Potential of Nanonutraceuticals in Increasing Immunity
Abstract
:1. Introduction
2. Vitamins
2.1. Vitamin C
2.2. Vitamin B12
2.3. Folic Acid
2.4. Vitamin D
2.5. Vitamin E
3. Minerals
3.1. Iron
3.2. Selenium
3.3. Zinc
4. Antioxidants
4.1. Carotenoids
β-Carotene
4.2. Coenzyme Q10
4.3. Polyphenols
4.3.1. Resveratrol
4.3.2. Catechins
4.4. Curcumin
5. Fatty Acids
6. Probiotics
7. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
References
- Elgert, K.D. Immunology: Understanding the Immune System; Wiley-Blackwell: Hoboken, NJ, USA, 2009. [Google Scholar]
- Neuschlova, M.; Novakova, E.; Kompanikova, J. Immunology—How the Immune System Works; Jessenius Faculty of Medicine in Martin, Comenius University in Bratislava: Bratislava, Slovakia, 2017. (In Slovak) [Google Scholar]
- Abbas, A.K.; Lichtman, A.H.; Pillai, S. Basic Immunology: Functions and Disorders of the Immune System, 6th ed.; Elsevier: Amsterdam, The Netherlands, 2019. [Google Scholar]
- Rao, C.V. An Introduction to Immunology; Alpha Science International: Pangbourne, India, 2002. [Google Scholar]
- Calder, P.C.; Yaqoob, P. Nutrient regulation of the immune response. In Present Knowledge in Nutrition, 11th ed.; Marriott, B.P., Birt, D.F., Stallings, V.A., Yates, A.A., Eds.; Academic Press: Cambridge, MA, USA, 2020; pp. 625–664. [Google Scholar]
- Redondo, N.; Nova, E.; Gomez-Martinez, S.; Diaz-Prieto, L.E.; Marcos, A. Diet, nutrition and the immune system. In Encyclopedia of Food Security and Sustainability; Ferranti, P., Berry, E.M., Anderson, J.R., Eds.; Elsevier: Amsterdam, The Netherlands, 2019; pp. 250–255. [Google Scholar]
- Lapik, I.A.; Galchenko, A.V.; Gapparova, K.M. Micronutrient status in obese patients: A narrative review. Obes. Med. 2020, 18, 100224. [Google Scholar] [CrossRef]
- Ashaolu, T.J. Immune boosting functional foods and their mechanisms: A critical evaluation of probiotics and prebiotics. Biomed. Pharmacother. 2020, 130, 110625. [Google Scholar] [CrossRef] [PubMed]
- Kersiene, M.; Jasutiene, I.; Eisinaite, V.; Venskutonis, P.R.; Leskauskaite, D. Designing multiple bioactives loaded emulsions for the formulations for diets of elderly. Food Funct. 2020, 11, 2195–2207. [Google Scholar] [CrossRef] [PubMed]
- Nasri, H.; Baradaran, A.; Shirzad, H.; Rafieian-Kopaei, M. New concepts in nutraceuticals as alternative for pharmaceuticals. Int. J. Prev. Med. 2014, 5, 1487–1499. [Google Scholar] [PubMed]
- Kohout, P. Possibilities of affecting the immune system with nutraceutics. Intern. Med. 2010, 12, 140–144. (In Czech) [Google Scholar]
- Vergallo, C. Nutraceutical vegetable oil nanoformulations for prevention and management of diseases. Nanomaterials 2020, 10, 1232. [Google Scholar] [CrossRef] [PubMed]
- Sachdeva, V.; Roy, A.; Bharadvaja, N. Current prospects of nutraceuticals: A review. Curr. Pharm. Biotechnol. 2020, 21, 884–896. [Google Scholar] [CrossRef] [PubMed]
- Das, L.; Bhaumik, E.; Raychaudhuri, U.; Chakraborty, R. Role of nutraceuticals in human health. J. Food Sci. Technol. 2012, 49, 173–183. [Google Scholar] [CrossRef] [Green Version]
- Aronson, J.K. Defining ‘nutraceuticals’: Neither nutritious nor pharmaceutical. Br. J. Clin. Pharmacol. 2017, 8, 19. [Google Scholar] [CrossRef]
- EU. Register of Nutrition and Health Claims Made on Foods. Available online: https://ec.europa.eu/food/safety/labelling_nutrition/claims/register/public/?event=register.home (accessed on 6 October 2020).
- European Commission—Health Claims. Available online: https://ec.europa.eu/food/safety/labelling_nutrition/claims/health_claims_en (accessed on 6 October 2020).
- Jayawardena, R.; Sooriyaarachchi, P.; Chourdakis, M.; Jeewandara, C.; Ranasinghe, P. Enhancing immunity in viral infections, with special emphasis on COVID-19: A review. Diabetes Metab. Syndr. 2020, 14, 367–382. [Google Scholar] [CrossRef]
- Shakoor, H.; Feehan, J.; Al Dhaheri, A.S.; Ali, H.I.; Platat, C.; Ismail, L.C.; Apostolopoulos, V.; Stojanovska, L. Immune-boosting role of vitamins D, C, E, zinc, selenium and omega-3 fatty acids: Could they help against COVID-19? Maturitas 2021, 143, 1–9. [Google Scholar] [CrossRef]
- Grant, W.B.; Lahore, H.; McDonnell, S.L.; Baggerly, C.A.; French, C.B.; Aliano, J.L.; Bhattoa, H.P. Evidence that vitamin D supplementation could reduce risk of influenza and COVID-19 infections and deaths. Nutrients 2020, 12, 988. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Martineau, A.R.; Forouhi, N.G. Vitamin D for COVID-19: A case to answer? Lancet Diabetes Endocrinol. 2020, 8, 735–736. [Google Scholar] [CrossRef]
- Jovic, T.H.; Ali, S.R.; Ibrahim, N.; Jessop, Z.M.; Tarassoli, S.P.; Dobbs, T.D.; Holford, P.; Thornton, C.A.; Whitaker, I.S. Could vitamins help in the fight against COVID-19? Nutrients 2020, 12, 2550. [Google Scholar] [CrossRef]
- Alkhatib, A. Antiviral functional foods and exercise lifestyle prevention of Coronavirus. Nutrients 2020, 12, 2633. [Google Scholar] [CrossRef] [PubMed]
- Calder, P.C.; Carr, A.C.; Gombart, A.F.; Eggersdorfer, M. Optimal nutritional status for a well-functioning immune system is an important factor to protect against viral infections. Nutrients 2020, 12, 1181. [Google Scholar] [CrossRef] [Green Version]
- Morais, A.H.A.; Passos, T.S.; Maciel, B.L.L.; da Silva-Maia, J.K. Can probiotics and diet promote beneficial immune modulation and purine control in Coronavirus infection? Nutrients 2020, 12, 1737. [Google Scholar] [CrossRef]
- Baud, D.; Agri, V.D.; Gibson, G.R.; Reid, G.; Giannoni, E. Using probiotics to flatten the curve of coronavirus disease COVID-2019 pandemic. Front. Public Health 2020, 8, 186. [Google Scholar] [CrossRef]
- Jampilek, J.; Kralova, K. Application of nanobioformulations for controlled release and targeted biodistribution of drugs. In Nanobiomaterials: Applications in Drug Delivery; Sharma, A.K., Keservani, R.K., Kesharwani, R.K., Eds.; CRC Press: Warentown, NJ, USA, 2018; pp. 131–208. [Google Scholar]
- Jampilek, J.; Kralova, K. Recent Advances in lipid nanocarriers applicable in the fight against cancer. In Nanoarchitectonics in Biomedicine; Grumezescu, A.M., Ed.; Elsevier: Amsterdam, The Netherlands, 2019; pp. 219–294. [Google Scholar]
- Jampilek, J.; Kralova, K. Nanotechnology based formulations for drug targeting to central nervous system. In Nanoparticulate Drug Delivery Systems; Keservani, R.K., Sharma, A.K., Eds.; Apple Academic Press & CRC Press: Warentown, NJ, USA, 2019; pp. 151–220. [Google Scholar]
- Jampilek, J.; Kralova, K.; Campos, E.V.R.; Fraceto, L.F. Bio-based nanoemulsion formulations applicable in agriculture, medicine and food industry. In Nanobiotechnology in Bioformulations; Prasad, R., Kumar, V., Kumar, M., Choudhary, D.K., Eds.; Springer: Cham, Switzerland, 2019; pp. 33–84. [Google Scholar]
- Pentak, D.; Kozik, V.; Bak, A.; Dybal, P.; Sochanik, A.; Jampilek, J. Methotrexate and cytarabine—Loaded nanocarriers for multidrug cancer therapy. Spectroscopic study. Molecules 2016, 21, 1689. [Google Scholar] [CrossRef] [Green Version]
- Placha, D.; Jampilek, J. Graphenic materials for biomedical applications. Nanomaterials 2019, 9, 1758. [Google Scholar] [CrossRef] [Green Version]
- Jampilek, J.; Kralova, K. Natural biopolymeric nanoformulations for brain drug delivery. In Nanocarriers for Brain Targetting: Principles and Applications; Raj, K., Keservani, A.K., Rajesh, S., Kesharwani, K., Eds.; Apple Academic Press & CRC Press: Warentown, NJ, USA, 2020; pp. 131–203. [Google Scholar]
- Jampilek, J.; Kralova, K. Nanoweapons against tuberculosis. In Nanoformulations in Human Health—Challenges and Approaches; Talegaonkar, S., Rai., M., Eds.; Springer Nature: Cham, Switzerland, 2020; pp. 469–502. [Google Scholar]
- Jampilek, J.; Kralova, K. Nanoformulations—Valuable tool in therapy of viral diseases attacking humans and animals. In Nanotheranostic—Applications and Limitations; Rai, M., Jamil, B., Eds.; Springer Nature: Cham, Switzerland, 2019; pp. 137–178. [Google Scholar]
- Jampilek, J.; Kralova, K.; Novak, P.; Novak, M. Nanobiotechnology in neurodegenerative diseases. In Nanobiotechnology in Neurodegenerative Diseases; Rai, M., Yadav, A., Eds.; Springer Nature: Cham, Switzerland, 2019; pp. 65–138. [Google Scholar]
- Jampilek, J.; Kos, J.; Kralova, K. Potential of nanomaterial applications in dietary supplements and foods for special medical purposes. Nanomaterials 2019, 9, 296. [Google Scholar] [CrossRef] [Green Version]
- Human Regulatory—Nanomedicines. European Medicines Agency. 2020. Available online: https://www.ema.europa.eu/en/human-regulatory/research-development/scientific-guidelines/multidisciplinary/multidisciplinary-nanomedicines (accessed on 25 October 2020).
- FDA’s Approach to Regulation of Nanotechnology Products. Available online: https://www.fda.gov/science-research/nanotechnology-programs-fda/fdas-approach-regulation-nanotechnology-products (accessed on 25 October 2020).
- Nanotechnology Guidance Documents. Available online: https://www.fda.gov/science-research/nanotechnology-programs-fda/nanotechnology-guidance-documents (accessed on 25 October 2020).
- Som, C.; Schmutz, M.; Borges, O.; Jesus, S.; Borchard, G.; Nguyen, V.; Perale, G.; Casalini, T.; Zinn, M.; Amstutz, V.; et al. Guidelines for Implementing a Safe-by-Design Approach for Medicinal Polymeric Nanocarriers, Empa St. Gallen. 2019. Available online: https://www.empa.ch/documents/56164/10586277/Guidelines/b0f2b20b-29d1-426b-8263-8d031b819c61 (accessed on 25 October 2020).
- Guidelines for Evaluation of Nanopharmaceuticals in India. Department of Biotechnology, Indian Society of Nanomedicine. 2019. Available online: https://www.birac.nic.in/webcontent/1550639649_guidelines_for_evaluation_of_Nanopharmaceuticals_in_India_20_02_2019.pdf (accessed on 25 October 2020).
