Targeted Metabolomics Analysis of Individuals Carrying the ANGPTL8 R59W Variant
Abstract
:1. Introduction
2. Materials and Methods
2.1. Participant Recruitment
2.2. Collection and Processing of Blood Samples
2.3. Targeted Genotyping of the ANGPTL8 Study Variant rs2278426
2.4. Metabolomics
2.5. Statistical Analysis
3. Results
3.1. Clinical Characteristics of the Study Cohort and the Sub-Cohorts
3.2. Differences in the Levels of Metabolites between Subjects with the Reference CC Genotype and Those with the Carrier (CT+TT) Genotypes
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Magliano, D.J.; Islam, R.M.; Barr, E.L.M.; Gregg, E.W.; Pavkov, M.E.; Harding, J.L.; Tabesh, M.; Koye, D.N.; Shaw, J.E. Trends in incidence of total or type 2 diabetes: Systematic review. BMJ 2019, 366, l5003. [Google Scholar] [CrossRef] [PubMed]
- Forouhi, N.G.; Wareham, N.J. Epidemiology of diabetes. Medicine 2014, 42, 698–702. [Google Scholar] [CrossRef] [PubMed]
- Zhang, R.; Abou-Samra, A.B. A dual role of lipasin (betatrophin) in lipid metabolism and glucose homeostasis: Consensus and controversy. Cardiovasc. Diabetol. 2014, 13, 133. [Google Scholar] [CrossRef]
- Ren, G.; Kim, J.Y.; Smas, C.M.; Solomon, G.; Atkins, A.; Shahar, R.; Gertler, A.; Monsonego-Ornan, E. Identification of RIFL, a novel adipocyte-enriched insulin target gene with a role in lipid metabolism. Am. J. Physiol. Endocrinol. Metab. 2012, 303, E334–E351. [Google Scholar] [CrossRef] [PubMed]
- Abu-Farha, M.; Abubaker, J.; Al-Khairi, I.; Cherian, P.; Noronha, F.; Hu, F.B.; Behbehani, K.; Elkum, N. Higher plasma betatrophin/ANGPTL8 level in Type 2 Diabetes subjects does not correlate with blood glucose or insulin resistance. Sci. Rep. 2015, 5, 10949. [Google Scholar] [CrossRef]
- Abu-Farha, M.; Abubaker, J.; Noronha, F.; Al-Khairi, I.; Cherian, P.; Alarouj, M.; Bennakhi, A.; Elkum, N. Lack of associations between betatrophin/ANGPTL8 level and C-peptide in type 2 diabetic subjects. Cardiovasc. Diabetol. 2015, 14, 112. [Google Scholar] [CrossRef]
- Yamada, H.; Saito, T.; Aoki, A.; Asano, T.; Yoshida, M.; Ikoma, A.; Kusaka, I.; Toyoshima, H.; Kakei, M.; Ishikawa, S.-E. Circulating betatrophin is elevated in patients with type 1 and type 2 diabetes. Endocr. J. 2015, 62, 417–421. [Google Scholar] [CrossRef]
- Fu, Z.; Berhane, F.; Fite, A.; Seyoum, B.; Abou-Samra, A.B.; Zhang, R. Elevated circulating lipasin/betatrophin in human type 2 diabetes and obesity. Sci. Rep. 2014, 4, 5013. [Google Scholar] [CrossRef]
- Hu, H.; Sun, W.; Yu, S.; Hong, X.; Qian, W.; Tang, B.; Wang, D.; Yang, L.; Wang, J.; Mao, C.; et al. Increased Circulating Levels of Betatrophin in Newly Diagnosed Type 2 Diabetic Patients. Diabetes Care 2014, 37, 2718–2722. [Google Scholar] [CrossRef]
