Thymus algeriensis and Thymus fontanesii: Chemical Composition, In Vivo Antiinflammatory, Pain Killing and Antipyretic Activities: A Comprehensive Comparison
Abstract
:1. Introduction
2. Materials and Methods
2.1. Plant Material and Extraction
2.2. HPLC-PDA-ESI-MS/MS
2.3. In Vitro Studies
2.3.1. Cyclooxygenase (COX) Inhibition Assay
2.3.2. Lipoxygenase (LOX) Inhibition Assay
2.3.3. Total Antioxidant Capacity (TAC) Assay
2.4. Animals
2.4.1. The Anti-Inflammatory Activity in Carrageenan-Induced Hind-Paw Edema Model
2.4.2. Leukocytes Recruitment into Peritoneal Cavity in Mice
2.4.3. Acetic Acid-Induced Vascular Permeability
2.4.4. Assay of Anti-Nociceptive Activity by Hot Plate Test
2.4.5. Assay of Anti-Nociceptive Activity by Acetic Acid-Induced Abdominal Writhing
2.4.6. Anti-Pyretic Activity in Brewer’s Yeast Induced Pyrexia in Mice
2.5. Molecular Modeling
2.6. Statistical Analysis
3. Results and Discussion
3.1. In Vitro Activities
3.1.1. Inhibition of Cyclooxygenase (COX-1/2), Lipoxygenase (5-LOX), and Total Antioxidant Capacity (TAC) Assay
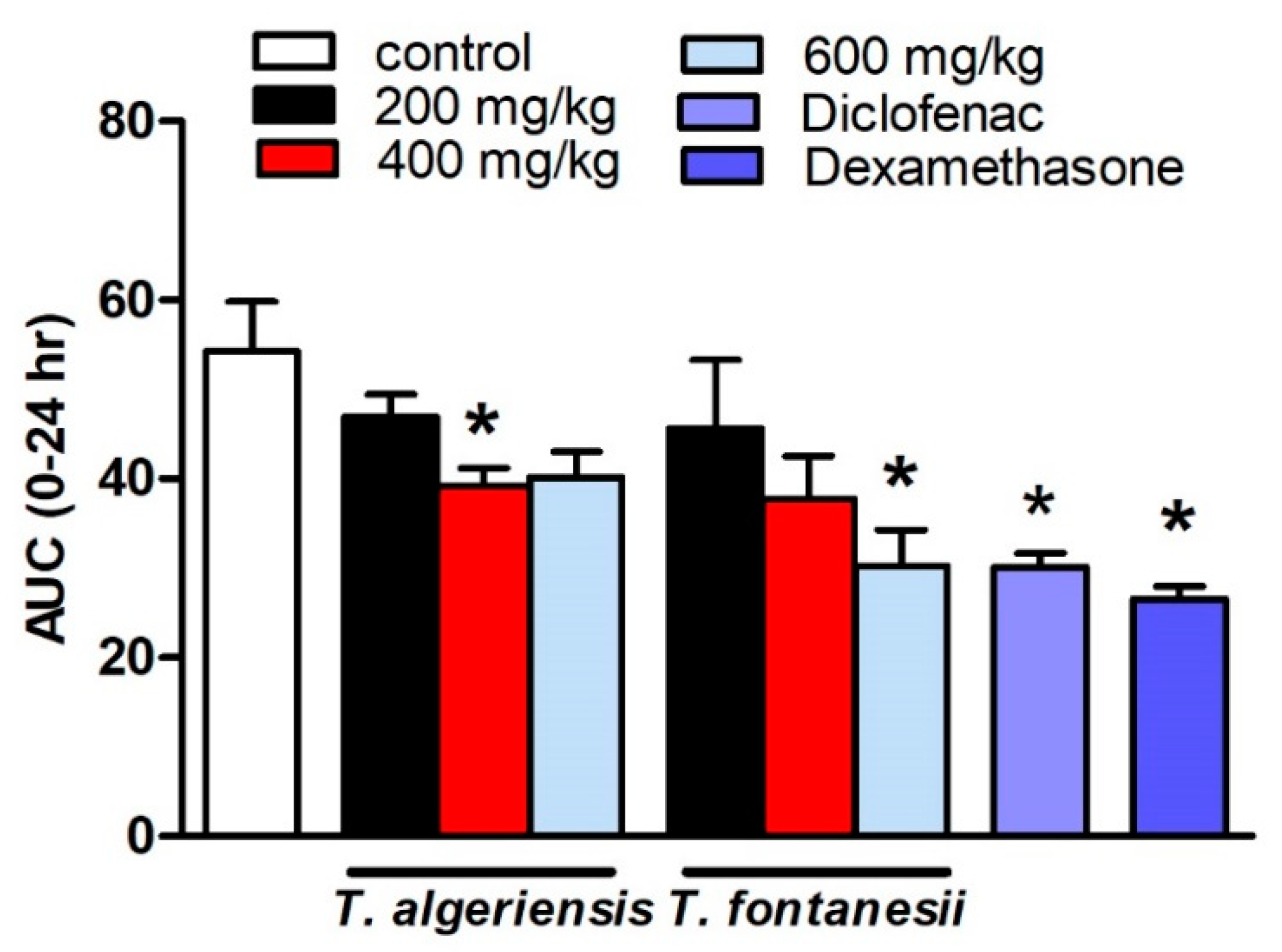
3.1.2. Effects of T. algeriensis and T. fontanesii Extracts on Carrageenan-Induced Paw Edema in Rats
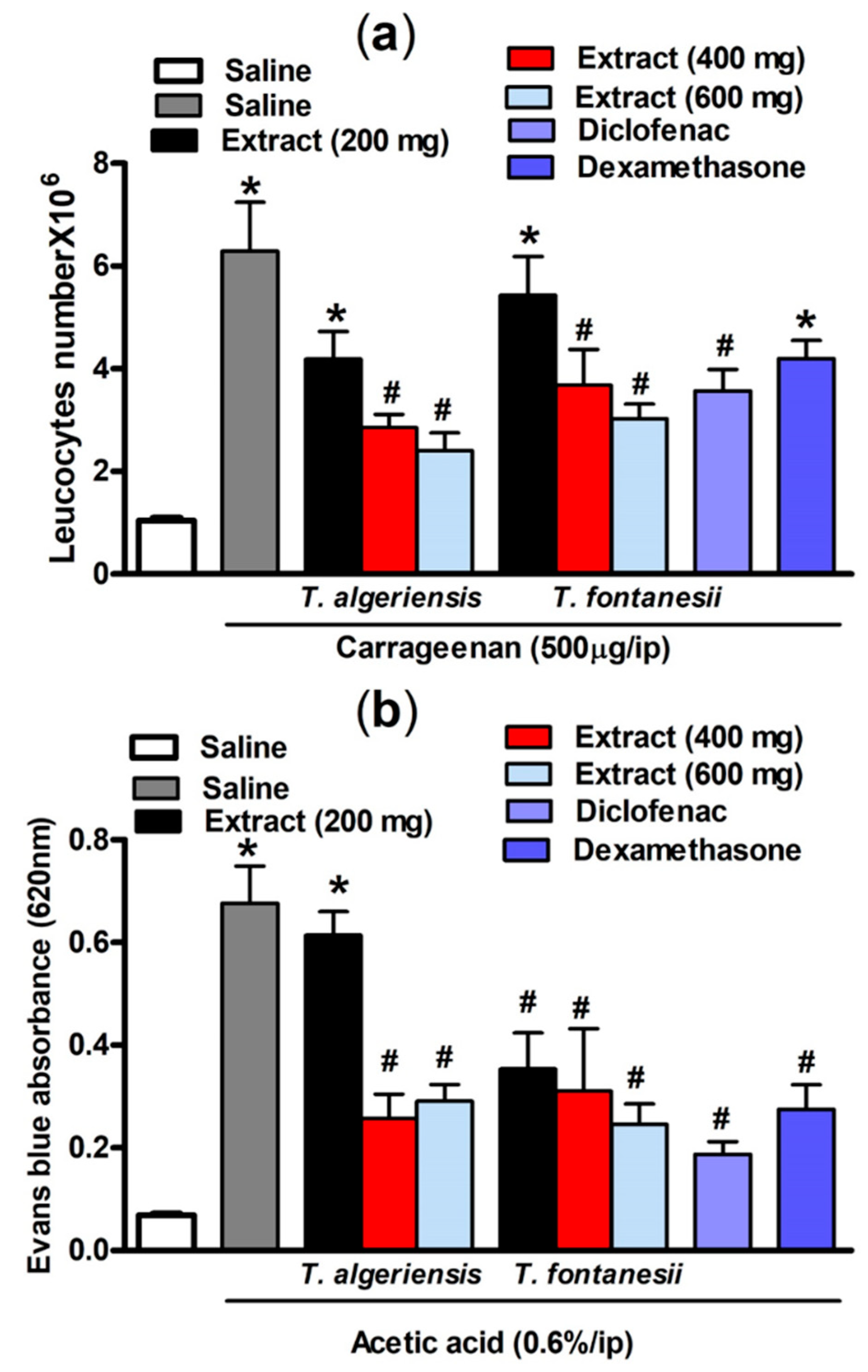
3.1.3. Effects on Carrageenan-Induced Leukocyte Migration into the Peritoneal Cavity in Mice
3.1.4. Effects on Acetic Acid-Induced Vascular Permeability in Mice
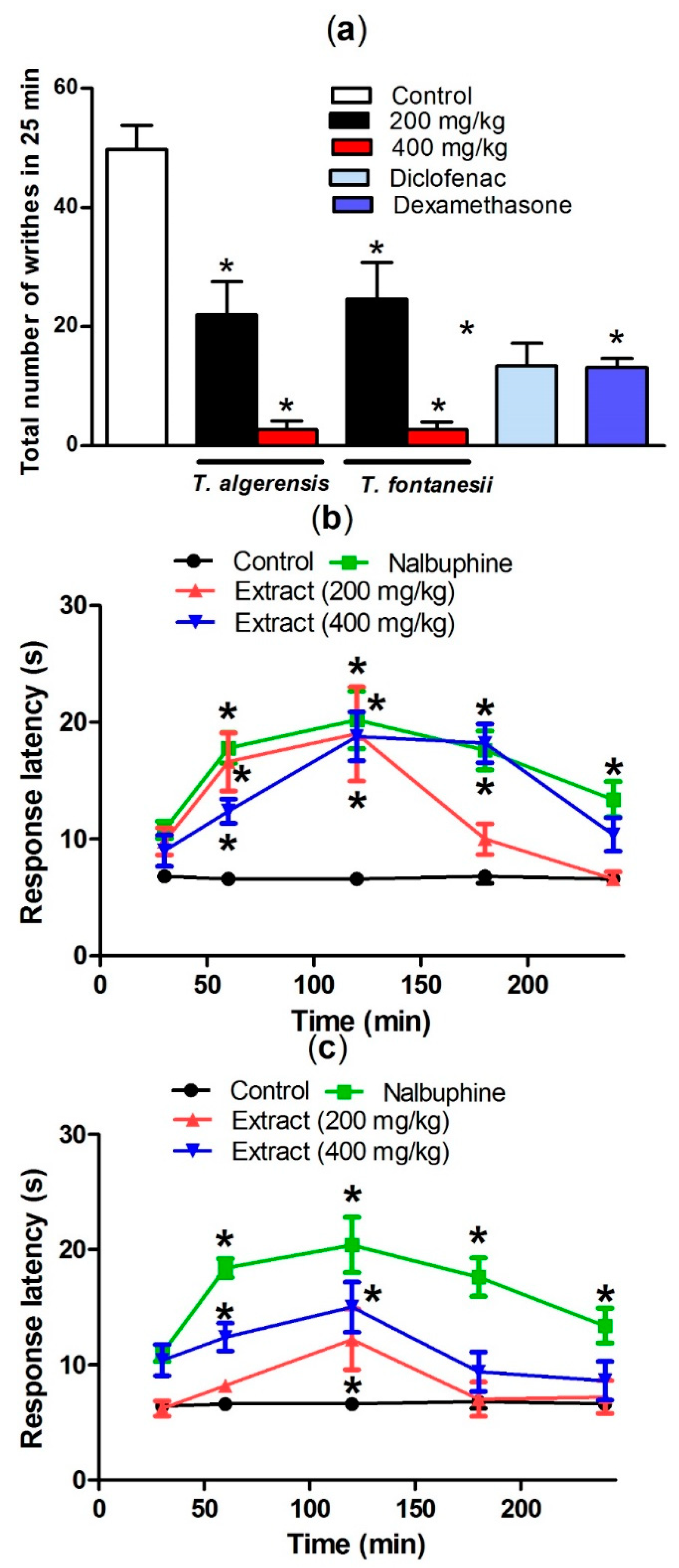
3.1.5. Effects on Acetic Acid-Induced Writhing and Hot Plate Test in Mice
3.1.6. Effect of Extracts on Brewer’s Yeast Induced Pyrexia in Mice
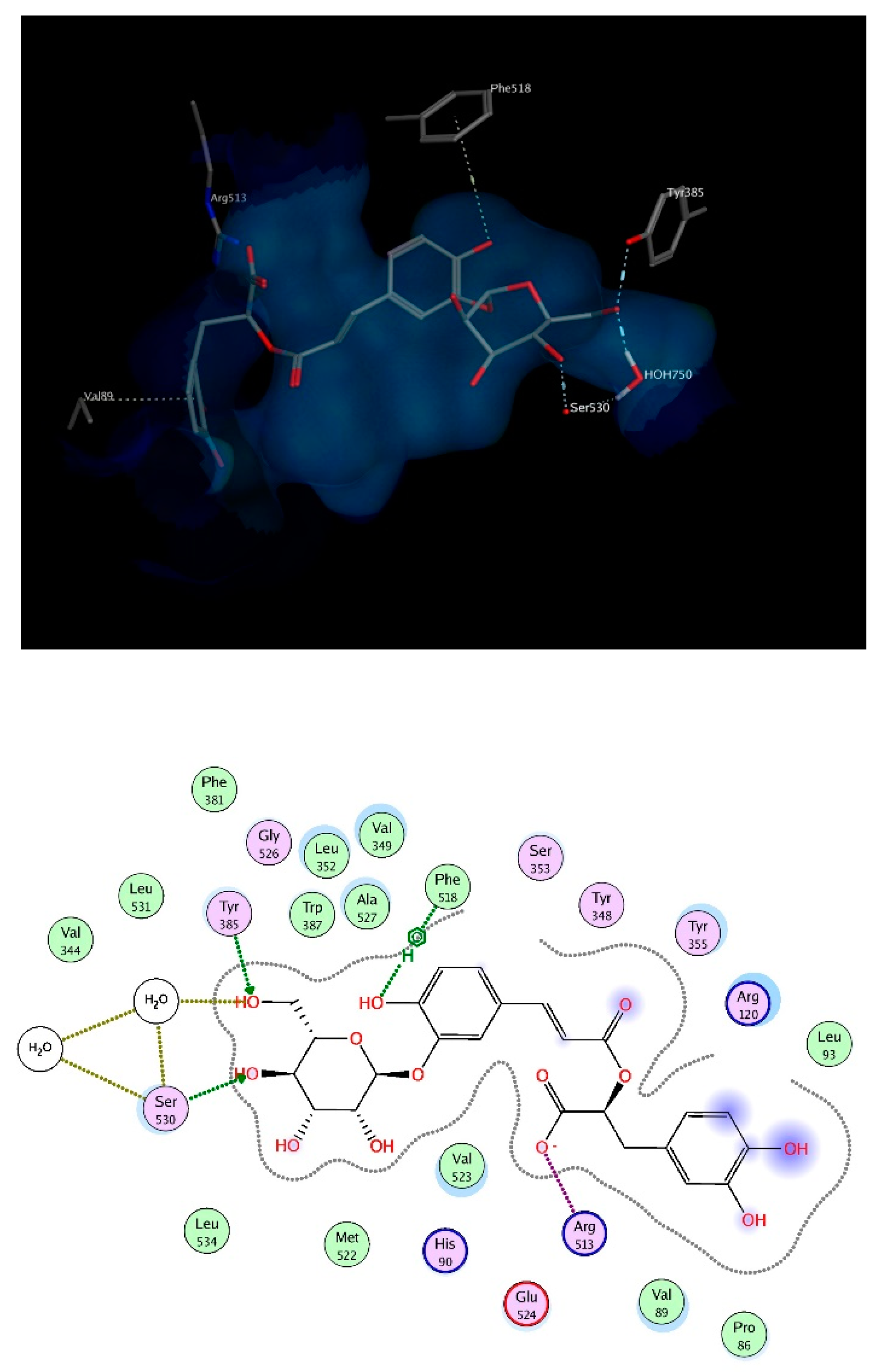
3.2. Molecular Docking
3.2.1. Cycloxygenase-1
3.2.2. Cycloxygenase-2 Protein
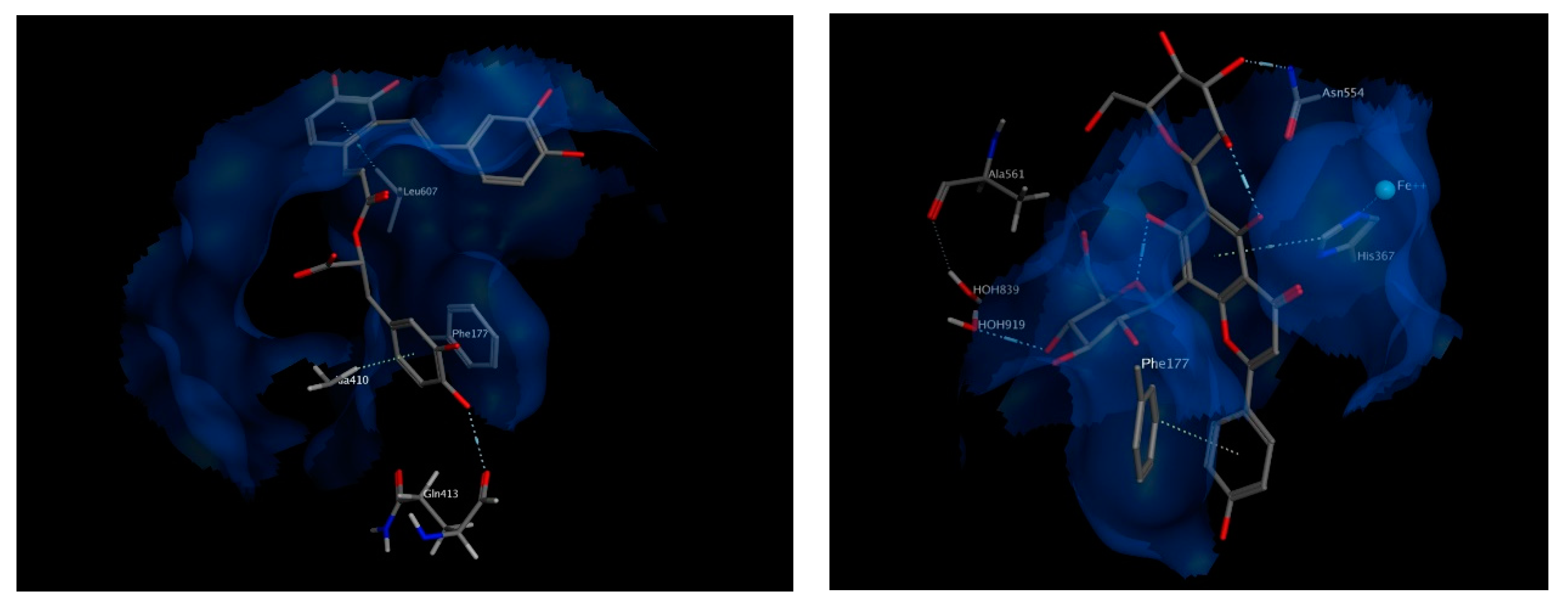
3.2.3. -Lipoxygenase Protein
3.2.4. 5 Lipoxygenase Activating Protein (FLAP)
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Nathan, C.F. Points of control in inflammation. Nature 2002, 420, 846–852. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Chen, P.; Tang, C.; Wang, Y.; Li, Y.; Zhang, H. Antinociceptive and anti-inflammatory activities of extract and two isolated flavonoids of Carthamus tinctorius L. J. Ethnopharmacol. 2014, 151, 944–950. [Google Scholar] [CrossRef] [PubMed]