- Zainal Abidin, H.F.; Hassan, K.H.; Zainol, Z.A. Regulating risk of nanomaterials for workers through soft law approach. Nanoethics 2020, 14, 155–167. [Google Scholar] [CrossRef]
- Souto, E.B.; Silva, G.F.; Dias-Ferreira, J.; Zielinska, A.; Ventura, F.; Durazzo, A.; Lucarini, M.; Novellino, E.; Santini, A. Nanopharmaceutics: Part I—Clinical trials legislation and Good Manufacturing Practices (GMP) of nanotherapeutics in the EU. Pharmaceutics 2020, 12, 146. [Google Scholar] [CrossRef] [Green Version]
- Souto, E.B.; Silva, G.F.; Dias-Ferreira, J.; Zielinska, A.; Ventura, F.; Durazzo, A.; Lucarini, M.; Novellino, E.; Santini, A. Nanopharmaceutics: Part II-production scales and clinically compliant production methods. Nanomaterials 2020, 10, 455. [Google Scholar] [CrossRef] [Green Version]
- Jampilek, J.; Kralova, K. Impact of nanoparticles on toxigenic fungi. In Nanomycotoxicology—Treating Mycotoxins in the Nano Way; Rai, M., Abd-Elsalam, K.A., Eds.; Academic Press & Elsevier: London, UK, 2020; pp. 309–348. [Google Scholar]
- Jampilek, J.; Kralova, K. Nanocomposites: Synergistic nanotools for management mycotoxigenic fungi. In Nanomycotoxicology—Treating Mycotoxins in the Nano Way; Rai, M., Abd-Elsalam, K.A., Eds.; Academic Press & Elsevier: London, UK, 2020; pp. 349–383. [Google Scholar]
- Jampilek, J.; Kralova, K.; Fedor, P. Bioactivity of nanoformulated synthetic and natural insecticides and their impact on the environment. In Nanopesticides—From Research and Development to Mechanisms of Action and Sustainable Use in Agriculture; Fraceto, L.F., de Castro, V.L., Grillo, R., Avila, D., Oliveira, H.C., de Lima, R., Eds.; Springer Nature: Cham, Switzerland, 2020; pp. 165–225. [Google Scholar]
- Su, S.; Kang, P.M. Systemic review of biodegradable nanomaterials in nanomedicine. Nanomaterials 2020, 10, 656. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zielinska, A.; Costa, B.; Ferreira, M.V.; Migueis, D.; Louros, J.M.S.; Durazzo, A.; Lucarini, M.; Eder, P.; Chaud, M.V.; Morsink, M.; et al. Nanotoxicology and nanosafety: Safety-by-design and testing at a glance. Int. J. Environ. Res. Public Health 2020, 17, 4657. [Google Scholar] [CrossRef] [PubMed]
- De Stefano, D.; Carnuccio, R.; Maiuri, M.C. Nanomaterials toxicity and cell death modalities. J. Drug Deliv. 2012, 2012, 167896. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sukhanova, A.; Bozrova, S.; Sokolov, P.; Berestovoy, M.; Karaulov, A.; Nabiev, I. Dependence of nanoparticle toxicity on their physical and chemical properties. Nanoscale Res. Lett. 2018, 13, 44. [Google Scholar] [CrossRef] [Green Version]
- European Food Safety Authority. Guidance on risk assessment of the application of nanoscience and nanotechnologies in the food and feed chain. EFSA J. 2018, 16, 5327. [Google Scholar]
- Jampilek, J.; Kralova, K. Benefits and potential risks of nanotechnology applications in crop protection. In Nanobiotechnology Applications in Plant Protection, Nanotechnology in the Life Sciences; Abd-Elsalam, K.A., Prasad, R., Eds.; Springer International Publishing: Singapore, 2018; pp. 189–246. [Google Scholar]
- Subramani, T.; Ganapathyswamy, H. An overview of liposomal nano-encapsulation techniques and its applications in food and nutraceutical. J. Food Sci. Technol. Mys. 2020, 57, 3545–3555. [Google Scholar] [CrossRef]
- Aswathanarayan, J.B.; Vittal, R.R. Nanoemulsions and their potential applications in food industry. Front. Sustain. Food Syst. 2019, 3, 95. [Google Scholar] [CrossRef] [Green Version]
- Haider, M.; Abdin, S.M.; Kamal, L.; Orive, G. Nanostructured lipid carriers for delivery of chemotherapeutics: A review. Pharmaceutics 2020, 12, 288. [Google Scholar] [CrossRef] [Green Version]
- Zhong, Q.X.; Zhang, L.H. Nanoparticles fabricated from bulk solid lipids: Preparation, properties, and potential food applications. Adv. Colloid Interface Sci. 2019, 273, 102033. [Google Scholar] [CrossRef]
- Assadpour, E.; Jafari, S.M. A systematic review on nanoencapsulation of food bioactive ingredients and nutraceuticals by various nanocarriers. Crit. Rev. Food Sci. Nutr. 2019, 59, 3129–3151. [Google Scholar] [CrossRef]
- Katouzian, I.; Jafari, S.M. Nano-encapsulation as a promising approach for targeted delivery and controlled release of vitamins. Trends Food Sci. Technol. 2016, 53, 34–48. [Google Scholar] [CrossRef]
- Ezhilarasi, P.N.; Karthik, P.; Chhanwal, N.; Anandharamakrishnan, C. Nanoencapsulation techniques for food bioactive components: A review. Food Bioprocess Technol. 2013, 6, 628–647. [Google Scholar] [CrossRef]
- Fathi, M.; Mozafari, M.R.; Mohebbi, M. Nanoencapsulation of food ingredients using lipid based delivery systems. Trends Food Sci. Technol. 2012, 23, 13–27. [Google Scholar] [CrossRef]
- Arenas-Jal, M.; Sune-Negre, J.M.; Garcia-Montoya, E. An overview of microencapsulation in the food industry: Opportunities, challenges, and innovations. Eur. Food Res. Technol. 2020, 246, 1371–1382. [Google Scholar] [CrossRef]
- Zam, W. Microencapsulation: A prospective to protect probiotics. Curr. Nutr. Food Sci. 2020, 16, 891–899. [Google Scholar] [CrossRef]
- Naidu, K.A. Vitamin C in human health and disease is still a mystery? An overview. Nutr. J. 2003, 2, 7. [Google Scholar] [CrossRef] [Green Version]
- Timoshnikov, V.A.; Kobzeva, T.V.; Polyakov, N.E.; Kontoghiorghes, G.J. Redox interactions of vitamin C and iron: Inhibition of the pro-oxidant activity by deferiprone. Int. J. Mol. Sci. 2020, 21, 3967. [Google Scholar] [CrossRef] [PubMed]
- Nantel, G.; Tontisirin, K. Human Vitamin and Mineral Requirements; FAO & WHO: Rome, Italy, 2002; Available online: http://www.fao.org/3/y2809e/y2809e00.pdf (accessed on 3 October 2020).
- Jiao, Z.; Wang, X.D.; Yin, Y.T.; Xia, J.X. Preparation and evaluation of vitamin C and folic acid-coloaded antioxidant liposomes. Particul. Sci. Technol. 2019, 37, 449–455. [Google Scholar] [CrossRef]
- Parhizkar, E.; Rashedinia, M.; Karimi, M.; Alipour, S. Design and development of vitamin C-encapsulated proliposome with improved in-vitro and ex-vivo antioxidant efficacy. J. Microencapsul. 2018, 35, 301–311. [Google Scholar] [CrossRef] [Green Version]
- Jiao, Z.; Wang, X.D.; Yin, Y.T.; Xia, J.X.; Mei, Y.N. Preparation and evaluation of a chitosan-coated antioxidant liposome containing vitamin C and folic acid. J. Microencapsul. 2018, 35, 272–280. [Google Scholar] [CrossRef]
- Liu, W.L.; Tian, M.M.; Kong, Y.Y.; Lu, J.M.; Li, N.; Han, J.Z. Multilayered vitamin C nanoliposomes by self-assembly of alginate and chitosan: Long-term stability and feasibility application in mandarin juice. LWT Food Sci. Technol. 2017, 75, 608–615. [Google Scholar] [CrossRef]
- Gautam, M.; Santhiya, D. Pectin/PEG food grade hydrogel blend for the targeted oral co-delivery of nutrients. Colloids Surf. A Physicochem. Eng. Asp. 2019, 577, 637–644. [Google Scholar] [CrossRef]
- Salaheldin, T.A.; Regheb, E.M. In-vivo nutritional and toxicological evaluation of nano iron fortified biscuits as food supplement for iron deficient anemia. J. Nanomed. Res. 2016, 3, 00049. [Google Scholar] [CrossRef] [Green Version]
- Charoenngam, N.; Holick, M.F. Immunologic effects of vitamin D on human health and disease. Nutrients 2020, 12, 2097. [Google Scholar] [CrossRef] [PubMed]
- O’Leary, D.; Samman, S. Vitamin B12 in health and disease. Nutrients 2010, 2, 299–316. [Google Scholar] [CrossRef]
- Gröber, U.; Kisters, K.; Schmidt, J. Neuroenhancement with vitamin B12–underestimated neurological significance. Nutrients 2013, 5, 5031–5045. [Google Scholar] [CrossRef] [Green Version]
- Zhu, K.; Chen, X.Y.; Yu, D.; He, Y.; Song, G.L. Preparation and characterisation of a novel hydrogel based on Auricularia polytricha β-glucan and its bio-release property for vitamin B12 delivery. J. Sci. Food Agric. 2018, 98, 2617–2623. [Google Scholar] [CrossRef]
- Liu, G.Y.; Yang, J.Q.; Wang, Y.X.; Liu, X.H.; Guan, L.L.; Chen, L.Y. Protein-lipid composite nanoparticles for the oral delivery of vitamin B12: Impact of protein succinylation on nanoparticle physicochemical and biological properties. Food Hydrocoll. 2019, 92, 189–197. [Google Scholar] [CrossRef]
- Mazzocato, M.C.; Thomazini, M.; Favaro-Trindade, C.S. Improving stability of vitamin B12 (Cyanocobalamin) using microencapsulation by spray chilling technique. Food Res. Int. 2019, 126, 108663. [Google Scholar] [CrossRef]
- Camilli, G.; Tabouret, G.; Quintin, J. The complexity of fungal β-glucan in health and disease: Effects on the mononuclear phagocyte system. Front. Immunol. 2018, 9, 673. [Google Scholar] [CrossRef] [Green Version]
- Stanger, O. Physiology of folic acid in health and disease. Curr. Drug Metab. 2002, 3, 211–223. [Google Scholar] [CrossRef]
- Crnivec, I.G.O.; Istenic, K.; Skrt, M.; Ulrih, N.P. Thermal protection and pH-gated release of folic acid in microparticles and nanoparticles for food fortification. Food Funct. 2020, 11, 1467–1477. [Google Scholar] [CrossRef]
- Acevedo-Fani, A.; Soliva-Fortuny, R.; Martin-Belloso, O. Photo-protection and controlled release of folic acid using edible alginate/chitosan nanolaminates. J. Food Eng. 2016, 229, 72–82. [Google Scholar] [CrossRef]
- Perez-Masia, R.; Lopez-Nicolas, R.; Periago, M.J.; Ros, G.; Lagaron, J.M.; Lopez-Rubio, A. Encapsulation of folic acid in food hydrocolloids through nanospray drying and electrospraying for nutraceutical applications. Food Chem. 2015, 168, 124–133. [Google Scholar] [CrossRef] [Green Version]
- do Evangelho, J.A.; Crizel, R.L.; Chaves, F.C.; Prietto, L.; Pinto, V.Z.; de Miranda, M.Z.; Dias, A.R.G.; Zavareze, E.D. Thermal and irradiation resistance of folic acid encapsulated in zein ultrafine fibers or nanocapsules produced by electrospinning and electrospraying. Food Res. Int. 2019, 124, 137–146. [Google Scholar] [CrossRef] [Green Version]
- Assadpour, E.; Maghsoudlou, Y.; Jafari, S.M.; Ghorbani, M.; Aalami, M. Evaluation of folic acid nano-encapsulation by double emulsions. Food Bioprocess Technol. 2016, 9, 2024–2032. [Google Scholar] [CrossRef]
