Assessment of the Antioxidant Capacity of Commercial Coffee Using Conventional Optical and Chromatographic Methods and an Innovative Electrochemical DNA-Based Biosensor
Abstract
:1. Introduction
2. Materials and Methods
2.1. Reagents and Solutions
2.2. Apparatus and Electrode
2.3. Samples
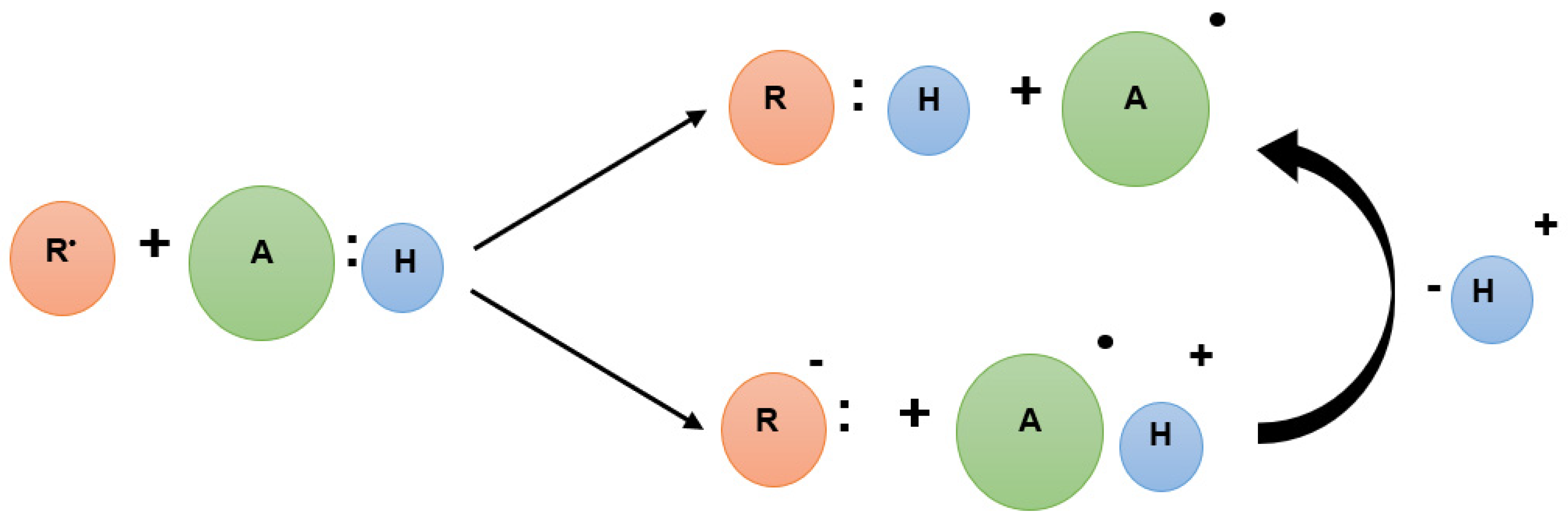
2.4. DNA-Based Biosensor Procedure


2.5. Conventional Optical Analytical Methods
2.5.1. Total Phenolic Content
2.5.2. Total Flavonoid Content
2.5.3. Ferric Reducing Antioxidant Power (FRAP) Assay
2.5.4. 2,2-Diphenyl-1-Picryl-Hydrazyl-Hydrate-Radical Scavenging Assay
2.6. HPLC-DAD
2.7. Statistical Analysis
3. Results and Discussion
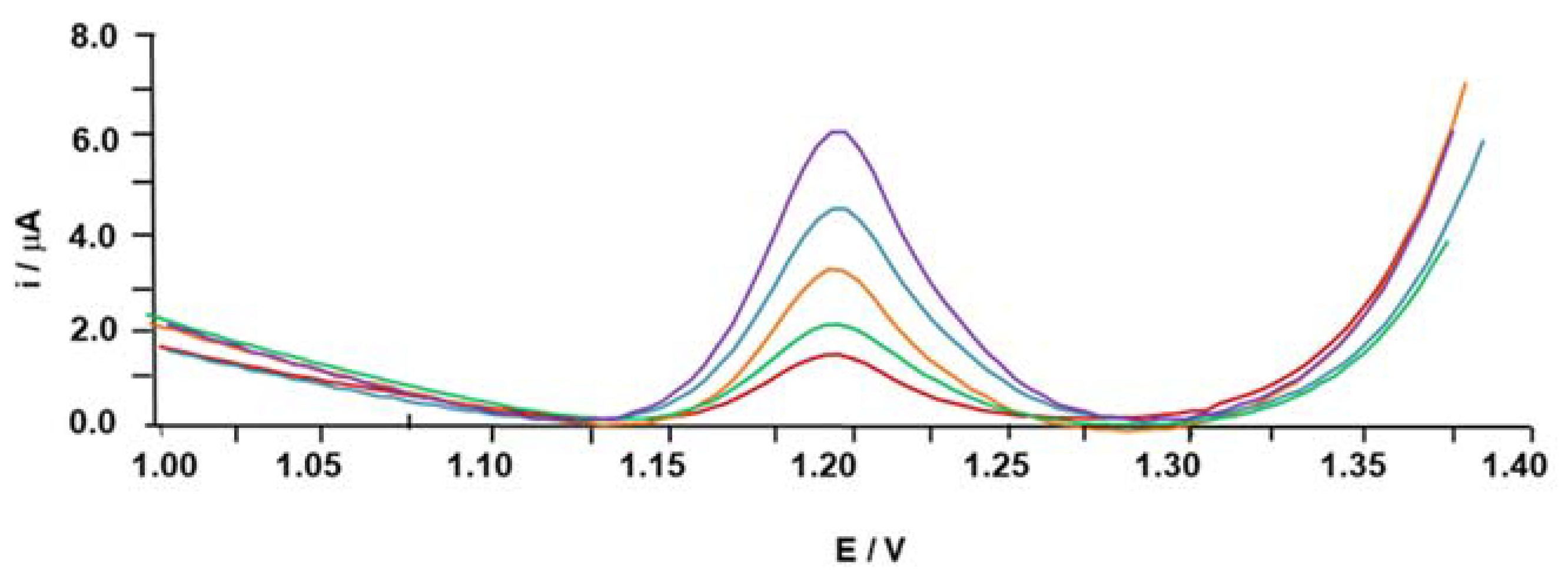
3.1. Electrochemical DNA-Based Biosensor
3.2. TAC Assessment
3.2.1. DNA-Based Biosensor

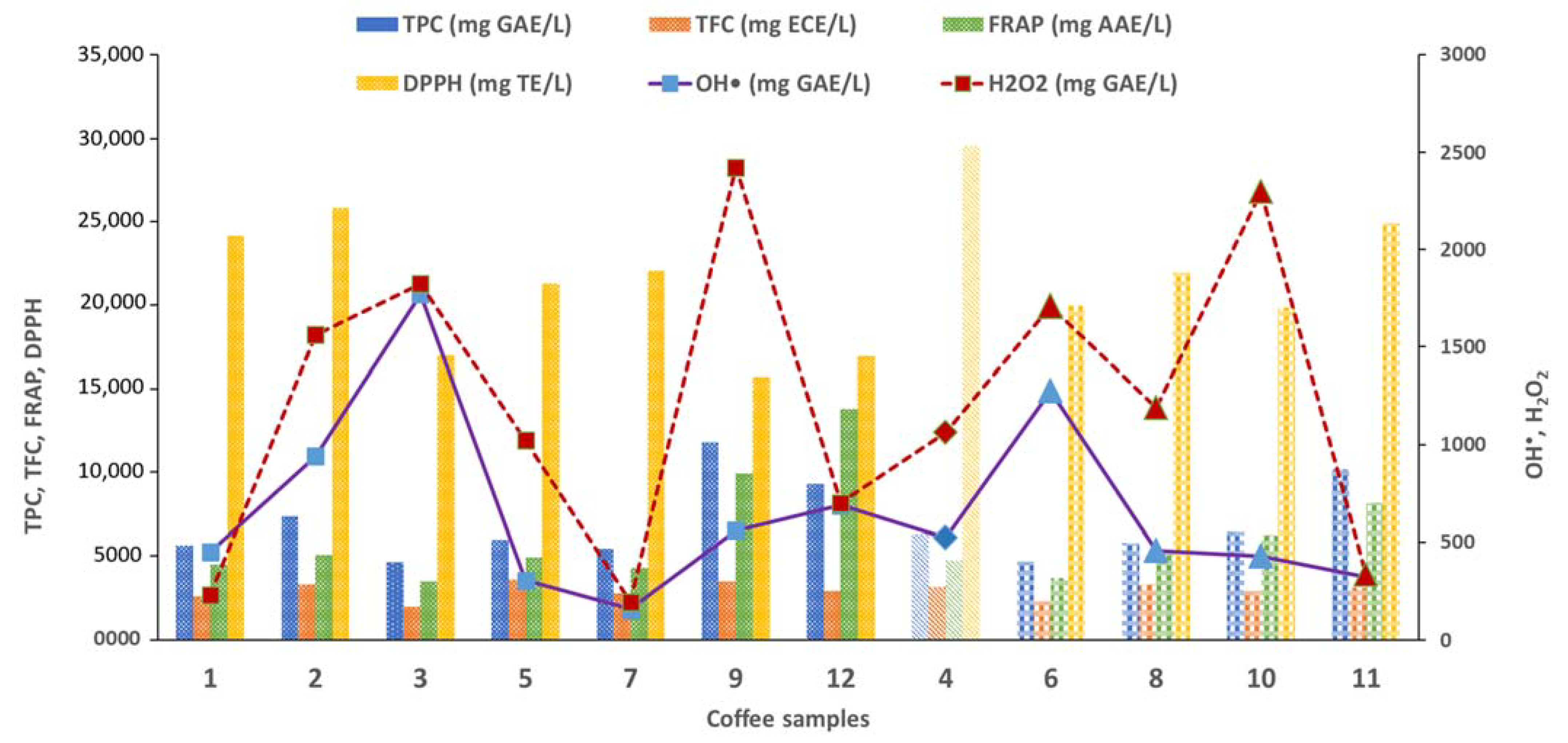
3.2.2. Optical Methods
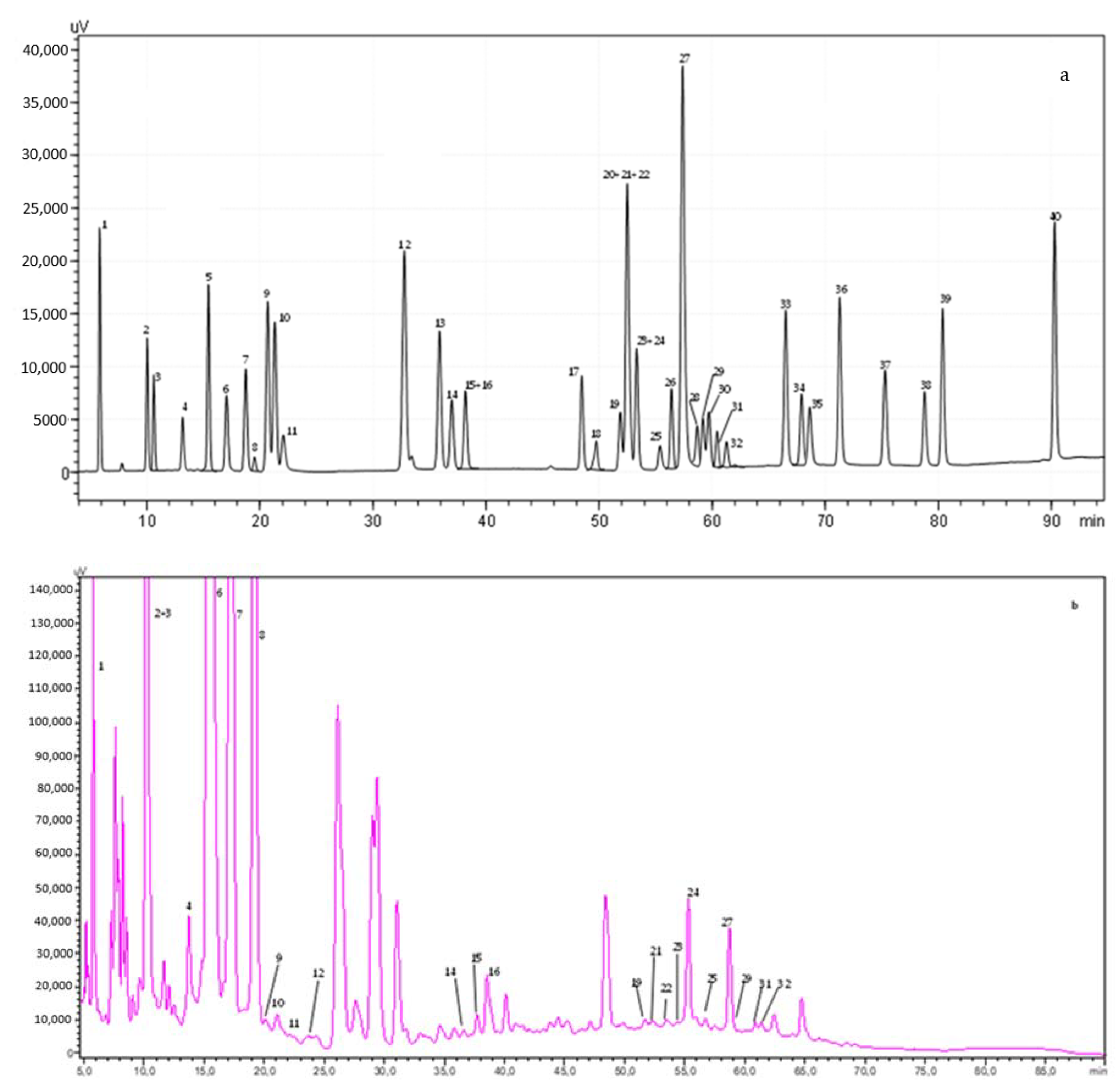
3.2.3. Phenolic Profile of Coffee Samples by HPLC-DAD
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Pizzino, G.; Irrera, N.; Cucinotta, M.; Pallio, G.; Mannino, F.; Arcoraci, V.; Squadrito, F.; Altavilla, D.; Bitto, A. Oxidative Stress: Harms and Benefits for Human Health. Oxidative Med. Cell. Longev. 2017, 2017, 8416763. [Google Scholar] [CrossRef]
- Liang, N.; Kitts, D.D. Antioxidant Property of Coffee Components: Assessment of Methods that Define Mechanisms of Action. Molecules 2014, 19, 19180–19208. [Google Scholar] [CrossRef]
- Socała, K.; Szopa, A.; Serefko, A.; Poleszak, E.; Wlaź, P. Neuroprotective Effects of Coffee Bioactive Compounds: A Review. Int. J. Mol. Sci. 2021, 22, 107. [Google Scholar] [CrossRef]
- Sharifi-Rad, M.; Anil Kumar, N.; Zucca, P.; Varoni, E.M.; Dini, L.; Panzarini, E.; Rajkovic, J.; Fokou, P.V.T.; Azzini, E.; Peluso, I.; et al. Lifestyle, Oxidative Stress, and Antioxidants: Back and Forth in the Pathophysiology of Chronic Diseases. Front. Physiol. 2020, 11, 694. [Google Scholar] [CrossRef]
