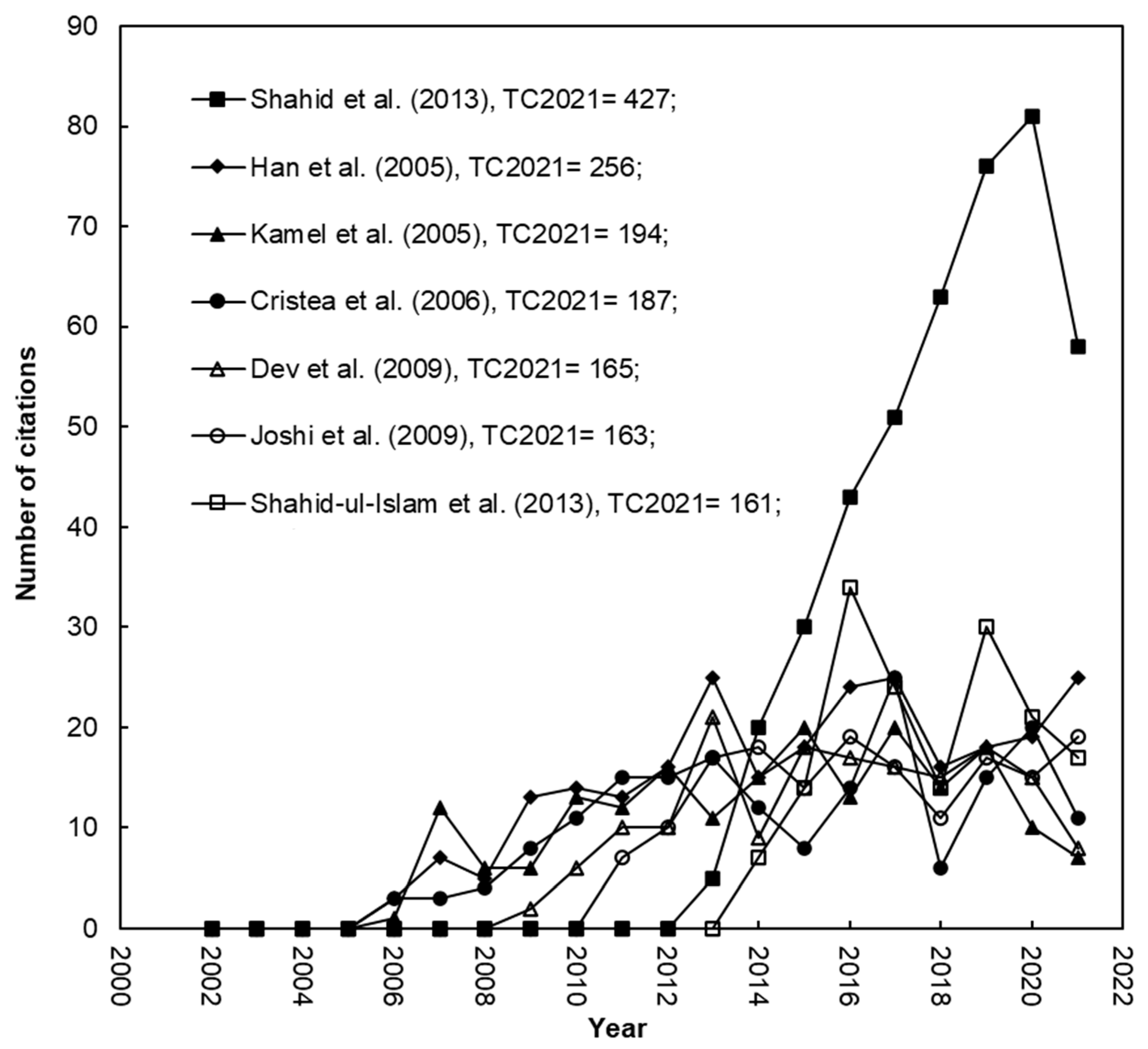
According to a number of bibliometric studies, the distribution of words in article titles, abstracts, author keywords, and KeyWords Plus over time can be used to evaluate key research goals and then reveal how they change over time in study subjects [
45]. For the period of the study, KeyWords Plus, author keywords, and article titles were all assessed and ranked in accordance with their frequency. More dyeing was used in subsequent research on the natural coloration of textile materials than other coloration methods such as printing (
Figure 5).