- Ochnio, M.E.; Martinez, J.H.; Allievi, M.C.; Palavecino, M.; Martinez, K.D.; Perez, O.E. Proteins as nano-carriers for bioactive compounds. The case of 7S and 11S soy globulins and folic acid complexation. Polymers 2018, 10, 149. [Google Scholar] [CrossRef] [Green Version]
- Zema, P.; Pilosof, A.M.R. On the binding of folic acid to food proteins performing as vitamin micro/nanocarriers. Food Hydrocoll. 2018, 79, 509–517. [Google Scholar] [CrossRef]
- Perez-Esteve, E.; Ruiz-Rico, M.; de la Torre, C.; Villaescusa, L.A.; Sancenon, F.; Marcos, M.D.; Amoros, P.; Martinez-Manez, R.; Barat, J.M. Encapsulation of folic acid in different silica porous supports: A comparative study. Food Chem. 2016, 196, 66–75. [Google Scholar] [CrossRef]
- Perez-Esteve, E.; Fuentes, A.; Coll, C.; Acosta, C.; Bernardos, A.; Amoros, P.; Marcos, M.D.; Sancenon, F.; Martinez-Manez, R.; Barat, J.M. Modulation of folic acid bioaccessibility by encapsulation in pH-responsive gated mesoporous silica particles. Micropor. Mesopor. Mat. 2015, 202, 124–132. [Google Scholar] [CrossRef]
- Perez-Esteve, E.; Ruiz-Rico, M.; Fuentes, A.; Marcos, M.D.; Sancenon, F.; Martinez-Manez, R.; Barat, J.M. Enrichment of stirred yogurts with folic acid encapsulated in pH-responsive mesoporous silica particles: Bioaccessibility modulation and physico-chemical characterization. LWT Food Sci. Technol. 2016, 72, 351–360. [Google Scholar] [CrossRef]
- Ruiz-Rico, M.; Perez-Esteve, E.; Lerma-Garcia, M.J.; Marcos, M.D.; Martinez-Manez, R.; Barat, J.M. Protection of folic acid through encapsulation in mesoporous silica particles included in fruit juices. Food Chem. 2017, 218, 471–478. [Google Scholar] [CrossRef]
- Pagano, C.; Tiralti, M.C.; Perioli, L. Nanostructured hybrids for the improvement of folic acid biopharmaceutical properties. J. Pharm. Pharmacol. 2016, 68, 1384–1395. [Google Scholar] [CrossRef]
- Lips, O. Vitamin D physiology. Prog. Biophys. Mol. Biol. 2006, 92, 4–8. [Google Scholar] [CrossRef]
- Maurya, V.K.; Bashir, K.; Aggarwal, M. Vitamin D microencapsulation and fortification: Trends and technologies. J. Steroid Biochem. Mol. Biol. 2020, 196, 105489. [Google Scholar] [CrossRef]
- Guttoff, M.; Saberi, A.H.; McClements, D.J. Formation of vitamin D nanoemulsion-based delivery systems by spontaneous emulsification: Factors affecting particle size and stability. Food Chem. 2015, 171, 117–122. [Google Scholar] [CrossRef]
- Kadappan, A.S.; Guo, C.; Gumus, C.E.; Bessey, A.; Wood, R.J.; McClements, D.J.; Liu, Z.H. The efficacy of nanoemulsion-based delivery to improve vitamin D absorption: Comparison of in vitro and in vivo studies. Mol. Nutr. Food Res. 2018, 62, 1700836. [Google Scholar] [CrossRef]
- Teng, Z.; Luo, Y.C.; Wang, Q. Carboxymethyl chitosan-soy protein complex nanoparticles for the encapsulation and controlled release of vitamin D3. Food Chem. 2013, 141, 524–532. [Google Scholar] [CrossRef]
- Li, W.J.; Peng, H.L.; Ning, F.J.; Yao, L.H.; Luo, M.; Zhao, Q.; Zhu, X.M.; Xiong, H. Amphiphilic chitosan derivative-based core-shell micelles: Synthesis, characterisation and properties for sustained release of vitamin D3. Food Chem. 2014, 152, 307–315. [Google Scholar] [CrossRef]
- Xiang, C.Y.; Gao, J.; Ye, H.X.; Ren, G.R.; Ma, X.J.; Xie, H.J.; Fang, S.; Lei, Q.F.; Fang, W.J. Development of ovalbumin-pectin nanocomplexes for vitamin D3 encapsulation: Enhanced storage stability and sustained release in simulated gastrointestinal digestion. Food Hydrocoll. 2020, 106, 105926. [Google Scholar] [CrossRef]
- Hasanvand, E.; Fathi, M.; Bassiri, A.; Javanmard, M.; Abbaszadeh, R. Novel starch based nanocarrier for vitamin D fortification of milk: Production and characterization. Food Bioprod. Process. 2015, 96, 264–277. [Google Scholar] [CrossRef]
- Winuprasith, T.; Khomein, P.; Mitbumrung, W.; Suphantharika, M.; Nitithamyong, A.; McClements, D.J. Encapsulation of vitamin D3 in pickering emulsions stabilized by nanofibrillated mangosteen cellulose: Impact on in vitro digestion and bioaccessibility. Food Hydrocoll. 2018, 83, 153–164. [Google Scholar] [CrossRef]
- Mitbumrung, W.; Suphantharika, M.; McClements, D.J.; Winuprasith, T. Encapsulation of vitamin D3 in Pickering emulsion stabilized by nanofibrillated mangosteen cellulose: Effect of environmental stresses. J. Food Sci. 2019, 84, 3213–3221. [Google Scholar] [CrossRef]
- Tan, Y.B.; Liu, J.N.; Zhou, H.L.; Mundo, J.M.; McClements, D.J. Impact of an indigestible oil phase (mineral oil) on the bioaccessibility of vitamin D3 encapsulated in whey protein-stabilized nanoemulsions. Food Res. Int. 2019, 120, 264–274. [Google Scholar] [CrossRef]
- Ozturk, B.; Argin, S.; Ozilgen, M.; McClements, D.J. Nanoemulsion delivery systems for oil-soluble vitamins: Influence of carrier oil type on lipid digestion and vitamin D3 bioaccessibility. Food Chem. 2015, 187, 499–506. [Google Scholar] [CrossRef]
- Maurya, V.K.; Aggarwal, M. Fabrication of nano-structured lipid carrier for encapsulation of vitamin D3 for fortification of ‘Lassi’; A milk based beverage. J. Steroid Biochem. Mol. Biol. 2019, 193, 105429. [Google Scholar] [CrossRef]
- Dalmoro, A.; Bochicchio, S.; Lamberti, G.; Bertoncin, P.; Janssens, B.; Barba, A.A. Micronutrients encapsulation in enhanced nanoliposomal carriers by a novel preparative technology. RSC Adv. 2019, 9, 19800–19812. [Google Scholar] [CrossRef] [Green Version]
- Mohammadi, M.; Pezeshki, A.; Abbasi, M.M.; Ghanbarzadeh, B.; Hamishehkar, H. Vitamin D3-loaded nanostructured lipid carriers as a potential approach for fortifying food beverages; in vitro and in vivo evaluation. Adv. Pharm. Bull. 2017, 7, 61–71. [Google Scholar] [CrossRef] [Green Version]
- Berino, R.P.; Baez, G.D.; Ballerini, G.A.; Llopart, E.E.; Busti, P.A.; Moro, A.; Delorenzi, N.J. Interaction of vitamin D3 with beta-lactoglobulin at high vitamin/protein ratios: Characterization of size and surface charge of nanoparticles. Food Hydrocoll. 2019, 90, 182–188. [Google Scholar] [CrossRef]
- Moeller, H.; Martin, D.; Schrader, K.; Hoffmann, W.; Lorenzen, P.C. Spray- or freeze-drying of casein micelles loaded with Vitamin D2: Studies on storage stability and in vitro digestibility. LWT Food Sci. Technol. 2018, 97, 87–93. [Google Scholar] [CrossRef]
- Loewen, A.; Chan, B.; Li-Chan, E.C.Y. Optimization of vitamins A and D3 loading in re-assembled casein micelles and effect of loading on stability of vitamin D3 during storage. Food Chem. 2018, 240, 472–481. [Google Scholar] [CrossRef]
- Cohen, Y.; Levi, M.; Lesmes, U.; Margier, M.; Reboul, E.; Livney, Y.D. Re-assembled casein micelles improve in vitro bioavailability of vitamin D in a Caco-2 cell model. Food Funct. 2017, 8, 2133–2141. [Google Scholar] [CrossRef]
- David, S.; Livney, Y.D. Potato protein based nanovehicles for health promoting hydrophobic bioactives in clear beverages. Food Hydrocoll. 2016, 57, 229–235. [Google Scholar] [CrossRef]
- Walia, N.; Chen, L.Y. Pea protein based vitamin D nanoemulsions: Fabrication, stability and in vitro study using Caco-2 cells. Food Chem. 2020, 305, 125475. [Google Scholar] [CrossRef] [PubMed]
- Jiang, S.S.; Yildiz, G.; Ding, J.Z.; Andrade, J.; Rababahb, T.M.; Almajwalc, A.; Abulmeatyc, M.M.; Feng, H. Pea protein nanoemulsion and nanocomplex as carriers for protection of cholecalciferol (vitamin D3). Food Bioprocess Technol. 2019, 12, 1031–1040. [Google Scholar] [CrossRef]
- Almajwal, A.M.; Abulmeaty, M.M.A.; Feng, H.; Alruwaili, N.W.; Dominguez-Uscanga, A.; Andrade, J.E.; Razak, S.; ElSadek, M.F. Stabilization of vitamin D in pea protein isolate nanoemulsions increases its bioefficacy in rats. Nutrients 2019, 11, 75. [Google Scholar] [CrossRef] [Green Version]
- Salvia-Trujillo, L.; Fumiaki, B.; Park, Y.; McClements, D.J. The influence of lipid droplet size on the oral bioavailability of vitamin D2 encapsulated in emulsions: An in vitro and in vivo study. Food Funct. 2017, 8, 767–777. [Google Scholar] [CrossRef] [PubMed]
- Mehmood, T.; Ahmed, A. Tween 80 and soya-lecithin-based food-grade nanoemulsions for the effective delivery of vitamin D. Langmuir 2020, 36, 2886–2892. [Google Scholar] [CrossRef] [PubMed]
- Mehmood, T.; Ahmed, A.; Ahmed, Z.; Ahmad, M.S. Optimization of soya lecithin and Tween 80 based novel vitamin D nanoemulsions prepared by ultrasonication using response surface methodology. Food Chem. 2019, 289, 664–670. [Google Scholar] [CrossRef] [PubMed]
- Gahruie, H.H.; Niakousari, M.; Parastouei, K.; Mokhtarian, M.; Es, I.; Khaneghah, A.M. Co-encapsulation of vitamin D3 and saffron petals’ bioactive compounds in nanoemulsions: Effects of emulsifier and homogenizer types. J. Food Process. Preserv. 2020, 44, 14629. [Google Scholar] [CrossRef]
- Zhou, H.L.; Tan, Y.B.; Lv, S.S.; Liu, J.N.; Mundo, J.L.M.; Bai, L.; Rojas, O.J.; McClements, D.J. Nanochitin-stabilized pickering emulsions: Influence of nanochitin on lipid digestibility and vitamin bioaccessibility. Food Hydrocoll. 2020, 106, 105878. [Google Scholar] [CrossRef]
- Golfomitsou, I.; Mitsou, E.; Xenakis, A.; Papadimitriou, V. Development of food grade O/W nanoemulsions as carriers of vitamin D for the fortification of emulsion based food matrices: A structural and activity study. J. Mol. Liq. 2018, 268, 734–742. [Google Scholar] [CrossRef]
- Otani, H.; Kihara, Y.; Park, M. The immunoenhancing property of a dietary casein phosphopeptide preparation in mice. Food Agric. Immunol. 2000, 12, 165–173. [Google Scholar] [CrossRef] [Green Version]
- Rizvi, S.; Raza, S.T.; Ahmed, F.; Ahmad, A.; Abbas, S.; Mahdi, F. The role of vitamin E in human health and some diseases. Sultan Qaboos Univ. Med. J. 2014, 14, e157–e165. [Google Scholar]
- Dietary Supplement Fact Sheets: Vitamin E, Office of Dietary Supplements, NIH, USA. Available online: https://ods.od.nih.gov/factsheets/VitaminE-HealthProfessional/ (accessed on 3 October 2020).
