Abstract
Many miRNAs and cancer-related mutations have been proposed as promising molecular markers of papillary thyroid carcinoma (PTC). However, there are limited data on the correlation between miRNA expression, BRAFV600E mutation, and PTC recurrence. Therefore, to evaluate the potential of BRAFV600E mutation and five selected miRNAs (-146b, -222, -21, -221, -181b) in predicting PTC recurrence, these molecular markers were analyzed in 400 formalin-fixed, paraffin-embedded PTC tissue specimens. The expression levels of miRNAs were measured using qRT-PCR. It was demonstrated that expression levels of all analyzed miRNAs are significantly higher in recurrent PTC than in non-recurrent PTC (p < 0.05). Moreover, higher expression levels of miR-146b, miR-222, miR-21, and miR-221 were associated with other clinicopathologic features of PTC, such as tumor size and lymph node metastases at initial surgery (p < 0.05). No significant differences in the frequency of BRAFV600E mutation in recurrent PTC and non-recurrent PTC were determined. Our results suggest that miRNA expression profile differs in PTC that is prone to recurrence when compared to PTC that does not reoccur after the initial surgery while BRAFV600E mutation frequency does not reflect the PTC recurrence status. However, the prognostic value of the analyzed miRNAs is rather limited in individual cases as the pattern of miRNA expression is highly overlapping between recurrent and non-recurrent PTC.
1. Introduction
Papillary thyroid carcinoma (PTC) is the most common type of thyroid cancer and it accounts for approximately 80% of thyroid cancer cases. For most PTC patients the prognosis is good, and the 5-year survival rate is about 90% [1]. Nevertheless, up to 10% of PTC patients after primary tumor removal and treatment suffer regional or distant metastatic recurrence [2]. Fine-needle aspiration biopsy (FNAB) is the most widely used method for presurgical diagnosis of PTC malignancy. However, this method has many limitations and is not very sensitive—for up to 20% FNAB diagnosis is unclear and surgery is needed to evaluate malignancy [3]. Therefore, the development of molecular markers of PTC recurrence such as miRNAs or BRAFV600E mutation has the potential to improve the clinical management of patients by assisting in risk stratification.
MicroRNAs (miRNAs) are single-stranded, non-coding small RNAs that are usually 21–25 nucleotides in length. MiRNAs bind to the 3‘UTR region of the target mRNAs and suppress their translation by degradation or inhibition. In this way, these molecules regulate the expression of many oncogenes or tumor suppressor genes [4]. Dysregulation of miRNA expression is related to several cancer types, including PTC [5]. Previous studies have reported that some miRNAs (such as miR-21, miR-146b, miR-9, miR-221, miR-222, miR181b, miR-155, miR-220, and others) are dysregulated in PTC compared to healthy patients [6,7,8,9,10]. There were also several studies that analyzed the association between the miRNA expression and PTC recurrence, but the results were not consistent as they included only small groups of PTC patients [8,11,12,13]. Thus, it is still unclear whether specific miRNAs could be used as PTC recurrence biomarkers.
BRAFV600E mutation is the transversion of thymine to adenine at nucleotide position 1799, therefore it leads to valine replacement by glutamic acid at codon 600. This mutation is the most common genetic lesion in PTC [14]. BRAFV600E mutation is found in 45%–80% of PTC cases [15,16,17]. Some studies revealed that this mutation is associated with clinicopathologic parameters of poor outcome in PTC [18,19,20,21], however, others have not shown the association [22]. It is supposed that BRAFV600E mutation might be related to miRNA expression in PTC but this association is still unclear. In previous studies, it was shown that the expression of some miRNAs is dysregulated in tissues with BRAFV600E mutation [8,13,23] while others have not determined any alterations [13,24].
Despite low mortality rates, recurrence in PTC occurs in up to 10% of patients [2]. Currently, PTC recurrence risk stratification is accomplished by using clinicopathologic factors. Molecular markers, such as cyclin D1, p27, p21, osteopontin, and E-cadherin, have been studied, however, none of them have been approved as PTC recurrence markers [25,26]. Thus, personalized treatment of patients is hard to achieve. Identification of new specific biomarkers might improve diagnostics of PTC, recurrence risk stratification, and would help to optimize PTC treatment for individual patients.
The aim of the current study was to identify specific miRNAs as biomarkers for predicting the recurrence of PTC. We determined the expression levels of five miRNAs (-146b, -222, -21, -221 and -181b) that were selected for this study based on previous reports [6,7,8,9,10] in a large number of paraffin-embedded PTC tissue samples. Additionally, we analyzed the association of miRNA expression levels with BRAFV600E mutation and clinicopathologic characteristics of PTC. For the first time, the expression level of miR-181b in recurrent and non-recurrent PTC was analyzed.
2. Materials and Methods
2.1. Human Tissue Samples
The formalin-fixed, paraffin-embedded (FFPE) PTC tissues (n = 400) were obtained from patients (aged from 18 to 83) who underwent total thyroidectomy at the Hospital of Lithuanian University of Health Sciences Kaunas Clinics between 2003 and 2017. After initial thyroid surgery all patients underwent postoperative radioactive iodine-131 (RAI) ablation and were treated with TSH-suppressive Levothyroxine doses. Patients were divided into two groups, depending on whether they had PTC recurrence after initial treatment or not. The non-recurrent group (NR-PTC, n = 313) comprised patients who had no signs of PTC recurrence after an initial treatment. They had undetectable levels of Tg and anti-Tg antibodies and a negative neck ultrasound after the initial surgery and RAI ablation at their latest follow-up. NR-PTC patients were selected randomly from the surgical database. Patients in the PTC recurrent group (Rc-PTC, n = 87) included those with a subsequent appearance of histologically confirmed PTC in the neck. The Rc-PTC group included only patients who had “true” PTC recurrence after the initial treatment. We defined disease as a “true” recurrence if a patient had local PTC recurrence at least 1 year after clinical and serological remission. Patients with persistent disease were not included in the study. All patients who had reoperative thyroid surgery due to recurrent PTC between 2003 and 2017 were included in the study.
The follow-up time for the patient was defined as the time period from the initial treatment to tumor recurrence or patient‘s death or, in the case of NR-PTC, to the most recent clinic visit. The follow-up time was censored in the disease-free survival analyses.
The study was approved by the Kaunas Regional Committee of Biomedical Research (Lithuania, approval No. BE-2-44; 2015-12-23). Written informed consent was obtained from each participant of the study after full explanation of the purpose and nature of all procedures used. This study was conducted in accordance with the Declaration of Helsinki.
2.2. RNA and DNA Extraction
Genomic DNA and RNA were extracted from 5–10 mm3 sections of FFPE PTC tissues using the QIAamp DNA FFPE Tissue Kit (Qiagen, Hilden, Germany) and miRNeasy FFPE Kit (Qiagen, Hilden, Germany), respectively, according to the manufacturer’s protocols. The PTC tissue samples were macrodissected from areas that contained over >90% of malignant tissue. RNA and DNA concentration and quality were examined by NanoDrop 2000 Spectrophotometer (ThermoFisher Scientific, Waltham, MA USA).
2.3. BRAFV600E Mutation Analysis
The sequence of BRAF gene encompassing codon 600 (fragment length 224 bp) was amplified by PCR. The composition of PCR mixture included Maxima Hot Start Master Mix (2×) (Thermo Fisher Scientific Baltics, Lithuania), forward (5′-TCA TAA TGC TTG CTC TGA TAG GA-3′) and reverse (5′-GGC CAA AAA TTT AAT CAG TGG A-3′) primers (10 μM), water and approximately 40 ng of purified genomic DNA. PCR reaction was run according to the following protocol: 95 °C for 5 min, 35 cycles: 95 °C 30 sec, 60 °C 1 min, 72 °C 1 min, and a final extension 72 °C 5 min. Amplification products were analyzed in ethidium bromide stained 2% agarose gel. Sequencing was performed by Applied Biosystems 3730xl DNA Analyzer (Baseclear, Leiden, The Netherlands).
2.4. qRT-PCR for miRNA Measurement
The expression levels of miRNAs were measured using quantitative reverse transcriptase-polymerase chain reaction (qRT-PCR) by TaqMan miRNA assays (Applied Biosystems, Foster City, Calif). Complementary DNA (cDNA) was synthesized from the purified total RNA using TaqMan miRNA Reverse Transcription Kit (Applied Biosystems, Foster City, Calif) following the manufacturer’s protocol. Real-time PCR was performed using TaqMan Universal PCR Master Mix (Applied Biosystems, Foster City, Calif) and Rotor-Gene 6000 PCR system (Corbett Research, Hilden, Germany). The fold expression changes were calculated by 2-ΔΔCt method. Relative expression was calculated using 2-ΔCt method where ΔCt is the difference in threshold cycles for target and reference genes. Firstly, purified RNA was reverse transcribed to cDNA using TaqMan miRNA primers according to the manufacturer’s instructions. As the starting material, 20 ng of tumor-derived RNA was used. Then the synthesized cDNA was amplified using TaqMan miRNA probes with the Rotor-Gene 6000 Real-time PCR System (Corbett Research, Germany). Three PCR reactions were run per sample according to the manufacturer’s instructions. We tested three endogenous controls for data normalization: RNU48, miRNA-16, and Let-7a. MiRNA Let-7a was chosen as an internal control for data normalization. As miRNA-181b expression level was very similar to miRNA Let-7a expression level and therefore had very small relative expression, in all presented figures miRNA-181b relative expression is plotted with a separate scale. Raw data of selected miRNA relative expression and clinical features is presented in Supplementary Table S1.
