1. Introduction
The lymphatic vascular system returns the interstitial fluid to the systemic circulation in the human body, which plays an essential role in homeostasis and immunization in the human body [
1,
2,
3,
4]. Changes or defects in the lymphatic vascular function can induce variable human body responses to a wide range of human diseases, such as obesity and Alzheimer’s disease [
4]. The lymphatic pathway is a common approach for the delivery of large therapeutic proteins, such as monoclonal antibodies, through subcutaneous (SC) injection [
2,
3,
5,
6]. SC injection has many merits, such as lower cost and convenience compared to intravascular (IV) injection [
7,
8,
9]. In a recent administration analysis, subcutaneous injection takes up
of all the US FDA-approved administration of therapeutic agents of high concentration [
10]. After being injected into the subcutaneous tissue, drug solutes transport into the initial lymphatics, deeper collecting lymphatics and then lymph nodes, and at last, into the systemic circulation [
2]. During the transport process, many internal and external factors affect drug absorption after subcutaneous injection, such as the physiological properties of the subcutaneous tissues, injection site, drug and formulation properties, and administration mode [
6]. The quantitative investigation on the subcutaneous absorption through physics-based computational modeling bridges the gap between the understanding of the absorption process through SC injection and the development of drug and drug delivery system.
In our previous works [
11,
12,
13], the convective transport of drug solutes is driven by the interstitial fluid pressure (IFP) into the lymphatic system, which plays an important role in drug absorption through SC injection. The mechanical process of the injection and its effect on pressure build-up and relaxation are essential to understand the fluid flow and drug clearance at the injection site [
12,
14]. The diffusion across the discrete heterogeneous lymphatic vessel network significantly affects the lymphatic uptake [
13]. However, the computation cost is expensive for a multi-scale problem with both a heterogeneous explicit vessel network and the constitutive equations included, where the vessels of radius
O (10
m) are embedded in a domain of size
O (1 cm). A time-efficient and accurate biomechanical model for pressure evolution can be valuable to such a complex computational system to study the transport and absorption of large drug molecules in biological tissues.
The pressure build-up in subcutaneous tissue during the subcutaneous injection has been investigated through experiments in several previous studies [
15,
16]. The counter pressure in the subcutaneous tissue is estimated based on experimental data, and a model for the pressure evolution is constructed [
15]. The experiments of subcutaneous injection show that controlling the injection rate could be used to partially prevent back pressure from increasing to unacceptable ranges, while changing the injection volume alone shows little influence on pressure evolution [
17]. The X-ray imaging technique is used to study the effects of injection conditions on the permeation of the drug in tissues [
18] in horizontal and vertical directions for different injection speeds. Due to the lack of in situ quantitative experiments on IFP and lymphatic uptake at the injection site, numerical simulations are used to improve our understanding of the effects of the tissue mechanical properties on drug delivery. The effect of the biomechanical responses of the soft tissue on the fluid flow is investigated by coupling Darcy’s law with stress–strain behaviors for the solid skeleton, i.e., the constitutive models. The poroelastic model is widely used to describe the deformation of biological tissues and hydrogels [
12,
14,
19,
20]. Our previous studies numerically investigate the contribution of large interstitial pressure to drug absorption during and after the injection [
11,
12]. A hybrid discrete-continuum vessel network model is developed to describe the diffusion and convection of drug solutes across the vessel wall [
13]. Although the poroelastic model can simulate the deformation of the soft subcutaneous tissue, the computation is expensive after coupling with transport equations and instability issues may occur [
12]. Including a three-dimensional (3D) heterogeneous vessel network in the simulations can make the computations more expensive. A few methods are used to simplify the computations in the literature. One approach is reducing the dimension of the numerical simulation. The mathematical equations for the interstitial fluid flow and soft tissue strain are solved in two-dimensional models using a finite element method to study the effect of strain relaxation on fluid drainage and solute transport [
21,
22], while one-dimensional mathematical models are also used to analyze the fluid flow and solute transport [
23]. Another approach is simplifying the stress–strain relation to make the computations more efficient. The linear poroelasticity based on small strain theory is often used to simplify the constitutive Equation [
24]. In [
25], the deformation and strain are obtained by simplifying the poroelastic constitutive equation to simulate the deformation of a hydrogel. In this paper, we develop a new approximate poroelastic model to mimic the relaxation of the pressure in soft tissues.