- Ziani, B.E.; Heleno, S.A.; Bachari, K.; Dias, M.I.; Alves, M.J.; Barros, L.; Ferreira, I.C. Phenolic compounds characterization by LC-DAD-ESI/MSn and bioactive properties of Thymus algeriensis Boiss. & Reut. and Ephedra alata Decne. Food Res. Int. 2019, 116, 312–319. [Google Scholar] [PubMed] [Green Version]
- Guesmi, F.; Beghalem, H.; Tyagi, A.K.; Ben Ali, M.; Ben Mouhoub, R.; Bellamine, H.; Landoulsi, A. Prevention of H2O2 induced oxidative damages of rat testis by Thymus algeriensis. Biomed. Environ. Sci. 2016, 29, 275–285. [Google Scholar]
- Ghareeb, M.; Sobeh, M.; El-Maadawy, W.H.; Mohammed, H.S.; Khalil, H.; Botros, S.S.; Wink, M. Chemical profiling of polyphenolics in Eucalyptus globulus and evaluation of its hepato-renal protective potential against cyclophosphamide induced toxicity in mice. Antioxidants 2019, 8, 415. [Google Scholar] [CrossRef] [Green Version]
- AbdelAll, E.; Lamie, P.F.; Ali, W.A. Cyclooxygenase-2 and 15-lipoxygenase inhibition, synthesis, anti-inflammatory activity and ulcer liability of new celecoxib analogues: Determination of region-specific pyrazole ring formation by NOESY. Bioorganic Med. Chem. Lett. 2016, 26, 2893–2899. [Google Scholar] [CrossRef]
- Sobeh, M.; Mahmoud, M.F.; Rezq, S.; Alsemeh, A.E.; Sabry, O.M.; Mostafa, I.; Abdelfattah, M.A.O.; El-Allem, K.A.; Shazly, A.M.; Yasri, A.; et al. Salix tetrasperma Roxb. extract alleviates neuropathic pain in rats via modulation of the NF-κB/TNF-α/NOX/iNOS pathway. Antioxidants 2019, 8, 482. [Google Scholar] [CrossRef] [Green Version]
- Liu, C.; Su, H.; Wan, H.; Qin, Q.; Wu, X.; Kong, X.; Lin, N. Forsythoside A exerts antipyretic effect on yeast-induced pyrexia mice via inhibiting transient receptor potential vanilloid 1 function. Int. J. Biol. Sci. 2017, 13, 65–75. [Google Scholar] [CrossRef] [Green Version]
- Nabet, N.; Gilbert-Lopez, B.; Madani, K.; Herrero, M.; Ibáñez, E.; Mendiola, J.A. Optimization of microwave-assisted extraction recovery of bioactive compounds from Origanum glandulosum and Thymus fontanesii. Ind. Crop. Prod. 2019, 129, 395–404. [Google Scholar] [CrossRef]