- Munteanu, I.G.; Apetrei, C. Analytical Methods Used in Determining Antioxidant Ac-tivity: A Review. Int. J. Mol. Sci. 2021, 22, 3380. [Google Scholar] [CrossRef]
- Carvalho, A.P.; Mendes, M.; Moreira, M.M.; Cruz, D.; Magalhães, J.M.C.S.; Barroso, M.F.; Ramalhosa, M.J.; Duarte, A.; Guido, L.; Gomes, A.M.; et al. Micro-wave-assisted extraction in goji berries: Effect on composition and bioactivity, evaluated through conventional and nonconventional methodologies. Int. J. Food Sci. Technol. 2016, 51, 1401–1408. [Google Scholar] [CrossRef]
- Carlsen, M.H.; Halvorsen, B.L.; Holte, K.; Bøhn, S.K.; Dragland, S.; Sampson, L.; Willey, C.; Senoo, H.; Umezono, Y.; Sanada, C.; et al. The total antioxidant content of more than 3100 foods, bev-erages, spices, herbs and supplements used worldwide. Nutr. J. 2010, 9, 3. [Google Scholar] [CrossRef]
- Hlavatá, L.; Vyskočil, V.; Beníková, K.; Borbélyová, M.; Labuda, J. DNA-based biosensors with external Nafion and chitosan membranes for the evaluation of the antioxidant activity of beer, coffee, and tea. Cent. Eur. J. Chem. 2014, 12, 604–611. [Google Scholar] [CrossRef]
- Cruz, D.; Barroso, M.F.; Alves, R.C.; González-García, M.B.; Ramalhosa, M.J.; Duarte, A.J.; Oliveira, M.B.P.P.; Delerue-Matos, C. Chapter 106—Assay of Total Antioxidant Capac-ity of Coffee: Use of a DNA-Based Biosensor. In Coffee in Health and Disease Prevention; Preedy, V.R., Ed.; Academic Press: Cambridge, MA, USA, 2015; pp. 963–970. [Google Scholar] [CrossRef]
- Tomac, I.; Šeruga, M.; Labuda, J. Evaluation of antioxidant activity of chlorogenic ac-ids and coffee extracts by an electrochemical DNA-based biosensor. Food Chem. 2020, 325, 126787. [Google Scholar] [CrossRef] [PubMed]
- Barroso, M.F.; Ramalhosa, M.J.; Alves, R.C.; Dias, A.; Soares, C.M.D.; Oliva-Teles, T.; Delerue-Matos, C. Total antioxidant capacity of plant infusions: Assessment using elec-trochemical DNA-based biosensor and spectrophotometric methods. Food Control 2016, 68, 153–161. [Google Scholar] [CrossRef]