- Parthasarathi, S.; Anandharamakrishnan, C. Enhancement of oral bioavailability of vitamin E by spray-freeze drying of whey protein microcapsules. Food Bioprod. Process. 2016, 100, 469–476. [Google Scholar]
- Jaberi, N.; Anarjan, N.; Jafarizadeh-Malmiri, H. Optimization the formulation parameters in preparation of α-tocopherol nanodispersions using low-energy solvent displacement technique. Int. J. Vitam. Nutr. Res. 2020, 90, 5–16. [Google Scholar] [CrossRef]
- Hategekirnana, J.; Masamba, K.G.; Ma, J.G.; Zhong, F. Encapsulation of vitamin E: Effect of physicochemical properties of wall material on retention and stability. Carbohydr. Polym. 2015, 124, 172–179. [Google Scholar] [CrossRef]
- Xia, S.Q.; Tan, C.; Xue, J.; Lou, X.W.; Zhang, X.M.; Feng, B.A. Chitosan/tripolyphosphate-nanoliposomes core-shell nanocomplexes as vitamin E carriers: Shelf-life and thermal properties. Int. J. Food Sci. Technol. 2014, 49, 1367–1374. [Google Scholar] [CrossRef]
- Huang, Z.G.; Brennan, C.S.; Zhao, H.; Liu, J.F.; Guan, W.Q.; Mohan, M.S.; Stipkovits, L.; Zheng, H.T.; Kulasiri, D. Fabrication and assessment of milk phospholipid-complexed antioxidant phytosomes with vitamin C and E: A comparison with liposomes. Food Chem. 2020, 324, 126837. [Google Scholar] [CrossRef]
- Saratale, R.G.; Lee, H.S.; Koo, Y.E.; Saratale, G.D.; Kim, Y.J.; Imm, J.Y.; Park, Y. Absorption kinetics of vitamin E nanoemulsion and green tea microstructures by intestinal in situ single perfusion technique in rats. Food Res. Int. 2018, 106, 149–155. [Google Scholar] [CrossRef]
- Hategekimana, J.; Chamba, M.V.M.; Shoemaker, C.F.; Majeed, H.; Zhong, F. Vitamin E nanoemulsions by emulsion phase inversion: Effect of environmental stress and long-term storage on stability and degradation in different carrier oil types. Colloids Surf. A Physicochem. Eng. Asp. 2015, 483, 70–80. [Google Scholar] [CrossRef]
- Saxena, V.; Hasan, A.; Sharma, S.; Pandey, L.M. Edible oil nanoemulsion: An organic nanoantibiotic as a potential biomolecule delivery vehicle. Int. J. Polym. Mater. Polym. Biomater. 2018, 67, 410–419. [Google Scholar] [CrossRef]
- He, J.B.; Shi, H.; Huang, S.S.; Han, L.J.; Zhang, W.N.; Zhong, Q.X. Core-shell nanoencapsulation of α-tocopherol by blending sodium oleate and rebaudioside A: Preparation, characterization, and antioxidant activity. Molecules 2018, 23, 3183. [Google Scholar] [CrossRef] [Green Version]
- Parthasarathi, S.; Muthukumar, S.P.; Anandharamakrishnan, C. The influence of droplet size on the stability, in vivo digestion, and oral bioavailability of vitamin E emulsions. Food Funct. 2016, 7, 2294–2302. [Google Scholar] [CrossRef]
- Lv, S.S.; Gu, J.Y.; Zhang, R.J.; Zhang, Y.H.; Tan, H.Y.; McClements, D.J. Vitamin E encapsulation in plant-based nanoemulsions fabricated using dual-channel microfluidization: Formation, stability, and bioaccessibility. J. Agric. Food Chem. 2018, 66, 10532–10542. [Google Scholar] [CrossRef]
- Ozturk, B.; Argin, S.; Ozilgen, M.; McClements, D.J. Formation and stabilization of nanoemulsion-based vitamin E delivery systems using natural surfactants: Quillaja saponin and lecithin. J. Food Eng. 2014, 142, 57–63. [Google Scholar] [CrossRef]
- Fang, Z.; Wusigale; Bao, H.Y.; Ni, Y.Z.; Choijilsuren, N.; Liang, L. Partition and digestive stability of α-tocopherol and resveratrol/naringenin in whey protein isolate emulsions. Int. Dairy J. 2019, 93, 116–123. [Google Scholar] [CrossRef]
- Schroder, A.; Sprakel, J.; Schroen, K.; Berton-Carabin, C.C. Chemical stability of α-tocopherol in colloidal lipid particles with various morphologies. Eur. J. Lipid Sci. Technol. 2020, 122, 2000012. [Google Scholar] [CrossRef] [Green Version]
- Sharif, H.R.; Goff, H.D.; Majeed, H.; Liu, F.; Nsor-Atindana, J.; Haider, J.; Liang, R.; Zhong, F. Physicochemical stability of β-carotene and α-tocopherol enriched nanoemulsions: Influence of carrier oil, emulsifier and antioxidant. Colloids Surf. A Physicochem. Eng. Asp. 2017, 529, 550–559. [Google Scholar] [CrossRef]
- Liu, Y.Q.; Hou, Z.Q.; Yang, J.; Gao, Y.X. Effects of antioxidants on the stability of β-carotene in O/W emulsions stabilized by gum arabic. J. Food Sci. Technol. Mys. 2015, 52, 3300–3311. [Google Scholar] [CrossRef] [Green Version]
- Kaur, K.; Kaur, J.; Kumar, R.; Mehta, S.K. Formulation and physiochemical study of α-tocopherol based oil in water nanoemulsion stabilized with non-toxic, biodegradable surfactant: Sodium stearoyl lactate. Ultrason. Sonochem. 2017, 38, 570–578. [Google Scholar] [CrossRef]
- Ramos, O.L.; Pereira, R.N.; Martins, A.; Rodrigues, R.; Fucinos, C.; Teixeira, J.A.; Pastrana, L.; Malcata, F.X.; Vicente, A.A. Design of whey protein nanostructures for incorporation and release of nutraceutical compounds in food. Crit. Rev. Food Sci. Nutr. 2017, 57, 1377–1393. [Google Scholar] [CrossRef] [Green Version]
- Martin, M.; Kopaliani, I.; Jannasch, A.; Mund, C.; Todorov, V.; Henle, T.; Deussen, A. Antihypertensive and cardioprotective effects of the dipeptide isoleucine–tryptophan and whey protein hydrolysate. Acta Physiol. 2015, 215, 167–176. [Google Scholar] [CrossRef]
- Corrochano, A.R.; Buckin, V.; Kelly, P.M.; Giblin, L. Whey proteins as antioxidants and promoters of cellular antioxidant pathways. J. Dairy Sci. 2018, 101, 4747–4761. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Abbaspour, N.; Hurrell, R.; Kelishadi, R. Review on iron and its importance for human health. J. Res. Med. Sci. 2014, 19, 164–174. [Google Scholar] [PubMed]
- Tsykhanovska, I.; Evlash, V.; Oleksandrov, O.; Gontar, T. Mechanism of fat-binding and fat-contenting of the nanoparticles of a food supplement on the basis of double oxide of two- and trivalent iron. Ukr. Food J. 2018, 7, 702–715. [Google Scholar] [CrossRef]
- Kruhlova, O.; Yevlash, T.; Evlash, V.; Tsykhanovska, I.; Potapov, V. Comprehensive analysis of food production efficiency using nanoparticles of nutritional supplements on the basis of oxides of two and three valence iron “Magnetofood”. Ukr. Food J. 2019, 8, 400–416. [Google Scholar] [CrossRef]
- Zimmermann, M.B.; Hilty, F.M. Nanocompounds of iron and zinc: Their potential in nutrition. Nanoscale 2011, 3, 2390–2398. [Google Scholar] [CrossRef]
- Rayman, M.P. Selenium and human health. Lancet 2012, 379, 1256–1268. [Google Scholar] [CrossRef]
- Michalke, B. Selenium. In Molecular and Integrative Toxicology; Springer: Cham, Switzerland, 2018. [Google Scholar]
- Gangadoo, S.; Bauer, B.W.; Bajagai, Y.S.; Van, T.T.H.; Moore, R.J.; Stanley, D. In vitro growth of gut microbiota with selenium nanoparticles. Anim. Nutr. 2019, 5, 424–431. [Google Scholar] [CrossRef]
- Mates, I.; Antoniac, I.; Laslo, V.; Vicas, S.; Brocks, M.; Fritea, L.; Milea, C.; Mohan, A.; Cavalu, S. Selenium nanoparticles: Production, characterization and possible applications in biomedicine and food science. Sci. Bull. B Chem. Mater. Sci. UPB 2019, 81, 205–216. [Google Scholar]
- Martinez, F.G.; Barrientos, M.E.C.; Mozzi, F.; Pescuma, M. Survival of selenium-enriched lactic acid bacteria in a fermented drink under storage and simulated gastro-intestinal digestion. Food Res. Int. 2019, 123, 115–124. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.W.; Yue, L.; Jiang, Q.X.; Xia, W.S. Effect of chitosan with different molecular weight on the stability, antioxidant and anticancer activities of well-dispersed selenium nanoparticles. IET Nanobiotechnol. 2019, 13, 30–35. [Google Scholar] [CrossRef] [PubMed]
- Bai, K.K.; Hong, B.H.; Huang, W.W.; He, J.L. Selenium-nanoparticles-loaded chitosan/chitooligosaccharide microparticles and their antioxidant potential: A chemical and in vivo investigation. Pharmaceutics 2020, 12, 43. [Google Scholar] [CrossRef] [Green Version]
- Bai, K.K.; Hong, B.H.; Tan, R.; He, J.L.; Hong, Z. Alcohol-induced gastric mucosal injury in rats: Rapid preparation, oral delivery, and gastroprotective potential of selenium nanoparticles. Int. J. Nanomed. 2020, 15, 1187–1203. [Google Scholar] [CrossRef] [Green Version]
- Qiu, W.Y.; Wang, Y.Y.; Wang, M.; Yan, J.K. Construction, stability, and enhanced antioxidant activity of pectin-decorated selenium nanoparticles. Colloids Surf. B Biointerfaces 2018, 170, 692–700. [Google Scholar] [CrossRef]
- Wu, Y.; Liu, H.; Li, Z.; Huang, D.Y.; Nong, L.Z.; Ning, Z.X.; Hu, Z.Z.; Xu, C.P.; Yan, J.K. Pectin-decorated selenium nanoparticles as a nanocarrier of curcumin to achieve enhanced physicochemical and biological properties. IET Nanobiotechnol. 2019, 13, 880–886. [Google Scholar] [CrossRef]
- Tang, H.Y.; Huang, Q.; Wang, Y.L.; Yang, X.Q.; Su, D.X.; He, S.; Tan, J.C.; Zeng, Q.Z.; Yuan, Y. Development, structure characterization and stability of food grade selenium nanoparticles stabilized by tilapia polypeptides. J. Food Eng. 2020, 275, 109878. [Google Scholar] [CrossRef]
- Roohani, N.; Hurrell, R.; Kelishadi, R.; Schulin, R. Zinc and its importance for human health: An integrative review. J. Res. Med. Sci. 2013, 18, 144–157. [Google Scholar]
- Livingstone, X. Zinc: Physiology, deficiency, and parenteral nutrition. Nutr. Clin. Pract. 2015, 30, 371–382. [Google Scholar] [CrossRef]
- Go, M.R.; Yu, J.; Bae, S.H.; Kim, H.J.; Choi, S.J. Effects of interactions between ZnO nanoparticles and saccharides on biological responses. Int. J. Mol. Sci. 2018, 19, 486. [Google Scholar]
- Yu, J.; Kim, H.J.; Go, M.R.; Bae, S.H.; Choi, S.J. ZnO interactions with biomatrices: Effect of particle size on ZnO-protein corona. Nanomaterials 2017, 7, 377. [Google Scholar] [CrossRef] [Green Version]
- Ebrahiminezhad, A.; Moeeni, F.; Taghizadeh, S.M.; Seifan, M.; Bautista, C.; Novin, D.; Ghasemi, Y.; Berenjian, A. Xanthan gum capped ZnO microstars as a promising dietary zinc supplementation. Foods 2019, 8, 88. [Google Scholar] [CrossRef] [Green Version]
- Swain, P.S.; Rao, S.B.N.; Rajendran, D.; Dominic, G.; Selvaraju, S. Nano zinc, an alternative to conventional zinc as animal feed supplement: A review. Anim. Nutr. 2016, 2, 134–141. [Google Scholar] [CrossRef]
- Hassan, M.A.; El-Nekeety, A.A.; Abdel-Aziem, S.H.; Hassan, N.S.; Abdel-Wahhab, M.A. Zinc citrate incorporation with whey protein nanoparticles alleviate the oxidative stress complication and modulate gene expression in the liver of rats. Food Chem. Toxicol. 2019, 125, 439–451. [Google Scholar] [CrossRef]
- Lamas, B.; Breyner, N.M.; Houdeau, E. Impacts of foodborne inorganic nanoparticles on the gut microbiota-immune axis: Potential consequences for host health. Part. Fibre Toxicol. 2020, 17, 19. [Google Scholar] [CrossRef]