2.5. Statistical Analysis
Chi-square (χ2) test was used for analysis of data when variables were categorical. Continuous variables were tested for normality according to the Kolmogorov–Smirnov test. The results were presented as means ± standard deviation (SD) if the distributions were normal or median and range (min–max) if the distributions did not meet the criteria of normality.
Student’s t-tests (for normally distributed variables) or the Mann–Whitney tests (for non-normal distributions) were used to compare two groups of continuous variables. Multivariate binary logistic regression analysis was performed to estimate associations between different variables and PTC recurrence.
Patients with miRNA expression below the median values were assigned as having low expression levels, and patients with miRNA above or equal to median assigned as having high expression levels. The association between miRNA expression levels, clinicopathological features and disease-free survival (DFS) were assessed by the Kaplan–Meier method. A log-rank test was used to estimate the statistical differences in Kaplan–Meier curves. Univariate Cox proportional hazard regression model was performed to evaluate significant clinopathological and molecular parameters for DFS. Multivariate Cox proportional hazard regression model with enter method was conducted to evaluate independent prognostic predictors for DFS.
Data were analyzed using STATISTICA 13.2, Microsoft Excel (2013), and IBM SPSS Statistics for Mac (V20.0). A p value less than 0.05 was considered as statistically significant.
3. Results
3.1. Characteristics of the Study Population
In total, 400 samples of FFPE PTC tissues were investigated in the study. Demographic and clinicopathological characteristics of the study population are presented in Table 1. The mean age of patients was 50 years and ranged from 18 to 83 years. More than three-quarters of patients were females, giving a ratio (male:female) of 1∶6.5. However, males made up a significantly higher percentage in the Rc-PTC group (23%) than in the NR-PTC group (10.6%), p = 0.003. NR-PTC consisted of 313 patients with a median age at initial surgery of 53 (18–83) years and Rc-PTC consisted of 87 patients with a median age of 47 (18–75) years (p < 0.001). Patients <45 years had higher frequency of PTC recurrence than patients ≥45 years, p = 0.001. Rc-PTC patients had more advanced tumor stage than NR-PTC patients. Moreover, 22.8% of the patients had regional lymph node metastases at the time of initial surgery with higher frequency in the Rc-PTC group than in the NR-PTC (49.4% vs. 15.4%, p < 0.001).

Table 1.
Demographic and clinicopathological characteristics of the study population.
3.2. miRNA Expression Levels in PTC and Their Association With PTC Recurrence
FFPE PTC tissue samples collected for this study were stored for up to 15 years. The median age of FFPE PTC tissue samples were 9.92 (4.67–14.08) and 9.83 (5.5–14) years, respectively, for NR-PTC and Rc-PTC groups (p = 0.207). The yields of total RNA isolated from these samples varied from 0.36 to 13.65 μg with an average yield 2.75 μg per sample. The average A260/280 ratio for all samples was 1.7. Using the same input of RNA quantity, the Ct range for control miRNA let-7a varied from 16.8 to 23.9 cycles.
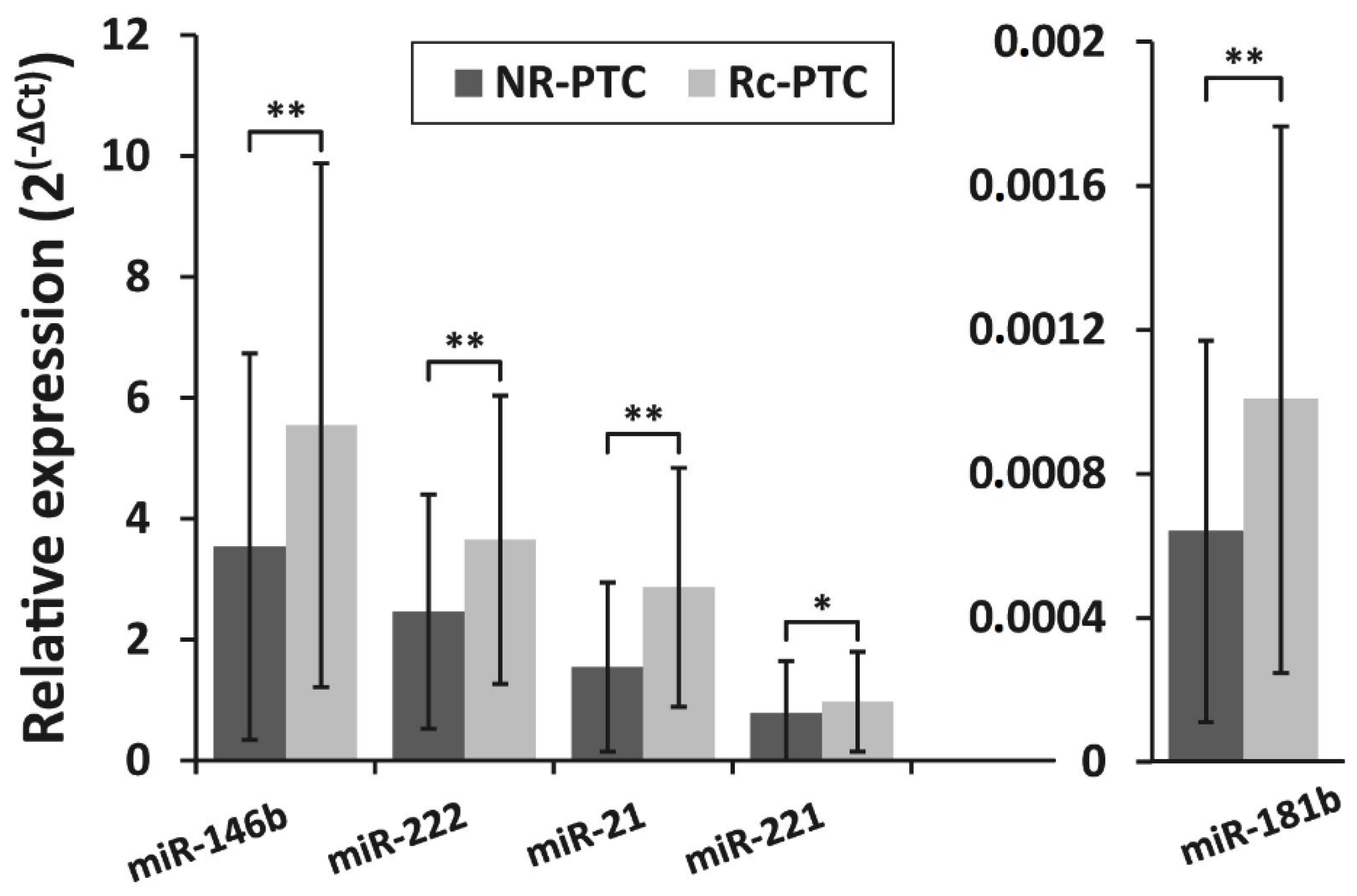
The expression levels of selected miRNAs (miR-146b, miR-222, miR-21, miR-221, miR-181b) were analyzed in Rc-PTC (n = 87) and NR-PTC (n = 313) groups. The expression levels of all miRNAs differed significantly between NR-PTC and Rc-PTC groups (p ˂ 0.05). MiR-146b, miR-222, miR-21, miR-221, and miR-181b were overexpressed 1.6-fold (p ˂ 0.01), 1.5-fold (p ˂ 0.01), 1.9-fold (p ˂ 0.01), 1.2-fold (p = 0.01), and 1.6-fold (p ˂ 0.01), respectively, in Rc-PTC compared to NR-PTC. Relative expression levels of all five miRNAs in NR-PTC and Rc-PTC groups are shown in Figure 1. As shown in this graph, there are substantial overlaps between miRNA profiles in NR-PTC and Rc-PTC groups. The overlap of miRNA expression profiles in Rc-PTC and NR-PTC groups can also be seen in Supplementary Figure S1. Although the samples with higher miRNA expression are more frequent in the Rc-PTC group than in the NR-PTC group this difference is too small to identify patients with high risk of PTC recurrence. ROC curve analysis also showed that all analyzed miRNAs are relatively weak biomarkers to stratify patients between Rc-PTC and NR-PTC groups (AUCs values for all miRNAs is less than 0.8) (Supplementary Figure S2).
Figure 1.
