Luminescent Guests Encapsulated in Metal–Organic Frameworks for Portable Fluorescence Sensor and Visual Detection Applications: A Review
Abstract
:1. Introduction
2. Synthetic Protocols and Design Guidelines
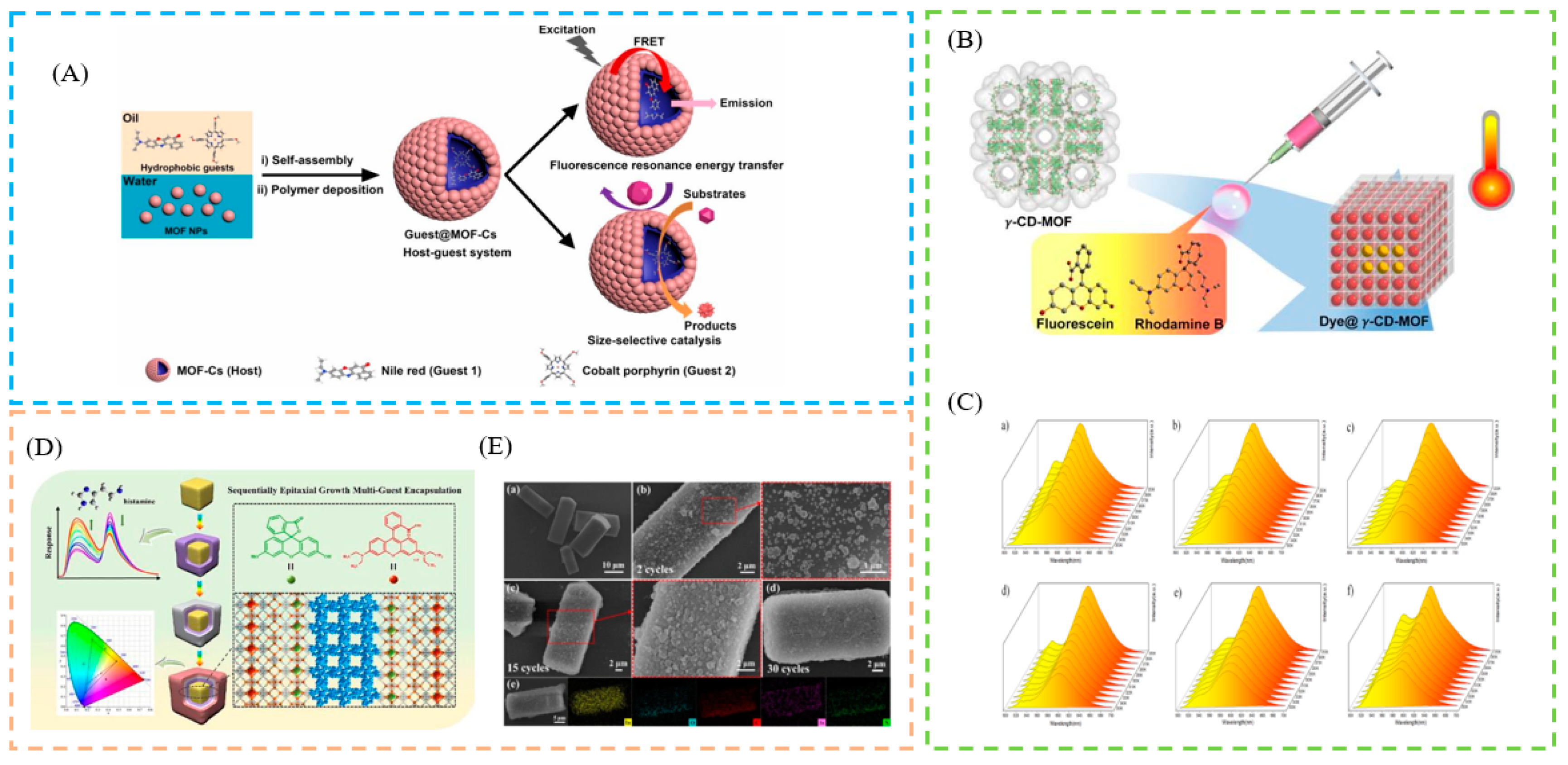
2.1. Post-Synthetic LG Confinement Methods
2.2. In Situ LG Confinement Methods
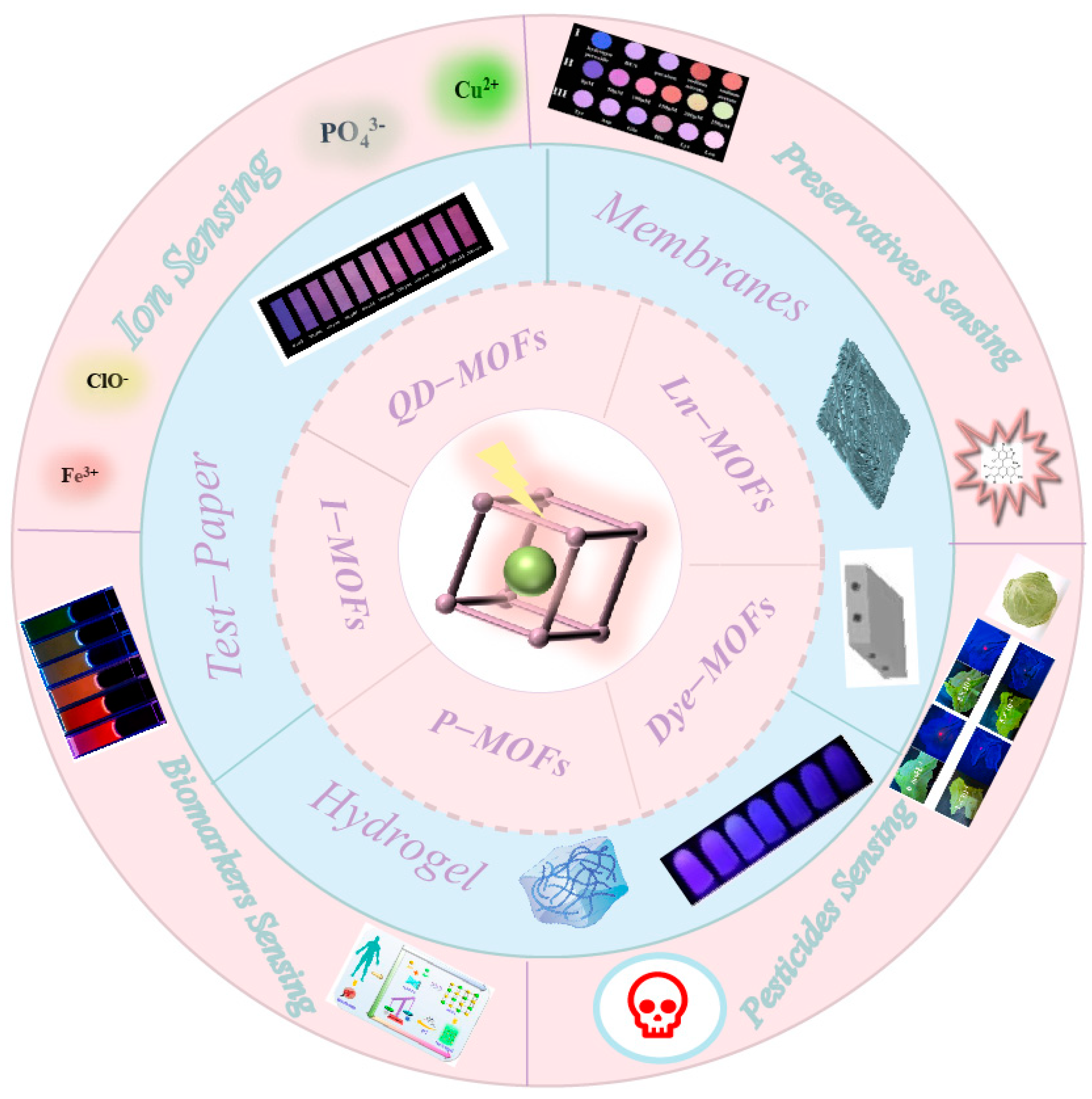
3. Encapsulation of Different Luminescent Guests in the MOFs
3.1. Lanthanide Ions
3.2. Quantum Dots
3.3. Luminescent Complexes
3.4. Organic Dyes
3.5. Metal Nanoclusters
4. Portable Detection Device Smartphone Platform
4.1. Surface Plasmon Coupled Emission (SPCE) Platform
4.2. Test Paper
4.3. Membranes
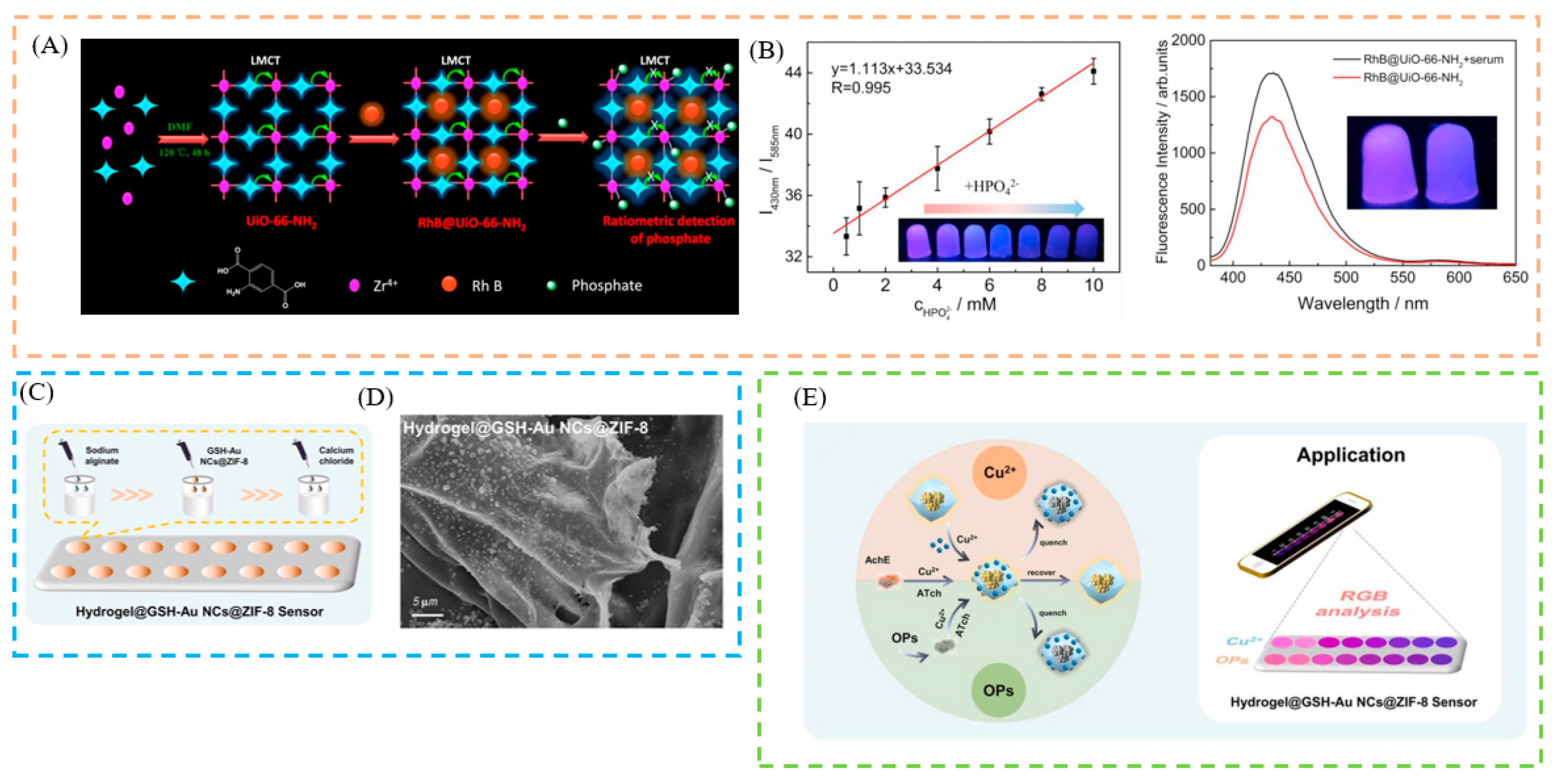
4.4. Hydrogel
5. Sensing Applications
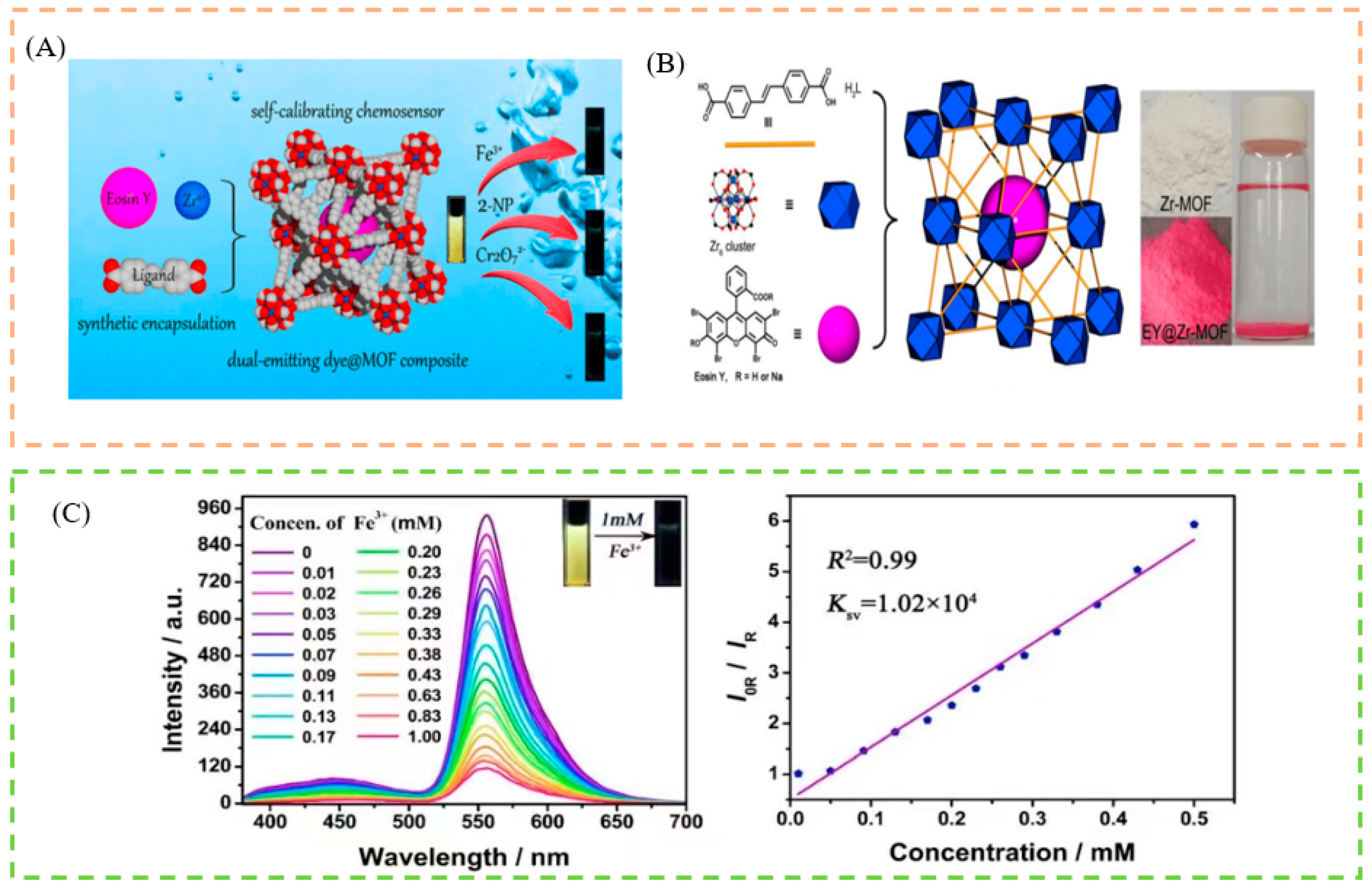
5.1. Sensing of Ions
5.2. Sensing of Biomarkers
5.3. Sensing of Pesticides
5.4. Sensing of Preservatives
5.5. Stability of Sensors
6. Future Scope and Perspectives
- (1)
- Synthesis: the luminescence properties of LG@MOFs are mainly dependent on the framework structure of the MOF materials and the synergistic effect between the photon units. The main limitation of the framework structure is that the pore size, structure, metal node, and organic ligand properties of MOFs must match the size, surface charge, and inherent properties of the fluorophore. Synergism refers to the relationship between the synergistic effects of different metal nodes such as photon units, organic ligands, and guest molecules. Therefore, the relationship between structure and properties, preparation strategies, host–guest interactions, and synergistic effects of LG@MOFs require further study.