- Richardson, T.G.; Leyden, G.M.; Wang, Q.; Bell, J.A.; Elsworth, B.; Davey Smith, G.; Holmes, M.V. Characterising metabolomic signatures of lipid-modifying therapies through drug target mendelian randomization. PLoS Biol. 2022, 20, e3001547. [Google Scholar] [CrossRef]
- Richardson, T.G.; Sanderson, E.; Palmer, T.M.; Ala-Korpela, M.; Ference, B.A.; Smith, G.D.; Holmes, M.V. Evaluating the relationship between circulating lipoprotein lipids and apolipoproteins with risk of coronary heart disease: A multivariable Mendelian randomisation analysis. PLOS Med. 2020, 17, e1003062. [Google Scholar] [CrossRef]
- Koskeridis, F.; Evangelou, E.; Said, S.; Boyle, J.J.; Elliott, P.; Dehghan, A.; Tzoulaki, I. Pleiotropic genetic architecture and novel loci for C-reactive protein levels. Nat. Commun. 2022, 13, 6939. [Google Scholar] [CrossRef] [PubMed]
- Sakaue, S.; Kanai, M.; Tanigawa, Y.; Karjalainen, J.; Kurki, M.; Koshiba, S.; Narita, A.; Konuma, T.; Yamamoto, K.; Akiyama, M.; et al. A cross-population atlas of genetic associations for 220 human phenotypes. Nat. Genet. 2021, 53, 1415–1424. [Google Scholar] [CrossRef]
- Moon, S.; Kim, Y.J.; Han, S.; Hwang, M.Y.; Shin, D.M.; Park, M.Y.; Lu, Y.; Yoon, K.; Jang, H.-M.; Kim, Y.K.; et al. The Korea Biobank Array: Design and Identification of Coding Variants Associated with Blood Biochemical Traits. Sci. Rep. 2019, 9, 1382. [Google Scholar] [CrossRef]
- Lee, C.-J.; Chen, T.-H.; Lim, A.M.W.; Chang, C.-C.; Sie, J.-J.; Chen, P.-L.; Chang, S.-W.; Wu, S.-J.; Hsu, C.-L.; Hsieh, A.-R.; et al. Phenome-wide analysis of Taiwan Biobank reveals novel glycemia-related loci and genetic risks for diabetes. Commun. Biol. 2022, 5, 1175. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.J.; Moon, S.; Hwang, M.Y.; Han, S.; Jang, H.-M.; Kong, J.; Shin, D.M.; Yoon, K.; Kim, S.M.; Lee, J.-E.; et al. The contribution of common and rare genetic variants to variation in metabolic traits in 288,137 East Asians. Nat. Commun. 2022, 13, 6642. [Google Scholar] [CrossRef] [PubMed]
- Kanai, M.; Akiyama, M.; Takahashi, A.; Matoba, N.; Momozawa, Y.; Ikeda, M.; Iwata, N.; Ikegawa, S.; Hirata, M.; Matsuda, K.; et al. Genetic analysis of quantitative traits in the Japanese population links cell types to complex human diseases. Nat. Genet. 2018, 50, 390–400. [Google Scholar] [CrossRef]
- Weissglas-Volkov, D.; Aguilar-Salinas, C.A.; Nikkola, E.; Deere, K.A.; Cruz-Bautista, I.; Arellano-Campos, O.; Muñoz-Hernandez, L.L.; Gomez-Munguia, L.; Ordoñez-Sánchez, M.L.; Reddy, P.M.V.L.; et al. Genomic study in Mexicans identifies a new locus for triglycerides and refines European lipid loci. J. Med. Genet. 2013, 50, 298–308. [Google Scholar] [CrossRef]
- Nielsen, J.B.; Rom, O.; Surakka, I.; Graham, S.E.; Zhou, W.; Roychowdhury, T.; Fritsche, L.G.; Taliun, S.A.G.; Sidore, C.; Liu, Y.; et al. Loss-of-function genomic variants highlight potential therapeutic targets for cardiovascular disease. Nat. Commun. 2020, 11, 6417. [Google Scholar] [CrossRef]