Table 6 lists the 20 most popular words in the titles and author keywords along with the KeyWords Plus rankings for each. The most popular search terms, such as dyeing, cloth, or natural, did not provide any information about the research hotspots. Cotton, a cellulosic textile fiber, wool, and silk, both protein fibers [
46] appeared as the 5th, 8th, and 12th top most frequently searched words respectively and not indicate a research hotspot. The outcomes of a word analysis produced a number of supporting terms for each word cluster. “Mordant”, “antibacterial”, “ultraviolet”, “fastness”, “antimicrobials”, and “ultrasonic” were the six potential areas of study for textile coloring using natural sources. Fastness and mordanting are discussed together since they are typically used to enhance the fastness qualities of colored substrates. Two of the six research clusters are significantly connected to the functionalization of textile materials. This is not unexpected given how frequently textile materials are functionalized in current textile research [
47]. The following sections provide summaries of influential and highly referenced articles that are relevant to certain research spots.
3.6.1. Fastness and Mordants
All articles containing mordant, mordanting, and fastness on their front page are displayed in
Figure 5, which shows that studies on improving fastness properties via mordanting rose rapidly and have taken the lead since 2007. Colorfastness is the primary requirement of any dyed textile goods. Due to the lower substantivity and durability [
48] and also the non-substantive nature of most of the natural dyes [
49], fatness improvement is required to obtain better and improved end products [
48]. Otherwise, textile coloration with natural sources will not be acceptable for commercial usage as a suitable alternative.
C. urucurana bark extract was used as a natural dye source for dyeing cotton and wool fabric. The fastness properties (fastness to light, wash, perspiration, and color staining) were generally good. Aluminum potassium sulfate (alum) and ferrous sulfate (6% owf) were used in a meta-mordanting process to improve the fastness properties. Notably, alum treatment decreased cotton’s light fastness, and ferrous sulfate showed better light fastness than the sample mordanted with alum. In all other fastness tests, the mordanted sample showed slightly better or similar fastness results [
1]. Dyeing silk fabric with turmeric extract using potassium aluminum sulfate, copper sulfate, and ferrous sulfate mordant showed a golden-yellow color, brown color, and brownish-grey color, respectively [
49]. Yusuf et al. [
48] conducted a similar study where a natural dye was extracted from R. cordifolia roots and found that it can be applied to obtain radiant red shades on wool with or without mordanting. However, mordanting with AlCl
3 or CaCl
2 increased the overall fastness properties, and the former was more efficient than the latter. It was observed that woolen yarn can be dyed with gallnut extract with or without mordanting to produce a bright ivory to light brownish-yellow color where good fastness properties to light, rubbing, and washing could be seen [
44].
Mixtures of red sandalwood and other natural dyes (jackfruit wood, sappan wood, manjistha, marigold, babool) were used to dye jute fabric pre-mordanted with myrobolan and then aluminum sulfate. For the use of the combinations of red sandalwood and babool, red sandalwood and marigold, and red sandalwood and sappan wood, a relatively lower wash fastness rating (grade 2–3) was observed. However, the wash fastness rating was relatively higher (typically grade 3–4) for the combinations of red sandalwood and manjistha and red sandalwood and jackfruit wood without any after-treatment [
50]. The fastness properties of flax fabric dyed with natural colorants were also improved using mordanting. Cellulase-pretreated flax fabric dyed with an extract from chestnut shell showed fair to good fastness to light, crocking, and washing when it was mordanted with aluminum potassium sulfate and stannous chloride [
8].