- (2)
- Detection: most detection methods are based on “off” processes, while LG@MOF materials based on “on” processes can improve selectivity and sensitivity. At the same time, most established LG@MOF sensors depend on the change in transmission signal strength from a single transmission center, which may lead to false responses due to the change in external conditions. The ratio fluorescence probe and self-calibration method can eliminate environmental interference and improve detection accuracy. In addition, it can also add an adsorption function and create a new luminous LG@MOF, combining detection and eradication. Due to the complexity of actual samples and the trace level of target pollutants, it is necessary to consider adding functional groups to LG@MOFs, for sample purification and target analyte enrichment.
- (3)
- Sensing mechanisms: the mechanism of fluorescence sensing still needs further investigation and theoretical calculations are needed to better understand the sensing process. The mechanisms proposed by many reported studies on LG@MOFs are either vague or speculative. The application of theoretical methods, such as density functional theory (DFT), should be encouraged for LG@MOF systems. The main challenges lie in modeling the dispersion correction and electronic structure of large-scale systems with spatial constraints, and using DFT to simulate excited-state events. Therefore, the recent development and implementation of a computationally efficient but accurate DFT method, are expected to simulate the structure–property relationship of large LG@MOF systems.
- (4)
- Application: the versatility of such composites and practical applications need further exploration, such as the activity and selectivity for catalyzing tandem reactions, optical imaging for biotherapeutics, diagnostics, and drug delivery characteristics. The use of LG@MOFs with customizable sizes and shapes for targeted drug delivery, living cell sensing, and imaging is also promising. In addition, sensing devices such as test papers and luminescent labs-on-a-chip, should be designed for convenient use, by the combination of the properties of LG@MOFs. Recent advances in high-resolution 3D printing, precision inkjet printing, electrospinning, and lithography, can also be combined, to expand LG@MOF-based applications. We believe that, with continuous research and improvement, a bright future for LG@MOFs in the field of fluorescence detection can be expected.
7. Conclusions
| Targets | MOF | Color Change | Linear Range | LOD | Ref. |
|---|---|---|---|---|---|
| 1-N | Eu3+@MOF-253 | red→green | 7 μg/mL | [167] | |
| OTC | EuUCBA | red→colorless | 0.118 μM | [168] | |
| CTC | red→colorless | 0.228 μM | |||
| MTC | red→colorless | 0.102 μM | |||
| MOC | red→colorless | 0.138 μM | |||
| TC | red→colorless | 0.206 μM | |||
| DOXY | red→dark | 0.078 μM | |||
| DCNA | Eu3+@Zn-MOF-NS | red→pink | 0.17 μM | [169] | |
| 4-NA | Eu3+@Zn-MOF | red→dark | 6.01 μM | [170] | |
| Rotenone | Eu@Zn-MOF | red→dark | 2.31 × 10−7 mol/L | [171] | |
| Carbaryl | Eu3+@MOF-253 | red→yellow | 0.2–200 µg/L | 0.14 μg/L | [172] |
| TBZ | Tb3+@MOF | green→blue | 0–80 μM | 0.271 μM | [173] |
| H2S | Tb3+@MOF | blue→green | 10–600 µM | 1.20 μM | [174] |
| H2O | Tb3+@pCDs/MOF | red→green | 0–30% | 0.28% | [175] |
| Pesticides | AuNCs@ZIF-8 | blue→colorless | 0.75 μg/L–100 mg/L | 0.4 μg/L | [158] |
| PA | RGH-Eu(BTC) | orange→yellow | 0–100 μM | 0.45 μM | [176] |
| ALP | SQDs@ZIF-8 | blue→colorless | 0.15–50 U/L | 0.044 U/L | [177] |
| GLP | N-CDs@MOF | blue→colorless | 0.01–6.67 mg/L | 9.06 µg/L | [178] |
| Ammonia | ZnQ@Zn-BTC | pink→green | 0.1–2 mg/L | 0.27 mg/L | [179] |
| Kanamycin | ZIF8@TPE/Aptamer | green→blue | 10–103 ng/mL | 7.3 ng/mL | [180] |
| OTC | NH2-BDC@FMIL-53(Al)-3 | blue→green | 0.3–4.0 μM | 0.18 μM | [181] |
| Targets | MOF | Color Change | Linear Range | LOD | Ref. |
|---|---|---|---|---|---|
| Fe3+ | CD@Eu-MOF | purple→red | 1–200 µM | 0.91 µM | [170] |
| Eu3+/CDs@MOF | red→colorless | 0–6 μM | 0.034 μM | [182] | |
| Tb3+@UiO6(COOH) | green→blue | 0–200 μM | 0.23 μM | [183] | |
| SRB@UiO-66 | red→pink | 0.1–0.9 mM | 3.693 μM | [184] | |
| Dye@bio-MOF-1 | green→dark | 10−5–10−2 M | [185] | ||
| Cu2+ | ZnS QDs@ZIF-8 | yellow→blue | 0.05–5 μM | 16 nM | [186] |
| BPEI-CQDs/ZIF-8 | blue→colorless | 2–1000 nM | 80 pM | [187] | |
| ZTMs@FITC | red→green | 0.1–5 μM | 5.61 nM | [188] | |
| Hg2+ | Eu3+/CDs@MOF-253 | blue→red | 0.065–150 μM | 13 μg/L | [82] |
| CDs@Eu-MOFs | blue→red | 0–300 μM | 0.12 nM | [189] | |
| Ag+ | Eu3+@MIL-121 | colorless→red | 0–100 μM | 0.1 μM | [190] |
| Pb2+ | CDs/QDs@ZIF-8 | red→blue | 0.04–60 μM | [79] | |
| PO43− | CDs/QDs@ZIF-8 | blue→red | 0.25–50 μM | 9.42 nM | [79] |
| Sl. No | Strategy/Approach Adopted | Nanomaterial Used | Colorimetric | LOD | Linear Range | Ref. |
|---|---|---|---|---|---|---|
| 1 | Electrochemical method | Cu-MOF | 14.5 fM | [191] | ||
| 2 | Electrochemical method | Tb-MOF | 4.84 μM | [192] | ||
| 3 | Electrochemical method | Iodide-enhanced Cu-MOF | 200 nM | [193] | ||
| 4 | SPCE platform | C-dots modified screen printed carbon electrode (SPCE) | 0.44 ppm | 0.5–25 ppm | [194] | |
| 5 | Spectrophotometry | Casein-capped gold nanoparticles (AuNPs) | 450 nM | 0.1–0.9 μM | [195] | |
| 6 | Surface-enhanced Raman spectroscopy (SERS) | Phenanthroline probe | 0.001 ppm | 0.01–0.001 ppm | [196] | |
| 7 | Voltammetry (cyclic voltammetry and differential pulse voltammetry) | Quercetin | 3.6 nM | 17.9–716.0 nM | [197] | |
| 8 | Electrochemical method | N, S doped GQD | 0.23 nM | 1–100 nM | [198] | |
| 9 | Ion chromatography | Ionic liquids | 0.09 ppm | 1–100 ppm | [199] | |
| 10 | Interferometric optical microfiber method | Ditrogen- and sulfur-codoped CDs | 0.77 μg/L | 0–300 μg/L | [200] | |
| 11 | Surface plasmon resonance optical detection | CTAB/hydroxylated graphene quantum dots | 0.1 ppm | [201] | ||
| 12 | Fluorescence | CD@Eu-MOF | blue→pink | 0.91 µM | 1–200 µM | [202] |
| 13 | Fluorescence | Acf@bioMOF | yellow→colorless | 1.33 μM | 0–370 μM | [139] |
| 14 | Fluorescence | EY@Zr-MOF | blue→yellow | 0.1 μM | 0–1 mM | [137] |
| Sl. No. | Strategy/Approach Adopted | Methodology in Brief | Advantages | Disadvantages | Ref. |
|---|---|---|---|---|---|
| 1 | Electrochemical method | Studying the interconversion of chemical and electrical energy and the related phenomena and laws in the process of conversion. |
|
| [203,204] |
| 2 | Spectrophotometry | Qualitative and quantitative analysis of a measured substance, by measuring the absorbance of light at a specific wavelength or in a certain wavelength range. |
|
| [205,206,207] |
| 3 | Surface plasmon coupled emission (SPCE) | Prism-coupling technique, where metallic thin film is coupled to the prism and the emission is monitored via a filter, a polarizer, an optic fiber, and a detector. |
|
| [208,209] |
| 4 | Surface-enhanced Raman spectroscopy (SERS) | Determination of samples adsorbed on colloidal metal particles such as silver, gold, or copper, or on the rough surfaces of these metal sheets. |
|
| [210,211,212] |
| 5 | Ion chromatography | It is a liquid chromatographic method for the analysis of anions and cations and belongs to the category of high-performance liquid chromatography (HPLC). |
|
| [213,214,215] |
| 6 | Fluorescence spectroscopy | The emission from the fluorescent molecules present in the cuvette is captured using a detector placed at 90° to the light source. |
|
| [215,216,217,218] |
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Lin, X.; Chen, Q.; Liu, W.; Li, H.; Lin, J.-M. A portable microchip for ultrasensitive and high-throughput assay of thrombin by rolling circle amplification and hemin/G-quadruplex system. Biosens. Bioelectron. 2014, 56, 71–76. [Google Scholar] [CrossRef] [PubMed]
- Chin, C.D.; Laksanasopin, T.; Cheung, Y.K.; Steinmiller, D.; Linder, V.; Parsa, H.; Wang, J.; Moore, H.; Rouse, R.; Umviligihozo, G.; et al. Microfluidics-based diagnostics of infectious diseases in the developing world. Nat. Med. 2011, 17, 1015–1019. [Google Scholar] [CrossRef] [PubMed]
- Lauwers, D.; Hutado, A.G.; Tanevska, V.; Moens, L.; Bersani, D.; Vandenabeele, P. Characterisation of a portable Raman spectrometer for in situ analysis of art objects. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2014, 118, 294–301. [Google Scholar] [CrossRef] [PubMed]
- Lee, W.G.; Kim, Y.-G.; Chung, B.G.; Demirci, U.; Khademhosseini, A. Nano/Microfluidics for diagnosis of infectious diseases in developing countries. Adv. Drug Deliv. Rev. 2010, 62, 449–457. [Google Scholar] [CrossRef] [Green Version]
- Xiang, Y.; Lu, Y. Portable and Quantitative Detection of Protein Biomarkers and Small Molecular Toxins Using Antibodies and Ubiquitous Personal Glucose Meters. Anal. Chem. 2012, 84, 4174–4178. [Google Scholar] [CrossRef] [Green Version]
