State-of-the-Art Optical Devices for Biomedical Sensing Applications—A Review
Abstract
:1. Introduction
2. Integrated Photonic Biosensing Platform
2.1. MZI and Young’s Interferometer Biomedical Devices
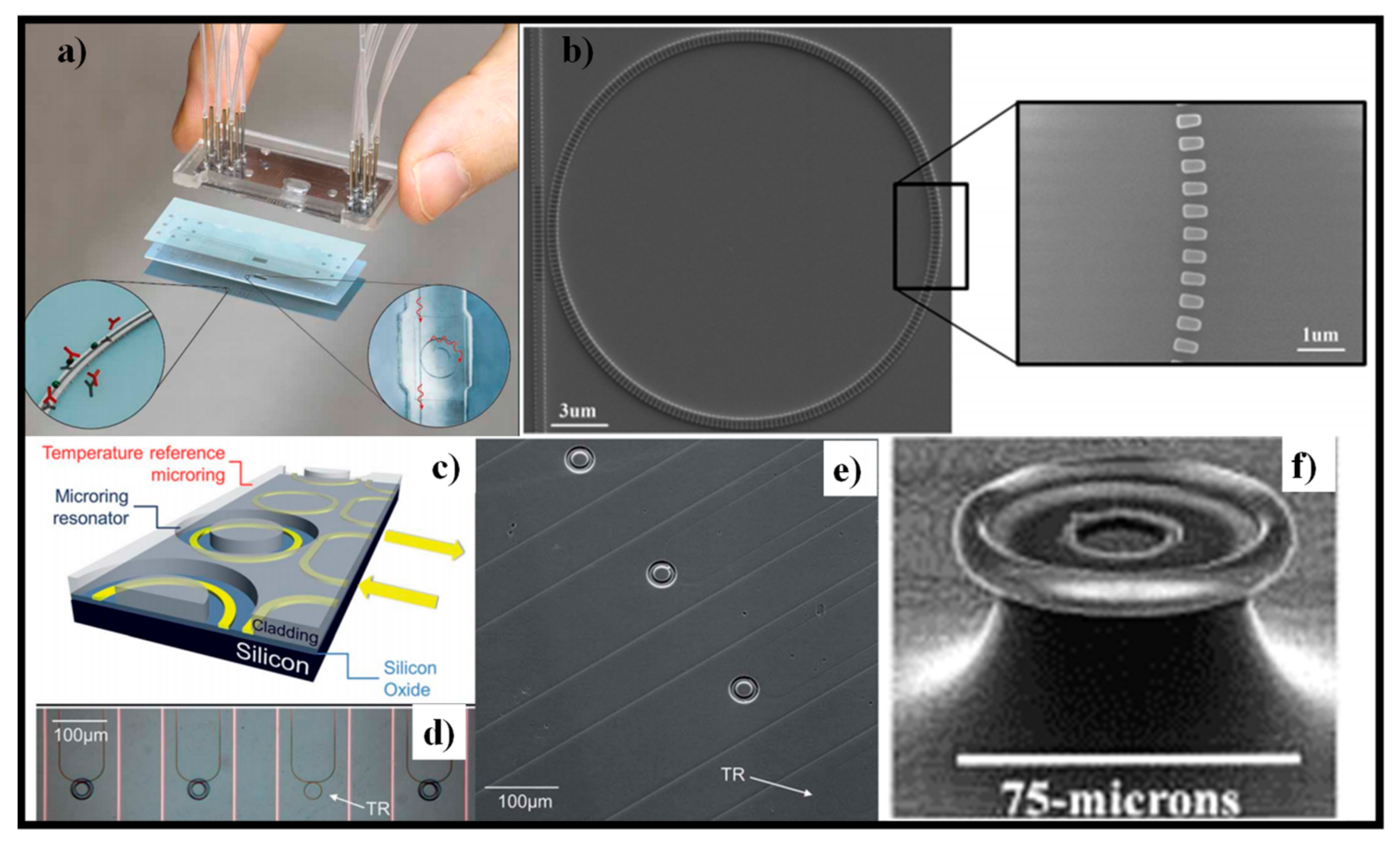
2.2. Silicon μ-RR Biosensors
2.3. Vertical Grating Coupler (VGC)
3. SPR-Optical Fiber-Based Biosensors
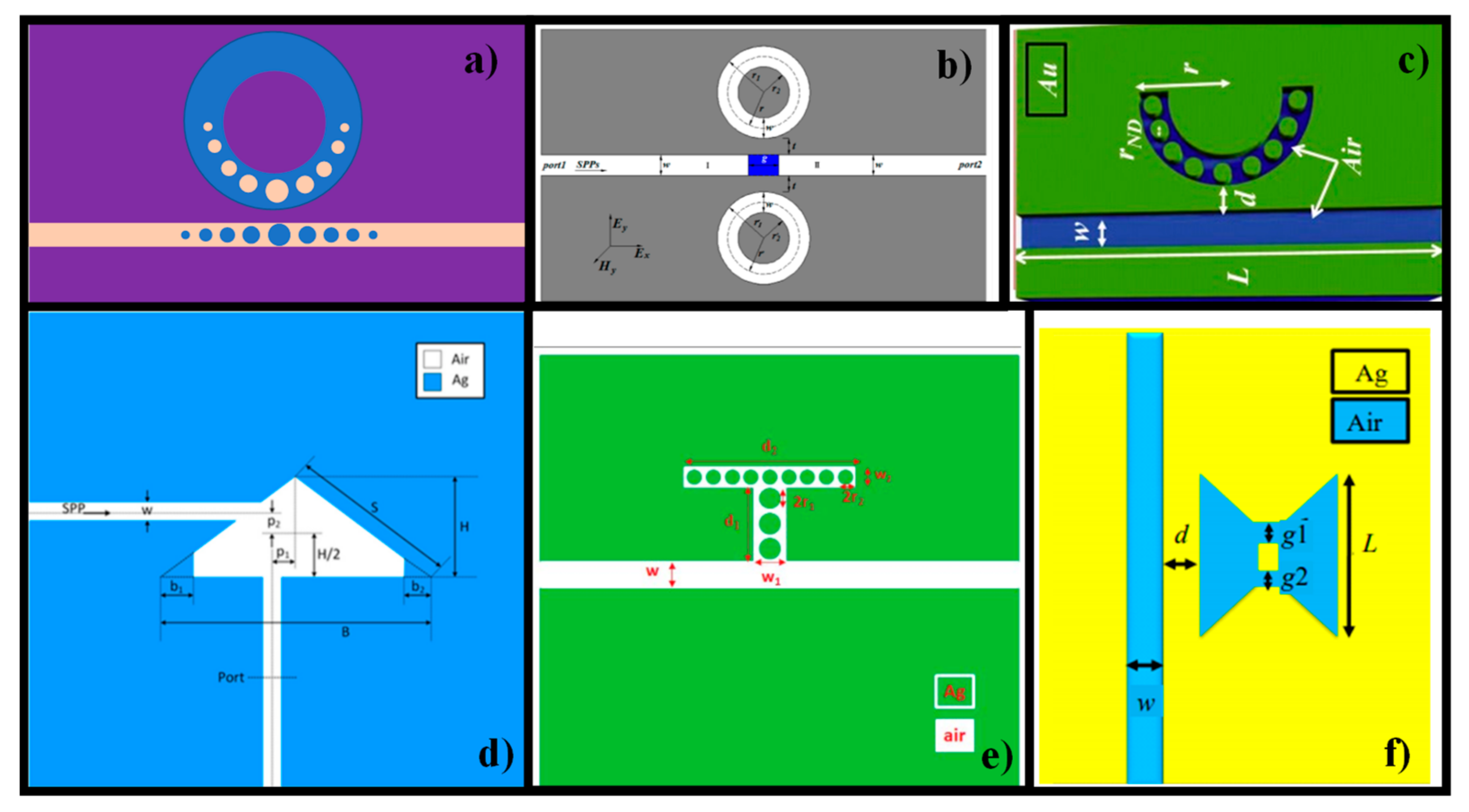
4. Metal-Insulator-Metal WG-Based Biosensors
5. Metasurfaces for Biosensing
6. Photonic Crystals-Based Biosensors
7. Photonic Biosensing with Thin Metal Films
8. Fiber Bragg Grating Biosensors
9. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Available online: globenewswire.com (accessed on 15 March 2021).
