Protector Role of Cx30.2 in Pancreatic β-Cell against Glucotoxicity-Induced Apoptosis
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Cell Line and Culture Conditions
2.2. Immunofluorescence Assays
2.3. Western Blot
2.4. TUNEL Assay
2.5. Flow Cytometry Assay of Apoptosis
2.6. Cell Viability
2.7. siRNAs Transfection
2.8. Scrape Loading/Dye Transfer Assay (SLDT)
2.9. Dye Transfer Studies
2.10. Statistical Analysis
3. Results
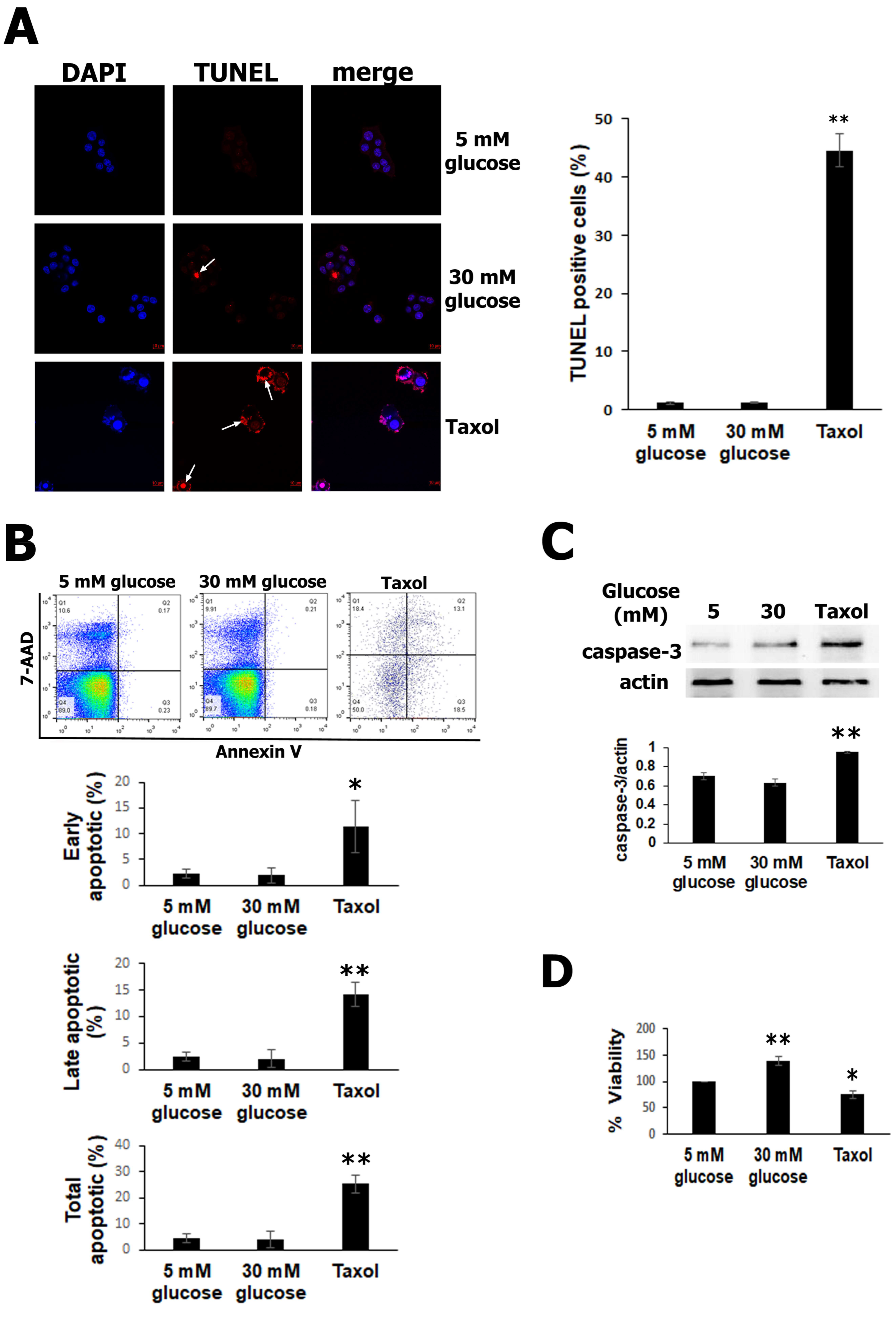
3.1. High Glucose Exposure Did Not Promote RIN-m5F Cells Apoptosis
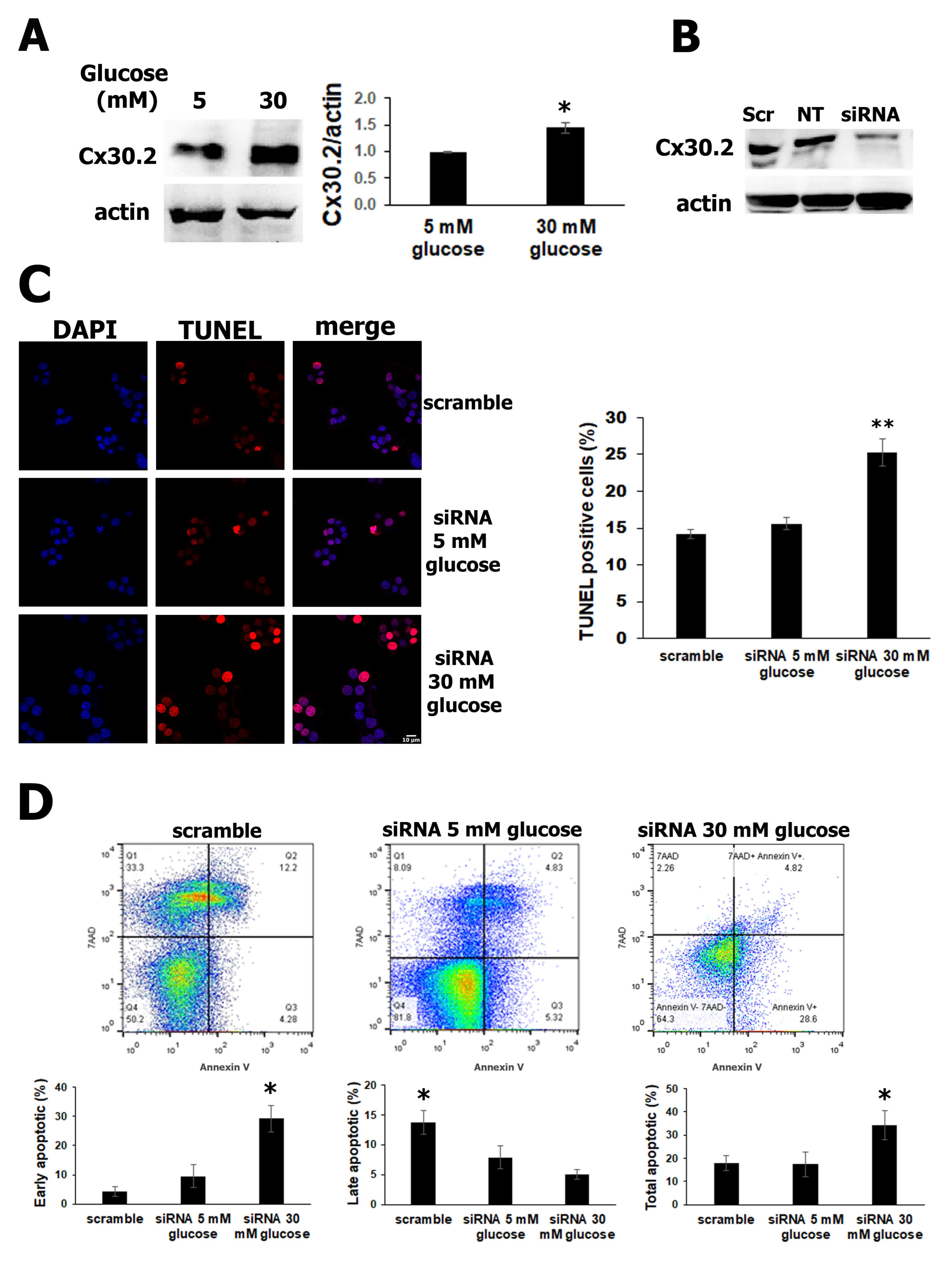
3.2. Silencing of Cx30.2 Increases Cell Apoptosis in RIN-m5F Cells
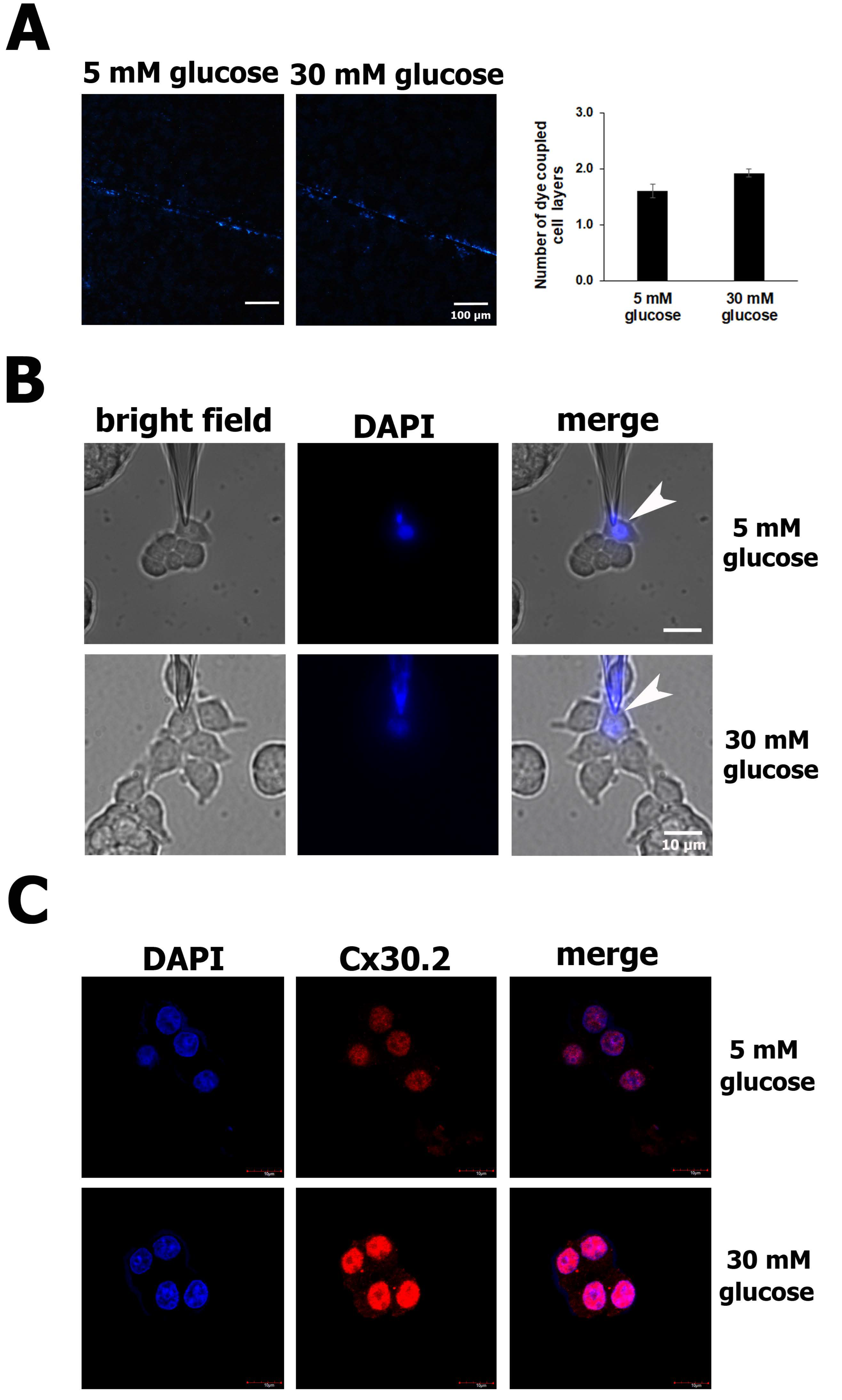
3.3. Increased Cx30.2 Protein Expression from High Glucose Levels Does Not Change GJIC Activity
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Rorsman, P.; Ashcroft, F.M. Pancreatic beta-Cell Electrical Activity and Insulin Secretion: Of Mice and Men. Physiol. Rev. 2018, 98, 117–214. [Google Scholar] [CrossRef] [PubMed]
- WHO. Diabetes. Available online: https://www.who.int/health-topics/diabetes (accessed on 6 October 2023).
