Heterogeneous Evolution of Breast Cancer Cells—An Endogenous Molecular-Cellular Network Study
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
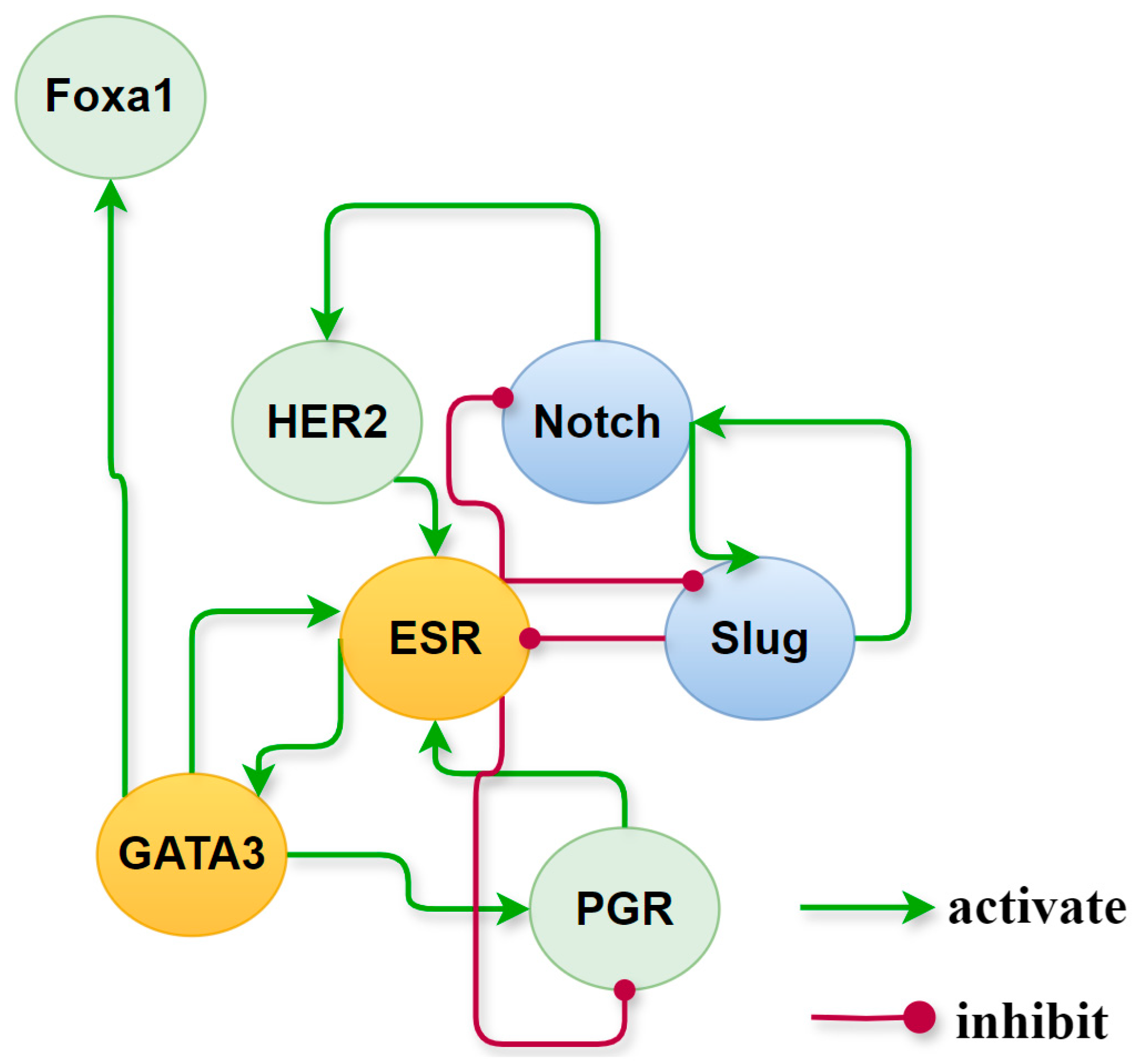
2.1. Biological Hypothesis and Principles of Network Construction
2.2. Quantitative Simulation of Endogenous Molecular–Cellular Network
2.3. Attractors for Cellular States
2.4. Validation of Modeling Results
2.5. Connectivity, Topology, and Phenotypic Landscape
3. Results
3.1. Construction of Core Endogenous Network for Breast Cancer
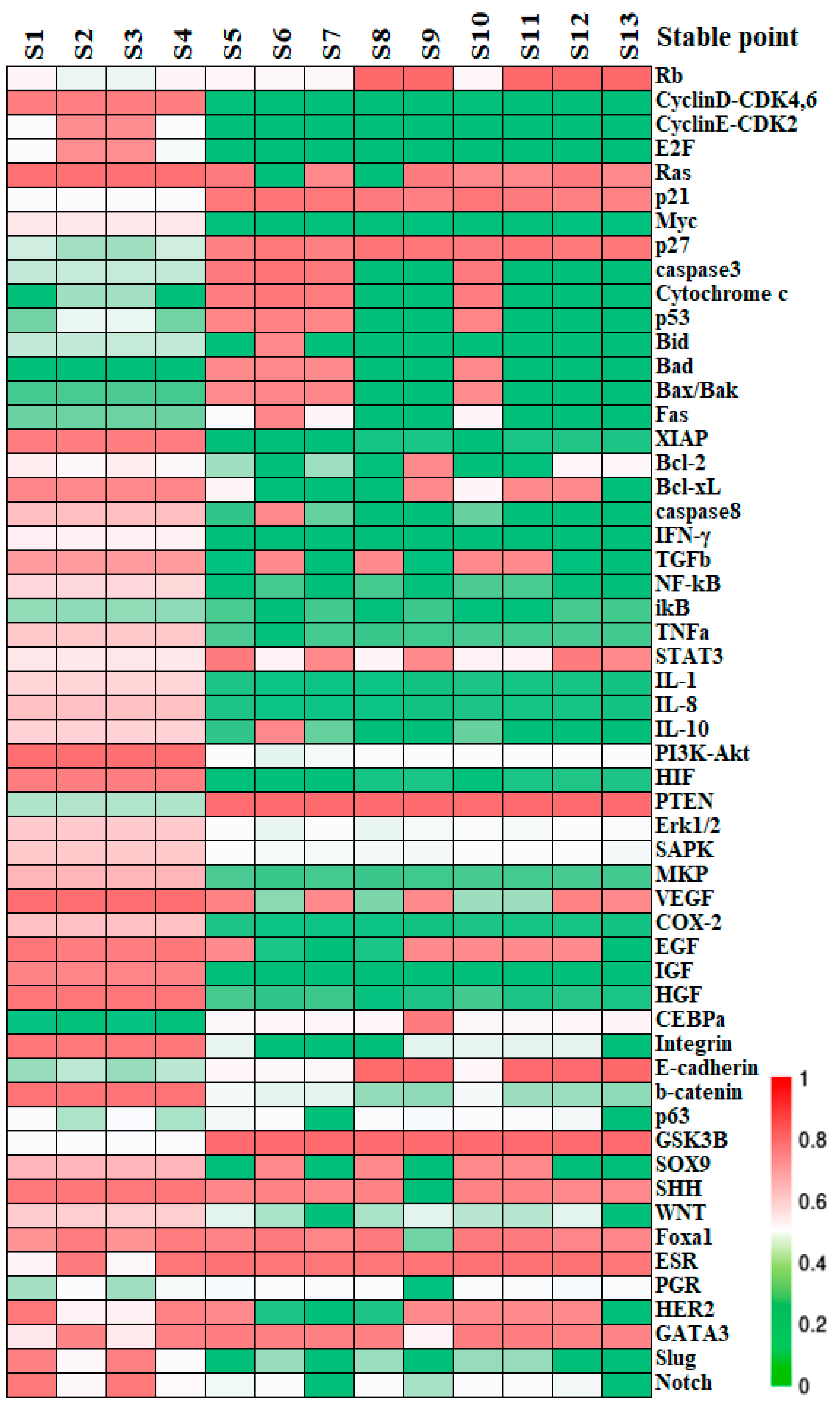
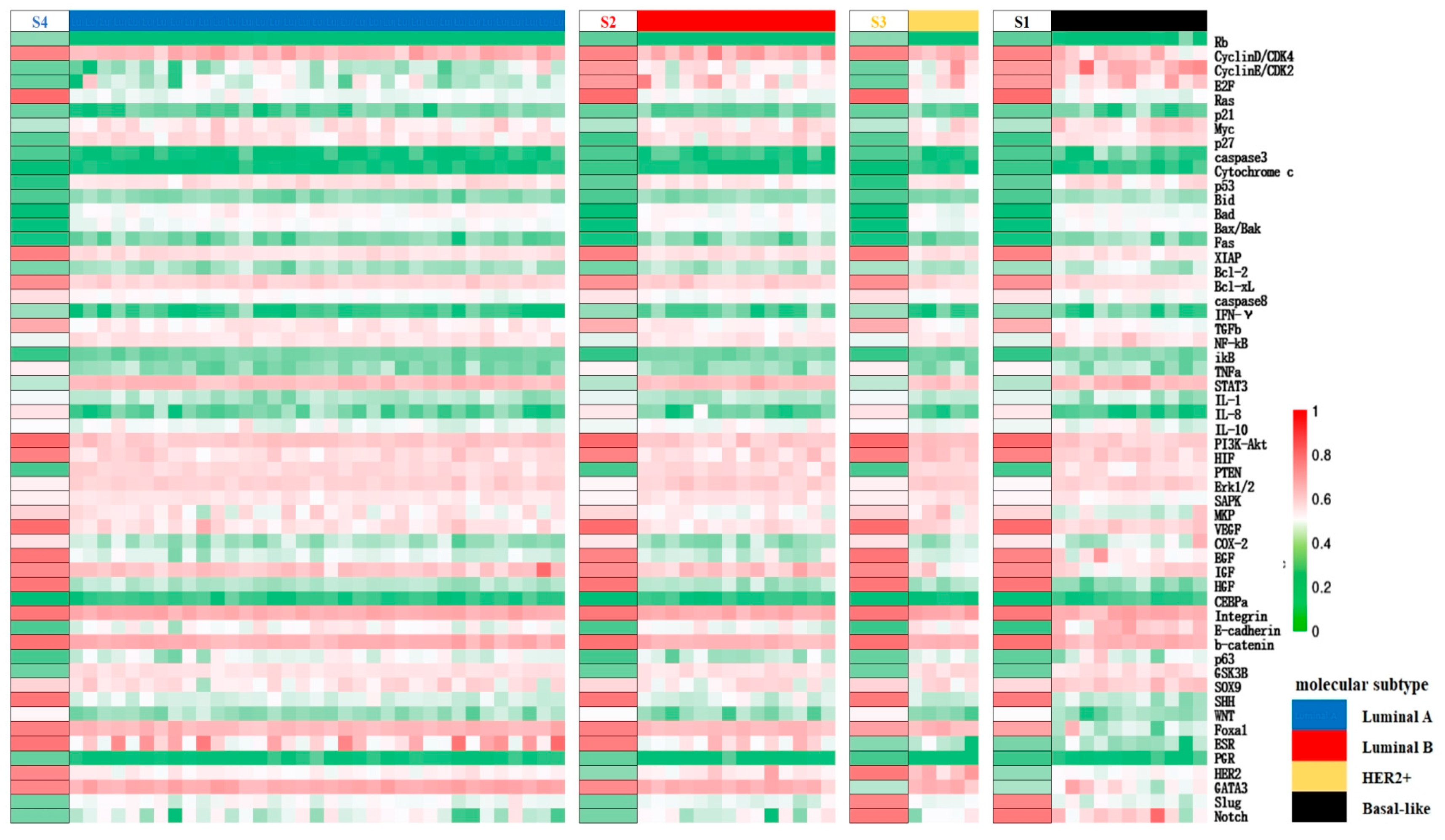
3.2. Identification of Cell States and Validation against Clinical Data
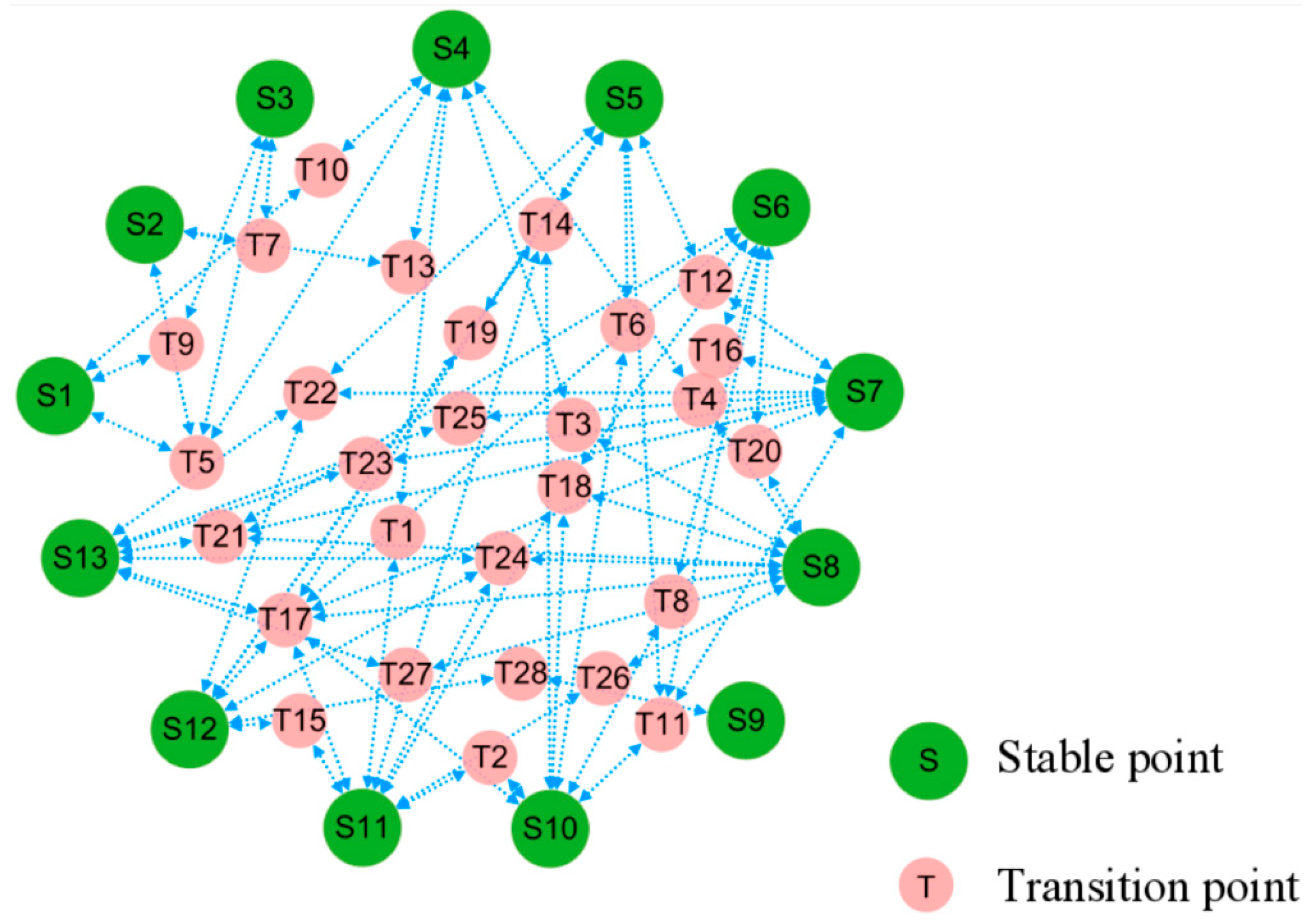
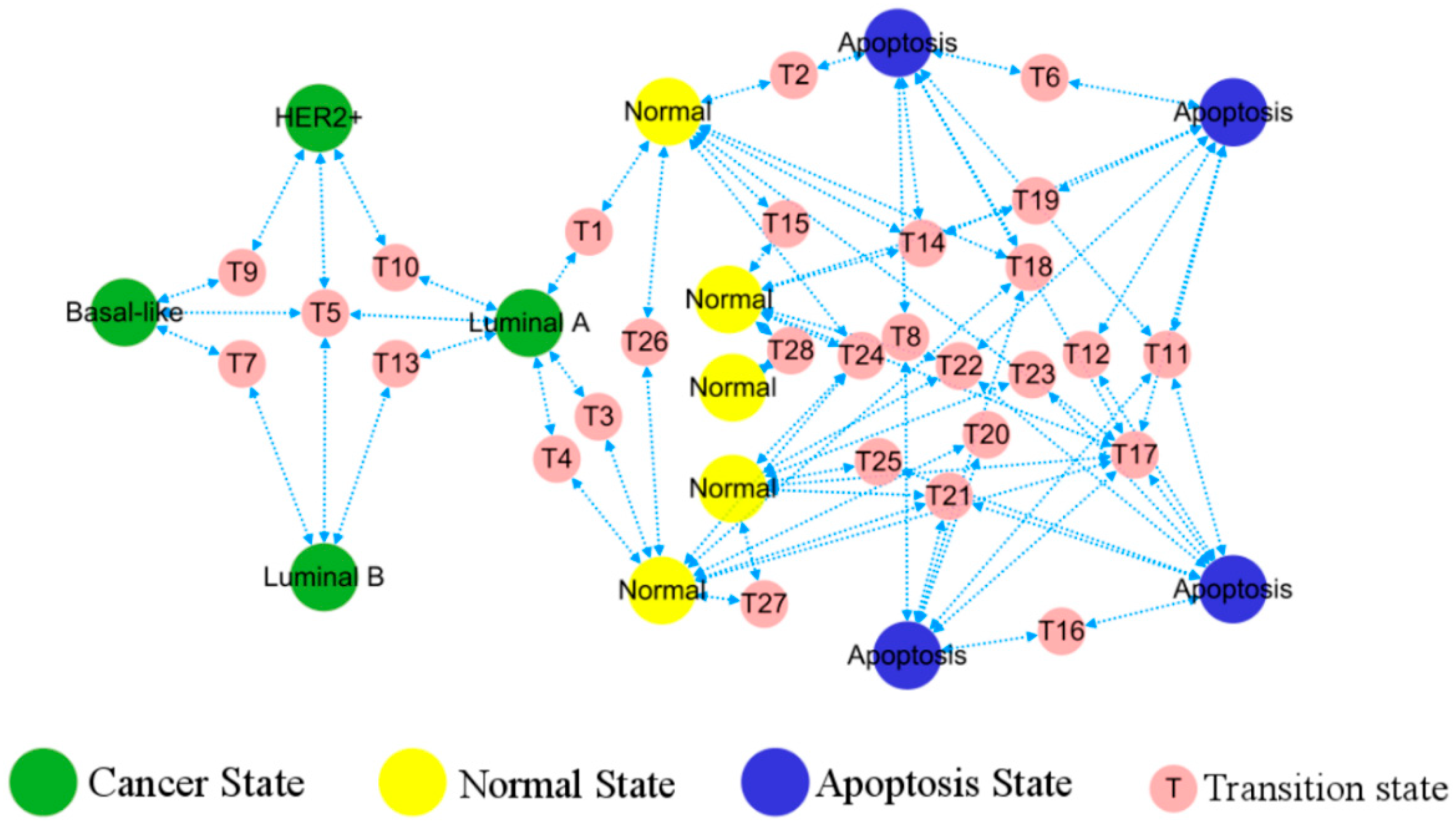
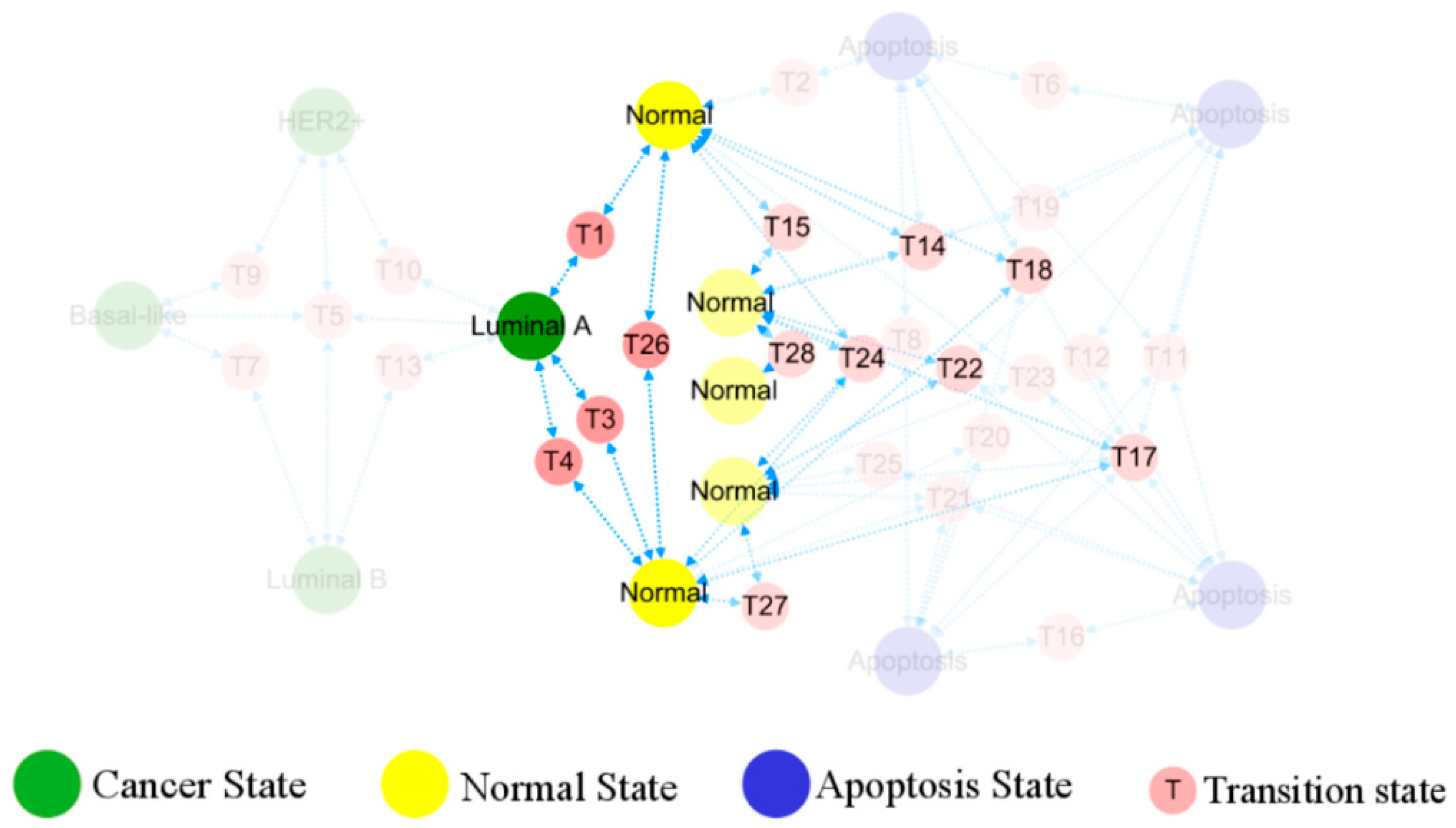
3.3. Topological Structure and Functional Landscape of Phenotype
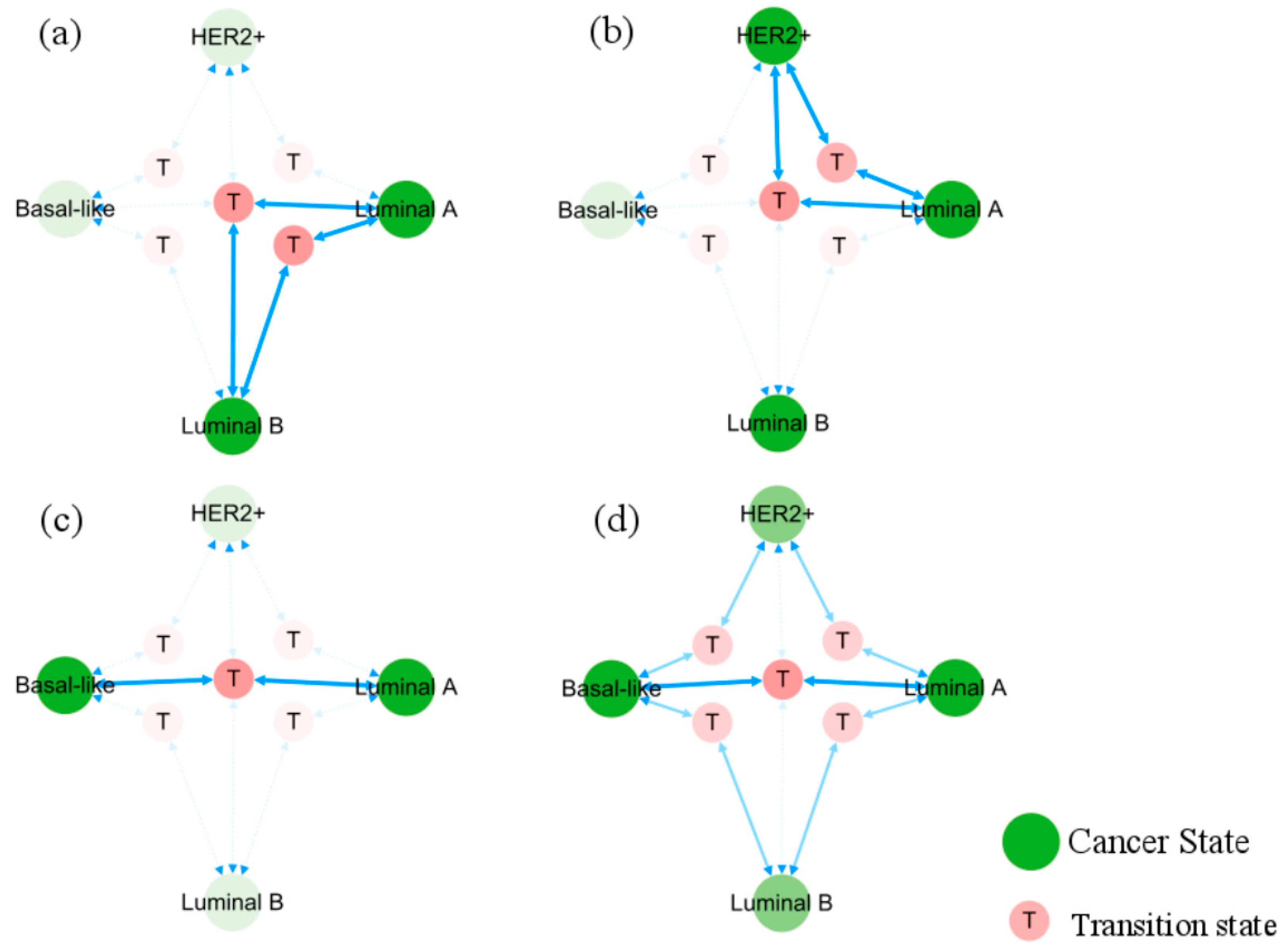
3.4. Cellular Origins and Heterogeneous Subtypes of Cancer
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J. Clin. 2021, 71, 209–249. [Google Scholar] [CrossRef]
- Siegel, R.L.; Miller, K.D.; Wagle, N.S.; Jemal, A. Cancer Statistics, 2023. CA Cancer J. Clin. 2023, 73, 17–48. [Google Scholar] [CrossRef] [PubMed]
- Torre, L.A.; Islami, F.; Siegel, R.L.; Ward, E.M.; Jemal, A. Global Cancer in Women: Burden and Trends. Cancer Epidemiol. Biomark. Prev. 2017, 26, 444–457. [Google Scholar] [CrossRef]
- Miller, K.D.; Nogueira, L.; Devasia, T.; Mariotto, A.B.; Yabroff, K.R.; Jemal, A.; Kramer, J.; Siegel, R.L. Cancer treatment and survivorship statistics, 2022. CA Cancer J. Clin. 2022, 72, 409–436. [Google Scholar] [CrossRef] [PubMed]
