Design of Beta-2 Microglobulin Adsorbent Protein Nanoparticles
Abstract
:1. Introduction
2. Methods
3. Results
3.1. Protein Design Approach
3.2. Testing and Design Optimization on Cages
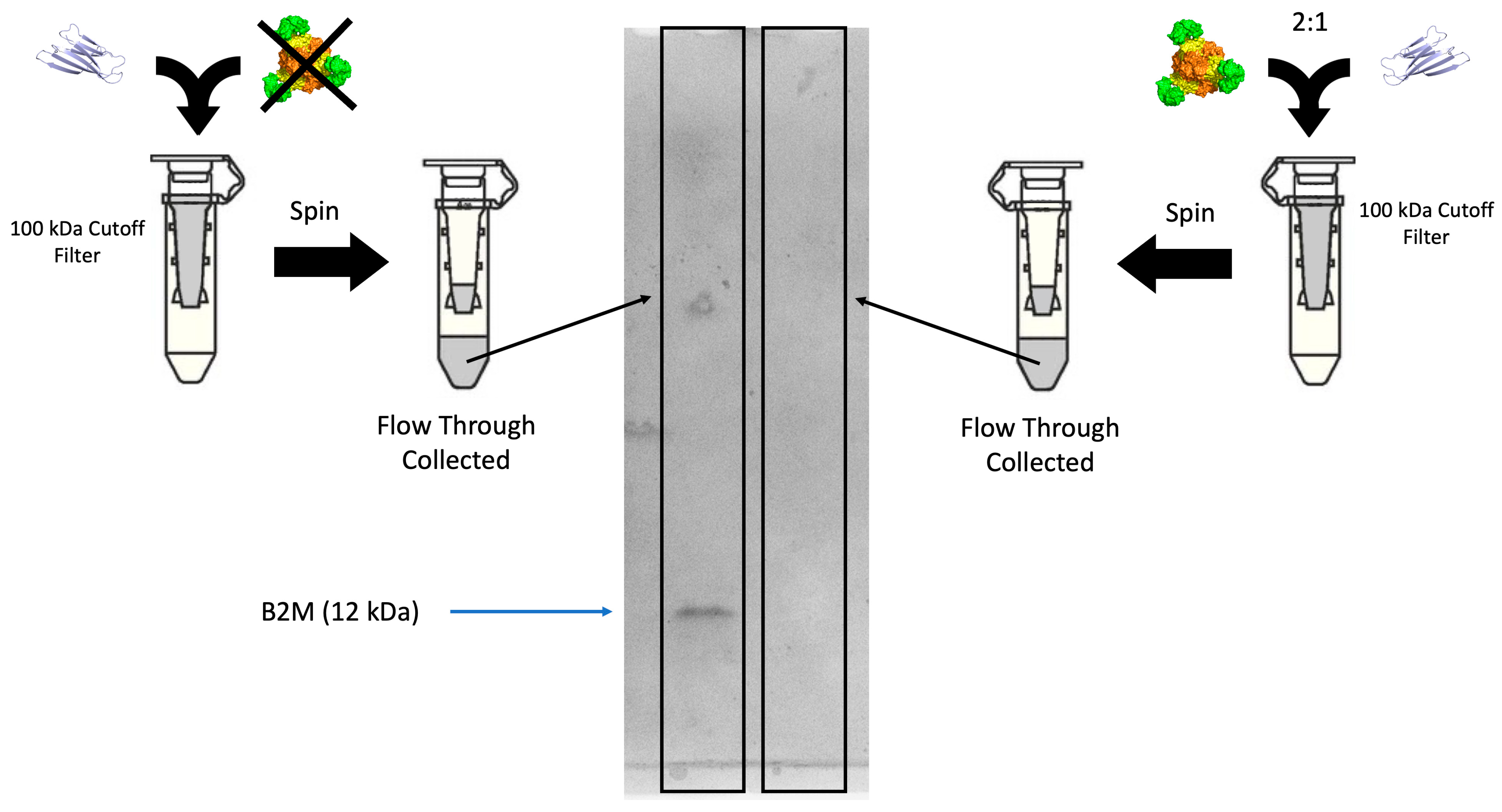
3.3. Assays on nbBAC1_LL for B2M Removal
4. Discussion
5. Patents
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Massy, Z.A.; Liabeuf, S. Middle-Molecule Uremic Toxins and Outcomes in Chronic Kidney Disease. In Contributions to Nephrology; Ronco, C., Ed.; S. Karger AG: Basel, Switzerland, 2017; Volume 191, pp. 8–17. [Google Scholar] [CrossRef]
- Eichner, T.; Kalverda, A.P.; Thompson, G.S.; Homans, S.W.; Radford, S.E. Conformational Conversion during Amyloid Formation at Atomic Resolution. Mol. Cell 2011, 41, 161–172. [Google Scholar] [CrossRef]
- Iadanza, M.G.; Silvers, R.; Boardman, J.; Smith, H.I.; Karamanos, T.K.; Debelouchina, G.T.; Su, Y.; Griffin, R.G.; Ranson, N.A.; Radford, S.E. The Structure of a Β2-Microglobulin Fibril Suggests a Molecular Basis for Its Amyloid Polymorphism. Nat. Commun. 2018, 9, 4517. [Google Scholar] [CrossRef] [Green Version]
- Winchester, J.F.; Salsberg, J.A.; Levin, N.W. Beta-2 Microglobulin in ESRD: An in-Depth Review. Adv. Ren. Replace. Ther. 2003, 10, 279–309. [Google Scholar] [CrossRef] [PubMed]
- Ohashi, K. Pathogenesis of β2-microglobulin Amyloidosis. Pathol. Int. 2001, 51, 1–10. [Google Scholar] [CrossRef]
- Floege, J.; Bartsch, A.; Schulze, M.; Shaldon, S.; Koch, K.M.; Smeby, L.C. Clearance and Synthesis Rates of Beta 2-Microglobulin in Patients Undergoing Hemodialysis and in Normal Subjects. J. Lab. Clin. Med. 1991, 118, 153–165. [Google Scholar]
- Barton, K.T.; Kakajiwala, A.; Dietzen, D.J.; Goss, C.W.; Gu, H.; Dharnidharka, V.R. Using the Newer Kidney Disease: Improving Global Outcomes Criteria, Beta-2-Microglobulin Levels Associate with Severity of Acute Kidney Injury. Clin. Kidney J. 2018, 11, 797–802. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Portales-Castillo, I.; Yee, J.; Tanaka, H.; Fenves, A.Z. Beta-2 Microglobulin Amyloidosis: Past, Present, and Future. Kidney360 2020, 1, 1447–1455. [Google Scholar] [CrossRef]
- Furuyoshi, S.; Nakatani, M.; Taman, J.; Kutsuki, H.; Takata, S.; Tani, N. New Adsorption Column (Lixelle) to Eliminate Beta2-Microglobulin for Direct Hemoperfusion. Ther. Apher. 1998, 2, 13–17. [Google Scholar] [CrossRef] [PubMed]
