Foodomic-Based Approach for the Control and Quality Improvement of Dairy Products
Abstract
:1. Introduction
2. Description and Application of Transcriptomics, Proteomics and Metabolomics in Food Analysis
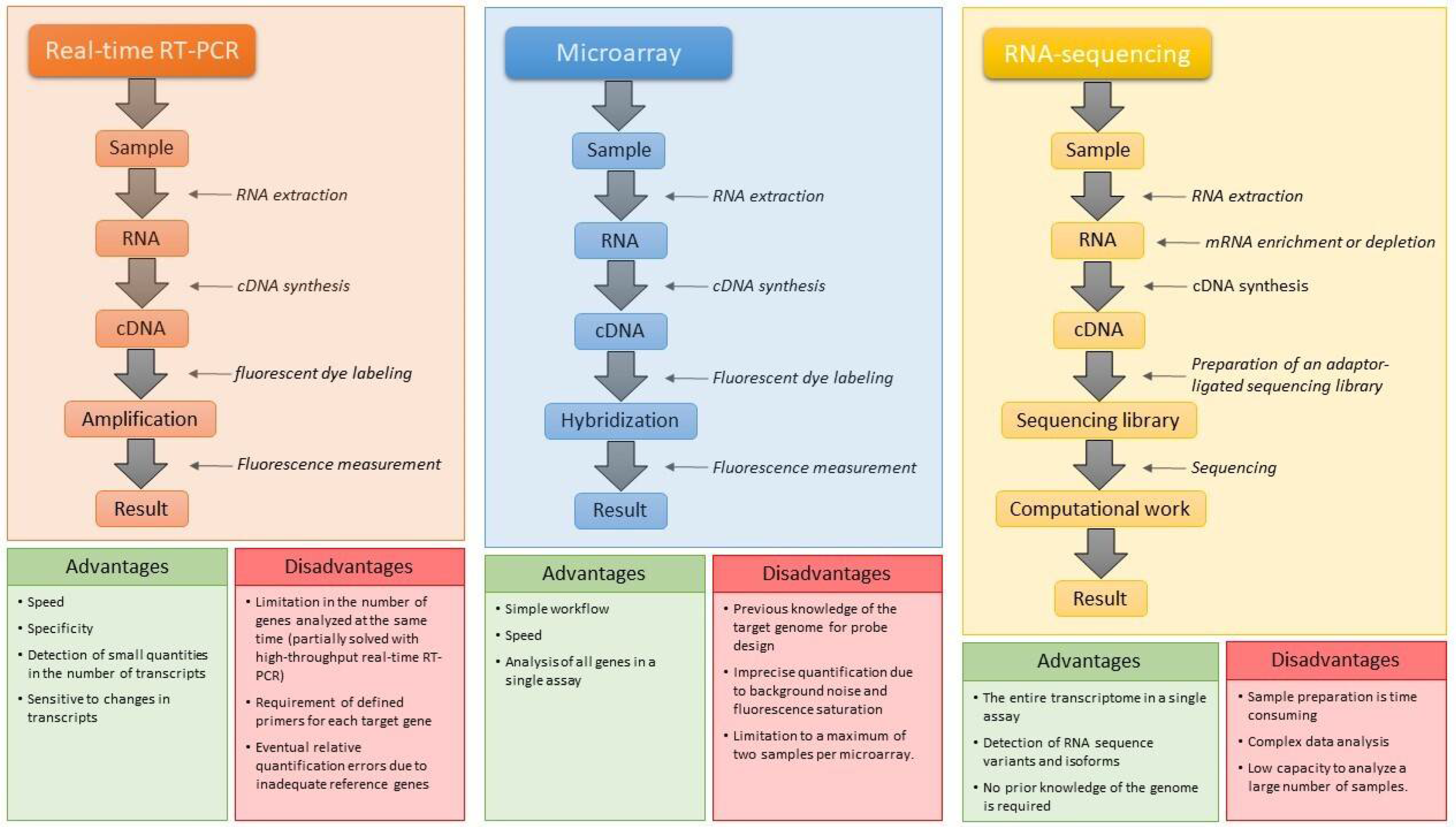
2.1. Transcriptomics
2.2. Proteomics
2.3. Metabolomics
3. Application of Foodomics for Quality Control in Dairy Products
3.1. Microbiological Contaminants. Detection of Pathogens and Their Toxins
3.2. Authenticity Assurance. Struggle against Food Fraud and the Presence of Undeclared Allergens
3.3. Evaluation of Industrial Processes for the Improvement of Dairy Products
Study of the Behavior of Microorganisms during the Fermentation and Maturation Stages
| Dairy Product or Culture Medium | Microorganism/s Involved | Omic/s Technologies | Applied Technologies and/or Techniques | Highlighted Findings | Reference |
|---|---|---|---|---|---|
| M17 broth | Streptococcus thermophilus TH1436 and TH1477 | Transcriptomics | RNA-sequencing | Overexpression of genes related to acid fermentation, phosphotransferase system, sugar transporter, and stress response in Gal+ species and underexpression of genes related to amino acids, protein metabolism, and CRISPR associated proteins in Gal− species. Modification of the metabolism in Gal+ strains depending on the environment. | Giaretta et al. [90] |
| Yogurt | Lactobacillus delbrueckii subsp. bulgaricus ATC11842 | Transcriptomics | RNA-sequencing and quantitative real-time PCR | Genes poorly expressed between the end of fermentation and the beginning of storage seem to be related to the post-acidification stage. The overexpression of the LDB_RS05285 gene could be involved in the reduction of lactic acid without affecting the growth of the strain. | Zhang et al. [91] |
| Swiss-type Maasdam cheese | Lactococcus lactis subsp. lactis, L. lactis subsp cremoris, Lactobacillus rhamnosus, Lactobacillus. helveticus, and Propionibacterium freudenreichii subsp. shermanii | Genomics and transcriptomics | Not specified | Lactococcus lactis is the dominant species of the cheese microbiota. Different pathways for the formation of flavor and production of free fatty acids, acetoin, diacetyl, acetate, ethanol, and propionate. Reduced center metabolism during cold storage except for Lactococcus lactis. | Duru et al. [85] |
| Raw ewes’ milk-based cheese | LAB community | Genomics and transcriptomics | PCR | Lactococcus is the dominant group of bacteria in cheese. Information on the role and properties of members of the Lactobacillus and Lactococcus genera | Pangallo et al. [84] |
| Fermented designed medium | Streptococcus thermophilus | Transcriptomics and proteomics | SDS-PAGE and iTRAQ HPLC-TOF MS/MS | Significant changes in the expression of 1396 genes and 876 proteins during fermentation. Relevant changes in heterofermentation, glycolysis, peptidoglycan biosynthesis, conversion between amino acids, and stress response occurred in the late-lag phase. | Qiao et al. [88] |
| M17 broth | Lactococcus lactic subsp. lactis IL1403, L. lactic subsp. lactis NCDO2118, L. lactic subsp. cremoris MG1363, and L. lactic subsp. cremoris NZ9000 | Proteomics | Label-free quantification nUPLC-nESI MS/MS | Probable involvement of the core proteome in resistance to stress and probiotic activity. The proteins related to the translation process are the most abundant in the core proteome. Presence of exclusive conserved proteins in some of the strains. Detection of a specific proteome in subsp. lactis NCDO2118. | Silva et al. [87] |
| Goat’s milk kefir | Kefir grains | Proteomics | UPLC-nESI MS/MS | Identification of 2238 unique peptides corresponding to 22 protein annotations. Maximum peptide release during the first 24 h of fermentation. Different digestion patterns according to the nature of the proteins. Identification of 11 peptides with recognized biological activity. | Izquierdo-gonzález et al. [86] |