- Rathnakumar, S.; Bhaskar, S.; Rai, A.; Saikumar, D.V.V.; Kambhampati, N.S.V.; Sivaramakrishnan, V.; Ramamurthy, S.S. Plasmon-Coupled Silver Nanoparticles for Mobile Phone-Based Attomolar Sensing of Mercury Ions. ACS Appl. Nano Mater. 2021, 4, 8066–8080. [Google Scholar] [CrossRef]
- Zheng, P.; Raj, P.; Wu, L.; Szabo, M.; Hanson, W.A.; Mizutani, T.; Barman, I. Leveraging Nanomechanical Perturbations in Raman Spectro-Immunoassays to Design a Versatile Serum Biomarker Detection Platform. Small 2022, 18, e2204541. [Google Scholar] [CrossRef]
- Imran, M.; Ahmed, S.; Abdullah, A.Z.; Hakami, J.; Chaudhary, A.A.; Rudayni, H.A.; Khan, S.; Khan, A.; Basher, N.S. Nanostructured material-based optical and electrochemical detection of amoxicillin antibiotic. Luminescence 2022. [Google Scholar] [CrossRef]
- Ahmed, S.; Ansari, A.; Haidyrah, A.S.; Chaudhary, A.A.; Imran, M.; Khan, A. Hierarchical Molecularly Imprinted Inverse Opal-Based Platforms for Highly Selective and Sensitive Determination of Histamine. ACS Appl. Polym. Mater. 2022, 4, 2783–2793. [Google Scholar] [CrossRef]
- Fang, L.; Liao, X.; Jia, B.; Shi, L.; Kang, L.; Zhou, L.; Kong, W. Recent progress in immunosensors for pesticides. Biosens. Bioelectron. 2020, 164, 112255. [Google Scholar] [CrossRef]
- Zhang, Y.; Wu, Q.; Sun, M.; Zhang, J.; Mo, S.; Wang, J.; Wei, X.; Bai, J. Magnetic-assisted aptamer-based fluorescent assay for allergen detection in food matrix. Sens. Actuators B Chem. 2018, 263, 43–49. [Google Scholar] [CrossRef]
- Luo, L.; Song, Y.; Zhu, C.; Fu, S.; Shi, Q.; Sun, Y.-M.; Jia, B.; Du, D.; Xu, Z.-L.; Lin, Y. Fluorescent silicon nanoparticles-based ratiometric fluorescence immunoassay for sensitive detection of ethyl carbamate in red wine. Sens. Actuators B Chem. 2017, 255, 2742–2749. [Google Scholar] [CrossRef]
- Shamsipur, M.; Molaabasi, F.; Hosseinkhani, S.; Rahmati, F. Detection of Early Stage Apoptotic Cells Based on Label-Free Cytochrome c Assay Using Bioconjugated Metal Nanoclusters as Fluorescent Probes. Anal. Chem. 2016, 88, 2188–2197. [Google Scholar] [CrossRef]
- Bik, E.; Mateuszuk, L.; Stojak, M.; Chlopicki, S.; Baranska, M.; Majzner, K. Menadione-induced endothelial inflammation detected by Raman spectroscopy. Biochim. Et Biophys. Acta (BBA)-Mol. Cell Res. 2020, 1868, 118911. [Google Scholar] [CrossRef]
- Mikac, L.; Kovačević, E.; Ukić, Š.; Raić, M.; Jurkin, T.; Marić, I.; Gotić, M.; Ivanda, M. Detection of multi-class pesticide residues with surface-enhanced Raman spectroscopy. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2021, 252, 119478. [Google Scholar] [CrossRef]
- Boardman, A.K.; Wong, W.S.; Premasiri, W.R.; Ziegler, L.D.; Lee, J.C.; Miljkovic, M.; Klapperich, C.M.; Sharon, A.; Sauer-Budge, A.F. Rapid Detection of Bacteria from Blood with Surface-Enhanced Raman Spectroscopy. Anal. Chem. 2016, 88, 8026–8035. [Google Scholar] [CrossRef] [Green Version]
- Ding, Y.; Zhang, X.; Yin, H.; Meng, Q.; Zhao, Y.; Liu, L.; Wu, Z.; Xu, H. Quantitative and Sensitive Detection of Chloramphenicol by Surface-Enhanced Raman Scattering. Sensors 2017, 17, 2962. [Google Scholar] [CrossRef] [Green Version]
- Liu, Y.; Chen, Y.; Zhang, Y.; Kou, Q.; Zhang, Y.; Wang, Y.; Chen, L.; Sun, Y.; Zhang, H.; MeeJung, Y. Detection and Identification of Estrogen Based on Surface-Enhanced Resonance Raman Scattering (SERRS). Molecules 2018, 23, 1330. [Google Scholar] [CrossRef] [Green Version]
- Lv, M.; Liu, Y.; Geng, J.; Kou, X.; Xin, Z.; Yang, D. Engineering nanomaterials-based biosensors for food safety detection. Biosens. Bioelectron. 2018, 106, 122–128. [Google Scholar] [CrossRef]
- Yarur, F.; Macairan, J.-R.; Naccache, R. Ratiometric detection of heavy metal ions using fluorescent carbon dots. Environ. Sci. Nano 2019, 6, 1121–1130. [Google Scholar] [CrossRef]
- Ze, Y.; Yi, C.; Jing, Z.; Yong, Z. Fluorescent sensor arrays for metal ions detection: A review. Measurement 2021, 187, 110355. [Google Scholar] [CrossRef]
- Guo, Y.; Zhang, L.; Zhang, S.; Yang, Y.; Chen, X.; Zhang, M. Fluorescent carbon nanoparticles for the fluorescent detection of metal ions. Biosens. Bioelectron. 2014, 63, 61–71. [Google Scholar] [CrossRef] [PubMed]
- Sharma, P.; Mehata, M.S. Rapid sensing of lead metal ions in an aqueous medium by MoS2 quantum dots fluorescence turn-off. Mater. Res. Bull. 2020, 131, 110978. [Google Scholar] [CrossRef]
- Singh, J.; Kaur, S.; Lee, J.; Mehta, A.; Kumar, S.; Kim, K.-H.; Basu, S.; Rawat, M. Highly fluorescent carbon dots derived from Mangifera indica leaves for selective detection of metal ions. Sci. Total Environ. 2020, 720, 137604. [Google Scholar] [CrossRef] [PubMed]
- Xu, Q.; Li, J.; Gong, X. Dual-emission carbon dots for sensitive fluorescence detection of metal ions and ethanol in water. Anal. Methods 2022, 14, 3562–3572. [Google Scholar] [CrossRef]
- Ma, S.; Ji, R.; Wang, X.; Yu, C.; Yu, Y.; Yang, X. Fluorescence detection of boscalid pesticide residues in grape juice. Optik 2018, 180, 236–239. [Google Scholar] [CrossRef]
- Watthaisong, P.; Kamutira, P.; Kesornpun, C.; Pongsupasa, V.; Phonbuppha, J.; Tinikul, R.; Maenpuen, S.; Wongnate, T.; Nishihara, R.; Ohmiya, Y.; et al. Luciferin Synthesis and Pesticide Detection by Luminescence Enzymatic Cascades. Angew. Chem. Int. Ed. 2022, 61, e202116908. [Google Scholar] [CrossRef]
- Ashrafi Tafreshi, F.; Fatahi, Z.; Ghasemi, S.F.; Taherian, A.; Esfandiari, N. Ultrasensitive fluorescent detection of pesticides in real sample by using green carbon dots. PLoS ONE 2020, 15, e0230646. [Google Scholar] [CrossRef] [Green Version]
- Huasheng, M.; Mamoun, C.; Xiangyang, W.; Lili, X.; Lijun, Y.; Jianying, H. Fluorescent Detection of Organophosphorus Pesticides Using Carbon Dots Derived from Broccoli. Arab. J. Sci. Eng. 2022, 1–10. [Google Scholar] [CrossRef]
- Lin, B.; Yan, Y.; Guo, M.; Cao, Y.; Yu, Y.; Zhang, T.; Huang, Y.; Wu, D. Modification-free carbon dots as turn-on fluorescence probe for detection of organophosphorus pesticides. Food Chem. 2017, 245, 1176–1182. [Google Scholar] [CrossRef]
- Nsibande, S.A.; Forbes, P.B.C. Fluorescence detection of pesticides using quantum dot materials—A review. Anal. Chim. Acta 2016, 945, 9–22. [Google Scholar] [CrossRef]
- Wang, J.; Zhang, J.; Wang, J.; Fang, G.; Liu, J.; Wang, S. Fluorescent peptide probes for organophosphorus pesticides detection. J. Hazard. Mater. 2020, 389, 122074. [Google Scholar] [CrossRef]
- Yang, J.; Chen, S.-W.; Zhang, B.; Tu, Q.; Wang, J.; Yuan, M.-S. Non-biological fluorescent chemosensors for pesticides detection. Talanta 2022, 240, 123200. [Google Scholar] [CrossRef]
- Haibo, L.; Yumin, W.; Li, Z.; Yujuan, C.; Manli, G.; Ying, Y.; Bixia, L. Construction of integrated and portable fluorescence sensor and the application for visual detection in situ. Sens. Actuators B Chem. 2022, 373, 132764. [Google Scholar] [CrossRef]
- Deb, A.; Nalkar, G.R.; Chowdhury, D. Biogenic carbon dot-based fluorescence-mediated immunosensor for the detection of disease biomarker. Anal. Chim. Acta 2023, 1242, 340808. [Google Scholar] [CrossRef]
- Li, Y.; Jia, D.; Ren, W.; Shi, F.; Liu, C. A Versatile Photoinduced Electron Transfer-Based Upconversion Fluorescent Biosensing Platform for the Detection of Disease Biomarkers and Nerve Agent. Adv. Funct. Mater. 2019, 29, 1903191. [Google Scholar] [CrossRef]
- Sun, Z.-H.; Zhang, X.-X.; Xu, D.; Liu, J.; Yu, R.-J.; Jing, C.; Han, H.-X.; Ma, W. Silver-amplified fluorescence immunoassay via aggregation-induced emission for detection of disease biomarker. Talanta 2020, 225, 121963. [Google Scholar] [CrossRef]
- Yao, B.; Giel, M.-C.; Hong, Y. Detection of kidney disease biomarkers based on fluorescence technology. Mater. Chem. Front. 2021, 5, 2124–2142. [Google Scholar] [CrossRef]
- Yao, C.; Liu, Q.; Zhao, N.; Liu, J.-M.; Fang, G.; Wang, S. Ratiometric determination of Cr(VI) based on a dual-emission fluorescent nanoprobe using carbon quantum dots and a smartphone app. Microchim. Acta 2021, 188, 1–12. [Google Scholar] [CrossRef]
- Kirchon, A.; Feng, L.; Drake, H.F.; Joseph, E.A.; Zhou, H.-C. From fundamentals to applications: A toolbox for robust and multifunctional MOF materials. Chem. Soc. Rev. 2018, 47, 8611–8638. [Google Scholar] [CrossRef]
- Zhou, H.-C.; Long, J.R.; Yaghi, O.M. Introduction to Metal–Organic Frameworks. Chem. Rev. 2012, 112, 673–674. [Google Scholar] [CrossRef] [PubMed]
- Cheng, X.-B.; Zhao, C.-Z.; Yao, Y.-X.; Liu, H.; Zhang, Q. Recent Advances in Energy Chemistry between Solid-State Electrolyte and Safe Lithium-Metal Anodes. Chem 2019, 5, 74–96. [Google Scholar] [CrossRef] [Green Version]
- Yan, F.; Wang, X.; Wang, Y.; Yi, C.; Xu, M.; Xu, J. Sensing performance and mechanism of carbon dots encapsulated into metal–organic frameworks. Microchim. Acta 2022, 189, 379. [Google Scholar] [CrossRef] [PubMed]
