Effect of Human Platelet Lysate as Cultivation Nutrient Supplement on Human Natal Dental Pulp Stem Cell In Vitro Expansion
Abstract
:1. Introduction
2. Materials and Methods
2.1. Natal Teeth Collection and hNDP-SCs Isolation
2.2. Culture Media
2.3. Effect on hNDP-SC Proliferation and Viability
2.4. Effect on hNDP-SC Phenotype Profile and Specific Factor Expressions
2.5. Effect on Relative Telomere Length
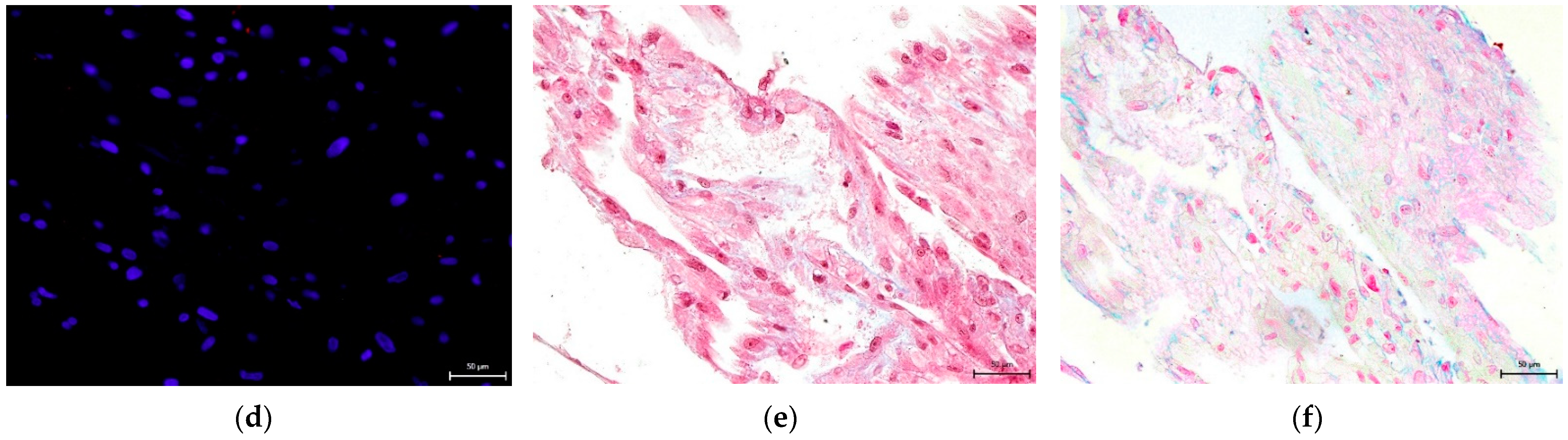
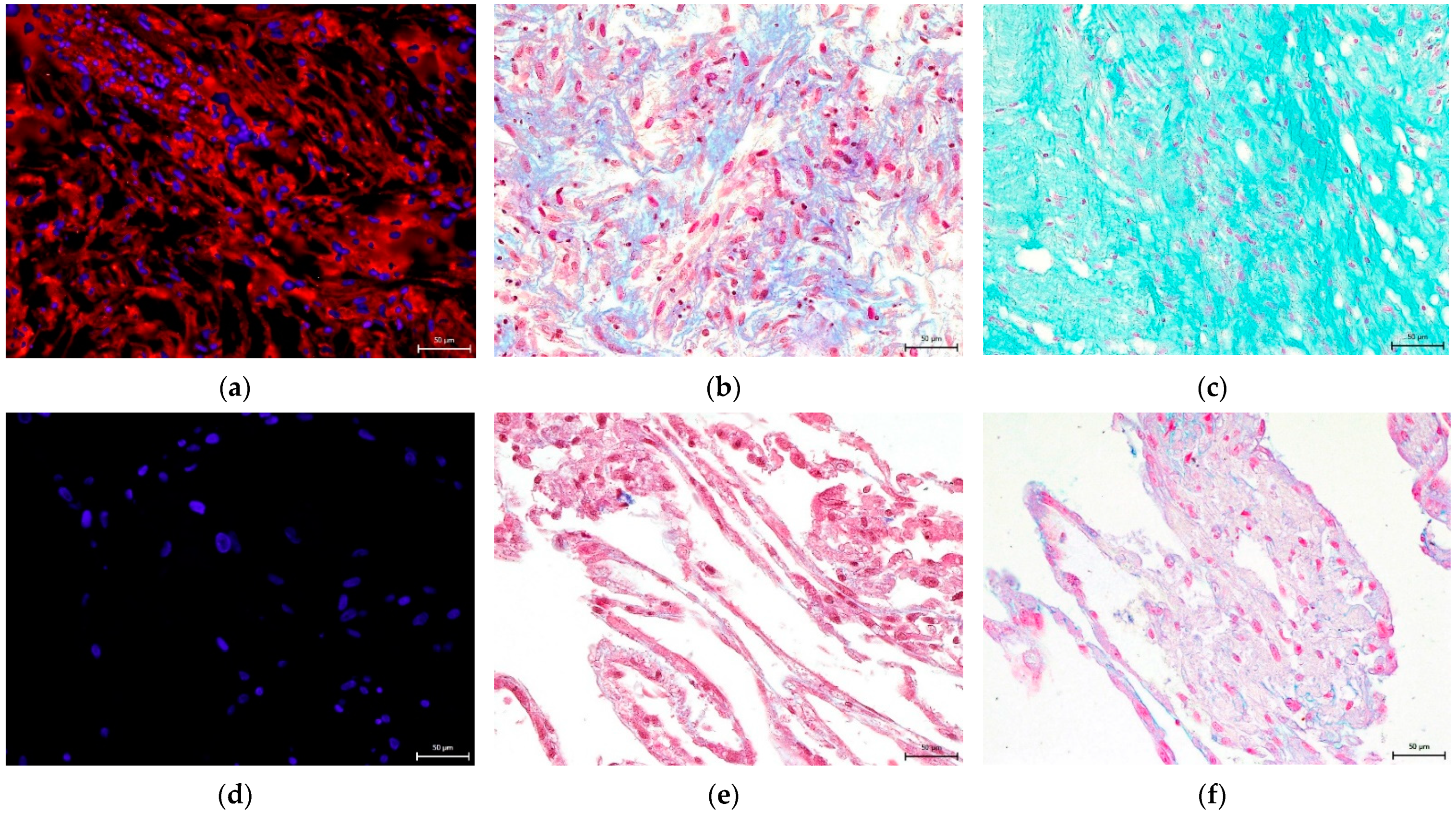
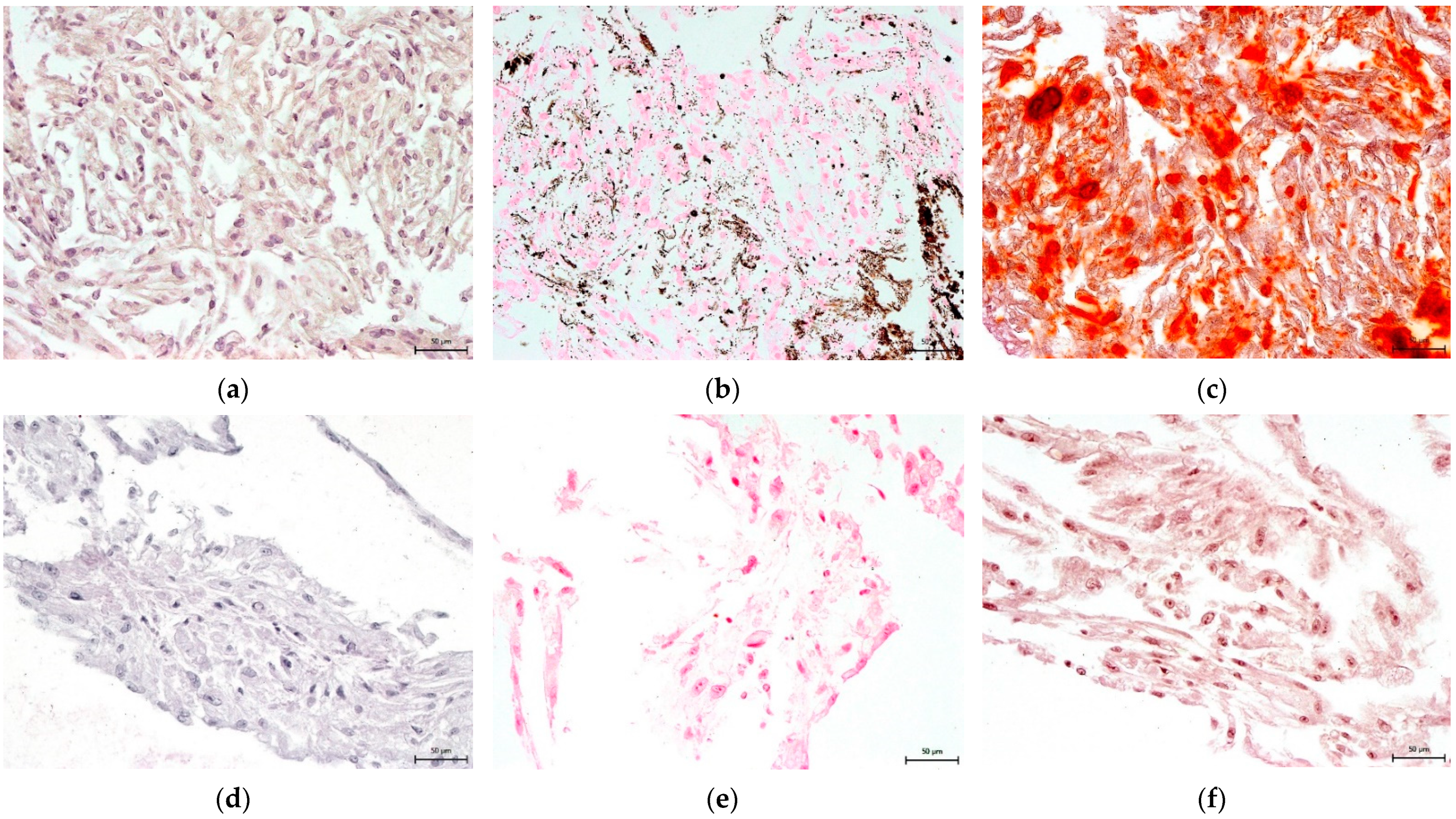
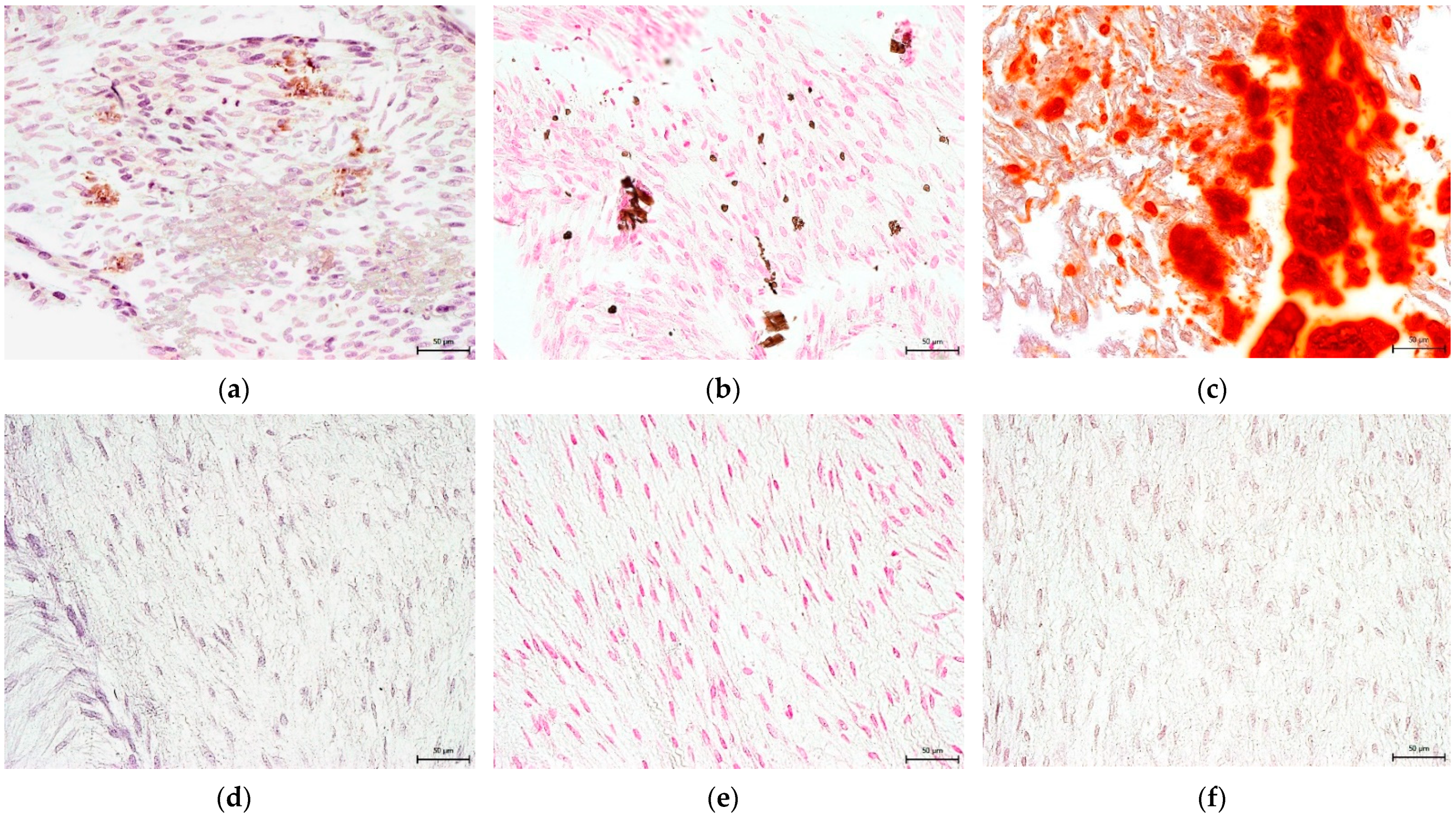
2.6. Effect on hNDP-SC Multipotency
2.6.1. Osteogenic Differentiation In Vitro
2.6.2. Chondrogenic Differentiation In Vitro
2.6.3. Adipogenic Differentiation In Vitro
2.7. Statistical Analysis
3. Results
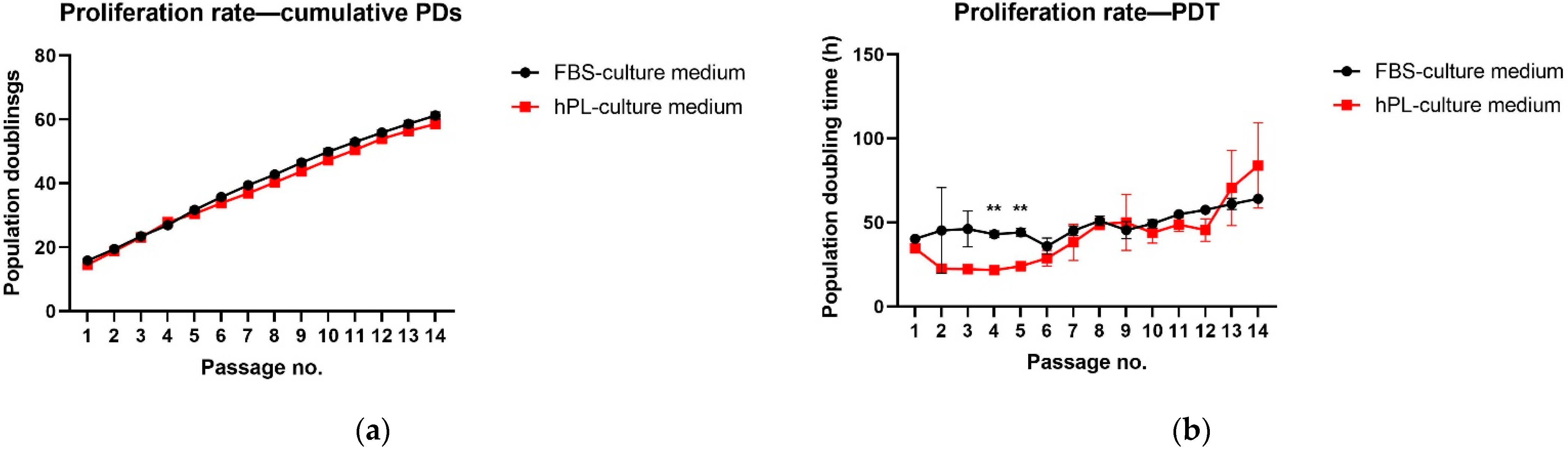
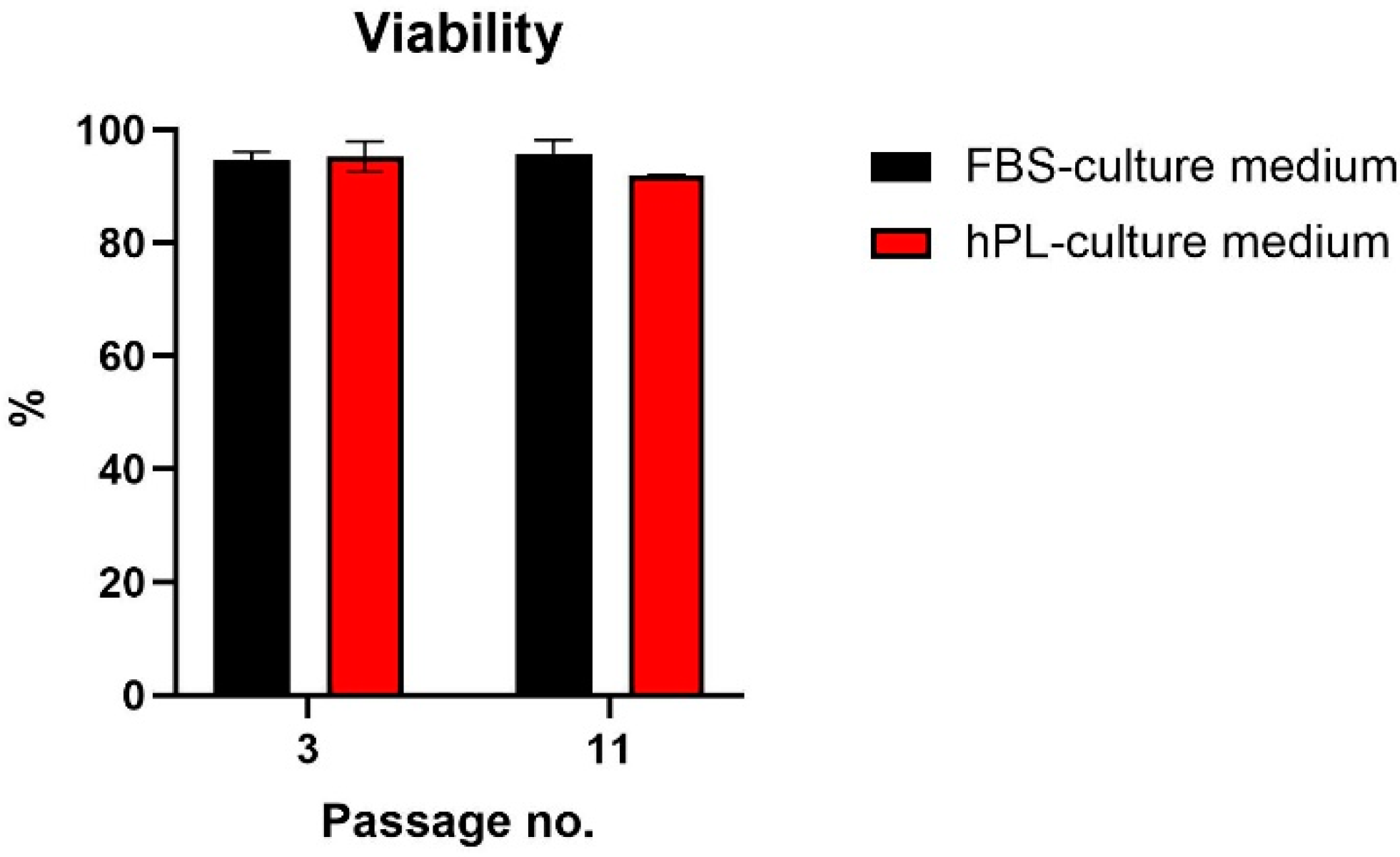
3.1. Effect on hNDP-SC Proliferation and Viability
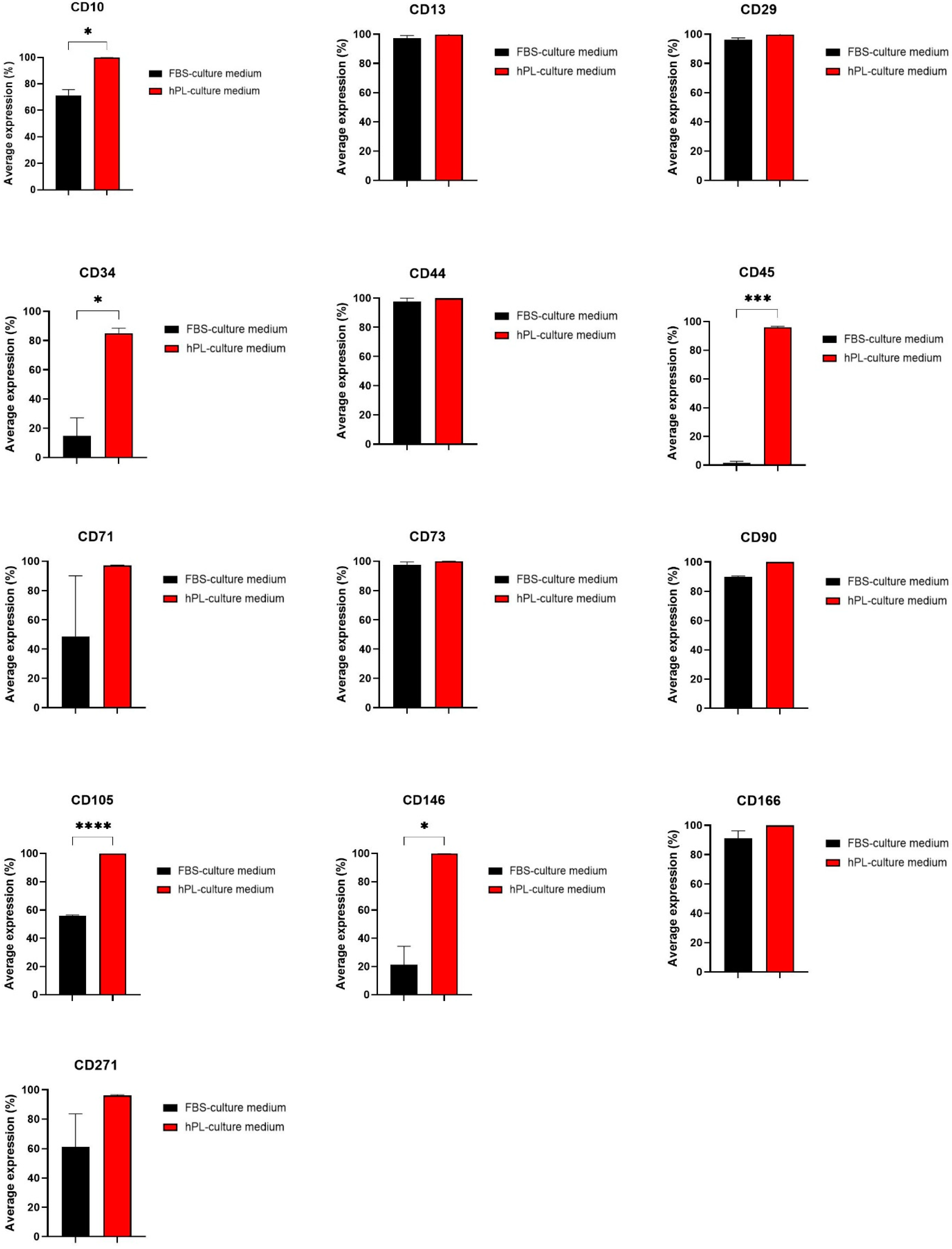
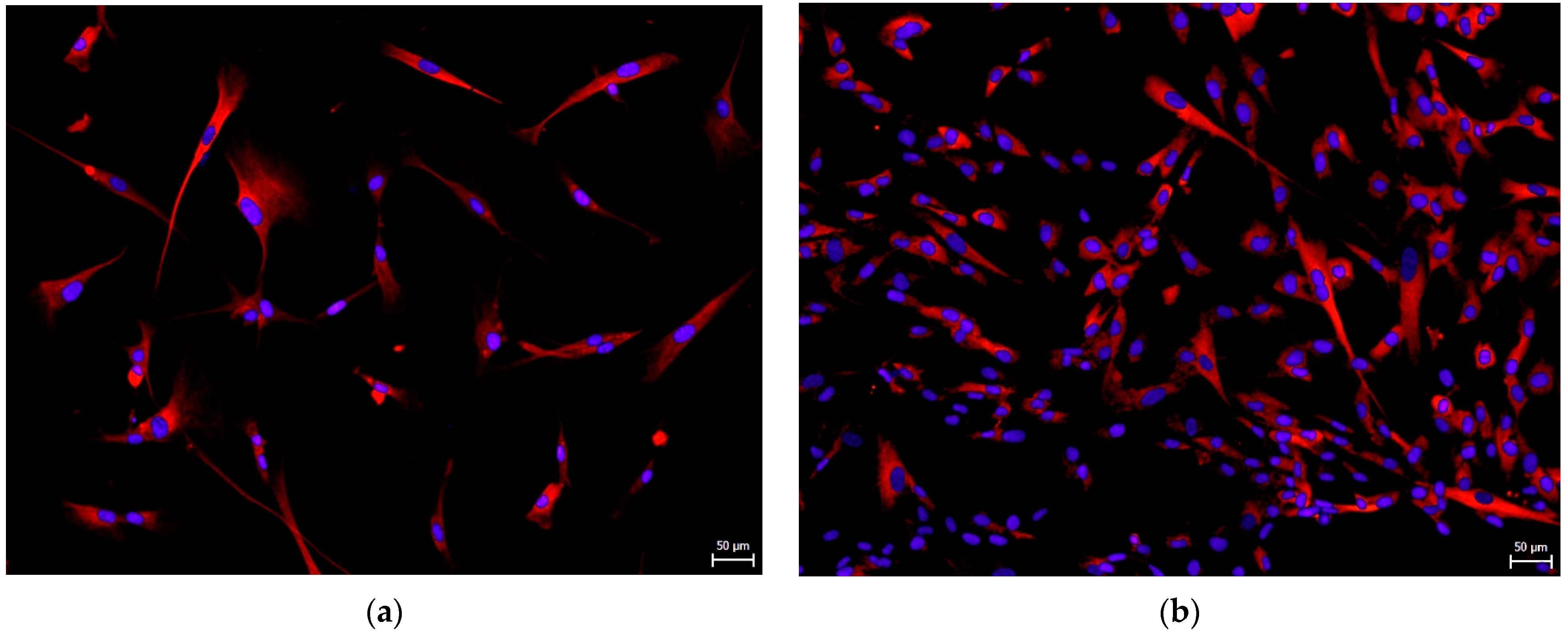
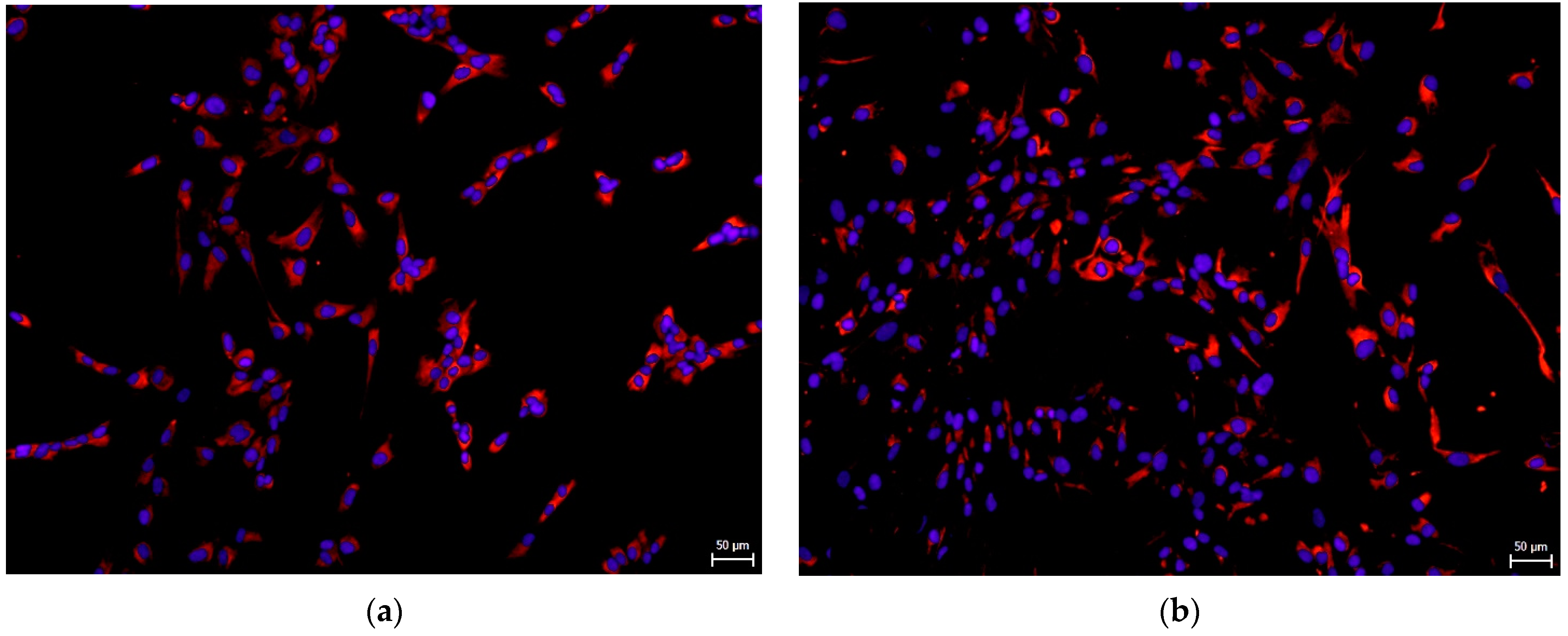
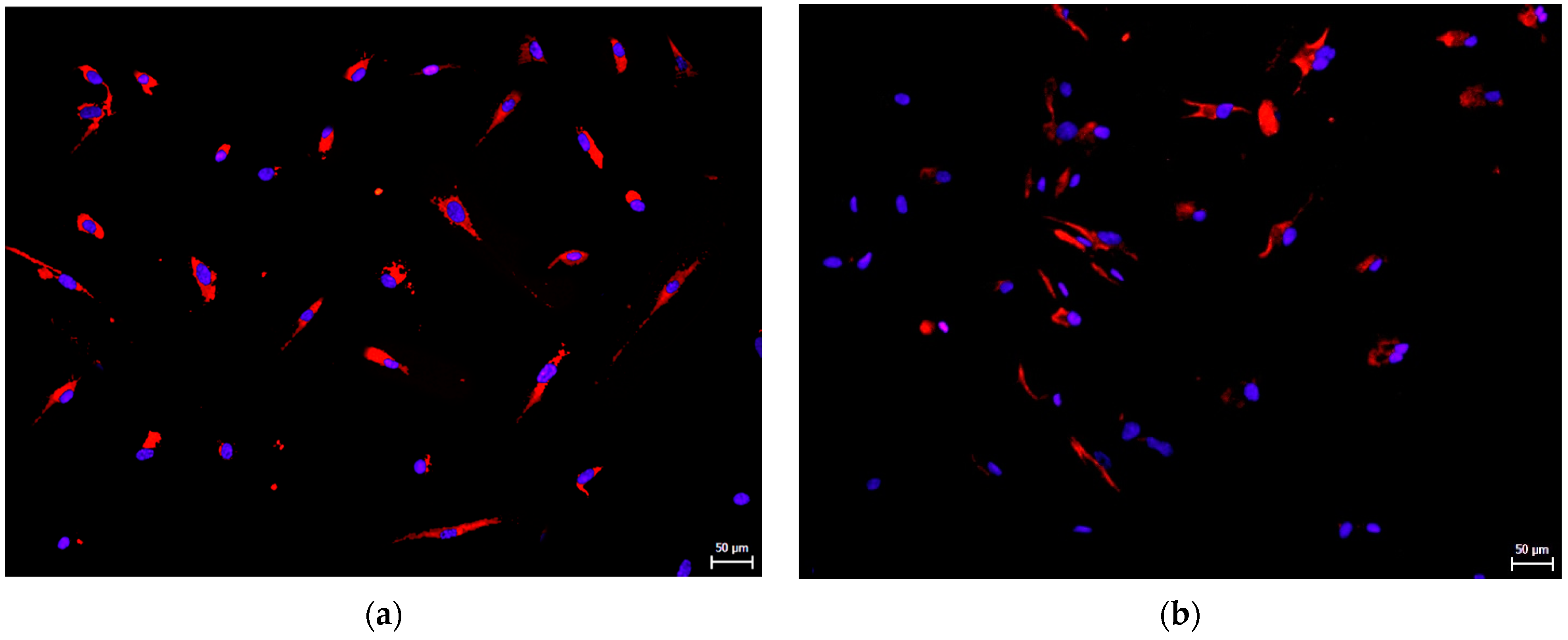
3.2. Effect on hNDP-SC Phenotype Profile and Specific Factor Expressions
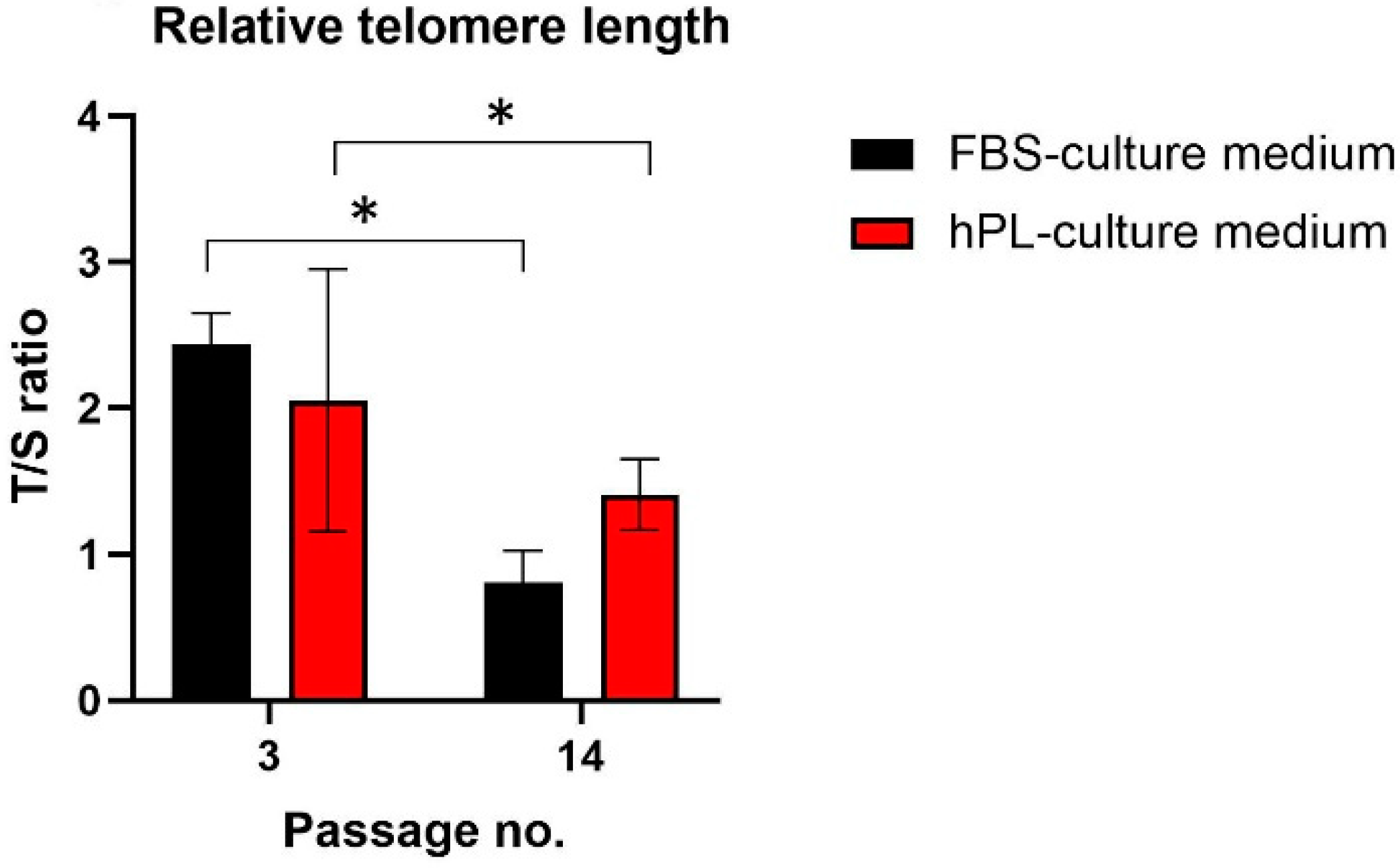
3.3. Effect on Relative Telomere Length
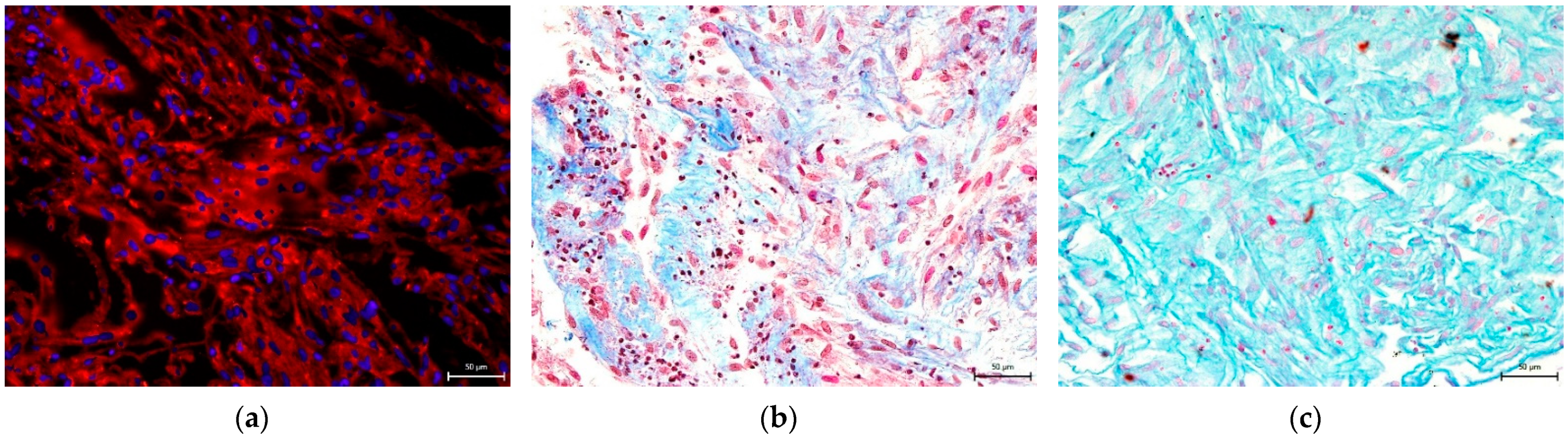
3.4. Effect on hNDP-SC Multipotency
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Mhaske, S.; Yuwanati, M.B.; Mhaske, A.; Ragavendra, R.; Kamath, K.; Saawarn, S. Natal and neonatal teeth: An overview of the literature. ISRN Pediatr. 2013, 2013, 956269. [Google Scholar] [CrossRef] [Green Version]
- Leung, A.K.; Robson, W.L. Natal teeth: A review. J. Natl. Med. Assoc. 2006, 98, 226–228. [Google Scholar]
- Leung, A.K. Natal teeth. Am. J. Dis. Child. 1986, 140, 249–251. [Google Scholar] [CrossRef]
- Seminario, A.L.; Ivancaková, R. Natal and neonatal teeth. Acta Med. 2004, 47, 229–233. [Google Scholar] [CrossRef] [Green Version]
- Kates, G.A.; Needleman, H.L.; Holmes, L.B. Natal and neonatal teeth: A clinical study. J. Am. Dent. Assoc. 1984, 109, 441–443. [Google Scholar] [CrossRef]
