Metallizing the Surface of Halloysite Nanotubes—A Review
Abstract
:1. Introduction
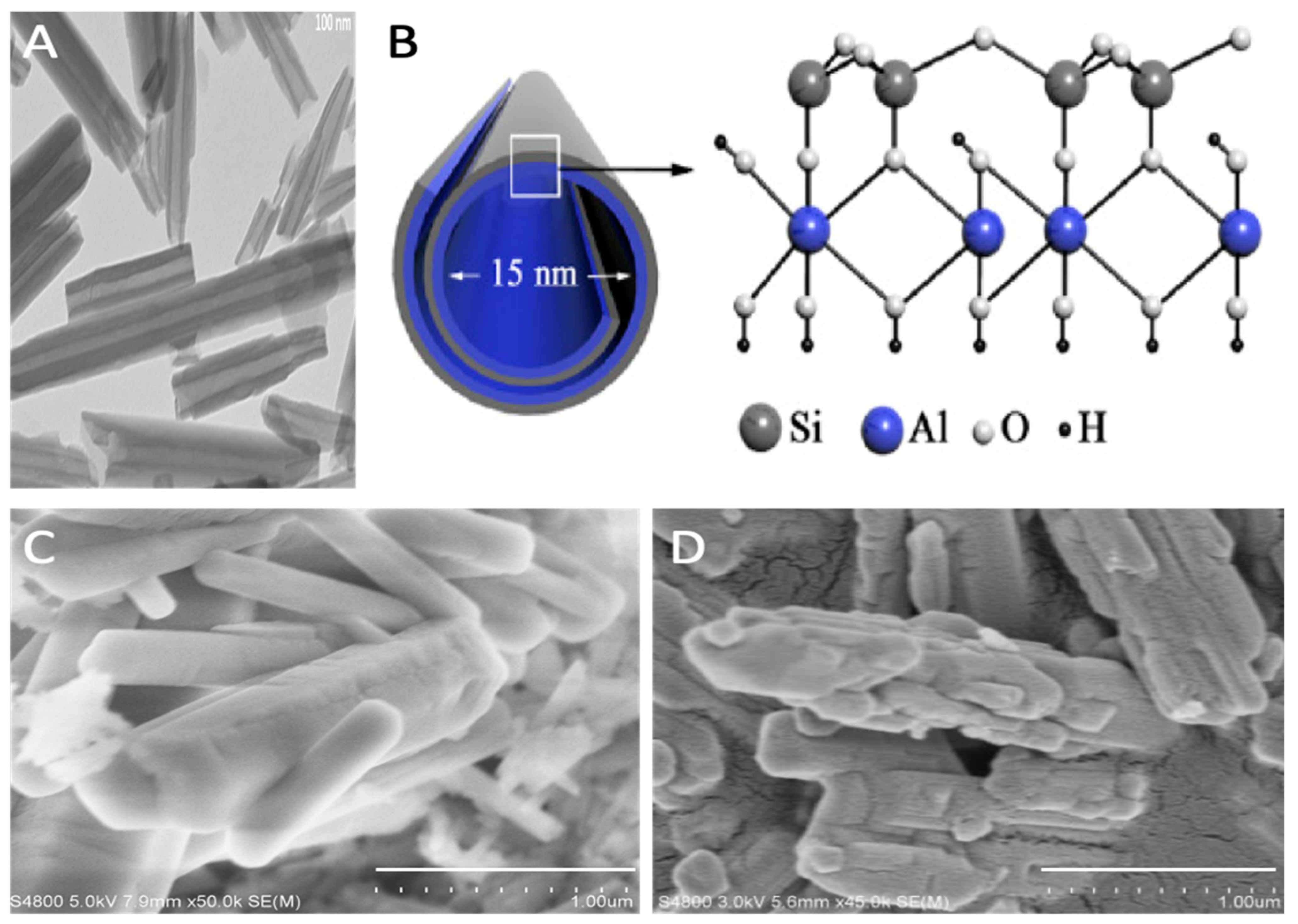
1.1. Structure and Properties of Halloysite
1.2. Surface Modification of Halloysite
1.3. Halloysite Cyto- and Biocompatibility

2. Metal Nanoparticles and Applications
3. Surface Modification of Halloysite Nanotubes with Metals
3.1. Calcination of Halloysite Nanotubes
3.2. Metal Sintering of Halloysite Nanotubes
3.3. Electrodeposition for Metallizing HNTs
3.4. Physical Adsorption or Self-Assembly
3.5. Solvothermal
4. Biomedical Applications
4.1. Antimicrobial Surfaces
4.2. 3D Printed Biomedical Devices
4.3. HNTs as a Drug Delivery System
4.4. Tissue Regeneration
5. Industrial and Military Applications
5.1. Radiation Absorptive Composites
5.2. Electronic Components
5.3. Toxic Waste Removal

5.4. Industrial Catalysts
6. Product Market Analysis and Projections
6.1. Metal and Metal Oxide Nanoparticles
6.2. Protective Films and Coatings
6.3. Textiles
7. Future Work and Impact of mHNTs
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Deepak, R.; Agrawal, Y. Multifarious applications of halloysite nanotubes: A review. Rev. Adv. Mater. Sci. 2012, 30, 282–295. [Google Scholar]
- Lvov, Y.M.; Shchukin, D.G.; Möhwald, H.; Price, R.R. Halloysite clay nanotubes for controlled release of protective agents. ACS Nano 2008, 2, 814–820. [Google Scholar] [CrossRef] [PubMed]
- Abdullayev, E.; Lvov, L. Functional polymer clay nanotube composites with sustained release of chemical agents. Prog. Polym. Sci. 2013, 38, 1690–1719. [Google Scholar]
- Veerabadran, N.G. Nanoengineered Templates for Controlled Delivery of Bioactive Compounds. Ph.D. Thesis, Louisiana Tech University, Ruston, LA, USA, 2008. [Google Scholar]
- Hu, Y.; Chen, J.; Li, X.; Sun, Y.; Huang, S.; Li, Y.; Liu, H.; Xu, J.; Zhong, S. Multifunctional halloysite nanotubes for targeted delivery and controlled release of doxorubicin in-vitro and in-vivo studies. Nanotechnology 2017, 28, 375101. [Google Scholar] [CrossRef]
- Bediako, E.G.; Nyankson, E.; Dodoo-Arhin, D.; Agyei-Tuffour, B.; Łukowiec, D.; Tomiczek, B.; Yaya, A.; Efavi, J.K. Modified halloysite nanoclay as a vehicle for sustained drug delivery. Heliyon 2018, 4, e00689. [Google Scholar] [CrossRef] [Green Version]
- Cheng, Z.-L.; Change, X.-Y.; Liu, Z. Surface modification of halloysite nanotubes grafted by dodecylamine and their application in reinforcing polytetrafluoroethylene. Clay Miner. 2019, 54, 219–225. [Google Scholar] [CrossRef]
- Karnik, S.; Mills, D.K. Clay nanotubes as a growth factor delivery vehicle for bone tissue engineering. J. Nanomed. Nanotechn. 2013, 4, 102. [Google Scholar]
- Fakhrullina, G.I.; Akhatova, F.S.; Lvov, Y.M.; Fakhrullin, R.F. Toxicity of halloysite clay nanotubes in vivo: A Caenorhabditis elegans Study. Environ. Sci. Nano 2015, 2, 54–59. [Google Scholar] [CrossRef]
- Kryuchkova, M.; Danilushkina, A.; Lvov, Y.; Fakhrullin, R. Evaluation of toxicity of nanoclays and graphene oxide in vivo: A Paramecium caudatum study. Environ. Sci. Nano 2016, 3, 442–452. [Google Scholar] [CrossRef] [Green Version]
- Lazzara, G.; Francesca Bruno, F.; Brancatoe, D.; Sturiale, V.; Grazia D’Amico, A.; Miloto, S.; Pasbakhsh, P.; D’Agata, V.; Saccone, S.; Federico, C. Biocompatibility analysis of halloysite clay nanotubes. Mat. Lett. 2023, 336, 133852. [Google Scholar] [CrossRef]
- Yaqoob, A.A.; Ahmad, H.; Parveen, T.; Ahmade, A.; Oves, M.; Ismail, I.M.; Qari, H.A.; Umar, K.; Mohamad Ibrahim, M.N. Recent advances in metal decorated nanomaterials and their various biological applications: A review. Front. Chem. 2020, 8, 341. [Google Scholar] [CrossRef]
