Altered L-Arginine Metabolic Pathways in Gastric Cancer: Potential Therapeutic Targets and Biomarkers
Abstract
:1. Introduction
2. Materials and Methods
2.1. Patients and Controls
2.1.1. Metabolomic Analysis
2.1.2. Transcriptomic Analysis
2.2. Analytical Methods
2.2.1. Metabolomic Analysis
Chemicals and Reagents
Sample Extraction
LC-QTOF-MS Analysis
2.2.2. Transcriptomic Analysis
2.2.3. Immunoassays
2.3. Statistical Analysis
3. Results
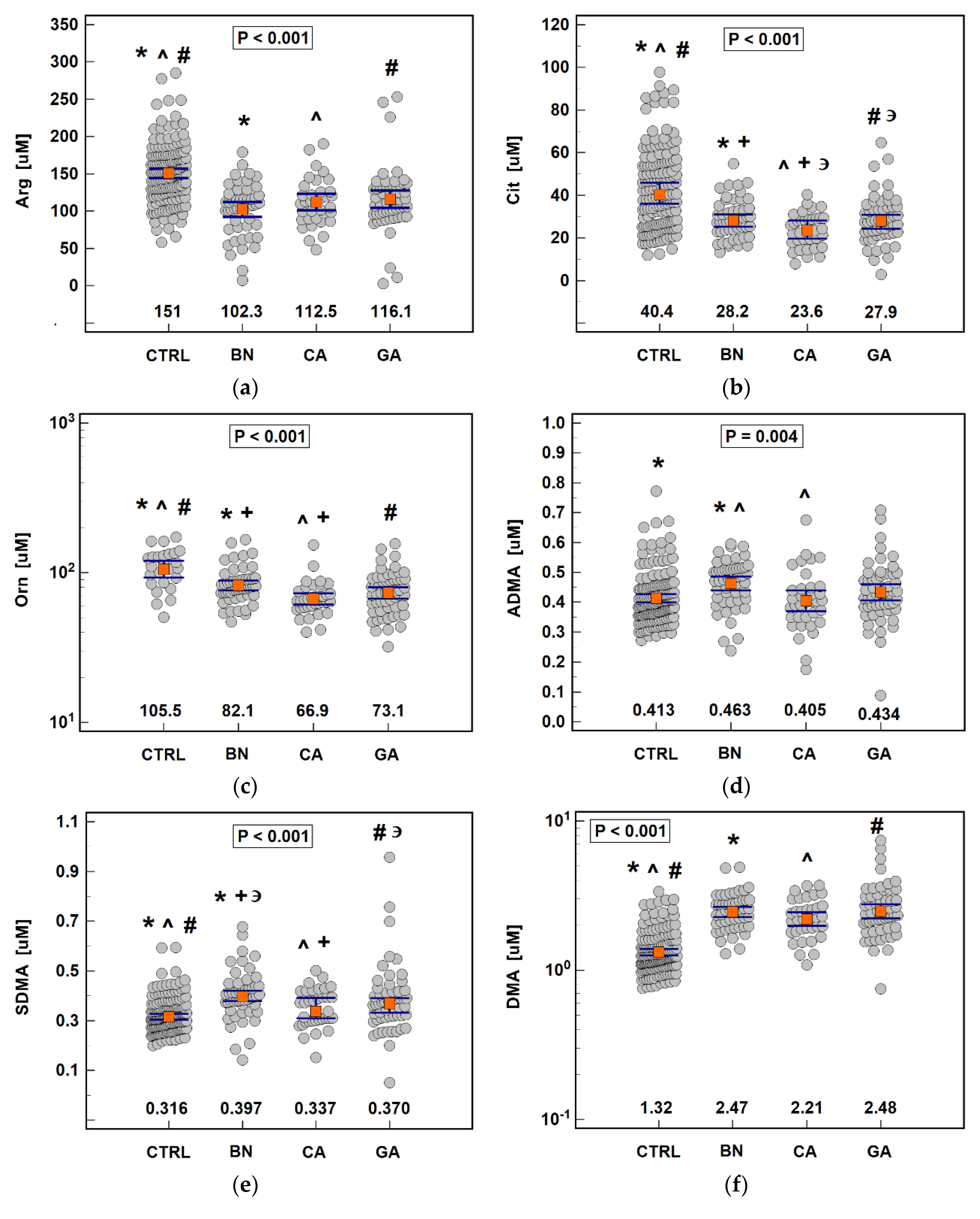
3.1. Serum Concentrations of Arg/NO Pathway Metabolites
3.1.1. Pathway Status in Gastric Cancers and Benign Disorders
3.1.2. Association with Cancer Pathology
3.1.3. Interplay between Pathway Metabolites, Cytokines and Growth Factors
3.1.4. Diagnostic Significance of the Pathway
3.2. Local Expression of Enzymes Associated with Arginine Metabolism
3.2.1. Pathway Enzymes in Gastric Cancers
3.2.2. Effect of Cancer Anatomical Site and Pathology on Pathway Enzymes
3.2.3. Interplay between Pathway Enzymes
3.2.4. Co-Expression with Markers of Proliferation, Survival, Inflammation, Angiogenesis, Metabolic Reprogramming, and Epithelial-Mesenchymal-Transition
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Sitarz, R.; Skierucha, M.; Mielko, J.; Offerhaus, G.J.A.; Maciejewski, R.; Polkowski, W.P. Gastric cancer: Epidemiology, prevention, classification, and treatment. Cancer Manag. Res. 2018, 10, 239–248. [Google Scholar] [CrossRef] [Green Version]
- Liu, X.; Meltzer, S.J. Gastric Cancer in the Era of Precision Medicine. Cell. Mol. Gastroenterol. Hepatol. 2017, 3, 348–358. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hanahan, D.; Weinberg, R.A. Hallmarks of Cancer: The Next Generation. Cell 2011, 144, 646–674. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Torresano, L.; Nuevo-Tapioles, C.; Santacatterina, F.; Cuezva, J.M. Metabolic reprogramming and disease progression in cancer patients. Biochim. Biophys. Acta Mol. Basis Dis. 2020, 1866, 165721. [Google Scholar] [CrossRef] [PubMed]
- Xiao, S.; Zhou, L. Gastric cancer: Metabolic and metabolomics perspectives (Review). Int. J. Oncol. 2017, 51, 5–17. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Faubert, B.; Solmonson, A.; DeBerardinis, R.J. Metabolic reprogramming and cancer progression. Science 2020, 368, eaaw5473. [Google Scholar] [CrossRef]
- Krzystek-Korpacka, M.; Szczęśniak-Sięga, B.; Szczuka, I.; Fortuna, P.; Zawadzki, M.; Kubiak, A.; Mierzchała-Pasierb, M.; Fleszar, M.G.; Lewandowski, Ł.; Serek, P.; et al. L-Arginine/Nitric Oxide Pathway Is Altered in Colorectal Cancer and Can Be Modulated by Novel Derivatives from Oxicam Class of Non-Steroidal Anti-Inflammatory Drugs. Cancers 2020, 12, 2594. [Google Scholar] [CrossRef]