- Broca, C.; Varin, E.; Armanet, M.; Tourrel-Cuzin, C.; Bosco, D.; Dalle, S.; Wojtusciszyn, A. Proteasome dysfunction mediates high glucose-induced apoptosis in rodent beta cells and human islets. PLoS ONE 2014, 9, e92066. [Google Scholar]
- Wu, J.; Wu, J.J.; Yang, L.J.; Wei, L.X.; Zou, D.J. Rosiglitazone protects against palmitate-induced pancreatic beta-cell death by activation of autophagy via 5′-AMP-activated protein kinase modulation. Endocrine 2013, 44, 87–98. [Google Scholar] [CrossRef]
- Lai, D.; Huang, M.; Zhao, L.; Tian, Y.; Li, Y.; Liu, D.; Wu, Y.; Deng, F. Delphinidin-induced autophagy protects pancreatic beta cells against apoptosis resulting from high-glucose stress via AMPK signaling pathway. Acta Biochim. Biophys. Sin. 2019, 51, 1242–1249. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.S.; Lee, H.A.; Han, J.S. Sargassum sagamianum extract protects INS-1 pancreatic beta cells against high glucose-induced apoptosis. Cytotechnology 2019, 71, 389–399. [Google Scholar] [CrossRef] [PubMed]
- Elumalai, S.; Karunakaran, U.; Moon, J.S.; Won, K.C. High glucose-induced PRDX3 acetylation contributes to glucotoxicity in pancreatic beta-cells: Prevention by Teneligliptin. Free Radic. Biol. Med. 2020, 160, 618–629. [Google Scholar] [CrossRef] [PubMed]
- Eiberger, J.; Degen, J.; Romualdi, A.; Deutsch, U.; Willecke, K.; Sohl, G. Connexin genes in the mouse and human genome. Cell Commun. Adhes. 2001, 8, 163–165. [Google Scholar] [CrossRef] [PubMed]
- Sohl, G.; Willecke, K. Gap junctions and the connexin protein family. Cardiovasc. Res. 2004, 62, 228–232. [Google Scholar] [CrossRef] [PubMed]
- Mese, G.; Richard, G.; White, T.W. Gap junctions: Basic structure and function. J. Investig. Dermatol. 2007, 127, 2516–2524. [Google Scholar] [CrossRef]
- Rackauskas, M.; Neverauskas, V.; Skeberdis, V.A. Diversity and properties of connexin gap junction channels. Medicina 2010, 46, 1. [Google Scholar] [CrossRef] [PubMed]
- Farnsworth, N.L.; Benninger, R.K. New insights into the role of connexins in pancreatic islet function and diabetes. FEBS Lett. 2014, 588, 1278–1287. [Google Scholar] [CrossRef] [PubMed]
- Saez, J.C.; Retamal, M.A.; Basilio, D.; Bukauskas, F.F.; Bennett, M.V. Connexin-based gap junction hemichannels: Gating mechanisms. Biochim. Biophys. Acta 2005, 1711, 215–224. [Google Scholar] [CrossRef] [PubMed]
- Laird, D.W.; Lampe, P.D. Therapeutic strategies targeting connexins. Nat. Rev. Drug Discov. 2018, 17, 905–921. [Google Scholar] [CrossRef] [PubMed]
- Lampe, P.D.; Laird, D.W. Recent advances in connexin gap junction biology. Fac. Rev. 2022, 11, 14. [Google Scholar] [CrossRef]
- Sosinsky, G.E.; Nicholson, B.J. Structural organization of gap junction channels. Biochim. Biophys. Acta 2005, 1711, 99–125. [Google Scholar] [CrossRef] [PubMed]
- Krysko, D.V.; Leybaert, L.; Vandenabeele, P.; D’Herde, K. Gap junctions and the propagation of cell survival and cell death signals. Apoptosis 2005, 10, 459–469. [Google Scholar] [CrossRef] [PubMed]