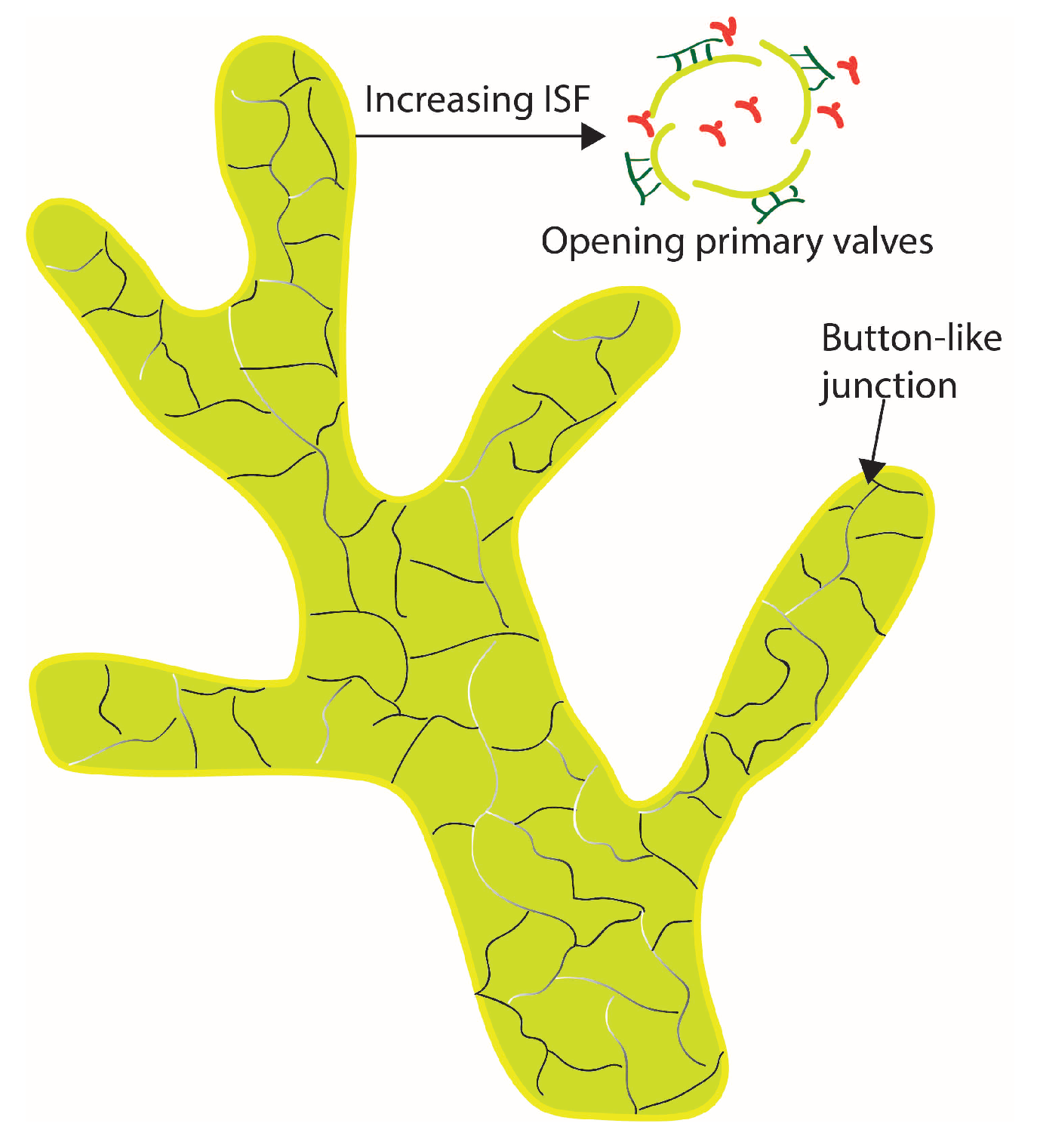
As one of the two vascular circulatory systems in the human body, the lymphatic vessel network consists of initial lymphatics with only one single layer of lymphatic endothelial cells (LECs), unlike blood vessels which have a complete basement membrane [
2,
3,
4]. For lymphatic vessels, LECs are loosely connected and have a primary valve structure to ensure the one-directional fluid flows from the interstitial space into the lymphatics. In our previous work [
13], three different transport conditions are used to investigate solute transport across the lymphatic vessel membrane, including the Kedem-Katchalsky model. In [
26,
27], an integral form of the modified Kedem–Katchalsky formulation is used to study dermal clearance through the blood and lymphatic vessels. To further address the special physiological structure of the lymphatic vessel membrane, in this paper, we develop an improved Kedem–Katchalsky model for solute transport across the explicit lymphatic vessel wall. The morphology of the initial lymphatic vessel network varies over the location and species [
3]. In human skin tissue, blind-ended and interconnected structures (reticular plexus) are observed for initial lymphatic vessels [
28,
29,
30,
31]. However, there is no quantitative investigation on the effect of various structures of the lymphatic vessel network on solute transport and absorption through the lymphatic vessel network.
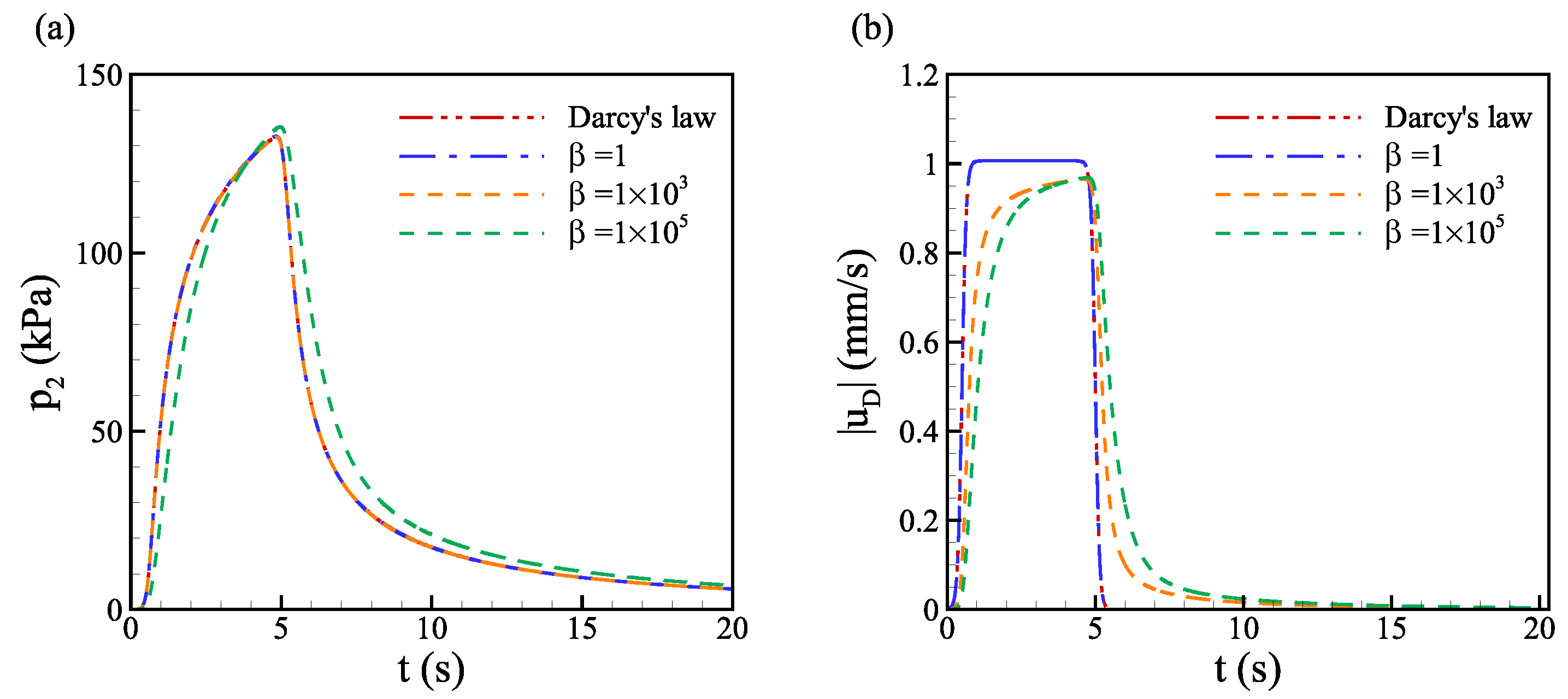