- Bednarz-Misa, I.; Fleszar, M.G.; Zawadzki, M.; Kapturkiewicz, B.; Kubiak, A.; Neubauer, K.; Witkiewicz, W.; Krzystek-Korpacka, M. L-Arginine/NO Pathway Metabolites in Colorectal Cancer: Relevance as Disease Biomarkers and Predictors of Adverse Clinical Outcomes Following Surgery. J. Clin. Med. 2020, 9, 1782. [Google Scholar] [CrossRef]
- Bednarz-Misa, I.; Fortuna, P.; Fleszar, M.G.; Lewandowski, Ł.; Diakowska, D.; Rosińczuk, J.; Krzystek-Korpacka, M. Esophageal Squamous Cell Carcinoma Is Accompanied by Local and Systemic Changes in L-arginine/NO Pathway. Int. J. Mol. Sci. 2020, 21, 6282. [Google Scholar] [CrossRef]
- Szefel, J.; Danielak, A.; Kruszewski, W.J. Metabolic pathways of L-arginine and therapeutic consequences in tumors. Adv. Med. Sci. 2019, 64, 104–110. [Google Scholar] [CrossRef]
- Clemente, G.S.; van Waarde, A.F.; Antunes, I.; Dömling, A.H.; Elsinga, P. Arginase as a Potential Biomarker of Disease Progression: A Molecular Imaging Perspective. Int. J. Mol. Sci. 2020, 21, 5291. [Google Scholar] [CrossRef] [PubMed]
- Hulin, J.A.; Gubareva, E.A.; Jarzebska, N.; Rodionov, R.N.; Mangoni, A.A.; Tommasi, S. Inhibition of Dimethylarginine Dimethylaminohydrolase (DDAH) Enzymes as an Emerging Therapeutic Strategy to Target Angiogenesis and Vasculogenic Mimicry in Cancer. Front. Oncol. 2020, 9, 1455. [Google Scholar] [CrossRef]
- Al-Koussa, H.; El Mais, N.; Maalouf, H.; Abi-Habib, R.; El-Sibai, M. Arginine deprivation: A potential therapeutic for cancer cell metastasis? A review. Cancer Cell Int. 2020, 20, 150. [Google Scholar] [CrossRef] [PubMed]
- Nanthakumaran, S.; Brown, I.; Heys, S.D.; Schofield, A.C. Inhibition of gastric cancer cell growth by arginine: Molecular mechanisms of action. Clin. Nutr. 2009, 28, 65–70. [Google Scholar] [CrossRef]
- Keshet, R.; Erez, A. Arginine and the metabolic regulation of nitric oxide synthesis in cancer. Dis. Models Mech. 2018, 11, dmm033332. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fulton, M.D.; Brown, T.; Zheng, Y.G. The Biological Axis of Protein Arginine Methylation and Asymmetric Dimethylarginine. Int. J. Mol. Sci. 2019, 20, 3322. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fleszar, M.G.; Wiśniewski, J.; Krzystek-Korpacka, M.; Misiak, B.; Frydecka, D.; Piechowicz, J.; Lorenc-Kukuła, K.; Gamian, A. Quantitative Analysis of l-Arginine, Dimethylated Arginine Derivatives, l-Citrulline, and Dimethylamine in Human Serum Using Liquid Chromatography-Mass Spectrometric Method. Chromatographia 2018, 81, 911–921. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hellemans, J.; Vandesompele, J. qPCR data analysis—Unlocking the secret to successful results. In PCR Troubleshooting and Optimization: The Essential Guide, 1st ed.; Kennedy, S., Oswald, N., Eds.; Caister Academic Press: Poole, UK, 2011. [Google Scholar]