- Yu, D.; Blankert, B.; Vire, J.; Kauffmann, J.; Blankert, J.P. Biosensors in Drug Discovery and Drug Analysis. Anal. Lett. 2005, 38, 1687–1701. [Google Scholar] [CrossRef]
- Thakur, M.S.; Ragavan, K.V. Biosensors in food processing. J. Food Sci. Technol. 2013, 50, 625–641. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gil, E.D.; Melo, G.R. Electrochemical biosensors in pharmaceutical analysis. Braz. J. Pharm. Sci. 2010, 46, 375–391. [Google Scholar] [CrossRef]
- Sadana, A.; Sadana, N. Market size and economics for biosensors. In Fractal Analysis of the Binding and Dissociation Kinetics for Different Analytes on Biosensor Surfaces; Elsevier: Amsterdam, The Netherlands, 2008; pp. 317–334. [Google Scholar]
- Clark, L. Monitor and control of blood and tissue oxygen tensions. Trans. Am. Soc. Artif. Internal Organs 1956, 2, 41–46. [Google Scholar]
- Chun, H.J.; Han, Y.D.; Park, Y.M.; Kim, K.R.; Lee, S.J.; Yoon, H.C. An optical biosensing strategy based on selective light absorption and wavelength filtering from chromogenic reaction. Materials 2018, 11, 388. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ligler, F.S. Fluorescence-based optical biosensors. In Biophotonics; Springer: Berlin/Heidelberg, Germany, 2008; pp. 199–215. [Google Scholar]
- Chen, Y.; Liu, J.; Yang, Z.; Wilkinson, J.S.; Zhou, X. Optical biosensors based on refractometric sensing schemes: A review. Biosens. Bioelectron. 2019, 144, 111693. [Google Scholar] [CrossRef] [PubMed]
- Maisonneuve, M.; Song, I.-H.; Patskovsky, S.; Meunier, M. Polarimetric total internal reflection biosensing. Opt. Express 2011, 19, 7410–7416. [Google Scholar] [CrossRef]
- Hutchinson, A.M. Evanescent wave biosensors. Mol. Biotechnol. 1995, 3, 47–54. [Google Scholar] [CrossRef]
- Taitt, C.R.; Anderson, G.P.; Ligler, F.S. Evanescent wave fluorescence biosensors: Advances of the last decade. Biosens. Bioelectron. 2016, 76, 103–112. [Google Scholar] [CrossRef] [Green Version]
- Huertas, C.S.; Calvo-Lozano, O.; Mitchell, A.; Lechuga, L.M. Advanced evanescent-wave optical biosensors for the detection of nucleic acids: An analytic perspective. Front. Chem. 2019, 7, 724. [Google Scholar] [CrossRef] [Green Version]
- Zhang, Z.; Li, Q.; Du, X.; Liu, M. Application of electrochemical biosensors in tumor cell detection. Thorac. Cancer 2020, 11, 840–850. [Google Scholar] [CrossRef] [PubMed]
- Villiger, M.; Stoop, R.; Vetsch, T.; Hohenauer, E.; Pini, M.; Clarys, P.; Pereira, F.; Clijsen, R. Evaluation and review of body fluids saliva, sweat and tear compared to biochemical hydration assessment markers within blood and urine. Eur. J. Clin. Nutr. 2017, 72, 69–76. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lee, S.H.; Cho, Y.C.; Choy, Y.B. Noninvasive self-diagnostic device for tear collection and glucose measurement. Sci. Rep. 2019, 9, 4747. [Google Scholar] [CrossRef] [Green Version]
- Kazanskiy, N.; Butt, M.; Khonina, S. Nanodots decorated MIM semi-ring resonator cavity for biochemical sensing applications. Photon. Nanostructures Fundam. Appl. 2020, 42, 100836. [Google Scholar] [CrossRef]
- Butt, M.A.; Kazanskiy, N.L.; Khonina, S.N. Label-free detection of ambient refractive index based on plasmonic Bragg gratings embedded resonator cavity sensor. J. Mod. Opt. 2019, 66, 1920–1925. [Google Scholar] [CrossRef]
- Xie, Y.; Zhang, M.; Dai, D. Design Rule of Mach-Zehnder Interferometer Sensors for Ultra-High Sensitivity. Sensors 2020, 20, 2640. [Google Scholar] [CrossRef]
- Kalbarczyk, A.; Jaroszewicz, L.R.; Bennis, N.; Chrusciel, M.; Marc, P. The Young Interferometer as an Optical System for a Variable Depolarizer Characterization. Sensors 2019, 19, 3037. [Google Scholar] [CrossRef] [Green Version]
- Yi, H.; Citrin, D.S.; Chen, Y.; Zhou, Z. Dual-microring-resonator interference sensor. Appl. Phys. Lett. 2009, 95, 191112. [Google Scholar] [CrossRef]
- Butt, M.A.; Khonina, S.N.; Kazanskiy, N.L. Device performance of standard strip, slot and hybrid plasmonic μ-ring resonator: A comparative study. Waves Random Complex Media 2020, 1–10. [Google Scholar] [CrossRef]
- Heideman, R.; Kooyman, R.; Greve, J. Development of an optical waveguide interferometric immunosensor. Sens. Actuators B Chem. 1991, 4, 297–299. [Google Scholar] [CrossRef] [Green Version]
- Liu, Q.; Tu, X.; Kim, K.W.; Kee, J.S.; Shin, Y.; Han, K.; Yoon, Y.-J.; Lo, G.-Q.; Park, M.K. Highly sensitive Mach–Zehnder interferometer biosensor based on silicon nitride slot waveguide. Sens. Actuators B Chem. 2013, 188, 681–688. [Google Scholar] [CrossRef]
- Mahmudin, D.; Huda, N.; Estu, T.T.; A Fathnan, A.; Daud, P.; Hardiati, S.; Hasanah, L.; Wijayanto, Y.N. Design of optical channel waveguide Mach-Zehnder interferometer (MZI) for environmental sensor applications. J. Phys. Conf. Ser. 2017, 817, 12036. [Google Scholar] [CrossRef] [Green Version]
- Lee, J.-M. Ultrahigh Temperature-Sensitive Silicon MZI with Titania Cladding. Front. Mater. 2015, 2, 36. [Google Scholar] [CrossRef] [Green Version]
- Lin, B.; Yi, Y.; Cao, Y.; Lv, J.; Yang, Y.; Wang, F.; Sun, X.; Zhang, D.; Lin, Y. A Polymer Asymmetric Mach–Zehnder Interferometer Sensor Model Based on Electrode Thermal Writing Waveguide Technology. Micromachines 2019, 10, 628. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Brandenburg, A.; Henninger, R. Integrated optical Young interferometer. Appl. Opt. 1994, 33, 5941–5947. [Google Scholar] [CrossRef] [PubMed]
- Brandenburg, A.; Krauter, R.; Künzel, C.; Stefan, M.; Schulte, H. Interferometric sensor for detection of surface-bound bioreactions. Appl. Opt. 2000, 39, 6396–6405. [Google Scholar] [CrossRef] [PubMed]
- Schmitt, K.; Schirmer, B.; Hoffmann, C.; Brandenburg, A.; Meyrueis, P. Interferometric biosensor based on planar optical waveguide sensor chips for label-free detection of surface bound bioreactions. Biosens. Bioelectron. 2007, 22, 2591–2597. [Google Scholar] [CrossRef]
- Uusitalo, S.; Känsäkoski, M.; Karkkainen, A.H.O.; Hannu-Kuure, M.; Aikio, S.; Kopola, H. Biosensing with Low-Index Waveguides: An Experimental Study of a Polymer-Based Young Interferometer Sensor. Micro Nanosyst. 2010, 2, 65–69. [Google Scholar] [CrossRef]
- Hiltunen, M.; Hiltunen, J.; Stenberg, P.; Aikio, S.; Kurki, L.; Vahimaa, P.; Karioja, P. Polymeric slot waveguide interferometer for sensor applications. Opt. Express 2014, 22, 7229–7237. [Google Scholar] [CrossRef]
- Luan, E.; Shoman, H.; Ratner, D.M.; Cheung, K.C.; Chrostowski, L. Silicon Photonic Biosensors Using Label-Free Detection. Sensors 2018, 18, 3519. [Google Scholar] [CrossRef] [Green Version]
- Janeiro, R.; Flores, R.; Viegas, J. Silicon photonics waveguide array sensor for selective detection of VOCs at room temperature. Sci. Rep. 2019, 9, 17099. [Google Scholar] [CrossRef] [PubMed]
- Zinoviev, K.; Carrascosa, L.G.; Del Río, J.S.; Sepúlveda, B.; Domínguez, C.; Lechuga, L.M. Silicon Photonic Biosensors for Lab-on-a-Chip Applications. Adv. Opt. Technol. 2008, 2008, 1–6. [Google Scholar] [CrossRef]
- Butt, M.A.; Khonina, S.N.; Kazanskiy, N.L. Hybrid plasmonic waveguide-assisted Metal–Insulator–Metal ring resonator for refractive index sensing. J. Mod. Opt. 2018, 65, 1135–1140. [Google Scholar] [CrossRef]
- Butt, M.A.; Khonina, S.N.; Kazanskiy, N.L. Highly sensitive refractive index sensor based on hybrid plasmonic waveguide microring resonator. Waves Random Complex Media 2018, 30, 292–299. [Google Scholar] [CrossRef]
- Sun, Y.; Fan, X. Optical ring resonators for biochemical and chemical sensing. Anal. Bioanal. Chem. 2010, 399, 205–211. [Google Scholar] [CrossRef] [PubMed]
- Su, J. Label-Free Biological and Chemical Sensing Using Whispering Gallery Mode Optical Resonators: Past, Present, and Future. Sensors 2017, 17, 540. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kazanskiy, N.; Butt, M.; Khonina, S. Silicon photonic devices realized on refractive index engineered subwavelength grating waveguides-A review. Opt. Laser Technol. 2021, 138, 106863. [Google Scholar] [CrossRef]
- Butt, M.A.; Khonina, S.N.; Kazanskiy, N.L. Ultrashort inverted tapered silicon ridge-to-slot waveguide coupler at 155 µm and 3392 µm wavelength. Appl. Opt. 2020, 59, 7821–7828. [Google Scholar] [CrossRef]
- Carlborg, C.F.; Gylfason, K.; Kazmierczak, A.; Dortu, F.; Polo, M.J.B.; Catala, A.M.; Kresbach, G.M.; Sohlström, H.; Moh, T.; Vivien, L.; et al. A packaged optical slot-waveguide ring resonator sensor array for multiplex label-free assays in labs-on-chips. Lab Chip 2010, 10, 281–290. [Google Scholar] [CrossRef] [Green Version]
- A Butt, M.; Khonina, S.N.; Kazanskiy, N.L. A highly sensitive design of subwavelength grating double-slot waveguide microring resonator. Laser Phys. Lett. 2020, 17, 076201. [Google Scholar] [CrossRef]
- Kazanskiy, N.L.; Khonina, S.N.; Butt, M.A. Subwavelength Grating Double Slot Waveguide Racetrack Ring Resonator for Refractive Index Sensing Application. Sensors 2020, 20, 3416. [Google Scholar] [CrossRef]
- Badri, S.H. Transmission resonances in silicon subwavelength grating slot waveguide with functional host material for sensing applications. Opt. Laser Technol. 2021, 136, 106776. [Google Scholar] [CrossRef]
- Flueckiger, J.; Schmidt, S.; Donzella, V.; Sherwali, A.; Ratner, D.M.; Chrostowski, L.; Cheung, K.C. Sub-wavelength grating for enhanced ring resonator biosensor. Opt. Express 2016, 24, 15672–15686. [Google Scholar] [CrossRef] [PubMed]
- Kirk, J.T.; Fridley, G.E.; Chamberlain, J.W.; Christensen, E.D.; Hochberg, M.; Ratner, D.M. Multiplexed inkjet functionalization of silicon photonic biosensors. Lab Chip 2011, 11, 1372–1377. [Google Scholar] [CrossRef] [PubMed]
- Armani, D.; Min, B.; Martin, A.; Vahala, K.J. Electrical thermo-optic tuning of ultrahigh-Q microtoroid resonators. Appl. Phys. Lett. 2004, 85, 5439. [Google Scholar] [CrossRef] [Green Version]
- Van Laere, F.; Roelkens, G.; Ayre, M.; Schrauwen, J.; Taillaert, D.; Van Thourhout, D.; Krauss, T.F.; Baets, R. Compact and Highly Efficient Grating Couplers between Optical Fiber and Nanophotonic Waveguides. J. Light. Technol. 2007, 25, 151–156. [Google Scholar] [CrossRef] [Green Version]
- Maire, G.; Vivien, L.; Sattler, G.; Kazmierczak, A.; Sanchez, B.; Gylfason, K.B.; Griol, A.; Marris-Morini, D.; Cassan, E.; Giannone, D.; et al. High efficiency silicon nitride surface grating couplers. Opt. Express 2008, 16, 328–333. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lukosz, W.; Tiefenthaler, K. Embossing technique for fabricating integrated optical components in hard inorganic waveguiding materials. Opt. Lett. 1983, 8, 537–539. [Google Scholar] [CrossRef] [Green Version]
- Tiefenthaler, K.; Lukosz, W. Sensitivity of grating couplers as integrated-optical chemical sensors. J. Opt. Soc. Am. B 1989, 6, 209–220. [Google Scholar] [CrossRef]
- Vörös, J.; Ramsden, J.; Csúcs, G.; Szendrő, I.; de Paul, S.; Textor, M.; Spencer, N. Optical grating coupler biosensors. Biomaterials 2002, 23, 3699–3710. [Google Scholar] [CrossRef]
- Szendro, I. Art and practice to emboss gratings into sol-gel waveguides. In Proceedings of the Volume 4284, Functional Integration of Opto-Electro-Mechanical Devices and Systems, San Jose, CA, USA, 15 May 2001; Volume 4284, pp. 80–88. [Google Scholar]
- Available online: https://www.owls-sensors.com/owls-system (accessed on 3 April 2021).
