Targeting TLR4 with ApTOLL Improves Heart Function in Response to Coronary Ischemia Reperfusion in Pigs Undergoing Acute Myocardial Infarction
Abstract
:1. Introduction
2. Materials and Methods
2.1. Reagents and Equipment
2.2. Animal Model of Coronary Ischemia/Reperfusion
2.3. Echocardiography
2.4. Evans Blue/TTC Staining
2.5. Confocal Microscopy
2.6. Determination of Cardiac TroponinI
2.7. Histology and Immunohistochemistry
2.8. Statistical Analysis
3. Results
3.1. ApTOLL Induces Cardiac Protection in Response to IR
3.2. ApTOLL Reduces Left Ventricle Necrosis and Fibrosis by Day 7 after IR
3.3. APTOLL Inhibits the Expression of MMP-9 by Day 7 after IR
3.4. ApTOLL Reduces the Expression of Pro-Inflammatory Cytokines 7 Days After Reperfusion
4. Discussion
5. Conclusions
6. Patents
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Benjamin, E.J.; Muntner, P.; Alonso, A.; Bittencourt, M.S.; Callaway, C.W.; Carson, A.P.; Chamberlain, A.M.; Chang, A.R.; Cheng, S.; Das, S.R.; et al. Heart Disease and Stroke Statistics—2019 Update: A Report From the American Heart Association. Circulation 2019, 139. [Google Scholar] [CrossRef]
- Prabhu, S.D.; Frangogiannis, N.G. The Biological Basis for Cardiac Repair After Myocardial Infarction: From Inflammation to Fibrosis. Circ. Res. 2016, 119, 91–112. [Google Scholar] [CrossRef]
- Nahrendorf, M.; Pittet, M.J.; Swirski, F.K. Monocytes: Protagonists of Infarct Inflammation and Repair After Myocardial Infarction. Circulation 2010, 121, 2437–2445. [Google Scholar] [CrossRef]
- Frangogiannis, N.G. Regulation of the Inflammatory Response in Cardiac Repair. Circ Res 2012, 110, 159–173. [Google Scholar] [CrossRef]
- Bhattacharya, D.; Yusuf, N. Expression of Toll-Like Receptors on Breast Tumors: Taking a Toll on Tumor Microenvironment. Int. J. Breast Cancer 2012, 2012, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Sun, N.; Wang, H.; Wang, L. Protective effects of ghrelin against oxidative stress, inducible nitric oxide synthase and inflammation in a mouse model of myocardial ischemia/reperfusion injury via the HMGB1 and TLR4/NF-?B pathway. Mol. Med. Rep. 2016, 14, 2764–2770. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Frangogiannis, N.G. Targeting the inflammatory response in healing myocardial infarcts. Curr. Med. Chem. 2006, 13, 1877–1893. [Google Scholar] [CrossRef] [PubMed]
- Avlas, O.; Srara, S.; Shainberg, A.; Aravot, D.; Hochhauser, E. Silencing cardiomyocyte TLR4 reduces injury following hypoxia. Exp. Cell Res. 2016, 348, 115–122. [Google Scholar] [CrossRef] [PubMed]
