Sensing of Proteins by ICD Response of Iron(II) Clathrochelates Functionalized by Carboxyalkylsulfide Groups
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Circular Dichroism Studies
2.3. ITC Experiments
2.4. Protein Fluorescence Quenching Studies
2.5. Electrospray Ionization Mass Spectrometry
2.6. Computer Modelling
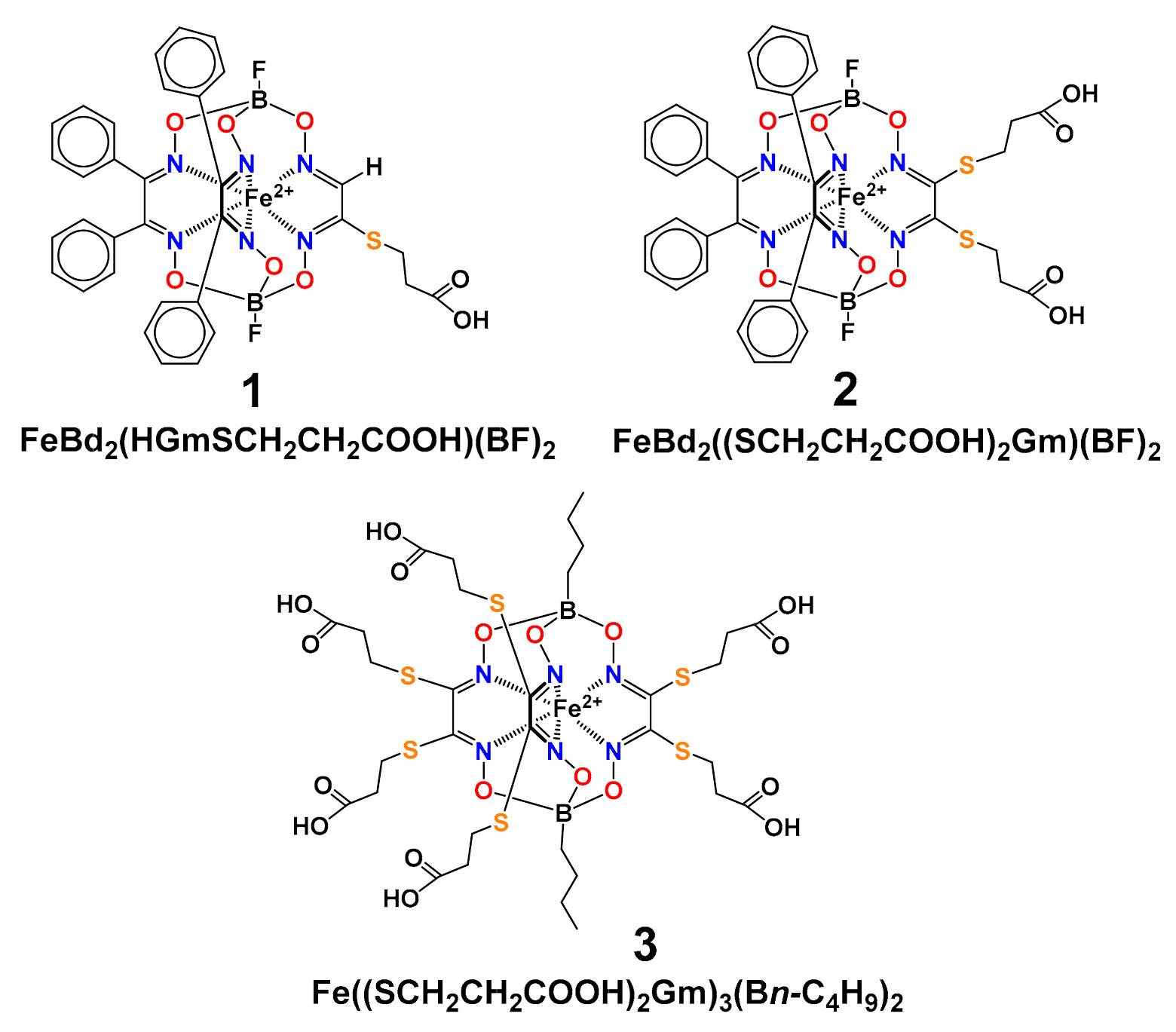
3. Results and Discussion
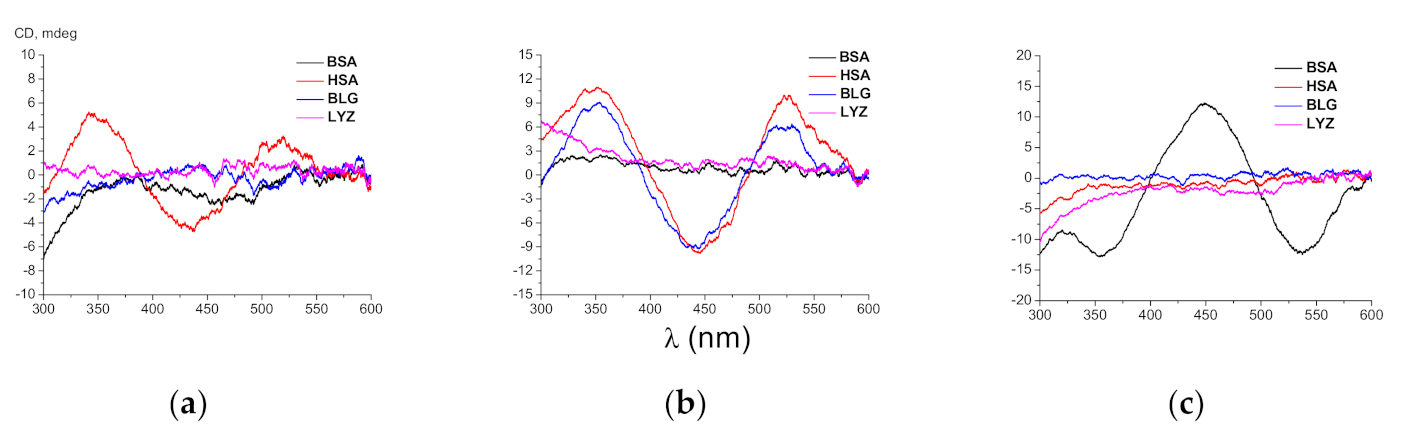
3.1. ICD Spectra
3.2. Native ESI-MS
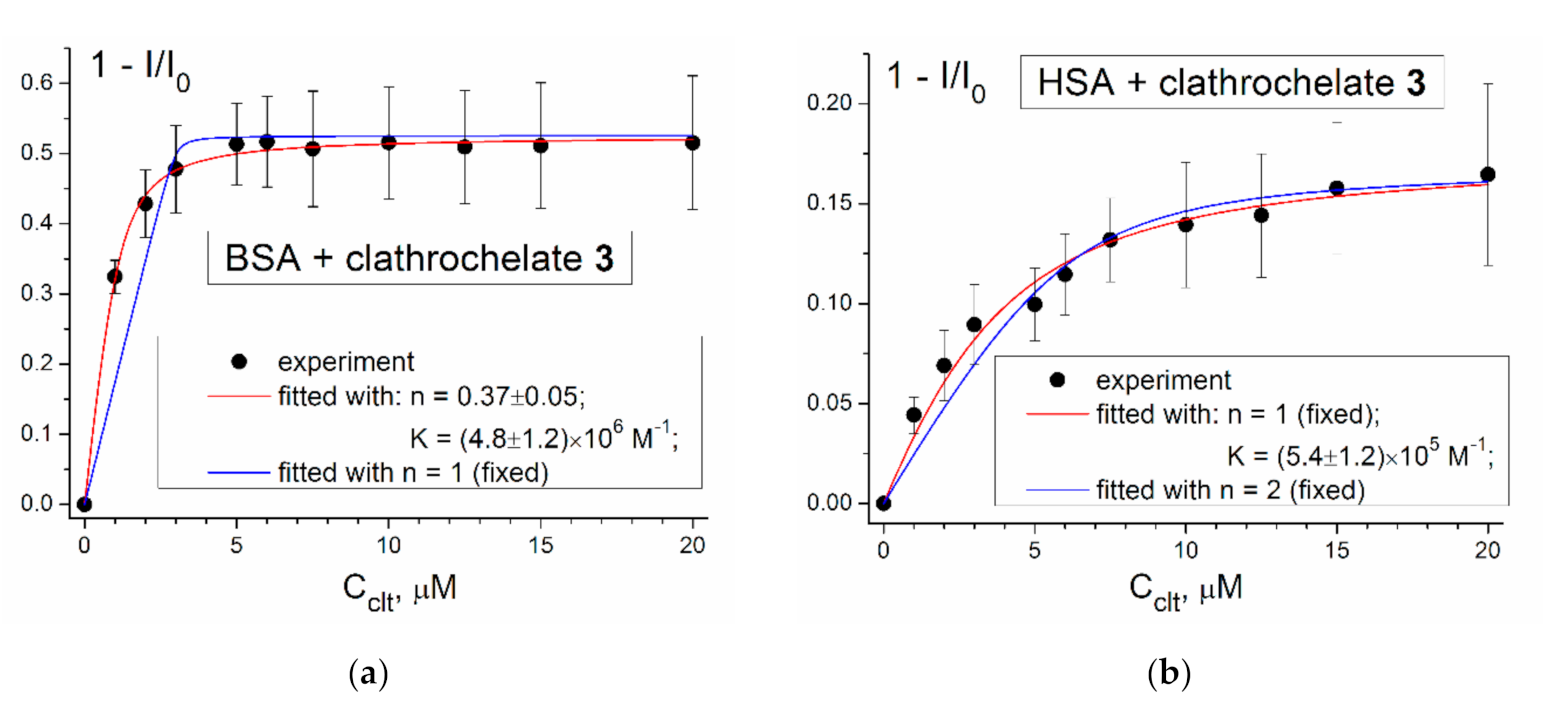
3.3. Protein Fluorescence Quenching Data
3.4. Thermodynamic Characteristics of the Supramolecular Clathrochelate 3–BSA Assembly
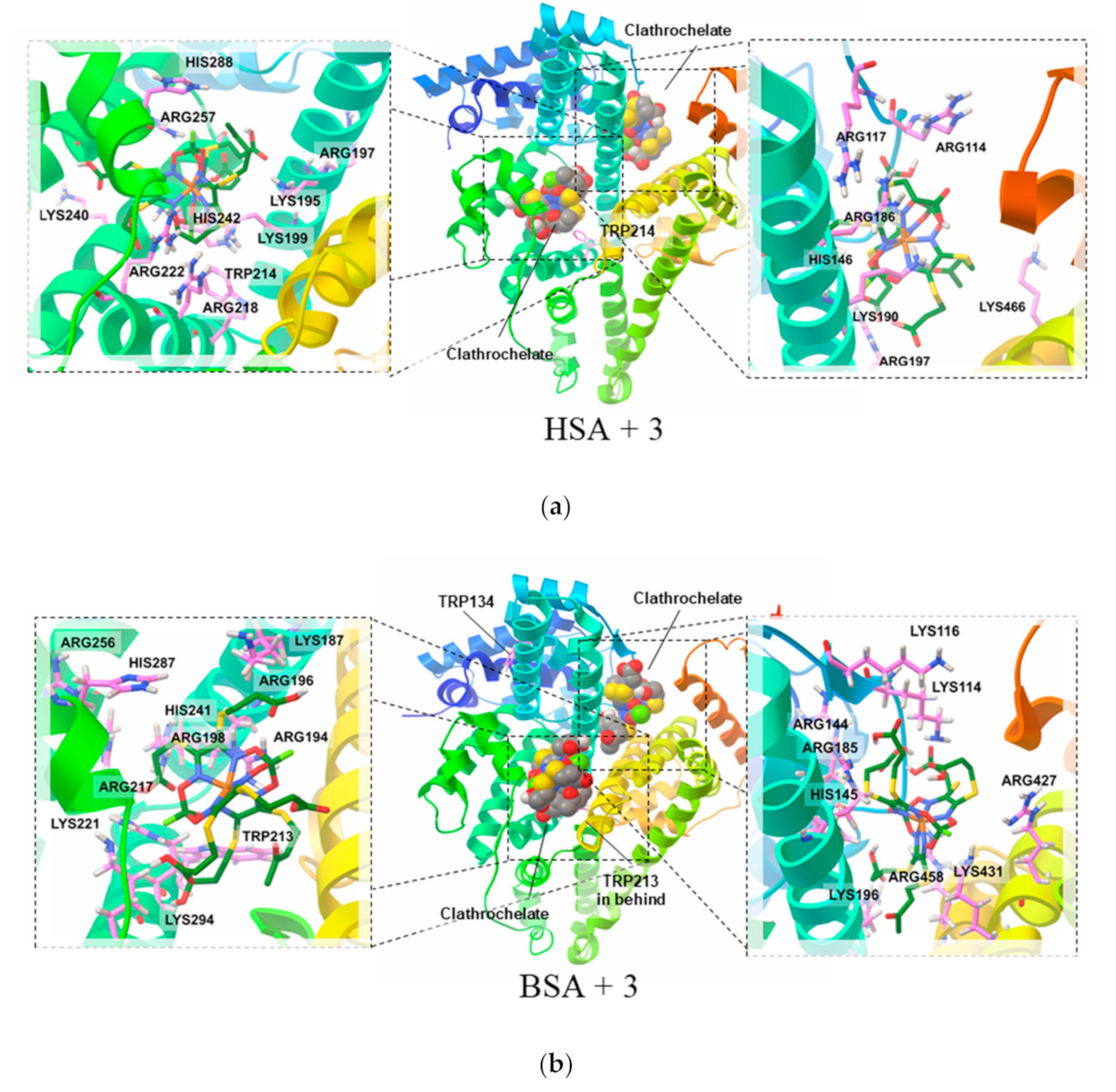
3.5. Molecular Modelling Calculations of the Clathrochelate–Albumin Assemblies
4. Conclusions
Supplementary Materials
Author Contributions
Funding

Conflicts of Interest
References
- Alberts, B.; Johnson, A.; Lewis, J.; Raff, M.; Roberts, K.; Walter, P. Molecular Biology of the Cell, 4th ed.; Garland Science: New York, NY, USA, 2002. [Google Scholar]
- Berg, J.M.; Tymoczko, J.L.; Stryer, L. Biochemistry, 5th ed.; W. H. Freeman: New York, NY, USA, 2002. [Google Scholar]
- Godbey, W.T. An Introduction to Biotechnology: The Science, Technology and Medical Applications, 1st ed.; Academic Press: Cambridge, MA, USA, 2015. [Google Scholar] [CrossRef]
- Engelking, L.R. Textbook of Veterinary Physiological Chemistry, 3rd ed.; Academic Press: Cambridge, MA, USA, 2015. [Google Scholar] [CrossRef]