- Karasiński, P. Sensor properties of planar waveguide structures with grating couplers. Opto Electron. Rev. 2007, 15, 168–178. [Google Scholar] [CrossRef]
- Karasiński, P. Embossable grating couplers for planar evanescent wave sensors. Opto Electron. Rev. 2011, 19, 10–21. [Google Scholar] [CrossRef]
- Kazmierczak, A.; Slowikowski, M.; Pavlov, K.; Filipiak, M.; Vervaeke, M.; Tyszkiewicz, C.; Ottevaere, H.; Piramidowicz, R.; Karasinski, P. Efficient, low-cost optical coupling mechansim for TiO2-SiO2 sol-gel derived slab waveguide surface grating coupler sensors. Opt. Appl. 2020, 50, 539–549. [Google Scholar]
- Brandenburg, A.; Gombert, A. Grating couplers as chemical sensors: A new optical configuration. Sens. Actuators B Chem. 1993, 17, 35–40. [Google Scholar] [CrossRef]
- Wiki, M.; Gao, H.; Juvet, M.; Kunz, R. Compact integrated optical sensor system. Biosens. Bioelectron. 2001, 16, 37–45. [Google Scholar] [CrossRef]
- Horvath, R.; Pedersen, H.C.; Skivesen, N.; Svanberg, C.; Larsen, N.B. Fabrication of reverse symmetry polymer waveguide sensor chips on nanoporous substrates using dip-floating. J. Micromech. Microengin. 2005, 15, 1260–1264. [Google Scholar] [CrossRef]
- Basu, D.; Sen, K.; Hossain, S.M.; Das, J. Instrumentation and Development of Grating Coupler Sensor for Cost-effective and Precision Measurement of Biomolecules. In Proceedings of the 2019 10th International Conference on Computing, Communication and Networking Technologies (ICCCNT), Kanpur, India, 6–8 July 2019; pp. 1–4. [Google Scholar]
- Li, H.-Y.; Hsu, W.-C.; Liu, K.-C.; Chen, Y.-L.; Chau, L.-K.; Hsieh, S.; Hsieh, W.-H. A low cost, label-free biosensor based on a novel double-sided grating waveguide coupler with sub-surface cavities. Sens. Actuators B Chem. 2015, 206, 371–380. [Google Scholar] [CrossRef]
- Xia, M.; Zhang, P.; Qiao, K.; Bai, Y.; Xie, Y.-H. Coupling SPP with LSPR for Enhanced Field Confinement: A Simulation Study. J. Phys. Chem. C 2015, 120, 527–533. [Google Scholar] [CrossRef] [Green Version]
- Wu, F.; Thomas, P.A.; Kravets, V.G.; Arola, H.O.; Soikkeli, M.; Iljin, K.; Kim, G.; Kim, M.; Shin, H.S.; Andreeva, D.V.; et al. Layered material platform for surface plasmon resonance biosensing. Sci. Rep. 2019, 9, 1–10. [Google Scholar] [CrossRef] [Green Version]
- Reinhard, I.; Miller, K.; Diepenheim, G.; Cantrell, K.; Hall, W.P. Nanoparticle Design Rules for Colorimetric Plasmonic Sensors. ACS Appl. Nano Mater. 2020, 3, 4342–4350. [Google Scholar] [CrossRef]
- Mauriz, E. Clinical applications of visual plasmoni colorimetric sensing. Sensors 2020, 20, 6214. [Google Scholar] [CrossRef]
- Bauch, M.; Toma, K.; Toma, M.; Zhang, Q.; Dostalek, J. Plasmon-Enhanced Fluorescence Biosensors: A Review. Plasmonics 2014, 9, 781–799. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fossati, S.; Hageneder, S.; Menad, S.; Maillart, E.; Dostalek, J. Multiresonant plasmonic nanostructure for ultrasensitive fluorescence biosensing. Nanophotonics 2020, 9, 3673–3685. [Google Scholar] [CrossRef]
- Langer, J.; Jimenez de Aberasturi, D.; Aizpurua, J.; Alvarez-Puebla, R.A.; Auguie, B.; Baumberg, J.J.; Bazan, G.C.; Bell, S.E.J.; Boisen, A.; Brolo, A.G.; et al. Present and Future of Surface Enhanced Raman Scattering. ACS Nano 2020, 14, 28–117. [Google Scholar] [CrossRef] [Green Version]
- Pilot, R.; Signorini, R.; Durante, C.; Orian, L.; Bhamidipati, M.; Fabris, L. A Review on Surface-Enhanced Raman Scattering. Biosensors 2019, 9, 57. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, T.; Dong, Z.; Koay, E.H.H.; Yang, J.K.W. Surface-Enhanced Infrared Absorption Spectroscopy Using Charge Transfer Plasmons. ACS Photon 2019, 6, 1272–1278. [Google Scholar] [CrossRef]
- Pereira, C.F.; Viegas, I.M.A.; Sobrinha, I.G.S.; Pereira, G.; Pereira, G.A.D.L.; Krebs, P.; Mizaikoff, B. Surface-enhanced infrared absorption spectroscopy using silver selenide quantum dots. J. Mater. Chem. C 2020, 8, 10448–10455. [Google Scholar] [CrossRef]
- Lin, L. Manipulation of Near Field Propagation and Far Field Radiation of Surface Plasmon Polariton; Springer: Singapore, 2017. [Google Scholar]
- Tabassum, R.; Mishra, S.K.; Gupta, B.D. Surface plasmon resonance-based fiber optic hydrogen sulphide gas sensor utilizing Cu-ZnO thin films. Phys. Chem. Chem. Phys. 2013, 15, 11868–11874. [Google Scholar] [CrossRef]
- Gupta, B.D.; Verma, R.K. Surface Plasmon Resonance-Based Fiber Optic Sensors: Principle, Probe Designs, and Some Applications. J. Sens. 2009, 2009, 1–12. [Google Scholar] [CrossRef]
- Lu, L.; Jiang, Z.; Hu, Y.; Zhou, H.; Liu, G.; Chen, Y.; Luo, Y.; Chen, Z. A portable optical fiber SPR temperature sensor based on a smart-phone. Opt. Express 2019, 27, 25420–25427. [Google Scholar] [CrossRef]
- Duarte, D.P.; Alberto, N.; Bilro, L.; Nogueira, R. Theoretical Design of a High Sensitivity SPR-Based Optical Fiber Pressure Sensor. J. Light. Technol. 2015, 33, 4606–4611. [Google Scholar] [CrossRef]
- Mi, H.; Wang, Y.; Jin, P.; Lei, L. Design of a ultrahigh-sensitivity SPR-based optical fiber pressure sensor. Optik 2013, 124, 5248–5250. [Google Scholar] [CrossRef]
- Kim, H.-M.; Park, J.-H.; Lee, S.-K. Fabrication and Measurement of Fiber Optic Surface Plasmon Resonance Sensor Based on Polymer Microtip and Gold Nanoparticles Composite. IEEE Sens. J. 2020, 20, 9895–9900. [Google Scholar] [CrossRef]
- Joe, H.-E.; Yun, H.; Jo, S.-H.; Jun, M.B.; Min, B.-K. A review on optical fiber sensors for environmental monitoring. Int. J. Precis. Eng. Manuf. Technol. 2018, 5, 173–191. [Google Scholar] [CrossRef]
- Homola, J. Surface plasmon resonance biosensors for food safety, in Optical Sensors. In Springer Series on Chemical Sensors and Biosensors (Methods and Applications), vol. 1; Springer: Berlin/Heidelberg, Germany, 2004. [Google Scholar]
- Zeni, L.; Perri, C.; Cennamo, N.; Arcadio, F.; D’Agostino, G.; Salmona, M.; Beeg, M.; Gobbi, M. A portable optical-fibre-based surface plasmon resonance biosensor for the detection of therapeutic antibodies in human serum. Sci. Rep. 2020, 10, 1–9. [Google Scholar] [CrossRef]
- Li, D.; Lu, B.; Zhu, R.; Yu, H.; Xu, K. An optofluidic system with volume measurement and surface plasmon resonance sensor for continuous glucose monitoring. Biomicrofluidics 2016, 10, 011913. [Google Scholar] [CrossRef] [Green Version]
- Zainuddin, N.; Ariannejad, M.; Arasu, P.; Harun, S.; Zakaria, R. Investigation of cladding thicknesses on silver SPR based side-polished optical fiber refractive-index sensor. Results Phys. 2019, 13, 102255. [Google Scholar] [CrossRef]
- Caucheteur, C.; Guo, T.; Albert, J. Review of plasmonic fiber optic biochemical sensors: Improving the limit of detection. Anal. Bioanal. Chem. 2015, 407, 3883–3897. [Google Scholar] [CrossRef]
- Mahmood, A.I.; Ibrahim, R.K.; Mahmood, A.I.; Ibrahim, Z.K. Design and simulation of surface plasmon resonance sensors for environmental monitoring. In Journal of Physics: Conference Series; IOP: Bristol, UK, 2018; Volume 1003. [Google Scholar]
