Sensitivity of Acute Myelocytic Leukemia Cells to the Dienone Compound VLX1570 Is Associated with Inhibition of the Ubiquitin-Proteasome System
Abstract
:1. Introduction
2. Materials and Methods
2.1. Cell Lines
2.2. Reagents and Antibodies
2.3. Cell Viability Assays
2.4. SDS PAGE and Immunoblotting
2.5. Glutathione Analysis
2.6. Developmental Toxicity Assay
2.7. Zebrafish Tumor Model
2.8. CMap Analysis
3. Results
3.1. Properties of Dienone Compounds
3.2. VLX1570 Induces the Loss of Viability of Acute Myelocytic Leukemia (AML) Cells
3.3. VLX1570 Induces Proteotoxic Stress and ER Stress in AML Cell Lines
3.4. VLX1570 Induces Glutathione Depletion and Formation of High-Molecular-Weight Protein Complexes
3.5. VLX1570 Inhibits Growth of AML Cells in Zebrafish Embryos
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Hossain, M.; Enci, C.E.; Dimmock, J.R.; Das, U. Discovery and investigation of 1-[4-(2-aminoethoxy)phenylcarbonyl]-3,5-bis-(benzylidene)-4-piperidones as candidate antineoplastic agents: Our last 15 years study. Curr. Med. Chem. 2020, 28, 2453–2464. [Google Scholar] [CrossRef] [PubMed]
- Bazzaro, M.; Linder, S. Dienone Compounds: Targets and Pharmacological Responses. J. Med. Chem. 2020, 63, 15075–15093. [Google Scholar] [CrossRef] [PubMed]
- Hossain, M.; Das, U.; Dimmock, J.R. Recent advances in alpha,beta-unsaturated carbonyl compounds as mitochondrial toxins. Eur. J. Med. Chem. 2019, 183, 111687. [Google Scholar] [CrossRef]
- Anchoori, R.K.; Karanam, B.; Peng, S.; Wang, J.W.; Jiang, R.; Tanno, T.; Orlowski, R.Z.; Matsui, W.; Zhao, M.; Rudek, M.A.; et al. A bis-benzylidine piperidone targeting proteasome ubiquitin receptor RPN13/ADRM1 as a therapy for cancer. Cancer Cell 2013, 24, 791–805. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- D’Arcy, P.; Brnjic, S.; Olofsson, M.H.; Fryknäs, M.; Lindsten, K.; De Cesare, M.; Perego, P.; Sadeghi, B.; Hassan, M.; Larsson, R.; et al. Inhibition of proteasome deubiquitinating activity as a new cancer therapy. Nat. Med. 2011, 17, 1636–1640. [Google Scholar] [CrossRef] [PubMed]
- Osei-Amponsa, V.; Sridharan, V.; Tandon, M.; Evans, C.N.; Klarmann, K.; Cheng, K.T.; Lack, J.; Chari, R.; Walters, K.J. Impact of losing hRpn13 Pru or UCHL5 on proteasome clearance of ubiquitinated proteins and RA190 cytotoxicity. Mol. Cell. Biol. 2020, 40, 122. [Google Scholar] [CrossRef]
- Adams, J. The development of proteasome inhibitors as anticancer drugs. Cancer Cell 2004, 5, 417–421. [Google Scholar] [CrossRef] [Green Version]
- Stanwell, C.; Burke, T.R., Jr.; Yuspa, S.H. The erbstatin analogue methyl 2,5-dihydroxycinnamate cross-links proteins and is cytotoxic to normal and neoplastic epithelial cells by a mechanism independent of tyrosine kinase inhibition. Cancer Res. 1995, 55, 4950–4956. [Google Scholar]