- Masakane, I.; Sakurai, K. Current Approaches to Middle Molecule Removal: Room for Innovation. Nephrol. Dial. Transplant. 2018, 33 (Suppl. S3), iii12–iii21. [Google Scholar] [CrossRef] [Green Version]
- Daniels, C.M.; Woolverton, E.M.; Sprague, S.M.; Ameer, G.A. Assessment of the Stability of an Immunoadsorbent for the Extracorporeal Removal of Beta-2-Microglobulin from Blood. Blood Purif. 2005, 23, 287–297. [Google Scholar] [CrossRef]
- Zhang, L.; Zang, B.; Huang, C.; Ren, J.; Jia, L. One-Step Preparation of a VHH-Based Immunoadsorbent for the Extracorporeal Removal of Β2-Microglobulin. Molecules 2019, 24, 2119. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Huang, C.; Ren, J.; Ji, F.; Muyldermans, S.; Jia, L. Nanobody-Based High-Performance Immunosorbent for Selective Beta 2-Microglobulin Purification from Blood. Acta Biomater. 2020, 107, 232–241. [Google Scholar] [CrossRef] [PubMed]
- Kang, S.; Yang, J.E.; Kim, J.; Ahn, M.; Koo, H.J.; Kim, M.; Lee, Y.-S.; Paik, S.R. Removal of Intact Β2-Microglobulin at Neutral Ph by Using Seed-Conjugated Polymer Beads Prepared with Β2-Microglobulin-Derived Peptide (58–67). Biotechnol. Prog. 2011, 27, 521–529. [Google Scholar] [CrossRef] [PubMed]
- Grovender, E.A.; Kellogg, B.; Singh, J.; Blom, D.; Ploegh, H.; Wittrup, K.D.; Langer, R.S.; Ameer, G.A. Single-Chain Antibody Fragment-Based Adsorbent for the Extracorporeal Removal of Beta2-Microglobulin. Kidney Int. 2004, 65, 310–322. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Studier, F.W. Protein Production by Auto-Induction in High Density Shaking Cultures. Protein Expr. Purif. 2005, 41, 207–234. [Google Scholar] [CrossRef] [PubMed]
- Trinh, C.H.; Smith, D.P.; Kalverda, A.P.; Phillips, S.E.V.; Radford, S.E. Crystal Structure of Monomeric Human β-2-Microglobulin Reveals Clues to Its Amyloidogenic Properties. Proc. Natl. Acad. Sci. USA 2002, 99, 9771–9776. [Google Scholar] [CrossRef]
- McParland, V.J.; Kad, N.M.; Kalverda, A.P.; Brown, A.; Kirwin-Jones, P.; Hunter, M.G.; Sunde, M.; Radford, S.E. Partially Unfolded States of β2-Microglobulin and Amyloid Formation in Vitro. Biochemistry 2000, 39, 8735–8746. [Google Scholar] [CrossRef]
- Cannon, K.A.; Ochoa, J.M.; Yeates, T.O. High-Symmetry Protein Assemblies: Patterns and Emerging Applications. Curr. Opin. Struct. Biol. 2019, 55, 77–84. [Google Scholar] [CrossRef] [Green Version]