A similar approach used turmeric rhizomes to dye a bleached and mercerized cotton fabric. The fabric and turmeric powder were treated with gamma irradiation, but to increase color fastness, alum was used as a mordant [
51]. In another study by Khan et al. [
43], red calico leaf powder and cotton fabric cotton were irradiated. The surface modification converted the hydroxyl groups of the cotton molecule to a carboxylic group, and it helped to form a strong interaction with the dye molecule. In their study, it was found that using 1% copper solution and 1% tannic acid as a pre-mordant and post-mordant, respectively, helped to get slightly better results for fastness properties. Moreover, the irradiation of powder (calico leaves) and fabric (cotton) before dyeing reduced the amount of mordanting required to achieve an acceptable color fastness and color strength. A similar result was seen when mercerized cotton fabric was dyed with chicken gizzard extract. Here, it was found that iron (5%) and tannic acid (1%) as a pre- and post-mordant, respectively, gave the best result. Iron stabilized in the complex formed by irradiation due to its low reduction power. The enhanced color strength was due to the carboxylic group in the irradiated cotton and the phenolic group in the natural dye. Tannic acid also formed a firm complex on the fabric upon reaction with dye molecules. A conjugated system and benzene ring in colorant improved the resistance to heat, light, detergent, and rubbing. Gamma irradiation on mercerized cotton increased the color fastness rating and color strength, saving time, money, and labor [
52].
In all these studies, the fastness properties were mostly improved by mordanting with metal salts, which has substantivity for both the fiber component and the dye molecules. They form an insoluble precipitate with the colorant on the fiber which results in better fastness properties [
53].
3.6.2. Ultrasonic
The number of articles mentioning ultrasonic, ultrasonication, and ultrasound on their first page has been increasing steadily since 2007 (
Figure 5). The conventional procedures are becoming less and less relevant due to their low extraction yield, prolonged use, degradation of the active ingredients, and poor fastness qualities. Modern extraction methods currently use gamma radiation, ultraviolet (UV) radiation, microwave radiation, ultrasonic radiation, and plasma treatment [
54]. Because of its many advantages, one of these contemporary procedures, ultrasonic-aided extraction, is referred to as a “Green Process.” Low temperatures produce the best results from the ultrasonic instrument, protecting the extract’s thermally delicate bioactive components. The ultrasonic method delivers a good extraction yield at a lower temperature due to sound cavitation, which is more efficient at lower temperatures. Another advantage of the ultrasonic treatment is that it allows for improved functional component isolation thanks to mass transfer kinetics.
In a study by Zia et al. [
55], the crude powder of neem bark was boiled for one hour in different solvents, including water, HCl (2%), and acidic methanol (2%). The mixtures were boiled and filtered, and the extracts were subjected to ultrasonic treatment for 15–60 min. Pristine and irradiated cotton samples were dyed at 75 °C by maintaining M:L = 1:25 using unirradiated and irradiated extracts. Adeel et al. [
54] used a similar extraction method that showed promising results where ultrasonic treatment was employed to extract natural colorant from neem bark to dye silk fabric. Neem-bark-based tannin, a natural coloring agent for silk dyeing, was separated from 8 g of crude powder in a methanolic medium for 30 min followed by 30 min of ultrasound treatment. This treated powder was then used to dye ultrasound-treated silk for 65 min at 75 °C. Compared to chemical mordants employed under mild extraction and dyeing circumstances, biomordants such as quercetin from acacia, lawsone from Heena, curcumin from turmeric, and tannin from pomegranate yielded good to exceptional ratings of fastness and color strength. It was concluded that ultrasonic treatment improved the separation of the functional moiety (tannin) and decreased the size of the colorant molecule, both of which resulted in a reduction in processing time and solvent usage. The amount of powder required for colorant extraction decreased thanks to ultrasonic radiation [
55].
