Diffuse Optical Monitoring of Cerebral Hemodynamics and Oxygen Metabolism during and after Cardiopulmonary Bypass: Hematocrit Correction and Neurological Vulnerability
Abstract
1. Introduction
2. Materials and Methods
2.1. Experimental Methods
2.1.1. Induction and Sedation
2.1.2. Monitoring Placement and Baseline Acquisition
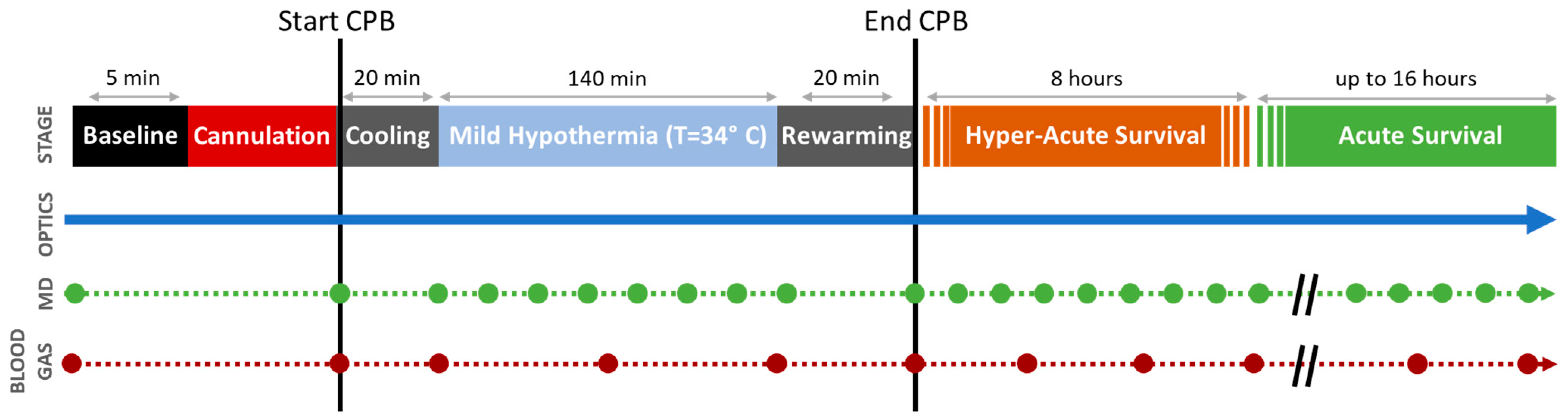
2.1.3. Initiation of Cardiopulmonary Bypass
2.1.4. Decannulation and Post-Operative Survival
2.2. Data Acquisition
2.2.1. Diffuse Optical Neuromonitoring
2.2.2. Cerebral Microdialysis
2.2.3. Blood Gas
2.3. Quantification of Cerebral Metabolic Rate of Oxygen (CMRO2)
2.3.1. Arterial Oxygen Content (CaO2)
2.3.2. Oxygen Extraction Fraction (OEF)
2.3.3. Cerebral Blood Flow (CBF)
2.4. Statistical Analysis
3. Results
3.1. Cardiopulmonary Bypass (CPB)
3.2. Hyper-Acute and Acute Survival Period
4. Discussion
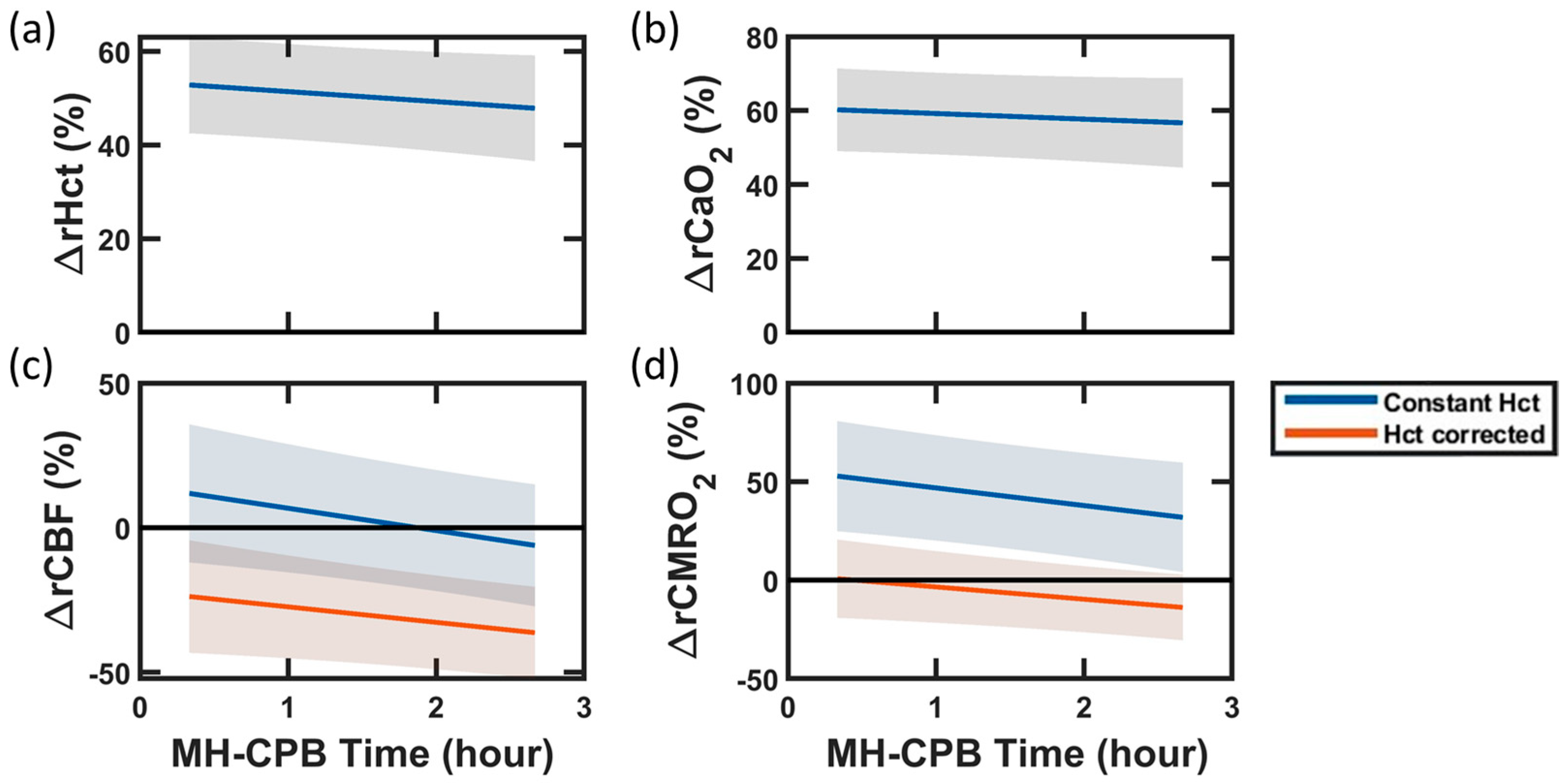
4.1. Improved Optical Quantification of CMRO2: Impact of Hematocrit
4.2. Effects of Cardiopulmonary Bypass
4.3. Cerebral Metabolism and Injury during Post-CPB Survival (0–24 h)
4.4. Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Huisenga, D.; La Bastide-Van Gemert, S.; Van Bergen, A.; Sweeney, J.; Hadders-Algra, M. Developmental outcomes after early surgery for complex congenital heart disease: A systematic review and meta-analysis. Dev. Med. Child Neurol. 2021, 63, 29–46. [Google Scholar] [CrossRef]
- Gaynor, J.W.; Stopp, C.; Wypij, D.; Andropoulos, D.B.; Atallah, J.; Atz, A.M.; Beca, J.; Donofrio, M.T.; Duncan, K.; Ghanayem, N.S.; et al. Neurodevelopmental Outcomes After Cardiac Surgery in Infancy. Pediatrics 2015, 135, 816–825. [Google Scholar] [CrossRef]
- Wernovsky, G.; Shillingford, A.J.; Gaynor, J.W. Central nervous system outcomes in children with complex congenital heart disease. Curr. Opin. Cardiol. 2005, 20, 94–99. [Google Scholar] [CrossRef] [PubMed]
- White, B.R.; Rogers, L.S.; Kirschen, M.P. Recent advances in our understanding of neurodevelopmental outcomes in congenital heart disease. Curr. Opin. Pediatr. 2019, 31, 783–788. [Google Scholar]
- Albers, E.L.; Bichell, D.P.; McLaughlin, B. New Approaches to Neuroprotection in Infant Heart Surgery. Pediatr. Res. 2010, 68, 1–9. [Google Scholar] [CrossRef]
- Kansy, A.; Tobota, Z.; Maruszewski, P.; Maruszewski, B. Analysis of 14,843 Neonatal Congenital Heart Surgical Procedures in the European Association for Cardiothoracic Surgery Congenital Database. Ann. Thorac. Surg. 2010, 89, 1255–1259. [Google Scholar] [CrossRef]
- Gaynor, J.W.; Stopp, C.; Wypij, D.; Andropoulos, D.B.; Atallah, J.; Atz, A.M.; Beca, J.; Donofrio, M.T.; Duncan, K.; Ghanayem, N.S.; et al. Impact of Operative and Postoperative Factors on Neurodevelopmental Outcomes After Cardiac Operations. Ann. Thorac. Surg. 2016, 102, 843–849. [Google Scholar] [CrossRef]
- Volk, L.E. Increased cerebral mitochondrial dysfunction and reactive oxygen species with cardiopulmonary bypass. Eur. J. Cardio-Thorac. Surg. 2021, 59, 1256–1264. [Google Scholar] [CrossRef]
- Mavroudis, C.D.; Ko, T.; Volk, L.E.; Smood, B.; Morgan, R.W.; Lynch, J.M.; Davarajan, M.; Boorady, T.W.; Licht, D.J.; Gaynor, J.W.; et al. Does supply meet demand? A comparison of perfusion strategies on cerebral metabolism in a neonatal swine model. J. Thorac. Cardiovasc. Surg. 2022, 163, e47–e58. [Google Scholar] [CrossRef] [PubMed]
- Parr, G.V.; Blackstone, E.H.; Kirklin, J.W. Cardiac performance and mortality early after intracardiac surgery in infants and young children. Circulation 1975, 51, 867–874. [Google Scholar] [CrossRef] [PubMed]
- Wernovsky, G.; Wypij, D.; Jonas, R.A.; MayerJr, J.E.; Hanley, F.L.; Hickey, P.R.; Walsh, A.Z.; Chang, A.C.; Castañeda, A.R.; Newburger, J.W.; et al. Postoperative Course and Hemodynamic Profile After the Arterial Switch Operation in Neonates and Infants. Circulation 1995, 92, 2226–2235. [Google Scholar] [CrossRef] [PubMed]
- Yuerek, M.; Rossano, J.W.; Mascio, C.E.; Shaddy, R.E. Postoperative management of heart failure in pediatric patients. Expert Rev. Cardiovasc. Ther. 2016, 14, 201–215. [Google Scholar] [CrossRef]
