Polym. Sci. 2013, 127, 2764–2775. [Google Scholar] [CrossRef]
- Park, S.W.; Yoo, S.H.; An, S.T.; Chang, S.H. Material characterization of glass/polypropylene composite bone plates according to the forming condition and performance evaluation under a simulated human body environment. Compos. B 2012, 43, 1101–1108. [Google Scholar] [CrossRef]
- Wu, I.T.; Kao, P.F.; Huang, Y.R.; Ding, S.J. In vitro and in vivo osteogenesis of gelatin-modified calcium silicate cement with washout resistance. Mater. Sci. Eng. C 2020, 117, 111297. [Google Scholar] [CrossRef]
- Huang, Y.R.; Wu, I.T.; Chen, C.C.; Ding, S.J. In vitro comparisons of microscale and nanoscale calcium silicate particles. J. Mater. Chem. B 2020, 8, 6034–6047. [Google Scholar] [CrossRef]
- Santos, J.M.; Coelho, C.M.; Sequeira, D.B.; Marques, J.A.; Pereira, J.F.; Sousa, V.; Palma, P.J.; Santos, A.C. Subcutaneous implantation assessment of new calcium-silicate based sealer for warm obturation. Biomedicines 2021, 9, 24. [Google Scholar] [CrossRef]
- Ding, S.J.; Chu, Y.H.; Chen, P.T. Mechanical biocompatibility, osteogenic activity and antibacterial efficacy of calcium silicate-zirconia biocomposites. ACS Omega 2021, 6, 7106–7118. [Google Scholar] [CrossRef]
- ASTM D 790; Standard Test Methods for Flexural Properties of Unreinforced and Reinforced Plastics and Electrical Insulating Materials. American Society of Testing and Material: West Conshohocken, PA, USA, 2003.
- Silver, I.A.; Murrills, R.J.; Etherington, D.J. Microelectrode studies on the acid microenvironment beneath adherent macrophages and osteoclasts. Exp. Cell Res. 1998, 175, 266–276. [Google Scholar] [CrossRef] [PubMed]
- Sun, L.; Xu, H.H.K.; Takagi, S.; Chow, L.C. Fast setting calcium phosphate cement-chitosan composite: Mechanical properties and dissolution rates. J. Biomater. Appl. 2007, 21, 299–315. [Google Scholar] [CrossRef] [PubMed]
- Wei, C.K.; Ding, S.J. Acid-resistant calcium silicate-based composite implants with high-strength as load-bearing bone graft substitutes and fracture fixation devices. J. Mech. Behav. Biomed. Mater. 2016, 62, 366–383. [Google Scholar] [CrossRef] [PubMed]