- Khazanov, N.A.; Carlson, H.A. Exploring the Composition of Protein-Ligand Binding Sites on a Large Scale. PLoS Comput. Biol. 2013, 9, e1003321. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bartlett, G.J.; Porter, C.T.; Borkakoti, N.; Thornton, J.M. Analysis of Catalytic Residues in Enzyme Active Sites. J. Mol. Biol. 2002, 324, 105–121. [Google Scholar] [CrossRef]
- Holliday, G.L.; Mitchell, J.B.O.; Thornton, J.M. Understanding the Functional Roles of Amino Acid Residues in Enzyme Catalysis. J. Mol. Biol. 2009, 390, 560–577. [Google Scholar] [CrossRef] [PubMed]
- Kupfer, L.; Hinrichs, W.; Groschup, M.H. Prion protein misfolding. Curr. Mol. Med. 2009, 9, 826–835. [Google Scholar] [CrossRef] [Green Version]
- Scheckel, C.; Aguzzi, A. Prions, prionoids and protein misfolding disorders. Nat. Rev. Genet. 2018, 19, 405–418. [Google Scholar] [CrossRef] [Green Version]
- Cohen, F.E. Protein misfolding and prion diseases. J. Mol. Biol. 1999, 293, 313–320. [Google Scholar] [CrossRef]
- Poggiolini, I.; Saverioni, D.; Parchi, P. Prion Protein Misfolding, Strains, and Neurotoxicity: An Update from Studies on Mammalian Prions. Int. J. Cell Biol. 2013, 2013, 24. [Google Scholar] [CrossRef] [Green Version]
- Uversky, V.N.; Fink, A.L. Conformational constraints for amyloid fibrillation: The importance of being unfolded. Biochim. Biophys. Acta 2004, 1698, 131–153. [Google Scholar] [CrossRef]