- Bojan, M.; Bonaveglio, E.; Dolcino, A.; Mirabile, C. Somatic and cerebral near infrared spectroscopy for the monitoring of perfusion during neonatal cardiopulmonary bypass. Interdiscip. CardioVascular Thorac. Surg. 2019, 29, 955–959. [Google Scholar] [CrossRef]
- Vranken, N.P.A.; Weerwind, P.W.; Sutedja, N.A.; Ševerdija, E.E.; Barenbrug, P.J.C.; Maessen, J.G. Cerebral Oximetry and Autoregulation during Cardiopulmonary Bypass: A Review. J. Extra-Corporeal Technol. 2017, 49, 182–191. [Google Scholar]
- Teng, Y.; Ding, H.; Gong, Q.; Jia, Z.; Huang, L. Monitoring cerebral oxygen saturation duringcardiopulmonary bypass using near-infraredspectroscopy: The relationships with bodytemperature and perfusion rate. Biomed. Opt. Express 2006, 11, 024016. [Google Scholar] [CrossRef]
- Feldmann, M.; Hagmann, C.; De Vries, L.; Disselhoff, V.; Pushparajah, K.; Logeswaran, T.; Jansen, N.J.G.; Breur, J.M.P.J.; Knirsch, W.; Benders, M.; et al. Neuromonitoring, neuroimaging, and neurodevelopmental follow-up practices in neonatal congenital heart disease: A European survey. Pediatr. Res. 2023, 93, 168–175. [Google Scholar] [CrossRef] [PubMed]
- Hoffman, G.M. Pro: Near-Infrared Spectroscopy Should Be Used for All Cardiopulmonary Bypass. J. Cardiothorac. Vasc. Anesth. 2006, 20, 606–612. [Google Scholar] [CrossRef]
- Kurth, C.D.; Steven, J.M.; Nicolson, S.C.; Chance, B.; Delivoria-Papadopoulos, M. Kinetics of Cerebral Deoxygenation during Deep Hypothermic Circulatory Arrest in Neonates. Anesthesiology 1992, 77, 656–661. [Google Scholar] [CrossRef]
- Wypij, D.; Newburger, J.W.; Rappaport, L.A.; Duplessis, A.J.; Jonas, R.A.; Wernovsky, G.; Lin, M.; Bellinger, D.C. The effect of duration of deep hypothermic circulatory arrest in infant heart surgery on late neurodevelopment: The Boston Circulatory Arrest Trial. J. Thorac. Cardiovasc. Surg. 2003, 126, 1397–1403. [Google Scholar] [CrossRef]
- Zaleski, K.L.; Kussman, B.D. Near-Infrared Spectroscopy in Pediatric Congenital Heart Disease. J. Cardiothorac. Vasc. Anesth. 2020, 34, 489–500. [Google Scholar] [CrossRef]
- Finucane, E.; Jooste, E.; Machovec, K.A. Neuromonitoring Modalities in Pediatric Cardiac Anesthesia: A Review of the Literature. J. Cardiothorac. Vasc. Anesth. 2020, 34, 3420–3428. [Google Scholar] [CrossRef]
- Jain, V.; Buckley, E.M.; Licht, D.J.; Lynch, J.M.; Schwab, P.J.; Naim, M.Y.; Lavin, N.A.; Nicolson, S.C.; Montenegro, L.M.; Yodh, A.G.; et al. Cerebral Oxygen Metabolism in Neonates with Congenital Heart Disease Quantified by MRI and Optics. J. Cereb. Blood Flow Metab. 2014, 34, 380–388. [Google Scholar] [CrossRef]
- Ko, T.S.; Mavroudis, C.D.; Baker, W.B.; Morano, V.C.; Mensh-Brown, K.; Boorady, T.W.; Schmidt, A.L.; Lynch, J.M.; Bush, D.R.; Gentile, J.; et al. Non-invasive optical neuromonitoring of the temperature-dependence of cerebral oxygen metabolism during deep hypothermic cardiopulmonary bypass in neonatal swine. J. Cereb. Blood Flow Metab. 2020, 40, 187–203. [Google Scholar] [CrossRef]
- Giovannella, M.; Andresen, B.; Andersen, J.B.; El-Mahdaoui, S.; Contini, D.; Spinelli, L.; Torricelli, A.; Greisen, G.; Durduran, T.; Weigel, U.M.; et al. Validation of diffuse correlation spectroscopy against 15O-water PET for regional cerebral blood flow measurement in neonatal piglets. J. Cereb. Blood Flow Metab. 2020, 40, 2055–2065. [Google Scholar] [CrossRef]
- Durduran, T.; Zhou, C.; Buckley, E.M.; Kim, M.N.; Yu, G.; Choe, R.; Gaynor, J.W.; Spray, T.L.; Durning, S.M.; Mason, S.E.; et al. Optical measurement of cerebral hemodynamics and oxygen metabolism in neonates with congenital heart defects. J. Biomed. Opt. 2010, 15, 037004. [Google Scholar] [CrossRef] [PubMed]
- Buckley, E.M.; Hance, D.; Pawlowski, T.; Lynch, J.; Wilson, F.B.; Mesquita, R.C.; Durduran, T.; Diaz, L.K.; Putt, M.E.; Licht, D.J.; et al. Validation of diffuse correlation spectroscopic measurement of cerebral blood flow using phase-encoded velocity mapping magnetic resonance imaging. J. Biomed. Opt. 2012, 17, 037007. [Google Scholar] [CrossRef]