- Ward, J.A.; Pinto-Fernandez, A.; Cornelissen, L.; Bonham, S.; Diaz-Saez, L.; Riant, O.; Huber, K.V.M.; Kessler, B.M.; Feron, O.; Tate, E.W. Re-Evaluating the Mechanism of Action of alpha,beta-Unsaturated Carbonyl DUB Inhibitors b-AP15 and VLX1570: A Paradigmatic Example of Unspecific Protein Cross-linking with Michael Acceptor Motif-Containing Drugs. J. Med. Chem. 2020, 63, 3756–3762. [Google Scholar] [CrossRef] [Green Version]
- Dohner, H.; Weisdorf, D.J.; Bloomfield, C.D. Acute Myeloid Leukemia. N. Engl. J. Med. 2015, 373, 1136–1152. [Google Scholar] [CrossRef] [Green Version]
- Mrozek, K. Cytogenetic, molecular genetic, and clinical characteristics of acute myeloid leukemia with a complex karyotype. Semin. Oncol. 2008, 35, 365–377. [Google Scholar] [CrossRef] [Green Version]
- Dohner, H.; Estey, E.H.; Amadori, S.; Appelbaum, F.R.; Buchner, T.; Burnett, A.K.; Dombret, H.; Fenaux, P.; Grimwade, D.; Larson, R.A.; et al. Diagnosis and management of acute myeloid leukemia in adults: Recommendations from an international expert panel, on behalf of the European LeukemiaNet. Blood J. Am. Soc. Hematol. 2010, 115, 453–474. [Google Scholar] [CrossRef]
- Hunter, A.M.; Sallman, D.A. Current status and new treatment approaches in TP53 mutated AML. Best Pract. Res. Clin. Haematol. 2019, 32, 134–144. [Google Scholar] [CrossRef]
- Rowinsky, E.K.; Paner, A.; Berdeja, J.G.; Paba-Prada, C.; Venugopal, P.; Porkka, K.; Gullbo, J.; Linder, S.; Loskog, A.; Richardson, P.G.; et al. Phase 1 study of the protein deubiquitinase inhibitor VLX1570 in patients with relapsed and/or refractory multiple myeloma. Investig. N. Drugs 2020, 38, 1448–1453. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cersosimo, U.; Sgorbissa, A.; Foti, C.; Drioli, S.; Angelica, R.; Tomasella, A.; Picco, R.; Semrau, M.S.; Storici, P.; Benedetti, F.; et al. Synthesis, characterization, and optimization for in vivo delivery of a nonselective isopeptidase inhibitor as new antineoplastic agent. J. Med. Chem. 2015, 58, 1691–1704. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kurozumi, N.; Tsujioka, T.; Ouchida, M.; Sakakibara, K.; Nakahara, T.; Suemori, S.I.; Takeuchi, M.; Kitanaka, A.; Shibakura, M.; Tohyama, K. VLX1570 induces apoptosis through the generation of ROS and induction of ER stress on leukemia cell lines. Cancer Sci. 2021, 112, 3302. [Google Scholar] [CrossRef] [PubMed]
- Pei, S.; Minhajuddin, M.; Callahan, K.P.; Balys, M.; Ashton, J.M.; Neering, S.J.; Lagadinou, E.D.; Corbett, C.; Ye, H.; Liesveld, J.L.; et al. Targeting aberrant glutathione metabolism to eradicate human acute myelogenous leukemia cells. J. Biol. Chem. 2013, 288, 33542–33558. [Google Scholar] [CrossRef] [Green Version]