- Vergara-Salinas, J.R.; Pérez-Jiménez, J.; Torres, J.L.; Agosin, E.; Pérez-Correa, J.R. Effects of temperature and time on polyphenolic content and antioxidant activity in the pressurized hot water extraction of deodorized Thyme (Thymus vulgaris). J. Agric. Food Chem. 2012, 60, 10920–10929. [Google Scholar] [CrossRef]
- El-Domiaty, M.M.; El-Shafae, A.M.; Abdel-Aal, M.M. A flavanol, flavanone and highly-oxygenated flavones from Thymus algeriensis Boiss. Alexanderia J. Pharm. Sci. 1997, 11, 13–17. [Google Scholar]
- Boutaoui, N.; Zaiter, L.; Benayache, S.; Benayache, S.; Carradori, S.; Cesa, S.; Giusti, A.M.; Campestre, C.; Menghini, L.; Innosa, D.; et al. Qualitative and quantitative phytochemical analysis of different extracts from Thymus algeriensis aerial parts. Molecules 2018, 23, 463. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Di Rosa, M.; Willoughby, D.A. Screens for anti-inflammatory drugs. J. Pharm. Pharmacol. 1971, 23, 297–298. [Google Scholar] [CrossRef] [PubMed]
- Vinegar, R.; Schreiber, W.; Hugo, R. Biphasic development of carrageenin edema in rats. J. Pharmacol. Exp. Ther. 1969, 166, 96–103. [Google Scholar]
- Meshram, G.G.; Kumar, A.; Rizvi, W.; Tripathi, C.; Khan, R. Evaluation of the anti-inflammatory activity of the aqueous and ethanolic extracts of the leaves of Albizzia lebbeck in rats. J. Tradit. Complement. Med. 2015, 6, 172–175. [Google Scholar] [CrossRef] [Green Version]
- Matsumoto, K.; Obara, S.; Kuroda, Y.; Kizu, J. Anti-inflammatory effects of linezolid on carrageenan-induced paw edema in rats. J. Infect. Chemother. 2015, 21, 889–891. [Google Scholar] [CrossRef]
- Wei, Y.; Chen, J.; Hu, Y.; Lu, W.; Zhang, X.; Wang, R.; Chu, K. Rosmarinic acid mitigates lipopolysaccharide-induced neuroinflammatory responses through the inhibition of TLR4 and CD14 expression and NF-κB and NLRP3 inflammasome activation. Inflammation 2018, 41, 732–740. [Google Scholar] [CrossRef]
- Mansourabadi, A.H.; Sadeghi, H.M.; Razavi, N.; Rezvani, E. Anti-inflammatory and analgesic properties of Salvigenin, Salvia officinalis flavonoid extracted. Adv. Herb. Med. 2015, 1, 31–34. [Google Scholar]
- Noori, S.; Hassan, Z.M.; Yaghmaei, B.; Dolatkhah, M. Antitumor and immunomodulatory effects of salvigenin on tumor bearing mice. Cell. Immunol. 2013, 286, 16–21. [Google Scholar] [CrossRef]