- Lynch, J.M.; Mavroudis, C.D.; Ko, T.S.; Jacobwitz, M.; Busch, D.R.; Xiao, R.; Nicolson, S.C.; Montenegro, L.M.; Gaynor, J.W.; Yodh, A.G.; et al. Association of Ongoing Cerebral Oxygen Extraction During Deep Hypothermic Circulatory Arrest With Postoperative Brain Injury. Semin. Thorac. Cardiovasc. Surg. 2022, 34, 1275–1284. [Google Scholar] [CrossRef] [PubMed]
- Busch, D.R.; Rusin, C.G.; Miller-Hance, W.; Kibler, K.; Baker, W.B.; Heinle, J.S.; Fraser, C.D.; Yodh, A.G.; Licht, D.J.; Brady, K.M. Continuous cerebral hemodynamic measurement during deep hypothermic circulatory arrest. Biomed. Opt. Express 2016, 7, 3461–3470. [Google Scholar] [CrossRef]
- Ferradal, S.L.; Yuki, K.; Vyas, R.; Ha, C.G.; Yi, F.; Stopp, C.; Wypij, D.; Cheng, H.H.; Newburger, J.W.; Kaza, A.K.; et al. Non-invasive Assessment of Cerebral Blood Flow and Oxygen Metabolism in Neonates during Hypothermic Cardiopulmonary Bypass: Feasibility and Clinical Implications. Sci. Rep. 2017, 7, 44117. [Google Scholar] [CrossRef]
- Zavriyev, A.I.; Kaya, K.; Farzam, P.; Farzam, P.Y.; Sunwoo, J.; Jassar, A.S.; Sundt, T.M.; Carp, S.A.; Franceschini, M.A.; Qu, J.Z. The role of diffuse correlation spectroscopy and frequency-domain near-infrared spectroscopy in monitoring cerebral hemodynamics during hypothermic circulatory arrests. JTCVS Tech. 2021, 7, 161–177. [Google Scholar] [CrossRef] [PubMed]
- Rajaram, A.; Milej, D.; Suwalski, M.; Yip, L.C.M.; Guo, L.R.; Chu, M.W.A.; Chui, J.; Diop, M.; Murkin, J.M.; Lawrence, K.S. Optical monitoring of cerebral perfusion and metabolism in adults during cardiac surgery with cardiopulmonary bypass. Biomed. Opt. Express 2020, 11, 2967–5981. [Google Scholar] [CrossRef] [PubMed]
- Buckley, E.M.; Lynch, J.M.; Goff, D.A.; Schwab, P.J.; Baker, W.B.; Durduran, T.; Busch, D.R.; Nicolson, S.C.; Montenegro, L.M.; Naim, M.Y.; et al. Early postoperative changes in cerebral oxygen metabolism following neonatal cardiac surgery: Effects of surgical duration. J. Thorac. Cardiovasc. Surg. 2013, 145, 196–205.e191. [Google Scholar] [CrossRef]
- Aronowitz, D.I.; Mavroudis, C.D.; Geoffrion, T.R.; Piel, S.; Benson, E.J.; Morton, S.R.; Starr, J.; Melchior, R.W.; Gaudio, H.A.; Degani, R.E.; et al. Early Impairment of Cerebral Bioenergetics after Cardiopulmonary Bypass in Neonatal Swine [Abstract]. In Proceedings of the 36th EACTS Annual Meeting, Milan, Italy, 5–8 October 2022. [Google Scholar]
- Gravlee, G.P.; Davis, R.F.; Hammon, J.; Kussman, B.D. Cardiopulmonary Bypass and Mechanical Support: Principles and Practice, 4th ed.; Lippincott Williams & Wilkins: Philadelphia, PA, USA, 2015; 912p. [Google Scholar]
- Wyatt, J.S.; Delpy, D.T.; Cope, M.; Wray, S.; Reynolds, E.O.R. Quantification of cerebral oxygenation and haemodynamics in sick newborn infants by near infrared spectrophotometry. Lancet 1986, 328, 1063–1066. [Google Scholar] [CrossRef]
- Anamaria, E.; Ekert Kabalin, A.; Balenović, T.; Valpotić, I.; Pavičić, Ž.; Valpotic, H. The influence of birth mass and age of suckling piglets on erythrocyte parameters. Vet. Arh. 2008, 78, 307–319. [Google Scholar]
- Ko, T.S.; Mavroudis, C.D.; Morgan, R.W.; Baker, W.B.; Marquez, A.M.; Boorady, T.W.; Devarajan, M.; Lin, Y.; Roberts, A.L.; Landis, W.P. Non-invasive diffuse optical neuromonitoring during cardiopulmonary resuscitation predicts return of spontaneous circulation. Sci. Rep. 2021, 11, 3828. [Google Scholar] [CrossRef]
- Durduran, T.; Choe, R.; Baker, W.B.; Yodh, A.G. Diffuse optics for tissue monitoring and tomography. Rep. Prog. Phys. 2010, 73, 076701. [Google Scholar] [CrossRef] [PubMed]
- Hutchinson, P.J.; Jalloh, I.; Helmy, A.; Carpenter, K.L.H.; Rostami, E.; Bellander, B.-M.; Boutelle, M.G.; Chen, J.W.; Claassen, J.; Dahyot-Fizelier, C.; et al. Consensus statement from the 2014 International Microdialysis Forum. Intensive Care Med. 2015, 41, 1517–1528. [Google Scholar] [CrossRef] [PubMed]
- Tisdall, M.M.; Smith, M. Cerebral microdialysis: Research technique or clinical tool. Br. J. Anaesth. 2006, 97, 18–25. [Google Scholar] [CrossRef] [PubMed]
- Nordström, C.-H. Cerebral energy metabolism and microdialysis in neurocritical care. Child’s Nerv. Syst. 2010, 26, 465–472. [Google Scholar] [CrossRef]