- Fink, A.L. The Aggregation and Fibrillation of α-Synuclein. Acc. Chem. Res. 2006, 39, 628–634. [Google Scholar] [CrossRef]
- Knowles, T.; Vendruscolo, M.; Dobson, C. The amyloid state and its association with protein misfolding diseases. Nat. Rev. Mol. Cell Biol. 2014, 15, 384–396. [Google Scholar] [CrossRef]
- Sweeney, P.; Park, H.; Baumann, M.; Dunlop, J.; Frydman, J.; Kopito, R.; McCampbell, A.; Leblanc, G.; Venkateswaran, A.; Nurmi, A.; et al. Protein misfolding in neurodegenerative diseases: Implications and strategies. Transl. Neurodegener. 2017, 6, 6. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chiti, F.; Dobson, C.M. Protein Misfolding, Amyloid Formation, and Human Disease: A Summary of Progress Over the Last Decade. Annu. Rev. Biochem. 2017, 86, 27–68. [Google Scholar] [CrossRef] [PubMed]
- Fredrickson, E.K.; Rosenbaum, J.C.; Locke, M.N.; Milac, T.I.; Gardner, R.G. Exposed hydrophobicity is a key determinant of nuclear quality control degradation. Mol. Biol. Cell 2011, 22, 2384–2395. [Google Scholar] [CrossRef] [PubMed]
- Buchberger, A.; Bukau, B.; Sommer, T. Protein Quality Control in the Cytosol and the Endoplasmic Reticulum: Brothers in Arms. Mol. Cell 2010, 40, 238–252. [Google Scholar] [CrossRef] [PubMed]
- Stefani, M. Protein misfolding and aggregation: New examples in medicine and biology of the dark side of the protein world. Biochim. Biophys. Acta (BBA) Mol. Basis Dis. 2004, 1739, 5–25. [Google Scholar] [CrossRef] [Green Version]