- Sherwood, E.R.; Tolliver-Kinsky, T. Mechanisms of the inflammatory response. Best Pr. Res. Clin. Anaesthesiol. 2004, 18, 385–405. [Google Scholar] [CrossRef]
- Ikeda, Y.; Ueno, A.; Naraba, H.; Oh-Ishi, S.; Ikeda-Matsuo, Y. Involvement of vanilloid receptor VR1 and prostanoids in the acid-induced writhing responses of mice. Life Sci. 2001, 69, 2911–2919. [Google Scholar] [CrossRef]
- Nidavani, R.B.; Mahalakshmi, A.M.; Shalawadi, M. Vascular permeability and evans blue dye: A physiological and pharmacological approach. J. Appl. Pharm. Sci. 2014, 4, 106–113. [Google Scholar]
- Khan, H.; Saeed, M.; Gilani, A.-H.; Khan, M.; Dar, A.; Khan, I. The antinociceptive activity of Polygonatum verticillatum rhizomes in pain models. J. Ethnopharmacol. 2010, 127, 521–527. [Google Scholar] [CrossRef] [PubMed]
- Malvar, D.D.C.; Soares, D.; Fabricio, A.S.C.; Kanashiro, A.; Machado, R.R.; Figueiredo, M.J.; Rae, G.A.; Souza, G. The antipyretic effect of dipyrone is unrelated to inhibition of PGE2 synthesis in the hypothalamus. Br. J. Pharmacol. 2011, 162, 1401–1409. [Google Scholar] [CrossRef] [Green Version]
- Malvar, D.D.C.; Aguiar, F.A.; Vaz, A.D.L.L.; Assis, D.C.R.; De Melo, M.C.C.; Jabor, V.A.P.; Kalapothakis, E.; Ferreira, S.H.; Clososki, G.; Souza, G. Dipyrone metabolite 4-MAA induces hypothermia and inhibits PGE2-dependent and -independent fever while 4-AA only blocks PGE2-dependent fever. Br. J. Pharmacol. 2014, 171, 3666–3679. [Google Scholar] [CrossRef] [Green Version]
- Lin, C.; Zhang, X.; Su, Z.; Xiao, J.; Lv, M.; Cao, Y.; Chen, Y. Carnosol improved lifespan and healthspan by promoting antioxidant capacity in Caenorhabditis elegans. Oxidative Med. Cell. Longev. 2019, 2019, 5958043. [Google Scholar] [CrossRef]
- Hsu, A.-L.; Murphy, C.T.; Kenyon, C. Regulation of aging and age-related disease by DAF-16 and heat-shock factor. Science 2003, 300, 1142–1145. [Google Scholar] [CrossRef] [Green Version]
- Munné-Bosch, S.; Schwarz, K.; Alegre, L. Response of abietane diterpenes to stress in Rosmarinus officinalis L.: New insights into the function of diterpenes in plants. Free. Radic. Res. 1999, 31, 107–112. [Google Scholar] [CrossRef]
- Johnson, J.J. Carnosol: A promising anti-cancer and anti-inflammatory agent. Cancer Lett. 2011, 305, 1–7. [Google Scholar] [CrossRef] [Green Version]