- Rámila, A.; Vallet-Regí, M. Static and dynamic in vitro study of a sol-gel glass bioactivity. Biomaterials 2001, 22, 2301–2306. [Google Scholar] [CrossRef]
- Huang, J.; Di Silvio, L.; Wang, M.; Tanner, K.E.; Bonfield, W. In vitro mechanical and biological assessment of hydroxyapatite-reinforced polyethylene composite. J. Mater. Sci. Mater. Med. 1997, 8, 775–779. [Google Scholar] [CrossRef]
- Brown, S.A.; Hastings, R.S.; Mason, J.J.; Moet, A. Characterization of short-fiber reinforced thermoplastics for fracture fixation devices. Biomaterials 1990, 11, 541–547. [Google Scholar] [CrossRef]
- Meyer, M.R.; Friedman, R.J.; Del Schutte, H., Jr.; Latour, R.A., Jr. Long-term durability of the interface in FRP composites after exposure to simulated physiologic saline environment. J. Biomed. Mater. Res. 1994, 28, 1221–1231. [Google Scholar] [CrossRef]
- Ekstrand, K.; Ruyter, I.E.; Wellendorf, H. Carbon/graphite fibre reinforced poly (methyl methacrylate): Properties under dry and wet conditions. J. Biomed. Mater. Res. 1987, 21, 1065–1080. [Google Scholar] [CrossRef]
- Heini, P.F.; Berlemann, U. Bone substitutes in vertebroplasty. Eur. Spine J. 2001, 10, S205–S213. [Google Scholar]
- Bunpetch, V.; Zhang, X.; Li, T.; Lin, J.; Maswikiti, E.; Wu, Y.; Cai, D.; Li, J.; Zhang, S.; Wu, C.; et al. Silicate-based bioceramic scaffolds for dual-lineage regeneration of osteochondral defect. Biomaterials 2019, 192, 323–333. [Google Scholar] [CrossRef]
- Emadi, H.; Baghani, M.; Masoudi Rad, M.; Hoomehr, B.; Baniassadi, M.; Lotfian, S. 3D-printed polycaprolactone-based containing calcium zirconium silicate: Bioactive scaffold for accelerating bone regeneration. Polymers 2024, 16, 1389. [Google Scholar] [CrossRef] [PubMed]
- Marco, F.; Milena, F.; Gianluca, G.; Vittoria, O. Peri-implant osteogenesis in health and osteoporosis. Micron 2005, 36, 630–644. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.; Lee, D.; Kye, M.; Ha, Y.-J.; Kim, S.-Y. Biocompatible Properties and mineralization potential of premixed calcium silicate-based cements and fast-set calcium silicate-based cements on human bone marrow-derived mesenchymal stem cells. Materials 2022, 15, 7595. [Google Scholar] [CrossRef] [PubMed]
