The Mechanisms of Action of Hyperbaric Oxygen in Restoring Host Homeostasis during Sepsis
Abstract
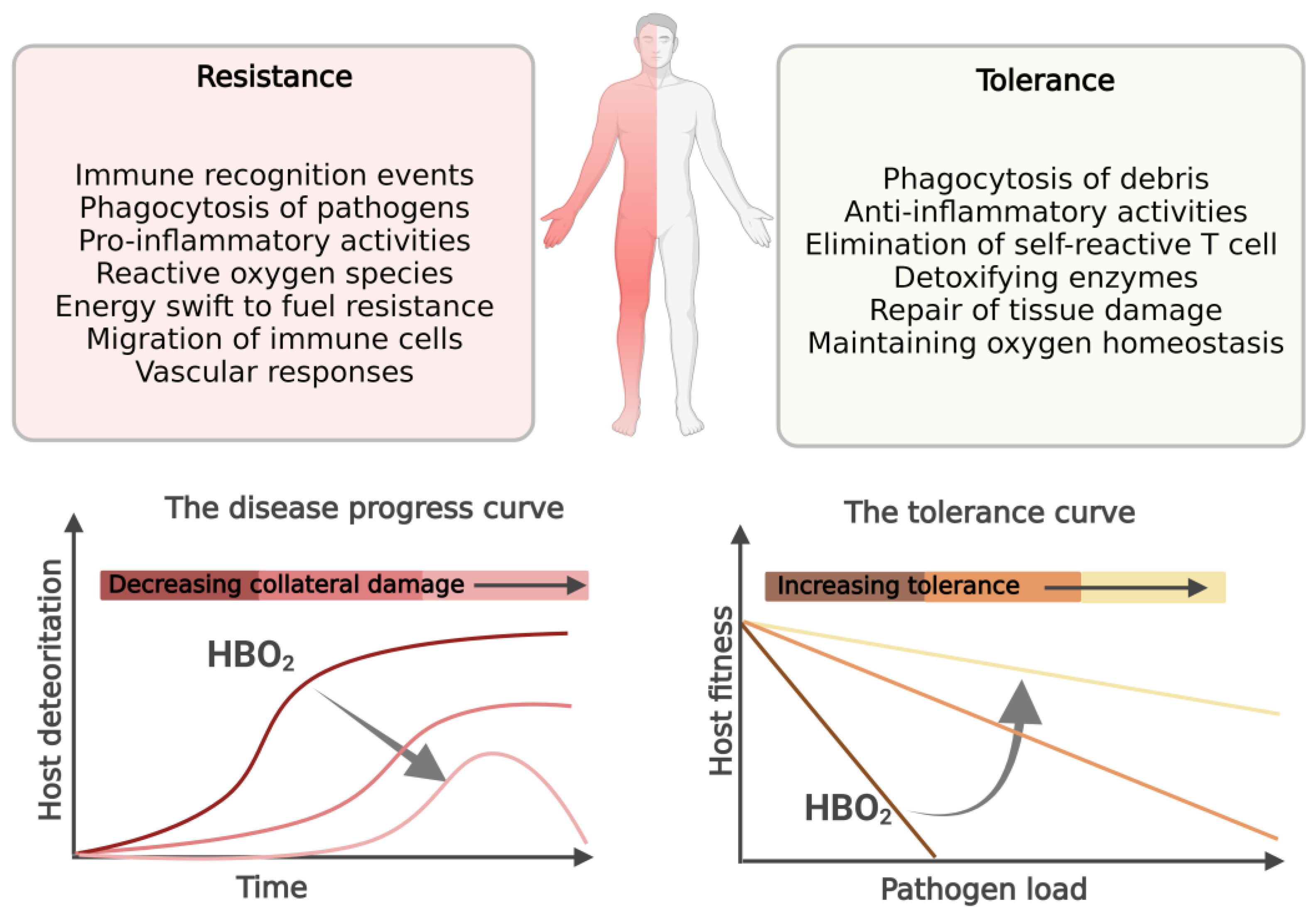
:1. Introduction
2. Resistance to Infection
2.1. Neutrophil Mediated Responses in Sepsis
2.2. Endothelial Dysfunction in Sepsis
2.3. The Cytokine Mediated Inflammatory Response in Sepsis
2.4. Switching Immune Cell Metabolism toward Glycolysis in Sepsis—The Warburg Effect
3. Tolerance to Infection
3.1. Iron Metabolism in the Pathogenesis of Sepsis
3.2. The Anti-Inflammatory Response in Sepsis
3.3. Hypoxia in the Pathogenesis of Sepsis
| Number of Hyperbaric Sessions | ||||||||
|---|---|---|---|---|---|---|---|---|
| 1–5 Sessions | 6–15 Sessions | 16–40 Sessions | ||||||
| mRNA | Protein | DNA Signal | mRNA | Protein | mRNA | Protein | ||
| Inflammatory stage | None | [113]*, [117]*, [118]*, [114] | [117]*, [118]*, [124], [125], [114], [17], [126] | [125], [114] | [124], [126] | |||
| [115]* | ||||||||
| Acute | [129], [130], [61]*, [131], [132], [133], [134], [135], | [61], | [136], [137], [139], | |||||
| [116] | [171], [172], [140], [141] | [141]* | [120], [121] | [120], [144], [145], [146], [147], [156], [148]*, [157] | ||||
| Chronic | [127]*, | [128], | ||||||
| [119]* | [119]*, [138], | [122]*, | [122]*, | |||||
| [142], [143] | [142] | [123] | [149], [151], [150], [152], [153] | [117] | [117], [154], [47], [155], [158], [159] | |||
| (a) | ||||||||
| Inflammatory stage | None | [117]* | [160], [17], [117] | |||||
| Acute | [161], | [162], | ||||||
| [163], [164], [165], [166], [46], [167] | [168] | [163], [169] | [163], [166], [169] | [166] | ||||
| Chronic | [153], [170], [149] | [117] | [47], [154], [158], [117] | |||||
| (b) | ||||||||
4. Discussion
5. Future Directions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Evans, L.; Rhodes, A.; Alhazzani, W.; Antonelli, M.; Coopersmith, C.M.; French, C.; Machado, F.R.; McIntyre, L.; Ostermann, M.; Prescott, H.C.; et al. Surviving sepsis campaign: International guidelines for management of sepsis and septic shock 2021. Intensive Care Med. 2021, 47, 1181–1247. [Google Scholar] [CrossRef]
- Fleischmann-Struzek, C.; Mellhammar, L.; Rose, N.; Cassini, A.; Rudd, K.E.; Schlattmann, P.; Allegranzi, B.; Reinhart, K. Incidence and mortality of hospital- and ICU-treated sepsis: Results from an updated and expanded systematic review and meta-analysis. Intensive Care Med. 2020, 46, 1552–1562. [Google Scholar] [CrossRef] [PubMed]
- Rudd, K.E.; Johnson, S.C.; Agesa, K.M.; Shackelford, K.A.; Tsoi, D.; Kievlan, D.R.; Colombara, D.V.; Ikuta, K.S.; Kissoon, N.; Finfer, S.; et al. Global, regional, and national sepsis incidence and mortality, 1990–2017: Analysis for the Global Burden of Disease Study. Lancet 2020, 395, 200–211. [Google Scholar] [CrossRef] [Green Version]
- Ackerman, M.H.; Ahrens, T.; Kelly, J.; Pontillo, A. Sepsis. Crit. Care Nurs. Clin. N. Am. 2021, 33, 407–418. [Google Scholar] [CrossRef] [PubMed]
