1. Introduction
RNA extraction of high integrity and purity is a prerequisite for downstream application. Robust and generic methods for isolating RNA from animal species cells have been well established [
1]. However, isolation of high-quality RNA from plant material remains a challenge due either to the degradation of the RNA or a very low yield. This is particularly true for tissues of roots and tubers, which present high levels of polysaccharides, polyphenols, and other secondary metabolites [
2,
3,
4]. TRIzol reagent (Invitrogen; Thermo Fisher Scientific, lnc., Waltham, MA, USA), a typical guanidine isothiocyanate (GITC)-based RNA extraction buffers, is a widely used commercial kit for nucleic acid extraction. TRIzol can remove protein and DNA contamination efficiently, but it cannot remove polysaccharide contamination in starch-rich plant tissues [
5,
6]. Several cetyltrimethylammonium bromide (CTAB)-based methods have also been developed specifically for the extraction of plant RNA with high levels of polysaccharides [
7]. However, even though the CTAB-based method works well for leaf and shoot tissues, isolation of nucleic acids from roots and tubers with dramatically elevated amount of starch and other contaminants still remains a challenge.
Transfer RNAs (tRNAs), a class of highly conserved small non-coding RNA of typically 76 to 90mer, play a crucial role in messenger RNA decoding in nearly all domains of life. In addition to their well-known roles in translation, tRNAs have been shown to perform diverse regulatory functions in other cellular processes, such as apoptosis modulating and stress response programs [
8,
9]. For a long time, great deal of research efforts have been put into discovering and characterizing the chemical modifications in tRNAs, which have been found to significantly impact structural and functional activities of these biomolecules [
10,
11]. The most efficient approach for discovering and characterizing tRNA modifications has been based on liquid chromatography tandem mass spectrometry (LC-MS/MS) [
12,
13,
14]. To date, more than 100 modified nucleosides have been identified in 715 tRNAs of 77 different organisms, including mainly bacteria, archaea, animal tissues, and plant leaves [
15]. However, tRNAs in plant roots or tubers that have been widely used for food (e.g., potatoes) and medical purpose (e.g., ginseng) for centuries are rarely studied. Fresh leaves are preferred materials for plant tRNA studies, because high-quality tRNA samples can be more easily extracted from leaves than roots and tubers [
16,
17]. As the epigenetic changes are distinct for different organs and cell types, studies on tRNAs in various organs, including roots and tubers, are imperative [
18].
The root of
Panax ginseng C.A. Mey is a globally well-known traditional Chinese medicine (TCM). Ginseng roots are rich in polysaccharides mainly composed of starch-like glucans and pectins [
19], which easily bind or co-precipitate with RNA during isolation by using ethanol, affecting the yield and quality of RNAs. Polysaccharases, such as α-amylase and pectinase commonly used for extraction or structure identification of ginseng polysaccharides [
19,
20], can hydrolyze polysaccharides efficiently yielding monosaccharides or disaccharides that are more soluble in organic solvents, thus, possibly being useful in removing polysaccharide contaminations from nucleic acid. So far, modification profiles of tRNAs (i.e., identities and locations of modifications in tRNA) in ginseng roots still remain unexplored. In this study, a polysaccharase-aided RNA isolation (PARI) method was developed for the extraction of high-quality RNA from ginseng roots, and the post-transcriptional modifications of tRNA
Gly are initially profiled by using a LC-MS/MS method.
2. Materials and Methods
2.1. Plant Materials
Fresh roots of Panax ginseng C. A. Mey collected from Fusong Town, Jilin, China, were rinsed with distilled water and frozen immediately in liquid nitrogen.
2.2. Chemicals and Reagents
TRIzol reagent (Invitrogen), mirVanaTM miRNA isolation kit, and SYBR® gold nucleic acid gel stain were purchased from Thermo Fisher Scientific (Waltham, MA, U.S.A.). Cetyltrimethylammonium bromide (CTAB) was purchased from Kingdin Industrial Co., Ltd. (Hong Kong, China). Water-saturated phenol was purchased from Leagene Co., Ltd. (Beijing, China). Chloroform and ethanol were purchased from Anaqua Chemicals Supply Inc. Ltd. (Houston, TX, USA). A-amylase was from Bacillus subtilis. Pectinase was from Aspergillus niger. Glucoamylase was from Rhizopus and guanidinium thiocyanate was purchased from Tokyo Chemical Industry Co., Ltd. (Tokyo, Japan). A low-rangessRNA ladder was purchased from New England Biolabs (Beverly, MA, USA). A 40% acrylamide/bis solution (19:1), ammonium persulphate (APS), and tetramethylethylenediamine (TEMED) were purchased from Bio-Rad Laboratories Inc. (Hercules, CA, USA). Biotinylated single-stranded DNA oligos (30 mer) were customized from BGI Tech Solutions (Beijing Liuhe) Co., Ltd. (Guangzhou, China).