- Sinnott-Armstrong, N.; Tanigawa, Y.; Amar, D.; Mars, N.; Benner, C.; Aguirre, M.; Venkataraman, G.R.; Wainberg, M.; Ollila, H.M.; Kiiskinen, T.; et al. Genetics of 35 blood and urine biomarkers in the UK Biobank. Nat. Genet. 2021, 53, 185–194. [Google Scholar] [CrossRef]
- Hu, Y.; Graff, M.; Haessler, J.; Buyske, S.; Bien, S.A.; Tao, R.; Highland, H.M.; Nishimura, N.K.; Zubair, N.; Lu, Y. Minority-centric meta-analyses of blood lipid levels identify novel loci in the Population Architecture using Genomics and Epidemiology (PAGE) study. PLoS Genet. 2020, 16, e1008684. [Google Scholar] [CrossRef] [PubMed]
- Wojcik, G.L.; Graff, M.; Nishimura, K.K.; Tao, R.; Haessler, J.; Gignoux, C.R.; Highland, H.M.; Patel, Y.M.; Sorokin, E.P.; Avery, C.L.; et al. Genetic analyses of diverse populations improves discovery for complex traits. Nature 2019, 570, 514–518. [Google Scholar] [CrossRef]
- Klarin, D.; Damrauer, S.M.; Cho, K.; Sun, Y.V.; Teslovich, T.M.; Honerlaw, J.; Gagnon, D.R.; Duvall, S.L.; Li, J.; Peloso, G.M.; et al. Genetics of blood lipids among ~300,000 multi-ethnic participants of the Million Veteran Program. Nat. Genet. 2018, 50, 1514–1523. [Google Scholar] [CrossRef]
- Kichaev, G.; Bhatia, G.; Loh, P.R.; Gazal, S.; Burch, K.; Freund, M.K.; Schoech, A.; Pasaniuc, B.; Price, A.L. Leveraging Polygenic Functional Enrichment to Improve GWAS Power. Am. J. Hum. Genet. 2019, 104, 65–75. [Google Scholar] [CrossRef] [PubMed]
- Abu-Farha, M.; Melhem, M.; Abubaker, J.; Behbehani, K.; Alsmadi, O.; Elkum, N. ANGPTL8/Betatrophin R59W variant is associated with higher glucose level in non-diabetic Arabs living in Kuwait. Lipids Health Dis. 2016, 15, 26. [Google Scholar] [CrossRef] [PubMed]
- Abu-Farha, M.; Abubaker, J.; Al-Khairi, I.; Cherian, P.; Noronha, F.; Kavalakatt, S.; Khadir, A.; Behbehani, K.; Alarouj, M.; Bennakhi, A.; et al. Circulating angiopoietin-like protein 8 (betatrophin) association with HsCRP and metabolic syndrome. Cardiovasc. Diabetol. 2016, 15, 25. [Google Scholar] [CrossRef]
- Abu-Farha, M.; Madhu, D.; Hebbar, P.; Mohammad, A.; Channanath, A.; Kavalakatt, S.; Alam-Eldin, N.; Alterki, F.; Taher, I.; Alsmadi, O.; et al. ANGPTL8 R59W variant influences inflammation through modulating NF-κB pathway under TNFα stimulation. bioRxiv 2023. [Google Scholar] [CrossRef]
- Schlosser, P.; Li, Y.; Sekula, P.; Raffler, J.; Grundner-Culemann, F.; Pietzner, M.; Cheng, Y.; Wuttke, M.; Steinbrenner, I.; Schultheiss, U.T.; et al. Genetic studies of urinary metabolites illuminate mechanisms of detoxification and excretion in humans. Nat. Genet. 2020, 52, 167–176. [Google Scholar] [CrossRef]
- Shin, S.Y.; Fauman, E.B.; Petersen, A.-K.; Krumsiek, J.; Santos, R.; Huang, J.; Arnold, M.; Erte, I.; Forgetta, V.; Yang, T.-P.; et al. An atlas of genetic influences on human blood metabolites. Nat. Genet. 2014, 46, 543–550. [Google Scholar] [CrossRef]