- Cecconi, M.; Evans, L.; Levy, M.; Rhodes, A. Sepsis and septic shock. Lancet 2018, 392, 75–87. [Google Scholar] [CrossRef] [PubMed]
- Larsen, R.; Gozzelino, R.; Jeney, V.; Tokaji, L.; Bozza, F.A.; Japiassu, A.M.; Bonaparte, D.; Cavalcante, M.M.; Chora, A.; Ferreira, A.; et al. A central role for free heme in the pathogenesis of severe sepsis. Sci. Transl. Med. 2010, 2, 51ra71. [Google Scholar] [CrossRef] [Green Version]
- Schneider, D.S.; Ayres, J.S. Two ways to survive infection: What resistance and tolerance can teach us about treating infectious diseases. Nat. Rev. Immunol. 2008, 8, 889–895. [Google Scholar] [CrossRef] [Green Version]
- Weis, S.; Carlos, A.R.; Moita, M.R.; Singh, S.; Blankenhaus, B.; Cardoso, S.; Larsen, R.; Rebelo, S.; Schauble, S.; Del Barrio, L.; et al. Metabolic Adaptation Establishes Disease Tolerance to Sepsis. Cell 2017, 169, 1263–1275.e14. [Google Scholar] [CrossRef] [Green Version]
- Soares, M.P.; Weiss, G. The Iron age of host-microbe interactions. EMBO Rep. 2015, 16, 1482–1500. [Google Scholar] [CrossRef] [Green Version]
- Moon, R.E.; Bakker, D.; Barnes, R.; Bennett, M.; Camporesi, E.; Cianci, P.; Clark, J.; Dodson, W.; Feldmeier, J.; Gesell, L.; et al. Hyperbaric Oxygen Therapy Indications, 14th ed.; Undersea and Hyperbaric Medical Society; Best Publishing Company: Palm Beach, FL, USA, 2019; p. 111. [Google Scholar]
- Dennis, T.J.; Mohr, N.M.; Bailey, O.E. The role of hyperbaric oxygen therapy in septic shock: Is it time for human studies? Undersea Hyperb. Med. 2022, 49, 43–55. [Google Scholar] [CrossRef]
- Thom, S.R. Oxidative stress is fundamental to hyperbaric oxygen therapy. J. Appl. Physiol. (1985) 2009, 106, 988–995. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schottlender, N.; Gottfried, I.; Ashery, U. Hyperbaric Oxygen Treatment: Effects on Mitochondrial Function and Oxidative Stress. Biomolecules 2021, 11, 1827. [Google Scholar] [CrossRef] [PubMed]
- Hadanny, A.; Efrati, S. The Hyperoxic-Hypoxic Paradox. Biomolecules 2020, 10, 958. [Google Scholar] [CrossRef] [PubMed]
- Gottfried, I.; Schottlender, N.; Ashery, U. Hyperbaric Oxygen Treatment-From Mechanisms to Cognitive Improvement. Biomolecules 2021, 11, 1520. [Google Scholar] [CrossRef] [PubMed]
- Fratantonio, D.; Cimino, F.; Speciale, A.; Virgili, F. Need (more than) two to Tango: Multiple tools to adapt to changes in oxygen availability. Biofactors 2018, 44, 207–218. [Google Scholar] [CrossRef] [Green Version]
- Fratantonio, D.; Virgili, F.; Zucchi, A.; Lambrechts, K.; Latronico, T.; Lafere, P.; Germonpre, P.; Balestra, C. Increasing Oxygen Partial Pressures Induce a Distinct Transcriptional Response in Human PBMC: A Pilot Study on the “Normobaric Oxygen Paradox”. Int. J. Mol. Sci. 2021, 22, 458. [Google Scholar] [CrossRef]