- Hirsch, T.; Zharnikov, M.; Shaporenko, A.; Stahl, J.; Weiss, D.; Wolfbeis, O.S.; Mirsky, V.M. Size-controlled electrochemical synthesis of metal nano particles on monomolecular templates. Angew. Chem. Int. Ed. 2005, 44, 6775–6778. [Google Scholar] [CrossRef]
- Okitsu, K.; Mizukoshi, Y.; Yamamoto, T.A.; Maeda, Y.; Nagata, Y. Sonochemical synthesis of gold nanoparticles on chitosan. Mater. Lett. 2007, 61, 3429. [Google Scholar] [CrossRef]
- Gutierrez, M.; Henglein, A. Formation of colloidal silver by “push-pull” reduction of silver(1+). J. Phys. Chem. 1993, 97, 11368. [Google Scholar] [CrossRef]
- Sato, T.; Onaka, H.; Yonezawa, Y. Sensitized photo reduction of silver ions in the presence of acetophenone. J. Photochem. Photobiol. A Chem. 1999, 127, 83–87. [Google Scholar] [CrossRef]
- Nicholson, J.C.; Weisman, J.A.; Boyer, C.J.; Wilson, C.G.; Mills, D.K. Dry sintered metal coating of halloysite nanotubes. Appl. Sci. 2016, 6, 265. [Google Scholar] [CrossRef] [Green Version]
- Hulkoti, N.I.; Taranath, T.C. Biosynthesis of nanoparticles using microbes—A review. Coll. Surf. B Biointerfac. 2014, 121, 474–483. [Google Scholar] [CrossRef]
- Shu, Z.; Zhang, Y.; Yang, Q.; Yang, H. Halloysite nanotubes supported Ag and ZnO nanoparticles with synergistically enhanced antibacterial activity. Nanoscale Res. Lett. 2017, 12, 135. [Google Scholar] [CrossRef] [Green Version]
- Abdullayev, E.; Sakakibara, K.; Okamoto, K.; Wei, W.; Ariga, K.; Lvov, Y. Natural tubule clay template synthesis of silver nanorods for antibacterial composite coating. ACS Appl. Mat. Interfac. 2011, 3, 4040–4046. [Google Scholar] [CrossRef]
- Chen, S.; Li, J.; Zhang, Y.; Zhang, D.; Zhu, J. Effect of preparation method on halloysite supported cobalt catalysts for Fischer-Tropsch synthesis. J. Nat. Gas Chem. 2012, 21, 426–430. [Google Scholar] [CrossRef]
- Liu, P.; Zhao, M. Silver nanoparticle supported on halloysite nanotubes catalyzed reduction of 4-nitrophenol (4-NP). Appl. Surf. Sci. 2009, 255, 3989–3993. [Google Scholar] [CrossRef]
- Tang, X.; Li, L.; Shen, B.; Wang, C. Halloysite-nanotubes supported FeNi alloy nanoparticles for catalytic decomposition of toxic phosphine gas into yellow phosphorous and hydrogen. Chemosphere 2013, 91, 1368–1373. [Google Scholar] [CrossRef] [PubMed]
- Geckler, K.E.; Shamsi, M.H. Gold Nanoparticle-Halloysite Nanotube and Method of Forming the Same. U.S. Patent #US20090092836A1, 24 July 2007. [Google Scholar]
- Jammalamadaka, U.; Tappa, K.; Weisman, J.A.; Nicholson, J.C.; Mills, D.K. Effect of barium-coated halloysite nanotube addition on the cytocompatibility, mechanical and contrast properties of poly(methyl methacrylate) cement. Nanotechnol. Sci. Appl. 2017, 10, 105–114. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Viel, C. Henri Moissan: L’homme, le collectionneur, l’enseignant [Henri Moissan: The man, the collector, the teacher]. Ann. Pharm. Fr. 2008, 66, 34–38. (In French) [Google Scholar] [CrossRef]
- Yin, L.; Allanore, A.; Sadoway, D.R. Towards Sustainable Metals Production by Molten Oxide Electrolysis. In Proceedings of the 222nd Electrochemical Society, Honolulu, HI, USA, 7–12 October 2012. [Google Scholar]
- Humayun, A.; Mills, D.K. Voltage regulated electrophoretic deposition of silver nanoparticles on halloysite nanotubes. Results Mater. 2020, 2020, 100112. [Google Scholar] [CrossRef]
- Humayun, A.; Luo, Y.Y.; Elumalai, A.; Mills, D.K. Differential antimicrobial and cellular response of electrolytically metalized halloysite nanotubes having different amounts of surface metallization. Mater. Adv. 2020, 1, 1705–1715. [Google Scholar] [CrossRef]
- Humayun, A.; Luo, Y.; Mills, D.K. Electrophoretic deposition of gentamicin-loaded ZnHNTs-chitosan on titanium. Coatings 2020, 10, 944. [Google Scholar] [CrossRef]