- Barroso, M.F.; de-los-Santos-Álvarez, N.; Lobo-Castañón, M.J.; Miranda-Ordieres, A.J.; Delerue-Matos, C.; Oliveira, M.B.P.P.; Tuñón-Blanco, P. DNA-based biosensor for the elec-trocatalytic determination of antioxidant capacity in beverages. Biosens. Bioelectron. 2011, 26, 2396–2401. [Google Scholar] [CrossRef]
- Czachor, J.; Miłek, M.; Galiniak, S.; Stępień, K.; Dżugan, M.; Mołoń, M. Coffee Extends Yeast Chronological Lifespan through Antioxidant Properties. Int. J. Mol. Sci. 2020, 21, 9510. [Google Scholar] [CrossRef]
- Ridder, M. Coffee—Worldwide. Statistica. 2022. Available online: https://www.statista.com/outlook/cmo/hot-drinks/coffee/worldwide (accessed on 1 May 2023).
- Zhu, M.; Long, Y.; Ma, Y.; Chen, Y.; Yu, Q.; Xie, J.; Li, B.; Tian, J. Comparison of chemi-cal and fatty acid composition of green coffee bean (Coffea arabica L.) from different geo-graphical origins. LWT 2021, 140, 110802. [Google Scholar] [CrossRef]
- Samsonowicz, M.; Regulska, E.; Karpowicz, D.; Leśniewska, B. Antioxidant properties of coffee substitutes rich in polyphenols and minerals. Food Chem. 2019, 278, 101–109. [Google Scholar] [CrossRef] [PubMed]
- Andrade, C.; Perestrelo, R.; Câmara, J.S. Bioactive Compounds and Antioxidant Activ-ity from Spent Coffee Grounds as a Powerful Approach for Its Valorization. Molecules 2022, 27, 7504. [Google Scholar] [CrossRef]
- Akomolafe, S.F.; Akinyemi, A.J.; Ogunsuyi, O.B.; Oyeleye, S.I.; Oboh, G.; Adeoyo, O.O.; Allismith, Y.R. Effect of caffeine, caffeic acid and their various combinations on enzymes of cholinergic, monoaminergic and purinergic systems critical to neurodegeneration in rat brain-In vitro. Neurotoxicology 2017, 62, 6–13. [Google Scholar] [CrossRef] [PubMed]
- Gökcen, B.B.; Şanlier, N. Coffee consumption and disease correlations. Crit. Rev. Food Sci. Nutr. 2019, 59, 336–348. [Google Scholar] [CrossRef]