- Bednarz-Misa, I.; Fortuna, P.; Diakowska, D.; Jamrozik, N.; Krzystek-Korpacka, M. Distinct Local and Systemic Molecular Signatures in the Esophageal and Gastric Cancers: Possible Therapy Targets and Biomarkers for Gastric Cancer. Int. J. Mol. Sci. 2020, 21, 4509. [Google Scholar] [CrossRef]
- Schober, P.; Boer, C.; Schwarte, L.A. Correlation Coefficients: Appropriate Use and Interpretation. Anesth. Analg. 2018, 126, 1763–1768. [Google Scholar] [CrossRef]
- Miyagi, Y.; Higashiyama, M.; Gochi, A.; Akaike, M.; Ishikawa, T.; Miura, T.; Saruki, N.; Bando, E.; Kimura, H.; Imamura, F.; et al. Plasma free amino acid profiling of five types of cancer patients and its application for early detection. PLoS ONE 2011, 6, e24143. [Google Scholar] [CrossRef] [Green Version]
- Wang, Y.Z.; Cao, Y.Q.; Wu, J.N.; Chen, M.; Cha, X.Y. Expression of nitric oxide synthase in human gastric carcinoma and its relation to p53, PCNA. World J. Gastroenterol. 2005, 11, 46–50. [Google Scholar] [CrossRef]
- Kus, K.; Kij, A.; Zakrzewska, A.; Jasztal, A.; Stojak, M.; Walczak, M.; Chlopicki, S. Alterations in arginine and energy metabolism, structural and signalling lipids in metastatic breast cancer in mice detected in plasma by targeted metabolomics and lipidomics. Breast Cancer Res. 2018, 20, 148. [Google Scholar] [CrossRef]
- Okuzumi, J.; Yamane, T.; Kitao, Y.; Tokiwa, K.; Yamaguchi, T.; Fujita, Y.; Nishino, H.; Iwashima, A.; Takahashi, T. Increased mucosal ornithine decarboxylase activity in human gastric cancer. Cancer Res. 1991, 51, 1448–1451. [Google Scholar] [PubMed]
- Chaturvedi, R.; de Sablet, T.; Coburn, L.A.; Gobert, A.P.; Wilson, K.T. Arginine and polyamines in Helicobacter pylori-induced immune dysregulation and gastric carcinogenesis. Amino Acids 2012, 42, 627–640. [Google Scholar] [CrossRef] [Green Version]
- Mao, L.; Clark, D. Molecular margin of surgical resections—Where do we go from here? Cancer 2015, 121, 1914–1916. [Google Scholar] [CrossRef] [PubMed]
- Dakubo, G.D.; Jakupciak, J.P.; Birch-Machin, M.A.; Parr, R.L. Clinical implications and utility of field cancerization. Cancer Cell Int. 2007, 7, 2. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Neubauer, K.; Bednarz-Misa, I.; Diakowska, D.; Kapturkiewicz, B.; Gamian, A.; Krzystek-Korpacka, M. Nampt/PBEF/visfatin upregulation in colorectal tumors, mirrored in normal tissue and whole blood of colorectal cancer patients, is associated with metastasis, hypoxia, IL1β, and anemia. BioMed Res. Int. 2015, 2015, 523930. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Krzystek-Korpacka, M.; Gorska, S.; Diakowska, D.; Kapturkiewicz, B.; Podkowik, M.; Gamian, A.; Bednarz-Misa, I. Midkine is up-regulated in both cancerous and inflamed bowel, reflecting lymph node metastasis in colorectal cancer and clinical activity of ulcerative colitis. Cytokine 2017, 89, 68–75. [Google Scholar] [CrossRef] [PubMed]