- Di Maro, M.; Faga, M.G.; Malucelli, G.; Mussano, F.D.; Genova, T.; Morsi, R.E.; Duraccio, D. Influence of chitosan on the mechanical and biological properties of HDPE for biomedical applications. Polym Test. 2020, 91, 106610. [Google Scholar] [CrossRef]
- Bello, S.A.; Egbanubi, O.E.; Alabi, A.G.F. Emerging hybrid particle-reinforced high-density polyethylene nanocomposite for bone replacement. Polym. Bull. 2024, 81, 1743–1766. [Google Scholar] [CrossRef]
- Zheng, J.; Zhao, H.; Ouyang, Z.; Zhou, X.; Kang, J.; Yang, C.; Sun, C.; Xiong, M.; Fu, M.; Jin, D.; et al. Additively-manufactured HA/PEEK porous scaffolds with excellent osteogenesis for bone tissue repairing. Compos. B Eng. 2022, 232, 109508. [Google Scholar] [CrossRef]
- Lowe, J.A.; Crist, B.D.; Bhandari, M.; Ferguson, T.A. Optimal treatment of femoral neck fractures according to patient’s physiologic age: An evidence-based review. Orthop. Clin. N. Am. 2010, 41, 157–166. [Google Scholar] [CrossRef]
- D’Ippolito, G.; Schiller, P.C.; Ricordi, C.; Roos, B.A.; Howard, G.A. Age-related osteogenic potential of mesenchymal stromal stem cells from human vertebral bone marrow. J. Bone Min. Res. 1999, 14, 1115–1122. [Google Scholar] [CrossRef]
- Pius, A.K.; Toya, M.; Gao, Q.; Lee, M.L.; Ergul, Y.S.; Chow, S.K.-H.; Goodman, S.B. Effects of aging on osteosynthesis at bone–implant interfaces. Biomolecules 2024, 14, 52. [Google Scholar] [CrossRef]
- Kammerlander, C.; Pfeufer, D.; Lisitano, L.A.; Mehaffey, S.; Böcker, W.; Neuerburg, C. Inability of older adult patients with hip fracture to maintain postoperative weight-bearing restrictions. J. Bone Jt. Surg. Am. 2018, 100, 936–941. [Google Scholar] [CrossRef]
- Maffulli, N.; Aicale, R. Proximal femoral fractures in the elderly: A few things to know, and some to forget. Medicina 2022, 58, 1314. [Google Scholar] [CrossRef]





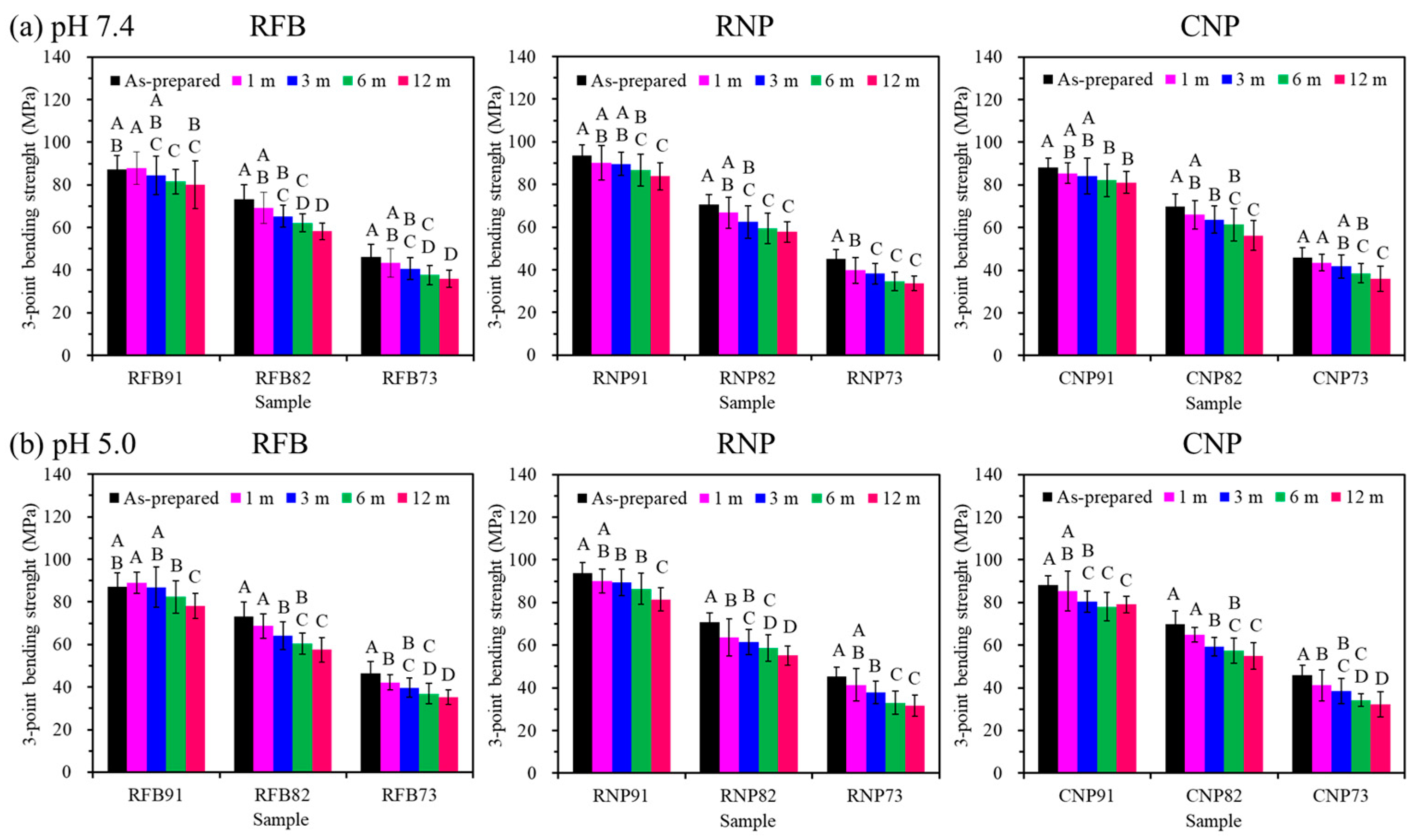


| Soaking Time (Month) | RFB | RNP | CNP | ||||||
|---|---|---|---|---|---|---|---|---|---|
| 91 | 82 | 73 | 91 | 82 | 73 | 91 | 82 | 73 | |
| pH 7.4 | |||||||||
| 0 | 87.1 ± 6.5 | 73.2 ± 6.8 | 46.4 ± 5.8 | 93.1 ± 4.9 | 70.6 ± 4.6 | 45.2 ± 4.4 | 88.2 ± 4.3 | 69.8 ± 6.1 | 46.0 ± 4.6 |