- Yin, H.-Q.; Yin, X.-B. Metal-Organic Frameworks with Multiple Luminescence Emissions: Designs and Applications. Acc. Chem. Res. 2020, 53, 485–495. [Google Scholar] [CrossRef]
- Harine, G.; Ajay, V.M.; Rajkumar, P.; Bhaskar, R.S. Metal-Organic Framework Derived Carbon-based Electrocatalysis for Hydrogen Evolution Reactions: A Review. Mater. Today Sustain. 2023. [Google Scholar] [CrossRef]
- Kundu, S.; Swaroop, A.K.; Selvaraj, J. Metal-Organic Framework in Pharmaceutical Drug Delivery. Curr. Top. Med. Chem. 2023. [Google Scholar] [CrossRef]
- Li, B.; Dong, J.-P.; Zhou, Z.; Wang, R.; Wang, L.-Y.; Zang, S.-Q. Robust lanthanide metal–organic frameworks with “all-in-one” multifunction: Efficient gas adsorption and separation, tunable light emission and luminescence sensing. J. Mater. Chem. C 2021, 9, 3429–3439. [Google Scholar] [CrossRef]
- Kaur, H.; Sundriyal, S.; Pachauri, V.; Ingebrandt, S.; Kim, K.-H.; Sharma, A.L.; Deep, A. Luminescent metal-organic frameworks and their composites: Potential future materials for organic light emitting displays. Coord. Chem. Rev. 2019, 401, 213077. [Google Scholar] [CrossRef]
- Lustig, W.P.; Li, J. Luminescent metal–organic frameworks and coordination polymers as alternative phosphors for energy efficient lighting devices. Coord. Chem. Rev. 2018, 373, 116–147. [Google Scholar] [CrossRef]
- Dong, J.; Zhao, D.; Lu, Y.; Sun, W.-Y. Photoluminescent metal–organic frameworks and their application for sensing biomolecules. J. Mater. Chem. A 2019, 7, 22744–22767. [Google Scholar] [CrossRef]
- Zhang, Y.; Yuan, S.; Day, G.; Wang, X.; Yang, X.; Zhou, H.-C. Luminescent sensors based on metal-organic frameworks. Coord. Chem. Rev. 2018, 354, 28–45. [Google Scholar] [CrossRef]
- Hardian, R.; Dissegna, S.; Ullrich, A.; Llewellyn, P.L.; Coulet, M.-V.; Fischer, R.A. Tuning the Properties of MOF-808 via Defect Engineering and Metal Nanoparticle Encapsulation. Chem. Eur. J. 2021, 27, 6804–6814. [Google Scholar] [CrossRef]
- Liu, X.; Hu, H.; Liu, Y.; Huang, Z.; Lu, Y.; Zhou, X.; Wang, J. Experimental investigation on fluorescence polarization properties of isomerical MOF⊃RhB crystals. J. Solid State Chem. 2020, 284, 121179. [Google Scholar] [CrossRef]
- Qiu, L.; Yu, C.; Wang, X.; Xie, Y.; Kirillov, A.M.; Huang, W.; Li, J.; Gao, P.; Wu, T.; Gu, X.; et al. Tuning the Solid-State White Light Emission of Postsynthetic Lanthanide-Encapsulated Double-Layer MOFs for Three-Color Luminescent Thermometry Applications. Inorg. Chem. 2019, 58, 4524–4533. [Google Scholar] [CrossRef]
- Li, G.; Zhao, S.; Zhang, Y.; Tang, Z. Metal–Organic Frameworks Encapsulating Active Nanoparticles as Emerging Composites for Catalysis: Recent Progress and Perspectives. Adv. Mater. 2018, 30, e1800702. [Google Scholar] [CrossRef]
- Mollick, S.; Mandal, T.N.; Jana, A.; Fajal, S.; Desai, A.V.; Ghosh, S.K. Ultrastable Luminescent Hybrid Bromide [email protected] Nanocomposites for the Degradation of Organic Pollutants in Water. ACS Appl. Energy Mater. 2019, 2, 1333–1340. [Google Scholar] [CrossRef]
- Xu, Z.; Zhang, J.; Pan, T.; Li, H.; Huo, F.; Zheng, B.; Zhang, W. Encapsulation of Hydrophobic Guests within Metal–Organic Framework Capsules for Regulating Host–Guest Interaction. Chem. Mater. 2020, 32, 3553–3560. [Google Scholar] [CrossRef]
- Peng, M.; Kaczmarek, A.M.; Van Hecke, K. Ratiometric Thermometers Based on Rhodamine B and Fluorescein Dye-Incorporated (Nano) Cyclodextrin Metal–Organic Frameworks. ACS Appl. Mater. Interfaces 2022, 14, 14367–14379. [Google Scholar] [CrossRef]
- Jiang, X.; Fan, R.; Zhang, J.; Fang, X.; Sun, T.; Zhu, K.; Zhou, X.; Xu, Y.; Yang, Y. Sequentially epitaxial growth multi-guest encapsulation strategy in MOF-on-MOF platform: Biogenic amine detection and systematic white light adjustment. Chem. Eng. J. 2022, 436, 135236. [Google Scholar] [CrossRef]
- Xia, Q.Q.; Wang, X.H.; Yu, J.L.; Xue, Z.Y.; Chai, J.; Wu, M.X.; Liu, X.M. Tunable fluorescence emission based on multi-layered MOF-on-MOF. Dalton Trans. 2022, 51, 9397–9403. [Google Scholar] [CrossRef]
- Xu, D.-D.; Dong, W.-W.; Li, M.-K.; Han, H.-M.; Zhao, J.; Li, D.-S.; Zhang, Q. Encapsulating Organic Dyes in Metal–Organic Frameworks for Color-Tunable and High-Efficiency White-Light-Emitting Properties. Inorg. Chem. 2022, 61, 21107–21114. [Google Scholar] [CrossRef] [PubMed]
- Yi, K.; Zhang, X.; Zhang, L. Eu3+@metal-organic frameworks encapsulating carbon dots as ratiometric fluorescent probes for rapid recognition of anthrax spore biomarker. Sci. Total Environ. 2020, 743, 140692. [Google Scholar] [CrossRef] [PubMed]
- Yin, H.-Q.; Yang, J.-C.; Yin, X.-B. Ratiometric Fluorescence Sensing and Real-Time Detection of Water in Organic Solvents with One-Pot Synthesis of [email protected](Al)–NH2. Anal. Chem. 2017, 89, 13434–13440. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.; Zhang, X.; Bai, J.; Huang, K.; Ren, L. Facile, controllable tune of blue shift or red shift of the fluorescence emission of solid-state carbon dots. Chem. Eng. J. 2019, 374, 787–792. [Google Scholar] [CrossRef]
- Rodrigues, M.O.; Paz, F.A.A.; Freire, R.O.; de Sá, G.F.; Galembeck, A.; Montenegro, M.C.B.S.M.; Araújo, A.N.; Alves, S. Modeling, Structural, and Spectroscopic Studies of Lanthanide-Organic Frameworks. J. Phys. Chem. B 2009, 113, 12181–12188. [Google Scholar] [CrossRef]
- Yin, Z.; Wan, S.; Yang, J.; Kurmoo, M.; Zeng, M.-H. Recent advances in post-synthetic modification of metal–organic frameworks: New types and tandem reactions. Coord. Chem. Rev. 2017, 378, 500–512. [Google Scholar] [CrossRef]
- Wang, F.; Zhang, F.; Zhao, Z.; Sun, Z.; Pu, Y.; Wang, Y.; Wang, X. Multifunctional MOF-based probes for efficient detection and discrimination of Pb2+, Fe3+ and Cr2O72−/CrO42−. Dalton Trans. 2021, 50, 12197–12207. [Google Scholar] [CrossRef]
- Liang, Y.-Y.; Luo, L.-J.; Li, Y.; Ling, B.-K.; Chen, B.-W.; Wang, X.-W.; Luan, T.-G. A Luminescent Probe for Highly Selective Cu2+ Sensing Using a Lanthanide-Doped Metal Organic Framework with Large Pores. Eur. J. Inorg. Chem. 2018, 2019, 206–211. [Google Scholar] [CrossRef]
- Xiao, J.; Song, L.; Liu, M.; Wang, X.; Liu, Z. Intriguing pH-modulated Luminescence Chameleon System based on Postsynthetic Modified Dual-emitting Eu3+@Mn-MOF and Its Application for Histidine Chemosensor. Inorg. Chem. 2020, 59, 6390–6397. [Google Scholar] [CrossRef]
- Luo, J.; Liu, B.-S.; Zhang, X.-R.; Liu, R.-T. A Eu3+ post-functionalized metal-organic framework as fluorescent probe for highly selective sensing of Cu2+ in aqueous media. J. Mol. Struct. 2019, 1177, 444–448. [Google Scholar] [CrossRef]
- Xu, X.-Y.; Yan, B. Eu(III)-Functionalized MIL-124 as Fluorescent Probe for Highly Selectively Sensing Ions and Organic Small Molecules Especially for Fe(III) and Fe(II). ACS Appl. Mater. Interfaces 2014, 7, 721–729. [Google Scholar] [CrossRef]
- Ji, G.; Gao, X.; Zheng, T.; Guan, W.; Liu, H.; Liu, Z. Postsynthetic Metalation Metal–Organic Framework as a Fluorescent Probe for the Ultrasensitive and Reversible Detection of PO43– Ions. Inorg. Chem. 2018, 57, 10525–10532. [Google Scholar] [CrossRef]
- Wu, J.-X.; Yan, B. Luminescent Hybrid Tb3+ Functionalized Metal–Organic Frameworks Act as Food Preservative Sensor and Water Scavenger for NO2–. Ind. Eng. Chem. Res. 2018, 57, 7105–7111. [Google Scholar] [CrossRef]
- Wang, Y.; Yang, H.; Cheng, G.; Wu, Y.; Lin, S. A new Tb(iii)-functionalized layer-like Cd MOF as luminescent probe for high-selectively sensing of Cr3+. CrystEngComm 2017, 19, 7270–7276. [Google Scholar] [CrossRef]
- Yu, W.W.; Qu, L.; Guo, W.; Peng, X. Experimental Determination of the Extinction Coefficient of CdTe, CdSe, and CdS Nanocrystals. Chem. Mater. 2003, 15, 2854–2860. [Google Scholar] [CrossRef]
- Bajorowicz, B.; Kobylański, M.P.; Gołąbiewska, A.; Nadolna, J.; Zaleska-Medynska, A.; Malankowska, A. Quantum dot-decorated semiconductor micro- and nanoparticles: A review of their synthesis, characterization and application in photocatalysis. Adv. Colloid Interface Sci. 2018, 256, 352–372. [Google Scholar] [CrossRef]
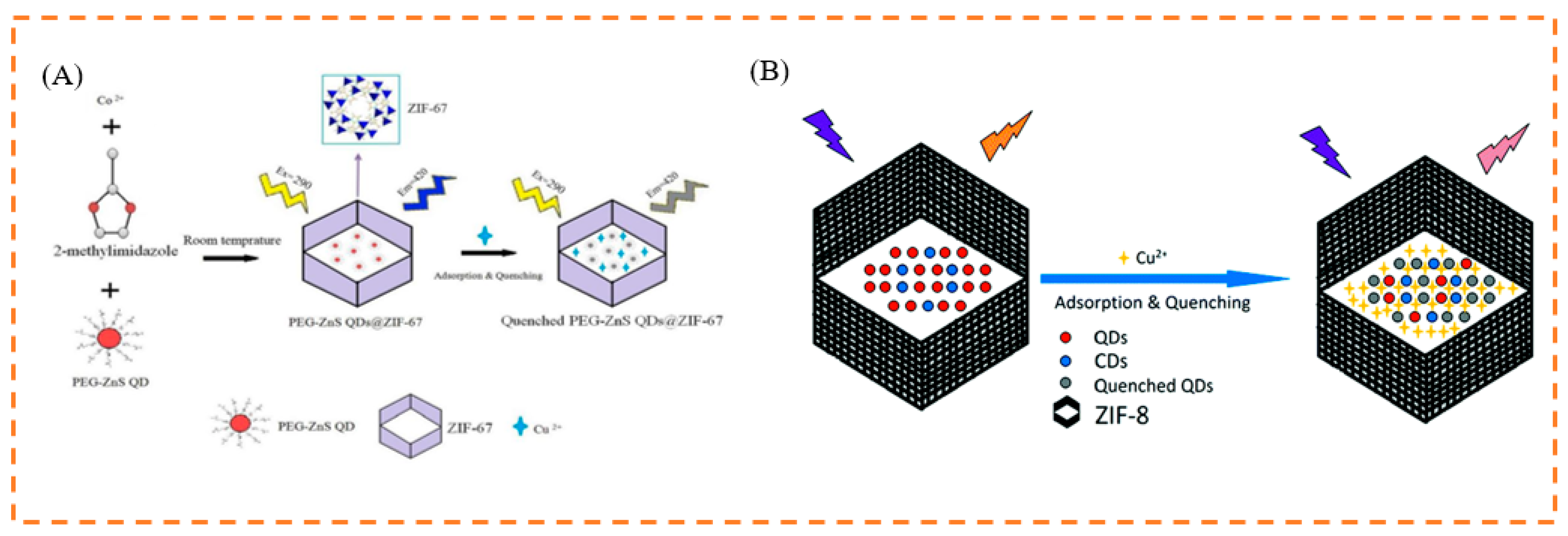
- Asadi, F.; Azizi, S.N.; Chaichi, M.J. Green synthesis of fluorescent PEG-ZnS QDs encapsulated into Co-MOFs as an effective sensor for ultrasensitive detection of copper ions in tap water. Mater. Sci. Eng. C 2019, 105, 110058. [Google Scholar] [CrossRef]
- Ma, Y.; Xu, G.; Wei, F.; Cen, Y.; Ma, Y.; Song, Y.; Xu, X.; Shi, M.; Muhammad, S.; Hu, Q. A dual-emissive fluorescent sensor fabricated by encapsulating quantum dots and carbon dots into metal–organic frameworks for the ratiometric detection of Cu2+ in tap water. J. Mater. Chem. C 2017, 5, 8566–8571. [Google Scholar] [CrossRef]
- Yi, K.; Zhang, L. Embedding dual fluoroprobe in metal-organic frameworks for continuous visual recognition of Pb2+ and PO43- via fluorescence 'turn-off-on' response: Agar test paper and fingerprint. J. Hazard. Mater. 2020, 389, 122141. [Google Scholar] [CrossRef]