- Ko, T.S.; Mavroudis, C.D.; Benson, E.J.; Forti, R.M.; Melchior, R.W.; Boorady, T.W.; Morano, V.C.; Mensah-Brown, K.; Lin, Y.; Aronowitz, D.; et al. Correlation of Cerebral Microdialysis with Non-Invasive Diffuse Optical Cerebral Hemodynamic Monitoring during Deep Hypothermic Cardiopulmonary Bypass. Metabolites 2022, 12, 737. [Google Scholar] [CrossRef]
- Marcoux, J.; McArthur, D.A.; Miller, C.; Glenn, T.C.; Villablanca, P.; Martin, N.A.; Hovda, D.A.; Alger, J.R.; Vespa, P. Persistent metabolic crisis as measured by elevated cerebral microdialysis lactate-pyruvate ratio predicts chronic frontal lobe brain atrophy after traumatic brain injury. Crit. Care Med. 2008, 36, 2871–2877. [Google Scholar] [CrossRef] [PubMed]
- Merenda, A.; Gugliotta, M.; Holloway, R.; Levasseur, J.E.; Alessandri, B.; Sun, D.; Bullock, M.R. Validation of brain extracellular glycerol as an indicator of cellular membrane damage due to free radical activity after traumatic brain injury. J. Neurotrauma 2008, 25, 527–537. [Google Scholar] [CrossRef]
- Hillered, L.; Valtysson, J.; Enblad, P.; Persson, L. Interstitial glycerol as a marker for membrane phospholipid degradation in the acutely injured human brain. J. Neurol. Neurosurg. Psychiatry 1998, 64, 486–491. [Google Scholar] [CrossRef] [PubMed]
- Reinstrup, P.; Ståhl, N.; Mellergård, P.; Uski, T.; Ungerstedt, U.; Nordström, C.-H. Intracerebral Microdialysis in Clinical Practice: Baseline Values for Chemical Markers during Wakefulness, Anesthesia, and Neurosurgery. Neurosurgery 2000, 47, 701–710. [Google Scholar] [PubMed]
- Zeiler, F.A.; Thelin, E.P.; Helmy, A.; Czosnyka, M.; Hutchinson, P.J.A.; Menon, D.K. A systematic review of cerebral microdialysis and outcomes in TBI: Relationships to patient functional outcome, neurophysiologic measures, and tissue outcome. Acta Neurochir. 2017, 159, 2245–2273. [Google Scholar] [CrossRef]
- Hirota, K.; Murata, M.; Shingu, K. A proposal for a new temperature-corrected formula for the oxygen content of blood. JA Clin. Rep. 2020, 6, 62. [Google Scholar] [CrossRef]
- Culver, J.P.; Durduran, T.; Furuya, D.; Cheung, C.; Greenberg, J.H.; Yodh, A.G. Diffuse Optical Tomography of Cerebral Blood Flow, Oxygenation, and Metabolism in Rat during Focal Ischemia. J. Cereb. Blood Flow Metab. 2003, 23, 911–924. [Google Scholar] [CrossRef]
- Watzman, H.M.; Kurth, C.D.; Montenegro, L.M.; Rome, J.; Steven, J.M.; Nicolson, S.C. Arterial and Venous Contributions to Near-infrared Cerebral Oximetry. Anesthesiology 2000, 93, 947–953. [Google Scholar] [CrossRef]
- Ito, H.; Kanno, I.; Iida, H.; Hatazawa, J.; Shimosegawa, E.; Tamura, H.; Okudera, T. Arterial fraction of cerebral blood volume in humans measured by positron emission tomography. Ann. Nucl. Med. 2001, 15, 111–116. [Google Scholar] [CrossRef]
- Sathialingam, E.; Williams, E.K.; Lee, S.Y.; McCracken, C.E.; Lam, W.A.; Buckley, E.M. Hematocrit significantly confounds diffuse correlation spectroscopy measurements of blood flow. Biomed. Opt. Express 2020, 11, 4786–4799. [Google Scholar] [CrossRef]
- Boas, D.A.; Sakadžic, S.; Selb, J.; Farzam, P.; Franceschini, M.A.; Carp, S.A. Establishing the diffuse correlation spectroscopy signal relationship with blood flow. Neurophotonics 2016, 3, 031412. [Google Scholar] [CrossRef]
- Goldsmith, H.L.; Marlow, J.C. Flow behavior of erythrocytes. II. Particle motions in concentrated suspensions of ghost cells. J. Colloid Interface Sci. 1979, 71, 383–407. [Google Scholar] [CrossRef]
- Tang, J.; Erdener, S.E.; Li, B.; Fu, B.; Sakadzic, S.; Carp, S.A.; Lee, J.; Boas, D.A. Shear-induced diffusion of red blood cells measured with dynamic light scattering-optical coherence tomography. J. Biophotonics 2018, 11, e201700070. [Google Scholar] [CrossRef]
- Zou, M.; Yu, L.; Lin, R.; Feng, J.; Zhang, M.; Ning, S.; Cui, Y.; Li, J.; Li, L.; Ma, L.; et al. Cerebral Autoregulation Status in Relation to Brain Injury on Electroencephalogram and Magnetic Resonance Imaging in Children Following Cardiac Surgery. J. Am. Heart Assoc. 2023, 12, e028147. [Google Scholar] [CrossRef] [PubMed]