In another study by Adeel et al. [
56], arjun bark was used as a source of natural coloring agent, and extracts were prepared in various media. The extracts and silk fabrics were subjected to ultrasonic exposure of 100 W and 40 KHz for about 15–60 min at 60 °C. Irradiated silk fabric dyed with natural color in the presence of 0.3 g/L salts for 65 min at 65 °C showed good color strength. The color properties were improved with pre- and postchemical mordants such as 5% aluminum sulfate and 9% iron sulfate. Biomordants such as pomegranate and turmeric extracts (9%) as pre- and post-mordants produced good coloring properties. In contrast to the ultrasonic treatment of both the extracts and silk in a basic medium, ultrasonic-assisted dyeing for 15 min in an aqueous medium yielded the best color strength. However, irradiation did not impact dyeing when using an acidic medium.
After extracting natural dye from pomegranate peel using an ultrasonic technique, several mordants were applied to lyocell fabrics. Afterward, exhaust dyeing was used to color the samples of mordanted fabric. A volume of 200 mL of solvent made of 80 mL distilled water and 120 mL ethanol was added to a beaker containing 10 g of powdered pomegranate peel at an M: L of 1:20. After that, the beakers were placed in the ultrasonic bath and sonicated for 1 h at a temperature of 50 °C, a frequency of 27–30 MHz, and a supply voltage of 160 V. Then, using a rotary evaporator, the solvents were evaporated from the solutions after being filtered through Whatman filter paper [
57].
Cotton did not show any affinity for the
P. quinquefolia natural dye. To increase its dyeability, cationic groups were applied to cotton fibers using the cationic chemical Croscolor DRT (formaldehyde-free dye-fixing agent). Ultrasonic extraction of the natural dye enabled the dyeing of cotton fabric. The natural dye extraction from
P. quinquefolia was performed utilizing ultrasonic energy at 500 W with a frequency of 20 kHz. A volume of 100 mL of water was added to a glass containing 5 g of P. quinquefolia fruits for extraction. This item was sonicated for 120 min in water. After complete extraction, the extract was run over filter paper [
58]. Ultrasonic energy was used to dye cotton fabric with red cabbage in the study by Ticha et al. [
59] where several cationizing agents were used to modify cotton, including the Rewin Os, Sera Fast, Denitex BC, and Acid Tannic. Ultrasonic power was used to carry out the dyeing process. pH 9 was maintained in the dye bath. The colorant was employed at a concentration of 30 g. The temperature was increased to 80 °C and held there for 60 min. Each of the investigated cationizing agents affected cotton. The sonicator dyeing tests were conducted in a glass beaker with a flat bottom equipped with a refrigerant. Ultrasonic power of 500 W at a 20 kHz frequency was employed. In a different approach, cotton fabric was pretreated with enzymes (pro-amyl, diasterase, lipase) before being dyed with the natural colorants catechu and tectona. The dye uptake capabilities of the fabrics treated with various enzymes were assessed, and the results were contrasted with a control sample. Protease and amylase were the enzymes for the catechu dye, but diasterase was best for tectona. Dye exhaustion increased by 39 and 52 percent with ultrasound. As a result, it was demonstrated that ultrasonography can be a useful technique for cleaner textile dyeing [
60]. In other research, the dye uptake of cationized cotton fabric with lac dye by ultrasound was successful. The improved effect after equilibrium dyeing was approximately 66.5% greater than that of traditional heating. In addition to reducing processing time and energy consumption, this method positively affects the environment because it facilitates increased dye uptake and effective dye bath reuse [
42].
The ideal conditions were used to extract almond shell powder (1 g/150 mL water) in an ultrasonic bath (Selecta, 40 kHz, 250 W) at 80 °C for 60 min. After being cooled to room temperature, the extracts were filtered to eliminate the insoluble residues. The ideal conditions for sonicator dyeing were an ultrasonic bath (Selecta, 40 kHz, 250 W) at 80 °C for 60 min. After thoroughly rinsing with water, the dyed samples were allowed to dry at room temperature [
61]. In this study, it was concluded that the process enhanced the dye extraction efficacy but the obtained shade was lighter in ultrasonic-assisted dyeing and could be improved by mordanting and biomordanting. In the case of biomordanting, rosemary could be an alternative to alum which could be used in pre- and post-mordanting conditions. In contrast, in terms of fastness properties, pomegranate rind and valex were equal to metallic mordants, i.e., alum, iron II sulfate, copper II sulfate, and potassium dichromate. Other notable studies using a biomordant for the natural coloration of silk [
62] and wool [
63] have also been reported.