| Fermented Elliker broth | Lactobacillus delbrueckii subsp. Bulgaricus ATCC11842, L. delbrueckii subsp. lactis LMG6401, and the isolate L. delbrueckii 23 from Mozzeralla di Bufala Campana | Metabolomics and proteomics | NMR spectroscopy (H-NMR analysis) and 2D LC-LTQ MS | Differences in the metabolic pathways of folate, amino acids, and sugar in the three strains. The origin of the strains is divergent. Lactobacillus delbrueckii subsp. Bulgaricus ATCC11842 showed probiotic properties. | Zanni et al. [89] |
| GABA-rich cheese | GABA-producing bacterial strain L. lactis subsp. Lactis biovar diacetylactis 01-7 and non-GABA-producing bacterial strain L. lactis subsp. cremoris 01-1 | Metabolomics | HPLC, LC-ESI MS/MS, and LC-LTQ Orbitrap MS | Presence of GABA and ornithine in cheese fermented with GABA-producing bacterial strain. Presence of citrate in the control cheese. Higher presence of peptides with antihypertensive activity and other functions in GABA-rich cheese. Lower amount of YL peptide in the GABA-rich cheese compared to the control. | Hagi et al. [92] |
| Yogurt | Lactobacillus delbrueckii subsp. Bulgaricus and Streptococcus thermophillus | Metabolomics | UPLC-Triple TOF MS/MS | Increase of 45 metabolites and decrease of another 47. Strict anaerobic fermentation promotes metabolic changes in bacteria and nutritional changes in yogurt. | Ding et al. [93] |
| Yogurt | Lactobacillus delbrueckii subsp. Bulgaricus, Streptococcus thermophillus, lactobacillus plantarum Taj-Apis362 UPMC90 and lactobacillus plantarum Taj-Apis362 UPMC91 | Metabolomics | NMR spectroscopy (H-NMR analysis) | Probable influence of Lactobacilus strains UPMC90 and UPMC91 on the metabolic profile. The addition of glucose increases the production of GABA by the UPMC90 and UPMC91 strains. GABA is produced by Lactobacillus delbrueckii subsp. Bulgaricus and Streptococcus thermophillus without the addition of glucose. Lower lactose content in GABA-rich yogurt. | Hussin et al. [94] |
| Fermented skim milk | L. plantarum P9 | Metabolomics | UPLC-QTOF MS/MS | Significant change in the metabolome after fermentation and cold storage (increase of 25 metabolites and decrease of another 10). These metabolites include fatty acids, peptides, and carbohydrates. Some of them could contribute functional attributes to the fermented product. | Zha et al. [95] |
4. Multi-Omics. The New Horizon for ‘Omic’ Technologies
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Prakash, B.; Singh, P.P.; Kumar, A.; Gupta, V. Prospects of omics technologies and bioinformatics approaches in food science. In Functional and Preservative Properties of Phytochemicals; Prakash, B., Ed.; Elsevier: Amsterdam, The Netherlands, 2020; pp. 317–340. ISBN 978-0-12-818593-3. [Google Scholar]
- Zheng, C.; Chen, A. System Biological Research on Food Quality for Personalised Nutrition and Health Using Foodomics Techniques: A Review. J. Food Nutr. Res. 2014, 2, 608–616. [Google Scholar] [CrossRef]
- Ellis, D.I.; Muhamadali, H.; Chisanga, M.; Goodacre, R. Omics Methods For the Detection of Foodborne Pathogens. In Encyclopedia of Food Chemistry; Melton, L., Shahidi, F., Varelis, P., Eds.; Elsevier: Amsterdam, The Netherlands, 2019; pp. 364–370. ISBN 978-0-12-814045-1. [Google Scholar]
- Ferranti, P. The future of analytical chemistry in foodomics. Curr. Opin. Food Sci. 2018, 22, 102–108. [Google Scholar] [CrossRef]
- Cifuentes, A. Food analysis and foodomics. J. Chromatogr. A 2009, 1216, 7109. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lamas, A.; Regal, P.; Vázquez, B.; Miranda, J.M.; Franco, C.M.; Cepeda, A. Transcriptomics: A powerful tool to evaluate the behavior of foodborne pathogens in the food production chain. Food Res. Int. 2019, 125, 108543. [Google Scholar] [CrossRef] [PubMed]
- Valdés, A.; Ibáñez, C.; Simó, C.; García-Cañas, V. Recent transcriptomics advances and emerging applications in food science. TrAC Trends Anal. Chem. 2013, 52, 142–154. [Google Scholar] [CrossRef]
- Lancova, K.; Dip, R.; Antignac, J.P.; Bizec, B.L.; Elliott, C.T.; Naegeli, H. Detection of hazardous food contaminants by transcriptomics fingerprinting. TrAC Trends Anal. Chem. 2011, 30, 181–191. [Google Scholar] [CrossRef]
- Lowe, R.; Shirley, N.; Bleackley, M.; Dolan, S.; Shafee, T. Transcriptomics technologies. PLoS Comput. Biol. 2017, 13, e1005457. [Google Scholar] [CrossRef] [Green Version]