- Fujiwara, M.; Matoba, T.; Koga, J.-I.; Okahara, A.; Funamoto, D.; Nakano, K.; Tsutsui, H.; Egashira, K. Nanoparticle incorporating Toll-like receptor 4 inhibitor attenuates myocardial ischaemia–reperfusion injury by inhibiting monocyte-mediated inflammation in mice. Cardiovasc. Res. 2019, 115, 1244–1255. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ogawa, K.; Hirooka, Y.; Kishi, T.; Ide, T.; Sunagawa, K. Partially Silencing Brain Toll-Like Receptor 4 Prevents in Part Left Ventricular Remodeling with Sympathoinhibition in Rats with Myocardial Infarction-Induced Heart Failure. PLoS ONE 2013, 8, e69053. [Google Scholar] [CrossRef]
- Leonard, M.; Zhang, D.; Zhang, X. Small Non-Coding RNAs and Aptamers in Diagnostics and Therapeutics. Adv. Struct. Saf. Stud. 2015, 1296, 225–233. [Google Scholar] [CrossRef]
- Jung, M.; Dodsworth, M.; Thum, T. Inflammatory cells and their non-coding RNAs as targets for treating myocardial infarction. Basic Res. Cardiol. 2018, 114, 4. [Google Scholar] [CrossRef] [Green Version]
- Fernández, G.; Moraga, A.; Cuartero, M.; García-Culebras, A.; Peña-Martínez, C.; Pradillo, J.M.; Hernández-Jiménez, M.; Sacristán, S.; Ayuso, M.I.; Gonzalo-Gobernado, R.; et al. TLR4-Binding DNA Aptamers Show a Protective Effect against Acute Stroke in Animal Models. Mol. Ther. 2018, 26, 2047–2059. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ramos, E.; Piñeiro, D.; Soto, M.; Abanades, D.R.; Martín, M.E.; Salinas, M.; Gonzalez, V.M.; Alvarez, M.S. A DNA aptamer population specifically detects Leishmania infantum H2A antigen. Lab. Investig. 2007, 87, 409–416. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- White, R.R.; Sullenger, B.A.; Rusconi, C.P. Developing aptamers into therapeutics. J. Clin. Investig. 2000, 106, 929–934. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bouchard, P.; Hutabarat, R.; Thompson, K. Discovery and Development of Therapeutic Aptamers. Annu. Rev. Pharmacol. Toxicol. 2010, 50, 237–257. [Google Scholar] [CrossRef] [PubMed]
- Rossello, X.; Rodríguez-Sinovas, A.; Vilahur, G.; Crisóstomo, V.; Jorge, I.; Zaragoza, C.; Zamorano, J.L.; Bermejo, J.; Ordoñez, A.; Boscá, L.; et al. CIBER-CLAP (CIBERCV Cardioprotection Large Animal Platform): A multicenter preclinical network for testing reproducibility in cardiovascular interventions. Sci. Rep. 2019, 9, 1–11. [Google Scholar] [CrossRef] [Green Version]
- Bøtker, H.E.; Hausenloy, D.; Andreadou, I.; Antonucci, S.; Boengler, K.; Davidson, S.M.; Deshwal, S.; Devaux, Y.; Di Lisa, F.; Di Sante, M.; et al. Practical guidelines for rigor and reproducibility in preclinical and clinical studies on cardioprotection. Basic Res. Cardiol. 2018, 113, 39. [Google Scholar] [CrossRef] [Green Version]
- Tarin, C.; Lavin, B.; Gomez, M.; Saura, M.; Diez-Juan, A.; Zaragoza, C. The extracellular matrix metalloproteinase inducer EMMPRIN is a target of nitric oxide in myocardial ischemia/reperfusion. Free. Radic. Boil. Med. 2011, 51, 387–395. [Google Scholar] [CrossRef]