- Sureshkumar, R.; McAulay, A.H. Natal and neonatal teeth. Arch. Dis. Child. Fetal Neonatal Ed. 2002, 87, F227. [Google Scholar] [CrossRef] [Green Version]
- Karaöz, E.; Doğan, B.N.; Aksoy, A.; Gacar, G.; Akyüz, S.; Ayhan, S.; Genç, Z.S.; Yürüker, S.; Duruksu, G.; Demircan, P.C.; et al. Isolation and in vitro characterisation of dental pulp stem cells from natal teeth. Histochem. Cell Biol. 2010, 133, 95–112. [Google Scholar] [CrossRef]
- Akpinar, G.; Kasap, M.; Aksoy, A.; Duruksu, G.; Gacar, G.; Karaoz, E. Phenotypic and proteomic characteristics of human dental pulp derived mesenchymal stem cells from a natal, an exfoliated deciduous, and an impacted third molar tooth. Stem Cells Int. 2014, 2014, 457059. [Google Scholar] [CrossRef]
- van der Valk, J.; Bieback, K.; Buta, C.; Cochrane, B.; Dirks, W.G.; Fu, J.; Hickman, J.J.; Hohensee, C.; Kolar, R.; Liebsch, M.; et al. Fetal Bovine Serum (FBS): Past—Present—Future. Altex 2018, 35, 99–118. [Google Scholar] [CrossRef] [Green Version]
- Heiskanen, A.; Satomaa, T.; Tiitinen, S.; Laitinen, A.; Mannelin, S.; Impola, U.; Mikkola, M.; Olsson, C.; Miller-Podraza, H.; Blomqvist, M.; et al. N-glycolylneuraminic acid xenoantigen contamination of human embryonic and mesenchymal stem cells is substantially reversible. Stem Cells 2007, 25, 197–202. [Google Scholar] [CrossRef]
- Sundin, M.; Ringdén, O.; Sundberg, B.; Nava, S.; Götherström, C.; Le Blanc, K. No alloantibodies against mesenchymal stromal cells, but presence of anti-fetal calf serum antibodies, after transplantation in allogeneic hematopoietic stem cell recipients. Haematologica 2007, 92, 1208–1215. [Google Scholar] [CrossRef]
- Lang, S.; Loibl, M.; Herrmann, M. Platelet-Rich Plasma in Tissue Engineering: Hype and Hope. Eur. Surg. Res. 2018, 59, 265–275. [Google Scholar] [CrossRef]
- Hawkes, P.W. Fetal bovine serum: Geographic origin and regulatory relevance of viral contamination. Bioresour. Bioprocess. 2015, 2, 34. [Google Scholar] [CrossRef] [Green Version]