- Marques, I.J.; Weiss, F.U.; Vlecken, D.H.; Nitsche, C.; Bakkers, J.; Lagendijk, A.K.; Partecke, L.I.; Heidecke, C.D.; Lerch, M.M.; Bagowski, C.P. Metastatic behaviour of primary human tumours in a zebrafish xenotransplantation model. BMC Cancer 2009, 9, 128. [Google Scholar] [CrossRef] [Green Version]
- Zhang, B.; Shimada, Y.; Kuroyanagi, J.; Umemoto, N.; Nishimura, Y.; Tanaka, T. Quantitative phenotyping-based in vivo chemical screening in a zebrafish model of leukemia stem cell xenotransplantation. PLoS ONE 2014, 9, e85439. [Google Scholar] [CrossRef]
- He, Y.; Li, W.; Hu, G.; Sun, H.; Kong, Q. Bioactivities of EF24, a Novel Curcumin Analog: A Review. Front. Oncol. 2018, 8, 614. [Google Scholar] [CrossRef] [Green Version]
- Adams, B.K.; Cai, J.; Armstrong, J.; Herold, M.; Lu, Y.J.; Sun, A.; Snyder, J.P.; Liotta, D.C.; Jones, D.P.; Shoji, M. EF24, a novel synthetic curcumin analog, induces apoptosis in cancer cells via a redox-dependent mechanism. Anti-Cancer Drugs 2005, 16, 263–275. [Google Scholar] [CrossRef]
- Liu, H.; Liang, Y.; Wang, L.; Tian, L.; Song, R.; Han, T.; Pan, S.; Liu, L. In vivo and in vitro suppression of hepatocellular carcinoma by EF24, a curcumin analog. PLoS ONE 2012, 7, e48075. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yin, D.L.; Liang, Y.J.; Zheng, T.S.; Song, R.P.; Wang, J.B.; Sun, B.S.; Pan, S.H.; Qu, L.D.; Liu, J.R.; Jiang, H.C.; et al. EF24 inhibits tumor growth and metastasis via suppressing NF-kappaB dependent pathways in human cholangiocarcinoma. Sci. Rep. 2016, 6, 32167. [Google Scholar] [CrossRef] [Green Version]
- Delaney, J.S. ESOL: Estimating aqueous solubility directly from molecular structure. J. Chem. Inf. Comput. Sci. 2004, 44, 1000–1005. [Google Scholar] [CrossRef] [PubMed]
- Daina, A.; Michielin, O.; Zoete, V. SwissADME: A free web tool to evaluate pharmacokinetics, drug-likeness and medicinal chemistry friendliness of small molecules. Sci. Rep. 2017, 7, 42717. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Subramanian, A.; Narayan, R.; Corsello, S.M.; Peck, D.D.; Natoli, T.E.; Lu, X.; Gould, J.; Davis, J.F.; Tubelli, A.A.; Asiedu, J.K.; et al. A Next Generation Connectivity Map: L1000 Platform and the First 1,000,000 Profiles. Cell 2017, 171, 1437–1452. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; D’Arcy, P.; Caulfield, T.R.; Paulus, A.; Chitta, K.; Mohanty, C.; Gullbo, J.; Chanan-Khan, A.; Linder, S. Synthesis and evaluation of derivatives of the proteasome deubiquitinase inhibitor b-AP15. Chem. Biol. Drug Des. 2015, 86, 1036–1048. [Google Scholar] [CrossRef] [Green Version]