- Marusyk, A.; Almendro, V.; Polyak, K. Intra-tumour heterogeneity: A looking glass for cancer? Nat. Rev. Cancer 2012, 12, 323–334. [Google Scholar] [CrossRef]
- Park, S.Y.; Gönen, M.; Kim, H.J.; Michor, F.; Polyak, K. Cellular and genetic diversity in the progression of in situ human breast carcinomas to an invasive phenotype. J. Clin. Investig. 2010, 120, 636. [Google Scholar] [CrossRef] [PubMed]
- Bravatà, V.; Minafra, L.; Cammarata, F.P.; Pisciotta, P.; Lamia, D.; Marchese, V.; Petringa, G.; Manti, L.; Cirrone, G.A.; Gilardi, M.C.; et al. Gene expression profiling of breast cancer cell lines treated with proton and electron radiations. Br. J. Radiol. 2018, 91, 20170934. [Google Scholar] [CrossRef]
- Harbeck, N.; Penault-Llorca, F.; Cortes, J.; Gnant, M.; Houssami, N.; Poortmans, P.; Ruddy, K.; Tsang, J.; Cardoso, F. Breast cancer. Nat. Rev. Dis. Primers. 2019, 5, 66. [Google Scholar] [CrossRef]
- Weigelt, B.; Horlings, H.M.; Kreike, B.; Hayes, M.M.; Hauptmann, M.; Wessels, L.F.; de Jong, D.; Van de Vijver, M.J.; Van’t Veer, L.J.; Peterse, J.L.; et al. Refinement of breast cancer classification by molecular characterization of histological special types. J. Pathol. 2008, 216, 141–150. [Google Scholar] [CrossRef]
- Harbeck, N. Breast cancer is a systemic disease optimally treated by a multidisciplinary team. Nat. Rev. Dis. Primers. 2020, 6, 30. [Google Scholar] [CrossRef]
- Yeo, S.K.; Guan, J.L. Breast Cancer: Multiple Subtypes within a Tumor? Trends Cancer 2017, 3, 753–760. [Google Scholar] [CrossRef] [PubMed]