- Bustin, S.A.; Nolan, T. Pitfalls of Quantitative Real-Time Reverse-Transcription Polymerase Chain Reaction. J. Biomol. Technol. 2004, 15, 155–166. [Google Scholar]
- Huggett, J.; Dheda, K.; Bustin, S.; Zumla, A. Real-time RT-PCR normalisation; strategies and considerations. Genes Immun. 2005, 6, 279–284. [Google Scholar] [CrossRef] [Green Version]
- Lenahan, M.; Sheridan, Á.; Morris, D.; Duffy, G.; Fanning, S.; Burgess, C.M. Transcriptomic analysis of triclosan-susceptible and-tolerant Escherichia coli O157:H19 in response to triclosan exposure. Microb. Drug Resist. 2014, 20, 91–103. [Google Scholar] [CrossRef] [PubMed]
- Kaliyappan, K.; Palanisamy, M.; Govindarajan, R.; Duraiyan, J. Microarray and its applications. J. Pharm. Bioallied Sci. 2012, 4, S310–S312. [Google Scholar] [CrossRef] [PubMed]
- Stark, R.; Grzelak, M.; Hadfield, J. RNA sequencing: The teenage years. Nat. Rev. Genet. 2019, 20, 631–656. [Google Scholar] [CrossRef]
- Gallardo, J.M.; Carrera, M.; Ortea, I. Proteomics in Food Science. In Foodomics: Advanced Mass Spectrometry in Modern Food Science and Nutrition; Cifuentes, A., Ed.; John Wiley & Sons: Hoboken, NJ, USA, 2013; pp. 125–165. ISBN 9781118169452. [Google Scholar]
- Tyers, M.; Mann, M. From genomics to proteomics. Nature 2003, 422, 193–197. [Google Scholar] [CrossRef] [PubMed]
- Rabilloud, T.; Chevallet, M.; Luche, S.; Lelong, C. Two-dimensional gel electrophoresis in proteomics: Past, present and future. J. Proteom. 2010, 73, 2064–2077. [Google Scholar] [CrossRef] [Green Version]
- Meleady, P. Two-Dimensional Gel Electrophoresis and 2D-DIGE. In Difference Gel Electrophoresis: Methods and Protocols, Methods in Molecular Biology; Ohlendieck, K., Ed.; Humana Press: New York, NY, USA, 2018; Volume 1664, ISBN 978-1-4939-7267-8. [Google Scholar]
- Dupree, E.J.; Jayathirtha, M.; Yorkey, H.; Mihasan, M.; Petre, B.A.; Darie, C.C. A critical review of bottom-up proteomics: The good, the bad, and the future of this field. Proteomes 2020, 8, 14. [Google Scholar] [CrossRef] [PubMed]
- Agregán, R.; Echegaray, N.; López-Pedrouso, M.; Aadil, R.M.; Hano, C.; Franco, D.; Lorenzo, J.M. Proteomic advances in cereal and vegetable crops. Molecules 2021, 26, 4924. [Google Scholar] [CrossRef] [PubMed]
- Gao, Y.; Yates, J.R. Protein Analysis by Shotgun Proteomics. In Mass Spectrometry-Based Chemical Proteomics; Tao, W.A., Zhang, Y., Eds.; John Wiley & Sons: Hoboken, NJ, USA, 2019; pp. 1–38. ISBN 9781118969557. [Google Scholar]
- Arora, A.; Somasundaram, K. Targeted Proteomics Comes to the Benchside and the Bedside: Is it Ready for Us? BioEssays 2019, 41, 1800042. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Idle, J.R.; Gonzalez, F.J. Metabolomics. Cell Metab. 2007, 6, 348–351. [Google Scholar] [CrossRef] [Green Version]
- Liu, X.; Locasale, J.W. Metabolomics: A Primer. Trends Biochem. Sci. 2017, 42, 274–284. [Google Scholar] [CrossRef] [Green Version]
- Castro-Puyana, M.; Pérez-Míguez, R.; Montero, L.; Herrero, M. Application of mass spectrometry-based metabolomics approaches for food safety, quality and traceability. TrAC Trends Anal. Chem. 2017, 93, 102–118. [Google Scholar] [CrossRef] [Green Version]
- Miggiels, P.; Wouters, B.; van Westen, G.J.P.; Dubbelman, A.C.; Hankemeier, T. Novel technologies for metabolomics: More for less. TrAC Trends Anal. Chem. 2019, 120, 115323. [Google Scholar] [CrossRef]
- Markley, J.L.; Brüschweiler, R.; Edison, A.S.; Eghbalnia, H.R.; Powers, R.; Raftery, D.; Wishart, D.S. The future of NMR-based metabolomics. Curr. Opin. Biotechnol. 2017, 43, 34–40. [Google Scholar] [CrossRef] [Green Version]
- Ren, J.L.; Zhang, A.H.; Kong, L.; Wang, X.J. Advances in mass spectrometry-based metabolomics for investigation of metabolites. RSC Adv. 2018, 8, 22335–22350. [Google Scholar] [CrossRef] [Green Version]
- Pal, M.; Mulu, S.; Tekle, M.; Pintoo, S.V.; Prajapati, J.P. Bacterial Contamination of Dairy Products. Beverage Food World 2016, 43, 40–43. [Google Scholar]
- Chen, S.; He, S.; Xu, X.; Wang, H. Transcriptomic responses of foodborne pathogens to the food matrix. Curr. Opin. Food Sci. 2021, 42, 23–30. [Google Scholar] [CrossRef]