- Tu, L.N.; Hsieh, L.; Kajimoto, M.; Charette, K.; Kibiryeva, N.; Forero, A.; Hampson, S.; Marshall, J.A.; O’Brien, J.; Scatena, M.; et al. Shear stress associated with cardiopulmonary bypass induces expression of inflammatory cytokines and necroptosis in monocytes. JCI Insight 2021, 6, e141341. [Google Scholar] [CrossRef] [PubMed]
- Robert, S.M.; Borasino, S.; Dabal, R.J.; Cleveland, D.C.; Hock, K.M.; Alten, J.A. Postoperative Hydrocortisone Infusion Reduces the Prevalence of Low Cardiac Output Syndrome after Neonatal Cardiopulmonary Bypass. Pediatr. Crit. Care Med. 2015, 16, 629–636. [Google Scholar] [CrossRef]
- Kreimeier, U.; Messmer, K. Perioperative hemodilution. Transfus. Apher. Sci. 2002, 27, 59–72. [Google Scholar] [CrossRef]
- Campbell, J.A.; Holt, D.W.; Shostrom, V.K.; Durham, S.J. Influence of Intraoperative Fluid Volume on Cardiopulmonary Bypass Hematocrit and Blood Transfusions in Coronary Artery Bypass Surgery. J. Extracorpor. Technol. 2008, 40, 99–108. [Google Scholar] [CrossRef]
- O’Dwyer, C.; Prough, D.S.; Johnston, W.E. Determinants of cerebral perfusion during cardiopulmonary bypass. J. Cardiothorac. Vasc. Anesth. 1996, 10, 54–65. [Google Scholar] [CrossRef]
- Fraser, C.D.; Andropoulos, D.B. Principles of Antegrade Cerebral Perfusion during Arch Reconstruction in Newborns/Infants. Semin. Thorac. Cardiovasc. Surg. Pediatr. Card. Surg. Annu. 2008, 11, 61–68. [Google Scholar] [CrossRef][Green Version]
- Andropoulos, D.B.; Stayer, S.A.; McKenzie, E.D.; Fraser, C.D. Novel cerebral physiologic monitoring to guide low-flow cerebral perfusion during neonatal aortic arch reconstruction. J. Thorac. Cardiovasc. Surg. 2003, 125, 491–499. [Google Scholar] [CrossRef]
- Bulte, D.P.; Chiarelli, P.A.; Wise, R.G.; Jezzard, P. Cerebral Perfusion Response to Hyperoxia. J. Cereb. Blood Flow Metab. 2007, 27, 69–75. [Google Scholar] [CrossRef]
- Engelman, R.; Baker, R.A.; Likosky, D.S.; Grigore, A.; Dickinson, T.A.; Shore-Lesserson, L.; Hammon, J.W. The Society of Thoracic Surgeons, The Society of Cardiovascular Anesthesiologists, and The American Society of ExtraCorporeal Technology: Clinical Practice Guidelines for Cardiopulmonary Bypass–Temperature Management During Cardiopulmonary Bypass. J. Cardiothorac. Vasc. Anesth. 2015, 29, 1104–1113. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Yenari, M.A. Therapeutic hypothermia: Neuroprotective mechanisms. Front. Biosci. 2007, 12, 816–825. [Google Scholar] [CrossRef]
- Polderman, K.H. Mechanisms of action, physiological effects, and complications of hypothermia. Crit. Care Med. 2009, 37, S186–S202. [Google Scholar] [CrossRef]
- Saad, H.; Aladawy, M. Temperature management in cardiac surgery. Glob. Cardiol. Sci. Pract. 2013, 2013, 7. [Google Scholar] [CrossRef] [PubMed]
- Jessen, K. An Assessment of Human Regulatory Nonshivering Thermogenesis. Acta Anaesthesiol. Scand. 1980, 24, 138–143. [Google Scholar] [CrossRef] [PubMed]
- Muizelaar, J.P.; Bouma, G.J.; Levasseur, J.E.; Kontos, H.A. Effect of hematocrit variations on cerebral blood flow and basilar artery diameter in vivo. Am. J. Physiol.-Heart Circ. Physiol. 1992, 262, H949–H954. [Google Scholar] [CrossRef] [PubMed]
- Dewey, R.C.; Pieper, H.P.; Hunt, W.E. Experimental cerebral hemodynamics. J. Neurosurg. 1974, 41, 597–606. [Google Scholar] [CrossRef] [PubMed]
- Ho, M.-L.; Rojas, R.; Eisenberg, R.L. Cerebral Edema. Am. J. Roentgenol. 2012, 199, W258–W273. [Google Scholar] [CrossRef]
- Levy, J.H.; Tanaka, K.A. Inflammatory Response to Cardiopulmonary Bypass. Ann. Thorac. Surg. 2003, 75, S715–S720. [Google Scholar] [CrossRef] [PubMed]
- Yoshimura, A.; Shichita, T. Post-Ischemic Inflammation in the Brain. Front. Immunol. 2012, 3, 132. [Google Scholar]
- Gobiet, W.; Grote, W.; Bock, W.J. The relation between intracranial pressure, mean arterial pressure and cerebral blood flow in patients with severe head injury. Acta Neurochir. 1975, 32, 13–24. [Google Scholar] [CrossRef] [PubMed]
- Warltier, D.C.; Laffey, J.G.; Boylan, J.F.; Cheng, D.C. The Systemic Inflammatory Response to Cardiac Surgery: Implications for the Anesthesiologist. Anesthesiology 2002, 97, 215–252. [Google Scholar] [CrossRef] [PubMed]