- Mollazadeh, S.; Fazly Bazzaz, B.S.; Kerachian, M.A. Role of apoptosis in pathogenesis and treatment of bone-related diseases. J. Orthop. Surg. Res. 2015, 10, 15. [Google Scholar] [CrossRef]
- Serre-Beinier, V.; Le Gurun, S.; Belluardo, N.; Trovato-Salinaro, A.; Charollais, A.; Haefliger, J.A.; Condorelli, D.F.; Meda, P. Cx36 preferentially connects beta-cells within pancreatic islets. Diabetes 2000, 49, 727–734. [Google Scholar] [CrossRef]
- Moreno, A.P.; Berthoud, V.M.; Perez-Palacios, G.; Perez-Armendariz, E.M. Biophysical evidence that connexin-36 forms functional gap junction channels between pancreatic mouse beta-cells. Am. J. Physiol. Endocrinol. Metab. 2005, 288, E948–E956. [Google Scholar] [CrossRef] [PubMed]
- Serre-Beinier, V.; Bosco, D.; Zulianello, L.; Charollais, A.; Caille, D.; Charpantier, E.; Gauthier, B.R.; Diaferia, G.R.; Giepmans, B.N.; Lupi, R.; et al. Cx36 makes channels coupling human pancreatic beta-cells, and correlates with insulin expression. Hum. Mol. Genet. 2009, 18, 428–439. [Google Scholar] [CrossRef]
- Le Gurun, S.; Martin, D.; Formenton, A.; Maechler, P.; Caille, D.; Waeber, G.; Meda, P.; Haefliger, J.A. Connexin-36 contributes to control function of insulin-producing cells. J. Biol. Chem. 2003, 278, 37690–37697. [Google Scholar] [CrossRef] [PubMed]
- Ravier, M.A.; Guldenagel, M.; Charollais, A.; Gjinovci, A.; Caille, D.; Sohl, G.; Wollheim, C.B.; Willecke, K.; Henquin, J.C.; Meda, P. Loss of connexin36 channels alters beta-cell coupling, islet synchronization of glucose-induced Ca2+ and insulin oscillations, and basal insulin release. Diabetes 2005, 54, 1798–1807. [Google Scholar] [CrossRef] [PubMed]
- Speier, S.; Gjinovci, A.; Charollais, A.; Meda, P.; Rupnik, M. Cx36-mediated coupling reduces beta-cell heterogeneity, confines the stimulating glucose concentration range, and affects insulin release kinetics. Diabetes 2007, 56, 1078–1086. [Google Scholar] [CrossRef] [PubMed]
- Wellershaus, K.; Degen, J.; Deuchars, J.; Theis, M.; Charollais, A.; Caille, D.; Gauthier, B.; Janssen-Bienhold, U.; Sonntag, S.; Herrera, P.; et al. A new conditional mouse mutant reveals specific expression and functions of connexin36 in neurons and pancreatic beta-cells. Exp. Cell Res. 2008, 314, 997–1012. [Google Scholar] [CrossRef]
- Head, W.S.; Orseth, M.L.; Nunemaker, C.S.; Satin, L.S.; Piston, D.W.; Benninger, R.K. Connexin-36 gap junctions regulate in vivo first- and second-phase insulin secretion dynamics and glucose tolerance in the conscious mouse. Diabetes 2012, 61, 1700–1707. [Google Scholar] [CrossRef] [PubMed]
- Coronel-Cruz, C.; Hernandez-Tellez, B.; Lopez-Vancell, R.; Lopez-Vidal, Y.; Berumen, J.; Castell, A.; Perez-Armendariz, E.M. Connexin 30.2 is expressed in mouse pancreatic beta cells. Biochem. Biophys. Res. Commun. 2013, 438, 772–777. [Google Scholar] [CrossRef] [PubMed]