- Alterki, A.; Joseph, S.; Thanaraj, T.A.; Al-Khairi, I.; Cherian, P.; Channanath, A.; Sriraman, D.; Ebrahim, M.A.K.; Ibrahim, A.; Tiss, A.; et al. Targeted Metabolomics Analysis on Obstructive Sleep Apnea Patients after Multilevel Sleep Surgery. Metabolites 2020, 10, 358. [Google Scholar] [CrossRef]
- McCoin, C.S.; Knotts, T.A.; Adams, S.H. Acylcarnitines—Old actors auditioning for new roles in metabolic physiology. Nat. Rev. Endocrinol. 2015, 11, 617–625. [Google Scholar] [CrossRef] [PubMed]
- Rutkowsky, J.M.; Knotts, T.A.; Ono-Moore, K.D.; McCoin, C.S.; Huang, S.; Schneider, D.; Singh, S.; Adams, S.H.; Hwang, D.H.; Shigiyama, F.; et al. Acylcarnitines activate proinflammatory signaling pathways. Am. J. Physiol. Metab. 2014, 306, E1378–E1387. [Google Scholar] [CrossRef] [PubMed]
- Treede, I.; Braun, A.; Jeliaskova, P.; Giese, T.; Füllekrug, J.; Griffiths, G.; Stremmel, W.; Ehehalt, R. TNF-alpha-induced up-regulation of pro-inflammatory cytokines is reduced by phosphatidylcholine in intestinal epithelial cells. BMC Gastroenterol. 2009, 9, 53. [Google Scholar] [CrossRef]
- Lagace, T.A. Phosphatidylcholine: Greasing the Cholesterol Transport Machinery. Lipid Insights 2016, 8 (Suppl. S1), 65–73. [Google Scholar] [CrossRef]
- Ludmir, J.; Alvarez, J.G.; Landon, M.B.; Gabbe, S.G.; Mennuti, M.T.; Touchstone, J.C. Amniotic fluid cholesteryl palmitate in pregnancies complicated by diabetes mellitus. Obstet. Gynecol. 1988, 72 3 Pt 1, 360–362. [Google Scholar]
- Mudgil, P.; Dennis, G.R.; Millar, T.J. Interactions of Poly(tert-butyl acrylate)−Poly(styrene) Diblock Copolymers with Lipids at the Air−Water Interface. Langmuir 2006, 22, 7672–7677. [Google Scholar] [CrossRef] [PubMed]
- Joshi-Barve, S.; Barve, S.S.; Amancherla, K.; Gobejishvili, L.; Hill, D.; Cave, M.; Hote, P.; McClain, C.J. Palmitic acid induces production of proinflammatory cytokines interleukin-6, interleukin-1β, and tumor necrosis factor-α via a NF-κB-dependent mechanism in HaCaT keratinocytes. Mediat. Inflamm. 2013, 2013, 530429. [Google Scholar]
- Wang, T.J.; Ngo, D.; Psychogios, N.; Dejam, A.; Larson, M.G.; Vasan, R.S.; Ghorbani, A.; O’sullivan, J.; Cheng, S.; Rhee, E.P.; et al. 2-Aminoadipic acid is a biomarker for diabetes risk. J. Clin. Investig. 2013, 123, 4309–4317. [Google Scholar] [CrossRef]
- Martin, F.P.J.; Montoliu, I.; Collino, S.; Scherer, M.; Guy, P.; Tavazzi, I.; Thorimbert, A.; Moco, S.; Rothney, M.P.; Ergun, D.L.; et al. Topographical body fat distribution links to amino acid and lipid metabolism in healthy obese women. PLoS ONE 2013, 8, e73445. [Google Scholar] [CrossRef]
- Wang, K.-Y.; Tanimoto, A.; Yamada, S.; Guo, X.; Ding, Y.; Watanabe, T.; Watanabe, T.; Kohno, K.; Hirano, K.-I.; Tsukada, H.; et al. Histamine Regulation in Glucose and Lipid Metabolism via Histamine Receptors: Model for Nonalcoholic Steatohepatitis in Mice. Am. J. Pathol. 2010, 177, 713–723. [Google Scholar] [CrossRef]