The relative expression levels of miR-146b, miR-222, miR-21, miR-221, and miR-181b in two patient groups—with recurrent papillary thyroid carcinoma (PTC) (Rc-PTC, n = 87) and without the recurrence of PTC (NR-PTC, n = 313). All data are presented as the mean ± SD. * p < 0.05, ** p < 0.01.
3.3. Detection of BRAFV600E Mutation in PTC Recurrence and Non-Recurrence Groups and Its Association With miRNA Expression
In total, 205 PTC samples were analyzed for BRAFV600E mutation by PCR and sequencing. The frequency of BRAFV600E mutation was analyzed in two patient groups: NR-PTC (n = 135) and Rc-PTC (n = 70). BRAFV600E mutation was detected in 62% of all PTC samples. Comparing NR-PTC and Rc-PTC groups, the frequency of BRAFV600E mutation was not significantly different (n = 83 (61.5%) and n = 44 (62.9%), p = 0.847, respectively).
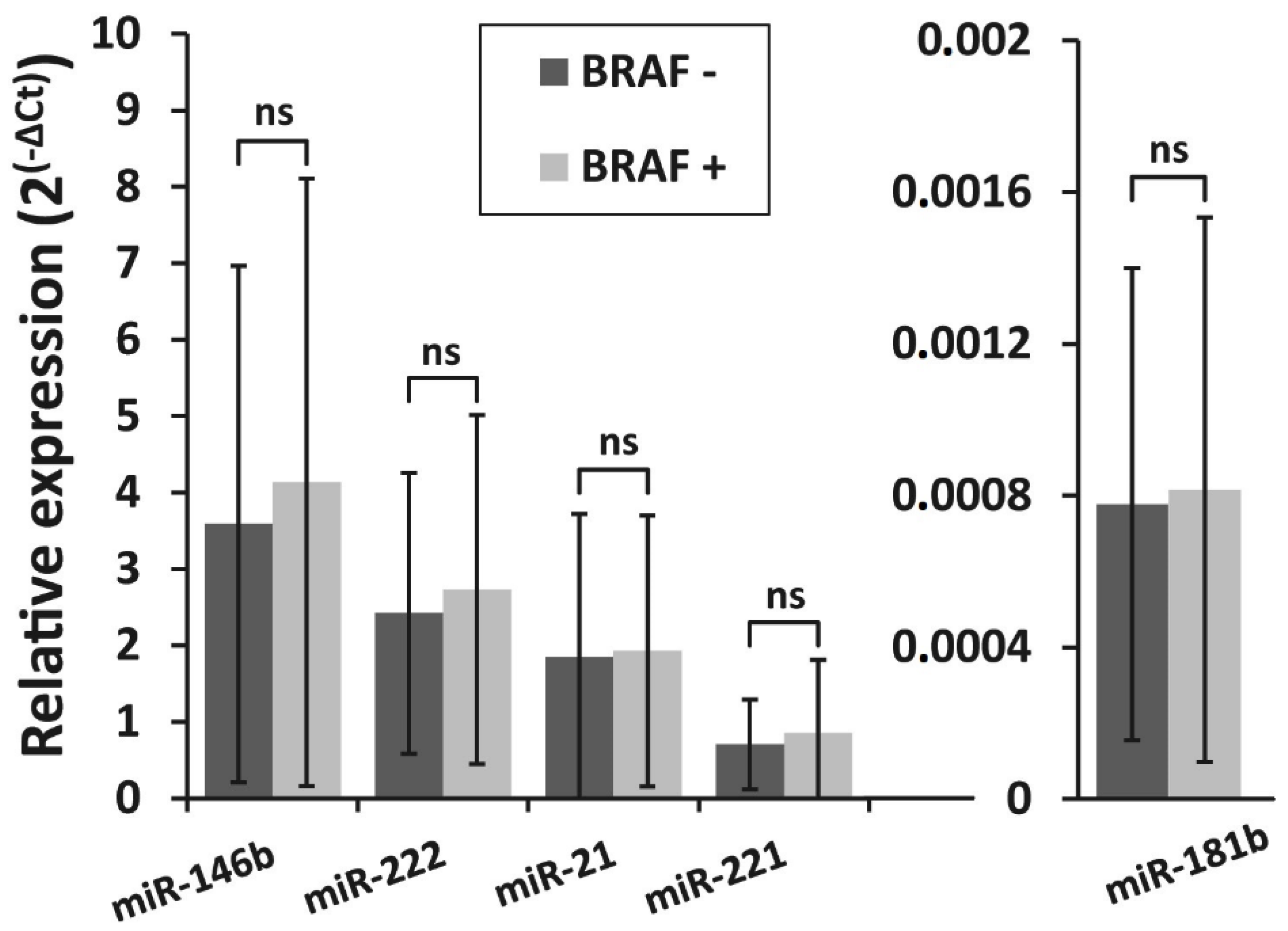
The correlation between BRAFV600E mutation and the expression levels of all five selected miRNAs was investigated in PTC samples. All samples were divided into two groups: BRAFV600E-positive (BRAF +; n = 127) and BRAFV600E-negative (BRAF −; n = 78). The expression levels of all miRNAs did not differ significantly between BRAFV600E-positive and BRAFV600E-negative PTC groups (p > 0.05) (Figure 2).
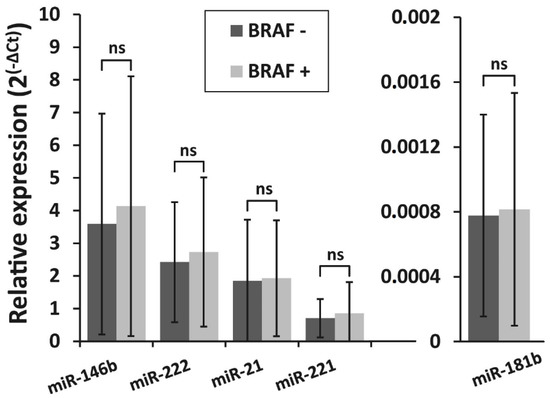
Figure 2.
Relative expression levels of miR-146b, miR-222, miR-21, miR-221, and miR-181b in two PTC groups—BRAFV600E-positive (BRAF +, n = 127) and BRAFV600E -negative (BRAF −, n = 78). All data are presented as the mean ± SD. ns—not significant.
3.4. Association of miRNAs Expression and BRAFV600E Mutation with Clinicopathologic Features of PTC
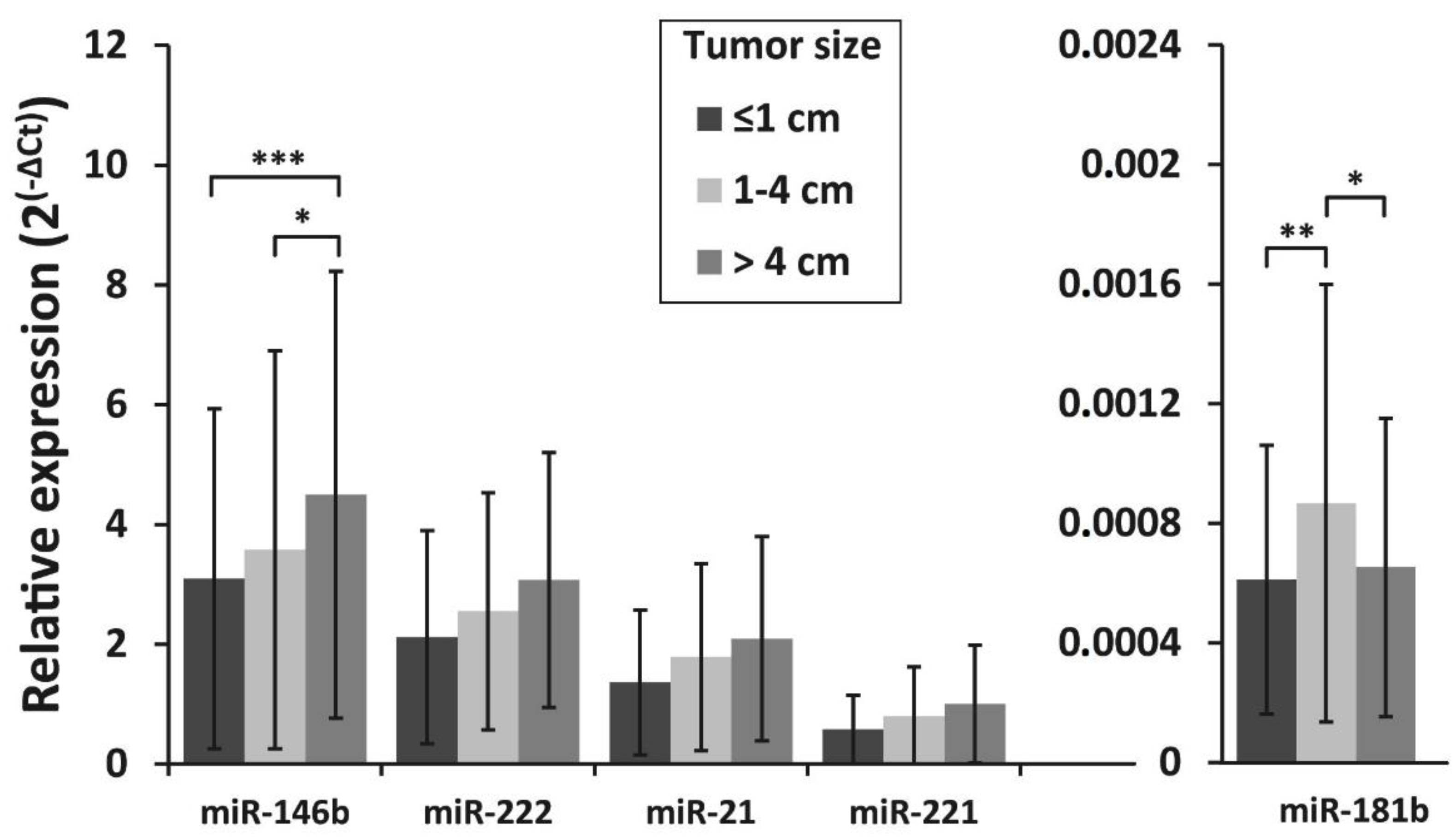
The association of miRNA expression with tumor size, lymph node metastasis status at initial surgery, and patient’s age was also investigated. To evaluate the association between tumor size and miRNA expression levels, all samples were divided into three groups according to the primary tumor size: tumors 1 cm or less in size (microcarcinomas) (n = 109), tumors >1–≤4 cm (n = 109) and tumors >4 cm (n =182). MiR-146b expression levels were similar between patients with PTC microcarcinoma and those who had tumors >1–≤4 cm (p = 0.405), however, a significantly higher expression of miR-146b was found in tumors >4cm than in tumors >1–≤4 cm or tumors ≤1cm (p = 0.012 and p < 0.001, respectively). For miR-181b, higher expression levels were found in tumors >1–≤4 cm than in smaller or larger tumors (p = 0.005 and p = 0.014, respectively) (Figure 3).