- Senapati, V.A.; Gupta, G.S.; Pandey, A.K.; Shanker, R.; Dhawan, A.; Kumar, A. Zinc oxide nanoparticle induced age dependent immunotoxicity in BALB/c mice. Toxicol. Res. 2017, 6, 342–352. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Akal, C. Benefits of whey proteins on human health. In Dairy in Human Health and Disease Across the Lifespan; Watson, R.R., Collier, R.J., Preedy, V.R., Eds.; Academic Press: Cambridge, MA, USA; Elsevier: Amsterdam, The Netherlands, 2017; pp. 363–372. [Google Scholar]
- Yamaguchi, M. Carotenoids: Food Sources, Production and Health Benefits; NOVA Science Publishers: Hauppauge, NY, USA, 2013. [Google Scholar]
- Eggersdorfer, M.; Wyss, A. Carotenoids in human nutrition and health. Arch. Biochem. Biophys. 2018, 15, 18–26. [Google Scholar] [CrossRef]
- Tan, C.; Feng, B.; Zhang, X.M.; Xia, W.S.; Xia, S.Q. Biopolymer-coated liposomes by electrostatic adsorption of chitosan (chitosomes) as novel delivery systems for carotenoids. Food Hydrocoll. 2016, 52, 774–784. [Google Scholar] [CrossRef]
- Rehman, A.; Tong, Q.Y.; Jafari, S.M.; Assadpour, E.; Shehzad, Q.; Aadil, R.M.; Iqbal, M.W.; Rashed, M.M.A.; Mushtaq, B.S.; Ashraf, W. Carotenoid-loaded nanocarriers: A comprehensive review. Adv. Colloid. Interface Sci. 2020, 275, 102048. [Google Scholar] [CrossRef]
- Choi, S.J.; McClements, D.J. Nanoemulsions as delivery systems for lipophilic nutraceuticals: Strategies for improving their formulation, stability, functionality and bioavailability. Food Sci. Biotechnol. 2020, 29, 149–168. [Google Scholar] [CrossRef]
- Nazemiyeh, E.; Eskandani, M.; Sheikhloie, H.; Nazemiyeh, H. Formulation and physicochemical characterization of lycopene-loaded solid lipid nanoparticles. Adv. Pharm. Bull. 2016, 6, 235–241. [Google Scholar] [CrossRef] [Green Version]
- de Campo, C.; Assis, R.Q.; da Silva, M.M.; Costa, T.M.H.; Paese, K.; Guterres, S.S.; Rios, A.D.; Floresa, S.H. Incorporation of zeaxanthin nanoparticles in yogurt: Influence on physicochemical properties, carotenoid stability and sensory analysis. Food Chem. 2019, 301, 125230. [Google Scholar] [CrossRef]
- Saravana, P.S.; Shanmugapriya, K.; Gereniu, C.R.N.; Chae, S.J.; Kang, H.W.; Woo, H.C.; Chun, B.S. Ultrasound-mediated fucoxanthin rich oil nanoemulsions stabilized by κ-carrageenan: Process optimization, bio-accessibility and cytotoxicity. Ultrason. Sonochem. 2019, 55, 105–116. [Google Scholar] [CrossRef]
- Liu, X.J.; Zhang, R.J.; McClements, D.J.; Li, F.; Liu, H.; Cao, Y.; Xiao, H. Nanoemulsion-based delivery systems for nutraceuticals: Influence of long-chain triglyceride (LCT) type on in vitro digestion and astaxanthin bioaccessibility. Food Biophys. 2018, 13, 412–421. [Google Scholar] [CrossRef]
- Shen, X.; Fang, T.Q.; Zheng, J.; Guo, M.R. Physicochemical properties and cellular uptake of astaxanthin-loaded emulsions. Molecules 2019, 24, 727. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, X.J.; McClements, D.J.; Cao, Y.; Xiao, H. Chemical and physical stability of astaxanthin-enriched emulsion-based delivery systems. Food Biophys. 2016, 11, 302–310. [Google Scholar] [CrossRef]
- Zhang, Z.P.; Zhang, R.J.; McClements, D.J. Encapsulation of β-carotene in alginate-based hydrogel beads: Impact on physicochemical stability and bioaccessibility. Food Hydrocoll. 2016, 61, 1–10. [Google Scholar] [CrossRef] [Green Version]
- Liu, F.G.; Ma, C.C.; Zhang, R.J.; Gao, Y.X.; McClements, D.J. Controlling the potential gastrointestinal fate of β-carotene emulsions using interfacial engineering: Impact of coating lipid droplets with polyphenol-protein-carbohydrate conjugate. Food Chem. 2017, 221, 395–403. [Google Scholar] [CrossRef]
- Mao, L.K.; Wang, D.; Liu, F.G.; Gao, Y.X. Emulsion design for the delivery of β-carotene in complex food systems. Crit. Rev. Food Sci. Nutr. 2018, 58, 770–784. [Google Scholar] [CrossRef]
- Moeller, H.; Martin, D.; Schrader, K.; Hoffmann, W.; Lorenzen, P.C. Native casein micelles as nanocarriers for β-carotene: pH-and temperature-induced opening of the micellar structure. Int. J. Food Sci. Technol. 2017, 52, 1122–1130. [Google Scholar] [CrossRef]
- Zhang, J.P.; Zhang, X.X.; Wang, X.Y.; Huang, Y.; Yang, B.B.; Pan, X.; Wu, C.B. The influence of maltodextrin on the physicochemical properties and stabilization of beta-carotene emulsions. AAPS PharmSciTech 2017, 18, 821–828. [Google Scholar] [CrossRef]
- Gu, L.P.; Su, Y.J.; Zhang, M.Q.; Chang, C.H.; Li, J.H.; McClements, D.J.; Yang, Y.J. Protection of β-carotene from chemical degradation in emulsion-based delivery systems using antioxidant interfacial complexes: Catechin-egg white protein conjugates. Food Res. Int. 2017, 96, 84–93. [Google Scholar] [CrossRef]
- Salvia-Trujillo, L.; McClements, D.J. Improvement of β-carotene bioaccessibility from dietary supplements using excipient nanoemulsions. J. Agric. Food Chem. 2016, 64, 4639–4647. [Google Scholar] [CrossRef]
- Liu, X.; Bi, J.F.; Xiao, H.; McClements, D.J. Lipid digestion products on bioaccessibility of carotenoids and phenolics from mangoes. J. Food Sci. 2016, 81, N754–N761. [Google Scholar] [CrossRef]
- Li, Q.; Li, T.; Liu, C.M.; Dai, T.T.; Zhang, R.J.; Zhang, Z.P.; McClements, D.J. Enhancement of carotenoid bioaccessibility from tomatoes using excipient emulsions: Influence of particle size. Food Biophys. 2017, 12, 172–185. [Google Scholar] [CrossRef]
- Mehrad, B.; Ravanfar, R.; Licker, J.; Regenstein, J.M.; Abbaspourrad, A. Enhancing the physicochemical stability of β-carotene solid lipid nanoparticle (SLNP) using whey isolate. Food Res. Int. 2018, 105, 962–969. [Google Scholar] [CrossRef]
- Molina, C.V.; Lima, J.G.; Moraes, I.C.F.; Pinho, S.C. Physicochemical characterization and sensory evaluation of yogurts incorporated with beta-carotene-loaded solid lipid microparticles stabilized with hydrolyzed soy protein isolate. Food Sci. Biotechnol. 2019, 28, 59–66. [Google Scholar] [CrossRef]
- Hernandez-Camacho, J.D.; Bernier, M.; Lopez-Lluch, G.; Navas, P. Coenzyme Q10 supplementation in aging and disease. Front. Physiol. 2018, 9, 44. [Google Scholar] [CrossRef] [Green Version]
- Saini, R. Coenzyme Q10: The essential nutrient. J. Pharm. Bioallied. Sci. 2011, 3, 466–467. [Google Scholar] [CrossRef]
- Martelli, A.; Testai, L.; Colletti, A.; Cicero, A.F.G. Coenzyme Q10: Clinical applications in cardiovascular diseases. Antioxidants 2020, 9, 341. [Google Scholar] [CrossRef] [Green Version]
- Zaki, N.M. Strategies for oral delivery and mitochondrial targeting of CoQ10. Drug Deliv. 2016, 23, 1868–1881. [Google Scholar] [CrossRef] [Green Version]
- Kumar, S.; Rao, R.; Kumar, A.; Mahant, S.; Nanda, S. Novel carriers for coenzyme Q10 delivery. Curr. Drug Deliv. 2016, 13, 1184–1204. [Google Scholar] [CrossRef]
- Uekaji, Y.; Terao, K. Bioavailability enhancement of hydrophobic nutraceuticals using γ-cyclodextrin. J. Incl. Phenom. Macrocycl. Chem. 2019, 93, 3–15. [Google Scholar] [CrossRef]
- Wei, Y.; Yang, S.F.; Zhang, L.; Dai, L.; Tai, K.D.; Liu, J.F.; Mao, L.K.; Yuan, F.; Gao, Y.X.; Mackie, A. Fabrication, characterization and in vitro digestion of food grade complex nanoparticles for co-delivery of resveratrol and coenzyme Q10. Food Hydrocoll. 2020, 105, 105791. [Google Scholar] [CrossRef]
- Chen, S.; Zhang, Y.H.; Qing, J.; Han, Y.H.; McClements, D.J.; Gao, Y.X. Core-shell nanoparticles for co-encapsulation of coenzyme Q10 and piperine: Surface engineering of hydrogel shell around protein core. Food Hydrocoll. 2020, 103, 105651. [Google Scholar] [CrossRef]
- Alavi, S.; Akhlaghi, S.; Dadashzadeh, S.; Haeri, A. Green formulation of triglyceride/phospholipid-based nanocarriers as a novel vehicle for oral coenzyme Q10 delivery. J. Food Sci. 2019, 84, 2572–2583. [Google Scholar] [CrossRef]
- Vatsa, P.; Sanchez, L.; Clement, C.; Baillieul, F.; Dorey, S. Rhamnolipid biosurfactants as new players in animal and plant defense against microbes. Int. J. Mol. Sci. 2010, 11, 5095–5108. [Google Scholar] [CrossRef] [Green Version]
- Ramirez-Garza, S.L.; Laveriano-Santos, E.P.; Marhuenda-Munoz, M.; Storniolo, C.E.; Tresserra-Rimbau, A.; Vallverdu-Queralt, A.; Lamuela-Raventos, R.M. Health effects of resveratrol: Results from human intervention trials. Nutrients 2018, 10, 1892. [Google Scholar] [CrossRef] [Green Version]
- Pannu, N.; Bhatnagar, A. Resveratrol: From enhanced biosynthesis and bioavailability to multitargeting chronic diseases. Biomed. Pharmacother. 2019, 109, 2237–2251. [Google Scholar] [CrossRef]
- De Amicis, F.; Chimento, A.; Montalto, F.I.; Casaburi, I.; Sirianni, R.; Pezzi, V. Steroid receptor signallings as targets for resveratrol actions in breast and prostate cancer. Int. J. Mol. Sci. 2019, 20, 1087. [Google Scholar] [CrossRef] [Green Version]
- Wei, Y.; Li, C.; Zhang, L.; Dai, L.; Yang, S.F.; Liu, J.F.; Mao, L.K.; Yuan, F.; Gao, Y.X. Influence of calcium ions on the stability, microstructure and in vitro digestion fate of zein-propylene glycol alginate-tea saponin ternary complex particles for the delivery of resveratrol. Food Hydrocoll. 2020, 106, 105886. [Google Scholar] [CrossRef]
- Huang, X.L.; Liu, Y.; Zou, Y.; Liang, X.; Peng, Y.Q.; McClements, D.J.; Hu, K. Encapsulation of resveratrol in zein/pectin core-shell nanoparticles: Stability, bioaccessibility, and antioxidant capacity after simulated gastrointestinal digestion. Food Hydrocoll. 2019, 93, 261–269. [Google Scholar] [CrossRef]
- Fan, Y.T.; Zeng, X.X.; Yi, J.; Zhang, Y.Z. Fabrication of pea protein nanoparticles with calcium-induced cross-linking for the stabilization and delivery of antioxidative resveratrol. Int. J. Biol. Macromol. 2020, 152, 189–198. [Google Scholar] [CrossRef]
- Liu, Y.X.; Fan, Y.T.; Gao, L.Y.; Zhang, Y.Z.; Yi, J. Enhanced pH and thermal stability, solubility and antioxidant activity of resveratrol by nanocomplexation with α-lactalbumin. Food Funct. 2018, 9, 4781–4790. [Google Scholar] [CrossRef]
- Xiong, W.F.; Ren, C.; Li, J.; Li, B. Enhancing the photostability and bioaccessibility of resveratrol using ovalbumin-carboxymethylcellulose nanocomplexes and nanoparticles. Food Funct. 2018, 9, 3788–3797. [Google Scholar] [CrossRef]
- Wu, W.H.; Kong, X.Z.; Zhang, C.M.; Hua, Y.F.; Chen, Y.M.; Li, X.F. Fabrication and characterization of resveratrol-loaded gliadin nanoparticles stabilized by gum Arabic and chitosan hydrochloride. LWT Food Sci. Technol. 2020, 129, 109532. [Google Scholar] [CrossRef]
- Qiu, C.; McClements, D.J.; Jin, Z.Y.; Qin, Y.; Hu, Y.; Xu, X.M.; Wang, J.P. Resveratrol-loaded core-shell nanostructured delivery systems: Cyclodextrin-based metal-organic nanocapsules prepared by ionic gelation. Food Chem. 2020, 317, 126328. [Google Scholar] [CrossRef]
