Anthelminthic Activity of Assassin Bug Venom against the Blood Fluke Schistosoma mansoni
Abstract
:1. Introduction
2. Results
2.1. Assassin Bug Venom Reduces Motility, Pairing, Attachment and Egg Production in S. mansoni
2.2. Proliferating Stem Cells Are Depleted by Assassin Bug Venom
2.3. Hemolytic Analysis of Assassin Bug Venom
3. Discussion
3.1. Antischistosomal Effects of Assassin Bug Venom
3.2. Antiproliferative Effect of Assassin Bug Venom
3.3. Venom as Source for Antischistosomal Compounds
4. Materials and Methods
4.1. Ethical Statement
4.2. Production of Adult Worms
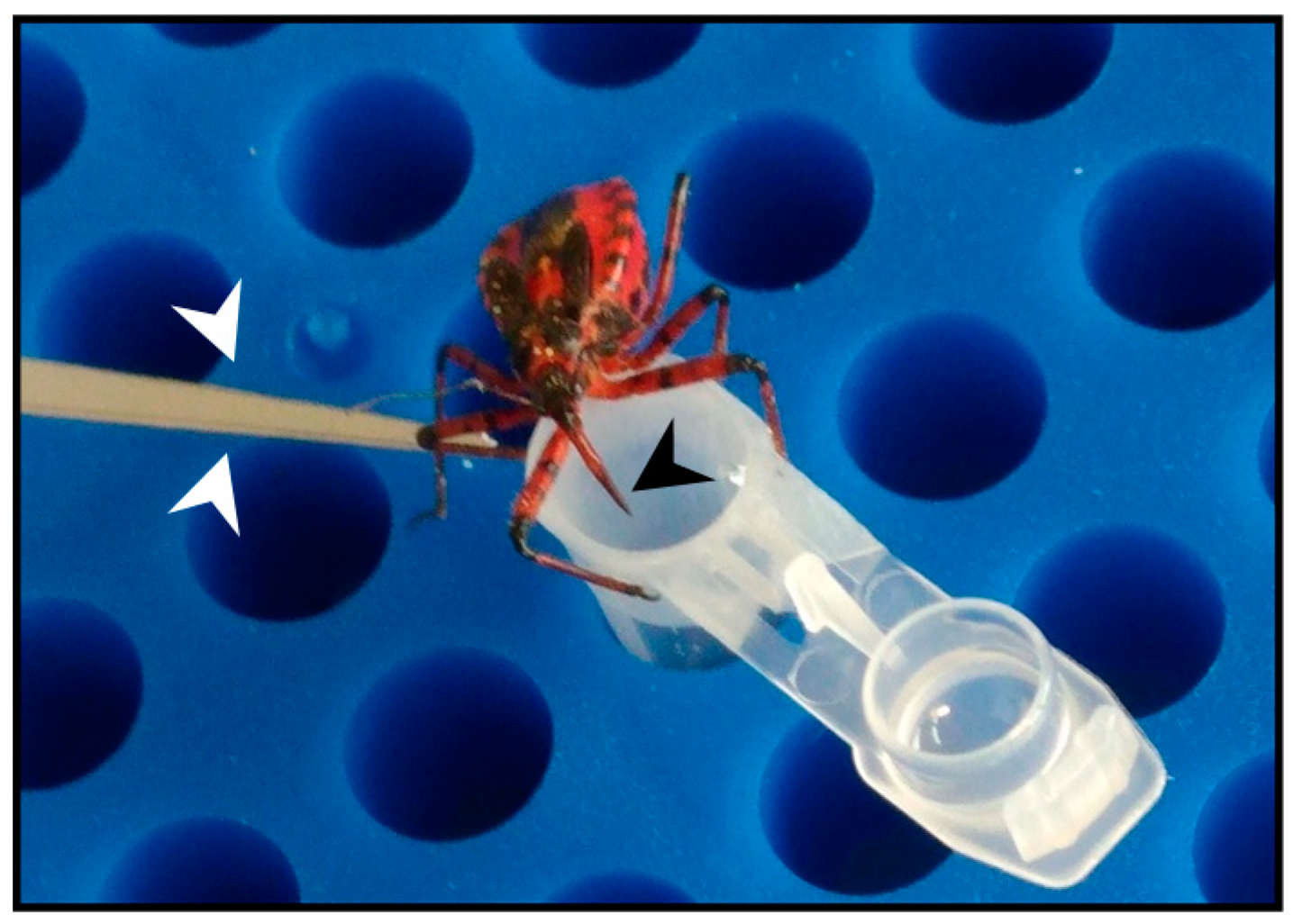
4.3. Assassin Bug Collection and Rearing
4.4. Venom Collection
4.5. Evaluation of the Physiological Effects of Venom
4.6. Proliferation Assay and CLSM
4.7. Hemolytic Activity Assay
4.8. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Hotez, P.J.; Bundy, D.A.P.; Beegle, K.; Brooker, S.; Drake, L.; de Silva, N.; Montresor, A.; Engels, D.; Jukes, M.; Chitsulo, L.; et al. Helminth infections: Soil-transmitted helminth infections and schistosomiasis. In Disease Control Priorities in Developing Countries; Jamison, D.T., Breman, J.G., Measham, A.R., Alleyne, G., Claeson, M., Evans, D.B., Jha, P., Mills, A., Musgrove, P., Eds.; Oxford University Press: Washington, DC, USA; New York, NY, USA, 2006. [Google Scholar]
- Feigin, V. Global, regional, and national incidence, prevalence, and years lived with disability for 310 diseases and injuries, 1990–2015: A systematic analysis for the Global Burden of Disease Study 2015. Lancet 2016, 388, 1545–1602. [Google Scholar]
- Newman, D.J.; Cragg, G.M. Natural products as sources of new drugs over the 30 years from 1981 to 2010. J. Nat. Prod. 2012, 75, 311–335. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Neves, B.J.; Andrade, C.H.; Cravo, P.V. Natural products as leads in schistosome drug discovery. Molecules 2015, 20, 1872–1903. [Google Scholar] [CrossRef] [PubMed]
- Moser, W.; Schindler, C.; Keiser, J. Drug Combinations against Soil-Transmitted Helminth Infections. Adv. Parasitol. 2019, 103, 91–115. [Google Scholar]
- Colley, D.G.; Bustinduy, A.L.; Secor, W.E.; King, C.H. Human schistosomiasis. Lancet 2014, 383, 2253–2264. [Google Scholar] [CrossRef]
- Hotez, P.J.; Alvarado, M.; Basanez, M.G.; Bolliger, I.; Bourne, R.; Boussinesq, M.; Brooker, S.J.; Brown, A.S.; Buckle, G.; Budke, C.M.; et al. The global burden of disease study 2010: Interpretation and implications for the neglected tropical diseases. PLoS Negl. Trop. Dis. 2014, 8, e2865. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cheever, A.W.; Macedonia, J.G.; Mosimann, J.E.; Cheever, E.A. Kinetics of egg production and egg excretion by Schistosoma mansoni and S. japonicum in mice infected with a single pair of worms. Am. J. Trop. Med. Hyg. 1994, 50, 281–295. [Google Scholar] [PubMed] [Green Version]
- Doenhoff, M.J.; Cioli, D.; Utzinger, J. Praziquantel: Mechanisms of action, resistance and new derivatives for schistosomiasis. Curr. Opin. Infect. Dis. 2008, 21, 659–667. [Google Scholar] [CrossRef]