- Cuadrado, I.; Castejon, B.; Martin, A.M.; Saura, M.; Reventún, P.; Zamorano, J.L.; Zaragoza, C. Nitric Oxide Induces Cardiac Protection by Preventing Extracellular Matrix Degradation through the Complex Caveolin-3/EMMPRIN in Cardiac Myocytes. PLoS ONE 2016, 11, e0162912. [Google Scholar] [CrossRef]
- Sadony, V.; Körber, M.; Albes, G.; Podtschaske, V.; Etgen, T.; Trösken, T.; Ravens, U.; Scheulen, M.E. Cardiac troponin I plasma levels for diagnosis and quantitation of perioperative myocardial damage in patients undergoing coronary artery bypass surgery. Eur. J. Cardio-Thoracic Surg. 1998, 13, 57–65. [Google Scholar] [CrossRef] [Green Version]
- Phatharajaree, W.; Phrommintikul, A.; Chattipakorn, N. Matrix metalloproteinases and myocardial infarction. Can. J. Cardiol. 2007, 23, 727–733. [Google Scholar] [CrossRef] [Green Version]
- Jeremias, A. The utility of troponin measurement to detect myocardial infarction: Review of the current findings. Vasc. Heal. Risk Manag. 2010, 691. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Satoh, M.; Shimoda, Y.; Maesawa, C.; Akatsu, T.; Ishikawa, Y.; Minami, Y.; Hiramori, K.; Nakamura, M. Activated toll-like receptor 4 in monocytes is associated with heart failure after acute myocardial infarction. Int. J. Cardiol. 2006, 109, 226–234. [Google Scholar] [CrossRef]
- Ma, H.; Du, J.; Feng, X.; Zhang, Y.; Wang, H.; Ding, S.; Huang, A.; Ma, J. Rosiglitazone alleviates myocardial apoptosis in rats with acute myocardial infarction via inhibiting TLR4/NF-κB signaling pathway. Exp. Ther. Med. 2020, 19, 2491–2496. [Google Scholar] [CrossRef] [Green Version]
- Oyama, J.-I.; Blais, C., Jr.; Liu, X.; Pu, M.; Kobzik, L.; Kelly, R.A.; Bourcier, T. Reduced Myocardial Ischemia-Reperfusion Injury in Toll-Like Receptor 4-Deficient Mice. Circulation 2004, 109, 784–789. [Google Scholar] [CrossRef] [Green Version]
- Li, H.; Xu, H.; Liu, S. Toll-like receptors 4 induces expression of matrix metalloproteinase-9 in human aortic smooth muscle cells. Mol. Boil. Rep. 2010, 38, 1419–1423. [Google Scholar] [CrossRef]
- Mann, D.L. The emerging role of innate immunity in the heart and vascular system: For whom the cell tolls. Circ. Res. 2011, 108, 1133–1145. [Google Scholar] [CrossRef]
- Ong, S.-B.; Hernández-Reséndiz, S.; Crespo-Avilan, G.E.; Mukhametshina, R.T.; Kwek, X.-Y.; Cabrera-Fuentes, H.A.; Hausenloy, D.J. Inflammation following acute myocardial infarction: Multiple players, dynamic roles, and novel therapeutic opportunities. Pharmacol. Ther. 2018, 186, 73–87. [Google Scholar] [CrossRef]
- Tapp, L.D.; Shantsila, E.; Wrigley, B.J.; Montoro-García, S.; Lip, G.Y.H. TLR4 expression on monocyte subsets in myocardial infarction. J. Intern. Med. 2012, 273, 294–305. [Google Scholar] [CrossRef] [Green Version]