2.3. Extraction of Total RNA from Ginseng Roots
Total RNA were extracted from ginseng roots according to the protocol of the polysaccharases-aided RNA isolation method (PARI), as well as two conventional methods based on CTAB and TRIzol reagent reported previously, with some minor modifications, respectively. To ensure the extraction to be carried out in an RNase-free environment, all surfaces and pipettors were cleaned with RNase Zap solution (Invitrogen; Thermo Fisher Scientific, lnc., Waltham, MA, USA) and plastic wares were autoclaved before use. For the PARI method, around 200 mg ginseng root tissues were ground into a fine powder in liquid nitrogen using a clean mortar and pestle, which was then homogenized in 1 mL TRIzol reagent using a digital dispersing device (IKA, Staufen, Germany). After the plant cells were fully lysed for 10 min at room temperature, an equal volume of chloroform was added to the tissue lysate, followed by vigorous vortex and centrifugation at 12,000× g for 15 min at 4 °C. The upper aqueous phase was collected and transferred carefully to a new Eppendorf tube. RNA and polysaccharides in the supernatant were precipitated by adding 1/25 volume of 5 M sodium chloride and 1.25 volume of cold absolute ethanol, and stored at −20 °C for 30 min. After centrifuging at 12,000× g for 15 min, polysaccharide contaminations in the pellet were resuspended in water and selectively degraded by polysaccharases, including α-amylase, pectinase, and glucoamylase, respectively, until the pellet was completely dissolved. The hydrolysate was mixed with 2× CTAB buffer (2.0% CTAB, 2% PVP, 2M NaCl, 100 Mm Tris-HCl (pH 8.0), 25 mM EDTA, 1.0% β-mercaptoethanol) and extracted with an equal volume of phenol-chloroform-IAA (50:48:1) by vortexing vigorously. Phases wereseparated by centrifuging at 12,000× g for 15 min, and upper phase was collected and extracted again with chloroform-IAA (24:1). The supernatant was collected and mixed with an equal volume of 6 M guanidinium thiocyanate buffer, followed by adding 100% ethanol to a final concentration of 55%. The total RNA in the mixture were isolated and enriched by passing through a filter cartridge containing a silicon gel membrane that immobilizes the RNA. The cartridge was then washed several times with 80% (v/v) ethanol solution and, finally, the total RNA was eluted with a low ionic-strength solution such as RNase-free water.
Protocols of CTAB and TRIzol methods modified from Chang et al. [
7] and Wang et al. [
2] were used in this study. Briefly, after ginseng samples (200 mg) was lysed with CTAB extraction buffer at 65 °C or TRIzol reagent at room temperature, the aqueous phase containing RNA were separated from the organic phase by adding equal volume of chloroform or chloroform-isoamyl alcohol. Total RNA in the supernatant were enriched and recovered by using a filter cartridge of silicon membrane described above in the present of guanidinium thiocyanate and ethanol.