- Taylor, A.D.; Ladd, J.; Homola, J.; Jiang, S. Surface plasmon resonance (SPR) sensors for the detection of bacterial pathogens. In Principles of Bacterial Detection:Biosensors, Recognition Receptors and Microsystems; Springer: New York, NY, USA, 2008; pp. 83–108. [Google Scholar]
- Fontana, E.; Dulman, H.; Doggett, D.; Pantell, R. Surface plasmon resonance on a single mode optical fiber. IEEE Trans. Instrum. Meas. 1998, 47, 168–173. [Google Scholar] [CrossRef]
- Mao, P.; Luo, Y.; Chen, X.; Fang, J.; Huang, H.; Chen, C.; Peng, S.; Zhang, J.; Tang, J.; Lu, H.; et al. Design and optimization of multimode fiber sensor based on surface plasmon resonance. In Proceedings of the Numerical Simulation of Optoelectronic Devices, Palma de Mallorca, Spain, 1–4 September 2014; pp. 119–120. [Google Scholar]
- Hoseinian, M.S.; Bolorizadeh, M.A. Design and Simulation of a Highly Sensitive SPR Optical Fiber Sensor. Photon Sens. 2018, 9, 33–42. [Google Scholar] [CrossRef] [Green Version]
- Xie, T.; He, Y.; Yang, Y.; Zhang, H.; Xu, Y. Highly Sensitive Surface Plasmon Resonance Sensor Based on Graphene-Coated U-shaped Fiber. Plasmonics 2021, 16, 205–213. [Google Scholar] [CrossRef]
- Verma, R.K.; Gupta, B.D. Theoretical modelling of a bi-dimensional U-shaped surface plasmon resonance based fiber optic sensor for sensitivity enhancement. J. Phys. D Appl. Phys. 2008, 41, 095106. [Google Scholar] [CrossRef]
- Yu, H.; Chong, Y.; Zhang, P.; Ma, J.; Li, D. A D-shaped fiber SPR sensor with a composite nanostructure of MoS2-graphene for glucose detection. Talanta 2020, 219, 121324. [Google Scholar] [CrossRef] [PubMed]
- Arasu, P.T.; Al-Qazwini, Y.; Onn, B.I.; Noor, A.S.M. Fiber Bragg grating based surface plasmon resonance sensor utilizing FDTD for alcohol detection applications. In Proceedings of the 2012 IEEE 3rd International Conference on Photonics, Pulau Pinang, Malaysia, 1–3 October 2012; pp. 93–97. [Google Scholar]
- Kim, Y.-C.; Peng, W.; Banerji, S.; Booksh, K.S. Tapered fiber optic surface plasmon resonance sensor for analyses of vapor and liquid phases. Opt. Lett. 2005, 30, 2218–2220. [Google Scholar] [CrossRef] [PubMed]
- Navarrete, M.-C.; Díaz-Herrera, N.; González-Cano, A.; Esteban, Ó. Surface plasmon resonance in the visible region in sensors based on tapered optical fibers. Sens. Actuators B Chem. 2014, 190, 881–885. [Google Scholar] [CrossRef]
- Aray, A.; Chiavaioli, F.; Arjmand, M.; Trono, C.; Tombelli, S.; Giannetti, A.; Cennamo, N.; Soltanolkotabi, M.; Zeni, L.; Baldini, F. SPR-based plastic optical fibre biosensor for the detection of C-reactive protein in serum. J. Biophotonics 2016, 9, 1077–1084. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Mai, Z.; Chen, Y.; Wang, J.; Li, L.; Su, Q.; Li, X.; Hong, X. A label-free fiber optic SPR biosensor for specific detection of C-reactive protein. Sci. Rep. 2017, 7, 1–8. [Google Scholar] [CrossRef]
- Tran, D.T.; Knez, K.; Janssen, K.P.; Pollet, J.; Spasic, D.; Lammertyn, J. Selection of aptamers against Ara h 1 protein for FO-SPR biosensing of peanut allergens in food matrices. Biosens. Bioelectron. 2013, 43, 245–251. [Google Scholar] [CrossRef]
- Luo, B.; Yan, Z.; Sun, Z.; Liu, Y.; Zhao, M.; Zhang, L. Biosensor based on excessively titled fiber grating in thin-cladding optical fiber for sensitive and selective detection of low glucose concentration. Opt. Express 2015, 23, 32429–32440. [Google Scholar] [CrossRef]
- Lu, J.; Van Stappen, T.; Spasic, D.; Delport, F.; Vermeire, S.; Gils, A.; Lammertyn, J. Fiber optic-SPR platform for fast and sensitive in fliximab detection in serum of inflammatory bowel disease patients. Biosens. Bioelectron. 2016, 79, 173–179. [Google Scholar] [CrossRef] [Green Version]
- Yeh, P.; Yariv, A.; Marom, E. Theory of Bragg fiber. J. Opt. Soc. Am. 1978, 68, 1196–1201. [Google Scholar] [CrossRef]
- Knight, J.C.; Birks, T.A.; Russell, P.S.J.; Atkin, D.M. All-silica single-mode optical fiber with photonic crystal cladding. Opt. Lett. 1996, 21, 1547–1549. [Google Scholar] [CrossRef]
- Pinto, A.M.R.; Lopez-Amo, M. Photonic Crystal Fibers for Sensing Applications. J. Sens. 2012, 2012, 1–21. [Google Scholar] [CrossRef] [Green Version]
- Kim, J.A.; Hwang, T.; Dugasani, S.R.; Amin, R.; Kulkarni, A.; Park, S.H.; Kim, T. Graphene based fiber optic surface plasmon resonance for bio-chemical sensor applications. Sens. Actuator B Chem. 2013, 187, 426–433. [Google Scholar] [CrossRef] [Green Version]
- Xu, H.; Wu, L.; Dai, X.; Gao, Y.; Xiang, Y. An ultra-high sensitivity surface plasmon resonance sensor based on graphene-aluminum-graphene sandwich-like structure. J. Appl. Phys. 2016, 120, 053101. [Google Scholar] [CrossRef]
- Lin, C.; Chen, S. Design of high-performance Au-Ag-dielectric-graphene based surface plasmon resonance biosensors using genetic algorithm. J. Appl. Phys. 2019, 125, 113101. [Google Scholar] [CrossRef]
- Chen, S.; Lin, C. Sensitivity comparison of graphene based surface plasmon resonance biosensor with Au, Ag and Cu in the visible region. Mater. Res. Express 2019, 6, 056503. [Google Scholar] [CrossRef]
- Shuai, B.; Xia, L.; Liu, D. Coexistence of positive and negative refractive index sensitivity in the liquid-core photonic crystal fiber based plasmonic sensor. Opt. Express 2012, 20, 25858–25866. [Google Scholar] [CrossRef] [PubMed]
- Mollah, A.; Islam, S.R.; Yousufali; Abdulrazak, L.F.; Hossain, M.B.; Amiri, I. Plasmonic temperature sensor using D-shaped photonic crystal fiber. Results Phys. 2020, 16, 102966. [Google Scholar] [CrossRef]
- Chen, N.; Chang, M.; Lu, X.; Zhou, J.; Zhang, X. Numerical Analysis of Midinfrared D-Shaped Photonic-Crystal-Fiber Sensor Based on Surface-Plasmon-Resonance Effect for Environmental Monitoring. Appl. Sci. 2020, 10, 3897. [Google Scholar] [CrossRef]
- Liu, C.; Su, W.; Liu, Q.; Lu, X.; Wang, F.; Sun, T.; Chu, P.K. Symmetrical dual D-shape photonic crystal fibers for surface plasmon resonance sensing. Opt. Express 2018, 26, 9039–9049. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Xie, Q.; Li, X.; Zhou, H.; Hong, X.; Geng, Y. Experimental realization of D-shaped photonic crystal fiber SPR sensor. J. Phys. D Appl. Phys. 2017, 50, 025101. [Google Scholar] [CrossRef]
- Yasli, A.; Ademgil, H.; Haxha, S. D-shaped photonic crystal fiber based surface plasmon resonance sensor. In Proceedings of the 26th Signal Processing and Communications Applications Conference (SIU), Izmir, Turkey, 2–5 May 2018. [Google Scholar]
- Lu, Y.; Yang, X.; Wang, M.; Yao, J. Surface plasmon resonance sensor based on hollow-core PCFs filled with silver nanowires. Electron. Lett. 2015, 51, 1675–1677. [Google Scholar] [CrossRef]
- Lu, Y.; Wang, M.; Hao, C.; Zhao, Z.; Yao, J. Temperature sensing using photonic crystal fiber filled with silver nanowires and liquid. Photonics J. IEEE 2014, 6, 1–7. [Google Scholar] [CrossRef]