- Bieback, K. Platelet lysate as replacement for fetal bovine serum in mesenchymal stromal cell cultures. Transfus. Med. HemoTher. 2013, 40, 326–335. [Google Scholar] [CrossRef] [Green Version]
- Burnouf, T.; Strunk, D.; Koh, M.B.; Schallmoser, K. Human platelet lysate: Replacing fetal bovine serum as a gold standard for human cell propagation? Biomaterials 2016, 76, 371–387. [Google Scholar] [CrossRef]
- Doucet, C.; Ernou, I.; Zhang, Y.; Llense, J.R.; Begot, L.; Holy, X.; Lataillade, J.J. Platelet lysates promote mesenchymal stem cell expansion: A safety substitute for animal serum in cell-based therapy applications. J. Cell. Physiol. 2005, 205, 228–236. [Google Scholar] [CrossRef]
- Schallmoser, K.; Bartmann, C.; Rohde, E.; Reinisch, A.; Kashofer, K.; Stadelmeyer, E.; Drexler, C.; Lanzer, G.; Linkesch, W.; Strunk, D. Human platelet lysate can replace fetal bovine serum for clinical-scale expansion of functional mesenchymal stromal cells. Transfusion 2007, 47, 1436–1446. [Google Scholar] [CrossRef]
- Marx, R.E.; Carlson, E.R.; Eichstaedt, R.M.; Schimmele, S.R.; Strauss, J.E.; Georgeff, K.R. Platelet-rich plasma: Growth factor enhancement for bone grafts. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endodontol. 1998, 85, 638–646. [Google Scholar] [CrossRef]
- Blair, P.; Flaumenhaft, R. Platelet alpha-granules: Basic biology and clinical correlates. Blood Rev. 2009, 23, 177–189. [Google Scholar] [CrossRef] [Green Version]
- Kettunen, P.; Karavanova, I.; Thesleff, I. Responsiveness of developing dental tissues to fibroblast growth factors: Expression of splicing alternatives of FGFR1, -2, -3, and of FGFR4; and stimulation of cell proliferation by FGF-2, -4, -8, and -9. Dev. Genet. 1998, 22, 374–385. [Google Scholar] [CrossRef]
- Shiba, H.; Fujita, T.; Doi, N.; Nakamura, S.; Nakanishi, K.; Takemoto, T.; Hino, T.; Noshiro, M.; Kawamoto, T.; Kurihara, H.; et al. Differential effects of various growth factors and cytokines on the syntheses of DNA, type I collagen, laminin, fibronectin, osteonectin/secreted protein, acidic and rich in cysteine (SPARC), and alkaline phosphatase by human pulp cells in culture. J. Cell. Physiol. 1998, 174, 194–205. [Google Scholar] [CrossRef]