- Subbaramaiah, K.; A Cole, P.; Dannenberg, A.J. Retinoids and carnosol suppress cyclooxygenase-2 transcription by CREB-binding protein/p300-dependent and -independent mechanisms. Cancer Res. 2002, 62, 2522–2530. [Google Scholar]
- Orlando, B.J.; Malkowski, M. Substrate-selective Inhibition of cyclooxygeanse-2 by fenamic acid derivatives is dependent on peroxide tone. J. Biol. Chem. 2016, 291, 15069–15081. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Blobaum, A.L.; Marnett, L.J. Structural and functional basis of cyclooxygenase inhibition. J. Med. Chem. 2007, 50, 1425–1441. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cingolani, G.; Panella, A.; Perrone, M.G.; Vitale, P.; Di Mauro, G.; Fortuna, C.G.; Scilimati, A. structural basis for selective inhibition of cyclooxygenase-1 (COX-1) by diarylisoxazolesmofezolac and 3-(5-chlorofuran-2-yl)-5-methyl-4-phenylisoxazole (P6). Eur. J. Med. Chem. 2017, 138, 661–668. [Google Scholar] [CrossRef] [PubMed]
- Rowlinson, S.W.; Crews, B.C.; Lanzo, C.A.; Marnett, L.J. The binding of arachidonic acid in the cyclooxygenase active site of mouse prostaglandin endoperoxide synthase-2 (COX-2) a putative L-shaped binding conformation utilizing the top channel region. J. Biol. Chem. 1999, 274, 23305–23310. [Google Scholar] [CrossRef] [Green Version]
- Tsolaki, E.; Eleftheriou, P.; Kartsev, V.; Geronikaki, A.; Saxena, A. Application of docking analysis in the prediction and biological evaluation of the lipoxygenase inhibitory action of thiazolyl derivatives of mycophenolic acid. Molecules 2018, 23, 1621. [Google Scholar] [CrossRef] [Green Version]
- Blevitt, J.M.; Hack, M.D.; Herman, K.; Chang, L.; Keith, J.M.; Mirzadegan, T.; Rao, N.L.; Lebsack, A.D.; Milla, M.E. A single amino acid difference between mouse and human 5-Lipoxygenase activating protein (FLAP) explains the speciation and differential pharmacology of novel FLAP inhibitors. J. Biol. Chem. 2016, 291, 12724–12731. [Google Scholar] [CrossRef] [Green Version]
- Evans, J.F.; Ferguson, A.D.; Mosley, R.T.; Hutchinson, J.H. What’s all the FLAP about? 5-Lipoxygenase-activating protein inhibitors for inflammatory diseases. Trends Pharmacol. Sci. 2008, 29, 72–78. [Google Scholar] [CrossRef]