- Nieber, K. The impact of coffee on health. Planta Med. 2017, 83, 1256–1263. [Google Scholar] [CrossRef] [PubMed]
- Wijarnpreecha, K.; Thongprayoon, C.; Thamcharoen, N.; Panjawatanan, P.; Cheungpasitporn, W. Association of coffee consumption and chronic kidney disease: A meta-analysis. Int. J. Clin. Pract. 2017, 71, e12919. [Google Scholar] [CrossRef]
- de Mendonça, A.; Cunha, R.A. Therapeutic opportunities for caffeine in Alzheimer’s disease and other neurodegenerative disorders. J. Alzheimer’s Dis. 2010, 20 (Suppl. S1), S1–S2. [Google Scholar] [CrossRef]
- Bidel, S.; Tuomilehto, J. The Emerging Health Benefits of Coffee with an Emphasis on Type 2 Diabetes and Cardiovascular Disease. Eur. J. Endocrinol. 2013, 9, 99–106. [Google Scholar] [CrossRef]
- Mineharu, Y.; Koizumi, A.; Wada, Y.; Iso, H.; Watanabe, Y.; Date, C.; Yamamoto, A.; Kikuchi, S.; Inaba, Y.; Toyoshima, H.; et al. Coffee, green tea, black tea and oolong tea consumption and risk of mortality from cardiovascular disease in Japanese men and women. J. Epidemiol. Community Health 2011, 65, 230–240. [Google Scholar] [CrossRef] [PubMed]
- Amorati, R.; Valgimigli, L. Methods to Measure the Antioxidant Activity of Phytochemicals and Plant Extracts. J. Agric. Food Chem. 2018, 66, 3324–3329. [Google Scholar] [CrossRef] [PubMed]
- Moreira, M.M.; Rodrigues, F.; Dorosh, O.; Pinto, D.; Costa, P.C.; Švarc-Gajić, J.; Dele-rue-Matos, C. Vine-Canes as a Source of Value-Added Compounds for Cosmetic Formulations. Molecules 2020, 25, 2969. [Google Scholar] [CrossRef]
- Losada-Barreiro, S.; Sezgin-Bayindir, Z.; Paiva-Martins, F.; Bravo-Díaz, C. Biochemis-try of Antioxidants: Mechanisms and Pharmaceutical Applications. Biomedicines 2022, 10, 3051. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Sevilla, E.; Palomar-Pardave, M.; Romero-Romo, M.; Ramírez-Silva, M.T. Modulating the analytical performance of an electrochemical biosensor through varying, at the working electrode, the surface area ratio between that covered by the enzyme and the enzyme-free. Anal. Methods 2015, 7, 8568–8571. [Google Scholar] [CrossRef]