- Nadeau, O.W.; Carlson, G.M. A Review of Methods Used for Identifying Structural Changes in a Large Protein Complex. In Allostery. Methods in Molecular Biology (Methods and Protocols); Fenton, A., Ed.; Springer: New York, NY, USA, 2012; Volume 796, pp. 117–132. [Google Scholar] [CrossRef] [Green Version]
- Lundblad, R.L. Approaches to the Conformational Analysis of Biopharmaceuticals, 1st ed.; CRC Press: Boca Raton, FL, USA, 2009; pp. 1–4. [Google Scholar]
- Santofimia-Castaño, P.; Rizzuti, B.; Xia, Y.; Abian, O.; Peng, L.; Velázquez-Campoy, A.; Neira, J.L.; Iovanna, J. Targeting intrinsically disordered proteins involved in cancer. Cell. Mol. Life Sci. 2020, 77, 1695–1707. [Google Scholar] [CrossRef] [Green Version]
- Voloshin, Y.; Belaya, I.; Krämer, R. Cage Metal Complexes: Clathrochelates Revisited, 1st ed.; Springer International Publishing AG: Berlin/Heidelberg, Germany, 2017. [Google Scholar] [CrossRef]
- Jansze, S.M.; Severin, K. Clathrochelate Metalloligands in Supramolecular Chemistry and Materials Science. Acc. Chem. Res. 2018, 51, 2139–2147. [Google Scholar] [CrossRef]
- Kovalska, V.B.; Vakarov, S.V.; Kuperman, M.V.; Losytskyy, M.Y.; Gumienna-Kontecka, E.; Voloshin, Y.Z.; Varzatskii, O.A. Induced chirality of cage metal complexes switched by their supramolecular and covalent binding. Dalton Trans. 2018, 47, 1036–1052. [Google Scholar] [CrossRef] [Green Version]