A study conducted by Baaka, Haddar et al. showed that wool fabrics could be dyed with the natural extract from grape pomace, and the use of ultrasound improved the dye exhaustion and fixation and ultimately the color yield (K/S) and fastness [
64].
Finally, it can be said that ultrasonic treatment is a time-, cost-, energy-, and labor-effective strategy in addition to being more uniform, clean, and sustainable and can be used to isolate colorant from several new plants that produce dye and to achieve darker colors with appropriate fastness qualities for natural fabrics [
54].
3.6.3. Ultraviolet (UV)
Due to their multipurpose qualities, such as UV protection, deodorizing performance, and antibacterial activity, natural dyes have recently sparked an increased interest in creating aesthetic and functional textiles [
65]. Among these properties, UV protective finishing has attracted the attention of many researchers. The UV-A band (320–400 nm), the UV-B band (290–320 nm), and the UV-C band make up the UVR band (200–290 nm). Only a small portion of these dangerous radiations (UV-A 94%, UV-B 6%) [
66] reach the earth’s surface since the atmosphere absorbs a large quantity of them. Generally speaking, these radiations cause harm including wrinkles, skin damage, and even skin cancer [
67]. Human skin’s melanin pigment reduces the resulting harm. To further protect skin from damaging UV rays, clothing has been regarded as a great shield. Clothing dyed with natural colorants usually show increased UV protection as most natural colorants absorb both the UV and visible regions and have good UV radiation-blocking properties [
68].
In a study, it was found that dyeing wool with marigold extract imparts excellent UV protection. Less than a 5 percent transmission rate for colored materials indicates adequate UV protection. All dyed wool fabric samples exhibit percentage transmittance values of 5 across the entire UV region. Still, undyed wool fabric exhibits poor UV protection with values of more than 10% in the UV-C area and rising to 40% in the UV-A region. The UV transmittance of wool is inversely correlated with the dye concentration; as the dye concentration grows, the transmittance decreases, and the protection increases. Alum and iron mordants reduced the UV transmittance; in the case of iron, it was found to be below 1.5% throughout the UV area. Undyed wool fabric has a 6.85 ultraviolet protection factor (UPF) rating, which is considered poor in terms of UV protection. Increased dye concentrations (10–20% o.w.f.) showed good 50+ UPF ratings, as the dyed materials’ UPF proportionally correlates to the extract% [
69], and even 5% o.w.f. marigold extract noticeably improved the UPF rating up to 35+ on wool. Due to the combined effects of the dye molecules and iron’s UV-screening properties, colored wool that had been iron-mordanted was discovered to be highly UV-protective. Tin mordant was shown to have a lower UPF, even though it was over 40, which is considered excellent [
67].
In another study that dyed cotton fabric with the extract of eucalyptus leaves, samples colored with extract concentrations of 20 g/L or more were considered to have good UV protection. Compared to samples treated with chitosan-untreated ones, the colored samples showed a considerable rise in their mean UPF values. Additionally, as the extract concentration in the dyeing increases, so do the UPF values. As a result, dyed materials (10–20 g/L extract) treated with chitosan had very good UPF indices, but they were excellent for specimens dyed with extracts at concentrations of 30 g/L or more. The mechanism involved in these two studies could be concluded as the increased uptake of the extract due to the chitosan treatment increasing the organic substrate, i.e., flavonoids increased the UPF rating [
69]. Still, in the first case, the addition of the organic contents was increased by mordanting, and also these mordants had UV absorbance and dissipation properties [
67]. Another approach to dyeing cotton fabric with tea extracts found that tea extract exhibits various behaviors depending on the type of tea being studied—green, red, or black. In terms of UPF, red and black tea displayed greater levels while green tea displayed the lowest value. Still, it must be noted that cotton fabrics colored with green tea have UPF values within the range where they can be considered to provide decent protection. Additionally, red and black tea extracts perform better when boiled than green tea extracts when cold [
70].