- Yokoyama, T.; Komori, A.; Nakamura, M.; Takii, Y.; Kamihira, T.; Shimoda, S.; Mori, T.; Fujiwara, S.; Koyabu, M.; Taniguchi, K.; et al. Human intrahepatic biliary epithelial cells function in innate immunity by producing IL-6 and IL-8 via the TLR4-NF-kappaB and -MAPK signaling pathways. Liver Int. 2006, 26, 467–476. [Google Scholar] [CrossRef] [PubMed]
- Cha, J.; Wang, Z.; Ao, L.; Zou, N.; Dinarello, C.A.; Banerjee, A.; Fullerton, D.A.; Meng, X. Cytokines Link Toll-Like Receptor 4 Signaling to Cardiac Dysfunction After Global Myocardial Ischemia. Ann. Thorac. Surg. 2008, 85, 1678–1685. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Montecucco, F.; Braunersreuther, V.; Lenglet, S.; Delattre, B.M.; Pelli, G.; Buatois, V.; Guilhot, F.; Galan, K.; Vuilleumier, N.; Ferlin, W.G.; et al. CC chemokine CCL5 plays a central role impacting infarct size and post-infarction heart failure in mice. Eur. Hear. J. 2011, 33, 1964–1974. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Neri, M.; Fineschi, V.; Paolo, M.; Pomara, C.; Riezzo, I.; Turillazzi, E.; Cerretani, D. Cardiac Oxidative Stress and Inflammatory Cytokines Response after Myocardial Infarction. Curr. Vasc. Pharmacol. 2015, 13, 26–36. [Google Scholar] [CrossRef] [PubMed]
- García-Culebras, A.; Durán-Laforet, V.; Peña-Martínez, C.; Moraga, A.; Ballesteros, I.; Cuartero, M.I.; De La Parra, J.; Palma-Tortosa, S.; Hidalgo, A.; Corbí, A.L.; et al. Role of TLR4 (Toll-Like Receptor 4) in N1/N2 Neutrophil Programming After Stroke. Stroke 2019, 50, 2922–2932. [Google Scholar] [CrossRef] [Green Version]
- Zhu, J.; Huang, J.; Dai, D.; Wang, X.; Gao, J.; Han, W.; Zhang, R. Recombinant human interleukin-1 receptor antagonist treatment protects rats from myocardial ischemia–reperfusion injury. Biomed. Pharmacother. 2019, 111, 1–5. [Google Scholar] [CrossRef]
- Kaplanski, G. Interleukin-18: Biological properties and role in disease pathogenesis. Immunol. Rev. 2017, 281, 138–153. [Google Scholar] [CrossRef] [Green Version]
- Coverstone, E.D.; Bach, R.G.; Chen, L.; Bierut, L.J.; Li, A.Y.; Lenzini, P.A.; O’Neill, H.C.; Spertus, J.A.; Sucharov, C.C.; Stitzel, J.A.; et al. A novel genetic marker of decreased inflammation and improved survival after acute myocardial infarction. Basic Res. Cardiol. 2018, 113, 38. [Google Scholar] [CrossRef]
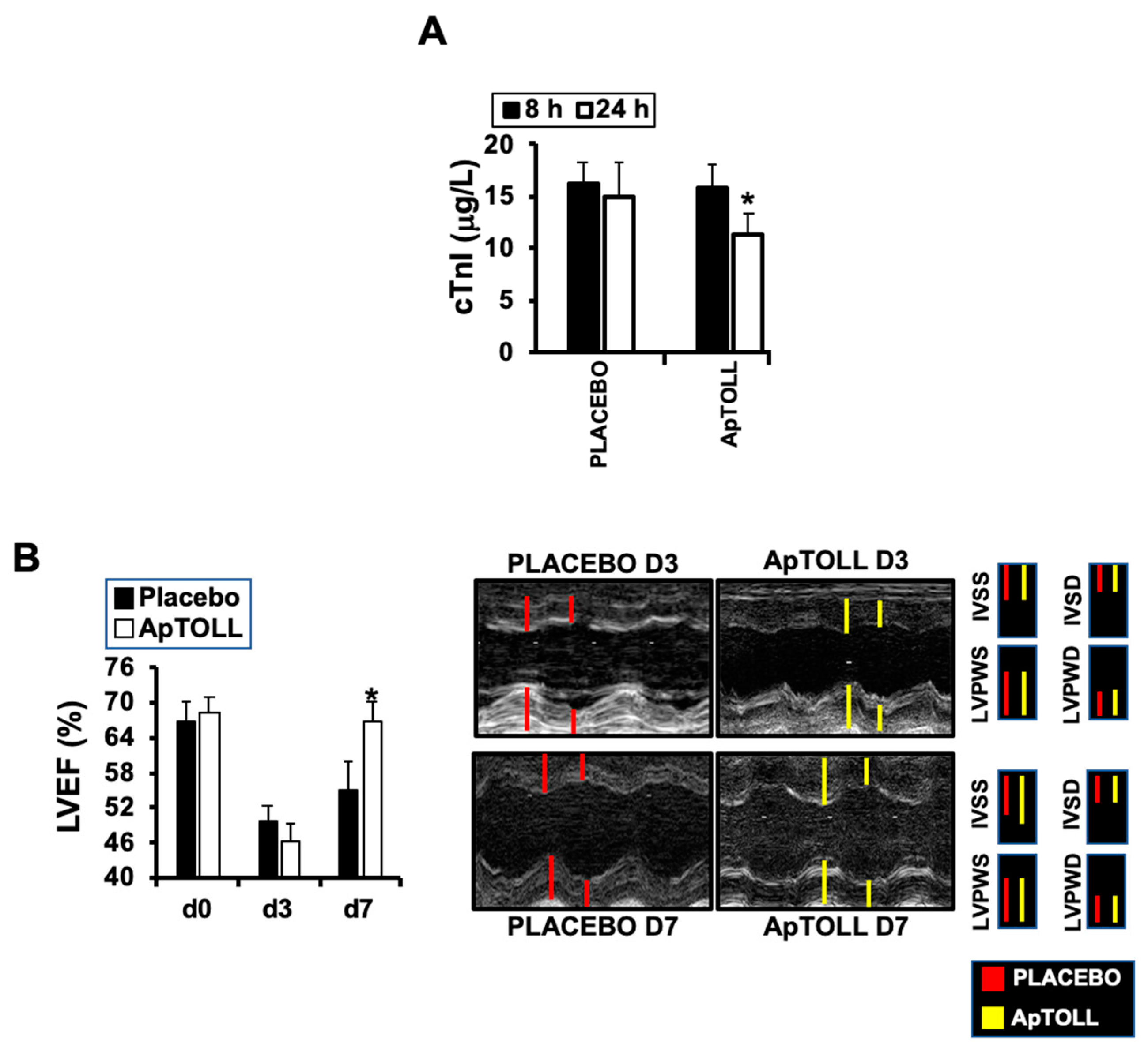




| PARAMETER | PLACEBO D3 | APTOLL D3 | PLACEBO D7 | APTOLL D7 | PARAMETER |
|---|---|---|---|---|---|
| IVSD (mm) | 11.68 ± 0.806 | 10.857 ± 0.61 | 10.124 ± 1.357 | 11.353 ± 1.276 | IVSD (mm) |
| LVIDD (mm) | 37.96 ± 4.219 | 39.511 ± 4.319 | 38.488 ± 8.667 | 38.067 ± 6.052 | LVIDD (mm) |
| LVPWD (mm) | 13.430 ± 2.394 | 9.954 ± 1.746 | 10.475 ± 0.67 | 11.093 ± 2.796 | LVPWD (mm) |