- Kovalska, V.; Kuperman, M.; Losytskyy, M.; Vakarov, S.; Potocki, S.; Yarmoluk, S.; Voloshin, Y.; Varzatskii, O.; Gumienna-Kontecka, E. Induced CD of iron(II) clathrochelates: Sensing of the structural and conformational alterations of serum albumins. Metallomics 2019, 11, 338–348. [Google Scholar] [CrossRef] [Green Version]
- Kovalska, V.; Vakarov, S.; Losytskyy, M.; Kuperman, M.; Chornenka, N.; Toporivska, Y.; Gumienna-Kontecka, E.; Voloshin, Y.; Varzatskii, O.; Mokhir, A. Dicarboxyl-terminated iron(II) clathrochelates as ICD-reporters for globular proteins. RSC Adv. 2019, 9, 24218–24230. [Google Scholar] [CrossRef] [Green Version]
- Voloshin, Y.Z.; Varzatskii, O.A.; Palchik, A.V.; Starikova, Z.A.; Antipin, M.Y.; Lebed, E.G.; Bubnov, Y.N. Mono- and trichloride clathrochelate iron (II) chloroglyoximates and their functionalization: The effect of the substituents in the clathrochelate framework on the reactivity of the chlorine-containing fragments in nucleophilic substitution reactions. Inorg. Chim. Acta 2006, 359, 553–569. [Google Scholar] [CrossRef]
- Voloshin, Y.Z.; Varzatskii, O.A.; Kron, T.E.; Belsky, V.K.; Zavodnik, V.E.; Palchik, A.V. Triribbed-Functionalized Clathrochelate Iron(II) Dioximates as a New and Promising Tool to Obtain Polynucleating and Polynuclear Compounds with Improved Properties. Inorg. Chem. 2000, 39, 1907–1918. [Google Scholar] [CrossRef] [PubMed]
- MOPAC®. Available online: http://openmopac.net/ (accessed on 30 September 2020).