- Semenova, D.; Gernaey, K.V.; Silina, Y.E. Exploring the potential of electroless and electroplated noble metal-semiconductor hybrids within bio- and environmental sensing. Analyst 2018, 143, 5646–5669. [Google Scholar] [CrossRef]
- Malinsky, P.; Slepicka, P.; Hnatowicz, V.; Svorcik, V. Early stages of growth of gold layers sputter deposited on glass and silicon substrates. Nanoscale Res. Lett. 2012, 7, 241. [Google Scholar] [CrossRef] [Green Version]
- Svorcik, V.; Siegel, J.; Sutta, P.; Mistrik, J.; Janicek, P.; Worsch, P.; Kolska, Z. Annealing of gold nanostructures sputtered on glass substrate. Appl. Phys. A 2011, 102, 605. [Google Scholar]
- Sennett, R.S.; Scott, G.D. The structure of evaporated metal films and their optical properties. J. Opt. Soc. Am. A Opt. Image Sci. Vis. 1950, 40, 203. [Google Scholar] [CrossRef]
- Khurgin, J.B. Replacing noble metals with alternative materials in plasmonics and metamaterials:How good an idea? Phil. Trans. R. Soc. A 2017, 375, 20160068. [Google Scholar] [CrossRef]
- Naik, G.V.; Shalaev, V.M.; Boltasseva, A. Alternative plasmonic materials:beyond gold and silver. Adv. Mater. 2013, 25, 3264–3294. [Google Scholar] [CrossRef]
- Shao, L.; Liu, Z.; Hu, J.; Gunawardena, D.; Tam, H.-Y. Optofluidics in microstructured optical fibers. Micromachines 2018, 9, 145. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Irawan, R.; Chuan, T.S.; Meng, T.C.; Ming, T.K. Rapid constructions of microstructures for optical fiber sensors using a commercial CO2 laser system. Open Biomed. Eng. J. 2008, 2, 28–35. [Google Scholar] [CrossRef] [PubMed]
- Argyros, A. Microstructures in polymer fibers for optical fibres, THz waveguides, and fibre-based metamaterials. Int. Sch. Res. Not. 2013, 2013, 785162. [Google Scholar]
- Rindorf, L.; Hoiby, P.E.; Jensen, J.B.; Pedersen, L.H.; Bang, O.; Geschke, O. Towards biochips using microstructured optical fiber sensors. Anal. Bioanal. Chem. 2006, 385, 1370–1375. [Google Scholar] [CrossRef] [PubMed]
- Lu, H.; Wang, G.; Liu, X. Manipulation of light in MIM plasmonic waveguide systems. Chin. Sci. Bull. 2013, 58, 3607–3616. [Google Scholar] [CrossRef] [Green Version]
- Hu, H.; Zeng, X.; Wang, L.; Xu, Y.; Song, G.; Gan, Q. Surface plasmon coupling efficiency from nanoslit apertures to metal-insulator-metal waveguides. Appl. Phys. Lett. 2012, 101, 121112. [Google Scholar] [CrossRef]
- Butt, M.A.; Khonina, S.N.; Kazanskiy, N.L. Ultra-short lossless plasmonic power splitter design based on metal-insulator-metal waveguide. Laser Phys. 2019, 30, 016201. [Google Scholar] [CrossRef]
- Hill, M.T.; Marell, M.; Leong, E.; Smalbrugge, B.; Zhu, Y.; Sun, M.; van Veldhoven, P.J.; Geluk, E.J.; Karouta, F.; Oei, Y.-S.; et al. Lasing in metal-insulator-metal sub-wavelength plasmonic waveguides. Opt. Express 2009, 17, 11107–11112. [Google Scholar] [CrossRef] [Green Version]
- Yu, Y.; Si, J.; Ning, Y.; Sun, M.; Deng, X. Plasmonic wavelength splitter based on a metal-insulator-metal waveguide with a graded grating coupler. Opt. Lett. 2017, 42, 187–190. [Google Scholar] [CrossRef]
- Khani, S.; Danaie, M.; Rezaei, P. Siza reduction of MIM surface plasmon based optical bandpass filters by the introduction of arrays of silver nano-rods. Phys. E Low Dimens. Syst. Nanostruct. 2019, 113, 25–34. [Google Scholar] [CrossRef]
- Butt, M.A. Numerical investigation of a small footprint plasmonic Bragg grating structure with a high extinction ratio. Photon. Lett. Pol. 2020, 12, 82–84. [Google Scholar] [CrossRef]
- Butt, M.A.; Khonina, S.N.; Kazanskiy, N.L. An array of nano-dots loaded MIM square ring resonator with enhanced sensitivity at NIR wavelength range. Optik 2020, 202, 163655. [Google Scholar] [CrossRef]
- Butt, M.A.A.; Kazanskiy, N. Enhancing the sensitivity of a standard plasmonic MIM square ring resonator by incorporating the Nano-dots in the cavity. Photon. Lett. Pol. 2020, 12, 1–3. [Google Scholar] [CrossRef]
- Butt, M.A.; Khonina, S.N.; Kazanskiy, N.L. Metal-insulator-metal nano square ring resonator for gas sensing applications. Waves Random Complex Media 2021, 31, 146–156. [Google Scholar] [CrossRef]
- A Butt, M.; Kazanskiy, N.L.; Khonina, S.N. Nanodots decorated asymmetric metal–insulator–metal waveguide resonator structure based on Fano resonances for refractive index sensing application. Laser Phys. 2020, 30, 076204. [Google Scholar] [CrossRef]
- Butt, M.A.; Khonina, S.N.; Kazanskiy, N.L. A plasmonic colour filter and refractive index sensor applications based on metal-insulator-metal square micro-ring cavities. Laser Phys. 2020, 30, 016205. [Google Scholar] [CrossRef]
- Butt, M.A.; Kazanskiy, N.L.; Khonina, S.N. Highly Sensitive Refractive Index Sensor Based on Plasmonic Bow Tie Configuration. Photon. Sens. 2020, 10, 223–232. [Google Scholar] [CrossRef]
- Butt, M.A.; Khonina, S.N.; Kazanskiy, N.L. Plasmonic refractive index sensor based on M-I-M square ring resonator. In Proceedings of the International Conference on Computing, Electronic and Electrical Engineering (ICE Cube), Quetta, Pakistan, 12–13 November 2018. [Google Scholar]
- Barizuddin, S.; Bok, S.; Gangopadhyay, S. Plasmonic sensors for disease detection-A review. Nanomed. Nanotechnol. 2016, 7, 1000373. [Google Scholar]
- A Butt, M.; Khonina, S.N.; Kazanskiy, N.L. A multichannel metallic dual nano-wall square split-ring resonator: Design analysis and applications. Laser Phys. Lett. 2019, 16, 126201. [Google Scholar] [CrossRef]
- Behnam, M.A.; Emami, F.; Sobhani, Z.; Koohi-Hosseinabadi, O.; Dehghanian, A.R.; Zebarjad, S.M.; Moghim, M.H.; Oryan, A. Novel Combination of Silver Nanoparticles and Carbon Nanotubes for Plasmonic Photo Thermal Therapy in Melanoma Cancer Model. Adv. Pharm. Bull. 2018, 8, 49–55. [Google Scholar] [CrossRef] [Green Version]
- Sagor, R.H.; Hassan, F.; Sharmin, S.; Adry, T.Z.; Emon, A.R. Numerical investigation of an optimized plasmonic on-chip refractive index sensor for temperature and blood group detection. Results Phys. 2020, 19, 103611. [Google Scholar] [CrossRef]
- Rakhshani, M.R. Optical refractive index sensor with two plasmonic double-square resonators for simultaneous sensing of human blood groups. Photon. Nanostruct. Fundam. Appl. 2020, 39, 100768. [Google Scholar] [CrossRef]
- Nejat, M.; Nozhat, N. Multi-band MIM refractive index biosensor based on Ag-air grating with equivalent circuit and T-matrix methods in near-infrared region. Sci. Rep. 2020, 10, 1–12. [Google Scholar] [CrossRef]
- Rakhshani, M.R. Refractive index sensor based on concentric triple racetrack resonators side-coupled to metal–insulator–metal waveguide for glucose sensing. J. Opt. Soc. Am. B 2019, 36, 2834–2842. [Google Scholar] [CrossRef]
- Liu, D.; Wang, J.; Zhang, F.; Pan, Y.; Lu, J.; Ni, X. Tunable Plasmonic Band-Pass Filter with Dual Side-Coupled Circular Ring Resonators. Sensors 2017, 17, 585. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rahmatiyar, M.; Afsahi, M. Design of a refractive index plasmonic sensor based on a ring resonator coupled to a MIM waveguide containing tapered defects. Plasmonics 2020, 15, 2169–2176. [Google Scholar] [CrossRef]
