Effects of Tryptophan Supplementation and Exercise on the Fate of Kynurenine Metabolites in Mice and Humans
Abstract
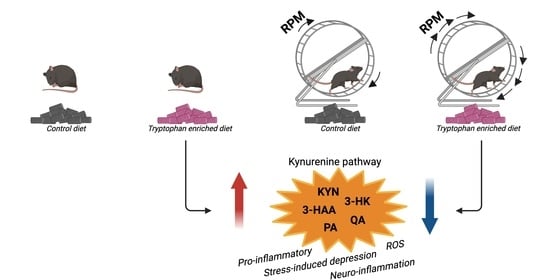
:1. Introduction
2. Results
3. Discussion
4. Materials and Methods
4.1. Animals
4.2. Diets
4.3. Free Wheel Running
4.4. Behavior
4.5. Gene Expression Analysis
4.6. Metabolic Cages
4.7. Body Composition
4.8. Glucose Tolerance Test
4.9. Human Exercise Study
4.10. Measurement of Metabolites in Plasma
4.11. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Maughan, R.J.; Burke, L.M.; Dvorak, J.; Larson-Meyer, D.E.; Peeling, P.; Phillips, S.M.; Rawson, E.S.; Walsh, N.P.; Garthe, I.; Geyer, H.; et al. IOC consensus statement: Dietary supplements and the high-performance athlete. Br. J. Sports Med. 2018, 52, 439–455. [Google Scholar] [CrossRef]
- Blomstrand, E. Amino acids and central fatigue. Amino Acids 2001, 20, 25–34. [Google Scholar] [CrossRef]
- Cervenka, I.; Agudelo, L.Z.; Ruas, J.L. Kynurenines: Tryptophan’s metabolites in exercise, inflammation, and mental health. Science 2017, 357, eaaf9794. [Google Scholar] [CrossRef] [Green Version]
- Opitz, C.A.; Litzenburger, U.M.; Sahm, F.; Ott, M.; Tritschler, I.; Trump, S.; Schumacher, T.; Jestaedt, L.; Schrenk, D.; Weller, M.; et al. An endogenous tumour-promoting ligand of the human aryl hydrocarbon receptor. Nature 2011, 478, 197–203. [Google Scholar] [CrossRef]
- Rothhammer, V.; Quintana, F.J. The aryl hydrocarbon receptor: An environmental sensor integrating immune responses in health and disease. Nat. Rev. Immunol. 2019, 19, 184–197. [Google Scholar] [CrossRef] [PubMed]
- Martin, K.S.; Azzolini, M.; Ruas, J. The kynurenine connection: How exercise shifts muscle tryptophan metabolism and affects energy homeostasis, the immune system, and the brain. Am. J. Physiol. Physiol. 2020, 318, C818–C830. [Google Scholar] [CrossRef] [PubMed]
- Eastman, C.L.; Guilarte, T.R. The role of hydrogen peroxide in the in vitro cytotoxicity of 3-hydroxykynurenine. Neurochem. Res. 1990, 15, 1101–1107. [Google Scholar] [CrossRef] [PubMed]
- Santamaría, A.; Galván-Arzate, S.; Lisý, V.; Ali, S.F.; Duhart, H.M.; Osorio-Rico, L.; Ríos, C.; Sut’Astný, F. Quinolinic acid induces oxidative stress in rat brain synaptosomes. NeuroReport 2001, 12, 871–874. [Google Scholar] [CrossRef]
- Lugo-Huitrón, R.; Blanco-Ayala, T.; Ugalde-Muñiz, P.; Carrillo-Mora, P.; Pedraza-Chaverrí, J.; Adaya, I.D.S.; Maldonado, P.D.; Torres, I.; Pinzón, E.; Ortiz-Islas, E.; et al. On the antioxidant properties of kynurenic acid: Free radical scavenging activity and inhibition of oxidative stress. Neurotoxicol. Teratol. 2011, 33, 538–547. [Google Scholar] [CrossRef]
- Stone, T.; Perkins, M. Quinolinic acid: A potent endogenous excitant at amino acid receptors in CNS. Eur. J. Pharmacol. 1981, 72, 411–412. [Google Scholar] [CrossRef]
- Birch, P.J.; Grossman, C.J.; Hayes, A.G. Kynurenic acid antagonises responses to NMDA via an action at the strychnine-insensitive glycine receptor. Eur. J. Pharmacol. 1988, 154, 85–87. [Google Scholar] [CrossRef]
- Divanovic, S.; Sawtell, N.M.; Trompette, A.; Warning, J.I.; Dias, A.; Cooper, A.M.; Yap, G.S.; Arditi, M.; Shimada, K.; DuHadaway, J.B.; et al. Opposing Biological Functions of Tryptophan Catabolizing Enzymes During Intracellular Infection. J. Infect. Dis. 2011, 205, 152–161. [Google Scholar] [CrossRef] [Green Version]
- Takikawa, O.; Yoshida, R.; Kido, R.; Hayaishi, O. Tryptophan degradation in mice initiated by indoleamine 2, 3-dioxygenase. J. Biol. Chem. 1986, 261, 3648–3653. [Google Scholar] [CrossRef]