- Cretenet, M.; Nouaille, S.; Thouin, J.; Rault, L.; Stenz, L.; François, P.; Hennekinne, J.A.; Piot, M.; Maillard, M.B.; Fauquant, J.; et al. Staphylococcus aureus virulence and metabolism are dramatically affected by Lactococcus lactis in cheese matrix. Environ. Microbiol. Rep. 2011, 3, 340–351. [Google Scholar] [CrossRef]
- Piras, C.; Roncada, P.; Rodrigues, P.M.; Bonizzi, L.; Soggiu, A. Proteomics in food: Quality, safety, microbes, and allergens. Proteomics 2016, 16, 799–815. [Google Scholar] [CrossRef]
- Karasu-Yalcin, S.; Soylemez-Milli, N.; Eren, O.; Eryasar-Orer, K. Reducing time in detection of Listeria monocytogenes from food by MALDI-TOF mass spectrometry. J. Food Sci. Technol. 2021, 58, 4102–4109. [Google Scholar] [CrossRef]
- Pyz-Łukasik, R.; Gondek, M.; Winiarczyk, D.; Michalak, K.; Paszkiewicz, W.; Piróg-Komorowska, A.; Policht, A.; Ziomek, M. Occurrence of Listeria monocytogenes in artisanal cheeses from poland and its identification by MALDI-TOF MS. Pathogens 2021, 10, 632. [Google Scholar] [CrossRef]
- Jadhav, S.; Sevior, D.; Bhave, M.; Palombo, E.A. Detection of Listeria monocytogenes from selective enrichment broth using MALDI-TOF Mass Spectrometry. J. Proteom. 2014, 97, 100–106. [Google Scholar] [CrossRef]
- Kačániová, M.; Terentjeva, M.; Godočíková, L.; Puchalski, C.; Kluz, M.; Kordiaka, R.; Kunová, S.; Haščík, P. Rapid Identification of Enterobacteriaceae in Milk and Dairy Products with the Matrix-Assisted Laser Desorption Ionization-Time of Flight Mass Spectrometry (MALDI-TOF MS). Sci. Pap. Anim. Sci. Biotechnol. 2017, 50, 41–46. [Google Scholar]
- Jadhav, S.R.; Shah, R.M.; Karpe, A.V.; Beale, D.J.; Kouremenos, K.A.; Palombo, E.A. Identification of Putative Biomarkers Specific to Foodborne Pathogens Using Metabolomics. In Foodborne Bacterial Pathogens: Methods and Protocols, Methods in Molecular Biology; Bridie, A., Ed.; Springer: New York, NY, USA, 2019; Volume 1918, pp. 149–164. [Google Scholar]
- Eshelli, M.; Qader, M.M.; Jambi, E.J.; Hursthouse, A.S.; Rateb, M.E. Current status and future opportunities of omics tools in mycotoxin research. Toxins 2018, 10, 433. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Becker-Algeri, T.A.; Castagnaro, D.; de Bortoli, K.; de Souza, C.; Drunkler, D.A.; Badiale-Furlong, E. Mycotoxins in Bovine Milk and Dairy Products: A Review. J. Food Sci. 2016, 81, R544–R552. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cakmakci, S.; Gurses, M.; Hayaloglu, A.A.; Cetin, B.; Sekerci, P.; Dagdemir, E. Mycotoxin production capability of Penicillium roqueforti in strains isolated from mould-ripened traditional Turkish civil cheese. Food Addit. Contam. Part A 2015, 32, 245–249. [Google Scholar] [CrossRef] [PubMed]
- Anelli, P.; Haidukowski, M.; Epifani, F.; Cimmarusti, M.T.; Moretti, A.; Logrieco, A.; Susca, A. Fungal mycobiota and mycotoxin risk for traditional artisan Italian cave cheese. Food Microbiol. 2019, 78, 62–72. [Google Scholar] [CrossRef]
- Mehta, R.; Shetty, S.; Young, M.F.; Ryan, P.B.; Rangiah, K. Quantification of Mycotoxins in Animal Milk from India (FS14-04-19). Curr. Dev. Nutr. 2019, 3, FS14-04-19. [Google Scholar] [CrossRef] [Green Version]
- Rocchetti, G.; Ghilardelli, F.; Masoero, F.; Gallo, A. Screening of Regulated and Emerging Mycotoxins in Bulk Milk Samples by High-Resolution Mass Spectrometry. Foods 2021, 10, 2025. [Google Scholar] [CrossRef]
- CEN-CENELEC Management Centre. Authentic and Fraud in the Feed and Food Chain—Concepts, Terms and Definitions; European Committee for Standarization: Brussels, Belgium, 2019. [Google Scholar]
- Ortea, I. Omics and Food Authentication. In Foodomics: Omic Strategies and Applications in Food Science; Barros-Velázquez, J., Ed.; RSC Publishing: Cambridge, UK, 2021; pp. 204–221. [Google Scholar]
- Böhme, K.; Calo-Mata, P.; Barros-Velázquez, J.; Ortea, I. Recent applications of omics-based technologies to main topics in food authentication. TrAC Trends Anal. Chem. 2019, 110, 221–232. [Google Scholar] [CrossRef]