- Polyak, K. Heterogeneity in breast cancer. J. Clin. Investig. 2011, 121, 3786–3788. [Google Scholar] [CrossRef] [PubMed]
- Tan, P.H.; Ellis, I.; Allison, K.; Brogi, E.; Fox, S.B.; Lakhani, S.; Lazar, A.J.; Morris, E.A.; Sahin, A.; Salgado, R.; et al. The 2019 World Health Organization classification of tumours of the breast. Histopathology 2020, 77, 181–185. [Google Scholar] [CrossRef] [PubMed]
- Weigelt, B.; Geyer, F.C.; Reis-Filho, J.S. Histological types of breast cancer: How special are they? Mol. Oncol. 2010, 4, 192–208. [Google Scholar] [CrossRef] [PubMed]
- Simpson, P.T.; Gale, T.; Fulford, L.G.; Reis-Filho, J.S.; Lakhani, S.R. The molecular evolution of breast cancer. J. Pathol. 2010, 220, 222–232. [Google Scholar] [CrossRef] [PubMed]
- Fu, N.Y.; Nolan, E.; Lindeman, G.J.; Visvader, J.E. Stem Cells and the Differentiation Hierarchy in Mammary Gland Development. Physiol. Rev. 2020, 100, 489–523. [Google Scholar] [CrossRef]
- Testa, U.; Castelli, G.; Pelosi, E. Breast Cancer: A Molecularly Heterogenous Disease Needing Subtype-Specific Treatments. Med. Sci. 2020, 8, 18. [Google Scholar] [CrossRef] [PubMed]
- Inman, J.L.; Robertson, C.; Mott, J.D.; Bissell, M.J. Mammary gland development: Cell fate specification, stem cells and the microenvironment. Development 2015, 142, 1028–1042. [Google Scholar] [CrossRef] [PubMed]
- Eroles, P.; Bosch, A.; Pérez-Fidalgo, J.A.; Lluch, A. Molecular biology in breast cancer: Intrinsic subtypes and signaling pathways. Cancer Treat. Rev. 2012, 38, 698–707. [Google Scholar] [CrossRef]