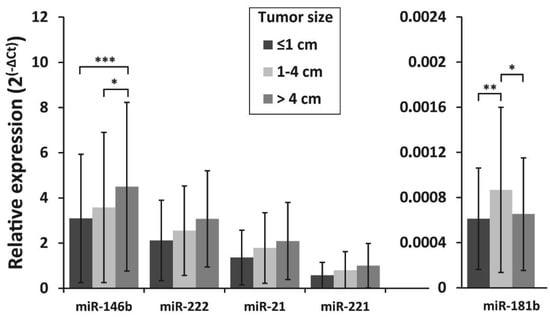
Figure 3.
Relative expression levels of miR-146b, miR-222, miR-21, miR-221, and miR-181b in differently sized PTC tumors. All data are presented as the mean ± SD. * p < 0.05, ** p < 0.01, and *** p < 0.001.
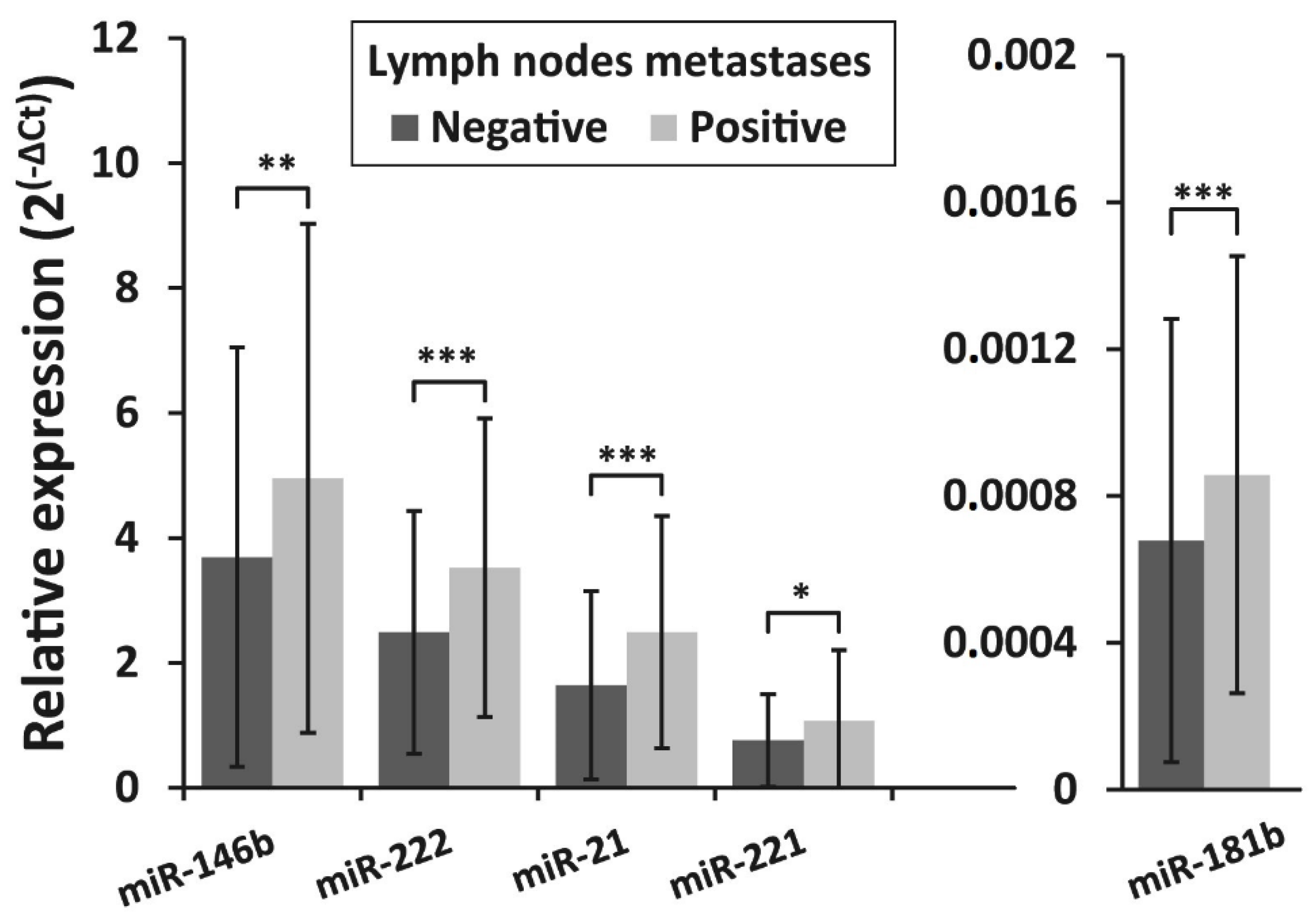
The expression levels of all investigated microRNAs were found to be associated with lymph node metastasis status at initial surgery. Expression levels of miR -146b, miR -222, miR -21, miR-221, and miR-181b were significantly higher in the lymph node metastases-positive group (n = 91) compared to the lymph node metastases-negative group (n = 309) (p = 0.004, p <0.001, p <0.001, p = 0.012, and p < 0.001, respectively) (Figure 4).
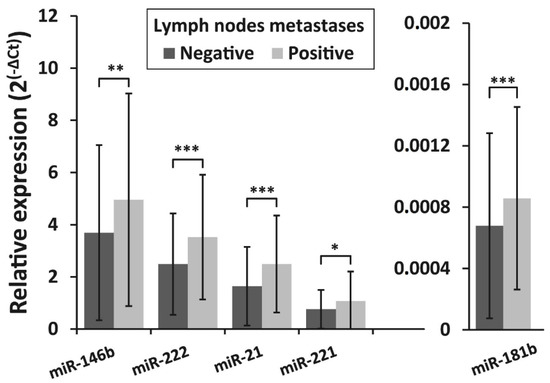
Figure 4.
Relative expression levels of miR-146b, miR-222, miR-21, miR-221, and miR-181b in PTC in patients with lymph node metastases at initial surgery and patients without metastases. All data are presented as the mean ± SD. * p < 0.05, ** p < 0.01, and *** p < 0.001.
Furthermore, patients were divided into two age groups: younger than 45 years at PTC diagnosis (n = 132) and 45 years and older (n = 268). Younger patients had similar expression levels of miR-146b, miR-222, miR-21, and miR-221 as compared to older patients (p = 0.11, p = 0.944, p = 0.485, p = 0.314, respectively). Only miR-181b levels were associated with patient’s age at PTC diagnosis. Higher expression of miR-181b was found in patients younger than 45 years compared to older patients (p = 0.007) (Figure 5).