- Chan, L.L.; Smith, T.; Kumph, K.A.; Kuksin, D.; Kessel, S.; Dery, O.; Cribbes, S.; Lai, N.; Qiu, J. A high-throughput AO/PI-based cell concentration and viability detection method using the Celigo image cytometry. Cytotechnology 2016, 68, 2015–2025. [Google Scholar] [CrossRef] [Green Version]
- Fleckenstein, D.S.; Uphoff, C.C.; Drexler, H.G.; Quentmeier, H. Detection of p53 gene mutations by single strand conformational polymorphism (SSCP) in human acute myeloid leukemia-derived cell lines. Leuk. Res. 2002, 26, 207–214. [Google Scholar] [CrossRef]
- Matsuo, Y.; MacLeod, R.A.; Uphoff, C.C.; Drexler, H.G.; Nishizaki, C.; Katayama, Y.; Kimura, G.; Fujii, N.; Omoto, E.; Harada, M.; et al. Two acute monocytic leukemia (AML-M5a) cell lines (MOLM-13 and MOLM-14) with interclonal phenotypic heterogeneity showing MLL-AF9 fusion resulting from an occult chromosome insertion, ins(11;9)(q23;p22p23). Leukemia 1997, 11, 1469–1477. [Google Scholar] [CrossRef] [Green Version]
- Weisberg, E.; Halilovic, E.; Cooke, V.G.; Nonami, A.; Ren, T.; Sanda, T.; Simkin, I.; Yuan, J.; Antonakos, B.; Barys, L.; et al. Inhibition of Wild-Type p53-Expressing AML by the Novel Small Molecule HDM2 Inhibitor CGM097. Mol. Cancer Ther. 2015, 14, 2249–2259. [Google Scholar] [CrossRef] [Green Version]
- Hamaguchi, H.; Suzukawa, K.; Nagata, K.; Yamamoto, K.; Yagasaki, F.; Morishita, K. Establishment of a novel human myeloid leukaemia cell line (HNT-34) with t(3;3)(q21;q26), t(9;22)(q34;q11) and the expression of EVI1 gene, P210 and P190 BCR/ABL chimaeric transcripts from a patient with AML after MDS with 3q21q26 syndrome. Br. J. Haematol. 1997, 98, 399–407. [Google Scholar] [CrossRef] [Green Version]
- Asou, H.; Tashiro, S.; Hamamoto, K.; Otsuji, A.; Kita, K.; Kamada, N. Establishment of a human acute myeloid leukemia cell line (Kasumi-1) with 8;21 chromosome translocation. Blood 1991, 77, 2031–2036. [Google Scholar] [CrossRef] [Green Version]
- Fribley, A.; Zeng, Q.; Wang, C.Y. Proteasome inhibitor PS-341 induces apoptosis through induction of endoplasmic reticulum stress-reactive oxygen species in head and neck squamous cell carcinoma cells. Mol. Cell. Biol. 2004, 24, 9695–9704. [Google Scholar] [CrossRef] [Green Version]
- Lebeaupin, C.; Yong, J.; Kaufman, R.J. The Impact of the ER Unfolded Protein Response on Cancer Initiation and Progression: Therapeutic Implications. Adv. Exp. Med. Biol. 2020, 1243, 113–131. [Google Scholar] [PubMed]
- Zhang, K.; Kaufman, R.J. The unfolded protein response: A stress signaling pathway critical for health and disease. Neurology 2006, 66 (Suppl. 1), S102–S109. [Google Scholar] [CrossRef]