- Perou, C.M.; Sørile, T.; Eisen, M.B.; Van De Rijn, M.; Jeffrey, S.S.; Rees, C.A.; Pollack, J.R.; Ross, D.T.; Johnsen, H.; Akslen, L.A.; et al. Molecular portraits of human breast tumours. Nature 2000, 406, 747–752. [Google Scholar] [CrossRef]
- Troester, M.A.; Hoadley, K.A.; Sørlie, T.; Herbert, B.-S.; Børresen-Dale, A.-L.; Lønning, P.E.; Shay, J.W.; Kaufmann, W.K.; Perou, C.M. Cell-type-specific responses to chemotherapeutics in breast cancer. Cancer Res. 2004, 64, 4218–4226. [Google Scholar] [CrossRef] [PubMed]
- Peppercorn, J.; Perou, C.M.; Carey, L.A. Molecular subtypes in breast cancer evaluation and management: Divide and conquer. Cancer Investig. 2008, 26, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Sorlie, T.; Perou, C.M.; Tibshirani, R.; Aas, T.; Geisler, S.; Johnsen, H.; Hastie, T.; Eisen, M.B.; van de Rijn, M.; Jeffrey, S.S.; et al. Gene expression patterns of breast carcinomas distinguish tumor subclasses with clinical implications. Proc. Natl. Acad. Sci. USA 2001, 98, 10869–10874. [Google Scholar] [CrossRef] [PubMed]
- Gusterson, B. Do ‘basal-like’ breast cancers really exist? Nat. Rev. Cancer 2009, 9, 128–134. [Google Scholar] [CrossRef] [PubMed]
- Wellings, S.R.; Jensen, H.M. On the origin and progression of ductal carcinoma in the human breast. J. Natl. Cancer Inst. 1973, 50, 1111–1118. [Google Scholar] [CrossRef] [PubMed]
- Wellings, S.R.; Jensen, H.M.; Marcum, R.G. An atlas of subgross pathology of the human breast with special reference to possible precancerous lesions. J. Natl. Cancer Inst. 1975, 55, 231–273. [Google Scholar] [PubMed]