- Jankovic, N.; Cselyuszka, N. Multiple Fano-like MIM plasmonic structure based on triangular resonator for refractive index sensing. Sensors 2018, 18, 287. [Google Scholar]
- Chau, Y.F.C.; Chao, C.T.C.; Huang, H.J.; Kumara, N.; Lim, C.M.; Chiang, H.P. Ultra-high refractive index sensing structure based on a metal-insulator-metal waveguide-coupled T-shaped cavity with metal nanorod defects. Nanomaterials 2019, 9, 1433. [Google Scholar]
- Kamada, S.; Okamoto, T.; El-Zohary, S.E.; Haraguchi, M. Design optimization and fabrication of Mach-Zehnder interferometer based on MIM plasmonic waveguides. Opt. Express 2016, 24, 16224–16231. [Google Scholar] [CrossRef]
- Butt, M.A.; Kazanskiy, N.L. Nanoblocks embedded in L-shaped nanocavity of a plasmonic sensor for best sensor performance. Opt. Appl. 2021, 51, 109–120. [Google Scholar]
- Cao, J.; Sun, T.; Grattan, K.T. Gold nanorod-based localized surface plasmon resonance biosensors:a review. Sens. Actuators B Chem. 2014, 195, 332–351. [Google Scholar] [CrossRef]
- Masson, J.F.; Live, L.S.; Murray-Methot, M.P. Nanohole arrays in chemical analysis:manufacturing methods and applications. Analyst 2010, 135, 1483–1489. [Google Scholar] [CrossRef]
- Kazanskiy, N.L.; Butt, M.A.; Degtyarev, S.A.; Khonina, S.N. Achievements in the development of plasmonic waveguide sensors for measuring the refractive index. Comput. Opt. 2020, 44, 295–318. [Google Scholar] [CrossRef]
- Kazanskiy, N.L.; Khonina, S.N.; Butt, M.A. Plasmonic sensors based on Metal-insulator-Metal waveguides for refractive index sensing applications: A brief review. Phys. E 2020, 117, 113798. [Google Scholar] [CrossRef]
- Veselago, V.G. Experimental demonstration of negative index of refraction. Sov. Phys. Usp. 1968, 10, 509. [Google Scholar]
- Pendry, J.B.; Holden, A.J.; Stewart, W.J.; Youngs, I. Extremely low frequency plasmons in metallic mesostructures. Phys. Rev. Lett. 1996, 76, 4773–4776. [Google Scholar] [CrossRef] [Green Version]
- Smith, D.R.; Padilla, W.J.; Vier, D.C.; Nemat-Nasser, S.C.; Schultz, S. Composite medium with simultaneously negative permeability and permittivity. Phys. Rev. Lett. 2000, 84, 4184–4187. [Google Scholar] [CrossRef] [Green Version]
- Cen, C.; Chen, Z.; Xu, D.; Jiang, L.; Chen, X.; Yi, Z.; Wu, P.; Li, G.; Yi, Y. High quality factor, high sensitivity metamaterial graphene-perfect absorber based on critical coupling theory and impedance matching. Nanomaterials 2020, 10, 95. [Google Scholar] [CrossRef] [Green Version]
- Butt, M.A.; Kazanskiy, N.L. Narrowband perfect metasurface absorber based on impedance matching. Photonics Lett. Poland 2020, 12, 88–90. [Google Scholar] [CrossRef]
- Wu, Z.; Kelp, G.; Yogeesh, M.N.; Li, W.; McNicholas, K.M.; Briggs, A.; Rajeeva, B.B.; Akinwande, D.; Bank, S.R.; Shvets, G.; et al. Dual-band moire metasurface patches for multifunctional biomedical applications. Nanoscale 2016, 8, 18461–18468. [Google Scholar] [CrossRef]
- Kazanskiy, N.L.; Butt, M.A.; Khonina, S.N. Carbon dioxide gas sensor based on polyhexamethylene biguanide polymer deposited on silicon nano-cylinders metasurface. Sensors 2021, 21, 378. [Google Scholar] [CrossRef]
- Meinzer, N.; Barnes, W.L.; Hooper, I.R. Plasmonic meta-atoms and metasurfaces. Nat. Photonics 2014, 8, 889–898. [Google Scholar] [CrossRef] [Green Version]
- Ding, J.; Xu, N.; Ren, H.; Lin, Y.; Zhang, W.; Zhang, H. Dual-wavelength terahertz metasurfaces with independent phase and amplitude control at each wavelength. Sci. Rep. 2016, 6, 34020. [Google Scholar] [CrossRef]
- Ahmadivand, A.; Gerislioglu, B.; Manickam, P.; Kaushik, A.; Bhansali, S.; Nair, M.; Pala, N. Rapid detection of infectious envelope proteins by magnetoplasmonic toroidal metasensors. ACS Sens. 2017, 2, 1359–1368. [Google Scholar] [CrossRef] [PubMed]
- Yesilkoy, F.; Arvelo, E.R.; Jahani, Y.; Liu, M.; Tittl, A.; Cevher, V.; Kivshar, Y.; Altug, H. Ultrasensitive hyperspectral imaging and biodetection enabled by dielectric metasurfaces. Nat. Photonics 2019, 13, 390–396. [Google Scholar] [CrossRef] [Green Version]
- Zhang, S.; Wong, C.L.; Zeng, S.; Bi, R.; Tai, K.; Dholakia, K.; Olivo, M. Metasurfaces for biomedical applications:imaging and sensing from a nanophotonics perspective. Nanophotonics 2020, 10, 259–293. [Google Scholar] [CrossRef]
- Serita, K.; Murakami, H.; Kawayama, I.; Tonouchi, M. A terahertz-microfluidic chip with a few arrays of asymmetric meta-atoms for the ultra-trace sensing of solutions. Photonics 2019, 6, 12. [Google Scholar] [CrossRef] [Green Version]
- Rodionov, S.A.; Remnev, M.A.; Klimov, V.V. Refractive index sensor based on all-dielectric gradient metasurface. Sens. Bio Sens. Res. 2019, 22, 100263. [Google Scholar] [CrossRef]
- Pahlevaninezhad, H.; Khorasaninejad, M.; Huang, Y.W.; Shi, Z.; Hariri, L.P.; Adams, D.C.; Ding, V.; Zhu, A.; Qiu, C.W.; Capasso, F.; et al. Nano-optic endoscope for high-resolution optical coherence tomography in vivo. Nat. Photonics 2018, 12, 540–547. [Google Scholar] [CrossRef]
- Chen, W.T.; Zhu, A.Y.; Capasso, F. Flat optics with dispersion-engineered metasurfaces. Nat. Rev. Mater. 2020, 5, 604–620. [Google Scholar] [CrossRef]
- Yu, N.; Capasso, F. Flat optics with designer metasurfaces. Nat. Mater. 2014, 13, 139–150. [Google Scholar] [CrossRef]
- Wu, P.C. Flat optics with nanophotonic metasurface. In JSAP-OSA Joint Symposia; OSA Publisher: Hokkaido, Japan, 2019. [Google Scholar]
- Lee, D.; Gwak, J.; Badloe, T.; Palomba, S.; Rho, J. Metasurfaces-based imaging and applications:from miniaturized optical components to functional imaging platforms. Nanoscale Adv. 2020, 2, 605–625. [Google Scholar] [CrossRef] [Green Version]
- Sung, J.; Lee, G.Y.; Lee, B. Progresses in the practical metasurface for holography and lens. Nanophotonics 2019, 8, 1701–1718. [Google Scholar] [CrossRef] [Green Version]
- Wang, Y.; Ali, M.A.; Chow, E.C.; Dong, L.; Lu, M. An optofluidic metasurface for lateral flow-through detection of breast cancer biomarker. Biosens. Bioelectron. 2018, 107, 224–229. [Google Scholar] [CrossRef]
- Geng, Z.; Zhang, X.; Fan, Z.; Lv, X.; Chen, H. A route to terahertz metamaterial biosensor integrated with microfluidics for liver cancer biomarker testing in early stage. Sci. Rep. 2017, 7, 16378. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yan, X.; Yang, M.; Zhang, Z.; Liang, L.; Wei, D.; Wang, M.; Zhang, M.; Wang, T.; Liu, L.; Xie, J.; et al. The terahertz electromagnetically induced transparency-like metamaterials for sensitive biosensors in the detection of cancer cells. Biosens. Bioelectron. 2019, 126, 485–492. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Zhao, C.; Wang, J.; Luo, X.; Xie, L.; Zhan, S.; Kim, J.; Wang, X.; Liu, X.; Ying, Y. Wearable plasmonic-metasurface sensor for noninvasive and universal molecular fingerprint detection on biointerfaces. Sci. Adv. 2021, 7, eabe4553. [Google Scholar]