- Miller, J.N.; Miller, J.C. Statistics and Chemometrics for Analytical Chemistry, 6th ed.; Pearson Education Limited: Gosport, UK, 2010; pp. 1–297. [Google Scholar]
- Hasbullah, U.H.A.; Umiyati, R. Antioxidant Activity and Total Phenolic Compounds of Arabica and Robusta Coffee at Different Roasting Levels. J. Phys. Conf. Ser. 2021, 1764, 012033. [Google Scholar] [CrossRef]
- Olechno, E.; Puścion-Jakubik, A.; Markiewicz-Żukowska, R.; Socha, K. Impact of Brewing Methods on Total Phenolic Content (TPC) in Various Types of Coffee. Molecules 2020, 25, 5274. [Google Scholar] [CrossRef]
- Schwarzmann, E.T.; Washington, M.P.; Rao, N.Z. Physicochemical Analysis of Cold Brew and Hot Brew Peaberry Coffee. Processes 2022, 10, 1989. [Google Scholar] [CrossRef]
- Catelani, T.A.; Páscoa, R.N.M.J.; Santos, J.R.; Pezza, L.; Pezza, H.R.; Lima, J.L.F.C.; Lopes, J.A. A Non-invasive Real-Time Methodology for the Quantification of Antioxidant Properties in Coffee During the Roasting Process Based on Near-Infrared Spectroscopy. Food Bioprocess Technol. 2017, 10, 630–638. [Google Scholar] [CrossRef]
- Baeza, G.; Sarriá, B.; Bravo, L.; Mateos, R. Exhaustive Qualitative LC-DAD-MSn Anal-ysis of Arabica Green Coffee Beans: Cinnamoyl-glycosides and Cinnamoylshikimic Acids as New Polyphenols in Green Coffee. J. Agric. Food Chem. 2016, 64, 9663–9674. [Google Scholar] [CrossRef]
- Vignoli, J.A.; Viegas, M.C.; Bassoli, D.G.; de Toledo-Benassi, M. Roasting process af-fects differently the bioactive compounds and the antioxidant activity of arabica and ro-busta coffees. Int. Food Res. J. 2014, 61, 279–285. [Google Scholar] [CrossRef]
- Vignoli, J.A.; Bassoli, D.G.; Benassi, M.T. Antioxidant activity, polyphenols, caffeine and melanoidins in soluble coffee: The influence of processing conditions and raw material. Food Chem. 2011, 124, 863–868. [Google Scholar] [CrossRef]