- Simpson, P.T.; Reis-Filho, J.S.; Gale, T.; Lakhani, S.R. Molecular evolution of breast cancer. J. Pathol. 2005, 205, 248–254. [Google Scholar] [CrossRef] [PubMed]
- Geyer, F.C.; Marchio, C.; Reis-Filho, J.S. The role of molecular analysis in breast cancer. Pathology 2009, 41, 77–88. [Google Scholar] [CrossRef]
- Khella, C.A.; Mehta, G.A.; Mehta, R.N.; Gatza, M.L. Recent Advances in Integrative Multi-Omics Research in Breast and Ovarian Cancer. J. Pers. Med. 2021, 11, 149. [Google Scholar] [CrossRef]
- Ao, P.; Galas, D.; Hood, L.; Zhu, X.M. Cancer as robust intrinsic state of endogenous molecular cellular network shaped by evolution. Med. Hypotheses 2008, 70, 678–684. [Google Scholar] [CrossRef]
- Yuan, R.S.; Zhu, X.M.; Wang, G.W.; Li, S.; Ao, P. Cancer as robust intrinsic state shaped by evolution: A key issues review. Rep. Prog. Phys. 2017, 80, 042701. [Google Scholar] [CrossRef] [PubMed]
- Zhu, X.M.; Yuan, R.S.; Hood, L.; Ao, P. Endogenous molecular-cellular hierarchical modeling of prostate carcinogenesis uncovers robust structure. Prog. Biophys. Mol. Biol. 2015, 117, 30–42. [Google Scholar] [CrossRef] [PubMed]
- Yuan, R.S.; Zhang, S.Z.; Yu, J.K.; Huang, Y.; Lu, D.; Cheng, R.; Huang, S.; Ao, P.; Zheng, S.; Hood, L.; et al. Beyond cancer genes: Colorectal cancer as robust intrinsic states formed by molecular interactions. Open Biol. 2017, 7, 2157–2166. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.Q.; Yuan, R.S.; Zhu, X.M.; Ao, P. Adaptive landscape shaped by core endogenous network coordinates complex early progenitor fate commitments in embryonic pancreas. Sci. Rep. 2020, 10, 1112. [Google Scholar] [CrossRef] [PubMed]