Grifoni et al. [
71] attempted to dye cotton and flax fabric using extracts from natural wild plants, including
D. gnidium L. (daphne),
L. stoechas L. (wild lavender),
R. peregrina L. (wild madder),
H. italicum (Roth) G. Don (curry plant), and
C. scolymus L. (artichoke). Undyed flax fabrics had lower transmittance values than undyed cotton fabrics, although both cotton and flax fabrics displayed higher transmittance values at low wavelengths. Vegetable fibers such as cotton and flax are not excellent UV filters, although their UV transmittance qualities are generally the same. Alum mordant treatment modestly decreased transmittance in cotton and flax textiles; in contrast, tannin mordant application significantly decreased transmittance over the whole UV range. Helichrysum-dyed flax fibers and Rubia-dyed and alum-mordanted fabrics maintained good UV protection even after numerous washings, but cotton fabrics did not achieve UV protection.
Natural dyes derived from Rheum root and
L. erythrorhizon root can successfully dye cotton fabric and silk. About 80% of the UV radiation is absorbed by the cotton and silk fabrics colored with these natural dyes. The fundamental explanation for the UV-protective qualities was that natural colors absorb UV light. These natural dyes were anticipated to be used in premium UV-protective clothing from cotton and silk materials [
72]. Worsted wool was dyed using orange peel extract, with aluminum mordant producing yellower and brighter hues (pre-mordant, one bath, and post-mordant), and iron mordant producing deeper and bluer shades (pre-mordant, one bath, and after mordant). The wool sample dyed directly with orange peel extract had a UPF value around seven times higher than the UPF value of the wool sample treated with synthetic dyes. Even after 30 home laundering cycles, the wool samples dyed with orange peel extract outperformed wool colored with synthetic dyes in terms of UV protection, despite the latter’s greater resistance to home laundering dyeing conditions [
73].
In an attempt by Zhou et al. [
65] to dye silk fabric with curcumin and modified curcumin, the UV absorber (2 g/L UV-Sun Cel, final concentration), sodium carbonate (2 g/L, final concentration), and curcumin (0.03 g, 0.08 mmol) were all added to a conical flask with a combined volume of 50 mL by adding distilled water. The mixture was then constantly shaken for 40 min at 60 °C in an XW-ZDR low-noise oscillated dyeing machine. The altered curcumin was then preserved in the dark for dyeing with silk. The original silk had a very low UPF of 4.91 and a high UV transmittance, indicating a poor UV protection capacity. After dyeing with modified curcumin, the silk demonstrated a high UPF of 50.65, which may be considered a “good protection” level compared to the UV absorber treatment. The silk dyed with both curcumin and modified curcumin displayed a high UPF of about 30 and maintained a “very good” UV protection performance after 30 washing cycles. Compared to undyed wool and silk, when dyed with a natural source (eucalyptus leaf) mordanted or unmordanted, wool fabric had better UV resistance properties due to its low porosity and high thickness and weight. Undyed silk and wool had a UPF value of 4.6 and 10.8, respectively, whereas treating them with eucalyptus extract increased it to 26 (unmordanted) and 53.3 (mordanted) in the case of silk and 53.1 (unmordanted) and 87.8 (mordanted) in the case of wool. The best result was obtained when samples dyed with natural extracts were mordanted with ferrous sulfate (FeSO
4) [
74].