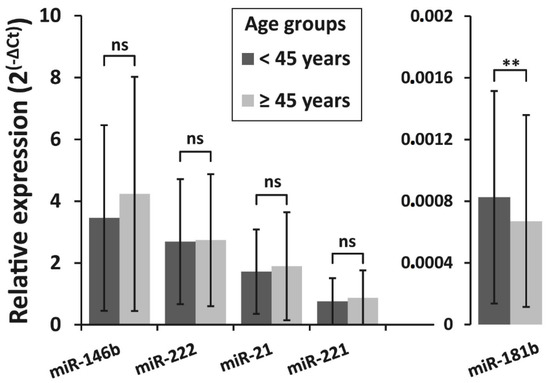
Figure 5.
Relative expression of miR-146b, miR-222, miR-21, miR-221, and miR-181b in different age groups of PTC patients. All data are presented as the mean ± SD. ** p < 0.01. ns—not significant.
The association of BRAFV600E mutation with patient’s age at diagnosis of PTC, primary tumor size and lymph node metastasis status were investigated (Figure 7). Patients of 45 years and older had the same frequency of BRAFV600E mutation as younger patients, whereas the significant difference was found in analysis limited within age groups. In 45 years and older patients group, a higher proportion of patients had BRAFV600E mutation (64.2% vs. 35.8%, p =0.002), whereas in the younger patients group the BRAFV600E positivity group did not significantly differ (58.5% vs. 41.5%, p = 0.122) (Figure 6A).
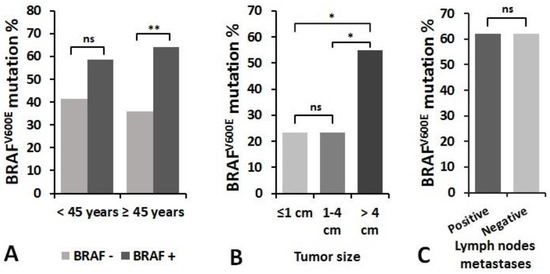
Figure 6.
The relationship between the frequency of BRAFV600E mutation and clinicopathological features of PTC: (A) patient’s age at diagnosis, (B) tumor size, and (C) the presence of regional lymph node metastases at initial surgery. * p < 0.05, ** p < 0.01. ns—not significant.
When assessing the relationship between BRAFV600E mutation and tumor size, samples were divided into three groups: tumor size ≤1 cm (n = 51), tumor size >1–≤4 cm (n = 56), and tumors >4 cm in size (n = 98). We found that BRAFV600E mutation occurred more frequently in a group of patients with primary tumor >4 cm in size compared to smaller tumors (p < 0.05) (Figure 6B). There was no significant relationship between BRAFV600E positivity and the presence of regional lymph nodes metastases at initial surgery. BRAFV600E mutation was detected in 62.0% of samples in the metastasis group (n = 31) and in 61.9% of samples in the group without metastases (n = 96) (p = 0.993) (Figure 6C).
The multivariate logistic regression analysis was performed to select the independent prognostic parameters for PTC recurrence. The analysis showed that positive lymph nodes and miR-21 were independently associated with elevated odds of PTC recurrence, OR 3.66 (95% CI 1.59–8.45) and OR 1.50 (95% CI 1.12–2.00), respectively (Table 2).

Table 2.
Multivariate analysis of associations of PTC recurrence with prognostic factors. Binary logistic regression model was used. Odds ratios adjusted by gender and age categories (<45 years vs. ≥45 years).
3.5. Influence of Clinicopathological Features and miRNA Expression on Disease-Free Survival (DFS)
The median follow-up time in NR-PTC group was 9 (3.08–14.08) years and median time to relapse in Rc-PTC group was 3.25 (1.17–11.5) years. To evaluate DFS association with miRNA expression levels patients were divided into high and low miRNAs expression groups. Univariate Cox regression hazards model analysis was performed that included clinicopathological features, BRAFV600E mutation status, and the expression levels (high/low) of each investigated miRNA (Table 3). Analysis revealed that females and patients with microcarcinomas have significantly lower hazard of recurrence, while younger patients, presence of metastatic lymph nodes, and high expression levels of all selected miRNAs were associated with increased hazard of shorter DFS. Variables that were chosen for inclusion in the multivariate analysis were those with significant univariate associations with DFS in this study, as well as those (i.e., BRAFV600E) found in other studies to be significant predictors for DFS. After multivariate Cox proportional regression hazard model analysis, only metastatic lymph nodes and high miR-21 expression level emerged as independent prognostic factors associated with shorter DFS (HR 1.94 (95% CI 1.12–3.36) and HR 5.84 (95% CI 2.79–12.28), respectively).

Table 3.
Univariate and multivariate analysis of disease-free survival (DFS) in patients with PTC. Cox proportional regression hazard model was used.
Kaplan–Meier plots for independent prognostic factors are reported in Figure 7 and for the remaining factors are presented in Supplementary Figure S3. The Log-rank test demonstrated significant differences in survival curves both for lymph node metastases (p < 0.001) and miRNA-21 expression (p < 0.001). Five-year disease-free survival was 92.1% in patients with negative lymph nodes and 65.7% in patients with metastatic lymph nodes, and 95.3% in patients with low miR-21 expression levels and 75.4% in patients with high miR-21 expression.
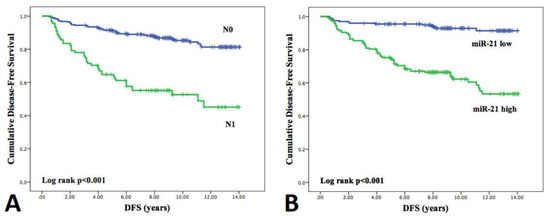
Figure 7.
Kaplan–Meier curves estimating disease-free survival according to (A) the presence of lymph node metastasis and (B) high/low expression levels of miR-21. DFS curves were compared using the Log rank test.
4. Discussion
In this study, we investigated five selected miRNAs (-146b, -222, -21, -221, and -181b) as biomarkers for predicting PTC recurrence and analyzed the associations of miRNAs expression with BRAFV600E mutation and clinicopathologic characteristics of PTC. Five miRNAs (-146b, -222, -21, -221, and -181b) were selected for this study based on previous reports [6,7,8,9,10]. Our investigation is the largest study as yet. We investigated 400 PTC samples, meanwhile, other studies involved fewer than 221 samples [8,11,12,24,28,29,30]. Most miRNAs are known to be very stable in tissue specimens making them a suitable marker to be analyzed in archived FFPE samples [31]. However, not much is known about the stability of widely used non-miRNA endogenous controls. It is likely that they are more prone to degradation because of their length and different functions in the cell. In the current study, we have tested three previously published endogenous controls for data normalization: RNU48 [8,10,11,24,28], miRNA-16 [2], and Let-7a [9,32]. When comparing the assay performance with these different controls, we have noticed that the commonly used endogenous control RNU48 degrades significantly faster in archived FFPE samples than any of our analyzed target miRNAs (our unpublished data). In contrast, Let-7a had similar stability as the target miRNAs. Therefore, we chose to use Let-7a as an endogenous control which was also used in several similar studies [9,32].
In our study, we first compared the expression profile of selected miRNA in PTC in general with miRNA expression in healthy thyroid tissue and demonstrated that four of five analyzed miRNAs (miR-146b, miR-222, miR-21, and miR-221) were significantly overexpressed in PTC. These results were in agreement with other reports [7,9,10,33,34]. Next, we divided PTC samples according to the PTC recurrence status and determined that expression levels of miR-146b, miR-222, miR-21, miR-221, and miR-181b were significantly higher in PTC recurrence group as compared to the non-recurrence group (p < 0.05). In line with these data, previous studies in smaller-sized groups of PTC patients have revealed enhanced expression of certain miRNAs in recurrent PTC [8,11,12,28,29,30]. In previous studies, the difference of miR-181b expression levels between Rc-PTC and NR-PTC groups has not been investigated. For the first time, we analyzed the association of miR-181b expression with PTC recurrence status and demonstrated different expression levels of miR-181b in NR-PTC and Rc-PTC groups (p < 0.05). In the present study, we found that metastatic lymph nodes at the time of diagnosis and expression of miR-21 are associated with PTC recurrence and were independent prognostic factors of DFS.
Although the levels of all studied miRNAs were significantly higher in the Rc-PTC group, an overlap of miRNA expression profiles in NR-PTC and Rc-PTC groups with a small difference between the peaks was observed. This indicates that the analyzed miRNA does not represent reliable prognostic markers that would allow identifying patients with a high risk of PTC recurrence. This observation was also confirmed by the ROC curve analysis. The AUCs for miR-146b, miR-222, miR-21, miR-221, and miR-181b were 0.685, 0.676, 0.762, 0.598, and 0.685, respectively.
BRAFV600E mutation is the most common genetic lesion in PTC [14]. In this study, the frequency of BRAFV600E mutation was investigated in 221 PTC samples. BRAFV600E mutation was detected in 61.5% of all PTC samples. In other studies, BRAFV600E mutation was identified in 45–80% of PTC cases [13,15,16,17,24]. Comparing NR-PTC and Rc-PTC groups, the frequency of BRAFV600E mutation was not significantly different (p = 0.641). There are limited data on the association of BRAFV600E mutation and PTC recurrence. We found only one study where the frequency of BRAFV600E mutation was analyzed in NR-PTC and Rc-PTC groups. In line with our study, it was reported that the frequency of BRAFV600E mutation was not different between NR-PTC and Rc-PTC groups (p > 0.05) [8].