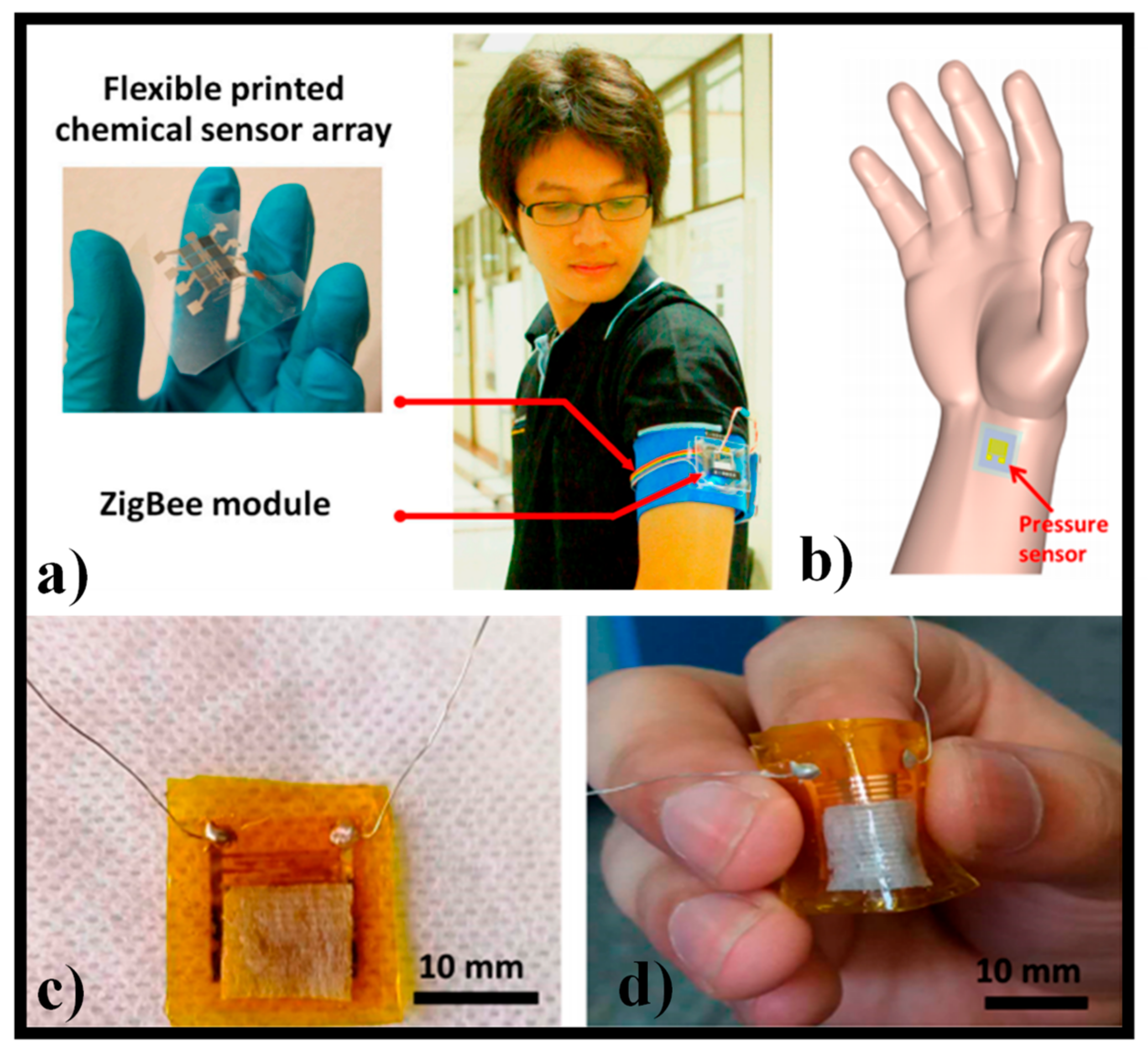
- Lorwongtragool, P.; Sowade, E.; Watthanawisuth, N.; Baumann, R.R.; Kerdcharoen, T. A novel wearable electronic nose for healthcare based on flexible printed chemical sensor array. Sensors 2014, 14, 19700–19712. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhan, Z.; Lin, R.; Tran, V.-T.; An, J.; Wei, Y.; Du, H.; Tran, T.; Lu, W. Paper/carbon nanotube-based wearable pressure sensor for physiological signal acquisition and soft robotic skin. ACS Appl. Mater. Interfaces 2017, 9, 37921–37928. [Google Scholar] [CrossRef]
- Chaghamirzaei, P.; Raeyani, D.; Khosravi, A.; Allahveisi, S.; Abdollahi-Kai, B.; Bayat, F.; Olyaeefar, B.; Ahmadi-Kandjani, S. Real-time detection of gas and chemical vapor flows by silica inverse-opals. IEEE Sens. J. 2019, 19, 7961–7967. [Google Scholar] [CrossRef]
- Pakchin, P.S.; Fathi, M.; Ghanbari, H.; Saber, R.; Omidi, Y. A novel electrochemical immunosensor for ultrasensitive detection of CA125 in ovarian cancer. Biosens. Bioelectron. 2020, 153, 112029. [Google Scholar]
- Xia, L.; Song, J.; Xu, R.; Liu, D.; Dong, B.; Xu, L.; Song, H. Zinc oxide inverse opal electrodes modified by glucose oxidase for electrochemical and photoelectrochemical biosensor. Biosens. Bioelectron. 2014, 59, 350–357. [Google Scholar] [CrossRef] [PubMed]
- Yablonovitch, E. Inhibited spontaneous emission in solid-state physics and electronics. Phys. Rev. Lett. 1987, 58, 2059. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- John, S. Strong localization of photons in certain disordered dielectric superlattices. Phys. Rev. Lett. 1987, 58, 2486. [Google Scholar] [CrossRef] [Green Version]
- Zhang, Y.; Li, B. Photonic crystal-based bending waveguides for optical interconnections. Opt. Express 2006, 14, 5723–5732. [Google Scholar] [CrossRef] [PubMed]
- Butt, M.A.; Khonina, S.N.; Kazanskiy, N.L. 2D-Photonic crystal heterostructures for the realization of compact photonic devices. Photon. Nanostruct. Fundam. Appl. 2021, 44, 100903. [Google Scholar] [CrossRef]
- Butt, M.A.; Kazanskiy, N.L. Two-dimensional photonic crystal heterostructure for light steering and TM-polarization maintaining applications. Laser Phys. 2021, 31, 036201. [Google Scholar] [CrossRef]
- Liu, B.; Liu, Y.F.; Jia, C.; He, X.D. All-optical diode structure based on asymmetrical coupling by a micro-cavity and FP cavity at two sides of photonic crystal waveguide. AIP Adv. 2016, 6, 065316. [Google Scholar] [CrossRef] [Green Version]
- Notomi, M. Negative refraction in photonic crystals. Opt. Quantum Electron. 2002, 34, 133–143. [Google Scholar] [CrossRef]
- Noori, M.; Soroosh, M.; Baghban, H. Self-Collimation in Photonic Crystals: Applications and Opportunities. Ann. Der Phys. 2018, 530, 1700049. [Google Scholar] [CrossRef] [Green Version]
- Nishijima, Y.; Ueno, K.; Juodkazis, S.; Mizeikis, V.; Misawa, H.; Tanimura, T.; Maeda, K. Inverse silica opal photonic crystals for optical sensing applications. Opt. Express 2007, 15, 12979–12988. [Google Scholar] [CrossRef] [PubMed]
- Cunningham, B.; Zhang, M.; Zhuo, Y.; Kwon, L.; Race, C. Review of recent advances in biosensing with photonic crystals. IEEE Sens. J. 2014, 16, 3349–3366. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rodriguez, G.A.; Markov, P.; Cartwright, A.P.; Choudhury, M.H.; Afzal, F.O.; Cao, T.; Halimi, S.I.; Retterer, S.T.; Kravchenko, I.I.; Weiss, S.M. Photonic crystal nanobeam biosensors based on porous silicon. Opt. Express 2019, 27, 9536–9549. [Google Scholar] [CrossRef]
- Fathi, F.; Rashidi, M.R.; Pakchin, P.S.; Ahmadi-Kandjani, S.; Nikniazi, A. Photonic crystal based biosensors: Emerging inverse opals for biomarker detection. Talanta 2021, 221, 121615. [Google Scholar] [CrossRef] [PubMed]
- Lee, W.S.; Kang, T.; Kim, S.H.; Jeong, J. An antibody-immobilized silica inverse opal nanostructure for label-free optical biosensors. Sensors 2018, 18, 307. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, J.; Zhao, X.; Wei, H.; Gu, Z.Z.; Lu, Z. Macroporous ordered titanium dioxide (TiO2) inverse opal as a new label-free immunosensor. Anal. Chim. Acta 2008, 625, 63–69. [Google Scholar] [CrossRef]
- Shen, W.; Li, M.; Wang, B.; Liu, J.; Li, Z.; Jiang, L.; Song, Y. Hierarchical optical antenna: Gold nanoparticle-modified photonic crystal for highly-sensitive label free DNA detection. In J. Mater. Chem.; 2012; Volume 22, pp. 8127–8133. [Google Scholar]
- Feng, X.; Xu, J.; Liu, Y.; Zhao, W. Visual sensors of an inverse opal hydrogel for the colorimetric detection of glucose. J. Mater. Chem. B 2019, 7, 3576–3581. [Google Scholar] [CrossRef]
- Zhao, X.; Xue, J.; Mu, Z.; Huang, Y.; Lu, M.; Gu, Z. Gold nanoparticle incorporated inverse opal photonic crystal capillaries for optofluidic surface enhanced Raman spectroscopy. Biosens. Bioelectron. 2015, 72, 268–274. [Google Scholar] [CrossRef]
- Jiang, Y.; Liu, D.; Yang, Y.; Xu, R.; Zhang, T.; Sheng, K.; Song, H. Photoelectrochemical detection of alpha-fetoprotein based on ZnO inverse opals structure electrodes modified by Ag2S nanoparticles. Sci. Rep. 2016, 6, 38400. [Google Scholar] [CrossRef] [PubMed]
- Choi, E.; Choi, Y.; Nejad, Y.H.; Shin, K.; Park, J. Label-free specific detection of immunoglobulin G antibody using nanoporous hydrogel photonic crystals. Sens. Actuators B Chem. 2013, 180, 107–113. [Google Scholar] [CrossRef]
- Inci, F.; Tokel, O.; Wang, S.; Gurkan, U.A.; Tasoglu, S.; Kuritzkes, D.R.; Demirci, U. Nanoplasmonic quantitative detection of intact viruses from unprocessed whole blood. ACS Nano 2013, 7, 4733–4745. [Google Scholar] [CrossRef] [PubMed]
- Shafiee, H.; Jahangir, M.; Inci, F.; Wang, S.; Willenbrecht, R.B.M.; Giguel, F.F.; Tsibris, A.M.N.; Kuritzkes, D.R.; Demirci, U. Acute On-Chip HIV Detection Through Label-Free Electrical Sensing of Viral Nano-Lysate. Small 2013, 9, 2553–2563. [Google Scholar] [CrossRef] [Green Version]
- Shamah, S.M.; Cunningham, B.T. Label-free cell-based assays using photonic crystal optical biosensors. Analyst 2011, 136, 12. [Google Scholar] [CrossRef]
- Endo, T.; Ozawa, S.; Okuda, N.; Yanagida, Y.; Tanaka, S.; Hatsuzawa, T. Reflectometric detection of influenza virus in human saliva using nanoimprint lithography-based flexible two-dimensional photonic crystal biosensor. Sens. Actuator B Chem. 2010, 148, 269–276. [Google Scholar] [CrossRef]
- Shafiee, H.; Lidstone, E.A.; Jahangir, M.; Inci, F.; Hanhauser, E.; Henrich, T.J.; Kuritzkes, D.R.; Cunningham, B.T.; Demirci, U. Nanostructured Optical Photonic Crystal Biosensor for HIV Viral Load Measurement. Sci. Rep. 2014, 4, 4116. [Google Scholar] [CrossRef] [Green Version]
- Holzgrafe, J.; Sinclair, N.; Zhu, D.; Shams-Ansari, A.; Colangelo, M.; Hu, Y.; Zhang, M.; Berggren, K.K.; Loncar, M. Cavity electro-optics in thin-film lithium niobate for efficient microwave-to-optical transduction. Optica 2020, 7, 1714–1720. [Google Scholar] [CrossRef]