- Stewart, J.J.P. Optimization of parameters for semiempirical methods VI: More modifications to the NDDO approximations and re-optimization of parameters. J. Mol. Model. 2013, 19, 1–32. [Google Scholar] [CrossRef] [Green Version]
- Neese, F. The ORCA program system. Wiley Interdiscip. Rev. Comput. Mol. Sci. 2012, 2, 73–78. [Google Scholar] [CrossRef]
- Neese, F. Software update: The ORCA program system, version 4.0. Wiley Interdiscip. Rev. Comput. Mol. Sci. 2018, 8, e1327. [Google Scholar] [CrossRef]
- Ekström, U.; Visscher, L.; Bast, R.; Thorvaldsen, A.J.; Ruud, K. Arbitrary-Order Density Functional Response Theory from Automatic Differentiation. J. Chem. Theory Comput. 2010, 6, 1971–1980. [Google Scholar] [CrossRef]
- Weigend, F.; Ahlrichs, R. Balanced basis sets of split valence, triple zeta valence and quadruple zeta valence quality for H to Rn: Design and assessment of accuracy. Phys. Chem. Chem. Phys. 2005, 7, 3297–3305. [Google Scholar] [CrossRef]
- Weigend, F. Accurate Coulomb-fitting basis sets for H to Rn. Phys. Chem. Chem. Phys. 2006, 8, 1057–1065. [Google Scholar] [CrossRef]
- Grimme, S.; Antony, J.; Ehrlich, S.; Krieg, H. A consistent and accurate ab initio parametrization of density functional dispersion correction (DFT-D) for the 94 elements H-Pu. J. Chem. Phys. 2010, 132, 154104. [Google Scholar] [CrossRef] [Green Version]
- Feinstein, W.P.; Brylinski, M. Calculating an optimal box size for ligand docking and virtual screening against experimental and predicted binding pockets. J. Cheminform. 2015, 7, 18. [Google Scholar] [CrossRef] [Green Version]