The associations of BRAFV600E mutation with clinicopathological features were also investigated. Study results revealed that tumor size >4 cm significantly associated with BRAFV600E mutation (p < 0.05). However, lymph node metastasis was not significantly different between patients with or without BRAFV600E mutation (p = 0.993). In previous reports, there are limited and controversial data on the association of BRAFV600E mutation with patient age, tumor size, and metastasis status. In line with our data, some studies demonstrated statistically significant correlations between BRAFV600E mutation and patient age [19,35,36,37,38,39], between BRAFV600E mutation and tumor size [24,35,39,40,41,42], and between BRAFV600E mutation and regional lymph nodes metastases [35,39,40,41,42]. However, other studies did not find a statistically significant correlation between BRAFV600E mutation and patient age [13,43,44,45,46], tumor size [13,22,37,47], and metastasis status [13,36,43]. We also did not find any significant association between BRAFV600E mutation and miRNA expression, which is in line with previous reports [24]. BRAFV600E usefulness as a prognostic marker in PTC remains controversial. Many previous studies reported a higher risk of PTC recurrence, metastases, and poor disease outcome in patients with BRAFV600E mutation, although there are plenty of studies that could not confirm any mutation-related associations [48,49,50]. These authors found that other factors, such as the presence of lymph node metastases, extrathyroidal invasion, or histologic type of PTC, provide higher accuracy in predicting disease outcome than BRAF mutation status alone [49,50,51].
Although miRNA expression profiles in PTC have been analyzed for more than a decade, little is known regarding the genes and pathways downstream that are regulated by miR-146b, miR-222, miR-221, miR-21, and miR-181b as only a few computationally predicted targets are validated in laboratory experiments. MiR-21 is one of the most extensively studied miRNAs in cancer and it was one of the first miRNAs described as a potential tumor biomarker. It is highly conserved across many species and its overexpression has been detected in many cancer types suggesting miR-21 role in gene regulation which disruption may contribute to cancer development. Studies with various cancer cell lines also showed that eliminating miR-21 expression inhibits the development of cancer-associated phenotypes. Moreover, IL-6, STAT3, AP-1, and TGFβ1 were described as molecules involved in the upregulation of miR-21 expression [52]. In the present study, we found that high levels miR-21 both with metastatic lymph nodes at the time of diagnosis are associated with PTC recurrence and were independent prognostic factors of DFS.
Another well-studied miRNA is miR-146b. Some of the detected downstream targets of miR-146b are SMAD4 and IRAK1 whose downregulation leads to an increased proliferation and migration activity and inhibits cell cycle arrest in PTC cell line [53,54]. MiR-221 and miR-222 are two highly homologous miRNAs that are also overexpressed in many types of cancer including PTC. The determined targets of these miRNAs are p27, p57, and PTEN which all have important roles in cell cycle regulation [55]. Among all dysregulated miRNAs in cancer, miR-181b has been found to be a critical regulatory miRNA linking inflammation and cancer by suppressing cylindromatosis expression. The expression of miR-181b is regulated by STAT3 and HMGA1 [56]. The functional significance of miR-146b, miR-222, miR-221, miR-21, and miR-181b in various tumors suggests that they exhibit great potential as predictive and prognostic biomarkers. Extensive efforts are underway to identify mRNA targets and the affected regulatory networks, which may be the key to providing a better understanding of PTC development and miRNA role in carcinogenesis in general.
5. Conclusions
Summarizing, the analysis of five miRNA (-146b, -222, -221, -21, and -181b) expression levels in a large number of well-characterized PTC samples (n = 400) revealed that these miRNAs are not only overexpressed in PTC, but they are also overexpressed in PTC with more aggressive features such as recurrence, bigger tumor size, lymph node metastasis, and shorter disease-free survival. However, the prognostic value of these miRNAs is rather limited in individual cases as the distribution of miRNA expression overlaps between patients with high and low risk of PTC recurrence. Additionally, more standardization is needed on tissues and different endogenous controls used in miRNA research as these factors have a significant impact on the final results. Different approaches such as analysis of circulating miRNAs, search for another more reliable molecular markers, or single-cell analysis might represent the future trends for improving the recognition of patients with recurrent PTC.
Supplementary Materials
The following are available online at https://www.mdpi.com/2218-273X/10/4/625/s1, Figure S1: Frequency distribution of (A) miR-146b, (B) miR-222, (C) miR-21, (D) miR-221, (E) miR-181b expression in NR-PTC and Rc-PTC groups, Figure S2: ROC curve analysis of miR-146b, miR-222, miR-21, miR-221, miR-181b for the stratification of patients who experienced PTC recurrence (Rc-PTC group) from patients who did not (NR-PTC group). The AUCs for miR-146b, miR-222, miR-21, miR-221 and miR-181b were 0.685, 0.676, 0.762, 0.598 and 0.685, respectively, Figure S3: Kaplan-Meier analysis of disease-free survival (DFS) according to the clinicopathological features, BRAF mutation, high/low expression levels of miRNA’s in patient’s cancer tissue: (A) Gender, (B) Age, (C) Tumor size, (D) BRAF mutation, (E) miR-146b, (F) miR-222, (G) miR-221, (H) miR-181b. DFS curves were compared using Log rank test, Table S1: Raw data of selected miRNA relative expression and clinical features.
Author Contributions
Conceptualization, A.Z. and B.Z.; methodology, V.S. (Vaida Simanaviciene); validation, D.P., A.D. and R.V.; formal analysis, D.P.; investigation, V.S. (Vaida Simanaviciene), D.P., D.D. and E.L.; resources, A.Z., B.Z. and V.S. (Valdas Sarauskas); data curation, D.P., D.D. and E.L.; writing—original draft preparation, V.S. (Vaida Simanaviciene); writing—review and editing, D.P., A.Z. and B.Z.; visualization, D.P. and A.D.; supervision, A.Z. and B.Z.; project administration, B.Z.; funding acquisition, B.Z. All authors have read and agreed to the published version of the manuscript.
Funding
This work was supported by the Lithuanian Research Council (Grant number SEN-14/2015).