- Ma, C.; Li, P.; Xia, L.; Qu, F.; Kong, R.-M.; Song, Z.-L. A novel ratiometric fluorescence nanoprobe for sensitive determination of uric acid based on CD@ZIF-CuNC nanocomposites. Microchim. Acta 2021, 188, 259. [Google Scholar] [CrossRef]
- Hao, J.; Liu, F.; Liu, N.; Zeng, M.; Song, Y.; Wang, L. Ratiometric fluorescent detection of Cu2+ with carbon dots chelated Eu-based metal-organic frameworks. Sens. Actuators B Chem. 2017, 245, 641–647. [Google Scholar] [CrossRef]
- Xu, X.-Y.; Yan, B. Fabrication and application of a ratiometric and colorimetric fluorescent probe for Hg2+ based on dual-emissive metal–organic framework hybrids with carbon dots and Eu3+. J. Mater. Chem. C 2016, 4, 1543–1549. [Google Scholar] [CrossRef]
- Chen, R.; Zhang, J.; Chelora, J.; Xiong, Y.; Kershaw, S.V.; Li, K.F.; Lo, P.-K.; Cheah, K.W.; Rogach, A.L.; Zapien, J.A.; et al. Ruthenium(II) Complex Incorporated UiO-67 Metal–Organic Framework Nanoparticles for Enhanced Two-Photon Fluorescence Imaging and Photodynamic Cancer Therapy. ACS Appl. Mater. Interfaces 2017, 9, 5699–5708. [Google Scholar] [CrossRef] [PubMed]
- Sun, C.-Y.; Wang, X.-L.; Zhang, X.; Qin, C.; Li, P.; Su, Z.-M.; Zhu, D.-X.; Shan, G.-G.; Shao, K.-Z.; Wu, H.; et al. Efficient and tunable white-light emission of metal-organic frameworks by iridium-complex encapsulation. Nat. Commun. 2013, 4, 2717. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhao, H.; Ni, J.; Zhang, J.-J.; Liu, S.-Q.; Sun, Y.-J.; Zhou, H.; Li, Y.-Q.; Duan, C.-Y. A trichromatic MOF composite for multidimensional ratiometric luminescent sensing. Chem. Sci. 2018, 9, 2918–2926. [Google Scholar] [CrossRef] [Green Version]
- Zhu, S.; Wang, S.; Xia, M.; Wang, B.; Huang, Y.; Zhang, D.; Zhang, X.; Wang, G. Intracellular Imaging of Glutathione with MnO2 Nanosheet@Ru(bpy)32+-UiO-66 Nanocomposites. ACS Appl. Mater. Interfaces 2019, 11, 31693–31699. [Google Scholar] [CrossRef]
- Fu, Y.; Finney, N.S. Small-molecule fluorescent probes and their design. RSC Adv. 2018, 8, 29051–29061. [Google Scholar] [CrossRef] [Green Version]
- Traven, V.F.; Cheptsov, D.A. Sensory effects of fluorescent organic dyes. Russ. Chem. Rev. 2020, 89, 713–749. [Google Scholar] [CrossRef]
- Xu, X.-Y.; Yan, B.; Lian, X. Wearable glove sensor for non-invasive organophosphorus pesticide detection based on a double-signal fluorescence strategy. Nanoscale 2018, 10, 13722–13729. [Google Scholar] [CrossRef]
- Liu, N.; Hao, J.; Chen, L.; Song, Y.; Wang, L. Ratiometric fluorescent detection of Cu2+ based on dual-emission ZIF-8@rhodamine-B nanocomposites. Luminescence 2019, 34, 193–199. [Google Scholar] [CrossRef]
- Feng, D.Y.; Zhang, T.; Zhong, T.Y.; Zhang, C.; Tian, Y.Y.; Wang, G. Coumarin-embedded MOF UiO-66 as a selective and sensitive fluorescent sensor for the recognition and detection of Fe3+ ions. J. Mater. Chem. C 2021, 9, 16978–16984. [Google Scholar] [CrossRef]
- Li, H.; Fu, F.; Yang, W.; Ding, L.; Dong, J.; Yang, Y.; Wang, F.; Pan, Q. A simple fluorescent probe for fast and sensitive detection of inorganic phosphate based on [email protected] composite. Sens. Actuators B Chem. 2019, 301, 127110. [Google Scholar] [CrossRef]
- Huang, C.J.; Ye, Y.X.; Zhao, L.W.; Li, Y.S.; Gu, J.L. One-Pot Trapping Luminescent Rhodamine 110 into the Cage of MOF-801 for Nitrite Detection in Aqueous Solution. J. Inorg. Organomet. Polym. Mater. 2019, 29, 1476–1484. [Google Scholar] [CrossRef]
- Jin, R. Quantum sized, thiolate-protected gold nanoclusters. Nanoscale 2009, 2, 343–362. [Google Scholar] [CrossRef] [PubMed]
- Burrows, P.E.; Sapochak, L.S.; McCarty, D.M.; Forrest, S.R.; Thompson, M.E. Metal ion dependent luminescence effects in metal tris-quinolate organic heterojunction light emitting devices. Appl. Phys. Lett. 1994, 64, 2718–2720. [Google Scholar] [CrossRef]
- Goswami, N.; Lin, F.; Liu, Y.; Leong, D.T.; Xie, J. Highly Luminescent Thiolated Gold Nanoclusters Impregnated in Nanogel. Chem. Mater. 2016, 28, 4009–4016. [Google Scholar] [CrossRef]
- Luo, Z.; Yuan, X.; Yu, Y.; Zhang, Q.; Leong, D.T.; Lee, J.Y.; Xie, J. From Aggregation-Induced Emission of Au(I)–Thiolate Complexes to Ultrabright Au(0)@Au(I)–Thiolate Core–Shell Nanoclusters. J. Am. Chem. Soc. 2012, 134, 16662–16670. [Google Scholar] [CrossRef]
- Han, B.; Hu, X.; Yu, M.; Peng, T.; Li, Y.; He, G. One-pot synthesis of enhanced fluorescent copper nanoclusters encapsulated in metal–organic frameworks. RSC Adv. 2018, 8, 22748–22754. [Google Scholar] [CrossRef] [Green Version]
- Pirot, S.M.; Omer, K.M. Designing of robust and sensitive assay via encapsulation of highly emissive and stable blue copper nanocluster into zeolitic imidazole framework (ZIF-8) with quantitative detection of tetracycline. J. Anal. Sci. Technol. 2022, 13, 22. [Google Scholar] [CrossRef]
- Chen, H.; Chang, Y.; Wei, R.; Zhang, P. Gold nanoclusters encapsulated into zinc-glutamate metal organic frameworks for efficient detection of H2O2. Anal. Methods 2022, 14, 1439–1444. [Google Scholar] [CrossRef]
- Liu, P.; Hao, R.; Sun, W.; Lin, Z.; Jing, T. One-pot synthesis of copper nanocluster/Tb-MOF composites for the ratiometric fluorescence detection of Cu2+. Luminescence 2022, 37, 1793–1799. [Google Scholar] [CrossRef]
- Jalili, R.; Irani-Nezhad, M.H.; Khataee, A.; Joo, S.W. A ratiometric fluorescent probe based on carbon dots and gold nanocluster encapsulated metal–organic framework for detection of cephalexin residues in milk. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2021, 262, 120089. [Google Scholar] [CrossRef]
- Hu, J.; Cui, X.; Gong, Y.; Xu, X.; Gao, B.; Wen, T.; Lu, T.J.; Xu, F. Portable microfluidic and smartphone-based devices for monitoring of cardiovascular diseases at the point of care. Biotechnol. Adv. 2016, 34, 305–320. [Google Scholar] [CrossRef]
- Jongs, N.; Jagesar, R.; Koning, I.; Ruhe, H.; Van Haren, N.; Vorstman, J.; Kas, M. Passive behavioural monitoring in neuropsychiatric disorders using smartphone technology. Eur. Neuropsychopharmacol. 2018, 28, S87–S88. [Google Scholar] [CrossRef]
- Majumder, S.; Deen, M.J. Smartphone Sensors for Health Monitoring and Diagnosis. Sensors 2019, 19, 2164. [Google Scholar] [CrossRef] [Green Version]
- Moses, J.C.; Adibi, S.; Shariful Islam, S.M.; Wickramasinghe, N.; Nguyen, L. Application of Smartphone Technologies in Disease Monitoring: A Systematic Review. Healthcare 2021, 9, 889. [Google Scholar] [CrossRef]
- Andrachuk, M.; Marschke, M.; Hings, C.; Armitage, D. Smartphone technologies supporting community-based environmental monitoring and implementation: A systematic scoping review. Biol. Conserv. 2019, 237, 430–442. [Google Scholar] [CrossRef]
- Chen, Z.s.; Liu, T.; Dong, J.f.; Chen, G.; Li, Z.; Zhou, J.l.; Chen, Z. Sustainable Application for Agriculture Using Biochar-Based Slow-Release Fertilizers: A Review. ACS Sustain. Chem. Eng. 2023, 11, 1–12. [Google Scholar] [CrossRef]
- Ramar, R.; Malaichamy, I. Simple smartphone merged rapid colorimetric platform for the environmental monitoring of toxic sulfide ions by cysteine functionalized silver nanoparticles. Microchem. J. 2021, 174, 107071. [Google Scholar] [CrossRef]
- Wang, Y.; Tan, R.; Xing, G.; Wang, J.; Tan, X.; Liu, X. Energy-Efficient Aquatic Environment Monitoring Using Smartphone-Based Robots. ACM Trans. Sens. Netw. 2016, 12, 1–28. [Google Scholar] [CrossRef]
- Deng, C.C.; Xu, Z.Y.; Sun, Z.; Xie, J.H.; Luo, H.Q.; Li, N.B. One-step synthesis of aldehyde-functionalized dual-emissive carbon dots for ratiometric fluorescence detection of bisulfite in food samples. Food Chem. 2022, 405, 134961. [Google Scholar] [CrossRef] [PubMed]
- Rateni, G.; Dario, P.; Cavallo, F. Smartphone-Based Food Diagnostic Technologies: A Review. Sensors 2017, 17, 1453. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Huang, H.; Song, G.; Huang, K.; Luo, Y.; Liu, Q.; He, X.; Cheng, N. Intelligent biosensing strategies for rapid detection in food safety: A review. Biosens. Bioelectron. 2022, 202, 114003. [Google Scholar] [CrossRef] [PubMed]
- Bhaskar, S.; Ramamurthy, S.S. Mobile Phone-Based Picomolar Detection of Tannic Acid on Nd2O3 Nanorod–Metal Thin-Film Interfaces. ACS Appl. Energy Mater. 2019, 2, 4613–4625. [Google Scholar] [CrossRef]
- Aayush, R.; Seemesh, B.; Kalathur Mohan, G.; Sai Sathish, R. Engineering of coherent plasmon resonances from silver soret colloids, graphene oxide and Nd2O3 nanohybrid architectures studied in mobile phone-based surface plasmon-coupled emission platform. Mater. Lett. 2021, 304, 130632. [Google Scholar] [CrossRef]
- Rai, A.; Bhaskar, S.; Reddy, N.; Ramamurthy, S.S. Cellphone-Aided Attomolar Zinc Ion Detection Using Silkworm Protein-Based Nanointerface Engineering in a Plasmon-Coupled Dequenched Emission Platform. ACS Sustain. Chem. Eng. 2021, 9, 14959–14974. [Google Scholar] [CrossRef]