- Flanders, T.M.; Lang, S.-S.; Ko, T.S.; Andersen, K.N.; Jahnavi, J.; Flibotte, J.J.; Licht, D.J.; Tasian, G.E.; Sotardi, S.T.; Yodh, A.G.; et al. Optical Detection of Intracranial Pressure and Perfusion Changes in Neonates With Hydrocephalus. J. Pediatr. 2021, 236, 54–61.e51. [Google Scholar] [CrossRef]
- Maruccia, F.; Tagliabue, S.; Fischer, J.B.; Kacprzak, M.; Pérez-Hoyos, S.; Rosas, K.; Álvarez, I.D.; Sahuquillo, J.; Durduran, T.; Poca, M.A. Transcranial optical monitoring for detecting intracranial pressure alterations in children with benign external hydrocephalus: A proof-of-concept study. Neurophotonics 2022, 9, 045005. [Google Scholar] [CrossRef]
- Ruesch, A.; Yang, J.; Schmitt, S.; Acharya, D.; Smith, M.A.; Kainerstorfer, J.M. Estimating intracranial pressure using pulsatile cerebral blood flow measured with diffuse correlation spectroscopy. Biomed. Opt. Express 2020, 11, 1462–1476. [Google Scholar] [CrossRef]
- Wu, K.-C.; Sunwoo, J.; Sheriff, F.; Farzam, P.; Farzam, P.Y.; Orihuela-Espina, F.; Larose, S.L.; Monk, A.D.; Aziz-Sultan, M.A.; Patel, N.; et al. Validation of diffuse correlation spectroscopy measures of critical closing pressure against transcranial Doppler ultrasound in stroke patients. J. Biomed. Opt. 2021, 26, 036008. [Google Scholar] [CrossRef]
- Urner, T.M.; Cowdrick, K.R.; Brothers, R.O.; Boodooram, T.; Zhao, H.; Goyal, V.; Sathialingam, E.; Quadri, A.; Turrentine, K.; Akbar, M.M.; et al. Normative cerebral microvascular blood flow waveform morphology assessed with diffuse correlation spectroscopy. Biomed. Opt. Express 2023, 14, 3635–3653. [Google Scholar] [CrossRef]
- Lafontant, A.; Mahanna Gabrielli, E.; Bergonzi, K.; Forti, R.M.; Ko, T.S.; Shah, R.M.; Arkles, J.S.; Licht, D.J.; Yodh, A.G.; Kofke, W.A.; et al. Comparison of optical measurements of critical closing pressure acquired before and during induced ventricular arrhythmia in adults. Neurophotonics 2022, 9, 035004. [Google Scholar] [CrossRef]
- Kawamura, S.; Yasui, N. Vascular response to hyperoxemia in rat brain surface microvessels. Neurol. Med.-Chir. 1996, 36, 156–161. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Beca, J.; Gunn, J.K.; Coleman, L.; Hope, A.; Reed, P.W.; Hunt, R.W.; Finucane, K.; Brizard, C.; Dance, B.; Shekerdemian, L.S. New White Matter Brain Injury After Infant Heart Surgery Is Associated With Diagnostic Group and the Use of Circulatory Arrest. Circulation 2013, 127, 971–979. [Google Scholar] [CrossRef]
- Lynch, J.M.; Ko, T.; Busch, D.R.; Newland, J.J.; Winters, M.E.; Mensah-Brown, K.; Boorady, T.W.; Xiao, R.; Nicolson, S.C.; Montenegro, L.M.; et al. Preoperative cerebral hemodynamics from birth to surgery in neonates with critical congenital heart disease. J. Thorac. Cardiovasc. Surg. 2018, 156, 1657–1664. [Google Scholar] [CrossRef] [PubMed]
- Takayama, J.; Teng, W.; Uyemoto, J.; Newman, T.; Pantell, R. Body temperature of newborns: What is normal? Clin. Pediatr. 2000, 39, 503–510. [Google Scholar] [CrossRef] [PubMed]
- Condo, S.G.; Corda, M.; Sanna, M.T.; Pellegrini, M.G.; Ruiz, M.P.; Castagnola, M.; Giardina, B. Molecular basis of low-temperature sensitivity in pig hemoglobins. Eur. J. Biochem. 1992, 209, 773–776. [Google Scholar] [CrossRef]
- Baik-Schneditz, N.; Schwaberger, B.; Urlesberger, B.; Wolfsberger, C.H.; Bruckner, M.; Pichler, G. Acid base and blood gas analysis in term neonates immediately after birth with uncomplicated neonatal transition. BMC Pediatr. 2022, 22, 271. [Google Scholar] [CrossRef] [PubMed]
- Jonas, R.; Wypij, D.; Roth, S.; Bellinger, D.; Visconti, K.; Du Plessis, A.; Goodkin, H.; Laussen, P.; Farrell, D.; Bartlett, J.; et al. The influence of hemodilution on outcome after hypothermic cardiopulmonary bypass: Results of a randomized trial in infants. J. Thorac. Cardiovasc. Surg. 2003, 126, 1765–1774. [Google Scholar] [CrossRef] [PubMed]
- Conrad, M.S.; Dilger, R.N.; Johnson, R.W. Brain Growth of the Domestic Pig (Sus scrofa) from 2 to 24 Weeks of Age: A Longitudinal MRI Study. Dev. Neurosci. 2012, 34, 291–298. [Google Scholar] [CrossRef] [PubMed]