- Edwardson, T.G.W.; Mori, T.; Hilvert, D. Rational Engineering of a Designed Protein Cage for SiRNA Delivery. J. Am. Chem. Soc. 2018, 140, 10439–10442. [Google Scholar] [CrossRef]
- Butterfield, G.L.; Lajoie, M.J.; Gustafson, H.H.; Sellers, D.L.; Nattermann, U.; Ellis, D.; Bale, J.B.; Ke, S.; Lenz, G.H.; Yehdego, A.; et al. Evolution of a Designed Protein Assembly Encapsulating Its Own RNA Genome. Nature 2017, 552, 415–420. [Google Scholar] [CrossRef] [Green Version]
- Edwardson, T.G.W.; Tetter, S.; Hilvert, D. Two-Tier Supramolecular Encapsulation of Small Molecules in a Protein Cage. Nat. Commun. 2020, 11, 5410. [Google Scholar] [CrossRef]
- Divine, R.; Dang, H.V.; Ueda, G.; Fallas, J.A.; Vulovic, I.; Sheffler, W.; Saini, S.; Zhao, Y.T.; Raj, I.X.; Morawski, P.A.; et al. Designed Proteins Assemble Antibodies into Modular Nanocages. Science 2021, 372, eabd9994. [Google Scholar] [CrossRef]
- McConnell, S.A.; Cannon, K.A.; Morgan, C.; McAllister, R.; Amer, B.R.; Clubb, R.T.; Yeates, T.O. Designed Protein Cages as Scaffolds for Building Multienzyme Materials. ACS Synth. Biol. 2020, 9, 381–391. [Google Scholar] [CrossRef] [PubMed]
- Kanekiyo, M.; Wei, C.-J.; Yassine, H.M.; McTamney, P.M.; Boyington, J.C.; Whittle, J.R.R.; Rao, S.S.; Kong, W.-P.; Wang, L.; Nabel, G.J. Self-Assembling Influenza Nanoparticle Vaccines Elicit Broadly Neutralizing H1N1 Antibodies. Nature 2013, 499, 102–106. [Google Scholar] [CrossRef]
- Marcandalli, J.; Fiala, B.; Ols, S.; Perotti, M.; de van der Schueren, W.; Snijder, J.; Hodge, E.; Benhaim, M.; Ravichandran, R.; Carter, L.; et al. Induction of Potent Neutralizing Antibody Responses by a Designed Protein Nanoparticle Vaccine for Respiratory Syncytial Virus. Cell 2019, 176, 1420–1431.e17. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Varner, C.T.; Rosen, T.; Martin, J.T.; Kane, R.S. Recent Advances in Engineering Polyvalent Biological Interactions. Biomacromolecules 2015, 16, 43–55. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cannon, K.A.; Park, R.U.; Boyken, S.E.; Nattermann, U.; Yi, S.; Baker, D.; King, N.P.; Yeates, T.O. Design and Structure of Two New Protein Cages Illustrate Successes and Ongoing Challenges in Protein Engineering. Protein Sci. 2020, 29, 919–929. [Google Scholar] [CrossRef]