- Seemesh, B.; Dipin, T.; Sai Sathish, R.; Chandramouli, S. Metal–Dielectric Interfacial Engineering with Mesoporous Nano-Carbon Florets for 1000-Fold Fluorescence Enhancements: Smartphone-Enabled Visual Detection of Perindopril Erbumine at a Single-molecular Level. ACS Sustain. Chem. Eng. 2022, 11, 78–91. [Google Scholar] [CrossRef]
- Bhaskar, S.; Ramamurthy, S.S. Synergistic coupling of titanium carbonitride nanocubes and graphene oxide for 800-fold fluorescence enhancements on smartphone based surface plasmon-coupled emission platform. Mater. Lett. 2021, 298, 130008. [Google Scholar] [CrossRef]
- Xiaoting, Z.; Ying, L.; Lei, Z. Developed ratiometric fluorescent probe as a logic platform for potential diagnosis of thyroid disease and diabetes and fluorescent ink. Microchem. J. 2021, 171, 106879. [Google Scholar] [CrossRef]
- Kou, X.; Tong, L.; Shen, Y.; Zhu, W.; Yin, L.; Huang, S.; Zhu, F.; Chen, G.; Ouyang, G. Smartphone-assisted robust enzymes@MOFs-based paper biosensor for point-of-care detection. Biosens. Bioelectron. 2020, 156, 112095. [Google Scholar] [CrossRef]
- Li, Z.; Xi, Y.; Zhao, A.; Jiang, J.; Li, B.; Yang, X.; He, J.; Li, F. Cobalt-imidazole metal-organic framework loaded with luminol for paper-based chemiluminescence detection of catechol with use of a smartphone. Anal. Bioanal. Chem. 2021, 413, 3541–3550. [Google Scholar] [CrossRef]
- Yang, L.; Liu, Y.; Chen, L.; Guo, L.; Lei, Y.; Wang, L. Stable dual-emissive fluorescin@UiO-67 metal-organic frameworks for visual and ratiometric sensing of Al3+ and ascorbic acid. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2021, 261, 120068. [Google Scholar] [CrossRef]
- Wang, B.H.; Yan, B. A dye@MOF crystalline probe serving as a platform for ratiometric sensing of trichloroacetic acid (TCA), a carcinogen metabolite in human urine. CrystEngComm 2019, 21, 4637–4643. [Google Scholar] [CrossRef]
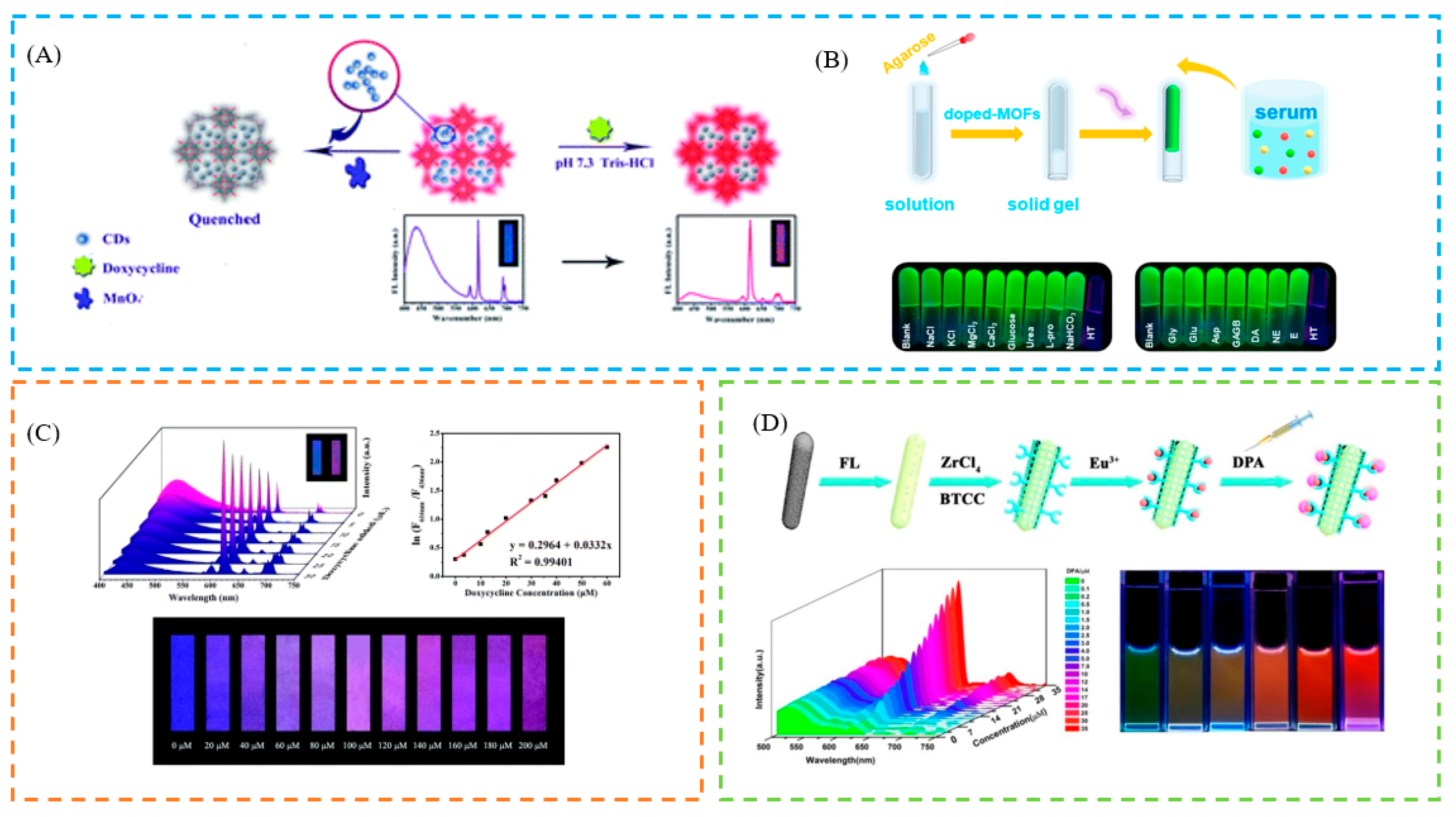
- Fu, X.; Lv, R.; Su, J.; Li, H.; Yang, B.; Gu, W.; Liu, X. A dual-emission nano-rod MOF equipped with carbon dots for visual detection of doxycycline and sensitive sensing of MnO4−. RSC Adv. 2018, 8, 4766–4772. [Google Scholar] [CrossRef] [Green Version]
- Wang, L.-B.; Wang, J.-J.; Yue, E.-L.; Tang, L.; Wang, X.; Hou, X.-Y.; Zhang, Y.; Ren, Y.-X.; Chen, X.-L. Highly selective detecting Aspartic acid, detecting Ornidazole and information encryption and decryption supported by a heterometallic anionic Cd (II)-K (I)-MOF. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2021, 265, 120340. [Google Scholar] [CrossRef]
- Xiaomeng, Z.; Xinjie, W.; Li, S. Ratiometric fluorescence and visual sensing of ATP based on gold nanocluster-encapsulated metal-organic framework with a smartphone. Chin. Chem. Lett. 2022. [Google Scholar] [CrossRef]
- Xiang-Juan, K.; Jing-Xuan, T.; Yan-Zhao, F.; Tao-Li, C.; Rui, Y.; Jia-Yu, H.; Zi-Yan, Z.; Qiang, X. Terbium metal-organic framework/bovine serum albumin capped gold nanoclusters-based dual-emission reverse change ratio fluorescence nanoplatform for fluorimetric and colorimetric sensing of heparin and chondroitin sulfate. Sens. Actuators B Chem. 2021, 356, 131331. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhang, Y.; Li, L.; Chen, J.; Li, P.; Huang, W. One-step in situ growth of high-density POMOFs films on carbon cloth for the electrochemical detection of bromate. J. Electroanal. Chem. 2020, 861, 113939. [Google Scholar] [CrossRef]
- Wang, C.; Tian, L.; Zhu, W.; Wang, S.Q.; Wang, P.; Liang, Y.; Zhang, W.L.; Zhao, H.W.; Li, G.T. Dye@bio-MOF-1 Composite as a Dual-Emitting Platform for Enhanced Detection of a Wide Range of Explosive Molecules. Acs Appl. Mater. Interfaces 2017, 9, 20076–20085. [Google Scholar] [CrossRef]
- Wang, H.J.; Sha, Z.J. Preparation of copper net-supported metal-organic framework-5 membranes for solid-state lasers. Sci. China-Chem. 2011, 54, 947–950. [Google Scholar] [CrossRef]
- Gao, N.; Huang, J.; Wang, L.Y.; Feng, J.Y.; Huang, P.C.; Wu, F.Y. Ratiometric fluorescence detection of phosphate in human serum with a metal-organic frameworks-based nanocomposite and its immobilized agarose hydrogels. Appl. Surf. Sci. 2018, 459, 686–692. [Google Scholar] [CrossRef]
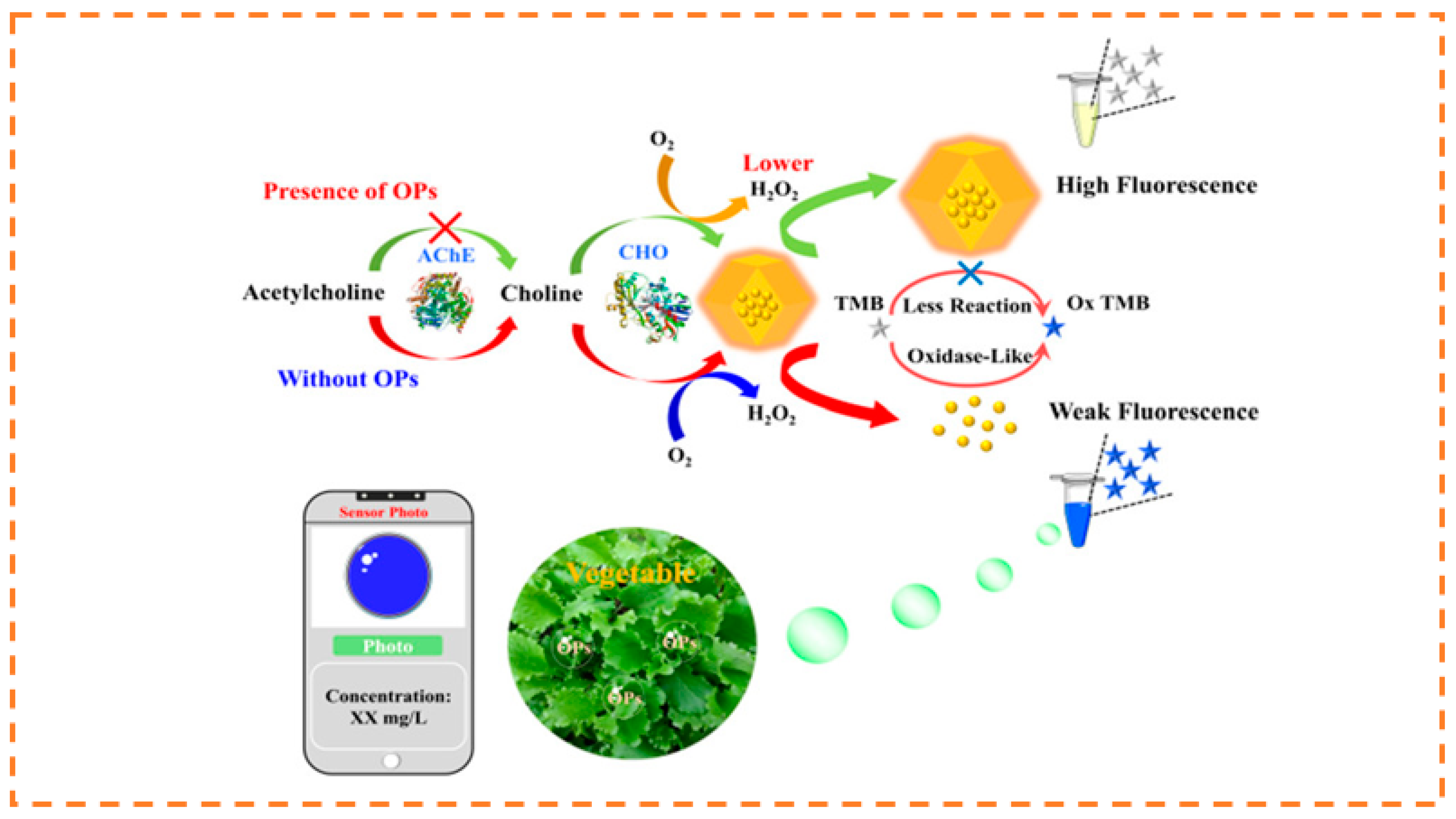
- Wei, D.; Li, M.; Wang, Y.; Zhu, N.; Hu, X.; Zhao, B.; Zhang, Z.; Yin, D. Encapsulating gold nanoclusters into metal–organic frameworks to boost luminescence for sensitive detection of copper ions and organophosphorus pesticides. J. Hazard. Mater. 2023, 441, 129890. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.N.; Rusling, J.F.; Papadimitrakopoulos, F. Carbon Nanotubes for Electronic and Electrochemical Detection of Biomolecules. Adv. Mater. 2007, 19, 3214–3228. [Google Scholar] [CrossRef] [Green Version]
- Chu, N.-C.; Taylor, R.N.; Chavagnac, V.R.; Nesbitt, R.W.; Boella, R.M.; Milton, J.A.; German, C.R.; Bayon, G.; Burton, K. Hf isotope ratio analysis using multi-collector inductively coupled plasma mass spectrometry: An evaluation of isobaric interference corrections. J. Anal. At. Spectrom. 2002, 17, 1567–1574. [Google Scholar] [CrossRef] [Green Version]
- Willis, J.B. Determination of Lead in Urine by Atomic Absorption Spectroscopy. Nature 1961, 191, 381–382. [Google Scholar] [CrossRef]
- Tao, Y.; Jiang, Y.; Huang, Y.; Liu, J.; Zhang, P.; Chen, X.; Fan, Y.; Wang, L.; Xu, J. Carbon dots@metal–organic frameworks as dual-functional fluorescent sensors for Fe3+ ions and nitro explosives. CrystEngComm 2021, 23, 4038–4049. [Google Scholar] [CrossRef]
- Li, Y.K.; Wei, Z.H.; Zhang, Y.; Guo, Z.F.; Chen, D.S.; Jia, P.Y.; Chen, P.; Xing, H.Z. Dual-Emitting EY@Zr-MOF Composite as Self-Calibrating Luminescent Sensor for Selective Detection of Inorganic Ions and Nitroaromatics. Acs Sustain. Chem. Eng. 2019, 7, 6196–6203. [Google Scholar] [CrossRef]
- Liu, M.F.; Yu, X.; Zhong, K.X.; Chen, X.Y.; Feng, L.J.; Yao, S. Dye-encapsulated nanocage-based metal-organic frameworks as luminescent dual-emitting sensors for selective detection of inorganic ions. Appl. Organomet. Chem. 2022, 36, e6692. [Google Scholar] [CrossRef]
- Liu, W.; Li, S.Q.; Shao, J.; Tian, J.L. A dual-emission Acf@bioMOF-1 platform as fluorescence sensor for highly efficient detection of inorganic ions. J. Solid State Chem. 2020, 290, 121580. [Google Scholar] [CrossRef]
- Zhang, Z.N.; Wei, Z.H.; Meng, F.Y.; Su, J.L.; Chen, D.S.; Guo, Z.F.; Xing, H.Z. RhB-Embedded Zirconium-Naphthalene-Based Metal-Organic Framework Composite as a Luminescent Self-Calibrating Platform for the Selective Detection of Inorganic Ions. Chem. Eur. J. 2020, 26, 1661–1667. [Google Scholar] [CrossRef]
- Que, E.L.; Domaille, D.W.; Chang, C.J. Metals in Neurobiology: Probing Their Chemistry and Biology with Molecular Imaging. Chem. Rev. 2008, 108, 4328. [Google Scholar] [CrossRef]
- Bora, T.; Aksoy, Ç.; Tunay, Z.; Aydın, F. Determination of trace elements in illicit spice samples by using ICP-MS. Microchem. J. 2015, 123, 179–184. [Google Scholar] [CrossRef]
- Cheng, W.-L.; Sue, J.-W.; Chen, W.-C.; Chang, J.-L.; Zen, J.-M. Activated Nickel Platform for Electrochemical Sensing of Phosphate. Anal. Chem. 2009, 82, 1157–1161. [Google Scholar] [CrossRef]
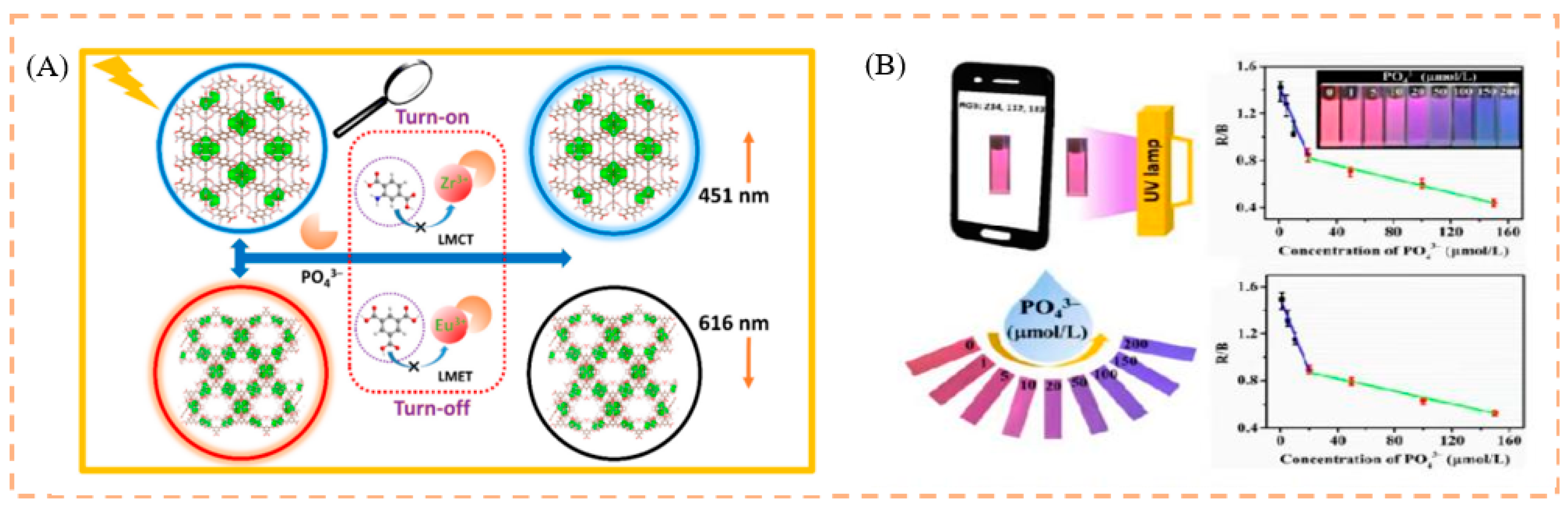
- Yi, K.; Zhang, X.; Zhang, L. Smartphone-based ratiometric fluorescent definable system for phosphate by merged metal−organic frameworks. Sci. Total Environ. 2021, 772, 144952. [Google Scholar] [CrossRef]
- Zhang, R.; Song, B.; Yuan, J. Bioanalytical Methods for Hypochlorous Acid Detection: Recent Advances and Challenges. Trends Anal. Chem. 2017, 99, 1–33. [Google Scholar] [CrossRef]
- Liu, L.; Zhu, G.; Zeng, W.; Lv, B.; Yi, Y. Highly sensitive and selective “off-on” fluorescent sensing platform for ClO− in water based on silicon quantum dots coupled with nanosilver. Anal. Bioanal. Chem. 2019, 411, 1561–1568. [Google Scholar] [CrossRef]
- Zhang, J.; Yang, X. A simple yet effective chromogenic reagent for the rapid estimation of bromate and hypochlorite in drinking water. Analyst 2012, 138, 434–437. [Google Scholar] [CrossRef]
- Xiong, J.; Xiao, Y.; Liang, J.; Sun, J.; Gao, L.; Zhou, Q.; Hong, D.; Tan, K. Dye-based dual-emission Eu-MOF synthesized by Post-modification for the sensitive ratio fluorescence visualization sensing of ClO. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2023, 285, 121863. [Google Scholar] [CrossRef]
- Broza, Y.Y.; Zhou, X.; Yuan, M.; Qu, D.; Zheng, Y.; Vishinkin, R.; Khatib, M.; Wu, W.; Haick, H. Disease Detection with Molecular Biomarkers: From Chemistry of Body Fluids to Nature-Inspired Chemical Sensors. Chem. Rev. 2019, 119, 11761–11817. [Google Scholar] [CrossRef]
- Turtoi, A.; Dumont, B.; Greffe, Y.; Blomme, A.; Mazzucchelli, G.; Delvenne, P.; Mutijima, E.N.; Lifrange, E.; De Pauw, E.; Castronovo, V. Novel comprehensive approach for accessible biomarker identification and absolute quantification from precious human tissues. J. Proteome Res. 2011, 10, 3160–3182. [Google Scholar] [CrossRef]