Acknowledgments
We gratefully acknowledge study funding source.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Jemal, A.; Siegel, R.; Ward, E.; Hao, Y.; Xu, J.; Thun, M.J. Cancer Statistics. Ca Cancer J. Clin. 2009, 59, 225–249. [Google Scholar] [CrossRef]
- Han, P.A.; Weng, C.-H.; Khawaja, H.T.; Nagarajan, N.; Schneider, E.B.; Umbricht, C.B.; Witwer, K.W.; Zeiger, M.A. MicroRNA Expression and Association with Clinicopathologic Features in Papillary Thyroid Cancer: A Systematic Review. Thyroid 2015, 25, 1322–1329. [Google Scholar] [CrossRef]
- Eszlinger, M.; Paschke, R. Molecular fine-needle aspiration biopsy diagnosis of thyroid nodules by tumor specific mutations and gene expression patterns. Mol. Cell. Endocrinol. 2010, 322, 29–37. [Google Scholar] [CrossRef] [PubMed]
- Lin, S.; Gregory, R.I. MicroRNA biogenesis pathways in cancer. Nat. Rev. Cancer 2015, 15, 321–333. [Google Scholar] [CrossRef] [PubMed]
- Wilson, C. Cancer: MicroRNA expression provides clues about the aggressiveness of papillary thyroid carcinoma. Nat. Rev. Endocrinol. 2010, 6, 416. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.-T.; Kitabayashi, N.; Zhou, X.K.; Fahey, T.J.; Scognamiglio, T. MicroRNA analysis as a potential diagnostic tool for papillary thyroid carcinoma. Mod. Pathol. 2008, 21, 1139–1146. [Google Scholar] [CrossRef] [PubMed]
- He, H.; Jazdzewski, K.; Li, W.; Liyanarachchi, S.; Nagy, R.; Volinia, S.; Calin, G.A.; Liu, C.-G.; Franssila, K.; Suster, S.; et al. The role of microRNA genes in papillary thyroid carcinoma. Proc. Natl. Acad. Sci. USA 2005, 102, 19075–19080. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.C.; Zhao, J.T.; Clifton-Bligh, R.; Gill, A.; Gundara, J.S.; Ip, J.C.; Glover, A.; Sywak, M.S.; Delbridge, L.W.; Robinson, B.G.; et al. MicroRNA-222 and MicroRNA-146b are tissue and circulating biomarkers of recurrent papillary thyroid cancer. Cancer 2013, 119, 4358–4365. [Google Scholar] [CrossRef]
- Nikiforova, M.N.; Tseng, G.C.; Steward, D.; Diorio, D.; Nikiforov, Y.E. MicroRNA expression profiling of thyroid tumors: Biological significance and diagnostic utility. J. Clin. Endocrinol. Metab. 2008, 93, 1600–1608. [Google Scholar] [CrossRef]
- Sheu, S.-Y.; Grabellus, F.; Schwertheim, S.; Worm, K.; Broecker-Preuss, M.; Schmid, U.-P.D.M.K.W. Differential miRNA expression profiles in variants of papillary thyroid carcinoma and encapsulated follicular thyroid tumours. Br. J. Cancer 2009, 102, 376–382. [Google Scholar] [CrossRef]
- Dai, L.; Wang, Y.; Chen, L.; Zheng, J.; Li, J.; Wu, X. MiR-221, a potential prognostic biomarker for recurrence in papillary thyroid cancer. World J. Surg. Oncol. 2017, 15, 11. [Google Scholar] [CrossRef] [PubMed]
- Yip, L.; Kelly, L.; Shuai, Y.; Armstrong, M.J.; Nikiforov, Y.E.; Carty, S.E.; Nikiforova, M.N.; Linwah, Y.; Yongli, S. MicroRNA signature distinguishes the degree of aggressiveness of papillary thyroid carcinoma. Ann. Surg. Oncol. 2011, 18, 2035–2041. [Google Scholar] [CrossRef] [PubMed]
- Chou, C.-K.; Chen, R.-F.; Chou, F.-F.; Chang, H.-W.; Chen, Y.-J.; Lee, Y.-F.; Yang, K.D.; Cheng, J.-T.; Huang, H.-Y.; Liu, R.-T. miR-146b is Highly Expressed in Adult Papillary Thyroid Carcinomas with High Risk Features Including Extrathyroidal Invasion and the BRAFV600EMutation. Thyroid 2010, 20, 489–494. [Google Scholar] [CrossRef] [PubMed]
- Tang, K.-T.; Lee, C.-H. BRAF Mutation in Papillary Thyroid Carcinoma: Pathogenic Role and Clinical Implications. J. Chin. Med Assoc. 2010, 73, 113–128. [Google Scholar] [CrossRef]
- Tufano, R.P.; Teixeira, G.; Bishop, J.; Carson, K.A.; Xing, M. BRAF Mutation in Papillary Thyroid Cancer and Its Value in Tailoring Initial Treatment. Medicine 2012, 91, 274–286. [Google Scholar] [CrossRef]
- Lassalle, S.; Hofman, V.; Ilie, M.; Butori, C.; Bozec, A.; Santini, J.; Vielh, P.; Hofman, P. Clinical impact of the detection of BRAF mutations in thyroid pathology: Potential usefulness as diagnostic, prognostic and theragnostic applications. Curr. Med. Chem. 2010, 17, 1839–1850. [Google Scholar] [CrossRef]
- Lee, J.-H.; Lee, E.-S.; Kim, Y.-S. Clinicopathologic significance ofBRAF V600E mutation in papillary carcinomas of the thyroid. Cancer 2007, 110, 38–46. [Google Scholar] [CrossRef]
- Xing, M. BRAF Mutation in Papillary Thyroid Cancer: Pathogenic Role, Molecular Bases, and Clinical Implications. Endocr. Rev. 2007, 28, 742–762. [Google Scholar] [CrossRef]
- Kim, S.-J.; Lee, K.E.; Myong, J.P.; Park, J.-H.; Jeon, Y.K.; Min, H.S.; Park, S.Y.; Jung, K.C.; Koo, D.H.; Youn, Y.-K. BRAFV600E Mutation is Associated with Tumor Aggressiveness in Papillary Thyroid Cancer. World J. Surg. 2011, 36, 310–317. [Google Scholar] [CrossRef]
- Chakraborty, A.; Narkar, A.; Mukhopadhyaya, R.; Kane, S.; D’Cruz, A.; Rajan, M.G.R. BRAF V600E Mutation in Papillary Thyroid Carcinoma: Significant Association with Node Metastases and Extra Thyroidal Invasion. Endocr. Pathol. 2011, 23, 83–93. [Google Scholar] [CrossRef]
- Nakayama, H.; Yoshida, A.; Nakamura, Y.; Hayashi, H.; Miyagi, Y.; Wada, N.; Rino, Y.; Masuda, M.; Imada, T. Clinical significance of BRAF (V600E) mutation and Ki-67 labeling index in papillary thyroid carcinomas. Anticancer. Res. 2007, 27, 3645–3649. [Google Scholar] [PubMed]
- Pelttari, H.; Schalin-Jantti, C.; Arola, J.; Löyttyniemi, E.; Knuutila, S.; Valimaki, M.J. BRAF V600E mutation does not predict recurrence after long-term follow-up in TNM stage I or II papillary thyroid carcinoma patients. APMIS 2011, 120, 380–386. [Google Scholar] [CrossRef] [PubMed]
- Cahill, S.; Smyth, P.; Denning, K.; Flavin, R.; Li, J.; Potratz, A.; Guenther, S.M.; Henfrey, R.; O’Leary, J.J.; Sheils, O. Effect of BRAFV600E mutation on transcription and post-transcriptional regulation in a papillary thyroid carcinoma model. Mol. Cancer 2007, 6, 21. [Google Scholar] [CrossRef] [PubMed]
- Sheu, S.-Y.; Schwertheim, S.; Handke, S.; Worm, K.; Grabellus, F.; Schmid, U.-P.D.M.K.W. Lack of Correlation between BRAF V600E Mutational Status and the Expression Profile of a Distinct Set of miRNAs in Papillary Thyroid Carcinoma. Horm. Metab. Res. 2009, 41, 482–487. [Google Scholar] [CrossRef]
- Cheng, S.; Serra, S.; Mercado, M.; Ezzat, S.; Asa, S.L. A High-Throughput Proteomic Approach Provides Distinct Signatures for Thyroid Cancer Behavior. Clin. Cancer Res. 2011, 17, 2385–2394. [Google Scholar] [CrossRef]
- Khoo, M.L.C.; Freeman, J.L.; Witterick, I.J.; Irish, J.C.; Rotstein, L.E.; Gullane, P.J.; Asa, S.L. Underexpression of p27/Kip in thyroid papillary microcarcinomas with gross metastatic disease. Arch. Otolaryngol. Head Neck Surg. 2002, 128, 253–257. [Google Scholar] [CrossRef]
- Vickery, A.L.; Carcangiu, M.L.; Johannessen, J.V.; Sobrinho-Simoes, M. Papillary carcinoma. Semin. Diagn. Pathol. 1985, 2, 2004 57. [Google Scholar]
- Sondermann, A.; Andreghetto, F.M.; Moulatlet, A.C.B.; Victor, E.D.S.; De Castro, M.G.; Nunes, F.D.; Brandão, L.G.; Severino, P. MiR-9 and miR-21 as prognostic biomarkers for recurrence in papillary thyroid cancer. Clin. Exp. Metastasis 2015, 32, 521–530. [Google Scholar] [CrossRef]
- Yang, Z.; Yuan, Z.; Fan, Y.; Deng, X.; Zheng, Q. Integrated analyses of microRNA and mRNA expression profiles in aggressive papillary thyroid carcinoma. Mol. Med. Rep. 2013, 8, 1353–1358. [Google Scholar] [CrossRef]
- Acibucu, F.; Dökmetaş, H.; Tutar, Y.; Elagoz, S.; Kilicli, F.; Acıbucu, F.; Kılıclı, F. Correlations between the Expression Levels of Micro-RNA146b, 221, 222 and p27Kip1 protein mRNA and the Clinicopathologic Parameters in Papillary Thyroid Cancers. Exp. Clin. Endocrinol. Diabetes 2014, 122, 137–143. [Google Scholar] [CrossRef]