- Bale, J.B.; Gonen, S.; Liu, Y.; Sheffler, W.; Ellis, D.; Thomas, C.; Cascio, D.; Yeates, T.O.; Gonen, T.; King, N.P.; et al. Accurate Design of Megadalton-Scale Two-Component Icosahedral Protein Complexes. Science 2016, 353, 389–394. [Google Scholar] [CrossRef] [Green Version]
- Domanska, K.; Vanderhaegen, S.; Srinivasan, V.; Pardon, E.; Dupeux, F.; Marquez, J.A.; Giorgetti, S.; Stoppini, M.; Wyns, L.; Bellotti, V.; et al. Atomic Structure of a Nanobody-Trapped Domain-Swapped Dimer of an Amyloidogenic Β2-Microglobulin Variant. Proc. Natl. Acad. Sci. USA 2011, 108, 1314–1319. [Google Scholar] [CrossRef]
- Brouwer, P.J.M.; Antanasijevic, A.; Ronk, A.J.; Müller-Kräuter, H.; Watanabe, Y.; Claireaux, M.; Perrett, H.R.; Bijl, T.P.L.; Grobben, M.; Umotoy, J.C.; et al. Lassa Virus Glycoprotein Nanoparticles Elicit Neutralizing Antibody Responses and Protection. Cell Host Microbe 2022, 30, 1759–1772.e12. [Google Scholar] [CrossRef]
- Liu, Y.; Huynh, D.T.; Yeates, T.O. A 3.8 Å Resolution Cryo-EM Structure of a Small Protein Bound to an Imaging Scaffold. Nat. Commun. 2019, 10, 1864. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Edwardson, T.G.W.; Levasseur, M.D.; Tetter, S.; Steinauer, A.; Hori, M.; Hilvert, D. Protein Cages: From Fundamentals to Advanced Applications. Chem. Rev. 2022, 122, 9145–9197. [Google Scholar] [CrossRef] [PubMed]
- Dundas, C.M.; Demonte, D.; Park, S. Streptavidin-Biotin Technology: Improvements and Innovations in Chemical and Biological Applications. Appl. Microbiol. Biotechnol. 2013, 97, 9343–9353. [Google Scholar] [CrossRef] [PubMed]




Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Miller, J.E.; Castells-Graells, R.; Arbing, M.A.; Munoz, A.; Jiang, Y.-X.; Espinoza, C.T.; Nguyen, B.; Moroz, P.; Yeates, T.O. Design of Beta-2 Microglobulin Adsorbent Protein Nanoparticles. Biomolecules 2023, 13, 1122. https://doi.org/10.3390/biom13071122
Miller JE, Castells-Graells R, Arbing MA, Munoz A, Jiang Y-X, Espinoza CT, Nguyen B, Moroz P, Yeates TO. Design of Beta-2 Microglobulin Adsorbent Protein Nanoparticles. Biomolecules. 2023; 13(7):1122. https://doi.org/10.3390/biom13071122
Chicago/Turabian StyleMiller, Justin E., Roger Castells-Graells, Mark A. Arbing, Aldo Munoz, Yi-Xiao Jiang, Charlize T. Espinoza, Brian Nguyen, Paul Moroz, and Todd O. Yeates. 2023. "Design of Beta-2 Microglobulin Adsorbent Protein Nanoparticles" Biomolecules 13, no. 7: 1122. https://doi.org/10.3390/biom13071122