| 1 | 87.8 ± 7.5 | 69.1 ± 7.2 | 43.3 ± 6.7 | 90.1 ± 8.2 | 66.7 ± 7.2 | 39.8 ± 6.1 | 85.5 ± 4.9 | 66.0 ± 6.7 | 43.8 ± 3.9 |
| 3 | 84.5 ± 9.1 | 65.3 ± 5.2 | 40.1 ± 5.1 | 89.7 ± 5.5 | 62.4 ± 7.6 | 38.2 ± 4.9 | 85.1 ± 8.4 | 63.7 ± 6.4 | 41.7 ± 5.4 |
| 6 | 81.5 ± 5.7 | 62.2 ± 4.3 | 37.7 ± 4.6 | 86.8 ± 7.4 | 59.4 ± 7.2 | 34.5 ± 4.4 | 82.2 ± 7.6 | 61.3 ± 7.5 | 38.6 ± 4.6 |
| 12 | 80.0 ± 11.2 | 58.2 ± 3.9 | 36.0 ± 4.0 | 83.8 ± 6.3 | 57.8 ± 4.8 | 33.6 ± 3.4 | 81.1 ± 5.1 | 56.2 ± 7.0 | 36.0 ± 5.8 |
| pH 5.0 | |||||||||
| 0 | 87.1 ± 6.5 | 73.2 ± 6.8 | 46.4 ± 5.8 | 93.1 ± 4.9 | 70.6 ± 4.6 | 45.2 ± 4.4 | 88.2 ± 4.26 | 69.8 ± 6.1 | 46.0 ± 4.6 |
| 1 | 88.9 ± 5.1 | 68.7 ± 5.8 | 42.2 ± 3.5 | 90.1 ± 5.5 | 63.7 ± 8.7 | 41.4 ± 7.6 | 85.3 ± 9.4 | 64.9 ± 3.3 | 41.2 ± 7.3 |
| 3 | 86.9 ± 9.5 | 64.1 ± 6.4 | 36.9 ± 4.6 | 39.3 ± 6.3 | 61.4 ± 5.9 | 37.9 ± 5.3 | 80.4 ± 5.1 | 59.4 ± 4.4 | 38.4 ± 5.9 |
| 6 | 82.4 ± 7.6 | 60.3 ± 5.0 | 36.9 ± 4.9 | 86.5 ± 7.4 | 58.7 ± 6.3 | 33.0 ± 5.5 | 78.1 ± 6.8 | 57.4 ± 6.0 | 34.2 ± 2.8 |
| 12 | 78.1 ± 5.8 | 57.4 ± 5.8 | 35.2 ± 3.4 | 81.5 ± 5.4 | 55.1 ± 4.6 | 31.7 ± 4.9 | 79.0 ± 3.9 | 55.0 ± 6.2 | 32.3 ± 5.9 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chang, C.-N.; Huang, Y.-R.; Ding, S.-J. Long-Term Stability and Osteogenic Activity of Recycled Polysulfone-Calcium Silicate Bone Implants In Vitro. J. Funct. Biomater. 2025, 16, 31. https://doi.org/10.3390/jfb16010031
Chang C-N, Huang Y-R, Ding S-J. Long-Term Stability and Osteogenic Activity of Recycled Polysulfone-Calcium Silicate Bone Implants In Vitro. Journal of Functional Biomaterials. 2025; 16(1):31. https://doi.org/10.3390/jfb16010031
Chicago/Turabian StyleChang, Chi-Nan, Yun-Ru Huang, and Shinn-Jyh Ding. 2025. "Long-Term Stability and Osteogenic Activity of Recycled Polysulfone-Calcium Silicate Bone Implants In Vitro" Journal of Functional Biomaterials 16, no. 1: 31. https://doi.org/10.3390/jfb16010031
APA StyleChang, C.-N., Huang, Y.-R., & Ding, S.-J. (2025). Long-Term Stability and Osteogenic Activity of Recycled Polysulfone-Calcium Silicate Bone Implants In Vitro. Journal of Functional Biomaterials, 16(1), 31. https://doi.org/10.3390/jfb16010031