In this paper, an approximate continuum poroelastic model is developed to simulate the pressure evolution in the soft subcutaneous tissue. The pressure evolution of the proposed model is quantitatively validated against the analytical solution and the numerical solution of the poroelasticity model. The effect of elasticity, variable porosity and permeability on pressure build-up is numerically investigated. The convective and diffusive transport of drug solute into the lymphatic vessels is investigated through an improved Kedem–Katchalsky model by implementing the hybrid vessel network in the continuum poroelastic model. At last, heterogeneous vessel networks with various structures are used to investigate the effect of the morphology of the lymphatics on solute transport into the lymphatic system in the multi-layer soft skin tissue.
5. Conclusions
Interstitial fluid pressure plays a crucial role in governing the convective transport and absorption of drug molecules in the tissue. An approximate continuum poroelasticity model is developed to mimic the pressure build-up and relaxation by introducing the time variation of pressure. The effects of elasticity, porosity, and permeability on pressure evolution are investigated after validating the proposed poroelastic model with the analytical solution and previous computational model. The advantage of this model lies in the simplicity of implementation while maintaining good accuracy in capturing the evolution of the interstitial pressure. The small elasticity of the tissue slows down the relaxation of pressure and lowers the magnitude of pressure, while the magnitude of pressure increases dramatically with decreasing hydraulic permeability. Pressure evolution for low hydraulic permeability tends to be more sensitive to small