- Slee, E.A.; Zhu, H.; Chow, S.C.; MacFarlane, M.; Nicholson, D.W.; Cohen, G.M. Benzyloxycarbonyl-Val-Ala-Asp (OMe) fluoromethylketone (Z-VAD.FMK) inhibits apoptosis by blocking the processing of CPP32. Biochem. J. 1996, 315, 21–24. [Google Scholar] [CrossRef] [Green Version]
- Brnjic, S.; Mazurkiewicz, M.; Fryknas, M.; Sun, C.; Zhang, X.; Larsson, R.; D’Arcy, P.; Linder, S. Induction of tumor cell apoptosis by a proteasome deubiquitinase inhibitor is associated with oxidative stress. Antioxid. Redox Signal. 2014, 21, 2271–2285. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pellegrini, P.; Selvaraju, K.; Faustini, E.; Mofers, A.; Zhang, X.; Ternerot, J.; Schubert, A.; Linder, S.; Padraig, D.A. Induction of ER Stress in Acute Lymphoblastic Leukemia Cells by the Deubiquitinase Inhibitor VLX1570. Int. J. Mol. Sci. 2020, 21, 4757. [Google Scholar] [CrossRef] [PubMed]
- Selvaraju, K.; Mofers, A.; Pellegrini, P.; Salomonsson, J.; Ahlner, A.; Morad, V.; Hillert, E.K.; Espinosa, B.; Arner, E.S.J.; Jensen, L.; et al. Cytotoxic unsaturated electrophilic compounds commonly target the ubiquitin proteasome system. Sci. Rep. 2019, 9, 9841. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Harrison, N.R.; Laroche, F.J.; Gutierrez, A.; Feng, H. Zebrafish Models of Human Leukemia: Technological Advances and Mechanistic Insights. Adv. Exp. Med. Biol. 2016, 916, 335–369. [Google Scholar]
- Hatcher, H.; Planalp, R.; Cho, J.; Torti, F.M.; Torti, S.V. Curcumin: From ancient medicine to current clinical trials. Cell. Mol. Life Sci. CMLS 2008, 65, 1631–1652. [Google Scholar] [CrossRef] [PubMed]
- Baell, J.; Walters, M.A. Chemistry: Chemical con artists foil drug discovery. Nature 2014, 513, 481–483. [Google Scholar] [CrossRef] [PubMed]
- Kasi, P.D.; Tamilselvam, R.; Skalicka-Wozniak, K.; Nabavi, S.F.; Daglia, M.; Bishayee, A.; Pazoki-Toroudi, H.; Nabavi, S.M. Molecular targets of curcumin for cancer therapy: An updated review. Tumour Biol. J. Int. Soc. Oncodevelopmental Biol. Med. 2016, 37, 13017–13028. [Google Scholar] [CrossRef]
- Nelson, K.M.; Dahlin, J.L.; Bisson, J.; Graham, J.; Pauli, G.F.; Walters, M.A. The Essential Medicinal Chemistry of Curcumin. J. Med. Chem. 2017, 60, 1620–1637. [Google Scholar] [CrossRef]
- Baell, J.B. Feeling Nature’s PAINS: Natural Products, Natural Product Drugs, and Pan Assay Interference Compounds (PAINS). J. Nat. Prod. 2016, 79, 616–628. [Google Scholar] [CrossRef] [PubMed]
- Contreras, L.; Calderon, R.I.; Varela-Ramirez, A.; Zhang, H.Y.; Quan, Y.; Das, U.; Dimmock, J.R.; Skouta, R.; Aguilera, R.J. Induction of apoptosis via proteasome inhibition in leukemia/lymphoma cells by two potent piperidones. Cell. Oncol. 2018, 41, 623–636. [Google Scholar] [CrossRef]
