1
I3S, Instituto de Investigação e Inovação em Saúde, 4200-135 Porto, Portugal
2
Ipatimup, Instituto de Patologia e Imunologia Molecular da Universidade do Porto, 4200-465 Porto, Portugal
3
INEB, Instituto de Engenharia Biomédica—Rua Alfredo Allen, 4200-135 Porto, Portugal
4
Department of Pathology and Oncology, Faculty of Medicine, University of Porto, 4200-319 Porto, Portugal
5
British Columbia Cancer Agency, Vancouver, BC V5Z4E6, Canada
†
These authors contributed equally to this work.
Biology 2022, 11(7), 1046; https://doi.org/10.3390/biology11071046 - 12 Jul 2022
Cited by 3 | Viewed by 2072
Abstract
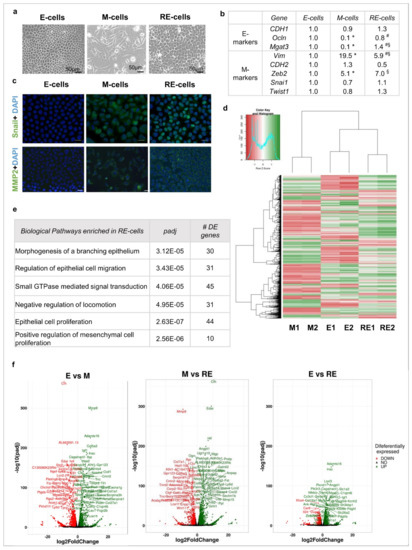
Transitions between epithelial and mesenchymal cellular states (EMT/MET) contribute to cancer progression. We hypothesize that EMT followed by MET promotes cell population heterogeneity, favouring tumour growth. We developed an EMT model by on and off exposure of epithelial EpH4 cells (E-cells) to TGFβ1
[...] Read more.
Transitions between epithelial and mesenchymal cellular states (EMT/MET) contribute to cancer progression. We hypothesize that EMT followed by MET promotes cell population heterogeneity, favouring tumour growth. We developed an EMT model by on and off exposure of epithelial EpH4 cells (E-cells) to TGFβ1 that mimics phenotypic EMT (M-cells) and MET. We aimed at understanding whether phenotypic MET is accompanied by molecular and functional reversion back to epithelia by using RNA sequencing, immunofluorescence (IF), proliferation, wound healing, focus formation and mamosphere formation assays as well as cell xenografts in nude mice. Phenotypic reverted epithelial cells (RE-cells) obtained after MET induction presented epithelial morphologies and proliferation rates resembling E cells. However, the RE transcriptomic profile and IF staining of epithelial and mesenchymal markers revealed a uniquely heterogeneous mixture of cell subpopulations with a high self-renewal ability. RE cell heterogeneity was stably maintained for long periods after TGFβ1 removal both in vitro and in large tumours derived from the nude mice. Overall, we show that phenotypic reverted epithelial cells (RE cells) do not return to the molecular and functional epithelial state and present mesenchymal features related to aggressiveness and cellular heterogeneity that favour tumour growth in vivo. This work strengthens epithelial cell reprogramming and cellular heterogeneity fostered by inflammatory cues as a tumour growth-promoting factor in vivo.
Full article
(This article belongs to the Special Issue Cytokines and Chemokines: Modulators of Epithelial Cell Biology in Health and Disease)
▼
Show Figures