2.4. Quality Assessment of Ginseng total RNA
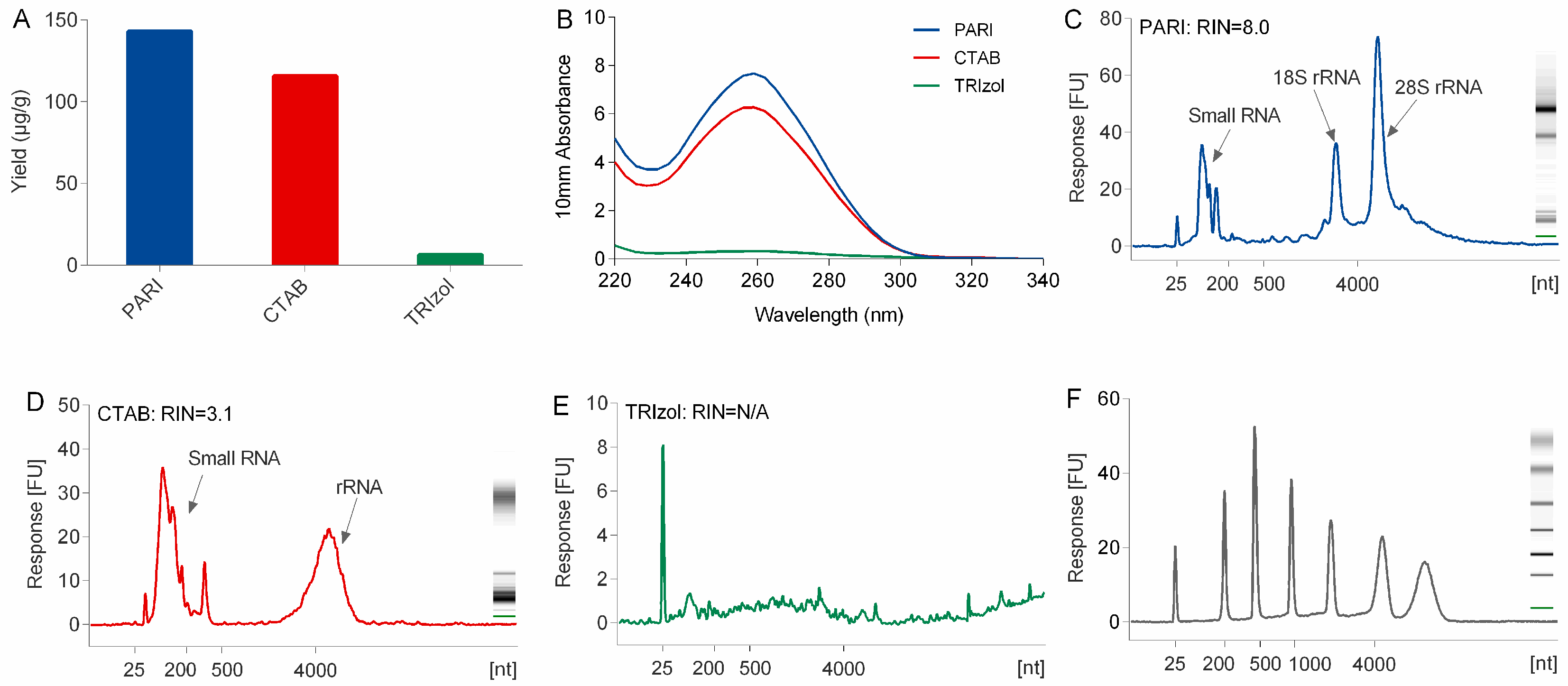
The yield and purity of the total RNA was spectrometrically assessed by using a NanoDrop spectrophotometer (Thermo Scientific, Waltham, MA, USA) at the wavelengths of 230, 260, and 280 nm. RNA integrity was assessed by its RNA integrity number (RIN) that was automatically generated by an Agilent 2100 BioAnalyzer (Agilent Technologies, Palo Alto, CA, USA).
The quality of total RNA was also confirmed by RT-PCR. For first-stand cDNA synthesis, 2 µg of total ginseng RNA in a volume of 20 μL was reverse-transcribed following instruction of Transcriptor Universal cDNA Master Kit (Roche). PCR amplifications were performed using primers described previously for cycloartenol synthase gene (Genebank accession number: AB009029) and β-amyrin synthase gene (Genebank accession number: AB014057). The β-actin gene was amplified as the reference gene (
Table S4). Thermocycling was carried out in a final volume of 50 μL containing 5 μL cDNA samples, 1 μL each of the primers (1μM of forward and reverse primers) and 25 μLDreamTaq Green PCR Master Mix (2X) (Thermo Scientific). A thermal cycling profile was conducted as showed in
Table S4. Reaction prodcts were analyzed by electrophoresis on SYBR-stained 2% agarose gels in TAE buffer and visualized with the Gel Doc XR imaging system (Bio-Rad Laboratories Inc., Hercules, CA, USA).