- Fallon, P.G.; Doenhoff, M.J. Drug-resistant schistosomiasis: Resistance to praziquantel and oxamniquine induced in Schistosoma mansoni in mice is drug specific. Am. J. Trop. Med. Hyg. 1994, 51, 83–88. [Google Scholar] [CrossRef]
- Botros, S.S.; Bennett, J.L. Praziquantel resistance. Expert Opin. Drug Discov. 2007, 2, S35–S40. [Google Scholar] [CrossRef]
- Mwangi, I.N.; Sanchez, M.C.; Mkoji, G.M.; Agola, L.E.; Runo, S.M.; Cupit, P.M.; Cunningham, C. Praziquantel sensitivity of Kenyan Schistosoma mansoni isolates and the generation of a laboratory strain with reduced susceptibility to the drug. Int. J. Parasitol. Drugs Drug Resist. 2014, 4, 296–300. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cioli, D.; Pica-Mattoccia, L.; Basso, A.; Guidi, A. Schistosomiasis control: Praziquantel forever? Mol. Biochem. Parasitol. 2014, 195, 23–29. [Google Scholar] [CrossRef] [PubMed]
- De Moraes, J. Natural products with antischistosomal activity. Future Med. Chem. 2015, 7, 801–820. [Google Scholar] [CrossRef] [PubMed]
- Herzig, V.; Cristofori-Armstrong, B.; Israel, M.R.; Nixon, S.A.; Vetter, I.; King, G.F. Animal toxins-Nature’s evolutionary-refined toolkit for basic research and drug discovery. Biochem. Pharmacol. 2020, 114096. [Google Scholar] [CrossRef]
- Mohamed Abd El-Aziz, T.; Garcia Soares, A.; Stockand, J.D. Snake venoms in drug discovery: Valuable therapeutic tools for life saving. Toxins 2019, 11, 564. [Google Scholar] [CrossRef] [Green Version]
- El-Asmar, M.F.; Swelam, N.; Abdel Aal, T.M.; Ghoneim, K.; Hodhod, S.S. Factor(s) in the venom of scorpions toxic to Schistosoma mansoni (intestinal belharzia) cercariae. Toxicon 1980, 18, 711–715. [Google Scholar] [CrossRef]
- Stábeli, R.G.; Amui, S.F.; Sant’Ana, C.D.; Pires, M.G.; Nomizo, A.; Monteiro, M.C.; Romão, P.R.; Guerra-Sá, R.; Vieira, C.A.; Giglio, J.R.; et al. Bothrops moojeni myotoxin-II, a Lys49-phospholipase A2 homologue: An example of function versatility of snake venom proteins. Comp. Biochem. Physiol. C Toxicol. Pharmacol. 2006, 142, 371–381. [Google Scholar]
- De Moraes, J.; Nascimento, C.; Miura, L.M.; Leite, J.R.; Nakano, E.; Kawano, T. Evaluation of the in vitro activity of dermaseptin 01, a cationic antimicrobial peptide, against Schistosoma mansoni. Chem. Biodivers. 2011, 8, 548–558. [Google Scholar] [CrossRef]
- Hassan, E.A.; Abdel-Rahman, M.A.; Ibrahim, M.M.; Soliman, M.F. In vitro antischistosomal activity of venom from the Egyptian snake Cerastes cerastes. Rev. Soc. Bras. Med. Trop. 2016, 49, 752–757. [Google Scholar] [CrossRef] [Green Version]
- Mohamed, A.H.; Hassab El-Nabi, S.E.; Bayomi, A.E.; Abdelaal, A.A. Effect of bee venom or proplis on molecular and parasitological aspects of Schistosoma mansoni infected mice. J. Parasit. Dis. 2016, 40, 390–400. [Google Scholar] [CrossRef] [Green Version]
- Bailey, P.C. The feeding behaviour of a sit-and wait-predator, Ranatra dispar (Heteroptera: Nepidae): Optimal foraging and feeding dynamics. Oecologia 1986, 68, 291–297. [Google Scholar] [CrossRef] [PubMed]