- Asplund, J.; Kauppila, J.H.; Mattsson, F.; Lagergren, J. Survival Trends in Gastric Adenocarcinoma: A Population-Based Study in Sweden. Ann. Surg. Oncol. 2018, 25, 2693–2702. [Google Scholar] [CrossRef] [PubMed]
- Kwiecien, S.; Ptak-Belowska, A.; Krzysiek-Maczka, G.; Targosz, A.; Jasnos, K.; Magierowski, M.; Szczyrk, U.; Brzozowski, B.; Konturek, S.J.; Konturek, P.C.; et al. Asymmetric dimethylarginine, an endogenous inhibitor of nitric oxide synthase, interacts with gastric oxidative metabolism and enhances stress-induced gastric lesions. J. Physiol. Pharmacol. 2012, 63, 515–524. [Google Scholar]
- Wang, L.; Zhou, Y.; Peng, J.; Zhang, Z.; Jiang, D.J.; Li, Y.J. Role of endogenous nitric oxide synthase inhibitor in gastric mucosal injury. Can. J. Physiol. Pharmacol. 2008, 86, 97–104. [Google Scholar] [CrossRef] [PubMed]
- Guo, Q.; Xu, J.; Huang, Z.; Yao, Q.; Chen, F.; Liu, H.; Zhang, Z.; Lin, J. ADMA mediates gastric cancer cell migration and invasion via Wnt/β-catenin signaling pathway. Clin. Transl. Oncol. 2021, 23, 325–334. [Google Scholar] [CrossRef]
- Brankovic, B.; Stanojevic, G.; Stojanovic, I.; Veljkovic, A.; Kocic, G.; Janosevic, P.; Nestorovic, M.; Petrovic, D.; Djindjic, B.; Pavlovic, D.; et al. Nitric oxide synthesis modulation—A possible diagnostic and therapeutic target in colorectal cancer. J. BUON 2017, 22, 162–169. [Google Scholar] [PubMed]
- Anestis, A.; Zoi, I.; Karamouzis, M.V. Current advances of targeting HGF/c-Met pathway in gastric cancer. Ann. Transl. Med. 2018, 6, 247. [Google Scholar] [CrossRef] [PubMed]
- Mazzoldi, E.L.; Pavan, S.; Pilotto, G.; Leone, K.; Pagotto, A.; Frezzini, S.; Nicoletto, M.O.; Amadori, A.; Pastò, A. A juxtacrine/paracrine loop between C-Kit and stem cell factor promotes cancer stem cell survival in epithelial ovarian cancer. Cell Death Dis. 2019, 10, 412. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ye, J.; Xu, J.; Li, Y.; Huang, Q.; Huang, J.; Wang, J.; Zhong, W.; Lin, X.; Chen, W.; Lin, X. DDAH1 mediates gastric cancer cell invasion and metastasis via Wnt/β-catenin signaling pathway. Mol. Oncol. 2017, 11, 1208–1224. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zeisel, S.H.; DaCosta, K.A.; Edrise, B.M.; Fox, J.G. Transport of dimethylamine, a precursor of nitrosodimethylamine, into stomach of ferret and dog. Carcinogenesis 1986, 7, 775–778. [Google Scholar] [CrossRef]