- Wang, Z.M.; Ho, J.X.; Ruble, J.R.; Rose, J.; Ruker, F.; Ellenburg, M.; Murphy, R.; Click, J.; Soistman, E.; Wilkerson, L.; et al. Structural studies of several clinically important oncology drugs in complex with human serum albumin. Biochim. Biophys. Acta 2013, 1830, 5356–5374. [Google Scholar] [CrossRef] [PubMed]
- Sekula, B.; Zielinski, K.; Bujacz, A. Crystallographic studies of the complexes of bovine and equine serum albumin with 3,5-diiodosalicylic acid. Int. J. Biol. Macromol. 2013, 60, 316–324. [Google Scholar] [CrossRef] [PubMed]
- Trott, O.; Olson, A.J. AutoDock Vina: Improving the speed and accuracy of docking with a new scoring function, efficient optimization, and multithreading. J. Comput. Chem. 2010, 31, 455–461. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Morris, G.M.; Huey, R.; Lindstrom, W.; Sanner, M.F.; Belew, R.K.; Goodsell, D.S.; Olson, A.J. AutoDock4 and AutoDockTools4: Automated docking with selective receptor flexibility. J. Comput. Chem. 2009, 30, 2785–2791. [Google Scholar] [CrossRef] [Green Version]
- Hartinger, C.G. Application of mass spectrometric techniques to delineate the modes-of-action of anticancer metallodrugs. Chem. Soc. Rev. 2013, 42, 6186–6199. [Google Scholar] [CrossRef]
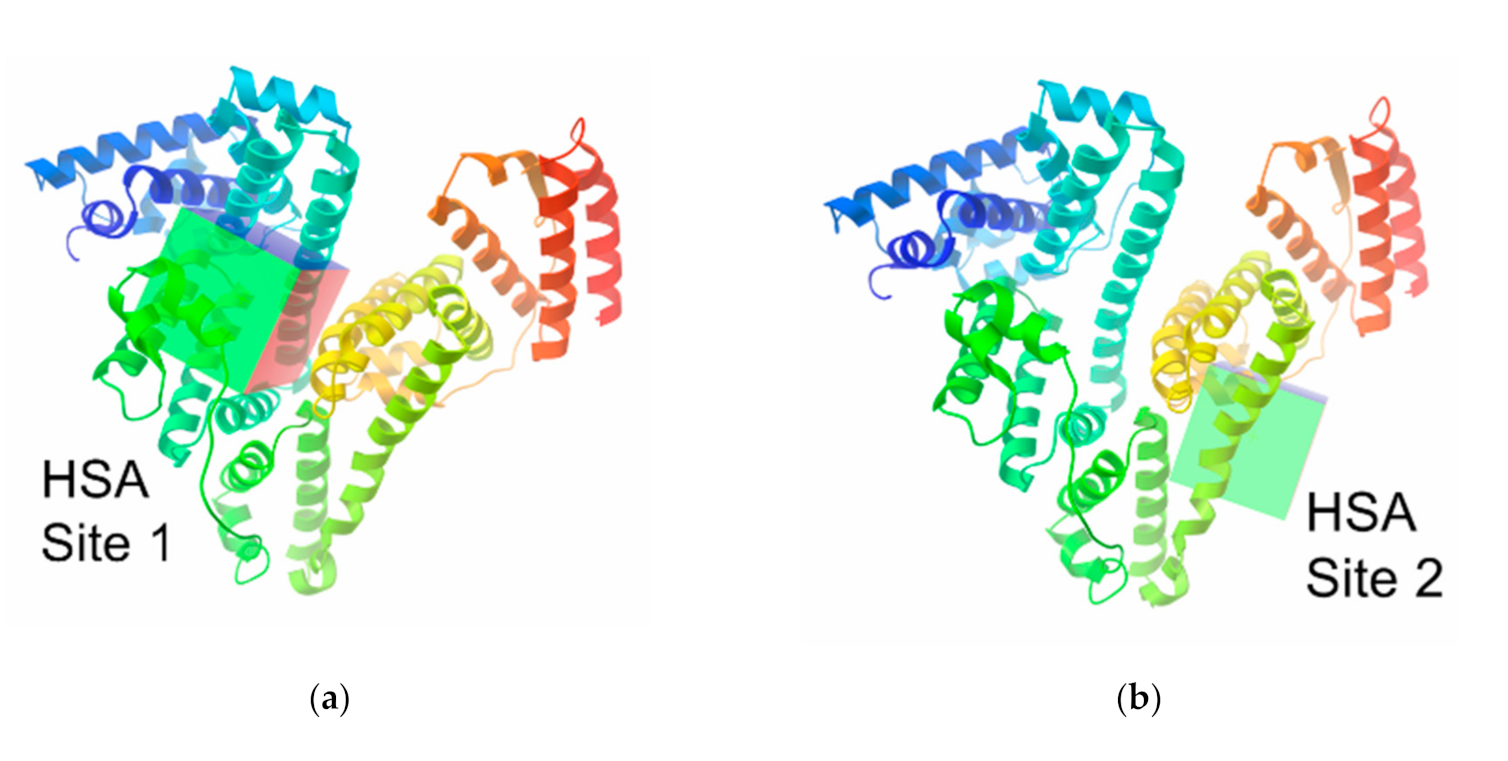
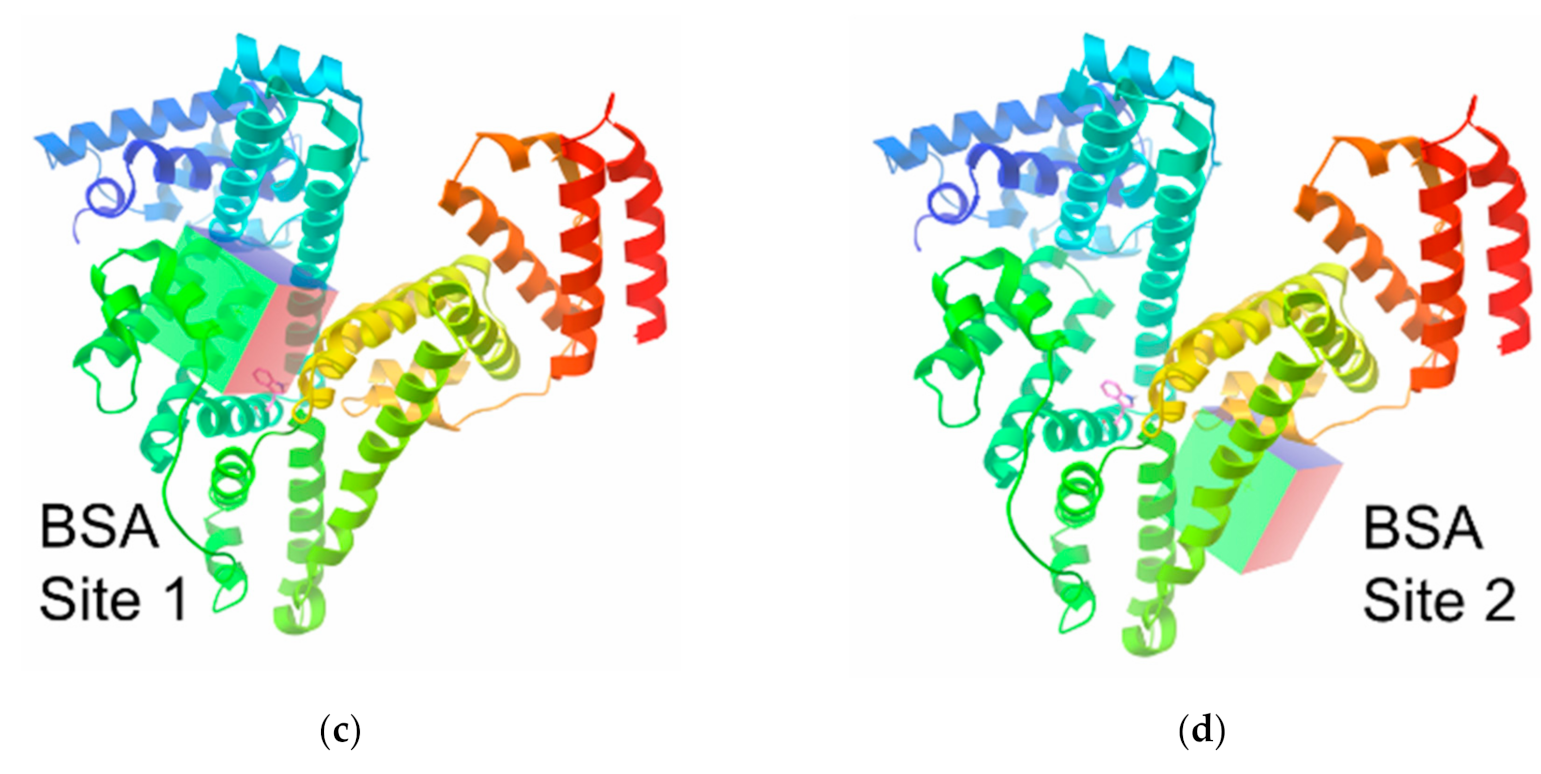


| HSA (RCSB ID: 4L8U) | BSA ((RCSB ID: 4JK4) | |||||
|---|---|---|---|---|---|---|
| x | y | z | x | y | z | |
| Site I | 28.8 | 15.2 | 17.6 | 95.9 | 18.5 | 20.7 |
| Site II | 3.7 | −3.8 | 6.9 | 112 | 40 | 37 |
| Complex | +BSA | +HSA | +LYZ | +BLG |
|---|---|---|---|---|