- Islam, S.; Thangadurai, D. Proteomic Authentication of Dairy Products. In Handbook of Dairy Foods Analysis; Fidel, T., Nollet, L.M.L., Eds.; CRC Press: Boca Raton, FL, USA, 2021; pp. 405–427. ISBN 9780367343132. [Google Scholar]
- Poonia, A.; Jha, A.; Sharma, R.; Singh, H.B.; Rai, A.K.; Sharma, N. Detection of adulteration in milk: A review. Int. J. Dairy Technol. 2017, 70, 23–42. [Google Scholar] [CrossRef]
- Arena, S.; Salzano, A.M.; Scaloni, A. Identification of protein markers for the occurrence of defrosted material in milk through a MALDI-TOF-MS profiling approach. J. Proteom. 2016, 147, 56–65. [Google Scholar] [CrossRef]
- Caira, S.; Pinto, G.; Nicolai, M.A.; Chianese, L.; Addeo, F. Simultaneously tracing the geographical origin and presence of bovine milk in Italian water buffalo Mozzarella cheese using MALDI-TOF data of casein signature peptides. Anal. Bioanal. Chem. 2016, 408, 5609–5621. [Google Scholar] [CrossRef] [PubMed]
- Fontenele, M.A.; Bastos, M.d.S.R.; dos Santos, K.M.O.; Bemquerer, M.P.; do Egito, A.S. Peptide profile of Coalho cheese: A contribution for Protected Designation of Origin (PDO). Food Chem. 2017, 219, 382–390. [Google Scholar] [CrossRef] [PubMed]
- Rau, J.; Korte, N.; Dyk, M.; Wenninger, O.; Schreiter, P.; Hiller, E. Rapid animal species identification of feta and mozzarella cheese using MALDI-TOF mass-spectrometry. Food Control 2020, 117, 107349. [Google Scholar] [CrossRef]
- Kandasamy, S.; Yoo, J.; Yun, J.; Kang, H.B.; Seol, K.H.; Ham, J.S. Rapid discrimination and authentication of Korean farmstead mozzarella cheese through MALDI-TOF and multivariate statistical analysis. Metabolites 2021, 11, 333. [Google Scholar] [CrossRef]
- Kritikou, A.S.; Aalizadeh, R.; Damalas, D.E.; Barla, I.V.; Baessmann, C.; Thomaidis, N.S. MALDI-TOF-MS integrated workflow for food authenticity investigations: An untargeted protein-based approach for rapid detection of PDO feta cheese adulteration. Food Chem. 2022, 370, 131057. [Google Scholar] [CrossRef]
- D’Auria, E.; Mameli, C.; Piras, C.; Cococcioni, L.; Urbani, A.; Zuccotti, G.V.; Roncada, P. Precision medicine in cow’s milk allergy: Proteomics perspectives from allergens to patients. J. Proteom. 2018, 188, 173–180. [Google Scholar] [CrossRef] [PubMed]
- Ji, J.; Zhu, P.; Pi, F.; Sun, C.; Sun, J.; Jia, M.; Ying, C.; Zhang, Y.; Sun, X. Development of a liquid chromatography-tandem mass spectrometry method for simultaneous detection of the main milk allergens. Food Control 2017, 74, 79–88. [Google Scholar] [CrossRef]
- Qi, K.; Liu, T.; Yang, Y.; Zhang, J.; Yin, J.; Ding, X.; Qin, W.; Yang, Y. A rapid immobilized trypsin digestion combined with liquid chromatography—Tandem mass spectrometry for the detection of milk allergens in baked food. Food Control 2019, 102, 179–187. [Google Scholar] [CrossRef]
- Zhu, D.; Kebede, B.; McComb, K.; Hayman, A.; Chen, G.; Frew, R. Milk biomarkers in relation to inherent and external factors based on metabolomics. Trends Food Sci. Technol. 2021, 109, 51–64. [Google Scholar] [CrossRef]
- Rocchetti, G.; Lucini, L.; Gallo, A.; Masoero, F.; Trevisan, M.; Giuberti, G. Untargeted metabolomics reveals differences in chemical fingerprints between PDO and non-PDO Grana Padano cheeses. Food Res. Int. 2018, 113, 407–413. [Google Scholar] [CrossRef]
- Salzano, A.; Manganiello, G.; Neglia, G.; Vinale, F.; De Nicola, D.; D’Occhio, M.; Campanile, G. A preliminary study on metabolome profiles of buffalo milk and corresponding mozzarella cheese: Safeguarding the authenticity and traceability of protected status buffalo dairy products. Molecules 2020, 25, 304. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tenori, L.; Santucci, C.; Meoni, G.; Morrocchi, V.; Matteucci, G.; Luchinat, C. NMR metabolomic fingerprinting distinguishes milk from different farms. Food Res. Int. 2018, 113, 131–139. [Google Scholar] [CrossRef] [PubMed]
- Sobolev, A.P.; Thomas, F.; Donarski, J.; Ingallina, C.; Circi, S.; Cesare Marincola, F.; Capitani, D.; Mannina, L. Use of NMR applications to tackle future food fraud issues. Trends Food Sci. Technol. 2019, 91, 347–353. [Google Scholar] [CrossRef] [Green Version]