- Sukowati, C.H.C.; Patti, R.; Pascut, D.; Ladju, R.B.; Tarchi, P.; Zanotta, N.; Comar, M.; Tiribelli, C.; Crocè, L.S. Serum Stem Cell Growth Factor Beta for the Prediction of Therapy Response in Hepatocellular Carcinoma. BioMed Res. Int. 2018, 2018, 6435482. [Google Scholar] [CrossRef]
- Rabinovich, S.; Adler, L.; Yizhak, K.; Sarver, A.; Silberman, A.; Agron, S.; Stettner, N.; Sun, Q.; Brandis, A.; Helbling, D.; et al. Diversion of aspartate in ASS1-deficient tumours fosters de novo pyrimidine synthesis. Nature 2015, 527, 379–383. [Google Scholar] [CrossRef]
- Silberman, A.; Goldman, O.; Boukobza Assayag, O.; Jacob, A.; Rabinovich, S.; Adler, L.; Lee, J.S.; Keshet, R.; Sarver, A.; Frug, J.; et al. Acid-Induced Downregulation of ASS1 Contributes to the Maintenance of Intracellular pH in Cancer. Cancer Res. 2019, 79, 518–533. [Google Scholar] [CrossRef] [Green Version]
- Lee, J.S.; Adler, L.; Karathia, H.; Carmel, N.; Rabinovich, S.; Auslander, N.; Keshet, R.; Stettner, N.; Silberman, A.; Agemy, L.; et al. Urea Cycle Dysregulation Generates Clinically Relevant Genomic and Biochemical Signatures. Cell 2018, 174, 1559–1570.e22. [Google Scholar] [CrossRef] [Green Version]
- Yang, Y.; Bedford, M.T. Protein arginine methyltransferases and cancer. Nat. Rev. Cancer 2013, 13, 37–50. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Wang, C.; Jiang, H.; Luo, C. A patent review of arginine methyltransferase inhibitors (2010–2018). Expert Opin. Ther. Pat. 2019, 29, 97–114. [Google Scholar] [CrossRef] [PubMed]
- Baldwin, R.M.; Morettin, A.; Côté, J. Role of PRMTs in cancer: Could minor isoforms be leaving a mark? World J. Biol. Chem. 2014, 5, 115–129. [Google Scholar] [CrossRef] [PubMed]
- Sato, H.; Ishihara, S.; Kawashima, K.; Moriyama, N.; Suetsugu, H.; Kazumori, H.; Okuyama, T.; Rumi, M.A.; Fukuda, R.; Nagasue, N.; et al. Expression of peroxisome proliferator-activated receptor (PPAR)gamma in gastric cancer and inhibitory effects of PPARgamma agonists. Br. J. Cancer 2000, 83, 1394–1400. [Google Scholar] [CrossRef] [Green Version]
- Pazienza, V.; Vinciguerra, M.; Mazzoccoli, G. PPARs Signaling and Cancer in the Gastrointestinal System. PPAR Res. 2012, 2012, 560846. [Google Scholar] [CrossRef] [Green Version]
- Zhong, J.; Chen, Y.J.; Chen, L.; Shen, Y.Y.; Zhang, Q.H.; Yang, J.; Cao, R.X.; Zu, X.Y.; Wen, G.B. PRMT2β, a C-terminal splice variant of PRMT2, inhibits the growth of breast cancer cells. Oncol. Rep. 2017, 38, 1303–1311. [Google Scholar] [CrossRef]





| Characteristics: | Controls | Benign Disorders | Cardia Subtype | Non-Cardia Subtype | p |
|---|---|---|---|---|---|
| N | 153 | 50 | 35 | 55 | - |
| Sex (F/M), n | 74/79 | 25/25 | 12/23 | 20/35 | 0.214 1 |
| Age (y), mean ± SD | 59.8 ± 12 | 58.8 ± 14 | 59.3 ± 8 | 62.3 ± 12 | 0.455 2 |
| Stage (I/II/III/IV) | na | na | 0/5/4/26 | 4/8/7/36 | 0.421 3 |