2.5. Preparation of Small RNA Fraction and tRNA-enriched Fraction (TEF)
The small RNA species (<200 mer) were isolated from the total RNA of ginseng roots by using a mirVana miRNA isolation kit following the manufacturer’s instruction. tRNAs were then gel-fractionated and purified from small RNA samples by electroelution as described previously with minor modifications [
21]. Briefly, 10 μg of small RNA were resolved by 6% urea-PAGE after denaturation for 2 min at 95 °C in loading buffer. After electrophoresis at 200 V in 1× TBE buffer for about 40 min until the bromophenol blue reached the bottom of the gel, RNA were visualized with SYBR
® gold nucleic gel stain under UV light. The region of the gel containing total tRNAs was cut out using a clean and sharp scalpel. The band was sliced and the total tRNAs were recovered from the gel by electroelution in dialysis bags of 3000 molecular weight cut-off (MWCO) at 100 V for 90 min in 1× TAE buffer. The eluents in the dialysis bags were collected and the TEF were desalted and concentrated by using the mirVana
TM miRNA isolation kit (Ambion; Thermo Fisher Scientific, lnc., Waltham, MA, USA).
2.6. TEF Library Construction and Sequencing
TEF libraries were constructed by using TruSeq small RNA Library Preparation Kit (Illumina, U.S.A.), followed by a round of adaptor ligation, reverse transcription and PCR enrichment (
Figure S3). PCR products were then purified and libraries were quantified on the Agilent Bioanalyzer 2100 system (Agilent Technologies, Palo Alto, CA, U.S.A.). The library preparations were sequenced at the Novogene Bioinformatics Institute (Beijing, China) on an Illumina HiSeq platform using the 150 bp paired-end (PE150) strategy.
2.7. Purification of Individual tRNA of Ginseng Roots
Ginseng tRNA
Gly(GCC) was isolated from small RNAs by immobilization of the target tRNA onto the streptavidin-coated magnetic beads with specific biotinylated capture DNA probes (
Table S5). Cognate DNA probes, which was designed based on the sequence information from NGS, were incubated with small RNA mixture at 65 °C for about 1.5 h in annealing buffer (1.2 M NaCl, 30 mM HEPES-KOH (pH 7.5), 15 mM EDTA, 0.5 mM DTT). Streptavidin-coated magnetic beads were then added to the mixture and incubated for 30 min at 65 °C. The biotinylated DNA/tRNA coated beads were separated with a magnet for 2 min and washed four times with washing buffer (0.1 M NaCl, 2.5 mM HEPES-KOH (pH 7.5), 1.25 mM EDTA, 0.5 mM DTT). The magnetic beads were re-suspended in RNase-free water and the immobilized tRNA molecules were released by incubation at 70 °C for 5 min. Purified tRNA were analyzed by electrophoresis on a 6% (
v/
v) polyacrylamide gels (PAGE) containing 8 M urea prepared according to the manufacturer’s protocol (Bio-Rad Laboratories Inc., Hercules, CA, USA).