- Davidov-Pardo, G.; McClements, D.J. Nutraceutical delivery systems: Resveratrol encapsulation in grape seed oil nanoemulsions formed by spontaneous emulsification. Food Chem. 2015, 167, 205–212. [Google Scholar] [CrossRef]
- Neves, A.R.; Lucio, M.; Martins, S.; Lima, J.L.C.; Reis, S. Novel resveratrol nanodelivery systems based on lipid nanoparticles to enhance its oral bioavailability. Int. J. Nanomed. 2013, 8, 177–187. [Google Scholar]
- Pando, D.; Beltran, M.; Gerone, I.; Matos, M.; Pazos, C. Resveratrol entrapped niosomes as yoghurt additive. Food Chem. 2015, 170, 281–287. [Google Scholar] [CrossRef]
- Seethu, B.G.; Pushpadass, H.A.; Emerald, F.M.E.; Nath, B.S.; Naik, N.L.; Subramanian, K.S. Electrohydrodynamic encapsulation of resveratrol using food-grade nanofibres: Process optimization, characterization and fortification. Food Bioprocess Technol. 2020, 13, 341–354. [Google Scholar] [CrossRef]
- Layman, D.K.; Lonnerdal, B.; Fernstrom, J.D. Applications for α-lactalbumin in human nutrition. Nutr. Rev. 2018, 76, 444–460. [Google Scholar] [CrossRef]
- Salehi, B.; Machin, L.; Monzote, L.; Sharifi-Rad, J.; Ezzat, S.M.; Salem, M.A.; Merghany, R.M.; El Mahdy, N.M.; Kılıç, C.S.; Sytar, O.; et al. Therapeutic potential of quercetin: New insights and perspectives for human health. ACS Omega 2020, 5, 11849–11872. [Google Scholar] [CrossRef]
- Nam, J.S.; Sharma, A.R.; Nguyen, L.T.; Chakraborty, C.; Sharma, G.; Lee, S.S. Application of bioactive quercetin in oncotherapy: From nutrition to nanomedicine. Molecules 2016, 21, 108. [Google Scholar] [CrossRef] [Green Version]
- Isemura, M. Catechin in human health and disease. Molecules 2019, 24, 528. [Google Scholar] [CrossRef] [Green Version]
- Ni, S.; Hu, C.B.; Sun, R.; Zhao, G.D.; Xia, Q. Nanoemulsions-based delivery systems for encapsulation of quercetin: Preparation, characterization, and cytotoxicity studies. J. Food Process Eng. 2017, 40, 12374. [Google Scholar] [CrossRef]
- Aditya, N.P.; Macedo, A.S.; Doktorovov, S.; Souto, E.B.; Kim, S.; Chang, P.S.; Ko, S. Development and evaluation of lipid nanocarriers for quercetin delivery: A comparative study of solid lipid nanoparticles (SLN), nanostructured lipid carriers (NLC), and lipid nanoemulsions (LNE). LWT Food Sci. Technol. 2014, 59, 115–121. [Google Scholar] [CrossRef]
- Azzi, J.; Jraij, A.; Auezova, L.; Fourmentin, S.; Greige-Gerges, H. Novel findings for quercetin encapsulation and preservation with cyclodextrins, liposomes, and drug-in-cyclodextrin-in-liposomes. Food Hydrocoll. 2018, 81, 328–340. [Google Scholar] [CrossRef]
- Sadeghi-Ghadi, Z.; Ebrahimnejad, P.; Talebpour Amiri, F.; Nokhodchi, A. Improved oral delivery of quercetin with hyaluronic acid containing niosomes as a promising formulation. J. Drug Target. 2020, in press. [Google Scholar] [CrossRef]
- Chen, S.; Han, Y.H.; Huang, J.Y.; Dai, L.; Du, J.; McClements, D.J.; Mao, L.K.; Liu, J.F.; Gao, Y.X. Fabrication and characterization of layer-by-layer composite nanoparticles based on zein and hyaluronic acid for codelivery of curcumin and quercetagetin. ACS Appl. Mater. Interfaces 2019, 11, 16922–16933. [Google Scholar] [CrossRef]
- Ghayour, N.; Hosseini, S.M.H.; Eskandari, M.H.; Esteghlal, S.; Nekoei, A.R.; Gahruie, H.H.; Tatar, M.; Naghibalhossaini, F. Nanoencapsulation of quercetin and curcumin in casein-based delivery systems. Food Hydrocoll. 2019, 87, 394–403. [Google Scholar] [CrossRef]
- Campbell, E.L.; Chebib, M.; Johnston, G.A. The dietary flavonoids apigenin and (−)-epigallocatechin gallate enhance the positive modulation by diazepam of the activation by GABA of recombinant GABAA receptors. Biochem. Pharmacol. 2004, 68, 1631–1638. [Google Scholar] [CrossRef] [PubMed]
- Adachi, N.; Tomonaga, S.; Tachibana, T.; Denbow, D.M.; Furuse, M. (−)-Epigallocatechin gallate attenuates acute stress responses through GABAergic system in the brain. Eur. J. Pharmacol. 2006, 531, 171–175. [Google Scholar] [CrossRef]
- Legeay, S.; Rodier, M.; Fillon, L.; Faure, S.; Clere, N. Epigallocatechin gallate: A review of its beneficial properties to prevent metabolic syndrome. Nutrients 2015, 7, 5443–5468. [Google Scholar] [CrossRef] [Green Version]
- Granja, A.; Frias, I.; Neved, A.R.; Pinheiro, M.; Reis, S. Therapeutic potential of epigallocatechin gallate nanodelivery systems. Biomed. Res. Int. 2017, 2017, 5813793. [Google Scholar] [CrossRef] [PubMed]
- Gani, A.; Benjakul, S.; ul Ashraf, Z. Nutraceutical profiling of surimi gel containing β-glucan stabilized virgin coconut oil with and without antioxidants after simulated gastro-intestinal digestion. J. Food Sci. Technol. Mys. 2020, 57, 3132–3141. [Google Scholar] [CrossRef]
- Shpigelman, A.; Israeli, G.; Livney, Y.D. Thermally-induced protein-polyphenol co-assemblies: Beta lactoglobulin-based nanocomplexes as protective nanovehicles for EGCG. Food Hydrocoll. 2010, 24, 735–743. [Google Scholar] [CrossRef]
- Wang, Q.; Li, W.R.; Liu, P.; Hu, Z.Z.; Qin, X.G.; Liu, G. A glycated whey protein isolate-epigallocatechin gallate nanocomplex enhances the stability of emulsion delivery of β-carotene during simulated digestion. Food Funct. 2019, 10, 6829–6839. [Google Scholar] [CrossRef]
- Zhang, G.H.; Wang, Q.; Chen, J.J.; Zhang, X.M.; Tam, S.C.; Zheng, Y.T. The anti-HIV-1 effect of scutellarin. Biochem. Biophys. Res. Commun. 2005, 334, 812–816. [Google Scholar] [CrossRef]
- Xiong, L.; Du, R.; Xue, L.L.; Jiang, Y.; Huang, J.; Chen, L.; Liu, J.; Wang, T.H. Anti-colorectal cancer effects of scutellarin revealed by genomic and proteomic analysis. Chin. Med. 2020, 15, 28. [Google Scholar] [CrossRef] [Green Version]
- Matos, A.L.; Bruno, D.F.; Ambrosio, A.F.; Santos, P.F. The benefits of flavonoids in diabetic retinopathy. Nutrients 2020, 12, 3169. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Tan, J.; Luo, J.; Huang, P.; Zhou, W.; Chen, L.; Long, L.; Zhang, L.M.; Zhu, B.; Yang, L.; et al. Enhancement of scutellarin oral delivery efficacy by vitamin B12-modified amphiphilic chitosan derivatives to treat type II diabetes induced-retinopathy. J. Nanobiotechnol. 2017, 15, 18. [Google Scholar] [CrossRef] [Green Version]
- Hewlings, S.J.; Kalman, D.S. Curcumin: A review of its effects on human health. Foods 2017, 6, 92. [Google Scholar] [CrossRef]
- Tsuda, T. Curcumin as a functional food-derived factor: Degradation products, metabolites, bioactivity, and future perspectives. Food Funct. 2018, 9, 705–714. [Google Scholar] [CrossRef]
- Lopresti, A.L. The problem of curcumin and its bioavailability: Could its gastrointestinal influence contribute to its overall health-enhancing effects? Adv. Nutr. 2018, 9, 41–50. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kotha, R.R.; Luthria, D.L. Curcumin: Biological, pharmaceutical, nutraceutical, and analytical aspects. Molecules 2019, 24, 2930. [Google Scholar] [CrossRef] [Green Version]
- Bansode, P.A.; Patil, P.V.; Birajdar, A.R.; Somasundaram, I.; Bachute, M.T.; Rashinkar, G.S. Anticancer, antioxidant and antiangiogenic activities of nanoparticles of bioactive dietary nutraceuticals. ChemistrySelect 2019, 4, 13792–13796. [Google Scholar] [CrossRef]
- Ipar, V.S.; Dsouza, A.; Devarajan, P.V. Enhancing curcumin oral bioavailability through nanoformulations. Eur. J. Drug Metab. Pharmacokinet. 2019, 44, 459–480. [Google Scholar] [CrossRef] [PubMed]
- Nasery, M.M.; Abadi, B.; Poormoghadam, D.; Zarrabi, A.; Keyhanvar, P.; Khanbabaei, H.; Ashrafizadeh, M.; Mohammadinejad, R.; Tavakol, S.; Sethi, G. Curcumin delivery mediated by bio-based nanoparticles: A review. Molecules 2020, 25, 689. [Google Scholar] [CrossRef] [Green Version]
- Kharat, M.; McClements, D.J. Recent advances in colloidal delivery systems for nutraceuticals: A case study—Delivery by Design of curcumin. J. Colloid Interface Sci. 2019, 557, 506–518. [Google Scholar] [CrossRef]
- Zheng, B.J.; Lin, H.; Zhang, X.Y.; McClements, D.J. Fabrication of curcumin-loaded dairy milks using the pH-shift method: Formation, stability, and bioaccessibility. J. Agric. Food Chem. 2019, 67, 12245–12254. [Google Scholar] [CrossRef]
- Zheng, B.J.; Peng, S.F.; Zhang, X.Y.; McClements, D.J. Impact of delivery system type on curcumin bioaccessibility: Comparison of curcumin-loaded nanoemulsions with commercial curcumin supplements. J. Agric. Food Chem. 2018, 66, 10816–10826. [Google Scholar] [CrossRef]
- Yerramilli, M.; Longmore, N.; Ghosh, S. Stability and bioavailability of curcumin in mixed sodium caseinate and pea protein isolate nanoemulsions. J. Am. Oil Chem. Soc. 2018, 95, 1013–1026. [Google Scholar] [CrossRef]
- Dharunya, G.; Duraipandy, N.; Lakra, R.; Korapatti, P.S.; Jayavel, R.; Kiran, M.S. Curcumin cross-linked collagen aerogels with controlled anti-proteolytic and pro-angiogenic efficacy. Biomed. Mater. 2016, 11, 045011. [Google Scholar] [CrossRef]
- Sneharani, A.H. Curcumin-sunflower protein nanoparticles-A potential antiinflammatory agent. J. Food Biochem. 2019, 43, 12909. [Google Scholar] [CrossRef]
- Araujo, J.F.; Bourbon, A.I.; Simoes, L.S.; Vicente, A.A.; Coutinho, P.J.G.; Ramos, O.L. Physicochemical characterisation and release behaviour of curcumin-loaded lactoferrin nanohydrogels into food simulants. Food Funct. 2020, 11, 305–317. [Google Scholar] [CrossRef]
- Liu, F.G.; Ma, D.; Luo, X.; Zhang, Z.Y.; He, L.L.; Gao, Y.X.; McClements, D.J. Fabrication and characterization of protein-phenolic conjugate nanoparticles for co-delivery of curcumin and resveratrol. Food Hydrocoll. 2018, 79, 450–461. [Google Scholar] [CrossRef]
- Dai, L.; Wei, Y.; Sun, C.X.; Mao, L.K.; McClements, D.J.; Gao, Y.X. Development of protein-polysaccharide-surfactant ternary complex particles as delivery vehicles for curcumin. Food Hydrocoll. 2018, 85, 75–85. [Google Scholar] [CrossRef]
- Chen, S.; Li, Q.; McClements, D.J.; Han, Y.H.; Dai, L.; Mao, L.K.; Gao, Y.X. Co-delivery of curcumin and piperine in zein-carrageenan core-shell nanoparticles: Formation, structure, stability and in vitro gastrointestinal digestion. Food Hydrocoll. 2020, 99, 105334. [Google Scholar] [CrossRef]
- Huang, X.X.; Huang, X.L.; Gong, Y.S.; Xiao, H.; McClements, D.J.; Hu, K. Enhancement of curcumin water dispersibility and antioxidant activity using core-shell protein-polysaccharide nanoparticles. Food Res. Int. 2016, 87, 1–9. [Google Scholar] [CrossRef] [Green Version]
- Silva, H.D.; Poejo, J.; Pinheiro, A.C.; Donsi, F.; Serra, A.T.; Duarte, C.M.M.; Ferrari, G.; Cerqueira, M.A.; Vicente, A.A. Evaluating the behaviour of curcumin nanoemulsions and multilayer nanoemulsions during dynamic in vitro digestion. J. Funct. Foods 2018, 48, 605–613. [Google Scholar] [CrossRef] [Green Version]