| Primary tumor, T (1/2/3/4) | na | na | 0/2/7/26 | 5/3/14/33 | 0.249 3 |
| Lymph node metastasis, N (no/yes) | na | na | 6/29 | 12/43 | 0.788 1 |
| Distant metastasis, M (no/yes) | na | na | 9/26 | 19/36 | 0.485 1 |
| Characteristics: | All | GA | CA | p |
|---|---|---|---|---|
| N | 29 | 17 | 12 | - |
| Sex (F/M), n | 16/13 | 10/7 | 6/6 | 0.716 1 |
| Age (y), mean (95% CI) | 65.2 (62–69) | 65.1 (60–70) | 65.4 (61–70) | 0.921 2 |
| Stage (I/II/III/IV) | 4/6/13/6 | 3/3/7/4 | 1/3/6/2 | 0.823 3 |
| Primary tumor, T (1-2/3/4) | 5/18/6 | 4/10/3 | 1/8/3 | 0.550 3 |
| Lymph node metastasis, N (no/yes) | 11/18 | 6/11 | 5/7 | 1.0 1 |
| Distant metastasis, M (no/yes) | 23/6 | 13/4 | 10/2 | 1.0 1 |
| Histological grade, G (1/2/3) | 3/13/13 | 2/6/9 | 1/7/4 | 0.469 3 |
| Metabolite | TNM | T | N0 vs. N(+) | M0 vs. M1 |
|---|---|---|---|---|
| Cit | ns | ns | 31.0 ± 13 vs. 25.6 ± 9.4, p = 0.048 2 | 30.5 ± 11 vs. 25.0 ± 9.7, p = 0.018 2 |
| Orn | ρ = −0.26, p = 0.012 1 | ρ = −0.22, p = 0.040 1 | 89.0 ± 23.1 vs. 70.2 ± 22.6, p = 0.002 2 | ns |
| ADMA | ρ = −0.21, p = 0.047 1 | ρ = −0.25, p = 0.017 1 | ns | ns |
| Metabolite | Cytokine | n | ρ | Cytokine | n | ρ |
|---|---|---|---|---|---|---|
| Arg | IL-2 | 28 | −0.49 2 | EOX1 | 82 | 0.24 1 |
| IL-15 | 23 | −0.51 1 | MIF | 26 | −0.57 2 | |
| IL-16 | 26 | −0.42 1 | SDF1α | 26 | −0.55 2 | |
| Cit | IL-4 | 82 | −0.26 1 | FGF2 | 82 | −0.36 3 |
| IL-6 | 82 | −0.33 2 | GM-CSF | 82 | −0.23 1 | |
| IL-7 | 82 | −0.23 1 | PDGF-BB | 82 | −0.23 1 | |
| IL-10 | 82 | −0.26 1 | VEGF-A | 82 | −0.26 1 | |
| IL-18 | 26 | −0.53 2 | TRAIL | 26 | −0.43 1 | |
| Orn | IL-1β | 82 | −0.34 2 | IL-17 | 82 | −0.41 3 |
| IL-4 | 82 | −0.25 1 | G-CSF | 82 | −0.25 1 | |
| IL-6 | 82 | −0.24 1 | IFNγ | 82 | −0.30 2 | |
| IL-7 | 82 | −0.38 3 | MIP-1α | 82 | −0.33 2 | |
| IL-10 | 82 | 0.22 1 | PDGF-BB | 82 | −0.26 1 | |
| IL-16 | 26 | 0.40 1 | ||||
| ADMA | G-CSF | 82 | 0.26 1 | IP-10 | 82 | 0.22 1 |
| HGF | 26 | 0.41 1 | MIP-1α | 82 | 0.23 1 | |
| IFNγ | 82 | 0.29 2 | ||||
| SDMA | HGF | 26 | 0.42 1 | SCF | 26 | 0.40 1 |
| DMA | IP-10 | 82 | 0.29 2 | RANTES | 82 | 0.23 1 |
| MIF | 26 | −0.43 1 | SCGFβ | 26 | 0.64 3 | |
| MIP-1β | 82 | 0.22 1 |
| Metabolite | Parameter | Cancers vs. CTRL | Cancer vs. BN | CA vs. GA |
|---|---|---|---|---|
| Arg | AUC (95%CI) | 0.770 (0.71–0.82) 1 | ns | ns |
| criterion | ≤142.9 μM | |||
| sens. and spec. | 88.9 and 58.2 | |||
| Cit | AUC (95%CI) | 0.758 (0.70–0.81) 1 | ns | 0.631 (0.52–0.73) 3 |
| criterion | ≤37.7 μM | ≤29.2 μM | ||
| sens. and spec. | 91.1 and 55.6 | 82.9 and 41.8 | ||
| Orn | AUC (95%CI) | 0.821 (0.74–0.89) 1 | 0.648 (0.56–0.73) 2 | ns |
| criterion | ≤91.8 μM | ≤70.6 μM | ||
| sens. and spec. | 84.4 and 72.0 | 54.4 and 78.0 | ||