- Semenas, E.; Nozari, A.; Wiklund, L. Sex differences in cardiac injury after severe haemorrhage and ventricular fibrillation in pigs. Resuscitation 2010, 81, 1718–1722. [Google Scholar] [CrossRef]
- Armstead, W.M.; Kiessling, J.W.; Kofke, W.A.; Vavilala, M.S. Impaired Cerebral Blood Flow Autoregulation During Postraumatic Arterial Hypotension After Fluid Percussion Brain Injury is Prevented by Phenylephrine in Female but Exacerbated in Male Piglets by ERK MAPK Upregulation. Crit. Care Med. 2012, 38, 1868–1874. [Google Scholar] [CrossRef]
- Vognsen, M.; Fabian-Jessing, B.K.; Secher, N.; Løfgren, B.; Dezfulian, C.; Andersen, L.W.; Granfeldt, A. Contemporary animal models of cardiac arrest: A systematic review. Resuscitation 2017, 113, 115–123. [Google Scholar] [CrossRef] [PubMed]
| Modality | Parameter | Baseline | Mild Hypothermic CPB | p-Value |
|---|---|---|---|---|
| Animal Characteristics | Length (cm) | 37 [36, 38], (n = 27) | - | - |
| Weight (kg) | 4.3 [4.1, 4.6], (n = 27) | - | - | |
| Vitals | MAP (mmHg) | 61 [53, 72], (n = 27) | 64 [57, 71], (n = 27) | 0.3 |
| Blood Gas | Hct (%) | 23 [20, 27], (n = 27) | 34 [33, 35], (n = 27) | <0.001 |
| CaO2 (mL O2/dL blood) | 10 [9, 12], (n = 26) | 16 [15, 16], (n = 26) | <0.001 | |
| SaO2 (%) | 98 [97, 98], (n = 26) | 100 [99.9, 100], (n = 26) | <0.001 | |
| PaO2 (mmHg) | 91 [84, 96], (n = 26) | 257 [237, 271], (n = 27) | <0.001 | |
| PaCO2 (mmHg) | 39 [37, 41], (n = 26) | 45 [42, 48], (n = 26) | <0.001 | |
| Glucose (mg/dL) | 102 [79, 117], (n = 27) | 150 [120, 184], (n = 27) | <0.001 | |
| Lactate (mmol/L) | 1.2 [1, 1.6], (n = 27) | 2.2 [1.6, 2.9], (n = 27) | <0.001 | |
| Optics: Physiologic Parameters | StO2 (%) | 54 [50, 57], (n = 27) | 61 [58, 64], (n = 27) | <0.001 |
| OEF (%) | 62 [56, 66], (n = 26) | 55 [50, 58], (n = 26) | 0.002 | |
| rCBF (% Baseline) | 100, (n = 27) | 63 [41, 89], (n = 27) | 0.004 | |
| rCMRO2 (% Baseline) | 100, (n = 26) | 91 [57, 135], (n = 26) | 0.5 | |
| CBV (μL/g brain tissue) | 53 [49, 67], (n = 27) | 48 [38, 51], (n = 27) | <0.001 | |
| THC (μM) | 68 [61, 82], (n = 27) | 82 [74, 89], (n = 27) | <0.001 | |
| Optics: Mie Scattering Parameters | A | 14 [12, 17], (n = 27) | 14 [10, 16], (n = 27) | 0.6 |
| b | 1.1 [0.95, 1.3], (n = 27) | 0.98 [0.80, 1.23], (n = 27) | 0.2 | |
| μs′(785 nm) | 8.3 [7.8, 9.7], (n = 27) | 8.3 [7.1, 9.8], (n = 27) | 0.8 | |
| Microdialysis | LPR | 17 [15, 20], (n = 20) | 15 [12, 20], (n = 25) | 0.12 |
| Lactate (mM) | 0.82 [0.63, 0.99], (n = 19) | 0.65 [0.45, 0.86], (n = 25) | 0.3 | |
| Pyruvate (μM) | 46 [33, 58], (n = 22) | 35 [28, 54], (n = 27) | 0.3 | |
| Glycerol (μM) | 21 [17, 31], (n = 21) | 22 [17, 24], (n = 27) | 0.7 | |
| Glucose (μM) | 12 [8, 19], (n = 20) | 11 [6, 15], (n = 26) | 0.17 |
| Parameter | LME Y-Intercept | LME Slope | ||||
|---|---|---|---|---|---|---|
| Value (%) | 95% CI | p-Value | Value (%/h) | 95% CI | p-Value | |
| ΔrMAP | +8 | (−2, 18) | 0.1 | −1.0 | (−4.1, 2.2) | 0.6 |
| ΔrHCT | +53 | (42, 63) | <0.001 | −2.1 | (−5.0, 0.9) | 0.16 |
| ΔrCaO2 | +60 | (48, 72) | <0.001 | −1.4 | (−4.4, 1.5) | 0.3 |
| ΔrStO2 | +13 | (7, 19) | <0.001 | −1.6 | (−3.4, 0.2) | 0.09 |
| ΔrOEF | −12 | (−16, −8) | <0.001 | +2.2 | (0.0, 4.4) | 0.046 |
| ΔrCBF | −22 | (−42, −2) | 0.03 | −5.5 | (−10.5, −0.5) | 0.03 |
| ΔrCMRO2 | +4 | (−17, 25) | 0.7 | −6.7 | (−12.2, −1.2) | 0.02 |
| ΔrCBV | −21 | (−26, −16) | <0.001 | +2.0 | (0.4, 3.6) | 0.01 |
| ΔrTHC | +17 | (13, 21) | <0.001 | +0.7 | (−0.6, 1.9) | 0.3 |
| ΔrA | −6 | (−11, −1) | 0.017 | −1.3 | (−3.0, 0.5) | 0.2 |
| Δrb | −8 | (−17, 1) | 0.1 | −1.8 | (−5.3, 1.7) | 0.3 |
| Δrμs’(785 nm) | −1.8 | (−5.0, 1.4) | 0.3 | 0.2 | (−5.8, 1.4) | 0.7 |
| ΔrLactate | +17 | (−27, 62) | 0.4 | −1.7 | (−20, 17) | 0.9 |
| ΔrPyruvate | −5 | (−27, 18) | 0.7 | 9.5 | (−8, 27) | 0.3 |
| ΔrLPR | +48 | (−42, 138) | 0.3 | −23 | (−65, 18) | 0.2 |
| ΔrGlycerol | +11 | (−205, 226) | 0.9 | +51 | (−86, 187) | 0.5 |
| ΔrGlucose | +47 | (−23, 122) | 0.2 | −18 | (−57, 24) | 0.4 |
| Parameter | Time after Decannulation | LME Y-Intercept | LME Slope | ||||
|---|---|---|---|---|---|---|---|
| Value (%) | 95% CI | p-Value | Value (%/h) | 95% CI | p-Value | ||
| ΔrMAP | 0–8 h | +12 | (−5, 29) | 0.2 | +0.6 | (−1.2, 2.4) | 0.5 |