- Luo, Y.; Humayun, A.; Mills, D.K. Surface modification of 3D printed PLA/halloysite composite scaffolds with antibacterial and osteogenic capabilities. Appl. Sci. 2020, 10, 3971. [Google Scholar] [CrossRef]
- Mellouk, S.; Souhila, C.; Mohamed, S.; Kheira, M.-K.; Abdelkader, B.; Jacques, S.; Amine, K. Intercalation of Halloysite from Djebel Debagh (Algeria) and Adsorption of Copper Ions. Appl. Clay Sci. 2009, 44, 230–236. [Google Scholar] [CrossRef]
- Yang, W.; Chen, X.; Yuan, X.; Yao, J.; Cai, W.; Peng, S.; Shuai, C. In-situ grown Ag on magnetic halloysite nanotubes in scaffolds, Antibacterial, biocompatibility and mechanical properties. Ceram. Int. 2021, 47, 32756–32765. [Google Scholar] [CrossRef]
- Dong, Y.; Liu, Z.; Chen, L. Removal of Zn(II) from aqueous solution by natural halloysite nanotubes. J. Radioanal. Nucl. Chem. 2012, 292, 435–443. [Google Scholar] [CrossRef]
- Kiani, G. High removal capacity of silver ions from aqueous solution onto halloysite nanotubes. Appl. Clay Sci. 2014, 90, 159–164. [Google Scholar] [CrossRef]
- Ankit, K.; Singh, N.K.; Soni, M.; Soni, A. Chapter 3—Deposition of Thin Films by Chemical Solution-Assisted Techniques. In Chemical Solution Synthesis for Materials Design and Thin Film Device Applications Dhara; Das, S., Sandip, B.T., Eds.; Elsevier: Amsterdam, The Netherlands, 2021; pp. 79–117. [Google Scholar] [CrossRef]
- Papoulis, D. Halloysite based nanocomposites and photocatalysis: A review. Appl. Clay Sci. 2019, 168, 164–174. [Google Scholar] [CrossRef]
- De Silva, R.T.; Pasbakhsh, P.; Lee, S.M.; Kit, A.Y. ZnO deposited/encapsulated halloysite-poly (lactic acid) (PLA) nanocomposites for high performance packaging films with improved mechanical and antimicrobial properties. Appl. Clay Sci. 2015, 111, 10–20. [Google Scholar] [CrossRef]
- Wang, R.; Jiang, G.; Ding, Y.; Wang, Y.; Sun, X.; Wang, X.; Chen, W. Photocatalytic activity of heterostructures based on TiO2 and halloysite nanotubes. ACS Appl. Mat. Interfac. 2011, 3, 4154–4158. [Google Scholar] [CrossRef]
- Cohen, M.L. Epidemiology of drug resistance: Implications for a post—Antimicrobial era. Science 1992, 257, 1050–1055. [Google Scholar] [CrossRef]
- Magiorakos, A.; Srinivasan, A.; Carey, R.B.; Carmeli, Y.; Falagas, M.E.; Giske, C.G.; Harbarth, S.; Hindler, J.F.; Kahlmeter, G.; Olsson-Liljequist, B. Multidrug-resistant, extensively drug-resistant and pandrug-resistant bacteria: An international expert proposal for interim standard definitions for acquired resistance. Clin. Microbiol. Infect. 2012, 18, 268–281. [Google Scholar] [CrossRef] [Green Version]
- Ventola, C.L. The antibiotic resistance crisis: Part 1: Causes and threats. Pharm. Ther. 2015, 40, 277–283. [Google Scholar]
- Zhou, Y.; Kong, Y.; Kundu, S.; Cirillo, J.D.; Liang, H. Antibacterial activities of gold and silver nanoparticles against Escherichia coli and Bacillus Calmette-Guérin. J. Nanobiotechnology 2012, 10, 19. [Google Scholar] [CrossRef] [Green Version]
- Hernández-Sierra, J.F.; Ruiz, F.; Pena, D.C.C.; Martínez-Gutiérrez, F.; Martínez, A.E.; de Guillén, A.J.P.; Tapia-Pérez, H.; Castañón, G.M. The antimicrobial sensitivity of Streptococcus mutans to nanoparticles of silver, zinc oxide, and gold. Nanomed. Nanotechnol. Biol. Med. 2008, 4, 237–240. [Google Scholar] [CrossRef]
- Fayaz, A.M.; Balaji, K.; Girilal, M.; Yadav, R.; Kalaichelvan, P.T.; Venketesan, R. Biogenic synthesis of silver nanoparticles and their synergistic effect with antibiotics: A study against gram-positive and gram-negative bacteria. Nanomed. Nanotechnol. Biol. Med. 2010, 6, 103–109. [Google Scholar] [CrossRef] [PubMed]