- López-Galilea, I.; De Peña, M.P.; Cid, C. Correlation of Selected Constituents with the Total Antioxidant Capacity of Coffee Beverages: Influence of the Brewing Procedure. J. Agric. Food Chem. 2007, 55, 6110–6117. [Google Scholar] [CrossRef] [PubMed]
- Parras, P.; Martínez-Tomé, M.; Jiménez, A.; Murcia, M. Antioxidant capacity of coffees of several origins brewed following three different procedures. Food Chem. 2007, 102, 582–592. [Google Scholar] [CrossRef]
- Garcia, L.F.; Benjamin, S.R.; Marreto, R.N.; Lopes, F.M. Laccase Carbon Paste Based Biosensors for Antioxidant Capacity. The Effect of Different Modifiers. Int. J. Electrochem. Sci. 2015, 10, 5650–5660. [Google Scholar] [CrossRef]
- Chávez, D.W.H.; Ascheri, J.L.R.; Carvalho, C.W.P.; Godoy, R.L.O.; Pacheco, S. Sorghum and roasted coffee blends as a novel extruded product: Bioactive compounds and antiox-idant capacity. J. Funct. Foods 2017, 29, 93–103. [Google Scholar] [CrossRef]
- Yashin, A.; Yashin, Y.; Wang, J.Y.; Nemzer, B. Antioxidant and Antiradical Activity of Coffee. Antioxidants 2013, 2, 230–245. [Google Scholar] [CrossRef]
- Soares, M.J.; Sampaio, G.R.; Guizellini, G.M.; Figueira, M.S.; Pinaffi, A.C.C.; Freitas, R.A.M.S.; Shahidi, F.; de Camargo, A.C.; Torres, E.A.F.S. Regular and decaffeinated espresso coffee capsules: Unravelling the bioaccessibility of phenolic compounds and their antioxidant properties in milk model system upon in vitro digestion. LWT 2021, 135, 110255. [Google Scholar] [CrossRef]
- Balzano, M.; Loizzo, M.R.; Tundis, R.; Lucci, P.; Nunez, O.; Fiorini, D.; Giardinieri, A.; Frega, N.G.; Pacetti, D. Spent espresso coffee grounds as a source of anti-proliferative and antioxidant compounds. Innov. Food Sci. Emerg. Technol. 2020, 59, 102254. [Google Scholar] [CrossRef]
- Vilas-Boas, A.A.; Oliveira, A.; Jesus, D.; Rodrigues, C.; Figueira, C.; Gomes, A.; Pintado, M. Chlorogenic acids composition and the impact of in vitro gastrointestinal digestion on espresso coffee from single-dose capsule. Food Res. Int. 2020, 134, 109223. [Google Scholar] [CrossRef]