- Onishi, T.; Kinoshita, S.; Shintani, S.; Sobue, S.; Ooshima, T. Stimulation of proliferation and differentiation of dog dental pulp cells in serum-free culture medium by insulin-like growth factor. Arch. Oral Biol. 1999, 44, 361–371. [Google Scholar] [CrossRef]
- Marcopoulou, C.E.; Vavouraki, H.N.; Dereka, X.E.; Vrotsos, I.A. Proliferative effect of growth factors TGF-beta1, PDGF-BB and rhBMP-2 on human gingival fibroblasts and periodontal ligament cells. J. Int. Acad. Periodontol. 2003, 5, 63–70. [Google Scholar]
- Fekete, N.; Gadelorge, M.; Fürst, D.; Maurer, C.; Dausend, J.; Fleury-Cappellesso, S.; Mailänder, V.; Lotfi, R.; Ignatius, A.; Sensebé, L.; et al. Platelet lysate from whole blood-derived pooled platelet concentrates and apheresis-derived platelet concentrates for the isolation and expansion of human bone marrow mesenchymal stromal cells: Production process, content and identification of active components. Cytotherapy 2012, 14, 540–554. [Google Scholar] [CrossRef] [Green Version]
- Griffiths, S.; Baraniak, P.R.; Copland, I.B.; Nerem, R.M.; McDevitt, T.C. Human platelet lysate stimulates high-passage and senescent human multipotent mesenchymal stromal cell growth and rejuvenation in vitro. Cytotherapy 2013, 15, 1469–1483. [Google Scholar] [CrossRef]
- Hemeda, H.; Giebel, B.; Wagner, W. Evaluation of human platelet lysate versus fetal bovine serum for culture of mesenchymal stromal cells. Cytotherapy 2014, 16, 170–180. [Google Scholar] [CrossRef] [Green Version]
- Pilbauerova, N.; Soukup, T.; Suchankova Kleplova, T.; Suchanek, J. Enzymatic Isolation, Amplification and Characterization of Dental Pulp Stem Cells. Folia Biol. 2019, 65, 124–133. [Google Scholar]
- Mokry, J.; Soukup, T.; Micuda, S.; Karbanova, J.; Visek, B.; Brcakova, E.; Suchanek, J.; Bouchal, J.; Vokurkova, D.; Ivancakova, R. Telomere attrition occurs during ex vivo expansion of human dental pulp stem cells. J. Biomed. Biotechnol. 2010, 2010, 673513. [Google Scholar] [CrossRef]