- Castro-Puyana, M.; Herrero, M. Metabolomics approaches based on mass spectrometry for food safety, quality and traceability. TrAC Trends Anal. Chem. 2013, 52, 74–87. [Google Scholar] [CrossRef]
- Corredig, M.; Dalgleish, D.G. Effect of temperature and pH on the interactions of whey proteins with casein micelles in skim milk. Food Res. Int. 1996, 29, 49–55. [Google Scholar] [CrossRef]
- Chen, D.; Li, X.; Zhao, X.; Qin, Y.; Wang, J.; Wang, C. Comparative proteomics of goat milk during heated processing. Food Chem. 2019, 275, 504–514. [Google Scholar] [CrossRef]
- Loveday, S.M. β-Lactoglobulin heat denaturation: A critical assessment of kinetic modelling. Int. Dairy J. 2016, 52, 92–100. [Google Scholar] [CrossRef]
- Meltretter, J.; Wüst, J.; Dittrich, D.; Lach, J.; Ludwig, J.; Eichler, J.; Pischetsrieder, M. Untargeted Proteomics-Based Profiling for the Identification of Novel Processing-Induced Protein Modifications in Milk. J. Proteome Res. 2020, 19, 805–818. [Google Scholar] [CrossRef]
- O’Brien, J. Non-Enzymatic Degradation Pathways of Lactose and Their Significance in Dairy Products. In Advanced Dairy Chemistry; McSweeney, P., Fox, P.F., Eds.; Springer: New York, NY, USA, 2009; Volume 3, pp. 231–294. ISBN 978-0-387-84864-8. [Google Scholar]
- Aalaei, K.; Rayner, M.; Sjöholm, I. Chemical methods and techniques to monitor early Maillard reaction in milk products; A review. Crit. Rev. Food Sci. Nutr. 2019, 59, 1829–1839. [Google Scholar] [CrossRef]
- Milkovska-Stamenova, S.; Hoffmann, R. Diversity of advanced glycation end products in the bovine milk proteome. Amino Acids 2019, 51, 891–901. [Google Scholar] [CrossRef]
- Meltretter, J.; Birlouez-Aragon, I.; Becker, C.M.; Pischetsrieder, M. Assessment of heat treatment of dairy products by MALDI-TOF-MS. Mol. Nutr. Food Res. 2009, 53, 1487–1495. [Google Scholar] [CrossRef] [PubMed]
- Milkovska-Stamenova, S.; Hoffmann, R. Influence of storage and heating on protein glycation levels of processed lactose-free and regular bovine milk products. Food Chem. 2017, 221, 489–495. [Google Scholar] [CrossRef]
- Chandra Roy, M.; Zhang, L.; Liu, X.; Zhou, P. Investigation of caprine milk serum proteome and glycated proteome changes during heat treatment using robust ion mobility time-of-flight proteomic techniques. Int. Dairy J. 2020, 110, 104798. [Google Scholar] [CrossRef]
- Liu, Y.; Xiong, L.; Kontopodi, E.; Boeren, S.; Zhang, L.; Zhou, P.; Hettinga, K. Changes in the milk serum proteome after thermal and non-thermal treatment. Innov. Food Sci. Emerg. Technol. 2020, 66, 102544. [Google Scholar] [CrossRef]
- Nasser, S.; De Sa Peixoto, P.; Moreau, A.; Croguennec, T.; Bray, F.; Rolando, C.; Tessier, F.J.; Hédoux, A.; Delaplace, G. Storage of Micellar Casein Powders with and without Lactose: Consequences on Color, Solubility, and Chemical Modifications. J. Agric. Food Chem. 2018, 66, 10274–10282. [Google Scholar] [CrossRef] [PubMed]
- Caboni, P.; Maxia, D.; Scano, P.; Addis, M.; Dedola, A.; Pes, M.; Murgia, A.; Casula, M.; Profumo, A.; Pirisi, A. A gas chromatography-mass spectrometry untargeted metabolomics approach to discriminate Fiore Sardo cheese produced from raw or thermized ovine milk. J. Dairy Sci. 2019, 102, 5005–5018. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.; Yan, D.; Zou, Y.; Mu, D.; Li, X.; Shi, H.; Luo, X.; Yang, M.; Yue, X.; Wu, R.; et al. Fermentation temperature affects yogurt quality: A metabolomics study. Food Biosci. 2021, 42, 101104. [Google Scholar] [CrossRef]
- Trimigno, A.; Lyndgaard, C.B.; Atladóttir, G.A.; Aru, V.; Engelsen, S.B.; Clemmensen, L.K.H. An NMR metabolomics approach to investigate factors affecting the yoghurt fermentation process and quality. Metabolites 2020, 10, 293. [Google Scholar] [CrossRef]
- Campos, D.C.D.S.; Neves, L.T.B.C.; Flach, A.; Costa, L.A.M.A.; de Sousa, B.O. Post-acidification and evaluation of anthocyanins stability and antioxidant activity in açaí fermented milk and yogurts (Euterpe oleracea Mart.). Rev. Bras. Frutic. 2017, 39, e871. [Google Scholar] [CrossRef] [Green Version]