- Erion, D.M.; Shulman, G.I. Diacylglycerol-mediated insulin resistance. Nat. Med. 2010, 16, 400–402. [Google Scholar] [CrossRef] [PubMed]
- Walters, R.O.; Arias, E.; Diaz, A.; Burgos, E.S.; Guan, F.; Tiano, S.; Mao, K.; Green, C.L.; Qiu, Y.; Shah, H.; et al. Sarcosine Is Uniquely Modulated by Aging and Dietary Restriction in Rodents and Humans. Cell Rep. 2018, 25, 663–676.e6. [Google Scholar] [CrossRef] [PubMed]
- Law, S.-H.; Chan, M.-L.; Marathe, G.K.; Parveen, F.; Chen, C.-H.; Ke, L.-Y. An Updated Review of Lysophosphatidylcholine Metabolism in Human Diseases. Int. J. Mol. Sci. 2019, 20, 1149. [Google Scholar] [CrossRef] [PubMed]
- Luo, M.; Peng, D. ANGPTL8: An Important Regulator in Metabolic Disorders. Front. Endocrinol. 2018, 9, 169. [Google Scholar] [CrossRef]
- Abu-Farha, M.; Abubaker, J.; Tuomilehto, J. ANGPTL8 (betatrophin) role in diabetes and metabolic diseases. Diabetes Metab. Res. Rev. 2017, 33, e2919. [Google Scholar] [CrossRef]
- Abu-Farha, M.; Ghosh, A.; Al-Khairi, I.; Madiraju, S.M.; Abubaker, J.; Prentki, M. The multi-faces of Angptl8 in health and disease: Novel functions beyond lipoprotein lipase modulation. Prog. Lipid Res. 2020, 80, 101067. [Google Scholar] [CrossRef] [PubMed]
- Buniello, A.; MacArthur, J.A.L.; Cerezo, M.; Harris, L.W.; Hayhurst, J.; Malangone, C.; McMahon, A.; Morales, J.; Mountjoy, E.; Sollis, E.; et al. The NHGRI-EBI GWAS Catalog of published genome-wide association studies, targeted arrays and summary statistics 2019. Nucleic Acids Res. 2019, 47, D1005–D1012. [Google Scholar] [CrossRef]
- Alenad, A.; Alenezi, M.M.; Alokail, M.S.; Wani, K.; Mohammed, A.K.; Alnaami, A.M.; Sulimani, M.; Zargar, S.; Clerici, M.; Al-Daghri, N.M. Association of ANGPTL8 (Betatrophin) Gene Variants with Components of Metabolic Syndrome in Arab Adults. Sci. Rep. 2020, 10, 6764. [Google Scholar] [CrossRef]
- Liu, J.; Yagi, K.; Nohara, A.; Chujo, D.; Ohbatake, A.; Fujimoto, A.; Miyamoto, Y.; Kobayashi, J.; Yamagishi, M. High frequency of type 2 diabetes and impaired glucose tolerance in Japanese subjects with the angiopoietin-like protein 8 R59W variant. J. Clin. Lipidol. 2018, 12, 331–337. [Google Scholar] [CrossRef]
- Treede, I.; Braun, A.; Sparla, R.; Kuhnel, M.; Giese, T.; Turner, J.R.; Anes, E.; Kulaksiz, H.; Füllekrug, J.; Stremmel, W.; et al. Anti-inflammatory effects of phosphatidylcholine. J. Biol. Chem. 2007, 282, 27155–27164. [Google Scholar] [CrossRef]
- He, M.; Zhang, W.; Dong, Y.; Wang, L.; Fang, T.; Tang, W.; Lv, B.; Chen, G.; Yang, B.; Huang, P.; et al. Pro-inflammation NF-κB signaling triggers a positive feedback via enhancing cholesterol accumulation in liver cancer cells. J. Exp. Clin. Cancer Res. 2017, 36, 15. [Google Scholar] [CrossRef]
- Branco, A.C.C.C.; Yoshikawa, F.S.Y.; Pietrobon, A.J.; Sato, M.N. Role of Histamine in Modulating the Immune Response and Inflammation. Mediat. Inflamm. 2018, 2018, 9524075. [Google Scholar] [CrossRef] [PubMed]