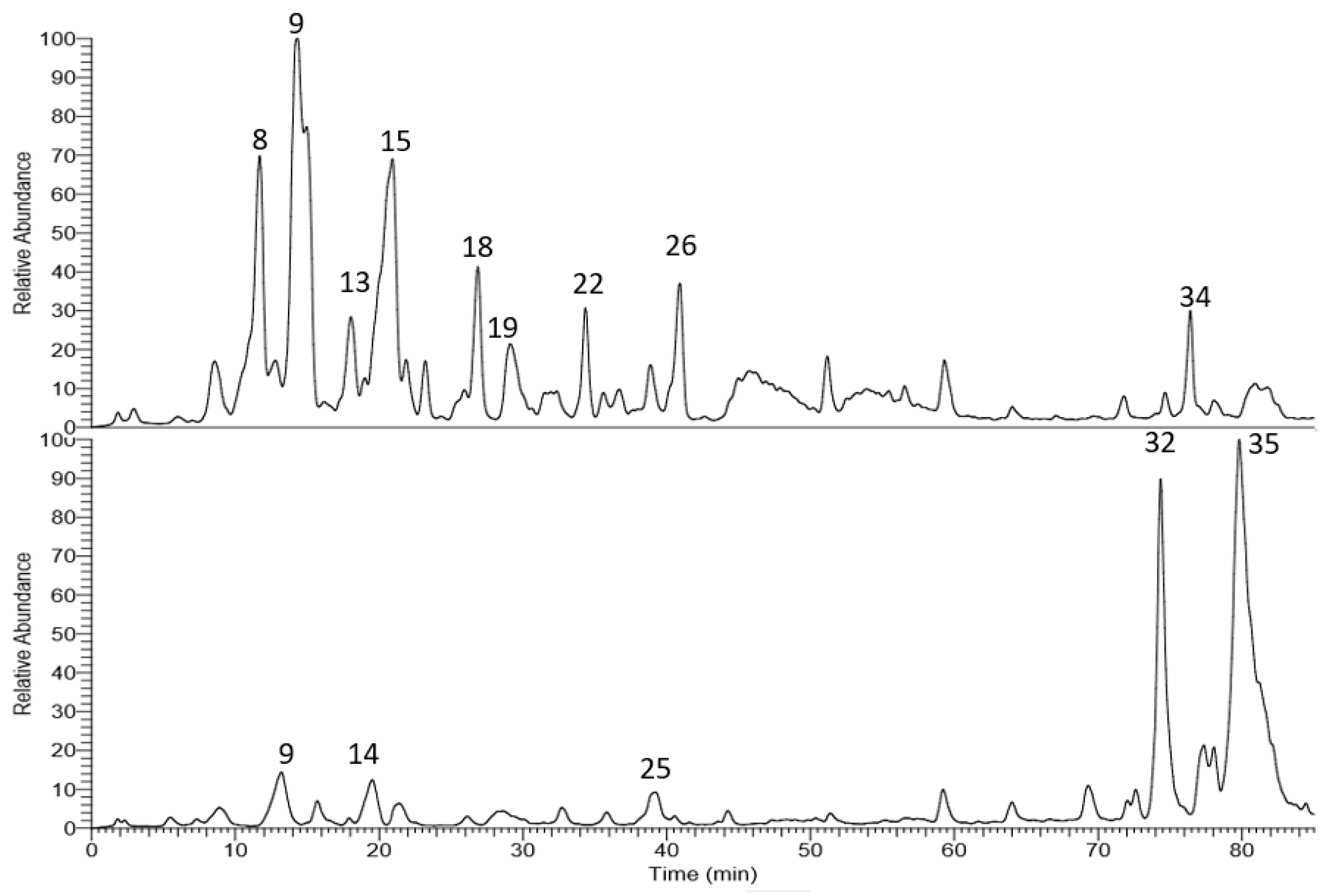



| No. | Rt | M-H | MS/MS | Relative Abundance | Proposed Compounds | |
|---|---|---|---|---|---|---|
| T. algeriensis | T. fontanesii | |||||
| 1 | 1.46 | 191 | + | - | Quinic acid a | |
| 2 | 1.60 | 133 | 115 | + | - | Malic acid |
| 3 | 1.87 | 341 | 179 | + | + | Caffeic acid glucoside a,b |
| 4 | 2.66 | 305 | 97, 225 | + | + | Gallocatechin a |
| 5 | 8.58 | 387 | 163, 179, 369 | + | + | 12-Hydroxyjasmonic acid 12-O-hexoside |
| 6 | 9.47 | 165 | 99, 149, 165 | + | + | Phloretic acid |
| 7 | 10.55 | 493 | 161, 179, 359 | + | + | Salvianolic Acid A |
| 8 | 11.68 | 555 | 161, 359, 493 | +++ | + | Salvianolic acid K c |
| 9 | 12.85 | 521 | 161, 359 | +++ | + | Rosmarinic acid glucoside c |
| 10 | 14.37 | 359 | 161, 179, 197 | + | + | Rosmarinic acid a,c |
| 11 | 16.90 | 385 | 153, 161, 223 | - | + | Sinapic acid glucoside |
| 12 | 17.58 | 623 | 179,315, 447 | + | - | Isorhamnetin pentosyl-glucuronide |
| 13 | 17.76 | 471 | 165, 309, 369 | + | - | Phloretic acid caffeoyl 3-hydroxy-3-methylglutaroyl |
| 14 | 18.17 | 327 | 161, 165 | - | + | Caffeoyl-phloretic acid |
| 15 | 20.12 | 461 | 175, 285 | +++ | + | Luteolin glucuronide c |
| 16 | 21.55 | 593 | 269, 353, 413, 473 | + | + | Apigenin 6,8-di-C-hexosides c |
| 17 | 25.79 | 449 | 151, 287 | + | - | Eriodictyol glucoside c |
| 18 | 26.91 | 551 | 161, 359, 519 | ++ | + | Schizotenuin F |
| 19 | 29.13 | 549 | 161, 387, 531 | + | + | Caffeoyl ethylrosmarinate |
| 20 | 32.33 | 303 | 125, 177, 285 | + | + | Dihydroquercetin |
| 21 | 34.26 | 563 | 193, 387, 531 | + | + | Feruloyl ethylrosmarinate |
| 22 | 35.53 | 637 | 285, 461 | ++ | - | Luteolin feruloyl glucuronide |
| 23 | 35.78 | 609 | 301, 447 | - | + | Querectin rhamnosyl-glucoside c |
| 24 | 38.52 | 579 | 285, 417, 447 | + | - | Luteolin pentosyl-glucoside |
| 25 | 38.85 | 433 | 301 | - | + | Quercetin pentoside |
| 26 | 40.79 | 447 | 161, 285 | ++ | + | Luteolin glucoside |
| 27 | 43.97 | 329 | 125, 229, 329 | - | + | Thymusin |
| 28 | 44.75 | 417 | 161, 285, 417 | + | - | Luteolin pentoside |
| 29 | 51.34 | 287 | 151, 269 | + | + | Eriodictyol d |
| 30 | 59.52 | 271 | 151, 177 | + | + | Naringenin a,e |