- Manasson, J.; Tien, T.; Moore, C.; Kumar, N.M.; Roy, S. High glucose-induced downregulation of connexin 30.2 promotes retinal vascular lesions: Implications for diabetic retinopathy. Investig. Ophthalmol. Vis. Sci. 2013, 54, 2361–2366. [Google Scholar] [CrossRef]
- Klee, P.; Allagnat, F.; Pontes, H.; Cederroth, M.; Charollais, A.; Caille, D.; Britan, A.; Haefliger, J.A.; Meda, P. Connexins protect mouse pancreatic beta cells against apoptosis. J. Clin. Investig. 2011, 121, 4870–4879. [Google Scholar] [CrossRef]
- Allagnat, F.; Klee, P.; Cardozo, A.K.; Meda, P.; Haefliger, J.A. Connexin36 contributes to INS-1E cells survival through modulation of cytokine-induced oxidative stress, ER stress and AMPK activity. Cell Death Differ. 2013, 20, 1742–1752. [Google Scholar] [CrossRef] [PubMed]
- Haefliger, J.A.; Martin, D.; Favre, D.; Petremand, Y.; Mazzolai, L.; Abderrahmani, A.; Meda, P.; Waeber, G.; Allagnat, F. Reduction of connexin36 content by ICER-1 contributes to insulin-secreting cells apoptosis induced by oxidized LDL particles. PLoS ONE 2013, 8, e55198. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez-Sanchez, I.; Lira-Rocha, A.; Navarrete, A.; Loza-Mejia, M.A.; Coronel-Cruz, C.; Mendoza-Rodriguez, C.A.; Cerbon, M.A. Synergistic anticancer activity of Thiazolo [5,4-b]quinoline derivative D3CLP in combination with cisplatin in human cervical cancer cells. Anticancer Res. 2012, 32, 5159–5165. [Google Scholar] [PubMed]
- Upham, B.L.; Sovadinova, I.; Babica, P. Gap Junctional Intercellular Communication: A Functional Biomarker to Assess Adverse Effects of Toxicants and Toxins, and Health Benefits of Natural Products. J. Vis. Exp. 2016, 2016, 54281. [Google Scholar]
- Han, D.; Yang, B.; Olson, L.K.; Greenstein, A.; Baek, S.H.; Claycombe, K.J.; Goudreau, J.L.; Yu, S.W.; Kim, E.K. Activation of autophagy through modulation of 5′-AMP-activated protein kinase protects pancreatic beta-cells from high glucose. Biochem. J. 2010, 425, 541–551. [Google Scholar] [CrossRef] [PubMed]
- Mir, S.U.; George, N.M.; Zahoor, L.; Harms, R.; Guinn, Z.; Sarvetnick, N.E. Inhibition of autophagic turnover in beta-cells by fatty acids and glucose leads to apoptotic cell death. J. Biol. Chem. 2015, 290, 6071–6085. [Google Scholar] [CrossRef] [PubMed]
- Stockert, J.C.; Horobin, R.W.; Colombo, L.L.; Blazquez-Castro, A. Tetrazolium salts and formazan products in Cell Biology: Viability assessment, fluorescence imaging, and labeling perspectives. Acta Histochem. 2018, 120, 159–167. [Google Scholar] [CrossRef]
- Allagnat, F.; Martin, D.; Condorelli, D.F.; Waeber, G.; Haefliger, J.A. Glucose represses connexin36 in insulin-secreting cells. J. Cell Sci. 2005, 118, 5335–5344. [Google Scholar] [CrossRef]
- Kreuzberg, M.M.; Sohl, G.; Kim, J.S.; Verselis, V.K.; Willecke, K.; Bukauskas, F.F. Functional properties of mouse connexin30.2 expressed in the conduction system of the heart. Circ. Res. 2005, 96, 1169–1177. [Google Scholar] [CrossRef]