- Macdonald, J.; Galley, H.F.; Webster, N.R. Oxidative stress and gene expression in sepsis. Br. J. Anaesth. 2003, 90, 221–232. [Google Scholar] [CrossRef] [Green Version]
- Thom, S.R.; Lauermann, M.W.; Hart, G.B. Intermittent hyperbaric oxygen therapy for reduction of mortality in experimental polymicrobial sepsis. J. Infect. Dis 1986, 154, 504–510. [Google Scholar] [CrossRef]
- Halbach, J.L.; Prieto, J.M.; Wang, A.W.; Hawisher, D.; Cauvi, D.M.; Reyes, T.; Okerblom, J.; Ramirez-Sanchez, I.; Villarreal, F.; Patel, H.H.; et al. Early hyperbaric oxygen therapy improves survival in a model of severe sepsis. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2019, 317, R160–R168. [Google Scholar] [CrossRef]
- Kot, J.; Lenkiewicz, E. Hyperbaric oxygen therapy in necrotizing soft tissue infections caused by Vibrio species from the Baltic Sea—Three clinical cases. Int. Marit. Health 2022, 73, 52–55. [Google Scholar] [CrossRef]
- Hedetoft, M.; Moser, C.; Jensen, P.O.; Vinkel, J.; Hyldegaard, O. Soluble ICAM-1 is modulated by hyperbaric oxygen treatment and correlates with disease severity and mortality in patients with necrotizing soft-tissue infection. J. Appl. Physiol. (1985) 2021, 130, 729–736. [Google Scholar] [CrossRef]
- Hedetoft, M.; Garred, P.; Madsen, M.B.; Hyldegaard, O. Hyperbaric oxygen treatment is associated with a decrease in cytokine levels in patients with necrotizing soft-tissue infection. Physiol. Rep. 2021, 9, e14757. [Google Scholar] [CrossRef]
- Hedetoft, M.; Jensen, P.O.; Moser, C.; Vinkel, J.; Hyldegaard, O. Hyperbaric oxygen treatment impacts oxidative stress markers in patients with necrotizing soft-tissue infection. J. Investig. Med. 2021, 69, 1330–1338. [Google Scholar] [CrossRef]
- Hedetoft, M.; Madsen, M.B.; Hyldegaard, O. Hyperbaric oxygen treatment in the management of necrotising soft-tissue infections: Results from a Danish nationwide registry study. BMJ Open 2023, 13, e066117. [Google Scholar] [CrossRef]
- Hedetoft, M.; Madsen, M.B.; Madsen, L.B.; Hyldegaard, O. Incidence, comorbidity and mortality in patients with necrotising soft-tissue infections, 2005-2018: A Danish nationwide register-based cohort study. BMJ Open 2020, 10, e041302. [Google Scholar] [CrossRef]
- Arina, P.; Singer, M. Pathophysiology of sepsis. Curr. Opin. Anaesthesiol. 2021, 34, 77–84. [Google Scholar] [CrossRef]
- van der Poll, T.; van de Veerdonk, F.L.; Scicluna, B.P.; Netea, M.G. The immunopathology of sepsis and potential therapeutic targets. Nat. Rev. Immunol. 2017, 17, 407–420. [Google Scholar] [CrossRef]
- Elsbach, P.; Weiss, J. Oxygen-dependent and oxygen-independent mechanisms of microbicidal activity of neutrophils. Immunol. Lett. 1985, 11, 159–163. [Google Scholar] [CrossRef]
- Nguyen, G.T.; Green, E.R.; Mecsas, J. Neutrophils to the ROScue: Mechanisms of NADPH Oxidase Activation and Bacterial Resistance. Front. Cell Infect. Microbiol. 2017, 7, 373. [Google Scholar] [CrossRef] [Green Version]