| ADMA | AUC (95%CI) | ns | 0.654 (0.57–0.73) 2 | ns |
| criterion | ≤0.454 μM | |||
| sens. and spec. | 70.0 and 62.0 | |||
| SDMA | AUC (95%CI) | 0.638 (0.57–0.70) 1 | 0.629 (0.54–0.71) 2 | ns |
| criterion | >0.356 μM | ≤0.373 μM | ||
| sens. and spec. | 55.6 and 72.5 | 53.3 and 72.0 | ||
| DMA | AUC (95%CI) | 0.887 (0.84–0.92) 1 | ns | ns |
| criterion | >1.5 μM | |||
| sens. and spec. | 93.3 and 73.2 | |||
| Panel (all metabolites) | AUC (95%CI) | 0.914 (0.85–0.96) 1 | 0.697 (0.61–0.77) 1 | 0.659 (0.55–0.76) 2 |
| criterion | >0.659 4 | >0.634 4 | >0.366 4 | |
| sens. and spec. | 91.1 and 88.0 | 76.6 and 68.0 | 77.1 and 66.0 |
| Gene | Normal Mean (95%CI) [NRQ] | Tumor Mean (95%CI) [NRQ] | p Value | Expression Ratio T/N (N/T) |
|---|---|---|---|---|
| ASL | 2.02 (1.65–2.88) | 1.05 (0.70–1.48) | 0.010 1 | 0.52 (1.9) |
| ARG1 | 2.29 (0.18–6.20) | 1.77 (0.42–3.47) | 0.456 1 | - |
| ARG2 | 1.08 (0.76–1.62) | 0.86 (0.55–2.35) | 0.981 1 | - |
| ASS1 | 1.16 (0.72–1.36) | 1.62 (0.84–1.87) | 0.071 1 | - |
| DDAH1 | 1.12 (1.01–1.69) | 0.70 (0.55–0.96) | 0.026 1 | 0.62 (1.6) |
| DDAH2 | 1.27 (0.84–1.84) | 1.10 (0.50–1.77) | 0.737 1 | - |
| NOS2 | 0.19 (0.07–0.54) | 3.09 (1.34–7.13) | <0.001 2 | 16.4 |
| ODC1 | 1.24 (0.67–1.71) | 0.81 (0.63–1.4) | 0.400 1 | - |
| ORNT1 | 2.00 (1.67–2.84) | 0.79 (0.59–1.62) | 0.003 1 | 0.39 (2.5) |
| PRMT1 | 1.49 (0.96–1.96) | 1.06 (0.71–1.50) | 0.524 1 | - |
| PRMT2 | 1.39 (1.13–2.53) | 0.84 (0.53–1.37) | 0.037 1 | 0.60 (1.7) |
| PRMT5 | 1.19 (0.90–1.72) | 0.92 (0.68–1.26) | 0.249 1 | - |
| Gene | ODC1 | DDAH1 | ARG2 | PRMT1 | DDAH2 | ORNT1 | PRMT2 | ASL | ASS1 | ARG1 | NOS2 |
|---|---|---|---|---|---|---|---|---|---|---|---|
| PRMT5 | 0.89 1 | 0.88 1 | 0.90 1 | 0.93 1 | 0.86 1 | 0.77 1 | 0.81 1 | 0.75 1 | 0.74 1 | 0.54 3 | ns |
| ODC1 | 0.80 1 | 0.93 1 | 0.78 1 | 0.77 1 | 0.83 1 | 0.71 1 | 0.74 1 | 0.73 1 | 0.53 3 | ns | |
| DDAH1 | 0.85 1 | 0.85 1 | 0.71 1 | 0.78 1 | 0.73 1 | 0.82 1 | 0.75 1 | ns | ns | ||
| ARG2 | 0.82 1 | 0.73 1 | 0.74 1 | 0.71 1 | 0.67 1 | 0.65 2 | 0.56 3 | ns | |||
| PRMT1 | 0.86 1 | 0.70 1 | 0.81 1 | 0.66 1 | 0.62 2 | 0.44 4 | ns | ||||
| DDAH2 | 0.70 1 | 0.93 1 | 0.64 2 | 0.67 2 | 0.51 3 | ns | |||||
| ORNT1 | 0.73 1 | 0.86 1 | 0.81 1 | 0.45 4 | ns | ||||||
| PRMT2 | 0.72 1 | 0.71 1 | 0.39 4 | ns | |||||||
| ASL | 0.82 1 | ns | ns | ||||||||
| ASS1 | 0.45 4 | ns | |||||||||
| ARG1 | ns |
| PRMT5 | PRMT1 | DDAH1 | PRMT2 | ORNT1 | ODC1 | ARG2 | ASL | ASS1 | NOS2 | ARG1 | |
|---|---|---|---|---|---|---|---|---|---|---|---|
| DDAH2 | 0.79 1 | 0.79 1 | 0.66 2 | 0.75 1 | 0.63 2 | 0.70 1 | 0.74 1 | 0.56 3 | 0.51 3 | 0.51 3 | 0.43 4 |
| PRMT5 | 0.93 1 | 0.81 1 | 0.87 1 | 0.66 2 | 0.52 3 | 0.57 3 | 0.60 2 | 0.43 4 | ns | ns | |