- Ramón-Gonçalves, M.; Gómez-Mejía, E.; Rosales-Conrado, N.; León-González, M.E.; Madrid, Y. Extraction, identification and quantification of polyphenols from spent coffee grounds by chromatographic methods and chemometric analyses. Waste Manag. 2019, 96, 15–24. [Google Scholar] [CrossRef] [PubMed]
- Lopes, G.R.; Petronilho, S.; Ferreira, A.S.; Pinto, M.; Passos, C.P.; Coelho, E.; Rodrigues, C.; Figueira, C.; Rocha, S.M.; Coimbra, M.A. Insights on Single-Dose Espresso Coffee Cap-sules’ Volatile Profile: From Ground Powder Volatiles to Prediction of Espresso Brew Aroma Properties. Foods 2021, 10, 2508. [Google Scholar] [CrossRef] [PubMed]
- Özyürek, M.; Bektaşoğlu, B.; Güçlü, K.; Apak, R. Hydroxyl radical scavenging assay of phenolics and flavonoids with a modified cupric reducing antioxidant capacity (CUPRAC) method using catalase for hydrogen peroxide degradation. Anal. Chim. Acta 2008, 616, 196–206. [Google Scholar] [CrossRef]
- Sroka, Z.; Cisowski, W.H. Hydrogen peroxide scavenging, antioxidant and anti-radical activity of some phenolic acids. Food Chem. Toxicol. 2003, 41, 753–758. [Google Scholar] [CrossRef] [PubMed]
- Gulcin, İ. Antioxidants and antioxidant methods: An updated overview. Arch. Toxicol. 2020, 94, 651–715. [Google Scholar] [CrossRef] [PubMed]
- Gobbi, L.; Maddaloni, L.; Prencipe, S.A.; Vinci, G. Bioactive Compounds in Different Coffee Beverages for Quality and Sustainability Assessment. Beverages 2023, 9, 3. [Google Scholar] [CrossRef]


| Sample | Presentation | Intensity (Degree of Roast) | Composition |
|---|---|---|---|
| 1 | Capsule | 9 | arabica + robusta |
| 2 | Capsule | 10 | arabica + robusta |
| 3 | Capsule (decaffeinated) | 7 | arabica + robusta |
| 4 | Capsule (power coffee) | 13 | blend coffee + guarana + ginseng |
| 5 | Capsule | 10 | arabica + robusta |