- Gao, S.; Quick, C.; Guasch-Ferre, M.; Zhuo, Z.; Hutchinson, J.M.; Su, L.; Hu, F.; Lin, X.; Christiani, D. The Association Between Inflammatory and Oxidative Stress Biomarkers and Plasma Metabolites in a Longitudinal Study of Healthy Male Welders. J. Inflamm. Res. 2021, 14, 2825–2839. [Google Scholar] [CrossRef]
- Samuel, V.T.; Petersen, K.F.; Shulman, G.I. Lipid-induced insulin resistance: Unravelling the mechanism. Lancet 2010, 375, 2267–2277. [Google Scholar] [CrossRef] [PubMed]
- Knuplez, E.; Marsche, G. An Updated Review of Pro- and Anti-Inflammatory Properties of Plasma Lysophosphatidylcholines in the Vascular System. Int. J. Mol. Sci. 2020, 21, 4501. [Google Scholar] [CrossRef] [PubMed]
- Feofanova, E.V.; Chen, H.; Dai, Y.; Jia, P.; Grove, M.L.; Morrison, A.C.; Qi, Q.; Daviglus, M.; Cai, J.; North, K.E.; et al. A Genome-wide Association Study Discovers 46 Loci of the Human Metabolome in the Hispanic Community Health Study/Study of Latinos. Am. J. Hum. Genet. 2020, 107, 849–863. [Google Scholar] [CrossRef] [PubMed]
- Vessichelli, M.; Mariggiò, S.; Varone, A.; Zizza, P.; Di Santo, A.; Amore, C.; Dell’Elba, G.; Cutignano, A.; Fontana, A.; Cacciapuoti, C.; et al. The natural phosphoinositide derivative glycerophosphoinositol inhibits the lipopolysaccharide-induced inflammatory and thrombotic responses. J. Biol. Chem. 2017, 292, 12828–12841. [Google Scholar] [CrossRef] [PubMed]
- Baker, R.G.; Hayden, M.S.; Ghosh, S. NF-κB, inflammation, and metabolic disease. Cell Metab. 2011, 13, 11–22. [Google Scholar] [CrossRef]
- Desine, S.; Gabriel, C.L.; Smith, H.M.; Antonetti, O.R.; Wang, C.; Calcutt, M.W.; Doran, A.C.; Silver, H.J.; Nair, S.; Terry, J.G.; et al. Association of alpha-aminoadipic acid (2-AAA) with cardiometabolic risk factors in healthy and high-risk individuals. medRxiv 2023. [Google Scholar] [CrossRef]
- Ohigashi, T.; Kanno, K.; Sugiyama, A.; Nguyen, P.T.; Kishikawa, K.N.; Otani, Y.; Kobayashi, T.; Matsuo, H.; Tazuma, S. Protective effect of phosphatidylcholine on lysophosphatidylcholine-induced cellular senescence in cholangiocyte. J. Hepatobiliary Pancreat. Sci. 2019, 26, 568–577. [Google Scholar] [CrossRef]
- Dambrova, M.; Makrecka-Kuka, M.; Kuka, J.; Vilskersts, R.; Nordberg, D.; Attwood, M.M.; Smesny, S.; Sen, Z.D.; Guo, A.C.; Oler, E.; et al. Acylcarnitines: Nomenclature, Biomarkers, Therapeutic Potential, Drug Targets, and Clinical Trials. Pharmacol. Rev. 2022, 74, 506–551. [Google Scholar] [CrossRef] [PubMed]
- Rafieian-Kopaei, M.; Setorki, M.; Doudi, M.; Baradaran, A.; Nasri, H. Atherosclerosis: Process, Indicators, Risk Factors and New Hopes. Int. J. Prev. Med. 2014, 5, 927–946. [Google Scholar] [PubMed]

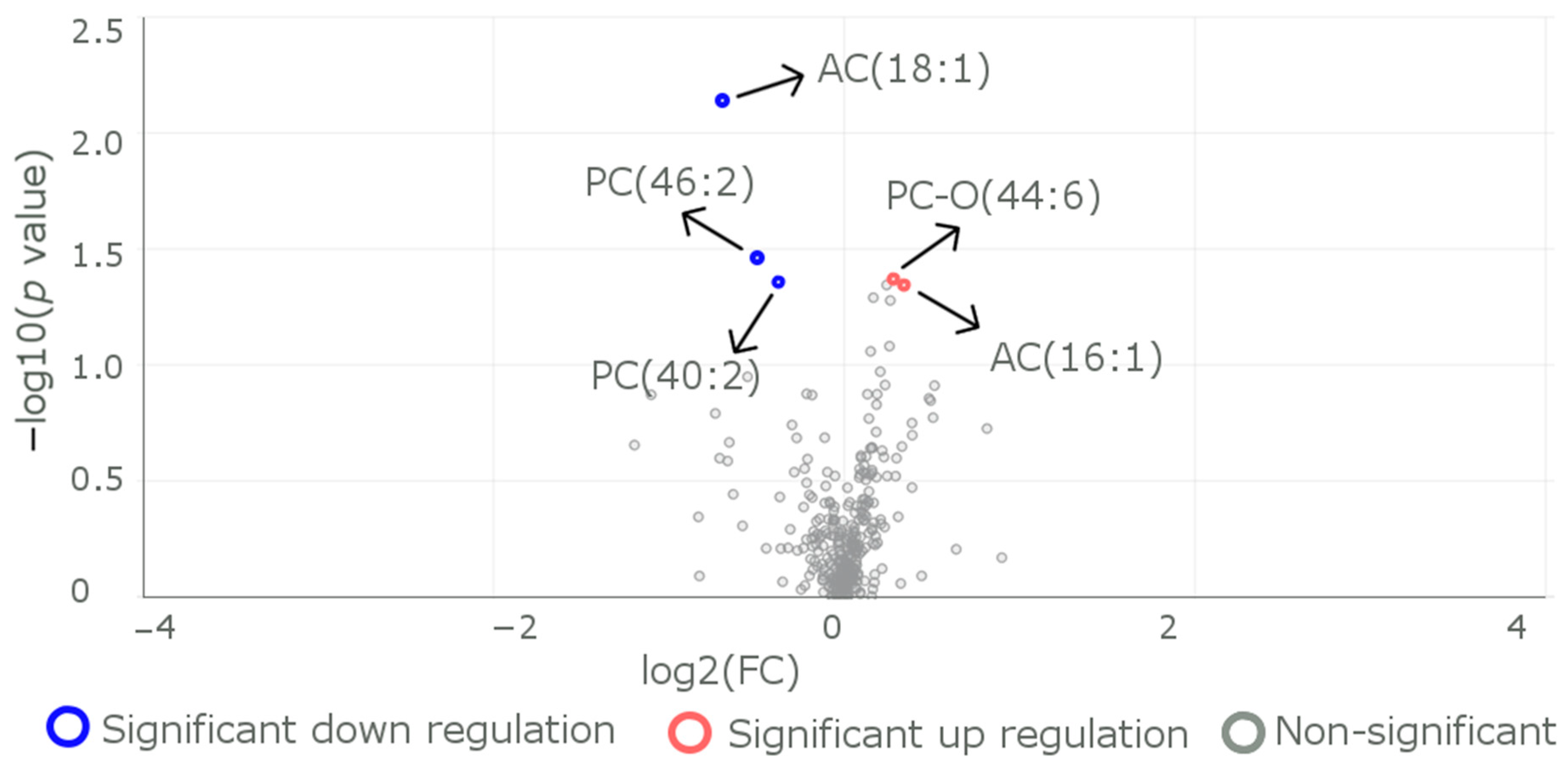
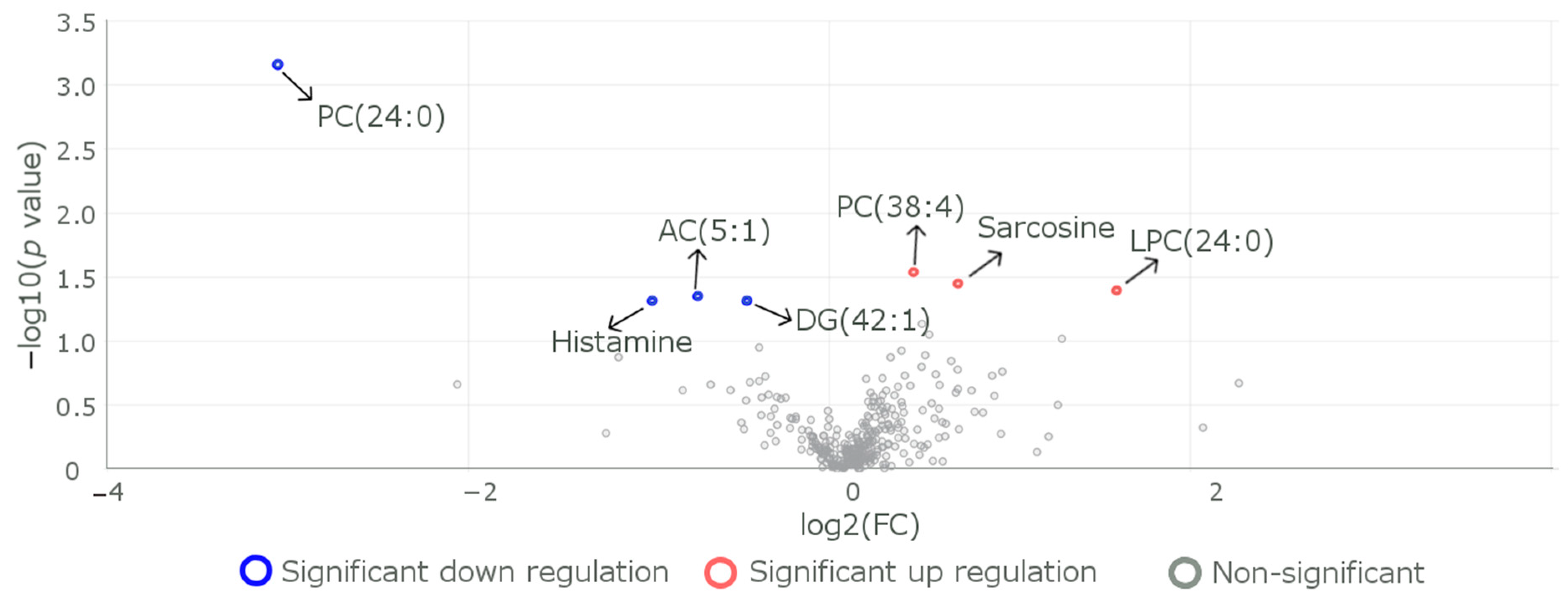