- Silver, S. Bacterial silver resistance: Molecular biology and uses and misuses of silver compounds. FEMS Microbiol. Rev. 2003, 27, 341–353. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mobaraki, M.; Karnik Li, Y.; Mills, D.K. Therapeutic applications of halloysite. Appl. Sci. 2022, 12, 87. [Google Scholar] [CrossRef]
- Devasconcellos, P.; Bose, S.; Beyenal, H.; Bandyopadhyay, A.; Zirkle, L. Antimicrobial particulate silver coatings on stainless steel implants for fracture management. Mater. Sci. Eng. C. 2012, 32, 1112–1120. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jaiswal, S.; McHale, P.; Duffy, B. Preparation and rapid analysis of antibacterial silver, copper, and zinc doped sol-gel surfaces. Coll. Surf. B Biointerfac. 2012, 94, 170–176. [Google Scholar] [CrossRef] [Green Version]
- Swaminathan, M.; Sharma, N.K. Antimicrobial activity of the engineered nanoparticles used as coating agents. Handb. Ecomater. 2019, 1, 549–563. [Google Scholar] [CrossRef]
- Kermavnar, T.; Shannon, A.; O’Sullivan, K.J.; McCarthy, C.; Dunne, C.P.; O’Sullivan, L.W. 3D printing and additive manufacturing. 3D Print. Addit. Manuf. 2021, 8, 366–408. [Google Scholar] [CrossRef]
- Tappa, K.; Jammalamadaka, U.; Weisman, J.A.; Ballard, D.B.; Wolford, D.D.; Alexander, J.S.; Mills, D.K.; Woodard, P.K. 3D printing custom bioactive and absorbable surgical screws, pins, and bone plates for localized drug delivery. J. Func. Biomater. 2019, 10, 17–24. [Google Scholar] [CrossRef] [Green Version]
- Humayun, A.; Luo, Y.; Elumalai, A.; Mills, D.K. 3D printed antimicrobial PLA constructs functionalized with zinc- coated halloysite nanotubes-Ag-chitosan oligosaccharide lactate. Mat. Tech. 2022, 37, 28–35. [Google Scholar]
- Boraei, S.; Nourmohammadi, J.; Mahdavi, F.S.; Zare, Y.; Rhee, K.Y.; Montero, A.F.; Herencia, A.J.; Ferrari, B. Osteogenesis capability of three-dimensionally printed poly(lactic acid)-halloysite nanotube scaffolds containing strontium ranelate. Nanotech. Rev. 2022, 11, 1901–1910. [Google Scholar] [CrossRef]
- Luo, Y.; Humayun, A.; Murray, T.A.; Kemp, B.S.; McFarland, A.; Liu, X.; Mills, D.K. Cellular analysis and chemotherapeutic potential of a bi-functionalized halloysite nanotube. Pharmaceutics 2020, 12, 962. [Google Scholar] [CrossRef]
- Boro, U.; Moholkar, V.S. Antimicrobial bionanocomposites of poly(lactic acid)/ZnO deposited halloysite nanotubes for potential food packaging applications. Mat. Today Commun. 2022, 33, 104787. [Google Scholar] [CrossRef]
- Fizir, M.; Dramou, P.; Dahiru, N.S.; Ruya, W.; Huang, T.; He, H. Halloysite nanotubes in analytical sciences and in drug delivery: A review. Mikrochim. Acta 2018, 25, 389. [Google Scholar] [CrossRef]
- Shutava, T.G.; Fakhrullin, R.F.; Lvov, Y.M. Spherical and tubule nanocarriers for sustained drug release. Curr. Opin. Pharmacol. 2014, 18, 141–148. [Google Scholar] [CrossRef] [Green Version]
- Rouster, P.; Dondelinger, M.; Galleni, M.; Nysten, B.; Jonas, A.M.; Glinel, K. Layer-by-layer assembly of enzyme-loaded halloysite nanotubes for the fabrication of highly active coatings. Coll. Surf. B Biointerfac. 2019, 178, 508–514. [Google Scholar] [CrossRef]
- Diekema, D.J.; Pfaller, M.A.; Schmitz, F.J.; Smayevsky, J.; Bell, J.; Jones, R.N.; Beach, M.; SENTRY Participants Group. Survey of infections due to Staphylococcus species: Frequency of occurrence and antimicrobial susceptibility of isolates collected in the United States, Canada, Latin America, Europe, and the Western Pacific region for the SENTRY Antimicrobial Surveillance. Clin. Infect. Dis. 2001, 32 (Suppl. 2), S114–S132. [Google Scholar] [CrossRef] [Green Version]