In the case of dyeing jute fabric with several natural sources such as babool, annatto, manjistha, and ratanjot, single mordanting was not enough to impart sufficient UV protection. Double mordanting with both biomordants—myrobolan (
T. chebula) and pomegranate (
P. granatum)—and chemical mordants—ferrous sulfate (FeSO
4.7H
2O) and potash alum (hydrated salt of potassium aluminum sulfate, K
2SO
4 Al
2(SO
4)3 24 H
2O)—exhibited improved UPF ratings, and transmittance was between 4.2 and 6.7%. The pretreatment of myrobolan-ferrous sulfate provided the greatest UV protection. After pre-mordanting with a mixture of a biomordant and chemical mordant, any naturally dyed jute cloth generated exceptionally good UV protection qualities. The order of the natural-colored jute fabric’s UV protection characteristics was babool > annatto > manjistha > ratanjot [
75].
3.6.4. Antimicrobial
Medical and antimicrobial textiles protect users from hygienic issues brought on by contact with pathogenic or odor-producing bacteria [
76]. Microorganism growth reduces functionality by causing unwanted aesthetic alterations or decaying damage [
77]. Since ancient times, plant extracts have been utilized to color textile substrates. They have been researched and reintroduced as coloring and functional agents due to their biodegradable and environmentally friendly character [
78]. Some of the natural dyes have lately been demonstrated to have good antibacterial action which can impede the growth of microorganisms without being harmful.
The antibacterial properties of four natural dyes—
K. lacca,
A. catechu,
R. cordifolia,
Q. infectoria, and
R. maritimus—were the subject of an exploratory investigation. The common bacteria
E. coli,
K. pneumoniae,
B. subtilis,
P. vulgaris, and
P. aeruginosa were tested on wool samples colored with these natural colors. The best antibacterial properties against all of the investigated microorganisms were demonstrated by the
Q. infectoria dye, which was also the most effective and demonstrated the largest zone of inhibition. However, because these natural colors were absorbed below the minimum inhibitory concentration (MIC) in the textile material, it was noticed that the textile material impregnated with them displayed decreased antibacterial action. Another study found that a dye’s antibacterial effectiveness varied depending on whether it was available in solution form or was held tightly by a textile material. The textile substrate less readily absorbed these natural dyes and their MIC values were lower. It was also discovered that cotton fabric might have increased antibacterial activity (50–90%) by using
Q. infectorie extract in conjunction with copper and alum mordants [
79].
In a study, 100% pure New Zealand semiworsted woolen yarn was dyed with extracts of T. arjuna dye mordanting with ferrous sulfate (FeSO
4·5H
2O), stannous chloride (SnCl
2·2H
2O), potash alum (K
2Al
2(SO
4)4·24H
2O), and magnesium sulfate (MgSO
4·5H
2O). Different bacterial strains such as
S. aureus,
P. aeruginosa,
E. coli, and
B. subtilis were used to test the effect of antimicrobial properties. The percentage of inhibition against all tested bacteria increased with the dye extract concentration from 5 mg/mL to 10 mg/mL, demonstrating a direct proportionality. Because of the interaction between functional groups and metal ions, the maximum activity was seen in unmordanted samples, followed by various metal-mordanted samples, resulting in minimal microbial–tannin interaction. After repeated washing, the mordanted specimen retained better antimicrobial activity than the other one. The most effective use of dyed woolen yarn against the bacterial isolates was against
B. subtilis, followed by
S. aureus,
E. coli, and
P. aeruginosa [
80]. Utilizing a pre-mordanting technique, 100% semiworsted 60-count wool yarn was dyed with powdered
R. emodi L. (5% and 10%) using ferrous sulfate (5%), stannous chloride (1%), and alum (10%) as mordants, and the antimicrobial efficacy was measured against
E. coli,
S. aureus,
C. albicans, and
C. tropicalis. Wool yarn colored with 5% of the dye inhibits bacterial growth by up to 72–77% and fungal growth by up to 85–88%. When 10% dye was employed, a considerable increase was seen in antibacterial activity (82–90% in bacteria and 93–95% in fungi). When samples with dye were inspected after being mordanted, the %-age inhibition decreased. Wool yarn that had been mordanted with 5% ferrous sulfate and then dyed with 10% dye exhibited a %-age inhibition ranging from 50 to 78%, whereas with 1% stannous chloride and colored with the same %-age of dye it exhibited a %-age inhibition of 66–73%. Wool yarn with 10% alum added has the best percentage suppression (70–83%) of microbial growth [
81]. In another approach to dyeing wool with natural dye henna, it was seen that dyeing solely with henna improved the antibacterial activities, and dyeing with pretreated chitosan further improved the performance [
39].