- Sharma, H.; Ramanathan, R. Gas chromatography-mass spectrometry based metabolomic approach to investigate the changes in goat milk yoghurt during storage. Food Res. Int. 2021, 140, 110072. [Google Scholar] [CrossRef]
- Zhu, D.; Kebede, B.; Chen, G.; McComb, K.; Frew, R. Impact of freeze-drying and subsequent storage on milk metabolites based on 1H NMR and UHPLC-QToF/MS. Food Control 2020, 116, 107017. [Google Scholar] [CrossRef]
- Ferrocino, I.; Rantsiou, K.; Cocolin, L. Microbiota of Milk and Dairy Foods: Structure and Function by -omics Approaches. In Encyclopedia of Dairy Sciences; McSweeney, P.L.H., McNamara, J.P., Eds.; Elsevier: Amsterdam, The Netherlands, 2022; pp. 313–318. ISBN 978-0-12-818767-8. [Google Scholar]
- Afshari, R.; Pillidge, C.J.; Dias, D.A.; Osborn, A.M.; Gill, H. Cheesomics: The future pathway to understanding cheese flavour and quality. Crit. Rev. Food Sci. Nutr. 2020, 60, 33–47. [Google Scholar] [CrossRef] [PubMed]
- Pangallo, D.; Kraková, L.; Puškárová, A.; Šoltys, K.; Bučková, M.; Koreňová, J.; Budiš, J.; Kuchta, T. Transcription activity of lactic acid bacterial proteolysis-related genes during cheese maturation. Food Microbiol. 2019, 82, 416–425. [Google Scholar] [CrossRef]
- Duru, I.C.; Laine, P.; Andreevskaya, M.; Paulin, L.; Kananen, S.; Tynkkynen, S.; Auvinen, P.; Smolander, O.P. Metagenomic and metatranscriptomic analysis of the microbial community in Swiss-type Maasdam cheese during ripening. Int. J. Food Microbiol. 2018, 281, 10–22. [Google Scholar] [CrossRef]
- Izquierdo-González, J.J.; Amil-Ruiz, F.; Zazzu, S.; Sánchez-Lucas, R.; Fuentes-Almagro, C.A.; Rodríguez-Ortega, M.J. Proteomic analysis of goat milk kefir: Profiling the fermentation-time dependent protein digestion and identification of potential peptides with biological activity. Food Chem. 2019, 295, 456–465. [Google Scholar] [CrossRef] [PubMed]
- Silva, W.M.; Sousa, C.S.; Oliveira, L.C.; Soares, S.C.; Souza, G.F.M.H.; Tavares, G.C.; Resende, C.P.; Folador, E.L.; Pereira, F.L.; Figueiredo, H.; et al. Comparative proteomic analysis of four biotechnological strains Lactococcus lactis through label-free quantitative proteomics. Microb. Biotechnol. 2019, 12, 265–274. [Google Scholar] [CrossRef]
- Qiao, Y.; Leng, C.; Liu, G.; Zhang, Y.; Lv, X.; Chen, H.; Sun, J.; Feng, Z. Transcriptomic and proteomic profiling revealed global changes in Streptococcus thermophilus during pH-controlled batch fermentations. J. Microbiol. 2019, 57, 769–780. [Google Scholar] [CrossRef] [PubMed]
- Zanni, E.; Schifano, E.; Motta, S.; Sciubba, F.; Palleschi, C.; Mauri, P.; Perozzi, G.; Uccelletti, D.; Devirgiliis, C.; Miccheli, A. Combination of metabolomic and proteomic analysis revealed different features among Lactobacillus delbrueckii subspecies bulgaricus and lactis strains while in vivo testing in the model organism Caenorhabditis elegans highlighted probiotic properties. Front. Microbiol. 2017, 8, 1206. [Google Scholar] [CrossRef] [Green Version]
- Giaretta, S.; Treu, L.; Vendramin, V.; Duarte, V.d.S.; Tarrah, A.; Campanaro, S.; Corich, V.; Giacomini, A. Comparative transcriptomic analysis of Streptococcus thermophilus TH1436 and TH1477 showing different capability in the use of galactose. Front. Microbiol. 2018, 9, 1765. [Google Scholar] [CrossRef] [Green Version]
- Zhang, S.; Li, D.; Zhang, X.; Sun, Y.; Xu, S.; Wang, X.; Zhang, N.; Wang, M.; Tian, H.; Li, C. Global transcriptomic analysis of Lactobacillus delbrueckii subsp. bulgaricus ATCC11842 reveals the role of LDB_RS05285 in the post-acidification of yogurt. Food Funct. 2021, 12, 9077–9086. [Google Scholar] [CrossRef]
- Hagi, T.; Nakagawa, H.; Ohmori, H.; Sasaki, K.; Kobayashi, M.; Narita, T.; Nomura, M. Characterization of unique metabolites in γ-aminobutyric acid-rich cheese by metabolome analysis using liquid chromatography-mass spectrometry. J. Food Biochem. 2019, 43, e13039. [Google Scholar] [CrossRef]
- Ding, R.; Li, M.; Zou, Y.; Wang, Y.; Yan, C.; Zhang, H.; Wu, R.; Wu, J. Effect of normal and strict anaerobic fermentation on physicochemical quality and metabolomics of yogurt. Food Biosci. 2021, 101368. [Google Scholar] [CrossRef]