- Sano-Martins, I.S.; González, C.; Anjos, I.V.; Díaz, J.; Gonçalves, L.R.C. Effectiveness of Lonomia antivenom in recovery from the coagulopathy induced by Lonomia orientoandensis and Lonomia casanarensis caterpillars in rats. PLoS Negl. Trop. Dis. 2018, 12, e0006721. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Arif, F.; Williams, M. Hymenoptera Stings (Bee, Vespids and Ants); StatPearls Publishing LLC: Treasure Island, FL, USA, 2020. [Google Scholar]
- Walker, A.A.; Weirauch, C.; Fry, B.G.; King, G.F. Venoms of heteropteran insects: A treasure trove of diverse pharmacological toolkits. Toxins 2016, 8, 43. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hwang, W.S.; Weirauch, C. Evolutionary history of assassin bugs (insecta: Hemiptera: Reduviidae): Insights from divergence dating and ancestral state reconstruction. PLoS ONE 2012, 7, e45523. [Google Scholar] [CrossRef] [Green Version]
- Edwards, J.S. The action and composition of the saliva of an assassin bug Platymeris rhadamanthus Gaerst (Hemiptera, Reduviidae). J. Exp. Biol. 1961, 38, 61–77. [Google Scholar]
- Fischer, G.; Conceicao, F.R.; Leite, F.P.; Dummer, L.A.; Vargas, G.D.; Hubner Sde, O.; Dellagostin, O.A.; Paulino, N.; Paulino, A.S.; Vidor, T. Immunomodulation produced by a green propolis extract on humoral and cellular responses of mice immunized with SuHV-1. Vaccine 2007, 25, 1250–1256. [Google Scholar] [CrossRef]
- Kellershohn, J.; Thomas, L.; Hahnel, S.R.; Grunweller, A.; Hartmann, R.K.; Hardt, M.; Vilcinskas, A.; Grevelding, C.G.; Haeberlein, S. Insects in anthelminthics research: Lady beetle-derived harmonine affects survival, reproduction and stem cell proliferation of Schistosoma mansoni. PLoS Negl. Trop. Dis. 2019, 13, e0007240. [Google Scholar] [CrossRef] [Green Version]
- Wendt, G.R.; Collins, J.J., 3rd. Schistosomiasis as a disease of stem cells. Curr. Opin. Genet. Dev. 2016, 40, 95–102. [Google Scholar] [CrossRef] [Green Version]
- Collins, J.J., 3rd; Wang, B.; Lambrus, B.G.; Tharp, M.E.; Iyer, H.; Newmark, P.A., 3rd; Wang, B.; Lambrus, B.G.; Tharp, M.E.; Iyer, H.; et al. Adult somatic stem cells in the human parasite Schistosoma mansoni. Nature 2013, 494, 476–479. [Google Scholar]
- Monincová, L.; Budesínský, M.; Slaninová, J.; Hovorka, O.; Cvacka, J.; Voburka, Z.; Fucík, V.; Borovicková, L.; Bednárová, L.; Straka, J.; et al. Novel antimicrobial peptides from the venom of the eusocial bee Halictus sexcinctus (Hymenoptera: Halictidae) and their analogs. Amino Acids 2010, 39, 763–775. [Google Scholar]
- Mortari, M.R.; do Couto, L.L.; dos Anjos, L.C.; Mourão, C.B.; Camargos, T.S.; Vargas, J.A.; Oliveira, F.N.; Gati Cdel, C.; Schwartz, C.A.; Schwartz, E.F. Pharmacological characterization of Synoeca cyanea venom: An aggressive social wasp widely distributed in the Neotropical region. Toxicon 2012, 59, 163–170. [Google Scholar] [CrossRef] [PubMed]