- Wang, X.; Mazurkiewicz, M.; Hillert, E.K.; Olofsson, M.H.; Pierrou, S.; Hillertz, P.; Gullbo, J.; Selvaraju, K.; Paulus, A.; Akhtar, S.; et al. The proteasome deubiquitinase inhibitor VLX1570 shows selectivity for ubiquitin-specific protease-14 and induces apoptosis of multiple myeloma cells. Sci. Rep. 2016, 6, 26979. [Google Scholar] [CrossRef]
- Hillert, E.-K.; Brnjic, S.; Zhang, X.; Mazurkiewicz, M.; Saei, A.A.; Mofers, A.; Selvaraju, K.; Zubarev, R.; Linder, S.; D’Arcy, P. Proteasome inhibitor b-AP15 induces enhanced proteotoxicity by inhibiting cytoprotective aggresome formation. Cancer Lett. 2019, 448, 70–83. [Google Scholar] [CrossRef] [Green Version]
- Bucciantini, M.; Giannoni, E.; Chiti, F.; Baroni, F.; Formigli, L.; Zurdo, J.; Taddei, N.; Ramponi, G.; Dobson, C.M.; Stefani, M. Inherent toxicity of aggregates implies a common mechanism for protein misfolding diseases. Nature 2002, 416, 507–511. [Google Scholar] [CrossRef]
- Bernard, K.; Wang, W.; Narlawar, R.; Schmidt, B.; Kirk, K.L. Curcumin cross-links cystic fibrosis transmembrane conductance regulator (CFTR) polypeptides and potentiates CFTR channel activity by distinct mechanisms. J. Biol. Chem. 2009, 284, 30754–30765. [Google Scholar] [CrossRef] [Green Version]
- Brown, C.J.; Cheok, C.F.; Verma, C.S.; Lane, D.P. Reactivation of p53: From peptides to small molecules. Trends Pharmacol. Sci. 2011, 32, 53–62. [Google Scholar] [CrossRef]
- Madan, E.; Parker, T.M.; Bauer, M.R.; Dhiman, A.; Pelham, C.J.; Nagane, M.; Kuppusamy, M.L.; Holmes, M.; Holmes, T.R.; Shaik, K.; et al. The curcumin analog HO-3867 selectively kills cancer cells by converting mutant p53 protein to transcriptionally active wildtype p53. J. Biol. Chem. 2018, 293, 4262–4276. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, C.Y.; Hsieh, F.S.; Chu, P.Y.; Tsai, W.C.; Huang, C.T.; Yu, Y.B.; Huang, T.T.; Ko, P.S.; Hung, M.H.; Wang, W.L.; et al. Carfilzomib induces leukaemia cell apoptosis via inhibiting ELK1/KIAA1524 (Elk-1/CIP2A) and activating PP2A not related to proteasome inhibition. Br. J. Haematol. 2017, 177, 726–740. [Google Scholar] [CrossRef] [Green Version]
- Csizmar, C.M.; Kim, D.H.; Sachs, Z. The role of the proteasome in AML. Blood Cancer J. 2016, 6, e503. [Google Scholar] [CrossRef] [Green Version]
- Mofers, A.; Perego, P.; Selvaraju, K.; Gatti, L.; Gullbo, J.; Linder, S.; D’Arcy, P. Analysis of determinants for in vitro resistance to the small molecule deubiquitinase inhibitor b-AP15. PLoS ONE 2019, 14, e0223807. [Google Scholar] [CrossRef] [PubMed] [Green Version]
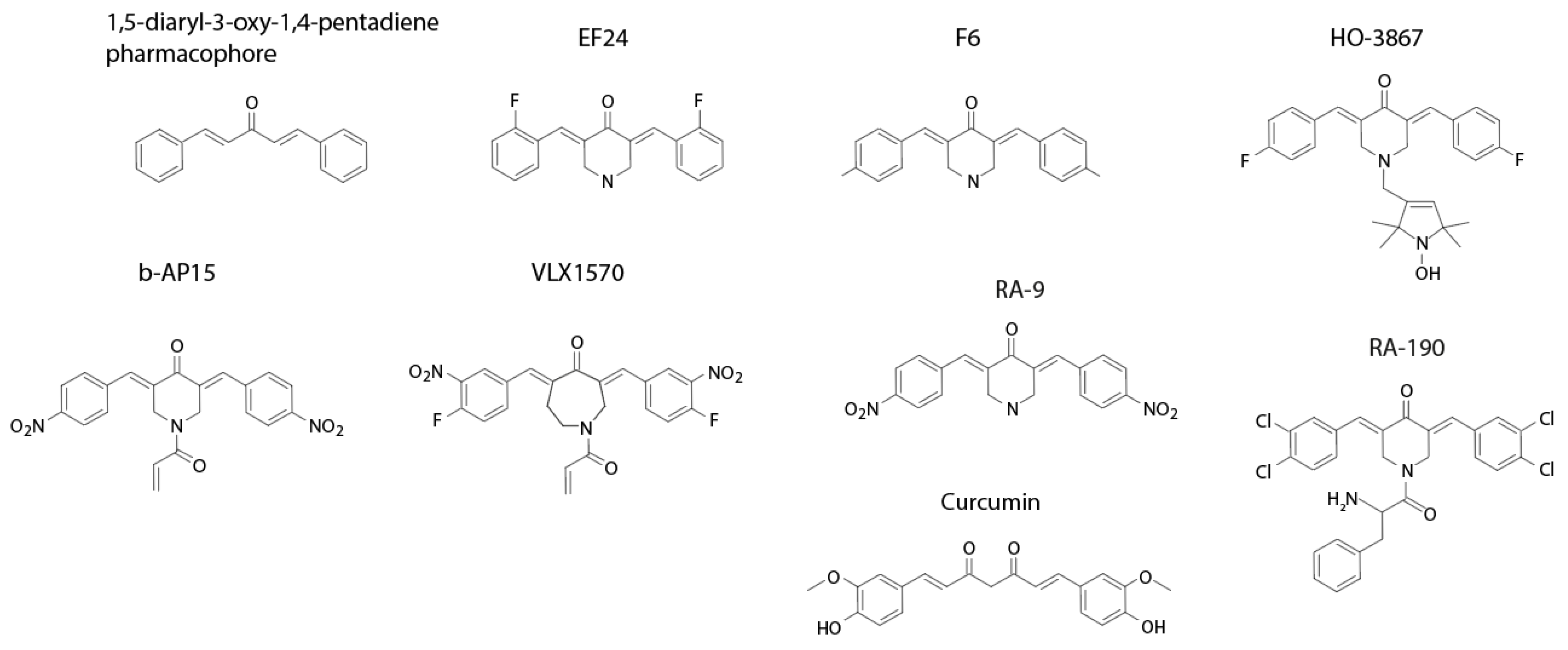