- Agudelo, L.Z.; Femenía, T.; Orhan, F.; Porsmyr-Palmertz, M.; Goiny, M.; Martinez-Redondo, V.; Correia, J.; Izadi, M.; Bhat, M.; Schuppe-Koistinen, I.; et al. Skeletal Muscle PGC-1α1 Modulates Kynurenine Metabolism and Mediates Resilience to Stress-Induced Depression. Cell 2014, 159, 33–45. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schlittler, M.; Goiny, M.; Agudelo, L.Z.; Venckunas, T.; Brazaitis, M.; Skurvydas, A.; Kamandulis, S.; Ruas, J.; Erhardt, S.; Westerblad, H.; et al. Endurance exercise increases skeletal muscle kynurenine aminotransferases and plasma kynurenic acid in humans. Am. J. Physiol. Cell Physiol. 2016, 310, C836–C840. [Google Scholar] [CrossRef] [Green Version]
- Agudelo, L.Z.; Ferreira, D.M.S.; Dadvar, S.; Cervenka, I.; Ketscher, L.; Izadi, M.; Zhengye, L.; Furrer, R.; Handschin, C.; Venckunas, T.; et al. Skeletal muscle PGC-1α1 reroutes kynurenine metabolism to increase energy efficiency and fatigue-resistance. Nat. Commun. 2019, 10, 1–12. [Google Scholar] [CrossRef]
- Joisten, N.; Kummerhoff, F.; Koliamitra, C.; Schenk, A.; Walzik, D.; Hardt, L.; Knoop, A.; Thevis, M.; Kiesl, D.; Metcalfe, A.J.; et al. Exercise and the Kynurenine pathway: Current state of knowledge and results from a randomized cross-over study comparing acute effects of endurance and resistance training. Exerc. Immunol. Rev. 2020, 26, 24–42. [Google Scholar]
- Agudelo, L.Z.; Ferreira, D.M.S.; Cervenka, I.; Bryzgalova, G.; Dadvar, S.; Jannig, P.R.; Pettersson-Klein, A.; Lakshmikanth, T.; Sustarsic, E.G.; Porsmyr-Palmertz, M.; et al. Kynurenic Acid and Gpr35 Regulate Adipose Tissue Energy Homeostasis and Inflammation. Cell Metab. 2018, 27, 378–392.e5. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lehnig, A.; Stanford, K.I. Exercise-induced adaptations to white and brown adipose tissue. J. Exp. Biol. 2018, 221. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Badawy, A.A.-B.; Bano, S. Tryptophan Metabolism in Rat Liver After Administration of Tryptophan, Kynurenine Metabolites, and Kynureninase Inhibitors. Int. J. Tryptophan Res. 2016, 9, 51–65. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rothschild, J.A.; Bishop, D.J. Effects of Dietary Supplements on Adaptations to Endurance Training. Sports Med. 2020, 50, 25–53. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Segura, R.; Ventura, J. Effect of L-Tryptophan Supplementation on Exercise Performance. Int. J. Sports Med. 1988, 9, 301–305. [Google Scholar] [CrossRef] [PubMed]
- Chaouloff, F. About the effect of L-tryptophan on exercise performance: Lacunae and pitfalls. Int. J. Sports Med. 1989, 10, 383. [Google Scholar] [CrossRef]
- Falabrègue, M.; Boschat, A.-C.; Jouffroy, R.; Derquennes, M.; Djemai, H.; Sanquer, S.; Barouki, R.; Coumoul, X.; Toussaint, J.-F.; Hermine, O.; et al. Lack of Skeletal Muscle Serotonin Impairs Physical Performance. Int. J. Tryptophan Res. 2021, 14. [Google Scholar] [CrossRef] [PubMed]
- Joisten, N.; Walzik, D.; Metcalfe, A.J.; Bloch, W.; Zimmer, P. Physical Exercise as Kynurenine Pathway Modulator in Chronic Diseases: Implications for Immune and Energy Homeostasis. Int. J. Tryptophan Res. 2020, 13, 13. [Google Scholar] [CrossRef]
- Pal, A.; Zimmer, P.; Clauss, D.; Schmidt, M.E.; Ulrich, C.M.; Wiskemann, J.; Steindorf, K. Resistance Exercise Modulates Kynurenine Pathway in Pancreatic Cancer Patients. Endoscopy 2021, 42, 33–40. [Google Scholar] [CrossRef]