- Weinberg, R.A. The Biology of Cancer, 2nd ed.; Garland Science: New York, NY, USA, 2014; pp. 275–390. [Google Scholar]
- Pellacani, D.; Tan, S.; Lefort, S.; Eaves, C.J. Transcriptional regulation of normal human mammary cell heterogeneity and its perturbation in breast cancer. EMBO J. 2019, 38, 14. [Google Scholar] [CrossRef] [PubMed]
- Hanahan, D.; Weinberg, R.A. Hallmarks of cancer: The next generation. Cell 2011, 144, 646–674. [Google Scholar] [CrossRef] [PubMed]
- Yuan, R.S.; Ma, Y.A.; Yuan, B.; Ao, P. Lyapunov function as potential function: A dynamical equivalence. Chin. Phys. B 2014, 23, 010505. [Google Scholar] [CrossRef]
- Shi, J.H.; Chen, T.Q.; Yuan, R.S.; Yuan, B.; Ao, P. Relation of a new interpretation of stochastic differential equations to Ito process. J. Stat. Phys. 2012, 184, 579–590. [Google Scholar] [CrossRef]
- Ao, P.; Galas, D.; Hood, L.; Yin, L.; Zhu, X.M. Towards predictive stochastic dynamical modeling of cancer genesis and progression. Interdiscip. Sci. Comput. Life Sci. 2010, 2, 140–144. [Google Scholar] [CrossRef] [PubMed]
- Macarthur, B.D.; Ma’ayan, A.; Lemischka, I.R. Systems biology of stem cell fate and cellular reprogramming. Nat. Rev. Mol. Cell Biol. 2009, 10, 672–681. [Google Scholar] [CrossRef]
- Kale, J.; Liu, Q.; Leber, B.; Andrews, D.W. Shedding Light on Apoptosis at Subcellular Membranes. Cell 2012, 151, 1179–1184. [Google Scholar] [CrossRef] [PubMed]
- Hanahan, D.; Weinberg, R.A. The hallmarks of cancer. Cell 2000, 100, 57–70. [Google Scholar] [CrossRef] [PubMed]