- Pilbauerova, N.; Soukup, T.; Suchankova Kleplova, T.; Schmidt, J.; Suchanek, J. The Effect of Cultivation Passaging on the Relative Telomere Length and Proliferation Capacity of Dental Pulp Stem Cells. Biomolecules 2021, 11, 464. [Google Scholar] [CrossRef]
- Foudah, D.; Monfrini, M.; Donzelli, E.; Niada, S.; Brini, A.T.; Orciani, M.; Tredici, G.; Miloso, M. Expression of neural markers by undifferentiated mesenchymal-like stem cells from different sources. J. Immunol. Res. 2014, 2014, 987678. [Google Scholar] [CrossRef] [Green Version]
- Pan, G.; Thomson, J.A. Nanog and transcriptional networks in embryonic stem cell pluripotency. Cell Res. 2007, 17, 42–49. [Google Scholar] [CrossRef]
- Jeon, B.G.; Kang, E.J.; Kumar, B.M.; Maeng, G.H.; Ock, S.A.; Kwack, D.O.; Park, B.W.; Rho, G.J. Comparative analysis of telomere length, telomerase and reverse transcriptase activity in human dental stem cells. Cell Transplant. 2011, 20, 1693–1705. [Google Scholar] [CrossRef]
- Serakinci, N.; Graakjaer, J.; Kolvraa, S. Telomere stability and telomerase in mesenchymal stem cells. Biochimie 2008, 90, 33–40. [Google Scholar] [CrossRef]
- Guiotto, M.; Raffoul, W.; Hart, A.M.; Riehle, M.O.; di Summa, P.G. Human platelet lysate to substitute fetal bovine serum in hMSC expansion for translational applications: A systematic review. J. Transl. Med. 2020, 18, 351. [Google Scholar] [CrossRef]
- Bieback, K.; Hecker, A.; Kocaömer, A.; Lannert, H.; Schallmoser, K.; Strunk, D.; Klüter, H. Human alternatives to fetal bovine serum for the expansion of mesenchymal stromal cells from bone marrow. Stem Cells 2009, 27, 2331–2341. [Google Scholar] [CrossRef]
- Li, J.; Xiang, L.; Guan, C.; Yang, X.; Hu, X.; Zhang, X.; Zhang, W. Effects of Platelet-Rich Plasma on Proliferation, Viability, and Odontogenic Differentiation of Neural Crest Stem-like Cells Derived from Human Dental Apical Papilla. Biomed. Res. Int. 2020, 2020, 4671989. [Google Scholar] [CrossRef]