Dried-up and de-gummed mulberry silk yarn was dyed using plant extracts made from discarded bark and leaves of the
S. asoca and
A. lebbeck species of plants. Later, the effectiveness of these substances’ antibacterial characteristics was examined in vitro against
A. niger,
K. pneumoniae, and
C. albicans. It was seen that A. lebbeck-dyed yarn had the greatest inhibitory effect on
K. pneumoniae (50%) and
A. niger (55%). The presence of all the yarns colored with different natural dyes made from the leaves and bark of both plants had the greatest impact on
A. niger’s growth. However, yarns colored with
A. lebbeck bark extract did not hinder the growth of
C. albicans,
K. pneumoniae, or
E. coli. The unmordanted colored yarns were initially treated with four cycles of washing in running water and soap solution. According to the findings, there was an average 5% reduction rate compared to yarns that just had one washing [
82].
Cotton fabric with a plain weave was dyed with a sample of
Q. infectoria dye, and its antimicrobial effectiveness against
B. subtilis and
E. coli was assessed. Fabshield AEM 5700, an antimicrobial agent, was used as a guide. It was clear that whereas cotton treated with 6%
Q. infectoria had a fairly low level of inhibition (12%), when 12%
Q. infectoria was applied, the activity was significantly increased (45% reduction) against
E. coli. The inhibition increased to 73% with the addition of 5% alum and to 57% with 5% copper. However, there was absolutely no activity while using ferrous salt. In cotton treated with Fabshield, 75% of microbial colonies were inhibited. Thus, it may be concluded that 12%
Q. infectoria combined with 5% alum can function effectively against Gram-negative bacteria
E. coli.
Q. infectoria exhibits comparable effects on the Gram-positive bacterium
B. subtilis. Comparing the activity to that against
E. coli, it is generally higher. Inhibiting microbial growth at 6%
Q. infectoria concentration is ineffective, while at 12%
Q. infectoria concentration,
B. subtilis is 60% inhibited. When combined with 5% alum and copper salts, this rises to 84% and 89%. The samples that were post-mordanted with ferrous sulfate did not exhibit any action. After the first washing, the samples treated with
Q. infectoria maintained roughly 50% of their activity, but after five launderings the action was almost lost. However, even after five launderings, the samples treated with
Q. infectoria in conjunction with 1% copper sulfate or 5% alum retained more than 75% of their activity [
83]. In another attempt to dye cotton fabric with Berberine (Natural Yellow 18) dye after pretreatment with a reactive anionic agent, the antimicrobial efficacy was assessed and evaluated with two methods: the AATCC Test Method 147–1998 and AATCC Test Method 100–1999. In the first method, the zone of bacterial suppression was not observed for the undyed control sample, which was solely treated with an anionic chemical. However, the zone of bacterial inhibition for Berberine coloring was plainly visible. This comparable result can be explained by the fact that the negatively charged bacterial cell membrane could be destroyed by the positively charged Berberine molecules, disrupting the equilibrium of charges in the cell membrane. At the same time, the Berberine-dyed sample exhibited robust antibacterial activity, with a 99.5% reduction in bacterial population compared to the undyed sample in the latter method [
84]. Similar studies on cotton to impart antibacterial functional properties were carried out by Aminoddin Haji’s group [
85,
86,
87].