- McFarland, A.W., Jr.; Elumalai, A.; Miller, C.C.; Humayun, A.; Mills, D.K. Effectiveness and applications of a metal-coated HNT/polylactic acid antimicrobial filtration system. Polymers 2022, 14, 1603. [Google Scholar] [CrossRef]
- Jana, S.; Kondakova, A.V.; Shevchenko, S.N.; Sheval, E.V.; Gonchar, K.A.; Timoshenko, V.Y.; Vasiliev, A.N. Halloysite nanotubes with immobilized silver nanoparticles for anti-bacterial application. Coll. Surf. B Biointerfac. 2017, 151, 249–254. [Google Scholar] [CrossRef]
- Saedi, S.; Shokri, M.; Roy, S.; Rhim, J. Silver loaded aminosilane modified halloysite for the preparation of carrageenan-based functional films. Appl. Clay Sci. 2021, 211, 106170. [Google Scholar] [CrossRef]
- Barot, T.; Rawtani, D.; Kulkarni, P. Physicochemical and biological assessment of silver nanoparticles immobilized halloysite nanotubes-based resin composite for dental applications. Heliyon 2020, 6, e03601. [Google Scholar] [CrossRef]
- Paul, A.; Augustine, R.; Hasan, A.; Zahid, A.A.; Thomas, S.; Agatemor, C.; Ghosal, K. Halloysite nanotube and chitosan polymer composites: Physicochemical and drug delivery properties. J. Drug Del. Sci. Tech. 2022, 72, 103380. [Google Scholar] [CrossRef]
- Azam, A.; Ahmed, A.S.; Oves, M.; Khan, M.S.; Habib, S.S.; Memic, A. Antimicrobial activity of metal oxide nanoparticles against Gram-positive and Gram-negative bacteria: A comparative study. Int. J. Nanomed. 2012, 7, 6003. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hasan, A.; Morshed, M.; Memic, A.; Hassan, S.; Webster, T.J.; Marei, H.E. Nanoparticles in tissue engineering: Applications, challenges and prospects. Int. J. Nanomed. 2018, 13, 5637–5655. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Deo, K.A.; Lokhande, L.; Akhilesh, K.; Gaharwar, A.K. Nanostructured hydrogels for tissue engineering and regenerative medicine. In Encyclopedia of Tissue Engineering and Regenerative Medicin; Elsevier: Amsterdam, The Netherlands, 2019; pp. 21–32. [Google Scholar]
- Luo, J.; Zhang, Y.; Zhu, S.; Tong, Y.; Ji, L.; Zhang, W.; Zhang, Q.; Bi, Q. The application prospect of metal/metal oxide nanoparticles in the treatment of osteoarthritis. Naunyn-Schmiedeberg’s Arch. Pharmacol. 2021, 39, 1991–2002. [Google Scholar] [CrossRef] [PubMed]
- Liu, M.; Shenb, Y.; Aoa, P.; Daib, L.; Liub, Z.; Changren Zhou, C. The improvement of hemostatic and wound healing property of chitosan by halloysite nanotubes. Mat. Adv. 2014, 4, 23540–23553. [Google Scholar] [CrossRef]
- Roux, S.; Faure, A.-C.; Mandon, C.; Dufort, S.; Rivière, C.; Bridot, J.-L.; Mutelet, B.; Marquette, C.A.; Josserand, V.; Le Duc, G.; et al. Multifunctional gadolinium oxide nanoparticles: Towards image-guided therapy. Imaging Med. 2010, 2, 211–223. [Google Scholar] [CrossRef]
- Kusiak, E.; Zaborski, M. Characteristic of natural rubber composites absorbing X-radiation. Compos. Interface 2012, 9, 433–439. [Google Scholar] [CrossRef]
- Burns, K.M.; Shoag, J.M.; Kahlon, S.S.; Parsons, P.J.; Bijur, P.E.; Taragin, B.H.; Markowitz, M. Lead Aprons Are a Lead Exposure Hazard. J. Am. Coll. Radiol. 2017, 14, 641–647. [Google Scholar] [CrossRef]
- Spillantini, P.; Casolino, M.; Durante, M.; Mueller-Mellin, R.; Reitz, G.; Rossi, L.; Shrurshakov, V.; Sorbi, M. Shielding from cosmic radiation for interplanetary mission: Active and passive methods. Radiat. Meas. 2007, 42, 14–23. [Google Scholar] [CrossRef]
- Moreno-Villanueva, M.; Wong, M.; Lu, T.; Zhang, Y.; Wu, H. Interplay of space radiation and microgravity in DNA damage and DNA damage response. Npj Microgravity 2017, 3, 14. [Google Scholar] [CrossRef] [Green Version]