2.8. Ribonuclease Digestion of tRNAGly(GCC)
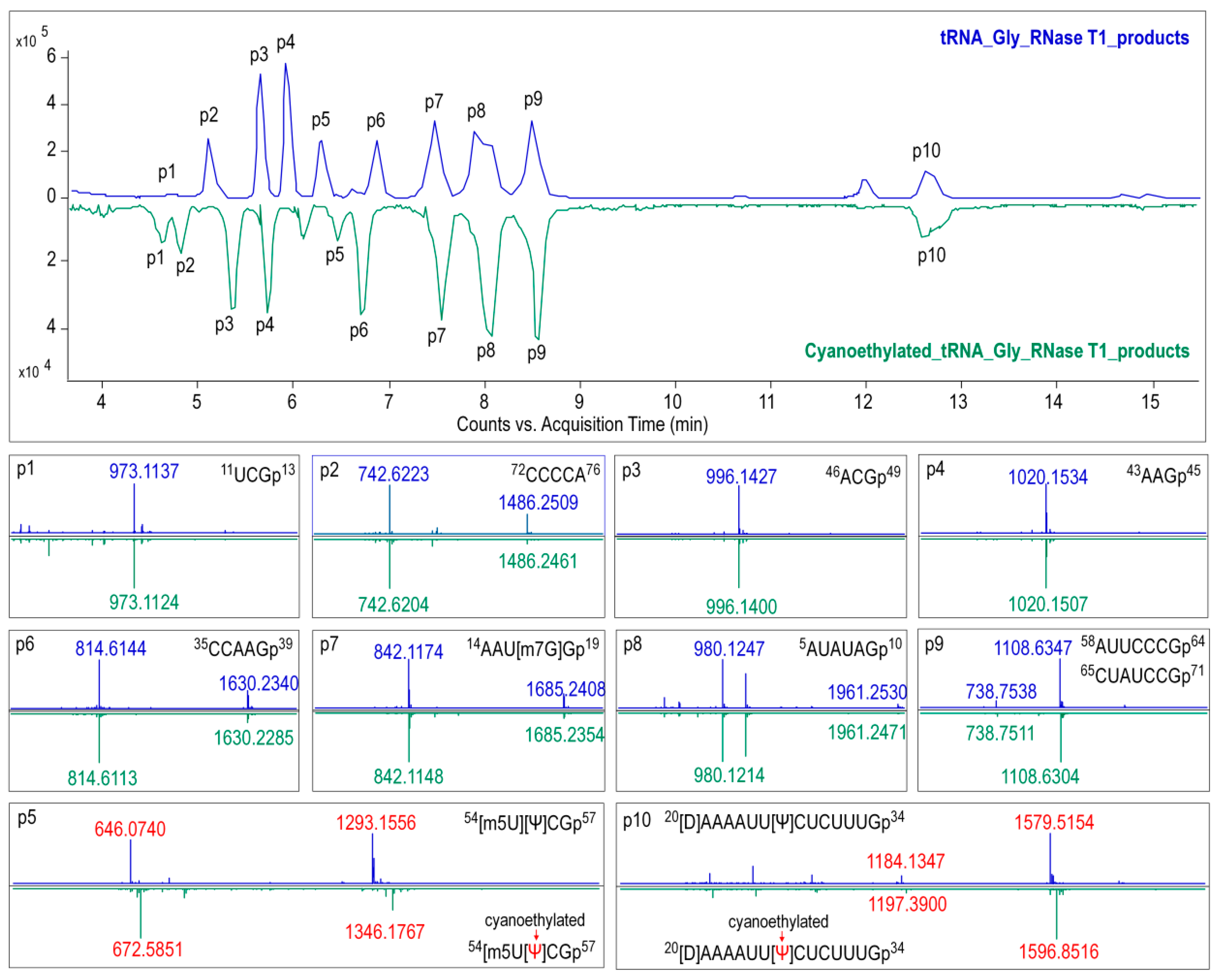
For RNase T1 digestion, 600 ng of tRNAGly(GCC) was added to 50 U of RNase T1 in 220 mM ammonium acetate buffer, and the mixture was then incubated at 37 °C for 1 h.Pseudouridine in the digestion products were cyanoethylated by reacting with acrylonitrile. The optimal conditions were as follows: 10 μL of RNase T1 digested tRNA was subjected to 26 μL of 41% ethanol/1.1 M triethyl ammonium acetate (pH 8.6) and 4 μL of acrylonitrile, then incubated at 70 °C for 2 h. Digestion samples treated with or without acrylonitrile were condensed for further analysis.
2.9. Preparation of Total Nucleosides
A total of 500 ng of tRNAGly(GCC) was added to 10 μL of 5× enzyme mixture containing 50 U of RNase I, 0.1 U of phosphodiesterase I, 30 U of bacterial alkaline phosphatase, 20 mM MgCl2, and 80 mM Tris-HCl (pH 8.0). The reaction system was adjusted to a final volume of 50 μL with RNase-free water, and the mixture was incubated at 37 °C for 3 h. The sample was lyophilized and rehydrated in mobile phase A (0.1% FA in water) described below.