- Heiden, M.G.V.; Cantley, L.C.; Thompson, C.B. Understanding the Warburg effect: The metabolic requirements of cell proliferation. Science 2009, 324, 1029–1033. [Google Scholar] [CrossRef] [PubMed]
- Wang, E.; Zaman, N.; McGee, S.; Milanese, J.-S.; Masoudi-Nejad, A.; O’Connor-McCourt, M. Predictive genomics: A cancer hallmark network framework for predicting tumor clinical phenotypes using genome sequencing data. Semin. Cancer Biol. 2015, 30, 4–12. [Google Scholar] [CrossRef] [PubMed]
- Phillips, S.; Prat, A.; Sedic, M.; Proia, T.; Wronski, A.; Mazumdar, S.; Skibinski, A.; Shirley, S.H.; Perou, C.M.; Gill, G.; et al. Cell-state transitions regulated by SLUG are critical for tissue regeneration and tumor initiation. Stem Cell Rep. 2014, 2, 633–647. [Google Scholar] [CrossRef]
- Zhang, X.; Chen, Y.C.; Yao, M.; Xiong, R.; Liu, B.; Zhu, X.; Ao, P. Potential therapeutic targets of gastric cancer explored under endogenous network modeling of clinical data. Sci. Rep. 2024, 14, 13127. [Google Scholar] [CrossRef]
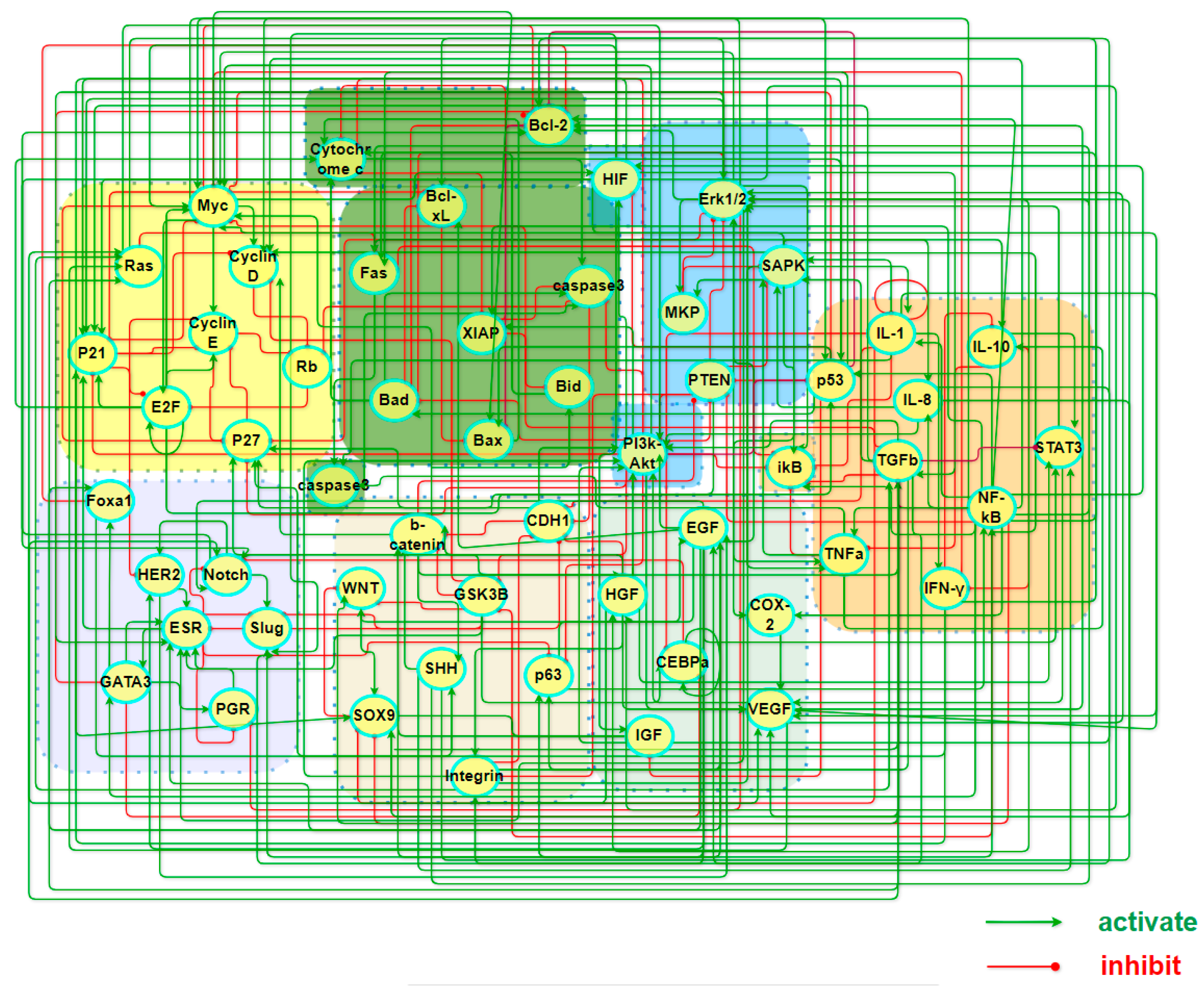
| Module | Factors |
|---|---|
| Cell cycle | Rb, CyclinD/CDK4, CyclinE/CDK2, E2F, Ras, p21, p27, Myc |
| Cell apoptosis | caspase3, Cytochrome c, Bid, Bad, Bax, Fas, XIAP, Bcl2, BclxL, caspase8 |
| Inflammation | IFN-γ, TGFb, NF-kB, p53, ikB, TNFa, STAT3, IL-1, IL-8, IL-10 |
| Metabolism | PI3K-Akt, HIF, PTEN, Erk1/2, SAPK, MKP |
| Angiogenesis | VEGF, COX-2, EGF, IGF, HGF, CEBPa |
| Adhesion factor | Integrin, E-cadherin, b-catenin, p63, GSK3B, SOX9, SHH, WNT |
| BC special factor | Foxa1, ER, PR, HER2, GATA3, Slug, Notch |
| Stable State | Cell Cycle | Cell Apoptosis | Inflammation | Metabolism | Angiogenesis | Adhesion Factor | Cell State |