- Alakija, F.; Elumalai, A.; Mills, D.K. Radiation Shielding Using Cerametals–Ceramic/Metal Nanoparticle Composites. In Proceedings of the Lunar Surface Science Meeting, Houston, TX, USA, 10 June 2020; Available online: https://lunarscience.arc.nasa.gov/lssw/falsrps/ (accessed on 12 January 2022).
- Liu, H.; Gao, J.; Huang, W.; Dai, K.; Zheng, G.; Liu, C.; Shen, C.; Yan, X.; Guo, J.; Guo, Z. Electrically conductive strain sensing polyurethane nanocomposites with synergistic carbon nanotubes and graphene bifillers. Nanoscale 2016, 8, 12977–12989. [Google Scholar] [CrossRef]
- Chen, Y.; Li, Q.; Li, C.; Dai, Z.; Yan, H.; Zhu, M.; Zhang, Y.; Yao, Y.; Li, Q. Regulation of multidimensional silver nanostructures for high-performance composite conductive adhesives. Compos. Part A Appl. Sci. Manuf. 2020, 37, 106025. [Google Scholar] [CrossRef]
- Catenacci, M.J.; Reyes, C.; Cruz, M.A.; Wiley, B.J. Stretchable conductive composites from Cu–Ag nanowire felt. ACS Nano 2018, 12, 3689–3698. [Google Scholar] [CrossRef]
- Sannicolo, T.; Lagrange, M.; Cabos, A.; Celle, C.; Simonato, J.-P.; Bellet, D. Metallic nanowire-based transparent electrodes for next generation flexible devices: A review. Wiley Small 2016, 12, 6052–6075. [Google Scholar] [CrossRef]
- Hong, I.; Lee, S.; Kim, D.; Cho, H.; Roh, Y.; An, H.; Hong, S.; Ko, S.H.; Han, S. Study on the oxidation of copper nanowire network electrodes for skin mountable flexible, stretchable and wearable electronics applications. Nanotechnology 2019, 30, 74001. [Google Scholar] [CrossRef]
- Cao, G.; Cai, S.; Zhang, H.; Chen, Y.; Tian, Y. High-performance conductive polymer composites by incorporation of polyaniline-wrapped halloysite nanotubes and silver microflakes. ACS Appl. Poly Mat. 2022, 4, 3352–3360. [Google Scholar] [CrossRef]
- Shao, L.; Wang, X.; Yang, B.; Wang, Q.; Tian, Q.; Ji, Z.; Zhang, J. A highly sensitive ascorbic acid sensor based on hierarchical polyaniline coated halloysite nanotubes prepared by electrophoretic deposition. Electrochim. Acta 2017, 255, 286–297. Available online: https://www.sciencedirect.com/science/article/pii/S0013468617320492 (accessed on 20 February 2023). [CrossRef]
- Karolina Pierchala, M.; Kadumudi, F.B.; Mehrali, M.; Zsurzsan, T.G.; Kempen, P.J.; Serdeczny, M.P.; Spangenberg, J.; Andresen, T.L.; Dolatshahi-Pirouz, A. Soft electronic materials with combinatorial properties generated via mussel-inspired chemistry and halloysite nanotube reinforcement. ACS Nano 2021, 15, 9531–9549. [Google Scholar] [CrossRef]
- Sudario, F.; Clemente, R.; Millare, J.; Jojo, A. Characterization and application of halloysite nanotubes in semiconductor. In Proceedings of the 28th Philippine Chemistry Congress, Dumaguete City, Philippines, 10–12 April 2013. [Google Scholar]
- Yuan, P.; Tan, D.; Annabi-Bergaya, F. Properties and applications of halloysite nanotubes: Recent research advances and future prospects. App Clay Sci. 2015, 112–113, 75–93. [Google Scholar] [CrossRef]
- Cavallaro, G.; Lazzara, G.; Rozhina, E.; Konnova, S.; Kryuchkova, M.; Khaertdinov, N.; Fakhrullin, R. Organic-nanoclay composite materials as removal agents for environmental decontamination. RSC Adv. 2019, 9, 40553–40564. [Google Scholar] [CrossRef] [Green Version]
- Yu, L.; Wang, H.; Zhang, Y.; Zhang, B.; Liu, J. Recent advances in halloysite nanotube derived composites for water treatment. Environ. Sci. Nano 2016, 3, 28–44. [Google Scholar] [CrossRef]
- Afzali, D.; Fayazi, M. Deposition of MnO2 nanoparticles on the magnetic halloysite nanotubes by hydrothermal method for lead(II) removal from aqueous solutions. J. Taiwan Inst. Chem. Eng. 2016, 63, 421–429. [Google Scholar] [CrossRef]