2.10. LC-MS Analysis
Intact tRNA and RNase T1 digestion products were analyzed by using a time-of-flight mass spectrometer (6545 QTOF, Agilent) coupled to the 1290 infinity UPLC system equipped with a diode array detector (DAD). Oligonucleotide separations were performed on a Waters Acquity OST C18 column (1.7 µm, 2.1 × 100 mm). Mobile phase A was 15 mM TEA and 100 mM HFIP (pH = 8.5) in MS-grade water, and mobile phase B was a mixture of methanol/water (50:50,
v/
v) containing 15 mM TEA and 100 mM HFIP. The chromatographic gradient was shown in
Table S6. Mass spectrums were recorded in negative mode over an m/z range of 600 to 2000 for MS1 scan and 100–1800 for MS2. Collision energy ranging from 10 to 45 V was used for precursor ion fragmentation. System operations and data acquisition were performed by Masshunter Data Acquisition Software (Version B.07.00, Agilent), and the instrument settings were as follows: gas temperature, 320 °C; gas flow, 12 L/min; nebulizer, 35 psi; sheath gas temperature, 350 °C; sheath gas flow, 12 L/min; and fragmenter, 220 V (
Table S7).
An Agilent 6550 Q-TOF mass spectrometer coupled with 1290 UHPLC was used for identifying isomers of modified nucleosides. The liquid chromatography separation was performed on an Agilent Poroshell 120 HPLC column (2.7 μm, 4.6 × 100 mm) at 35 °C with a mobile phase flow rate of 0.4 mL/min. Nucleotides were eluted using mobile phase A (0.1% formic acid in water) and B (0.1% formic acid in acetonitrile). The gradient started at 1.5% B and increased as follows: 4% B at 4 min, 15% B at 12 min, 25% B at 18 min, then returning to 1.5% B and holding for 3 min. Mass spectra were recorded in positive mode over an m/z range of 100 to 1000 for MS1 scan, and the instrument settings were summarized in
Tables S8 and S9.
2.11. Data Analysis
Precursor ions of tRNA and digestion products in MS1 data were identified by matching them to their expected m/z values, which were calculated using the MongoOligo on-line calculator (
https://mods.rna.albany.edu/masspec/Mongo-Oligo). Sequence informative product ions in LC-MS/MS data of each digestion product were analyzed by RNAModmapper (RAMM) in a variable mode. The mass tolerance was set to 0.8 Da, and
p-scores of 55 and above were considered to be significant [
14].
4. Discussion
In this study, polysaccharases were unprecedentedly integrated into the process of RNA extraction of ginseng roots. Three enzymes, including α-amylase, pectinase, and glucoamylase were selected based on the types of ginseng polysaccharides. It can be expected that α-amylase will work better for ginseng roots than other polysaccharases because starch-type polysaccharides readily hydrolyzed by α-amylase make up over 60% of water-soluble polysaccharides of ginseng roots, while substrates for pectinases including various pectins with different pectic domains account for only a small portion (<9%) of ginseng’s polysaccharides (
Figure S4) [
19]. Actually, α-amylase made a real difference to ginseng RNA isolation in our lab. As shown in the left tube (No. 1) in
Figure 1F, large amounts of polysaccharides released from ginseng tissues clumped up in ethanol-water solution to form a sticky and chunky wad wrapping almost all the nucleic acid up in it. When α-amylase was added, these polysaccharides were rapidly and specifically hydrolyzed to monoses that are freely soluble in high concentration of ethanol (tube no. 2 in
Figure 1F), keeping the RNA free from contamination and degradation.
Compared with TRIzol and CTAB method, the PARI method is better at the isolation of high-quality RNA from polysaccharide-rich planttissues. The low yield problem of the TRIzol method is due mainly to the co-precipitation of a large portion of ginseng RNA with polysaccharides that were difficult to dissolve when they were exposed to a high concentration (>30%,
v/
v) of alcohol. CTAB naturally holds the power to segregate RNA from polysaccharide. However, severe degradation of ginseng RNA in CTAB extraction buffer was observed in this study (
Figure 2). This is mainly caused by heating the lysis buffer to 65 °C at the first step of the CTAB protocol [
7], which is necessary and inevitable for releasing nucleic acid out of cells completely. Although several modified CTAB methods with heating have been developed for many plant species, none of them could be applied successfully in our lab for the extraction of ginseng RNA because they could be more susceptible to degradation by heating even within two minutes. In the PARI method, the ginseng root was treated at room temperature, thus avoiding degradation of RNA.