- Erasmus, D.A. A comparative study of the reproductive system of mature, immature and “unisexual” female Schistosoma mansoni. Parasitology 1973, 67, 165–183. [Google Scholar] [CrossRef] [PubMed]
- Cornara, L.; Biagi, M.; Xiao, J.; Burlando, B. Therapeutic properties of bioactive compounds from different honeybee products. Front. Pharmacol. 2017, 8, 412. [Google Scholar] [CrossRef]
- Vilcinskas, A.; Stoecker, K.; Schmidtberg, H.; Rohrich, C.R.; Vogel, H. Invasive harlequin ladybird carries biological weapons against native competitors. Science 2013, 340, 862–863. [Google Scholar] [CrossRef]
- Van Hellemond, J.J.; Retra, K.; Brouwers, J.F.; van Balkom, B.W.; Yazdanbakhsh, M.; Shoemaker, C.B.; Tielens, A.G. Functions of the tegument of schistosomes: Clues from the proteome and lipidome. Int. J. Parasitol. 2006, 36, 691–699. [Google Scholar] [CrossRef]
- Tonk, M.; Institute for Insect Biotechnology, Justus Liebig University of Giessen, Giessen, Germany. Personal communication, 2020.
- Koziol, U.; Rauschendorfer, T.; Zanon Rodriguez, L.; Krohne, G.; Brehm, K. The unique stem cell system of the immortal larva of the human parasite Echinococcus multilocularis. Evodevo 2014, 5, 10. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhou, H.; Li, D.; Shi, C.; Xin, T.; Yang, J.; Zhou, Y.; Hu, S.; Tian, F.; Wang, J.; Chen, Y. Effects of Exendin-4 on bone marrow mesenchymal stem cell proliferation, migration and apoptosis in vitro. Sci. Rep. 2015, 5, 12898. [Google Scholar] [CrossRef]
- Miao, Z.; Lu, Z.; Luo, S.; Lei, D.; He, Y.; Wu, H.; Zhao, J.; Zheng, L. Murine and Chinese cobra venom-derived nerve growth factor stimulate chondrogenic differentiation of BMSCs in vitro: A comparative study. Mol. Med. Rep. 2018, 18, 3341–3349. [Google Scholar] [CrossRef]
- Assimakopoulos, S.F.; Tsamandas, A.C.; Georgiou, C.D.; Vagianos, C.E.; Scopa, C.D. Bombesin and neurotensin exert antiproliferative effects on oval cells and augment the regenerative response of the cholestatic rat liver. Peptides 2010, 31, 2294–2303. [Google Scholar] [CrossRef]
- Liu, J.; Zhang, Y.; Sun, S.; Zhang, G.; Jiang, K.; Sun, P.; Zhang, Y.; Yao, B.; Sui, R.; Chen, Y.; et al. Bufalin induces apoptosis and improves the sensitivity of human glioma stem-like sells to temozolamide. Oncol. Res. 2019, 27, 475–486. [Google Scholar] [CrossRef]
- White, J. Bites and stings from venomous animals: A global overview. Ther. Drug Monit. 2000, 22, 65–68. [Google Scholar] [CrossRef] [PubMed]
- Walker, A.A.; Madio, B.; Jin, J.; Undheim, E.A.; Fry, B.G.; King, G.F. Melt with this kiss: Paralyzing and liquefying venom of the assassin bug Pristhesancus plagipennis (hemiptera: Reduviidae). Mol. Cell. Proteom. 2017, 16, 552–566. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Walker, A.A.; Robinson, S.D.; Undheim, E.A.B.; Jin, J.; Han, X.; Fry, B.G.; Vetter, I.; King, G.F. Missiles of mass disruption: Composition and glandular origin of venom used as a projectile defensive weapon by the assassin bug Platymeris rhadamanthus. Toxins 2019, 11, 673. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gönnert, R. Schistosomiasis-Studien. II. Über die Eibildung bei Schistosoma mansoni und das Schicksal der Eier im Wirtsorganismus. Z. Trop. Parasitol. 1955, 6, 33–52. [Google Scholar]