- Lee, U.L.; Jeon, S.H.; Park, J.Y.; Choung, P.H. Effect of platelet-rich plasma on dental stem cells derived from human impacted third molars. Regen. Med. 2011, 6, 67–79. [Google Scholar] [CrossRef]
- Stute, N.; Holtz, K.; Bubenheim, M.; Lange, C.; Blake, F.; Zander, A.R. Autologous serum for isolation and expansion of human mesenchymal stem cells for clinical use. Exp. Hematol. 2004, 32, 1212–1225. [Google Scholar] [CrossRef]
- Suchanek, J.; Browne, K.Z.; Nasry, S.A.; Kleplova, T.S.; Pilbauerova, N.; Schmidt, J.; Soukup, T. Characteristics of Human Natal Stem Cells Cultured in Allogeneic Medium. Braz. Dent. J. 2018, 29, 427–434. [Google Scholar] [CrossRef] [Green Version]
- Ballesteros, O.R.; Brooks, P.T.; Haastrup, E.K.; Fischer-Nielsen, A.; Munthe-Fog, L.; Svalgaard, J.D. Adipose-Derived Stromal/Stem Cell Culture: Effects of Different Concentrations of Human Platelet Lysate in Media. Cells Tissues Organs 2020, 209, 257–265. [Google Scholar] [CrossRef]
- Chen, B.; Sun, H.H.; Wang, H.G.; Kong, H.; Chen, F.M.; Yu, Q. The effects of human platelet lysate on dental pulp stem cells derived from impacted human third molars. Biomaterials 2012, 33, 5023–5035. [Google Scholar] [CrossRef]
- Suchánková Kleplová, T.; Soukup, T.; Řeháček, V.; Suchánek, J. Human plasma and human platelet-rich plasma as a substitute for fetal calf serum during long-term cultivation of mesenchymal dental pulp stem cells. Acta Med. (Hradec Kralove) 2014, 57, 119–126. [Google Scholar] [CrossRef] [Green Version]
- Karadjian, M.; Senger, A.S.; Essers, C.; Wilkesmann, S.; Heller, R.; Fellenberg, J.; Simon, R.; Westhauser, F. Human Platelet Lysate Can Replace Fetal Calf Serum as a Protein Source to Promote Expansion and Osteogenic Differentiation of Human Bone-Marrow-Derived Mesenchymal Stromal Cells. Cells 2020, 9, 918. [Google Scholar] [CrossRef] [Green Version]
- Vennila, R.; Raja Sundari, M.S.; Selvaraj, S.; Srinivasan, P.; Pathak, S.; Rupert, S.; Rajagopal, S. Effect of Human Platelet Lysate in Differentiation of Wharton’s Jelly Derived Mesenchymal Stem Cells. Endocr. Metab. Immune Disord. Drug Targets 2019, 19, 1177–1191. [Google Scholar] [CrossRef]