- Chang, M.S.; Burlamacchi, P.; Hu, C.; Whinnery, J.R. Light amplification in a thin film. Appl. Phys. Lett. 1972, 20, 313. [Google Scholar]
- Nikoleli, G.P.; Nikolelis, D.P.; Siontorou, C.G.; Nikolelis, M.T.; Karapetis, S. The application of lipid membranes in biosensing. Membranes 2018, 8, 108. [Google Scholar] [CrossRef] [Green Version]
- Nikoleli, G.P.; Nikolelis, D.P.; Siontorou, C.G.; Karapetis, S.; Nikolelis, M.T. Application of biosensors based on lipid membranes for the rapid detection of toxins. Biosensors 2018, 8, 61. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mueller, P.; Rudin, D.O. Action potentials induced in biomolecular lipid membranes. Nature 1968, 217, 713. [Google Scholar] [CrossRef]
- Sugawara, M.; Kojima, K.; Sazawa, H.; Umezawa, Y. Ion-channel sensors. Anal Chem. 1987, 59, 2842–2846. [Google Scholar] [CrossRef]
- Holden, M.A.; Needham, D.; Bayley, H. Functional biometworks from nanoliter water droplets. J. Am. Chem. Soc. 2007, 129, 8650–8655. [Google Scholar] [CrossRef]
- Zhong, X.B.; Reynolds, R.; Kidd, J.R.; Kidd, K.K.; Jenison, R.; Marlar, R.A.; Ward, D.C. Single-nucleotide polymorphism genotyping on optical thin-film biosensor chips. Proc. Natl. Acad. Sci. USA 2003, 100, 11559–11564. [Google Scholar] [CrossRef] [Green Version]
- Ceylan, K.H.; Kulah, H.; Ozgen, C. Thin film biosensors. In Thin Films and Coatings in Biology. Biological and Medical Physics, Biomedical Engineering; Springer: Dordrecht, The Netherlands, 2013. [Google Scholar]
- Sasaki, T.; Kasai, H.; Nishibori, E. Tightly binding valence electron in aluminum observed through X-ray charge density study. Sci. Rep. 2018, 8, 11964. [Google Scholar] [CrossRef]
- Tseng, M.L.; Yang, J.; Semmlinger, M.; Zhang, C.; Nordlander, P.; Halas, N.J. Two-dimensional active tuning of an aluminum plasmonic array for full-spectrum response. Nano Lett. 2017, 17, 6034–6039. [Google Scholar] [CrossRef] [PubMed]
- Lambert, A.S.; Valiulis, S.N.; Malinick, A.S.; Tanabe, I.; Cheng, Q. Plasmonic biosensing with aluminum thin films under the Kretschmann configuration. Anal. Chem. 2020, 92, 8654–8659. [Google Scholar] [CrossRef] [PubMed]
- Handoyo, T.; Kondoh, J. Development of gold thin-film for optical-based biosensor. In AIP Conference Proceedings; AIP Publishing: College Park, MD, USA, 2020; Volume 2230, p. 020008. [Google Scholar]
- Iftimie, N.; Savin, A.; Steigmann, R.; Faktorova, D.; Salaoru, I. ZnO thin film as MSG for sensitive biosensor. IOP Conf. Ser. Mater. Sci. Eng. 2016, 145, 042030. [Google Scholar] [CrossRef]
- Moirangthem, R.S.; Chang, Y.C.; Wei, P.K. Ellipsometry study on gold-nanoparticle-coated gold thin film for biosensing application. Biomed. Opt. Express 2011, 2, 2569. [Google Scholar] [CrossRef]
- Zhao, Y.; Tong, R.J.; Xia, F.; Peng, Y. Current status of optical fiber biosensor based on surface plasmon resonance. Biosens. Bioelectron. 2019, 142, 111505. [Google Scholar] [CrossRef]
- Jang, H.S.; Park, K.N.; Kim, J.P.; Sim, S.J.; Kwon, O.J.; Han, Y.G.; Lee, K.S. Sensitive DNA biosensor based on a long-period grating formed on the side-polished fiber surface. Opt. Express 2009, 17, 3855–3860. [Google Scholar] [CrossRef] [PubMed]
- Chiavaioli, F.; Baldini, F.; Tombelli, S.; Trono, C.; Giannetti, A. Biosensing with optical fiber gratings. Nanophotonics 2017, 6, 663–679. [Google Scholar] [CrossRef]
- Coelho, L.; Almeida, J.; Santos, J.L.; Jorge, P.; Martins, M.C.; Viegas, D.; Queiros, R.B. Aptamer-based fiber sensor for thrombin detection. J. Biomed. Opt. 2016, 21, 87005. [Google Scholar] [CrossRef] [PubMed]
- Shevchenko, Y.; Francis, T.J.; Blair, D.A.; Walsh, R.; DeRosa, M.C.; Albert, J. In Situ Biosensing with a surface plasmon resonance fiber grating aptasensor. Anal. Chem. 2011, 83, 7027–7034. [Google Scholar] [CrossRef] [PubMed]
- Liu, M.; Li, J.; Li, B.X. A colorimetric aptamer biosensor based on cationic polythiophene derivative as peroxidase mimetics for the ultrasensitive detection of thrombin. Talanta 2017, 175, 224–228. [Google Scholar] [CrossRef]
- Li, S.; Zhang, D.; Zhang, Q.; Lu, Y.L.; Li, N.; Chen, Q.W.; Liu, Q.J. Electrophoresis-enhanced localized surface plasmon resonance sensing based on nanocup array for thrombin detection. Sens. Actuators B Chem. 2016, 232, 219–225. [Google Scholar] [CrossRef]
- Villatoro, J.; Monzon-Hernandez, D. Low-cost optical fiber refractive-index sensor based on core diameter mismatch. J. Light. Technol. 2006, 24, 1409–1413. [Google Scholar] [CrossRef]
- Owji, E.; Mokhtari, H.; Ostovari, F.; Darazereshki, B.; Shakiba, N. 2D materials coated on etched optical fibers as humidity sensor. Sci. Rep. 2021, 11, 1771. [Google Scholar] [CrossRef] [PubMed]
- Komanec, M.; Nemecek, T.; Vidner, P.M.; Martan, T.; Lahodny, F.; Zvanovec, S. Structurally-modified tapered optical fiber sensors for long-term detection of liquids. Opt. Fiber Technol. 2019, 47, 187–191. [Google Scholar] [CrossRef]
- Asseh, A.; Sandgren, S.; Ahlfeldt, H.; Sahlgren, B.; Stubbe, R.; Edwall, G. Fiber optical Bragg grating refractometer. Fiber Integr. Opt. 1998, 17, 51–62. [Google Scholar]
- Ladicicco, A.; Cusano, A.; Campopiano, S.; Cutolo, A.; Giordano, M. Thinned fiber Bragg gratings as refractive index sensors. IEEE Sens. J. 2005, 5, 1288–1295. [Google Scholar] [CrossRef]
- Chryssis, A.N.; Saini, S.S.; Lee, S.M.; Yi, H.; Bentley, W.E.; Dagenais, M. Detecting hybridization of DNA by highly sensitive evanescent field etched core fiber Bragg grating sensors. IEEE J. Sel. Top. Quantum Electron. 2005, 11, 864. [Google Scholar] [CrossRef]
- Guo, T.; Liu, F.; Liu, Y.; Chen, N.K.; Guan, B.O.; Albert, J. In-situ detection of density alteration in non-physiological cells with polarimetric tilted fiber grating sensors. Biosens. Bioelectron. 2014, 55, 452–458. [Google Scholar] [CrossRef] [PubMed] [Green Version]











Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kazanskiy, N.L.; Khonina, S.N.; Butt, M.A.; Kaźmierczak, A.; Piramidowicz, R. State-of-the-Art Optical Devices for Biomedical Sensing Applications—A Review. Electronics 2021, 10, 973. https://doi.org/10.3390/electronics10080973
Kazanskiy NL, Khonina SN, Butt MA, Kaźmierczak A, Piramidowicz R. State-of-the-Art Optical Devices for Biomedical Sensing Applications—A Review. Electronics. 2021; 10(8):973. https://doi.org/10.3390/electronics10080973
Chicago/Turabian StyleKazanskiy, N. L., S. N. Khonina, M. A. Butt, A. Kaźmierczak, and R. Piramidowicz. 2021. "State-of-the-Art Optical Devices for Biomedical Sensing Applications—A Review" Electronics 10, no. 8: 973. https://doi.org/10.3390/electronics10080973
APA StyleKazanskiy, N. L., Khonina, S. N., Butt, M. A., Kaźmierczak, A., & Piramidowicz, R. (2021). State-of-the-Art Optical Devices for Biomedical Sensing Applications—A Review. Electronics, 10(8), 973. https://doi.org/10.3390/electronics10080973