| 8–24 h | +13 | (1, 24) | 0.03 | −0.0 | (−0.1, −0.0) | <0.001 | |
| ΔrHct | 0–8 h | +53 | (50, 55) | <0.001 | −0.3 | (−1.2, 0.7) | 0.6 |
| 8–24 h | +51 | (40, 62) | <0.001 | −0.4 | (−0.5, −0.2) | <0.001 | |
| ΔrCaO2 | 0–8 h | +60 | (49, 71) | <0.001 | −0.5 | (−0.6, −0.4) | <0.001 |
| 8–24 h | +57 | (46, 68) | <0.001 | −0.6 | (−0.7, −0.5) | <0.001 | |
| ΔrStO2 | 0–8 h | +6 | (2, 10) | 0.003 | −1.0 | (−1.4, −0.6) | <0.001 |
| 8–24 h | −2 | (−6, 1) | 0.2 | −0.0 | (−0.6, 0.6) | 0.9 | |
| ΔrOEF | 0–8 h | −2 | (−6, 3) | 0.5 | +1.0 | (0.6, 1.4) | <0.001 |
| 8–24 h | +7 | (2, 12) | 0.003 | −0.1 | (−0.8, 0.6) | 0.8 | |
| ΔrCBF | 0–8 h | −28 | (−42, −14) | <0.001 | −2.1 | (−3.6, −0.6) | 0.005 |
| 8–24 h | −44 | (−56, −33) | <0.001 | +0.7 | (−0.0, 1.4) | 0.05 | |
| ΔrCMRO2 | 0–8 h | +9 | (−10, 28) | 0.3 | −3.0 | (−5.2, −0.8) | 0.008 |
| 8–24 h | −12 | (−27, 2) | 0.09 | +0.6 | (−0.2, 1.4) | 0.1 | |
| ΔrCBV | 0–8 h | −20 | (−28, −11) | <0.001 | −1.0 | (−1.5, −0.5) | <0.001 |
| 8–24 h | −28 | (−38, −18) | <0.001 | +0.1 | (−0.4, 0.6) | 0.7 | |
| ΔrTHC | 0–8 h | +18 | (11, 26) | <0.001 | −1.7 | (−2.4, −1.1) | <0.001 |
| 8–24 h | +4 | (−6, 14) | 0.5 | +0.1 | (−0.6, 0.8) | 0.8 | |
| ΔrA | 0–8 h | −6 | (−13, 0.6) | 0.08 | +0.1 | (−1.0, 1.2) | 0.8 |
| 8–24 h | −7 | (−18, 5) | 0.2 | +0.7 | (−1.3, 2.7) | 0.5 | |
| Δrb | 0–8 h | −6 | (−23, 10) | 0.4 | +1.1 | (−1.4, 3.6) | 0.4 |
| 8–24 h | −1 | (−20, 18) | 0.9 | +1.1 | (−2.5, 4.7) | 0.6 | |
| Δrμs’(785 nm) | 0–8 h | −1 | (−6, 3) | 0.5 | −0.3 | (−1.3, 0.6) | 0.5 |
| 8–24 h | −5 | (−15, 5) | 0.3 | +0.7 | (−1.3, 2.7) | 0.2 | |
| ΔrLactate | 0–8 h | +127 | (−152, 406) | 0.4 | +19 | (−20, 57) | 0.3 |
| 8–24 h | +191 | (−144, 526) | 0.3 | −0.2 | (−4.3, 3.9) | 0.9 | |
| ΔrPyruvate | 0–8 h | +87 | (−21, 195) | 0.1 | +1.5 | (−2.2, 5.3) | 0.4 |
| 8–24 h | +97 | (−33, 228) | 0.1 | −4.2 | (−9.6, 1.1) | 0.1 | |
| ΔrLPR | 0–8 h | −5 | (−40, 29) | 0.8 | +5.0 | (−0.7, 10.7) | 0.08 |
| 8–24 h | +2 | (−39, 43) | 0.9 | +4.0 | (−11.7, 19.6) | 0.6 | |
| ΔrGlycerol | 0–8 h | +48 | (7, 90) | 0.02 | +9.3 | (−14.7, 33.3) | 0.4 |
| 8–24 h | +25 | (−15, 64) | 0.2 | −2.0 | (−8.0, 4.1) | 0.5 | |
| ΔrGlucose | 0–8 h | −0 | (−94, 93) | >0.99 | +2.1 | (−21.6, 25.8) | 0.9 |
| 8–24 h | +60 | (−101, 221) | 0.5 | +1.8 | (−10.3, 14.0) | 0.8 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Benson, E.J.; Aronowitz, D.I.; Forti, R.M.; Lafontant, A.; Ranieri, N.R.; Starr, J.P.; Melchior, R.W.; Lewis, A.; Jahnavi, J.; Breimann, J.; et al. Diffuse Optical Monitoring of Cerebral Hemodynamics and Oxygen Metabolism during and after Cardiopulmonary Bypass: Hematocrit Correction and Neurological Vulnerability. Metabolites 2023, 13, 1153. https://doi.org/10.3390/metabo13111153
Benson EJ, Aronowitz DI, Forti RM, Lafontant A, Ranieri NR, Starr JP, Melchior RW, Lewis A, Jahnavi J, Breimann J, et al. Diffuse Optical Monitoring of Cerebral Hemodynamics and Oxygen Metabolism during and after Cardiopulmonary Bypass: Hematocrit Correction and Neurological Vulnerability. Metabolites. 2023; 13(11):1153. https://doi.org/10.3390/metabo13111153
Chicago/Turabian StyleBenson, Emilie J., Danielle I. Aronowitz, Rodrigo M. Forti, Alec Lafontant, Nicolina R. Ranieri, Jonathan P. Starr, Richard W. Melchior, Alistair Lewis, Jharna Jahnavi, Jake Breimann, and et al. 2023. "Diffuse Optical Monitoring of Cerebral Hemodynamics and Oxygen Metabolism during and after Cardiopulmonary Bypass: Hematocrit Correction and Neurological Vulnerability" Metabolites 13, no. 11: 1153. https://doi.org/10.3390/metabo13111153
APA StyleBenson, E. J., Aronowitz, D. I., Forti, R. M., Lafontant, A., Ranieri, N. R., Starr, J. P., Melchior, R. W., Lewis, A., Jahnavi, J., Breimann, J., Yun, B., Laurent, G. H., Lynch, J. M., White, B. R., Gaynor, J. W., Licht, D. J., Yodh, A. G., Kilbaugh, T. J., Mavroudis, C. D., ... Ko, T. S. (2023). Diffuse Optical Monitoring of Cerebral Hemodynamics and Oxygen Metabolism during and after Cardiopulmonary Bypass: Hematocrit Correction and Neurological Vulnerability. Metabolites, 13(11), 1153. https://doi.org/10.3390/metabo13111153