| PRMT1 | 0.79 1 | 0.78 1 | 0.58 2 | 0.52 3 | 0.62 2 | 0.49 3 | 0.49 3 | 0.42 4 | ns | ||
| DDAH1 | 0.64 2 | 0.66 2 | 0.60 2 | 0.54 3 | 0.65 2 | 0.39 4 | ns | ns | |||
| PRMT2 | 0.51 3 | 0.39 4 | 0.49 3 | 0.63 2 | ns | ns | ns | ||||
| ORNT1 | 0.67 1 | 0.48 4 | 0.75 1 | 0.37 4 | 0.51 3 | ns | |||||
| ODC1 | 0.77 1 | 0.48 4 | 0.52 3 | 0.43 4 | 0.46 4 | ||||||
| ARG2 | ns | 0.55 3 | ns | 0.45 4 | |||||||
| ASL | ns | 0.55 3 | ns | ||||||||
| ASS1 | ns | 0.66 2 | |||||||||
| NOS2 | 0.39 4 |
| Gene | ASS1 | ARG2 | ASL | DDAH1 | DDAH2 | ODC1 | ORNT1 | PRMT1 | PRMT2 | PRMT5 |
|---|---|---|---|---|---|---|---|---|---|---|
| Ki67 | 0.68 2 | 0.72 2 | ns | 0.70 2 | 0.71 2 | 0.61 3 | ns | 0.72 2 | 0.73 2 | 0.85 1 |
| HIF1A | 0.56 3 | 0.70 2 | ns | ns | 0.76 2 | ns | ns | 0.60 3 | 0.77 2 | 0.84 1 |
| CDKN1A | ns | 0.60 3 | ns | ns | ns | 0.67 3 | ns | ns | ns | 0.67 3 |
| BCLXL | 0.54 3 | 0.69 2 | ns | 0.74 2 | 0.75 2 | 0.58 3 | ns | 0.72 2 | 0.73 2 | 0.88 1 |
| PTGS2 | ns | ns | 0.57 3 | ns | ns | 0.61 3 | 0.65 2 | 0.60 3 | 0.59 3 | 0.73 2 |
| CCL2 | ns | 0.61 3 | ns | 0.56 3 | 0.76 2 | ns | ns | ns | 0.81 1 | 0.80 1 |
| GLUT1 | 0.84 1 | 0.64 3 | ns | ns | ns | 0.56 3 | ns | 0.64 2 | ns | 0.66 3 |
| VEGFA | ns | ns | ns | ns | ns | 0.64 3 | 0.58 3 | 0.67 2 | ns | 0.70 2 |
| CLDN2 | ns | ns | ns | 0.65 3 | 0.57 3 | ns | ns | 0.58 3 | 0.59 3 | 0.75 2 |
| TJP1 | ns | 0.65 3 | ns | ns | 0.63 3 | 0.56 3 | ns | ns | 0.66 3 | 0.63 3 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bednarz-Misa, I.; Fleszar, M.G.; Fortuna, P.; Lewandowski, Ł.; Mierzchała-Pasierb, M.; Diakowska, D.; Krzystek-Korpacka, M. Altered L-Arginine Metabolic Pathways in Gastric Cancer: Potential Therapeutic Targets and Biomarkers. Biomolecules 2021, 11, 1086. https://doi.org/10.3390/biom11081086
Bednarz-Misa I, Fleszar MG, Fortuna P, Lewandowski Ł, Mierzchała-Pasierb M, Diakowska D, Krzystek-Korpacka M. Altered L-Arginine Metabolic Pathways in Gastric Cancer: Potential Therapeutic Targets and Biomarkers. Biomolecules. 2021; 11(8):1086. https://doi.org/10.3390/biom11081086
Chicago/Turabian StyleBednarz-Misa, Iwona, Mariusz G. Fleszar, Paulina Fortuna, Łukasz Lewandowski, Magdalena Mierzchała-Pasierb, Dorota Diakowska, and Małgorzata Krzystek-Korpacka. 2021. "Altered L-Arginine Metabolic Pathways in Gastric Cancer: Potential Therapeutic Targets and Biomarkers" Biomolecules 11, no. 8: 1086. https://doi.org/10.3390/biom11081086
APA StyleBednarz-Misa, I., Fleszar, M. G., Fortuna, P., Lewandowski, Ł., Mierzchała-Pasierb, M., Diakowska, D., & Krzystek-Korpacka, M. (2021). Altered L-Arginine Metabolic Pathways in Gastric Cancer: Potential Therapeutic Targets and Biomarkers. Biomolecules, 11(8), 1086. https://doi.org/10.3390/biom11081086