- Li, L.; Wang, F.; Lv, Y.; Liu, J.; Zhang, D.; Shao, Z. Halloysite nanotubes and Fe3O4 nanoparticles enhanced adsorption removal of heavy metal using electrospun membranes. Appl. Clay Sci. 2018, 161, 225–234. [Google Scholar] [CrossRef]
- Wang, X.S.; Zhou, Y.; Jiang, Y.; Sun, C. The removal of basic dyes from aqueous solutions using agricultural by-products. J. Hazard. Mat. 2008, 157, 374–385. [Google Scholar] [CrossRef] [PubMed]
- Xie, Y.; Qian, D.; Wu, D.; Ma, X. Magnetic halloysite nanotubes/iron oxide composites for the adsorption of dyes. Chem. Eng. J. 2011, 168, 959–963. [Google Scholar] [CrossRef]
- Jiang, L.; Zhang, C.; Wei, J.; Tjiu, W.; Pan, J.; Chen, Y.; Liu, T. Surface modifications of halloysite nanotubes with superparamagnetic Fe3O4 nanoparticles and carbonaceous layers for efficient adsorption of dyes in water treatment. Chem. Res. Chin. Univ. 2014, 30, 971–977. [Google Scholar] [CrossRef]
- Li, X.; Yao, C.; Lu, X.; Hu, Z.; Yin, Y.; Ni, C. Halloysite–CeO2–AgBr nanocomposite for solar light photodegradation of methyl orange. Appl. Clay Sci. 2015, 104, 74–78. [Google Scholar] [CrossRef]
- Zou, M.; Du, M.; Zhu, H.; Xu, C.; Fu, Y. Green synthesis of halloysite nanotubes supported Ag nanoparticles for photocatalytic decomposition of methylene blue. J. Phys. D Appl. Phy. 2012, 45, 325302. [Google Scholar] [CrossRef]
- Xianchu, L.; Xiuyun, C.; Aiping, W.; Feiyu, K. Microstructure and photocatalytic decomposition of methylene blue by TiO2-mounted halloysite, a natural tubular mineral. Acta Geol. Sin. Engl. Ed. 2006, 80, 278–284. [Google Scholar] [CrossRef]
- Peng, L.; Lei, L.; Liu, Y.; Du, L. Mechanical and sound absorption performance of addition type liquid silicone rubber reinforced with halloysite nanotubes. Mater. Res. Express 2021, 8, 015309. [Google Scholar] [CrossRef]
- Barrientos-Ramírez, S.; de Oca-Ramírez, G.M.; Ramos-Fernández, E.V.; Sepúlveda-Escribano, A.; Pastor-Blas, M.M.; González-Montiel, A. Surface modification of natural halloysite clay nanotubes with aminosilanes. Application as catalyst supports in the atom transfer radical polymerization of methyl methacrylate. App. Catal. A Gen. 2011, 406, 2–33. [Google Scholar] [CrossRef]
- Guo, X.; Ye, W.; Sun, H.; Zhang, Q.; Yang, J. A dealloying process of core–shell Au@AuAg nanorods for porous nanorods with enhanced catalytic activity. Nanoscale 2013, 5, 12582–12588. [Google Scholar] [CrossRef] [PubMed]
- Fu, X.; Ding, Z.; Zhang, X.; Weng, W.; Xu, Y.; Liao, J.; Xie, Z. Preparation of halloysite nanotube-supported gold nanocomposite for solvent-free oxidation of benzyl alcohol. Nano Res. Lett. 2014, 9, 1–4. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cheng, Z.L.; Sun, W. Preparation of nano-CuO-loaded halloysite nanotubes with high catalytic activity for selective oxidation of cyclohexene. Chin. Chem. Lett. 2016, 27, 81–84. [Google Scholar] [CrossRef]
- Carrillo, A.M.; Carriazo, J.G. Cu and Co oxides supported on halloysite for the total oxidation of toluene. Appl. Catal. B Environ. 2015, 164, 443–452. [Google Scholar] [CrossRef]
- Long, Y.; Yuan, B.; Ma, J. Epoxidation of alkenes efficiently catalyzed by Mo salen supported on surface-modified halloysite nanotubes. Chin. J. Catal. 2015, 36, 348–354. [Google Scholar] [CrossRef]
- Du, Y.; Zheng, P. Adsorption and photodegradation of methylene blue on TiO2-halloysite adsorbents. Korean J. Chem. Eng. 2014, 31, 2051–2056. [Google Scholar] [CrossRef]
- Ukkund, S.J.; Puthiyillam, P.; Anqi, A.E.; Taqui, S.N.; Ali, M.A.; Syed, U.T.; Alghamdi, M.N.; Siddiqui, M.I.; Alshehri, H.M.; Safaei, M.R.; et al. A recent study on remediation of Direct Blue 15 dye using halloysite nanotubes. Appl. Sci. 2021, 11, 8196. [Google Scholar] [CrossRef]