- Guo, C.J.; Yin, J.G.; Chen, D.Q. Co-encapsulation of curcumin and resveratrol into novel nutraceutical hyalurosomes nano-food delivery system based on oligo-hyaluronic acid-curcumin polymer. Carbohydr. Polym. 2018, 181, 1033–1037. [Google Scholar] [CrossRef]
- Aadinath, W.; Bhushani, A.; Anandharamakrishnan, C. Synergistic radical scavenging potency of curcumin-in-β-cyclodextrin-in-nanomagnetoliposomes. Mater. Sci. Eng. C Mater. Biol. Appl. 2016, 64, 293–302. [Google Scholar] [CrossRef]
- Peng, S.F.; Li, Z.L.; Zou, L.Q.; Liu, W.; Liu, C.M.; McClements, D.J. Enhancement of curcumin bioavailability by encapsulation in sophorolipid-coated nanoparticles: An in vitro and in vivo study. J. Agric. Food Chem. 2018, 66, 1488–1497. [Google Scholar] [CrossRef] [PubMed]
- Peng, S.F.; Li, Z.L.; Zou, L.Q.; Liu, W.; Liu, C.M.; McClements, D.J. Improving curcumin solubility and bioavailability by encapsulation in saponin-coated curcumin nanoparticles prepared using a simple pH-driven loading method. Food Funct. 2018, 9, 1829–1839. [Google Scholar] [CrossRef]
- Li, Z.L.; Peng, S.F.; Chen, X.; Zhu, Y.Q.; Zou, L.Q.; Liu, W.; Liu, C.M. Pluronics modified liposomes for curcumin encapsulation: Sustained release, stability and bioaccessibility. Food Res. Int. 2018, 108, 246–253. [Google Scholar] [CrossRef]
- Zarate, R.; Jaber-Vazdekis, N.; Tejera, N.; Perez, J.A.; Rodriguez, C. Significance of long chain polyunsaturated fatty acids in human health. Clin. Transl. Med. 2017, 6, 25. [Google Scholar] [CrossRef] [Green Version]
- Serini, S.; Calviello, G. Omega-3 PUFA responders and non-responders and the prevention of lipid dysmetabolism and related diseases. Nutrients 2020, 12, 1363. [Google Scholar] [CrossRef]
- Lunn, J.; Theobald, H.E. The health effects of dietary unsaturated fatty acids. Nutr. Bull. 2006, 31, 178–224. [Google Scholar] [CrossRef]
- Serini, S.; Cassano, R.; Trombino, S.; Calviello, G. Nanomedicine-based formulations containing ω-3 polyunsaturated fatty acids: Potential application in cardiovascular and neoplastic diseases. Int. J. Nanomed. 2019, 14, 2809–2828. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Valenzuela, A.; Valenzuela, R.; Sanhueza, J.; de la Barra, F.; Morales, G. Phospholipids from marine origin: A new alternative for supplementing omega-3 fatty acids. Rev. Chil. Nutr. 2014, 41, 433–438. [Google Scholar]
- Gulotta, A.; Saberi, A.H.; Nicoli, M.C.; McClements, D.J. Nanoemulsion-based delivery systems for polyunsaturated (ω-3) oils: Formation using a spontaneous emulsification method. J. Agric. Food Chem. 2014, 62, 1720–1725. [Google Scholar] [CrossRef]
- Uluata, S.; McClements, D.J.; Decker, E.A. Physical stability, autoxidation, and photosensitized oxidation of ω-3 oils in nanoemulsions prepared with natural and synthetic surfactants. J. Agric. Food Chem. 2015, 63, 9333–9340. [Google Scholar] [CrossRef]
- Walker, R.M.; Gumus, C.E.; Decker, E.A.; McClements, D.J. Improvements in the formation and stability of fish oil-in-water nanoemulsions using carrier oils: MCT, thyme oil, & lemon oil. J. Food Eng. 2017, 211, 60–68. [Google Scholar]
- Esquerdo, V.M.; Silva, P.P.; Dotto, G.L.; Pinto, L.A.A. Nanoemulsions from unsaturated fatty acids concentrates of carp oil using chitosan, gelatin, and their blends as wall materials. Eur. J. Lipid Sci. Technol. 2018, 120, 1700240. [Google Scholar] [CrossRef]
- Dey, T.K.; Koley, H.; Ghosh, M.; Dey, S.; Dhar, P. Effects of nano-sizing on lipid bioaccessibility and ex vivo bioavailability from EPA-DHA rich oil in water nanoemulsion. Food Chem. 2019, 275, 135–142. [Google Scholar] [CrossRef]
- Li, Y.; Li, M.D.; Qi, Y.M.; Zheng, L.; Wu, C.L.; Wang, Z.J.; Teng, F. Preparation and digestibility of fish oil nanoemulsions stabilized by soybean protein isolate-phosphatidylcholine. Food Hydrocoll. 2020, 100, 105310. [Google Scholar] [CrossRef]
- Hwang, J.Y.; Ha, H.K.; Lee, M.R.; Kim, J.W.; Kim, H.J.; Lee, W.J. Physicochemical property and oxidative stability of whey protein concentrate multiple nanoemulsion containing fish oil. J. Food Sci. 2017, 82, 437–444. [Google Scholar] [CrossRef]
- Prieto, C.; Lagaron, J.M. Nanodroplets of docosahexaenoic acid-enriched algae oil encapsulated within microparticles of hydrocolloids by emulsion electrospraying assisted by pressurized gas. Nanomaterials 2020, 10, 270. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Torres-Giner, S.; Martinez-Abad, A.; Ocio, M.J.; Lagaron, J.M. Stabilization of a nutraceutical omega-3 fatty acid by encapsulation in ultrathin electrosprayed zein prolamine. J. Food Sci. 2010, 75, N69–N79. [Google Scholar] [CrossRef] [PubMed]
- Dey, T.K.; Banerjee, P.; Chatterjee, R.; Dhar, P. Designing of ω-3 PUFA enriched biocompatible nanoemulsion with sesame protein isolate as a natural surfactant: Focus on enhanced shelf-life stability and biocompatibility. Colloids Surf. A Physicochem. Eng. Asp. 2018, 538, 36–44. [Google Scholar] [CrossRef]
- Zimet, P.; Rosenberg, D.; Livney, Y.D. Re-assembled casein micelles and casein nanoparticles as nano-vehicles for ω-3 polyunsaturated fatty acids. Food Hydrocoll. 2011, 25, 1270–1276. [Google Scholar] [CrossRef]
- Semenova, M.G.; Antipova, A.S.; Zelikina, D.V.; Martirosova, E.I.; Plashchina, I.G.; Palmina, N.P.; Binyukov, V.I.; Bogdanova, N.G.; Kasparov, V.V.; Shumilina, E.A.; et al. Biopolymer nanovehicles for essential polyunsaturated fatty acids: Structure-functionality relationships. Food Res. Int. 2016, 88, 70–78. [Google Scholar] [CrossRef]
- Zimet, P.; Livney, Y.D. Beta-lactoglobulin and its nanocomplexes with pectin as vehicles for ω-3 polyunsaturated fatty acids. Food Hydrocoll. 2009, 23, 1120–1126. [Google Scholar] [CrossRef]
- Loughrill, E.; Thompson, S.; Owusu-Ware, S.; Snowden, M.J.; Douroumis, D.; Zand, N. Controlled release of microencapsulated docosahexaenoic acid (DHA) by spray-drying processing. Food Chem. 2019, 286, 368–375. [Google Scholar] [CrossRef]
- Hashemi, F.S.; Farzadnia, F.; Aghajani, A.; NobariAzar, F.A.; Pezeshki, A. Conjugated linoleic acid loaded nanostructured lipid carrier as a potential antioxidant nanocarrier for food applications. Food Sci. Nutr. 2020, 8, 4185–4195. [Google Scholar] [CrossRef]
- Yaghmur, A.; Ghazal, A.; Ghazal, R.; Dimaki, M.; Svendsen, W.E. A hydrodynamic flow focusing microfluidic device for the continuous production of hexosomes based on docosahexaenoic acid monoglyceride. Phys. Chem. Chem. Phys. 2019, 21, 13005–13013. [Google Scholar] [CrossRef]
- Shao, X.R.; Bor, G.; Al-Hosayni, S.; Salentinig, S.; Yaghmur, A. Structural characterization of self-assemblies of new omega-3 lipids: Docosahexaenoic acid and docosapentaenoic acid monoglycerides. Phys. Chem. Chem. Phys. 2018, 20, 23928–23941. [Google Scholar] [CrossRef]
- Zarrabi, A.; Abadi, M.A.A.; Khorasani, S.; Mohammadabadi, M.R.; Jamshidi, A.; Torkaman, S.; Taghavi, E.; Mozafari, M.R.; Rasti, B. Nanoliposomes and tocosomes as multifunctional nanocarriers for the encapsulation of nutraceutical and dietary molecules. Molecules 2020, 25, 638. [Google Scholar] [CrossRef] [Green Version]
- Gill, H.; Guarner, F. Probiotics and human health: A clinical perspective. Postgrad. Med. J. 2004, 80, 516–526. [Google Scholar] [CrossRef] [Green Version]
- Wan, M.L.Y.; Forsythe, S.J.; El-Nezami, H. Probiotics interaction with foodborne pathogens: A potential alternative to antibiotics and future challenges. Crit. Rev. Food Sci. Nutr. 2019, 59, 3320–3333. [Google Scholar] [CrossRef]
- Kerry, R.G.; Patra, J.K.; Gouda, S.; Park, Y.; Shin, H.S.; Das, G. Benefaction of probiotics for human health: A review. J. Food Drug Anal. 2018, 26, 927–939. [Google Scholar] [CrossRef] [Green Version]
- Sanders, M.E.; Merenstein, D.; Merrifield, C.A.; Hutkins, R. Probiotics for human use. Nutr. Bull. 2018, 43, 212–225. [Google Scholar] [CrossRef]
- Coghetto, C.C.; Brinques, G.B.; Ayub, M.A. Probiotics production and alternative encapsulation methodologies to improve their viabilities under adverse environmental conditions. Int. J. Food Sci. Nutr. 2016, 67, 929–943. [Google Scholar] [CrossRef]
- Kavitake, D.; Kandasamy, S.; Devi, P.B.; Shetty, P.H. Recent developments on encapsulation of lactic acid bacteria as potential starter culture in fermented foods—A review. Food Biosci. 2018, 21, 34–44. [Google Scholar] [CrossRef]
- Anal, A.K.; Singh, H. Recent advances in microencapsulation of probiotics for industrial applications and targeted delivery. Trends Food Sci. Technol. 2007, 18, 240–251. [Google Scholar] [CrossRef]
- Kwiecien, I.; Kwiecien, M. Application of polysaccharide-based hydrogels as probiotic delivery systems. Gels 2018, 4, 47. [Google Scholar] [CrossRef] [Green Version]
- Pathak, K.; Akhtar, N. Nanoprobiotics: Progress and Issues. In Nanonutraceuticals, 1st ed.; Singh, B., Ed.; CRC Press: Boca Raton, FL, USA, 2018; Chapter 9; 18p. [Google Scholar]
- Durazzo, A.; Nazhand, A.; Lucarini, M.; Atanasov, A.G.; Souto, E.B.; Novellino, E.; Capasso, R.; Santini, A. An updated overview on nanonutraceuticals: Focus on nanoprebiotics and nanoprobiotics. Int. J. Mol. Sci. 2020, 21, 2285. [Google Scholar] [CrossRef] [Green Version]
- Anselmo, A.C.; McHugh, K.J.; Webster, J.; Langer, R.; Jaklenec, A. Layer-by-layer encapsulation of probiotics for delivery to the microbiome. Adv. Mater. 2016, 28, 9486–9490. [Google Scholar] [CrossRef] [Green Version]
- Liu, H.; Cui, S.W.; Chen, M.; Li, Y.; Liang, R.; Xu, F.F.; Zhong, F. Protective approaches and mechanisms of microencapsulation to the survival of probiotic bacteria during processing, storage and gastrointestinal digestion: A review. Crit. Rev. Food Sci. Nutr. 2019, 59, 2863–2878. [Google Scholar] [CrossRef] [PubMed]
- Qi, W.; Liang, X.; Yun, T.; Guo, W. Growth and survival of microencapsulated probiotics prepared by emulsion and internal gelation. J. Food Sci. Technol. 2019, 56, 1398–1404. [Google Scholar] [CrossRef] [Green Version]
- Hansen, L.T.; Allan-Wojtas, P.M.; Jin, Y.L.; Paulson, A.T. Survival of Ca-alginate microencapsulated Bifidobacterium spp. in milk and simulated gastrointestinal conditions. Food Microbiol. 2002, 19, 35–45. [Google Scholar] [CrossRef]
- Holkem, A.T.; Raddatz, G.C.; Barin, J.S.; Flores, E.M.M.; Muller, E.I.; Codevilla, C.F.; Jacob-Lopes, E.; Grosso, R.F.; Menezes, C.R. Production of microcapsules containing Bifidobacterium BB-12 by emulsification/internal gelation. LWT Food Sci. Technol. 2017, 76, 216–221. [Google Scholar] [CrossRef]