elasticity, i.e., the relaxation time is longer for low hydraulic permeability at the same elasticity. The effect of variable porosity-dependent permeability is found to alleviate the high pressure due to increased porosity and permeability near the injection site.
To better address the microscopic structure of the lymphatic vessel membrane, an improved Kedem–Katchalsky model is developed for solute transport across the explicit vessel wall. The transport and lymphatic uptake of drug solutes in a multi-layered medium are investigated to elucidate the effect of heterogeneity of multi-layered structure and the heterogeneous vessel network. We investigate the convective diffusion of various macromolecules through the hybrid lymphatic vessel network by varying the Stokes radius and solute diffusivity across the opening lymphatic valves. With a smaller Stokes radius and larger solute diffusivity, the lymphatic uptake is significantly fast. Finally, the effect of the lymphatic vessel network structure is investigated through the fractal trees and Voronoi structures. We find that the Voronoi structure in the subcutaneous layer is more efficient in drug absorption because there are more interactions between the drug molecules and the vessel surfaces in the dermis and hypodermis layers.
For future work, the computational model can be further coupled with the mathematical model for the lymph flow inside the lymphatic vessels. In this way, the effect of the lymph flow rate can be incorporated into the model. The computational models developed here can also be coupled with the pharmacokinetic (PK) models to better predict drug bioavailability.