| IVSS (mm) | 15.77 ± 2.264 | 13.562 ± 0.932 | 13.153 ± 2.806 | 14.177 ± 1.678 | IVSS (mm) |
| LVIDS (mm) | 26.28 ± 4.36 | 29.126 ± 3.493 | 27.316 ± 5.860 | 24.705 ± 4.129 | LVIDS (mm) |
| LVPWS (mm) | 15.77 ± 3.383 | 11.024 ± 1.339 | 10.926 ± 1.704 | 11.90 ± 1.722 | LVPWS |
| EF (%) | 49.07 ± 5.91 | 46.31 ± 2.60 | 55.14 ± 4.947 | 66.85 ± 3.196 * | EF (%) |
| FS (%) | 21.25 ± 3.77 | 20.272 ± 1.443 | 28.55 ± 2.530 | 34.75 ± 2.492 * | FS (%) |
| HR (bpm) | 99 ± 31.494 | 111.1O1 ± 27.31 | 103.444 ± 31.269 | 91.875 ± 11.849 | HR (bpm) |
| CO (mL/sec) | 3614 ± 2068.651 | 3940.33 ± 1217.115 | 3801.111 ± 1.826 | 3918.875 ± 1356 | CO (mL/sec) |
| CYTOKINE | PLACEBO | APTOLL |
|---|---|---|
| CCL5 | 1.108 ± 0.141 | 0.27 ± 0.070 |
| CXCL1 | 0.429 ± 0.024 | 0.105 ± 0.023 |
| CXCL10 | 1.542 ± 0.237 | 0.37 ± 0.07 |
| G-CSF | 0.372 ± 0.088 | 0.0732 ± 0.014 |
| GM-CSF | 0.261 ± 0.057 | 0.031 ± 0.009 |
| ICAM-1 | 0.154 ± 0.030 | 0.045 ± 0.009 |
| INF-g | 0.092 ± 0.019 | 0 |
| IL1-a | 0.255 ± 0.043 | 0.108 ± 0.018 |
| ILI-b | 0.616 ± 0.146 | 0.047 ± 0.146 |
| IL-IRA | 0.193 ± 0.023 | 0.421 ± 0.123 |
| IL2 | 0.493 ± 0.074 | 0.141 ± 0.036 |
| IL5 | 0.197 ± 0.020 | 0.014 ± 0.004 |
| IL6 | 0.309 ± 0.040 | 0.165 ± 0.043 |
| IL8 | 0.317 ± 0.042 | 0.183 ± 0.047 |
| IL12 | 0.392 ± 0.054 | 0.213 ± 0.054 |
| IL16 | 0.306 ± 0.031 | 0.16 ± 0.046 |
| IL17-A | 0.42 ± 0.042 | 0.109 ± 0.032 |
| IL18 | 0.311 ± 0.077 | 0.504 ± 0.116 |
| IL21 | 0.466 ± 0.074 | 0.159 ± 0.029 |
| IL27 | 0.348 ± 0.037 | 0 |
| MIF | 1.359 ± 0.200 | 0.527 ± 0.089 |
| SERPIN-E1 | 0.876 ± 0.095 | 0.454 ± 0.076 |
| TNF-a | 0.52 ± 0.040 | 0.152 ± 0.045 |
| TREM-1 | 0.4192 ± 0.039 | 0.105 ± 0.031 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ramirez-Carracedo, R.; Tesoro, L.; Hernandez, I.; Diez-Mata, J.; Piñeiro, D.; Hernandez-Jimenez, M.; Zamorano, J.L.; Zaragoza, C. Targeting TLR4 with ApTOLL Improves Heart Function in Response to Coronary Ischemia Reperfusion in Pigs Undergoing Acute Myocardial Infarction. Biomolecules 2020, 10, 1167. https://doi.org/10.3390/biom10081167
Ramirez-Carracedo R, Tesoro L, Hernandez I, Diez-Mata J, Piñeiro D, Hernandez-Jimenez M, Zamorano JL, Zaragoza C. Targeting TLR4 with ApTOLL Improves Heart Function in Response to Coronary Ischemia Reperfusion in Pigs Undergoing Acute Myocardial Infarction. Biomolecules. 2020; 10(8):1167. https://doi.org/10.3390/biom10081167
Chicago/Turabian StyleRamirez-Carracedo, Rafael, Laura Tesoro, Ignacio Hernandez, Javier Diez-Mata, David Piñeiro, Macarena Hernandez-Jimenez, Jose Luis Zamorano, and Carlos Zaragoza. 2020. "Targeting TLR4 with ApTOLL Improves Heart Function in Response to Coronary Ischemia Reperfusion in Pigs Undergoing Acute Myocardial Infarction" Biomolecules 10, no. 8: 1167. https://doi.org/10.3390/biom10081167