- Massaro, M.; Colletti, C.G.; Lazzara, G.; Milioto, S.; Noto, R.; Riela, S. Halloysite nanotubes as support for metal-based catalysts. J. Mater. Chem. A 2017, 5, 13276–13293. [Google Scholar] [CrossRef]
- Singh, K.; Nayak, V.; Singh, J.; Singh, A.; Singh, R. Potentialities of bioinspired metal and metal oxide nanoparticles in biomedical sciences. RSC Adv. 2021, 11, 24722–24746. [Google Scholar] [CrossRef]
- Nikolova, M.P.; Chavali, M.S. Metal oxide nanoparticles as biomedical materials. Biomimetics 2020, 5, 27. [Google Scholar] [CrossRef]
- Available online: https://www.marketwatch.com/press-release/metal-metal-oxide-nanoparticles-market-2022-industry-size-revenue-growth-development-business-opportunities-future-trends-top-key-players-covid-19-impact-global-analysis-forecast-till-2028-2022-08-02 (accessed on 20 February 2023).
- Available online: https://www.grandviewresearch.com/industry-analysis/nanotechnology-and-nanomaterials-market (accessed on 20 February 2023).
- Gkouma, E.; Gianni, E.; Avgoustakis, K.; Papoulis, D. Applications of halloysite in tissue engineering. Appl. Clay Sci. 2021, 214, 106291. [Google Scholar] [CrossRef]
- Karewicz, A.; Machowska, A.; Kasprzyk, M.; Ledwójcik, G. Application of halloysite nanotubes in cancer therapy—A review. Materials 2021, 14, 2943. [Google Scholar] [CrossRef]
- Biddeci, G.; Spinelli, G.; Colomba, P.; Di Blasi, F. Nanomaterials: A review about halloysite nanotubes, properties, and application in the biological field. Int. J. Mol. Sci. 2022, 23, 11518. [Google Scholar] [CrossRef]
- Coetzee, D.; Venkataraman, M.; Militky, J.; Petru, M. Influence of nanoparticles on thermal and electrical conductivity of composites. Polymers 2020, 12, 742. [Google Scholar] [CrossRef] [Green Version]
- Góral-Kowalczyk, M. Application of metal nanoparticles for production of self-sterilizing coatings. Coatings 2022, 12, 480. [Google Scholar] [CrossRef]
- Kumar, A.; Nath, K.; Parekh, Y.; Enayathullah, M.G.; Bokara, K.K.; Sinhamahapatra, A. Antimicrobial silver nanoparticle-photodeposited fabrics for SARS-CoV-2 destruction. Colloid. Interface Sci. Commun. 2021, 45, 100542. [Google Scholar] [CrossRef]
- Franco, D.; Calabrese, G.; Guglielmino, S.P.P.; Conoci, S. Metal-based nanoparticles: Antibacterial mechanisms and biomedical application. Microorganisms 2022, 10, 1778. [Google Scholar] [CrossRef]
- Nanotextiles Market Size, Share, Growth, and Industry Analysis by Type. Available online: https://www.businessresearchinsights.com/market-reports/nanotextiles-market-101664 (accessed on 20 February 2023).
- Parvin, F.; Islam, S.; Urmy ZAhmed, S.; Saiful Islam, A.K.M. A Study on the Solutions of Environment Pollutions and Worker’s Health Problems Caused by Textile Manufacturing Operations. Biomed. J. Sci. Tech. Res. 2020, 28, 21831–21844. [Google Scholar]








Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Masoud, A.-R.; Alakija, F.; Perves Bappy, M.J.; Mills, P.A.S.; Mills, D.K. Metallizing the Surface of Halloysite Nanotubes—A Review. Coatings 2023, 13, 542. https://doi.org/10.3390/coatings13030542
Masoud A-R, Alakija F, Perves Bappy MJ, Mills PAS, Mills DK. Metallizing the Surface of Halloysite Nanotubes—A Review. Coatings. 2023; 13(3):542. https://doi.org/10.3390/coatings13030542
Chicago/Turabian StyleMasoud, Abdul-Razak, Femi Alakija, Mohammad Jabed Perves Bappy, Patrick A. S. Mills, and David K. Mills. 2023. "Metallizing the Surface of Halloysite Nanotubes—A Review" Coatings 13, no. 3: 542. https://doi.org/10.3390/coatings13030542
APA StyleMasoud, A.-R., Alakija, F., Perves Bappy, M. J., Mills, P. A. S., & Mills, D. K. (2023). Metallizing the Surface of Halloysite Nanotubes—A Review. Coatings, 13(3), 542. https://doi.org/10.3390/coatings13030542