| 31 | 64.01 | 269 | 149, 225, 269 | - | + | Apigenin a |
| 32 | 74.31 | 329 | 270, 286, 329 | + | ++++ | Carnosol a,b |
| 33 | 76.29 | 283 | 179, 266, 283 | + | + | Genkwanin |
| 34 | 77.36 | 343 | 179, 300, 325 | + | + | Xanthomicrol |
| 35 | 79.16 | 327 | 285, 299, 327 | - | ++++ | Salvigenin |
| Treatment | COX-1 | COX-2 | SI | 5-LOX | TAC |
|---|---|---|---|---|---|
| IC50 (µM) | IC50 (µM) | U/L | |||
| T. algeriensis Extract | 12.4 ± 0.49 | 0.05 ± 0.01 * | 248 | 2.70 ± 0.23 | 39.27± 3.47 |
| T. fontanesii Extract | 12.88 ± 0.94 | 0.04 ± 0.002 * | 322 | 2.5 ± 0.4 | 44.33 ± 4.6 @ |
| Celecoxib | 15.97 ± 1.03 | 0.06 ± 0.01 * | 266.2 | - | - |
| Diclofenac | 4.06 ± 0.22 | 0.76 ± 0.06 | 5.34 | 2.60 ± 0.21 | - |
| Indomethacin | 0.1 ± 0.01 | 0.72 ± 0.06 | 0.14 | - | - |
| Zileuton | - | - | - | 3.20 ± 0.15 | - |
| Ascorbic Acid | - | - | - | - | 27.6 ± 1.40 |
| Extract | Dose (mg/kg) | Rectal Temperature # | Rectal Temperature Recorded Following Different Treatments | ||||
|---|---|---|---|---|---|---|---|
| 30 min | 1 h | 2 h | 3 h | 24 h | |||
| Control | - | 38.36 ±0.27 | 38.68 ± 0.15 | 38.66 ± 0.19 | 38.80 ± 0.20 | 38.84 ± 0.30 | 38.26 ± 0.18 |
| T. algeriensis | 200 | 38.78 ± 0.12 | 38.56 ± 0.31 | 38.80 ± 0.40 | 39.00 ± 0.23 | 38.46 ± 0.20 | 37.88 ± 0.53 |
| 400 | 38.06 ± 0.52 | 38.14 ± 0.34 | 38.54 ± 0.29 | 38.54 ± 0.19 | 38.24 ± 0.16 | 37.52 ± 0.26 | |
| 600 | 39.18 ± 0.34 | 38.58 ± 0.29 | 37.85 ± 0.30 | 37.8 ± 0.25 | 38.10 ± 0.15 | 37.73 ± 0.18 | |
| T. fontanesii | 200 | 38.42 ± 0.29 | 37.90 ± 0.48 | 38.44 ± 0.42 | 38.52 ± 0.15 | 38.30 ± 0.13 | 37.28 ± 0.16 * |
| 400 | 38.52 ± 0.37 | 38.14 ± 0.28 | 38.44 ± 0.37 | 38.02 ± 0.09 | 37.82 ± 0.11 * | 37.24 ± 0.22 * | |
| 600 | 38.48 ± 0.24 | 38.15 ± 0.14 | 37.58 ± 0.15 * | 37.35 ± 0.16 * | 37.6 ± 0.12 * | 37.10 ± 0.23 * | |
| Paracetamol | 150 | 38.66 ± 0.18 | 38.18 ± 0.20 | 37.56 ± 0.30 | 37.06 ± 0.29 * | 36.94 ± 0.25 * | 36.54 ± 0.23 * |
| Compound Name | Docking Score (kcal/mol) | |||
|---|---|---|---|---|
| COX-1 | COX-2 | 5-LOX | FLAP | |
| Quinic acid | −9.29 | −11.98 | −13.78 | −9.20 |
| Malic acid | −9.54 | −9.89 | −8.85 | −7.48 |
| Caffeic acid glucoside | −15.79 | −18.14 | −12.90 | −16.35 |
| Phloretic acid | −9.39 | −10.55 | −9.40 | −12.37 |