- Zhang, L.; Wan, S.; Jiang, Y.; Wang, Y.; Fu, T.; Liu, Q.; Cao, Z.; Qiu, L.; Tan, W. Molecular Elucidation of Disease Biomarkers at the Interface of Chemistry and Biology. J. Am. Chem. Soc. 2017, 139, 2532–2540. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yi, K.; Li, H.; Zhang, X.; Zhang, L. Designed Tb(III)-Functionalized MOF-808 as Visible Fluorescent Probes for Monitoring Bilirubin and Identifying Fingerprints. Inorg. Chem. 2021, 60, 3172–3180. [Google Scholar] [CrossRef] [PubMed]
- Song, L.; Tian, F.; Liu, Z. Lanthanide doped metal-organic frameworks as a ratiometric fluorescence biosensor for visual and ultrasensitive detection of serotonin. J. Solid State Chem. 2022, 312, 123231. [Google Scholar] [CrossRef]
- Jia, L.; Chen, X.; Xu, J.; Zhang, L.; Guo, S.; Bi, N.; Zhu, T. A smartphone-integrated multicolor fluorescence probe of bacterial spore biomarker: The combination of natural clay material and metal-organic frameworks. J. Hazard. Mater. 2020, 402, 123776. [Google Scholar] [CrossRef] [PubMed]
- Sun, N.; Yan, B. Fluorescence detection of urinary N-methylformamide for biomonitoring of human occupational exposure to N,N-dimethylformamide by Eu(III) functionalized MOFs. Sens. Actuators B Chem. 2018, 261, 153–160. [Google Scholar] [CrossRef]
- Wei, Z.H.; Chen, D.S.; Guo, Z.F.; Jia, P.Y.; Xing, H.Z. Eosin Y-Embedded Zirconium-Based Metal-Organic Framework as a Dual-Emitting Built-In Self-Calibrating Platform for Pesticide Detection. Inorg. Chem. 2020, 59, 5386–5393. [Google Scholar] [CrossRef]
- Liu, L.; Chen, X.-L.; Shang, L.; Cai, M.; Cui, H.-L.; Yang, H.; Wang, J.-J. Eu3+-postdoped MOFs are used for fluorescence sensing of TNP, TC and pesticides and for anti-counterfeiting ink application. Dye. Pigment. 2022, 202, 110253. [Google Scholar] [CrossRef]
- Cai, Y.; Zhu, H.; Zhou, W.; Qiu, Z.; Chen, C.; Qileng, A.; Li, K.; Liu, Y. Capsulation of AuNCs with AIE Effect into Metal–Organic Framework for the Marriage of a Fluorescence and Colorimetric Biosensor to Detect Organophosphorus Pesticides. Anal. Chem. 2021, 93, 7275–7282. [Google Scholar] [CrossRef]
- Sun, X.Y.; Zhang, H.J.; Sun, Q.; Gao, E.Q. PB@UiO-67-CDC-(CH3)(2) as an Ultrasensitive Ratiometric Fluorescence Sensor: Visible “Turn-On” Effect for Detecting Preservatives and Amino Acids. Cryst. Growth Des. 2021, 21, 7218–7229. [Google Scholar] [CrossRef]
- Xiong, T.; Zhang, Y.; Donà, L.; Gutiérrez, M.; Möslein, A.F.; Babal, A.S.; Amin, N.; Civalleri, B.; Tan, J.-C. Tunable Fluorescein-Encapsulated Zeolitic Imidazolate Framework-8 Nanoparticles for Solid-State Lighting. ACS Appl. Nano Mater. 2021, 4, 10321–10333. [Google Scholar] [CrossRef]
- Jia, W.; Fan, R.; Zhang, J.; Zhu, K.; Gai, S.; Nai, H.; Guo, H.; Wu, J.; Yang, Y. Home-made multifunctional auxiliary device for in-situ imaging detection and removal of quinclorac residues through MOF decorated gel refills. Chem. Eng. J. 2022, 450, 138303. [Google Scholar] [CrossRef]
- Lu, Z.; Li, M.; Chen, M.; Wang, Q.; Wu, C.; Sun, M.; Su, G.; Wang, X.; Wang, Y.; Zhou, X.; et al. Deep learning-assisted smartphone-based portable and visual ratiometric fluorescence device integrated intelligent gel label for agro-food freshness detection. Food Chem. 2023, 413, 135640. [Google Scholar] [CrossRef]
- Pei, J.; Xuemei, H.; Jiayu, Y.; Xinyu, S.; Tong, B.; Yuting, Z.; Li, W. Dual–emission MOF–based ratiometric platform and sensory hydrogel for visible detection of biogenic amines in food spoilage. Sens. Actuators B Chem. 2023, 374, 132803. [Google Scholar] [CrossRef]
- Seemesh Bhaskar. Biosensing Technologies: A Focus Review on Recent Advancements in Surface Plasmon Coupled Emission. Micromachines 2023, 14, 574. [Google Scholar] [CrossRef]
- Mondal, S.; Subramaniam, C. Xenobiotic Contamination of Water by Plastics and Pesticides Revealed through Real-Time, Ultrasensitive, and Reliable Surface-Enhanced Raman Scattering. ACS Sustain. Chem. Eng. 2020, 8, 7639–7648. [Google Scholar] [CrossRef]
- Rai, A.; Bhaskar, S.; Ganesh, K.M.; Ramamurthy, S.S. Hottest Hotspots from the Coldest Cold: Welcome to Nano 4.0. ACS Appl. Nano Mater. 2022, 5, 12245–12264. [Google Scholar] [CrossRef]
- Qin, S.-J.; Yan, B. A facile indicator box based on Eu3+ functionalized MOF hybrid for the determination of 1-naphthol, a biomarker for carbaryl in urine. Sens. Actuators B Chem. 2017, 259, 125–132. [Google Scholar] [CrossRef]
- He, J.-X.; Yuan, H.-Q.; Zhong, Y.-F.; Peng, X.-X.; Xia, Y.-F.; Liu, S.-Y.; Fan, Q.; Yang, J.-L.; Deng, K.; Wang, X.-Y.; et al. A luminescent Eu3+-functionalized MOF for sensitive and rapid detection of tetracycline antibiotics in swine wastewater and pig kidney. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2022, 277, 121252. [Google Scholar] [CrossRef]
- Qin, G.; Kong, Y.; Gan, T.; Ni, Y. Ultrathin 2D Eu3+@Zn-MOF Nanosheets: A Functional Nanoplatform for Highly Selective, Sensitive, and Visualized Detection of Organochlorine Pesticides in a Water Environment. Inorg. Chem. 2022, 61, 8966–8975. [Google Scholar] [CrossRef]
- Sun, Z.; Li, Y.; Liu, J.; Zhao, Z.; Wang, F.; Wang, X. Lanthanide-Functionalized Metal−Organic Framework as Ratiometric Probe for Selective Detection of 4-NA and Fe3+. J. Inorg. Organomet. Polym. Mater. 2022, 32, 2953–2960. [Google Scholar] [CrossRef]
- Yingmin, J.; Xin, X.; Wanpeng, M.; Bing, Y. An Eu3+-functionalized metal–organic framework (Eu@Zn-MOF) for the highly sensitive detection of rotenone in serum. New J. Chem. 2022, 46, 19168–19173. [Google Scholar] [CrossRef]
- Liu, C.; Wang, H.; Hu, X.; Cao, Y.; Fang, G. Construction of an ECL Detection Platform for Sensitive Detection of Carbaryl Based on an Eu3+-Functionalized Metal–Organic Framework Encapsulated with Nanogold. Foods 2022, 11, 1487. [Google Scholar] [CrossRef] [PubMed]
- Peng, X.-X.; Bao, G.-M.; Zhong, Y.-F.; Zhang, L.; Zeng, K.-B.; He, J.-X.; Xiao, W.; Xia, Y.-F.; Fan, Q.; Yuan, H.-Q. Highly sensitive and rapid detection of thiabendazole residues in oranges based on a luminescent Tb3+-functionalized MOF. Food Chem. 2020, 343, 128504. [Google Scholar] [CrossRef] [PubMed]
- Zheng, X.; Fan, R.; Song, Y.; Wang, A.; Xing, K.; Du, X.; Wang, P.; Yang, Y. A highly sensitive turn-on ratiometric luminescent probe based on postsynthetic modification of Tb3+@Cu-MOF for H2S detection. J. Mater. Chem. C 2017, 5, 9943–9951. [Google Scholar] [CrossRef]
- Wu, J.-X.; Yan, B. A dual-emission probe to detect moisture and water in organics based on green-Tb3+ post-coordinated metal−organic frameworks with red-carbon dots. Dalton Trans. 2017, 46, 7098–7105. [Google Scholar] [CrossRef]
- Zhang, Y.; Li, B.; Ma, H.; Zhang, L.; Zhang, W. An RGH–MOF as a naked eye colorimetric fluorescent sensor for picric acid recognition. J. Mater. Chem. C 2017, 5, 4661–4669. [Google Scholar] [CrossRef]
- Jiang, X.-X.; Li, P.; Zhao, M.-Y.; Chen, R.-C.; Wang, Z.-G.; Xie, J.-X.; Lv, Y.-K. In situ encapsulation of SQDs by zinc ion-induced ZIF-8 growth strategy for fluorescent and colorimetric dual-signal detection of alkaline phosphatase. Anal. Chim. Acta 2022, 1221, 340103. [Google Scholar] [CrossRef]
- Luo, X.; Huang, G.; Bai, C.; Wang, C.; Yu, Y.; Tan, Y.; Tang, C.; Kong, J.; Huang, J.; Li, Z. A versatile platform for colorimetric, fluorescence and photothermal multi-mode glyphosate sensing by carbon dots anchoring ferrocene metal-organic framework nanosheet. J. Hazard. Mater. 2023, 443, 130277. [Google Scholar] [CrossRef]
- Wong, D.; Phani, A.; Homayoonnia, S.; Park, S.S.; Kim, S.; Abuzalat, O. Manipulating Active Sites of 2D Metal–Organic Framework Nanosheets with Fluorescent Materials for Enhanced Colorimetric and Fluorescent Ammonia Sensing. Adv. Mater. Interfaces 2022, 9, 2102086. [Google Scholar] [CrossRef]
- Liu, S.; Chen, Y.; Ruan, Z.; Lin, J.; Kong, W. Development of label-free fluorescent biosensor for the detection of kanamycin based on aptamer capped metal-organic framework. Environ. Res. 2022, 206, 112617. [Google Scholar] [CrossRef]
- Peng-Chen, S.; Long, Y.; Mi, Y.; Ling-Xiao, W.; Ming-Tai, S.; Wei-Jie, H.; Hua, T.; Su-Hua, W. Dye-encapsulated metal–organic framework composites for highly sensitive and selective sensing of oxytetracycline based on ratiometric fluorescence. Chem. Pap. 2022, 77, 2295–2308. [Google Scholar] [CrossRef]
- Guo, X.; Pan, Q.; Song, X.; Guo, Q.; Zhou, S.; Qiu, J.; Dong, G. Embedding carbon dots in Eu3+-doped metal-organic framework for label-free ratiometric fluorescence detection of Fe3+ ions. J. Am. Ceram. Soc. 2020, 104, 886–895. [Google Scholar] [CrossRef]
- Peng, X.-X.; Bao, G.-M.; Zhong, Y.-F.; He, J.-X.; Zeng, L.; Yuan, H.-Q. Highly selective detection of Cu2+ in aqueous media based on Tb3+-functionalized metal-organic framework. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2020, 240, 118621. [Google Scholar] [CrossRef]
- Ruan, B.; Yang, J.; Zhang, Y.J.; Ma, N.; Shi, D.A.; Jiang, T.; Tsai, F.C. UiO-66 derivate as a fluorescent probe for Fe3+ detection. Talanta 2020, 218, 121207. [Google Scholar] [CrossRef]