| Characteristics | All the Participants, n = 60 1 | Participants with the Variant R59W, n = 29 1 | Participants with the Reference Genotype, n = 31 1 | p Value 2 |
|---|---|---|---|---|
| Genotype at the variant | <0.001 | |||
| CC | 31 (52%) | 0 (0%) | 31 (100%) | |
| CT | 26 (43%) | 26 (90%) | 0 (0%) | |
| TT | 3 (5.0%) | 3 (10%) | 0 (0%) | |
| Age: years | 42 (37, 52) | 42 (38, 53) | 43 (37, 51) | 0.8 |
| Sex: male | 60 (100%) | 29 (100%) | 31 (100%) | |
| Height: cm | 170 (166, 175) | 171 (169, 176) | 169 (164, 175) | 0.2 |
| Weight: Kg | 86 (78, 96) | 88 (80, 99) | 82 (77, 95) | 0.14 |
| BMI: Kg/m2 | 29.0 (26.6, 32.2) | 30.8 (26.5, 32.5) | 28.0 (26.7, 31.3) | 0.3 |
| WC (cm) | 94 (90, 106) | 94 (92, 103) | 98 (90, 107) | 0.8 |
| Hip (cm) | 103 (101, 110) | 103 (102, 110) | 104 (101, 109) | >0.9 |
| SBP | 38 (0, 136) | 103 (0, 135) | 0 (0, 136) | 0.5 |
| DBP | 26 (0, 87) | 59 (0, 88) | 0 (0, 82) | 0.6 |
| FBG: mg/dL | 90.81 (82.88, 98.90) | 89.19 (82.88, 99.64) | 92.61 (83.24, 97.30) | 0.9 |
| FBG: mmol/L | 5.04 (4.60, 5.47) | 4.95 (4.60, 5.53) | 5.14 (4.62, 5.40) | 0.9 |
| HbA1C: % | 5.50 (5.30, 5.70) | 5.56 (5.38, 5.62) | 5.40 (5.00, 5.77) | 0.6 |
| TC: mmol/L | 5.47 (4.65, 5.70) | 5.47 (4.75, 5.70) | 5.45 (4.21, 5.83) | 0.9 |
| TG: mmol/L | 1.21 (0.97, 2.33) | 1.61 (0.97, 2.50) | 1.13 (0.99, 2.04) | 0.6 |
| HDL: mmol/L | 0.99 (0.83, 1.19) | 1.04 (0.84, 1.30) | 0.97 (0.84, 1.05) | 0.6 |
| LDL: mmol/L | 3.62 (2.78, 3.98) | 3.55 (2.92, 3.85) | 3.66 (2.73, 3.98) | 0.6 |
| Non-diabetic | 46 (77%) | 22 (76%) | 24 (77%) | |
| Pre-diabetes | 14 (23%) | 7 (24%) | 7 (23%) |
| Metabolite | Reference (n = 31) | R59W (n = 29) | p Value |
|---|---|---|---|
| AC.14.1.DC. | |||
| Mean (SD) | 0.0401 (0.0129) | 0.0338 (0.0114) | 0.049 |
| Median (Min, Max) | 0.0410 (0.0130, 0.0660) | 0.0330 (0.0130, 0.0590) | |
| AC.16.1. | |||
| Mean (SD) | 0.0102 (0.00380) | 0.00807 (0.00393) | 0.015 |
| Median (Min, Max) | 0.00900 (0.00400, 0.0230) | 0.00600 (0.00300, 0.0190) | |
| AC.16.2.OH. | |||
| Mean (SD) | 0.00661 (0.00230) | 0.00569 (0.00154) | 0.014 |
| Median (Min, Max) | 0.00600 (0.00500, 0.0140) | 0.00500 (0.00500, 0.0130) | |
| AC.18.1. | |||
| Mean (SD) | 0.0182 (0.0208) | 0.0306 (0.0279) | <0.001 |
| Median (Min, Max) | 0.0110 (0.00800, 0.0830) | 0.0150 (0.0100, 0.0860) | |
| alpha.AAA | |||
| Mean (SD) | 2.12 (0.515) | 2.62 (0.714) | 0.007 |
| Median (Min, Max) | 2.14 (0, 2.97) | 2.45 (1.69, 4.57) | |
| PC.46.2. | |||
| Mean (SD) | 0.748 (0.587) | 1.13 (0.954) | 0.034 |
| Median (Min, Max) | 0.725 (0.00400, 2.48) | 1.06 (0.108, 4.96) | |
| Missing | 2 (6.5%) | 6 (20.7%) | |
| PC.O.40.4. | |||
| Mean (SD) | 1.01 (0.395) | 0.828 (0.306) | 0.021 |
| Median (Min, Max) | 0.902 (0.372, 2.57) | 0.764 (0.340, 1.72) | |
| Missing | 0 (0%) | 1 (3.4%) | |
| PC.O.42.4. | |||
| Mean (SD) | 0.436 (0.149) | 0.369 (0.142) | 0.032 |
| Median (Min, Max) | 0.389 (0.253, 0.933) | 0.322 (0.172, 0.727) | |