- Trepci, A.; Imbeault, S.; Wyckelsma, V.L.; Westerblad, H.; Hermansson, S.; Andersson, D.C.; Piehl, F.; Venckunas, T.; Brazaitis, M.; Kamandulis, S.; et al. Quantification of Plasma Kynurenine Metabolites Following One Bout of Sprint Interval Exercise. Int. J. Tryptophan Res. 2020, 13. [Google Scholar] [CrossRef] [PubMed]
- Saran, T.; Turska, M.; Kocki, T.; Zawadka, M.; Zieliński, G.; Turski, W.A.; Gawda, P. Effect of 4-week physical exercises on tryptophan, kynurenine and kynurenic acid content in human sweat. Sci. Rep. 2021, 11, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Schenk, A.; Esser, T.; Knoop, A.; Thevis, M.; Herden, J.; Heidenreich, A.; Bloch, W.; Joisten, N.; Zimmer, P. Effect of a Single Bout of Aerobic Exercise on Kynurenine Pathway Metabolites and Inflammatory Markers in Prostate Cancer Patients—A Pilot Randomized Controlled Trial. Metabolites 2020, 11, 4. [Google Scholar] [CrossRef] [PubMed]
- Mina, A.; LeClair, R.A.; LeClair, K.B.; Cohen, D.E.; Lantier, L.; Banks, A.S. CalR: A Web-Based Analysis Tool for Indirect Calorimetry Experiments. Cell Metab. 2018, 28, 656–666.e1. [Google Scholar] [CrossRef] [Green Version]
- Jacobs, K.; Lim, E.; Blennow, K.; Zetterberg, H.; Chatterjee, P.; Martins, R.; Brew, B.J.; Guillemin, G.J.; Lovejoy, D.B. Correlation between plasma and CSF concentrations of kynurenine pathway metabolites in Alzheimer’s disease and relationship to amyloid-β and tau. Neurobiol. Aging 2019, 80, 11–20. [Google Scholar] [CrossRef] [PubMed]
- Midttun, Ø.; Hustad, S.; Ueland, P.M. Quantitative profiling of biomarkers related to B-vitamin status, tryptophan metabolism and inflammation in human plasma by liquid chromatography/tandem mass spectrometry. Rapid Commun. Mass Spectrom. 2009, 23, 1371–1379. [Google Scholar] [CrossRef] [PubMed]
- Motulsky, H.J.; Brown, E.R. Detecting outliers when fitting data with nonlinear regression–a new method based on robust nonlinear regression and the false discovery rate. BMC Bioinform. 2006, 7, 123. [Google Scholar] [CrossRef] [PubMed] [Green Version]






Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Valente-Silva, P.; Cervenka, I.; Ferreira, D.M.S.; Correia, J.C.; Edman, S.; Horwath, O.; Heng, B.; Chow, S.; Jacobs, K.R.; Guillemin, G.J.; et al. Effects of Tryptophan Supplementation and Exercise on the Fate of Kynurenine Metabolites in Mice and Humans. Metabolites 2021, 11, 508. https://doi.org/10.3390/metabo11080508
Valente-Silva P, Cervenka I, Ferreira DMS, Correia JC, Edman S, Horwath O, Heng B, Chow S, Jacobs KR, Guillemin GJ, et al. Effects of Tryptophan Supplementation and Exercise on the Fate of Kynurenine Metabolites in Mice and Humans. Metabolites. 2021; 11(8):508. https://doi.org/10.3390/metabo11080508
Chicago/Turabian StyleValente-Silva, Paula, Igor Cervenka, Duarte M. S. Ferreira, Jorge C. Correia, Sebastian Edman, Oscar Horwath, Benjamin Heng, Sharron Chow, Kelly R. Jacobs, Gilles J. Guillemin, and et al. 2021. "Effects of Tryptophan Supplementation and Exercise on the Fate of Kynurenine Metabolites in Mice and Humans" Metabolites 11, no. 8: 508. https://doi.org/10.3390/metabo11080508
APA StyleValente-Silva, P., Cervenka, I., Ferreira, D. M. S., Correia, J. C., Edman, S., Horwath, O., Heng, B., Chow, S., Jacobs, K. R., Guillemin, G. J., Blomstrand, E., & Ruas, J. L. (2021). Effects of Tryptophan Supplementation and Exercise on the Fate of Kynurenine Metabolites in Mice and Humans. Metabolites, 11(8), 508. https://doi.org/10.3390/metabo11080508