| Salvianolic Acid A | −21.08 | −23.32 | −20.87 | −17.37 |
| Salvianolic acid K | −19.21 | −24.15 | −19.25 | −13.77 |
| Rosmarinic acid glucoside | −17.00 | −25.93 | −10.60 | −14.82 |
| Rosmarinic acid | −16.20 | −19.46 | −12.72 | −15.09 |
| Sinapic acid glucoside | −16.87 | −20.64 | −18.27 | −16.00 |
| Phloretic acid caffeoyl 3-hydroxy-3-methylglutaroyl | −16.88 | −22.27 | −1.06 | −10.56 |
| Caffeoyl-phloretic acid | −16.63 | −18.03 | −19.66 | −14.35 |
| Apigenin 6,8-di-C-hexosides | −20.48 | −25.36 | −22.19 | −14.06 |
| Schizotenuin F | −15.91 | −25.75 | −19.45 | −14.92 |
| Caffeoylethylrosmarinate | −18.63 | −23.83 | −19.27 | −15.37 |
| Feruloyl ethylrosmarinate | −15.63 | −22.04 | −14.17 | −14.62 |
| Quercetin pentoside | −17.20 | −18.85 | −19.57 | −16.79 |
| Luteolin glucoside | −17.89 | −24.48 | −18.46 | −14.39 |
| Naringenin | −13.96 | −14.61 | −11.34 | −12.80 |
| Carnosol | −12.78 | −13.47 | −12.88 | −13.23 |
| Genkwanin | −13.68 | −14.47 | −10.61 | −11.22 |
| Xanthomicrol | −15.16 | −16.07 | −14.87 | −14.71 |
| Salvigenin | −17.62 | −17.23 | −14.15 | −12.42 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sobeh, M.; Rezq, S.; Cheurfa, M.; Abdelfattah, M.A.O.; Rashied, R.M.H.; El-Shazly, A.M.; Yasri, A.; Wink, M.; Mahmoud, M.F. Thymus algeriensis and Thymus fontanesii: Chemical Composition, In Vivo Antiinflammatory, Pain Killing and Antipyretic Activities: A Comprehensive Comparison. Biomolecules 2020, 10, 599. https://doi.org/10.3390/biom10040599
Sobeh M, Rezq S, Cheurfa M, Abdelfattah MAO, Rashied RMH, El-Shazly AM, Yasri A, Wink M, Mahmoud MF. Thymus algeriensis and Thymus fontanesii: Chemical Composition, In Vivo Antiinflammatory, Pain Killing and Antipyretic Activities: A Comprehensive Comparison. Biomolecules. 2020; 10(4):599. https://doi.org/10.3390/biom10040599
Chicago/Turabian StyleSobeh, Mansour, Samar Rezq, Mohammed Cheurfa, Mohamed A.O. Abdelfattah, Rasha M.H. Rashied, Assem M. El-Shazly, Abdelaziz Yasri, Michael Wink, and Mona F. Mahmoud. 2020. "Thymus algeriensis and Thymus fontanesii: Chemical Composition, In Vivo Antiinflammatory, Pain Killing and Antipyretic Activities: A Comprehensive Comparison" Biomolecules 10, no. 4: 599. https://doi.org/10.3390/biom10040599