- Hussin, F.S.; Chay, S.Y.; Hussin, A.S.M.; Wan Ibadullah, W.Z.; Muhialdin, B.J.; Abd Ghani, M.S.; Saari, N. GABA enhancement by simple carbohydrates in yoghurt fermented using novel, self-cloned Lactobacillus plantarum Taj-Apis362 and metabolomics profiling. Sci. Rep. 2021, 11, 9417. [Google Scholar] [CrossRef]
- Zha, M.; Li, K.; Zhang, W.; Sun, Z.; Kwok, L.Y.; Menghe, B.; Chen, Y. Untargeted mass spectrometry-based metabolomics approach unveils molecular changes in milk fermented by Lactobacillus plantarum P9. LWT 2021, 140, 110759. [Google Scholar] [CrossRef]
- Subramanian, I.; Verma, S.; Kumar, S.; Jere, A.; Anamika, K. Multi-omics Data Integration, Interpretation, and Its Application. Bioinform. Biol. Insights 2020, 14. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Beale, D.J.; Karpe, A.V.; Ahmed, W. Beyond Metabolomics: A Review of Multi-Omics-Based Approaches. In Microbial Metabolomics; Beale, D., Kouremenos, K., Palombo, E., Eds.; Springer International Publishing: Cham, Switzerland, 2016; ISBN 978-3-319-46324-7. [Google Scholar]
- Bellassi, P.; Rocchetti, G.; Nocetti, M.; Lucini, L.; Masoero, F.; Morelli, L. A combined metabolomic and metagenomic approach to discriminate raw milk for the production of hard cheese. Foods 2021, 10, 109. [Google Scholar] [CrossRef] [PubMed]
- Sirén, K.; Mak, S.S.T.; Fischer, U.; Hansen, L.H.; Gilbert, M.T.P. Multi-omics and potential applications in wine production. Curr. Opin. Biotechnol. 2019, 56, 172–178. [Google Scholar] [CrossRef]
- Kim, M.; Tagkopoulos, I. Data integration and predictive modeling methods for multi-omics datasets. Mol. Omi. 2018, 14, 8–25. [Google Scholar] [CrossRef]
- Conesa, A.; Beck, S. Making multi-omics data accessible to researchers. Sci. Data 2019, 6, 251. [Google Scholar] [CrossRef] [Green Version]
- Belda, I.; Zarraonaindia, I.; Perisin, M.; Palacios, A.; Acedo, A. From vineyard soil to wine fermentation: Microbiome approximations to explain the “terroir” Concept. Front. Microbiol. 2017, 8, 821. [Google Scholar] [CrossRef] [Green Version]


| Aim of the Study | Type of Cheese | Biomarkers Found | Reference |
|---|---|---|---|
| Identify the use of frozen milk in cheese production | Mozzarela di Bufala Campana | GLYCAM1-derived phosphopeptides | Arena et al. [49] |
| β-casein-derived phosphopeptides | |||
| γ-Casein/β-casein peptides | |||
| α-Lactalbumin peptides | |||
| β-Lactoglobulin peptides | |||
| Unknown peptides | |||
| Identify the origin and presence of cow’s milk in a cheese made of water buffalo milk | Italian water buffalo Mozzarella cheese | αs1-Casein peptides | Aira et al. [50] |
| β-Casein peptides | |||
| Define the original geographical location and PDO of a traditional cheese | Coalho cheese | αs1-Casein peptides | Fontenele et al. [51] |
| αs2-Casein peptides | |||
| β-Casein peptides | |||
| βA2-Casein peptides | |||
| βA3-Casein peptides | |||
| κ-Casein peptides | |||
| Identify the species of milk used in making cheeses | Feta and Mozzarella cheeses | Not specified | Rau et al. [52] |
| Authenticate and discriminate different cheeses | Korean Mozzarella cheese | αs1-Casein peptides | Kandasamy et al. [53] |
| β-Casein peptides | |||
| αs2-Casein peptides | |||
| κ-Casein peptides | |||
| Unknown peptides | |||
| Identify the species of milk used in making cheeses | Feta cheese | Not specified | Kritikou et al. [54] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Agregán, R.; Echegaray, N.; Nawaz, A.; Hano, C.; Gohari, G.; Pateiro, M.; Lorenzo, J.M. Foodomic-Based Approach for the Control and Quality Improvement of Dairy Products. Metabolites 2021, 11, 818. https://doi.org/10.3390/metabo11120818
Agregán R, Echegaray N, Nawaz A, Hano C, Gohari G, Pateiro M, Lorenzo JM. Foodomic-Based Approach for the Control and Quality Improvement of Dairy Products. Metabolites. 2021; 11(12):818. https://doi.org/10.3390/metabo11120818
Chicago/Turabian StyleAgregán, Rubén, Noemí Echegaray, Asad Nawaz, Christophe Hano, Gholamreza Gohari, Mirian Pateiro, and José M. Lorenzo. 2021. "Foodomic-Based Approach for the Control and Quality Improvement of Dairy Products" Metabolites 11, no. 12: 818. https://doi.org/10.3390/metabo11120818