| 6 | Capsule | 9 | 100% arabica |
| 7 | Capsule | 8 | arabica + robusta |
| 8 | Capsule | 5 | 100% arabica |
| 9 | Capsule | 7 | arabica + robusta |
| 10 | Ground coffee | 7 | 100% arabica |
| 11 | Capsule (decaffeinated) | 9 | 100% arabica |
| 12 | Instant coffee | 8 | arabica + robusta |
| TPC | TFC | FRAP | DPPH | OH• | H2O2 | |
|---|---|---|---|---|---|---|
| TPC | 1 | 0.5380 ** | 0.8140 ** | −0.1900 | −0.2970 | 0.1060 |
| TFC | 0.5380 ** | 1 | 0.3260 | 0.2230 | −0.6150 ** | 0.0274 |
| FRAP | 0.8140 ** | 0.3260 | 1 | −0.3710 | −0.2250 | −0.0095 |
| DPPH | −0.1900 | 0.2230 | −0.3710 | 1 | −0.2750 | −0.4180 * |
| OH• | −0.2970 | −0.6150 ** | −0.2250 | −0.2750 | 1 | 0.4530 * |
| H2O2 | 0.1060 | 0.0274 | −0.0095 | −0.4180 * | 0.4530 * | 1 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Morais, S.L.; Rede, D.; Ramalhosa, M.J.; Correia, M.; Santos, M.; Delerue-Matos, C.; Moreira, M.M.; Soares, C.; Barroso, M.F. Assessment of the Antioxidant Capacity of Commercial Coffee Using Conventional Optical and Chromatographic Methods and an Innovative Electrochemical DNA-Based Biosensor. Biosensors 2023, 13, 840. https://doi.org/10.3390/bios13090840
Morais SL, Rede D, Ramalhosa MJ, Correia M, Santos M, Delerue-Matos C, Moreira MM, Soares C, Barroso MF. Assessment of the Antioxidant Capacity of Commercial Coffee Using Conventional Optical and Chromatographic Methods and an Innovative Electrochemical DNA-Based Biosensor. Biosensors. 2023; 13(9):840. https://doi.org/10.3390/bios13090840
Chicago/Turabian StyleMorais, Stephanie L., Diana Rede, Maria João Ramalhosa, Manuela Correia, Marlene Santos, Cristina Delerue-Matos, Manuela M. Moreira, Cristina Soares, and Maria Fátima Barroso. 2023. "Assessment of the Antioxidant Capacity of Commercial Coffee Using Conventional Optical and Chromatographic Methods and an Innovative Electrochemical DNA-Based Biosensor" Biosensors 13, no. 9: 840. https://doi.org/10.3390/bios13090840