- Meng, W.; McElroy, J.P.; Volinia, S.; Palatini, J.; Warner, S.; Ayers, L.W.; Palanichamy, K.; Chakravarti, A.; Lautenschlaeger, T. Comparison of MicroRNA Deep Sequencing of Matched Formalin-Fixed Paraffin-Embedded and Fresh Frozen Cancer Tissues. PLoS ONE 2013, 8, e64393. [Google Scholar] [CrossRef]
- Agretti, P.; Ferrarini, E.; Rago, T.; Candelieri, A.; De Marco, G.; Dimida, A.; Niccolai, F.; Molinaro, A.; Di Coscio, G.; Pinchera, A.; et al. MicroRNA expression profile helps to distinguish benign nodules from papillary thyroid carcinomas starting from cells of fine-needle aspiration. Eur. J. Endocrinol. 2012, 167, 393–400. [Google Scholar] [CrossRef] [PubMed]
- Mančíková, V.; Castelblanco, E.; Piñeiro-Yáñez, E.; Perales-Patón, J.; A De Cubas, A.; Inglada, L.; Matias-Guiu, X.; Capel, I.; Bella, M.; Lerma, E.; et al. MicroRNA deep-sequencing reveals master regulators of follicular and papillary thyroid tumors. Mod. Pathol. 2015, 28, 748–757. [Google Scholar] [CrossRef] [PubMed]
- Tetzlaff, M.T.; Liu, A.; Xu, X.; Master, S.R.; Baldwin, N.A.; Tobias, J.W.; Livolsi, V.A.; Baloch, Z. Differential Expression of miRNAs in Papillary Thyroid Carcinoma Compared to Multinodular Goiter Using Formalin Fixed Paraffin Embedded Tissues. Endocr. Pathol. 2007, 18, 163–173. [Google Scholar] [CrossRef]
- Sykorova, V.; Dvořáková, Š.; Ryska, A.; Vcelak, J.; Vaclavikova, E.; Laco, J.; Kodetova, D.; Kodet, R.; Cibula, A.; Dušková, J.; et al. BRAFV600E mutation in the pathogenesis of a large series of papillary thyroid carcinoma in Czech Republic. J. Endocrinol. Investig. 2010, 33, 318–324. [Google Scholar] [CrossRef]
- Kebebew, E.; Weng, J.; Bauer, J.; Ranvier, G.; Clark, O.H.; Duh, Q.-Y.; Shibru, D.; Bastian, B.C.; Griffin, A. The Prevalence and Prognostic Value of BRAF Mutation in Thyroid Cancer. Ann. Surg. 2007, 246, 466–471. [Google Scholar] [CrossRef]
- Zhou, Y.-L.; Liu, C.; Dai, X.-X.; Zhang, X.-H.; Wang, O.-C. Overexpression of miR-221 is associated with aggressive clinicopathologic characteristics and the BRAF mutation in papillary thyroid carcinomas. Med Oncol. 2012, 29, 3360–3366. [Google Scholar] [CrossRef]
- Rosenbaum, E.; Hosler, G.; Zahurak, M.; Cohen, Y.; Sidransky, D.; Westra, W.H. Mutational activation of BRAF is not a major event in sporadic childhood papillary thyroid carcinoma. Mod. Pathol. 2004, 18, 898–902. [Google Scholar] [CrossRef]
- Pelizzo, M.R.; Boschin, I.M.; Barollo, S.; Pennelli, G.; Toniato, A.; Zambonin, L.; Vianello, F.; Piotto, A.; Ide, E.C.; Pagetta, C.; et al. BRAF analysis by fine needle aspiration biopsy of thyroid nodules improves preoperative identification of papillary thyroid carcinoma and represents a prognostic factor. A mono-institutional experience. Clin. Chem. Lab. Med. 2011, 49, 325–329. [Google Scholar] [CrossRef]
- Basolo, F.; Torregrossa, L.; Giannini, R.; Miccoli, M.; Lupi, C.; Sensi, E.; Berti, P.; Elisei, R.; Vitti, P.; Baggiani, A.; et al. Correlation between the BRAF V600E Mutation and Tumor Invasiveness in Papillary Thyroid Carcinomas Smaller than 20 Millimeters: Analysis of 1060 Cases. J. Clin. Endocrinol. Metab. 2010, 95, 4197–4205. [Google Scholar] [CrossRef]
- Frasca, F.; Nucera, C.; Pellegriti, G.; Gangemi, P.; Attard, M.; Stella, M.; Loda, M.; Vella, V.; Giordano, C.; Trimarchi, F.; et al. BRAF(V600E) mutation and the biology of papillary thyroid cancer. Endocrine-Related Cancer 2008, 15, 191–205. [Google Scholar] [CrossRef] [PubMed]
- Guan, H.; Ji, M.; Bao, R.; Yu, H.; Wang, Y.; Hou, P.; Zhang, Y.; Shan, Z.; Teng, W.; Xing, M. Association of High Iodine Intake with the T1799ABRAFMutation in Papillary Thyroid Cancer. J. Clin. Endocrinol. Metab. 2009, 94, 1612–1617. [Google Scholar] [CrossRef]
- Ito, Y.; Yoshida, H.; Maruo, R.; Morita, S.; Takano, T.; Hirokawa, M.; Yabuta, T.; Fukushima, M.; Inoue, H.; Tomoda, C.; et al. BRAF mutation in papillary thyroid carcinoma in a Japanese population: Its lack of correlation with high-risk clinicopathological features and disease-free survival of patients. Endocr. J. 2008, 56, 89–97. [Google Scholar] [CrossRef] [PubMed]
- Goutas, N.; Vlachodimitropoulos, D.; Bouka, M.; Lazaris, A.C.; Nasioulas, G.; Gazouli, M. BRAF and K-RAS mutation in a Greek papillary and medullary thyroid carcinoma cohort. Anticancer. Res. 2008, 28, 305–308. [Google Scholar] [PubMed]
- Oler, G.; Cerutti, J. High prevalence ofBRAFmutation in a Brazilian cohort of patients with sporadic papillary thyroid carcinomas. Cancer 2009, 115, 972–980. [Google Scholar] [CrossRef] [PubMed]
- Fang, W.; Ye, L.; Zhang, X.; Shen, L.; Han, R.; Wei, Q.; Fei, X.; Chen, X.; Wang, W.; Wang, S.; et al. BRAF mutation correlates with recurrent papillary thyroid carcinoma in Chinese patients. Curr. Oncol. 2014, 21, 740–e747. [Google Scholar] [CrossRef]
- Barbaro, D.; Incensati, R.M.; Materazzi, G.; Boni, G.; Grosso, M.; Panicucci, E.; Lapi, P.; Pasquini, C.; Miccoli, P. The BRAF V600E mutation in papillary thyroid cancer with positive or suspected pre-surgical cytological finding is not associated with advanced stages or worse prognosis. Endocrine 2013, 45, 462–468. [Google Scholar] [CrossRef]
- Kim, T.H.; Park, Y.J.; Lim, J.A.; Ahn, H.Y.; Lee, E.-K.; Lee, E.-K.; Kim, K.W.; Hahn, S.K.; Youn, Y.-K.; Kim, K.H.; et al. The association of the BRAFV600E mutation with prognostic factors and poor clinical outcome in papillary thyroid cancer. Cancer 2011, 118, 1764–1773. [Google Scholar] [CrossRef]
- Czarniecka, A.; Kowal, M.; Rusinek, D.; Krajewska, J.; Jarząb, M.; Stobiecka, E.; Chmielik, E.; Zembala-Nozyńska, E.; Poltorak, S.; Sacher, A.; et al. The Risk of Relapse in Papillary Thyroid Cancer (PTC) in the Context of BRAFV600E Mutation Status and Other Prognostic Factors. PLoS ONE 2015, 10, e0132821. [Google Scholar] [CrossRef]
- Nair, C.G.; Babu, M.; Biswas, L.; Jacob, P.; Ramachandran, R.; Revathy, A.K.; Nair, K. Lack of Association of B-type Raf Kinase V600E Mutation with High-risk Tumor Features and Adverse Outcome in Conventional and Follicular Variants of Papillary Thyroid Carcinoma. Indian J. Endocrinol. Metab. 2017, 21, 329–333. [Google Scholar] [CrossRef]
- Yarchoan, M.; Livolsi, V.A.; Brose, M.S. BRAF Mutation and Thyroid Cancer Recurrence. J. Clin. Oncol. 2015, 33, 7–8. [Google Scholar] [CrossRef] [PubMed]
- Selcuklu, S.D.; Donoghue, M.T.; Spillane, C. miR-21 as a key regulator of oncogenic processes. Biochem. Soc. Trans. 2009, 37, 918–925. [Google Scholar] [CrossRef] [PubMed]
- Geraldo, M.V.; Yamashita, A.S.; Kimura, E.T. MicroRNA miR-146b-5p regulates signal transduction of TGF-β by repressing SMAD4 in thyroid cancer. Oncogene 2011, 31, 1910–1922. [Google Scholar] [CrossRef] [PubMed]
- Chou, C.-K.; Chi, S.-Y.; Huang, C.-H.; Chou, F.-F.; Huang, C.-C.; Liu, R.-T.; Kang, H.-Y. IRAK1, a Target of miR-146b, Reduces Cell Aggressiveness of Human Papillary Thyroid Carcinoma. J. Clin. Endocrinol. Metab. 2016, 101, 4357–4366. [Google Scholar] [CrossRef] [PubMed]
- Song, J.; Ouyang, Y.; Che, J.; Li, X.; Zhao, Y.; Yang, K.; Zhao, X.; Chen, Y.; Fan, C.; Yuan, W.-E. Potential Value of miR-221/222 as Diagnostic, Prognostic, and Therapeutic Biomarkers for Diseases. Front. Immunol. 2017, 8, 29. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Shi, W.; Wu, C.; Ju, J.; Jiang, J. miR-181b as a key regulator of the oncogenic process and its clinical implications in cancer (Review). Biomed. Rep. 2013, 2, 7–11. [Google Scholar] [CrossRef] [PubMed]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).