| Missing | 0 (0%) | 1 (3.4%) | |
| PC.O.42.6. | |||
| Mean (SD) | 0.869 (0.243) | 0.731 (0.198) | 0.018 |
| Median (Min, Max) | 0.807 (0.529, 1.60) | 0.711 (0.473, 1.28) | |
| Missing | 1 (3.2%) | 2 (6.9%) | |
| PC.O.44.6. | |||
| Mean (SD) | 1.14 (0.354) | 0.924 (0.237) | 0.024 |
| Median (Min, Max) | 1.06 (0.703, 2.11) | 0.891 (0.504, 1.47) | |
| Missing | 4 (12.9%) | 3 (10.3%) | |
| CE.20.4. | |||
| Mean (SD) | 2430 (785) | 2070 (678) | 0.015 |
| Median (Min, Max) | 2320 (719, 4920) | 1900 (1270, 4330) | |
| Missing | 0 (0%) | 1 (3.4%) |
| Metabolite | Change in Metabolite Level in the R59W Group | Disorder for which Risk Increases Due to the Observed Change in the Metabolite Level. | Reference |
|---|---|---|---|
| α-AAA | Increased | Alpha-aminoadipic acid (2-AAA) is associated with the development of type 2 diabetes (T2D) and atherosclerosis, as well as cardiometabolic disorders. | [38,59] |
| Phosphatidylcholine | Decreased | Masks the protective role against lysophosphatidylcholine-induced cytotoxicity and cellular senescence. | [60] |
| Acylcarnitine AC 18.1 | Increased | Important indicators of metabolic disorders, including diabetes, cardiovascular disorders, and certain types of cancers. | [61] |
| Sarcosine | Decreased | It is a potential functional biomarker of the aging and diet restriction metabolic phenotype. | [42] |
| Cholesteryl Ester | Decreased | Accumulation of cholesterol esters in the arterial intima is a characteristic feature of atherosclerosis. This may have a protective effect due to the variant. | [62] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Abu-Farha, M.; Joseph, S.; Mohammad, A.; Channanath, A.; Taher, I.; Al-Mulla, F.; Mujammami, M.; Thanaraj, T.A.; Abubaker, J.; Abdel Rahman, A.M. Targeted Metabolomics Analysis of Individuals Carrying the ANGPTL8 R59W Variant. Metabolites 2023, 13, 972. https://doi.org/10.3390/metabo13090972
Abu-Farha M, Joseph S, Mohammad A, Channanath A, Taher I, Al-Mulla F, Mujammami M, Thanaraj TA, Abubaker J, Abdel Rahman AM. Targeted Metabolomics Analysis of Individuals Carrying the ANGPTL8 R59W Variant. Metabolites. 2023; 13(9):972. https://doi.org/10.3390/metabo13090972
Chicago/Turabian StyleAbu-Farha, Mohamed, Shibu Joseph, Anwar Mohammad, Arshad Channanath, Ibrahim Taher, Fahd Al-Mulla, Muhammad Mujammami, Thangavel Alphonse Thanaraj, Jehad Abubaker, and Anas M. Abdel Rahman. 2023. "Targeted Metabolomics Analysis of Individuals Carrying the ANGPTL8 R59W Variant" Metabolites 13, no. 9: 972. https://doi.org/10.3390/metabo13090972
APA StyleAbu-Farha, M., Joseph, S., Mohammad, A., Channanath, A., Taher, I., Al-Mulla, F., Mujammami, M., Thanaraj, T. A., Abubaker, J., & Abdel Rahman, A. M. (2023). Targeted Metabolomics Analysis of Individuals Carrying the ANGPTL8 R59W Variant. Metabolites, 13(9), 972. https://doi.org/10.3390/metabo13090972