|---|---|---|---|---|---|---|---|
| S1 | OFF | OFF | ON | ON | ON | OFF | Cancer |
| S2 | ON | OFF | ON | ON | ON | OFF | Cancer |
| S3 | OFF | OFF | ON | ON | ON | OFF | Cancer |
| S4 | ON | OFF | ON | ON | ON | OFF | Cancer |
| S5 | OFF | ON | OFF | OFF | OFF | OFF | Apoptosis |
| S6 | OFF | ON | OFF | OFF | OFF | OFF | Apoptosis |
| S7 | OFF | ON | OFF | OFF | OFF | OFF | Apoptosis |
| S8 | OFF | OFF | OFF | OFF | OFF | ON | Normal |
| S9 | OFF | OFF | OFF | OFF | OFF | ON | Normal |
| S10 | OFF | ON | OFF | OFF | OFF | OFF | Apoptosis |
| S11 | OFF | OFF | OFF | OFF | OFF | ON | Normal |
| S12 | OFF | OFF | OFF | OFF | OFF | ON | Normal |
| S13 | OFF | OFF | OFF | OFF | OFF | ON | Normal |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, T.; Chen, Y.-C.; Ao, P. Heterogeneous Evolution of Breast Cancer Cells—An Endogenous Molecular-Cellular Network Study. Biology 2024, 13, 564. https://doi.org/10.3390/biology13080564
Li T, Chen Y-C, Ao P. Heterogeneous Evolution of Breast Cancer Cells—An Endogenous Molecular-Cellular Network Study. Biology. 2024; 13(8):564. https://doi.org/10.3390/biology13080564
Chicago/Turabian StyleLi, Tianqi, Yong-Cong Chen, and Ping Ao. 2024. "Heterogeneous Evolution of Breast Cancer Cells—An Endogenous Molecular-Cellular Network Study" Biology 13, no. 8: 564. https://doi.org/10.3390/biology13080564
APA StyleLi, T., Chen, Y.-C., & Ao, P. (2024). Heterogeneous Evolution of Breast Cancer Cells—An Endogenous Molecular-Cellular Network Study. Biology, 13(8), 564. https://doi.org/10.3390/biology13080564