| 1 | 3.0 | 9.8 | 1.0 | 1.5 |
| 2 | 2.5 | 20.8 | 1.1 | 18.5 |
| 3 | 24.9 | 0.9 | 0.8 | 0.5 |
| Species | Deconvoluted Mass [Da] | Mass Difference to Reference [Da] | Theoretical Mass of Compound [Da] |
|---|---|---|---|
| Delipidated HSA | 66,802 ± 21 | ||
| +1 | 67,721 ± 34 | 919 | 782 |
| +2× 1 | 68,328 ± 32 | 2 × 763 | 2 × 782 |
| +2 | 67,800 ± 29 | 998 | 886 |
| +2× 2 | 68,573 ± 36 | 2 × 885 | 2 × 886 |
| +3 | 68,052 ± 58 | 1250 | 1075 |
| +2× 3 | 69,085 ± 64 | 2 × 1142 | 2 × 1075 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Losytskyy, M.; Chornenka, N.; Vakarov, S.; Meier-Menches, S.M.; Gerner, C.; Potocki, S.; Arion, V.B.; Gumienna-Kontecka, E.; Voloshin, Y.; Kovalska, V. Sensing of Proteins by ICD Response of Iron(II) Clathrochelates Functionalized by Carboxyalkylsulfide Groups. Biomolecules 2020, 10, 1602. https://doi.org/10.3390/biom10121602
Losytskyy M, Chornenka N, Vakarov S, Meier-Menches SM, Gerner C, Potocki S, Arion VB, Gumienna-Kontecka E, Voloshin Y, Kovalska V. Sensing of Proteins by ICD Response of Iron(II) Clathrochelates Functionalized by Carboxyalkylsulfide Groups. Biomolecules. 2020; 10(12):1602. https://doi.org/10.3390/biom10121602
Chicago/Turabian StyleLosytskyy, Mykhaylo, Nina Chornenka, Serhii Vakarov, Samuel M. Meier-Menches, Christopher Gerner, Slawomir Potocki, Vladimir B. Arion, Elzbieta Gumienna-Kontecka, Yan Voloshin, and Vladyslava Kovalska. 2020. "Sensing of Proteins by ICD Response of Iron(II) Clathrochelates Functionalized by Carboxyalkylsulfide Groups" Biomolecules 10, no. 12: 1602. https://doi.org/10.3390/biom10121602