The PARI method provides a reliable and efficient way to collect high purity and integrity of RNA from ginseng roots, which is critical for the overall success of downstream application. In our lab, the PARI method can successfully be up-scaled to extract hundreds of milligrams of total RNA from various plant tissues including ginseng roots, ligustrum fruits and notoginseng roots, which further illustrate the versatility of the method. This protocol is supposed to be universally applicable to isolation of not only RNA but also DNA from other plant tissues rich in polysaccharides, no matter what kind and how many of them, with reasonable amounts of proper polysaccharases.
tRNA sequencing is important for structural and functional study of tRNAs, because nearly half of tRNA genes remain silent in life entitiesand the genome data cannot describe the practical transcriptional profile of tRNAs in cells [
25]. So far, tRNAs in ginseng root have never been sequenced and little genomic information is available. In this study, ginseng tRNAs were first sequenced by NGS, because high-quality tRNA samples were prepared by using the PARI method. These tRNA-seq results initially confirmed the successful transcription of chloroplast tRNAs in ginseng root, and additionally provided some new tRNA sequence information besides chloroplast tRNAs, which could be helpful for ginseng tRNA studies such as structure and modification characterizations. These new ginseng tRNAs were found to be highly homologous with cytoplasmic or mitochondrial tRNAs of other plant species (e.g.,
Arabidopsis thaliana and
Ilex pubescens), which could be attribute to the high evolutionary conservation of tRNAs across plant species [
26].
It is a well-known fact that synonymous codons are not used with equal frequencies, which shape the tRNAomes in both eukaryotes and prokaryotes. A statistical analysis of the chloroplast genome in
Panax ginseng based on codon count (relative synonymous codon usage, RSCU) indicated that 31 codons in ginseng chloroplast genome with RSCU larger than 1.0 were preferred [
22]. However, we surprisingly found that many corresponding anticodons were obviously completely absent in the genome (KF431956.1) or minimally transcribed (
Figure 4C), suggesting a low correlation between the practical codon usage bias and the codon frequencies in genomes. On the other hand, there is evidence of a strong positive relationship between codon usage bias and tRNA abundance, which may partially explain the variability of tRNA transcripts [
23]. However, this correlation was not observed in
Panax ginseng, mainly because of a fact that certain tRNAs with heavy modifications could be significantly underrepresented during the NGS process.
In the present study, primary structure with post-transcriptional modifications of chloroplastictRNA
Gly(GCC) in ginseng root was characterized by using a LC-MS/MS method. Since the tRNA genehad been confirmed by NGS, correlations between modified sequence and CID product ions became conceptually straightforward so that we could map the modification onto the tRNAs with high confidence. Two contiguous modifications, m
5U-Ψ, were identified at positions 54 and 55, respectively, in the T-loop of tRNA
Gly(GCC) (
Figure 8A), which were consistent with the conserved location of TΨ in thecanonical cloverleaf structure [
24,
27]. An atypical D-stem of only 2bp was observed in tRNA
Gly(GCC), resulting in a larger D-loop that, as expected, contained a D at position 20.This result parallels published findings that D commonly occurs at position 16, 17, and 20,which flank the two highly conserved guanines at positions 18 and 19 (
Figure 8) [
24,
28]. Unexpectedly, an m
7G was also found in D-loop at a novel position 18. The m
7G is catalyzed by tRNA (guanine-
N7-)-methyltransferase commonly at position 46 guanine in a variable loop, which is critically dependent on the structure motif of a loop (the variable loop) inserting into two stems (the anticodon-stem and T-stem) [
29,
30]. Herein, due to the truncated D-stem, a similar motif was formed in the D-loop region combined with anticodon- and acceptor-stem that fits in the methyltransferase, which was supposed to facilitate the
N7-methylation of guanine at position 18.
Wobble position 34 in anticodon region and position 37 are two most frequently modified tRNA residues that are essential for translational efficiency and fidelity. However, these modifications are completely absent in ginseng tRNA
Gly(GCC) (
Figure 8). tRNAs lacking modifications have been found in many other species, especially in organelles and single cell organisms with small genomes. It has been reported that tRNA
Gly(GCC) without any base modifications in anticodon regions mainly participates in transcriptional regulation and transcription. Instead, the tRNA
Gly(UCC), which was also identified in ginseng chloroplasts (
Table S10), more often participates in translation by decoding all four glycine codons by “superwobbling” [
31].