- Rackauskas, M.; Verselis, V.K.; Bukauskas, F.F. Permeability of homotypic and heterotypic gap junction channels formed of cardiac connexins mCx30.2, Cx40, Cx43, and Cx45. Am. J. Physiol. Heart Circ. Physiol. 2007, 293, H1729–H1736. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Rojas, J.; Bermudez, V.; Palmar, J.; Martinez, M.S.; Olivar, L.C.; Nava, M.; Tomey, D.; Rojas, M.; Salazar, J.; Garicano, C.; et al. Pancreatic Beta Cell Death: Novel Potential Mechanisms in Diabetes Therapy. J. Diabetes Res. 2018, 2018, 9601801. [Google Scholar] [CrossRef] [PubMed]
- Lytrivi, M.; Castell, A.L.; Poitout, V.; Cnop, M. Recent Insights Into Mechanisms of beta-Cell Lipo- and Glucolipotoxicity in Type 2 Diabetes. J. Mol. Biol. 2020, 432, 1514–1534. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Zhou, X.A.; Liu, C.; Shen, Q.; Wu, Y. Vitamin B6 Inhibits High Glucose-Induced Islet beta Cell Apoptosis by Upregulating Autophagy. Metabolites 2022, 12, 1048. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, R.; Girao, H.; Pereira, P. High glucose down-regulates intercellular communication in retinal endothelial cells by enhancing degradation of connexin 43 by a proteasome-dependent mechanism. J. Biol. Chem. 2004, 279, 27219–27224. [Google Scholar] [CrossRef] [PubMed]
- Howarth, F.C.; Chandler, N.J.; Kharche, S.; Tellez, J.O.; Greener, I.D.; Yamanushi, T.T.; Billeter, R.; Boyett, M.R.; Zhang, H.; Dobrzynski, H. Effects of streptozotocin-induced diabetes on connexin43 mRNA and protein expression in ventricular muscle. Mol. Cell. Biochem. 2008, 319, 105–114. [Google Scholar] [CrossRef] [PubMed]
- Wang, G.Y.; Bi, Y.G.; Liu, X.D.; Han, J.F.; Wei, M.; Zhang, Q.Y. Upregulation of connexin 43 and apoptosis-associated protein expression by high glucose in H9c2 cells was improved by resveratrol via the autophagy signaling pathway. Mol. Med. Report. 2017, 16, 3262–3268. [Google Scholar] [CrossRef] [PubMed]
- Shi, W.; Meng, Z.; Luo, J. Connexin 43 (Cx43) regulates high-glucose-induced retinal endothelial cell angiogenesis and retinal neovascularization. Front. Endocrinol. (Lausanne) 2022, 13, 909207. [Google Scholar] [CrossRef] [PubMed]
- Muto, T.; Tien, T.; Kim, D.; Sarthy, V.P.; Roy, S. High glucose alters Cx43 expression and gap junction intercellular communication in retinal Muller cells: Promotes Muller cell and pericyte apoptosis. Investig. Ophthalmol. Vis. Sci. 2014, 55, 4327–4337. [Google Scholar] [CrossRef] [PubMed]
- Zou, H.; Zhuo, L.; Han, T.; Hu, D.; Yang, X.; Wang, Y.; Yuan, Y.; Gu, J.; Bian, J.; Liu, X.; et al. Autophagy and gap junctional intercellular communication inhibition are involved in cadmium-induced apoptosis in rat liver cells. Biochem. Biophys. Res. Commun. 2015, 459, 713–719. [Google Scholar] [CrossRef] [PubMed]
- De Pina-Benabou, M.H.; Szostak, V.; Kyrozis, A.; Rempe, D.; Uziel, D.; Urban-Maldonado, M.; Benabou, S.; Spray, D.C.; Federoff, H.J.; Stanton, P.K.; et al. Blockade of gap junctions in vivo provides neuroprotection after perinatal global ischemia. Stroke 2005, 36, 2232–2237. [Google Scholar] [CrossRef] [PubMed]