- Muraglia, A.; Todeschi, M.R.; Papait, A.; Poggi, A.; Spanò, R.; Strada, P.; Cancedda, R.; Mastrogiacomo, M. Combined platelet and plasma derivatives enhance proliferation of stem/progenitor cells maintaining their differentiation potential. Cytotherapy 2015, 17, 1793–1806. [Google Scholar] [CrossRef]
- Mohammadi, S.; Nikbakht, M.; Malek Mohammadi, A.; Zahed Panah, M.; Ostadali, M.R.; Nasiri, H.; Ghavamzadeh, A. Human Platelet Lysate as a Xeno Free Alternative of Fetal Bovine Serum for the In Vitro Expansion of Human Mesenchymal Stromal Cells. Int. J. Hematol. Oncol. Stem Cell Res. 2016, 10, 161–171. [Google Scholar]
- Ding, L.; Vezzani, B.; Khan, N.; Su, J.; Xu, L.; Yan, G.; Liu, Y.; Li, R.; Gaur, A.; Diao, Z.; et al. CD10 expression identifies a subset of human perivascular progenitor cells with high proliferation and calcification potentials. Stem Cells 2020, 38, 261–275. [Google Scholar] [CrossRef]
- Lin, C.S.; Ning, H.; Lin, G.; Lue, T.F. Is CD34 truly a negative marker for mesenchymal stromal cells? Cytotherapy 2012, 14, 1159–1163. [Google Scholar] [CrossRef] [Green Version]
- Lin, C.S.; Xin, Z.C.; Dai, J.; Lue, T.F. Commonly used mesenchymal stem cell markers and tracking labels: Limitations and challenges. Histol. Histopathol. 2013, 28, 1109–1116. [Google Scholar] [CrossRef]
- Yeh, S.P.; Chang, J.G.; Lo, W.J.; Liaw, Y.C.; Lin, C.L.; Lee, C.C.; Chiu, C.F. Induction of CD45 expression on bone marrow-derived mesenchymal stem cells. Leukemia 2006, 20, 894–896. [Google Scholar] [CrossRef] [Green Version]
- Ma, L.; Huang, Z.; Wu, D.; Kou, X.; Mao, X.; Shi, S. CD146 controls the quality of clinical grade mesenchymal stem cells from human dental pulp. Stem Cell. Res. Ther. 2021, 12, 488. [Google Scholar] [CrossRef]
- Kulikova, B.; Kovac, M.; Bauer, M.; Tomkova, M.; Olexikova, L.; Vasicek, J.; Balazi, A.; Makarevich, A.V.; Chrenek, P. Survivability of rabbit amniotic fluid-derived mesenchymal stem cells post slow-freezing or vitrification. Acta Histochem. 2019, 121, 491–499. [Google Scholar] [CrossRef]