- Dettman, C.D.; Higgins-Opitz, S.B.; Saikoolal, A. Enhanced efficacy of the paddling method for schistosome infection of rodents by a four-step pre-soaking procedure. Parasitol. Res. 1989, 76, 183–184. [Google Scholar] [CrossRef]
- Grevelding, C.G. The female-specific W1 sequence of the Puerto Rican strain of Schistosoma mansoni occurs in both genders of a Liberian strain. Mol. Biochem. Parasitol. 1995, 71, 269–272. [Google Scholar] [CrossRef]
- Ramirez, B.; Bickle, Q.; Yousif, F.; Fakorede, F.; Mouries, M.A.; Nwaka, S. Schistosomes: Challenges in compound screening. Expert Opin. Drug Discov. 2007, 2, S53–S61. [Google Scholar] [CrossRef]
- Hahnel, S.; Quack, T.; Parker-Manuel, S.J.; Lu, Z.; Vanderstraete, M.; Morel, M.; Dissous, C.; Cailliau, K.; Grevelding, C.G. Gonad RNA-specific qRT-PCR analyses identify genes with potential functions in schistosome reproduction such as SmFz1 and SmFGFRs. Front. Genet. 2014, 5, 170. [Google Scholar] [CrossRef] [Green Version]
- Neves, R.H.; de Lamare Biolchini, C.; Machado-Silva, J.R.; Carvalho, J.J.; Branquinho, T.B.; Lenzi, H.L.; Hulstijn, M.; Gomes, D.C. A new description of the reproductive system of Schistosoma mansoni (Trematoda: Schistosomatidae) analyzed by confocal laser scanning microscopy. Parasitol. Res. 2005, 95, 43–49. [Google Scholar] [CrossRef]
- Beckmann, S.; Grevelding, C.G. Imatinib has a fatal impact on morphology, pairing stability and survival of adult Schistosoma mansoni in vitro. Int. J. Parasitol. 2010, 40, 521–526. [Google Scholar] [CrossRef]
- Tonk, M.; Pierrot, C.; Cabezas-Cruz, A.; Rahnamaeian, M.; Khalife, J.; Vilcinskas, A. The Drosophila melanogaster antimicrobial peptides Mtk-1 and Mtk-2 are active against the malarial parasite Plasmodium falciparum. Parasitol. Res. 2019, 118, 1993–1998. [Google Scholar] [CrossRef] [PubMed]
- Marx, A.; Backes, C.; Meese, E.; Lenhof, H.P.; Keller, A. EDISON-WMW: Exact dynamic programing solution of the Wilcoxon-Mann-Whitney test. Genom. Proteom. Bioinform. 2016, 14, 55–61. [Google Scholar] [CrossRef] [PubMed] [Green Version]






© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tonk, M.; Vilcinskas, A.; Grevelding, C.G.; Haeberlein, S. Anthelminthic Activity of Assassin Bug Venom against the Blood Fluke Schistosoma mansoni. Antibiotics 2020, 9, 664. https://doi.org/10.3390/antibiotics9100664
Tonk M, Vilcinskas A, Grevelding CG, Haeberlein S. Anthelminthic Activity of Assassin Bug Venom against the Blood Fluke Schistosoma mansoni. Antibiotics. 2020; 9(10):664. https://doi.org/10.3390/antibiotics9100664
Chicago/Turabian StyleTonk, Miray, Andreas Vilcinskas, Christoph G. Grevelding, and Simone Haeberlein. 2020. "Anthelminthic Activity of Assassin Bug Venom against the Blood Fluke Schistosoma mansoni" Antibiotics 9, no. 10: 664. https://doi.org/10.3390/antibiotics9100664