- Akopian, A.; Atlasz, T.; Pan, F.; Wong, S.; Zhang, Y.; Volgyi, B.; Paul, D.L.; Bloomfield, S.A. Gap junction-mediated death of retinal neurons is connexin and insult specific: A potential target for neuroprotection. J. Neurosci. 2014, 34, 10582–10591. [Google Scholar] [CrossRef] [PubMed]
- Luo, C.; Yuan, D.; Yao, W.; Cai, J.; Zhou, S.; Zhang, Y.; Hei, Z. Dexmedetomidine protects against apoptosis induced by hypoxia/reoxygenation through the inhibition of gap junctions in NRK-52E cells. Life Sci. 2015, 122, 72–77. [Google Scholar] [CrossRef] [PubMed]
- Sai, K.; Kang, K.S.; Hirose, A.; Hasegawa, R.; Trosko, J.E.; Inoue, T. Inhibition of apoptosis by pentachlorophenol in v-myc-transfected rat liver epithelial cells: Relation to down-regulation of gap junctional intercellular communication. Cancer Lett. 2001, 173, 163–174. [Google Scholar] [CrossRef] [PubMed]
- Dang, X.; Doble, B.W.; Kardami, E. The carboxy-tail of connexin-43 localizes to the nucleus and inhibits cell growth. Mol. Cell. Biochem. 2003, 242, 35–38. [Google Scholar] [CrossRef] [PubMed]
- Kotini, M.; Barriga, E.H.; Leslie, J.; Gentzel, M.; Rauschenberger, V.; Schambony, A.; Mayor, R. Gap junction protein Connexin-43 is a direct transcriptional regulator of N-cadherin in vivo. Nat. Commun. 2018, 9, 3846. [Google Scholar] [CrossRef] [PubMed]
- Giardina, S.F.; Mikami, M.; Goubaeva, F.; Yang, J. Connexin 43 confers resistance to hydrogen peroxide-mediated apoptosis. Biochem. Biophys. Res. Commun. 2007, 362, 747–752. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ortega-Cuellar, D.; González-Sánchez, I.; Piñón-Zárate, G.; Cerbón, M.A.; De la Rosa, V.; Franco-Juárez, Y.; Castell-Rodríguez, A.; Islas, L.D.; Coronel-Cruz, C. Protector Role of Cx30.2 in Pancreatic β-Cell against Glucotoxicity-Induced Apoptosis. Biology 2024, 13, 468. https://doi.org/10.3390/biology13070468
Ortega-Cuellar D, González-Sánchez I, Piñón-Zárate G, Cerbón MA, De la Rosa V, Franco-Juárez Y, Castell-Rodríguez A, Islas LD, Coronel-Cruz C. Protector Role of Cx30.2 in Pancreatic β-Cell against Glucotoxicity-Induced Apoptosis. Biology. 2024; 13(7):468. https://doi.org/10.3390/biology13070468
Chicago/Turabian StyleOrtega-Cuellar, Daniel, Ignacio González-Sánchez, Gabriela Piñón-Zárate, Marco A. Cerbón, Víctor De la Rosa, Yuliana Franco-Juárez, Andrés Castell-Rodríguez, León D. Islas, and Cristina Coronel-Cruz. 2024. "Protector Role of Cx30.2 in Pancreatic β-Cell against Glucotoxicity-Induced Apoptosis" Biology 13, no. 7: 468. https://doi.org/10.3390/biology13070468
APA StyleOrtega-Cuellar, D., González-Sánchez, I., Piñón-Zárate, G., Cerbón, M. A., De la Rosa, V., Franco-Juárez, Y., Castell-Rodríguez, A., Islas, L. D., & Coronel-Cruz, C. (2024). Protector Role of Cx30.2 in Pancreatic β-Cell against Glucotoxicity-Induced Apoptosis. Biology, 13(7), 468. https://doi.org/10.3390/biology13070468