- Zhang, N.; Zhang, D.; Zhao, J.; Xia, Z. Fabrication of a dual-emitting dye-encapsulated metal–organic framework as a stable fluorescent sensor for metal ion detection. Dalton Trans. 2019, 48, 6794–6799. [Google Scholar] [CrossRef]
- Yang, W.; Yang, Y.; Li, H.; Lin, D.; Yang, W.; Guo, D.; Pan, Q. Integration of Cd:ZnS QDs into ZIF-8 for enhanced selectivity toward Cu2+ detection. Inorg. Chem. Front. 2020, 7, 3718–3726. [Google Scholar] [CrossRef]
- Lin, X.; Gao, G.; Zheng, L.; Chi, Y.; Chen, G. Encapsulation of Strongly Fluorescent Carbon Quantum Dots in Metal–Organic Frameworks for Enhancing Chemical Sensing. Anal. Chem. 2013, 86, 1223–1228. [Google Scholar] [CrossRef]
- Hou, J.; Jia, P.; Yang, K.; Bu, T.; Zhao, S.; Li, L.; Wang, L. Fluorescence and Colorimetric Dual-Mode Ratiometric Sensor Based on Zr–Tetraphenylporphyrin Tetrasulfonic Acid Hydrate Metal–Organic Frameworks for Visual Detection of Copper Ions. ACS Appl. Mater. Interfaces 2022, 14, 13848–13857. [Google Scholar] [CrossRef]
- Guo, H.; Wang, X.; Wu, N.; Xu, M.; Wang, M.; Zhang, L.; Yang, W. In-situ Synthesis of Carbon Dots-embedded Europium Metal-Organic Frameworks for Ratiometric Fluorescence Detection of Hg2+ in Aqueous Environment. Anal. Chim. Acta 2021, 1141, 13–20. [Google Scholar] [CrossRef]
- Hao, J.-N.; Yan, B. Highly sensitive and selective fluorescent probe for Ag+ based on a Eu3+ post-functionalized metal–organic framework in aqueous media. J. Mater. Chem. A 2014, 2, 18018–18025. [Google Scholar] [CrossRef]
- Wu, X.; Xi, J.; Wei, X.; Yin, C. An ultra-fast UV-electrochemical sensor based on Cu-MOF for highly sensitive and selective detection of ferric ions. Analyst 2023, 148, 366–373. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Feng, L.; Ma, S.; Xia, T.; Jiao, F.; Kong, Z.; Duan, X. A microporous Tb-based MOF for multifunctional detection of the α-CHC, Cu2+ and Fe3+. J. Solid State Chem. 2022, 312, 123232. [Google Scholar] [CrossRef]
- Guan, Y.; Zhao, X.-L.; Li, Q.-X.; Huang, L.; Yang, J.-M.; Yang, T.; Yang, Y.-H.; Hu, R. Iodide-enhanced Cu-MOF nanomaterials for the amplified colorimetric detection of Fe3+. Anal. Methods 2021, 13, 5851–5858. [Google Scholar] [CrossRef] [PubMed]
- Tan, S.C.; Chin, S.F.; Pang, S.C. Disposable Carbon Dots Modified Screen Printed Carbon Electrode Electrochemical Sensor Strip for Selective Detection of Ferric Ions. J. Sens. 2017, 2017, 1–7. [Google Scholar] [CrossRef] [Green Version]
- Kim, D.-Y.; Shinde, S.; Saratale, R.; Syed, A.; Ameen, F.; Ghodake, G. Spectrophotometric determination of Fe(III) by using casein-functionalized gold nanoparticles. Microchim. Acta 2017, 184, 4695–4704. [Google Scholar] [CrossRef]
- Chen, L.; Ma, N.; Park, Y.; Jin, S.; Hwang, H.; Jiang, D.; Jung, Y.M. Highly sensitive determination of iron (III) ion based on phenanthroline probe: Surface-enhanced Raman spectroscopy methods. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2017, 197, 43–46. [Google Scholar] [CrossRef]
- Olgaç, N.; Karakuş, E.; Şahin, Y.; Liv, L. Voltammetric Method for Determining Ferric Ions with Quercetin. Electroanalysis 2021, 33, 2115–2121. [Google Scholar] [CrossRef]
- Kalhori, S.; Ahour, F.; Aurang, P. Determination of trace amount of iron cations using electrochemical methods at N, S doped GQD modified electrode. Sci. Rep. 2023, 13, 1557. [Google Scholar] [CrossRef]
- Wen, X.-Z.; Yu, H.; Ma, Y.-J. Separation and indirect ultraviolet detection of ferrous and trivalent iron ions by using ionic liquids in ion chromatography. J. Sep. Sci. 2019, 42, 3432–3438. [Google Scholar] [CrossRef]
- Yap, S.H.K.; Chan, K.K.; Zhang, G.; Tjin, S.C.; Yong, K.-T. Carbon Dot-functionalized Interferometric Optical Fiber Sensor for Detection of Ferric Ions in Biological Samples. ACS Appl. Mater. Interfaces 2019, 11, 28546–28553. [Google Scholar] [CrossRef]
- Anas, N.A.A.; Fen, Y.W.; Yusof, N.A.; Omar, N.A.S.; Daniyal, W.M.E.M.M.; Ramdzan, N.S.M. Highly sensitive surface plasmon resonance optical detection of ferric ion using CTAB/hydroxylated graphene quantum dots thin film. J. Appl. Phys. 2020, 128, 083105. [Google Scholar] [CrossRef]
- Zheng, Y.; Wang, X.; Guan, Z.; Deng, J.; Liu, X.; Li, H.; Zhao, P. Application of CD and Eu3+ Dual Emission MOF Colorimetric Fluorescent Probe Based on Neural Network in Fe3+ Detection. Part. Part. Syst. Charact. 2022, 39, 2200124. [Google Scholar] [CrossRef]
- Manoj, D.; Theyagarajan, K.; Saravanakumar, D.; Senthilkumar, S.; Thenmozhi, K. Aldehyde functionalized ionic liquid on electrochemically reduced graphene oxide as a versatile platform for covalent immobilization of biomolecules and biosensing. Biosens. Bioelectron. 2017, 103, 104–112. [Google Scholar] [CrossRef]
- Devaraj, M.; Deivasigamani, R.K.; Jeyadevan, S. Enhancement of the electrochemical behavior of CuO nanoleaves on MWCNTs/GC composite film modified electrode for determination of norfloxacin. Colloids Surf. B Biointerfaces 2012, 102, 554–561. [Google Scholar] [CrossRef]
- He, W.-y.; Wang, K.-p.; Yang, J.-y. Spectrophotometric methods for determination of vanadium: A review. Toxicol. Environ. Chem. 2018, 100, 20–31. [Google Scholar] [CrossRef]
- Li, D.; Xu, X.; Li, Z.; Wang, T.; Wang, C. Detection methods of ammonia nitrogen in water: A review. Trends Anal. Chem. 2020, 127, 115890. [Google Scholar] [CrossRef]
- Singh, P.; Singh, M.K.; Beg, Y.R.; Nishad, G.R. A review on spectroscopic methods for determination of nitrite and nitrate in environmental samples. Talanta 2018, 191, 364–381. [Google Scholar] [CrossRef]
- Dutta Choudhury, S.; Badugu, R.; Lakowicz, J.R. Directing Fluorescence with Plasmonic and Photonic Structures. Acc. Chem. Res. 2015, 48, 2171–2180. [Google Scholar] [CrossRef] [Green Version]
- Gryczynski, I.; Malicka, J.; Gryczynski, Z.; Lakowicz, J.R. Radiative decay engineering 4. Experimental studies of surface plasmon-coupled directional emission. Anal. Biochem. 2004, 324, 170–182. [Google Scholar] [CrossRef] [Green Version]
- Chao, L.; Di, X.; Xuan, D.; Qing, H. A review: Research progress of SERS-based sensors for agricultural applications. Trends Food Sci. Technol. 2022, 128, 90–101. [Google Scholar] [CrossRef]
- Shintaro, P.; Tianxi, Y.; Lili, H. Review of surface enhanced raman spectroscopic (SERS) detection of synthetic chemical pesticides. Trends Anal. Chem. 2016, 85, 73–82. [Google Scholar]
- Sun, X.; Li, H. A Review: Nanofabrication of Surface-Enhanced Raman Spectroscopy (SERS) Substrates. Curr. Nanosci. 2016, 12, 175–183. [Google Scholar] [CrossRef] [Green Version]
- Michalski, R.; Pecyna-Utylska, P.; Kernert, J. Determination of ammonium and biogenic amines by ion chromatography. A review. J. Chromatogr. A 2021, 1651, 462319. [Google Scholar] [CrossRef] [PubMed]
- Paull, B.; Barron, L. Using ion chromatography to monitor haloacetic acids in drinking water: A review of current technologies. J. Chromatogr. A 2004, 1046, 1–9. [Google Scholar] [CrossRef]
- Chang, C.-C. Recent Advancements in Aptamer-Based Surface Plasmon Resonance Biosensing Strategies. Biosensors 2021, 11, 233. [Google Scholar] [CrossRef]
- Lakowicz, J.R.; Ray, K.; Chowdhury, M.; Szmacinski, H.; Fu, Y.; Zhang, J.; Nowaczyk, K. Plasmon-controlled fluorescence: A new paradigm in fluorescence spectroscopy. Analyst 2008, 133, 1308–1346. [Google Scholar] [CrossRef] [Green Version]
- Meng, L.; Yang, Z. Directional surface plasmon-coupled emission of tilted-tip enhanced spectroscopy. Nanophotonics 2018, 7, 1325–1332. [Google Scholar] [CrossRef]
- Hoang Minh, N.; Yoon, J.S.; Kang, D.H.; Yoo, Y.-E.; Kim, K. Assembling Vertical Nanogap Arrays with Nanoentities for Highly Sensitive Electrical Biosensing. Langmuir 2023, 39, 2274–2280. [Google Scholar] [CrossRef]


Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Xu, X.; Ma, M.; Sun, T.; Zhao, X.; Zhang, L. Luminescent Guests Encapsulated in Metal–Organic Frameworks for Portable Fluorescence Sensor and Visual Detection Applications: A Review. Biosensors 2023, 13, 435. https://doi.org/10.3390/bios13040435
Xu X, Ma M, Sun T, Zhao X, Zhang L. Luminescent Guests Encapsulated in Metal–Organic Frameworks for Portable Fluorescence Sensor and Visual Detection Applications: A Review. Biosensors. 2023; 13(4):435. https://doi.org/10.3390/bios13040435
Chicago/Turabian StyleXu, Xu, Muyao Ma, Tongxin Sun, Xin Zhao, and Lei Zhang. 2023. "Luminescent Guests Encapsulated in Metal–Organic Frameworks for Portable Fluorescence Sensor and Visual Detection Applications: A Review" Biosensors 13, no. 4: 435. https://doi.org/10.3390/bios13040435
APA StyleXu, X., Ma, M., Sun, T., Zhao, X., & Zhang, L. (2023). Luminescent Guests Encapsulated in Metal–Organic Frameworks for Portable Fluorescence Sensor and Visual Detection Applications: A Review. Biosensors, 13(4), 435. https://doi.org/10.3390/bios13040435