- Stanko, P.; Altanerova, U.; Jakubechova, J.; Repiska, V.; Altaner, C. Dental Mesenchymal Stem/Stromal Cells and Their Exosomes. Stem Cells Int. 2018, 2018, 8973613. [Google Scholar] [CrossRef] [Green Version]


| Components | FBS-Culture Medium | hPL-Culture Medium |
|---|---|---|
| Eagle Minimum Essential Medium Alpha (α-Mem, Sigma-Aldrich) | 94.96 mL | 94.96 mL |
| Fetal bovine serum (PAA Laboratories, Dartmouth, MA, USA) | 1.94 mL (2%) | - |
| human Platelet lysate (Transfusion Department, University Hospital Hradec Kralove, Czech Republic) | - | 1.94 mL (2%) |
| Insulin-Transferrin-Selenium (ITS, Invitrogen) | 10 µL/mL | 10 µL/mL |
| EPGF (PeproTech, London, UK) | 10 ng/mL | 10 ng/mL |
| PDGF (PeproTech) | 10 ng/mL | 10 ng/mL |
| Dexamethasone (Bieffe Medital) | 8 µL/mL | 8 µL/mL |
| L-ascorbic acid (Bieffe Medital) | 1 mL | 1 mL |
| Glutamine (Invitrogen) | 1.9 mL | 1.9 mL |
| Streptomycin/Penicillin (Invitrogen) | 0.6 mL | 0.6 mL |
| Gentamycin (Invitrogen) | 0.5 mL | 0.5 mL |
| Amphotericin (Sigma-Aldrich) | 2.5 µg/mL | 2.5 µg/mL |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pilbauerova, N.; Schmidt, J.; Suchankova Kleplova, T.; Soukup, T.; Suchanek, J. Effect of Human Platelet Lysate as Cultivation Nutrient Supplement on Human Natal Dental Pulp Stem Cell In Vitro Expansion. Biomolecules 2022, 12, 1091. https://doi.org/10.3390/biom12081091
Pilbauerova N, Schmidt J, Suchankova Kleplova T, Soukup T, Suchanek J. Effect of Human Platelet Lysate as Cultivation Nutrient Supplement on Human Natal Dental Pulp Stem Cell In Vitro Expansion. Biomolecules. 2022; 12(8):1091. https://doi.org/10.3390/biom12081091
Chicago/Turabian StylePilbauerova, Nela, Jan Schmidt, Tereza Suchankova Kleplova, Tomas Soukup, and Jakub Suchanek. 2022. "Effect of Human Platelet Lysate as Cultivation Nutrient Supplement on Human Natal Dental Pulp Stem Cell In Vitro Expansion" Biomolecules 12, no. 8: 1091. https://doi.org/10.3390/biom12081091