| Compound | Described Target(s) | Calculated Solubility a | PAINS b | LogP |
|---|---|---|---|---|
| b-AP15 | USP14/UCHL5 | −4.43 | yes | 2.08 |
| VLX1570 | USP14/UCHL5 | −5.14 | yes | 2.91 |
| RA-9 | USP14/UCHL5 | −3.95 | yes | 1.75 |
| RA-190 | Rpn13/UCHL5 | −8.22 | yes | 5.49 |
| F6 (NSC632839) | Deubiquitinases | −5.15 | yes | 3.60 |
| EF24 | NFkB, MAPK, HIF-1α | −4.86 | yes | 3.62 |
| Curcumin | Multiple targets | −3.94 | no | 3.03 |
| NSC-632839 | Curcumin | |||
|---|---|---|---|---|
| Rank | Gene | Description | Gene | Description |
| 1 | PSMA1 | Proteasome subunit | TP53RK | Bud32 family |
| 2 | PSMD1 | Proteasome subunit | CD46 | CD molecules |
| 3 | PSMA3 | Proteasome subunit | ZNF418 | Zinc finger |
| 4 | PSMD3 | Proteasome subunit | KLF2 | Kruppel-like transcription factor |
| 5 | PSMB2 | Proteasome subunit | RGS18 | Regulator of G-protein signaling |
| 6 | VCP | AAA ATPase | PI3KCB | Phosphatidylinositol kinase |
| 7 | PSMB5 | Proteasome subunit | ZNF350 | Zinc finger |
| 8 | HSPA5 | Heat Shock Protein | ZNF67 | Zinc finger |
| 9 | CEBPG | Basic Leucine Zipper | ATP6V0A1 | V-ATPase |
| 10 | RYK | Type XV RTK | ACACA | Carboxylase |
| Cell Line | Cell Line Properties | VLX1570 IC50 (nM) |
|---|---|---|
| KG1a | Complex karyotype [11], p53mut [29] | 307 ± 31 |
| MOLM-14 | 49,<2n>,XY,+6,+8, ins(11;9)(q23;p22p23), del(14)(q13.2q31.3) fusion gene MLL-AF9 [30], p53wt [30,31] | 204 ± 73 |
| HNT34 | 46,XX, t(3;3)(q21;q26), t(9;22)(q34;q11),20q− [32] | 273 ± 61 |
| Kasumi-1 | Human hypodiploid karyotype—45,<2n>,X,−Y,−9,−13,−16, +3mar, t(8;21)(q22;q22), der(9)t(9;?)(p22;?), der(15)t(?9;15)((?q11;?p11) [33] | 260 ± 27 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Selvaraju, K.; Lotfi, K.; Gubat, J.; Miquel, M.; Nilsson, A.; Hill, J.; Jensen, L.D.; Linder, S.; D’Arcy, P. Sensitivity of Acute Myelocytic Leukemia Cells to the Dienone Compound VLX1570 Is Associated with Inhibition of the Ubiquitin-Proteasome System. Biomolecules 2021, 11, 1339. https://doi.org/10.3390/biom11091339
Selvaraju K, Lotfi K, Gubat J, Miquel M, Nilsson A, Hill J, Jensen LD, Linder S, D’Arcy P. Sensitivity of Acute Myelocytic Leukemia Cells to the Dienone Compound VLX1570 Is Associated with Inhibition of the Ubiquitin-Proteasome System. Biomolecules. 2021; 11(9):1339. https://doi.org/10.3390/biom11091339
Chicago/Turabian StyleSelvaraju, Karthik, Kourosh Lotfi, Johannes Gubat, Maria Miquel, Amanda Nilsson, Julia Hill, Lasse D. Jensen, Stig Linder, and Pádraig D’Arcy. 2021. "Sensitivity of Acute Myelocytic Leukemia Cells to the Dienone Compound VLX1570 Is Associated with Inhibition of the Ubiquitin-Proteasome System" Biomolecules 11, no. 9: 1339. https://doi.org/10.3390/biom11091339