- Wang, J.; Korber, D.R.; Low, N.H.; Nickerson, M.T. Encapsulation of Bifidobacterium adolescentis cells with legume proteins and survival under stimulated gastric conditions and during storage in commercial fruit juices. Food Sci. Biotechnol. 2015, 24, 383–391. [Google Scholar] [CrossRef]
- Patrignani, F.; Siroli, L.; Serrazanetti, D.I.; Braschi, G.; Betoret, E.; Reinheimer, J.A.; Lanciotti, R. Microencapsulation of functional strains by high pressure homogenization for a potential use in fermented milk. Food Res. Int. 2017, 97, 250–257. [Google Scholar] [CrossRef]
- Atia, A.; Gomaa, A.; Fliss, I.; Beyssac, E.; Garrait, G.; Subirade, M. A prebiotic matrix for encapsulation of probiotics: Physicochemical and microbiological study. J. Microencapsul. 2016, 33, 89–101. [Google Scholar] [CrossRef] [PubMed]
- Coghetto, C.C.; Brinques, G.B.; Siqueira, N.M.; Pletsch, J.; Soarea, N.D.; Ayub, M.A.Z. Electrospraying microencapsulation of Lactobacillus plantarum enhances cell viability under refrigeration storage and simulated gastric and intestinal fluids. J. Funct. Foods 2016, 24, 316–326. [Google Scholar] [CrossRef]
- Silva, K.C.G.; Cezarino, E.C.; Michelon, M.; Sato, A.C.K. Symbiotic microencapsulation to enhance Lactobacillus acidophilus survival. LWT Food Sci. Technol. 2018, 89, 503–509. [Google Scholar] [CrossRef]
- Yao, M.F.; Li, B.; Ye, H.W.; Huang, W.H.; Luo, Q.X.; Xiao, H.; McClements, D.J.; Li, L.J. Enhanced viability of probiotics (Pediococcus pentosaceus Li05) by encapsulation in microgels doped with inorganic nanoparticles. Food Hydrocoll. 2018, 83, 246–252. [Google Scholar] [CrossRef]
- Poletto, G.; Raddatz, G.C.; Cichoski, A.J.; Zepla, L.Q.; Lopse, E.J.; Barin, J.S.; Wagner, R.; Menezes, C.R. Study of viability and storage stability of Lactobacillus acidophillus when encapsulated with the prebiotics rice bran, inulin and Hi-maize. Food Hydrocoll. 2019, 95, 238–244. [Google Scholar] [CrossRef]
- Huq, T.; Fraschini, C.; Khan, A.; Riedl, B.; Bouchard, J.; Lacroix, M. Alginate based nanocomposite for microencapsulation of probiotic: Effect of cellulose nanocrystal (CNC) and lecithin. Carbohydr. Polym. 2017, 168, 61–69. [Google Scholar] [CrossRef]
- Pitigraisorn, P.; Srichaisupakit, K.; Wongpadungkiat, N.; Wongsasulak, S. Encapsulation of Lactobacillus acidophilus in moist-heat-resistant multilayered microcapsules. J. Food Eng. 2017, 192, 11–18. [Google Scholar] [CrossRef]
- Ji, R.; Wu, J.; Zhang, J.L.; Wang, T.; Zhang, X.D.; Shai, L.; Chen, D.J.; Wang, J. Extending viability of Bifidobacterium longum in chitosan-coated alginate microcapsules using emulsification and internal gelation encapsulation technology. Front. Microbiol. 2019, 10, 1389. [Google Scholar] [CrossRef]
- Riaz, T.; Iqbal, M.W.; Saeed, M.; Yasmin, I.; Hassanin, H.A.M.; Mahmood, S.; Rehman, A. In vitro survival of Bifidobacterium bifidum microencapsulated in zein-coated alginate hydrogel microbeads. J. Microencapsul. 2019, 36, 192–203. [Google Scholar] [CrossRef]
- Ramos, P.E.; Abrunhosa, L.; Pinheiro, A.; Cerqueira, M.A.; Motta, C.; Castanheira, I.; Chandra-Hioe, M.V.; Arcot, J.; Teixeira, J.A.; Vicente, A.A. Probiotic-loaded microcapsule system for human in situ folate production: Encapsulation and system validation. Food Res. Int. 2016, 90, 25–32. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ramos, P.E.; Cerqueira, M.A.; Teixeira, J.A.; Vicente, A.A. Physiological protection of probiotic microcapsules by coatings. Crit. Rev. Food Sci. Nutr. 2018, 58, 1864–1877. [Google Scholar] [CrossRef] [PubMed]
- Calinoiu, L.-F.; Ştefănescu, B.E.; Pop, I.D.; Muntean, L.; Vodnar, D.C. Chitosan coating applications in probiotic microencapsulation. Coatings 2019, 9, 194. [Google Scholar] [CrossRef] [Green Version]
- Ebrahimnejad, P.; Khavarpour, M.; Khalilid, S. Survival of Lactobacillus acidophilus as probiotic bacteria using chitosan nanoparticles. IJE Trans. Basics 2017, 30, 456–463. [Google Scholar]
- Kim, J.U.; Kim, B.; Shahbaz, H.M.; Lee, S.H.; Park, D.; Park, J.Y. Encapsulation of probiotic Lactobacillus acidophilus by ionic gelation with electrostatic extrusion for enhancement of survival under simulated gastric conditions and during refrigerated storage. Int. J. Food Sci. Technol. 2017, 52, 519–530. [Google Scholar] [CrossRef]
- Chen, L.; Yang, T.; Song, Y.; Shu, G.W.; Chen, H. Effect of xanthan-chitosan-xanthan double layer encapsulation on survival of Bifidobacterium BB01 in simulated gastrointestinal conditions, bile salt solution and yogurt. LWT Food Sci. Technol. 2017, 81, 274–280. [Google Scholar] [CrossRef]
- Priya, A.J.; Vijayalakshmi, S.P.; Raichur, A.M. Enhanced survival of probiotic Lactobacillus acidophilus by encapsulation with nanostructured polyelectrolyte layers through layer-by-layer approach. J. Agric. Food Chem. 2011, 59, 11838–11845. [Google Scholar] [CrossRef]
- Shah, A.; Gani, A.; Ahmad, M.; Ashwar, B.A.; Masoodi, F.A. β-Glucan as an encapsulating agent: Effect on probiotic survival in simulated gastrointestinal tract. Int. J. Biol. Macromol. 2016, 82, 217–222. [Google Scholar] [CrossRef]
- Nawong, S.; Oonsivilai, R.; Boonkerd, N.; Truelstrup Hansen, L. Entrapment in food-grade transglutaminase cross-linked gelatin-maltodextrin microspheres protects Lactobacillus spp. during exposure to simulated gastro-intestinal juices. Food Res. Int. 2016, 85, 191–199. [Google Scholar] [CrossRef]
- Nunes, G.L.; Etchepare, M.A.; Cichoski, A.J.; Zepka, L.Q.; Lopes, E.J.; Barin, J.S.; Flores, E.M.D.M.; Silva, C.D.B.; Menezes, C.R. Inulin, hi-maize, and trehalose as thermal protectants for increasing viability of Lactobacillus acidophilus encapsulated by spray drying. LWT Food Sci. Technol. 2018, 89, 128–133. [Google Scholar] [CrossRef]
- Krithika, B.; Preetha, R. Formulation of protein based inulin incorporated synbiotic nanoemulsion for enhanced stability of probiotic. Mat. Res. Express 2019, 6, 114003. [Google Scholar] [CrossRef]
- Rodrigues, D.; Sousa, S.; Rocha-Santos, T.; Silva, J.P.; Sousa Lobo, J.M.; Costa, R.; Amaral, M.H.; Pintado, M.M.; Gomes, A.M.; Malcata, F.X.; et al. Influence of l-cysteine, oxygen and relative humidity upon survival throughout storage of probiotic bacteria in whey protein-based microcapsules. Int. Dairy J. 2011, 21, 869–876. [Google Scholar] [CrossRef] [Green Version]
- Gonzalez-Ferrero, C.; Irache, J.M.; Gonzalez-Navarro, C.J. Soybean protein-based microparticles for oral delivery of probiotics with improved stability during storage and gut resistance. Food Chem. 2018, 239, 879–888. [Google Scholar] [CrossRef] [PubMed]
- Mao, L.; Pan, Q.; Yuan, F.; Gao, Y. Formation of soy protein isolate-carrageenan complex coacervates for improved viability of Bifidobacterium longum during pasteurization and in vitro digestion. Food Chem. 2019, 276, 307–314. [Google Scholar] [CrossRef]
- Zupancic, S.; Skrlec, K.; Kocbek, P.; Kristl, J.; Berlec, A. Effects of electrospinning on the viability of ten species of lactic acid bacteria in poly(ethylene oxide) nanofibers. Pharmaceutics 2019, 11, 483. [Google Scholar] [CrossRef] [Green Version]
- Pedroso, D.L.; Thomazini, M.; Heinemann, R.J.B.; Favaro-Trindade, C.S. Protection of Bifidobacterium lactis and Lactobacillus acidophilus by microencapsulation using spray-chilling. Int. Dairy J. 2012, 26, 127–132. [Google Scholar] [CrossRef]
- Pedroso, D.L.; Dogenski, M.; Thomazini, M.; Heinemann, R.J.B.; Favaro-Trindade, C.S. Microencapsulation of Bifidobacterium animalis subsp. lactis and Lactobacillus acidophilus in cocoa butter using spray chilling technology. Braz. J. Microbiol. 2013, 44, 777–783. [Google Scholar] [CrossRef] [Green Version]
- de Matos Junior, F.E.; Silva, M.P.; Kasemodel, M.G.C.; Santosm, T.T.; Burns, P.; Reinheimer, J.; Vinderola, G.; Favaro-Trindade, C.S. Evaluation of the viability and the preservation of the functionality of microencapsulated Lactobacillus paracasei BGP1 and Lactobacillus rhamnosus 64 in lipid particles coated by polymer electrostatic interaction. J. Funct. Foods 2019, 54, 98–108. [Google Scholar] [CrossRef]
- Paula, D.A.; Martins, E.M.F.; Costa, N.A.; Oliveira, P.M.; Oliveira, E.B.; Ramos, A.M. Use of gelatin and gum arabic for microencapsulation of probiotic cells from Lactobacillus plantarum by a dual process combining double emulsification followed by complex coacervation. Int. J. Biol. Macromol. 2019, 133, 722–731. [Google Scholar] [CrossRef] [PubMed]
- Okuro, P.K.; Thomazini, M.; Balieiro, J.C.C.; Liberal, R.D.C.O.; Favaro-Trindade, C.S. Co-encapsulation of Lactobacillus acidophilus with inulin or polydextrose in solid lipid microparticles provides protection and improves stability. Food Res. Int. 2013, 53, 96–103. [Google Scholar] [CrossRef]
- Amakiri, A.C.; Kalombo, L.; Thantsha, M.S. Lyophilised vegetal BM 297 ATO-inulin lipid-based synbiotic microparticles containing Bifidobacterium longum LMG 13197: Design and characterisation. J. Microencapsul. 2015, 32, 820–827. [Google Scholar] [CrossRef]
- Verruck, S.; de Carvalho, M.W.; de Liz, G.R.; Amante, E.R.; Vieira, C.R.W.; Amboni, R.D.D.C.; Prudencio, E.S. Bifidobacterium BB-12 microencapsulated with full-fat goat’s milk and prebiotics when exposed to simulated gastrointestinal conditions and thermal treatments. Small Rumin. Res. 2017, 153, 48–56. [Google Scholar] [CrossRef]
- Nagy, Z.K.; Wagner, I.; Suhajda, A.; Tobak, T.; Harsztos, A.H.; Vigh, T.; Soti, P.L.; Pataki, K.; Molnar, K.; Marosi, G. Nanofibrous solid dosage form of living bacteria prepared by electrospinning. Express Polym. Lett. 2014, 8, 352–361. [Google Scholar] [CrossRef] [Green Version]
- Ceylan, Z.; Uslu, E.; Ispirli, H.; Meral, R.; Gavgali, M.; Yilmaz, M.T.; Dertli, E. A novel perspective for Lactobacillus reuteri: Nanoencapsulation to obtain functional fish fillets. LWT Food Sci. Technol. 2019, 115, 108427. [Google Scholar] [CrossRef]
- Shoaib, M.; Shehzad, A.; Omar, M.; Rakha, A.; Raza, H.; Rizwan Sharif, H.; Shakeel, A.; Ansari, A.; Niazi, S. Inulin: Properties, health benefits and food applications. Carbohydr. Polym. 2016, 147, 444–454. [Google Scholar] [CrossRef]





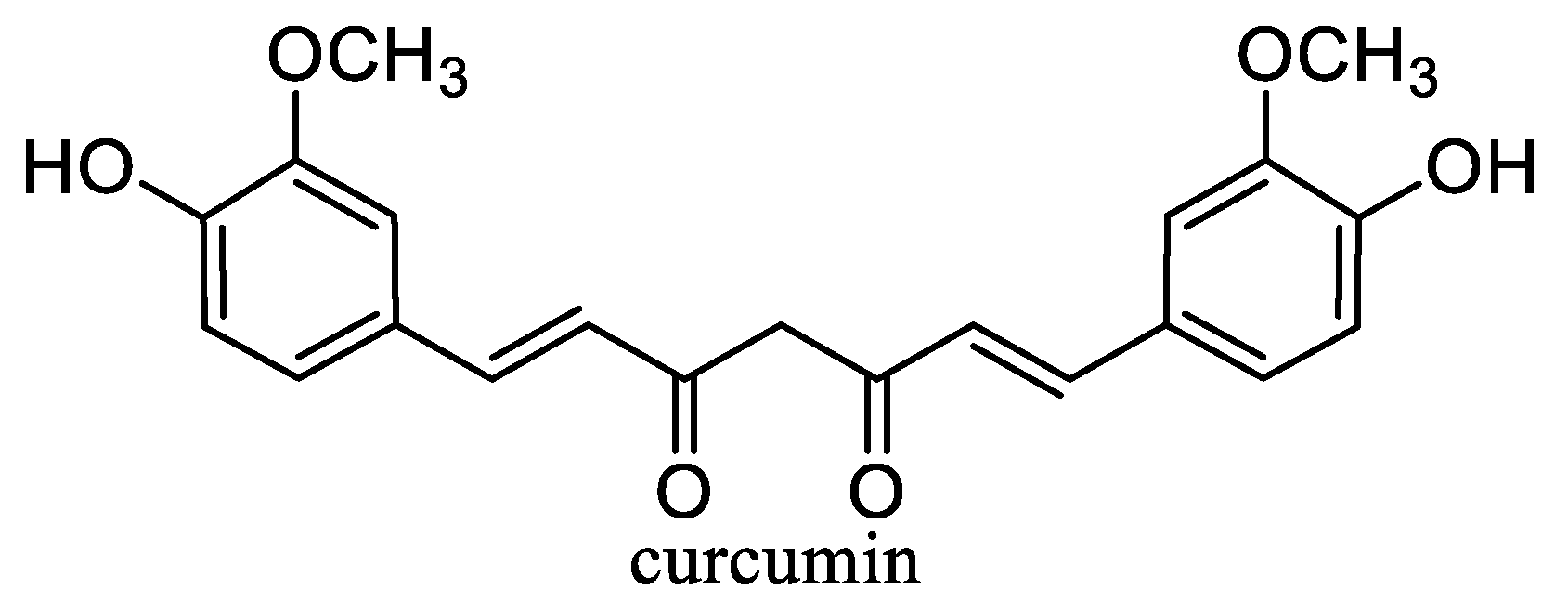
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jampilek, J.; Kralova, K. Potential of Nanonutraceuticals in Increasing Immunity. Nanomaterials 2020, 10, 2224. https://doi.org/10.3390/nano10112224
Jampilek J, Kralova K. Potential of Nanonutraceuticals in Increasing Immunity. Nanomaterials. 2020; 10(11):2224. https://doi.org/10.3390/nano10112224
Chicago/Turabian StyleJampilek, Josef, and Katarina Kralova. 2020. "Potential of Nanonutraceuticals in Increasing Immunity" Nanomaterials 10, no. 11: 2224. https://doi.org/10.3390/nano10112224