- Nussler, A.K.; Wittel, U.A.; Nussler, N.C.; Beger, H.G. Leukocytes, the Janus cells in inflammatory disease. Langenbecks Arch. Surg. 1999, 384, 222–232. [Google Scholar] [CrossRef]
- Grimberg-Peters, D.; Buren, C.; Windolf, J.; Wahlers, T.; Paunel-Gorgulu, A. Hyperbaric Oxygen Reduces Production of Reactive Oxygen Species in Neutrophils from Polytraumatized Patients Yielding in the Inhibition of p38 MAP Kinase and Downstream Pathways. PLoS ONE 2016, 11, e0161343. [Google Scholar] [CrossRef] [PubMed]
- de Wolde, S.D.; Hulskes, R.H.; de Jonge, S.W.; Hollmann, M.W.; van Hulst, R.A.; Weenink, R.P.; Kox, M. The Effect of Hyperbaric Oxygen Therapy on Markers of Oxidative Stress and the Immune Response in Healthy Volunteers. Front. Physiol. 2022, 13, 826163. [Google Scholar] [CrossRef] [PubMed]
- Ince, C.; Mayeux, P.R.; Nguyen, T.; Gomez, H.; Kellum, J.A.; Ospina-Tascon, G.A.; Hernandez, G.; Murray, P.; De Backer, D.; Workgroup, A.X. The Endothelium in Sepsis. Shock 2016, 45, 259–270. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Baiula, M.; Greco, R.; Ferrazzano, L.; Caligiana, A.; Hoxha, K.; Bandini, D.; Longobardi, P.; Spampinato, S.; Tolomelli, A. Integrin-mediated adhesive properties of neutrophils are reduced by hyperbaric oxygen therapy in patients with chronic non-healing wound. PLoS ONE 2020, 15, e0237746. [Google Scholar] [CrossRef]
- Mittal, M.; Siddiqui, M.R.; Tran, K.; Reddy, S.P.; Malik, A.B. Reactive oxygen species in inflammation and tissue injury. Antioxid. Redox Signal. 2014, 20, 1126–1167. [Google Scholar] [CrossRef] [Green Version]
- Zamboni, W.A.; Roth, A.C.; Russell, R.C.; Graham, B.; Suchy, H.; Kucan, J.O. Morphologic analysis of the microcirculation during reperfusion of ischemic skeletal muscle and the effect of hyperbaric oxygen. Plast. Reconstr. Surg. 1993, 91, 1110–1123. [Google Scholar] [CrossRef]
- Kalns, J.; Lane, J.; Delgado, A.; Scruggs, J.; Ayala, E.; Gutierrez, E.; Warren, D.; Niemeyer, D.; George Wolf, E.; Bowden, R.A. Hyperbaric oxygen exposure temporarily reduces Mac-1 mediated functions of human neutrophils. Immunol. Lett. 2002, 83, 125–131. [Google Scholar] [CrossRef]
- Thom, S.R.; Bhopale, V.M.; Mancini, D.J.; Milovanova, T.N. Actin S-nitrosylation inhibits neutrophil beta2 integrin function. J. Biol. Chem. 2008, 283, 10822–10834. [Google Scholar] [CrossRef] [Green Version]
- Buras, J.A.; Stahl, G.L.; Svoboda, K.K.; Reenstra, W.R. Hyperbaric oxygen downregulates ICAM-1 expression induced by hypoxia and hypoglycemia: The role of NOS. Am. J. Physiol. Cell Physiol. 2000, 278, C292–C302. [Google Scholar] [CrossRef]
- Liu, T.; Zhang, L.; Joo, D.; Sun, S.C. NF-kappaB signaling in inflammation. Signal Transduct. Target Ther. 2017, 2, 17023. [Google Scholar] [CrossRef] [Green Version]
- Gharamti, A.A.; Samara, O.; Monzon, A.; Montalbano, G.; Scherger, S.; DeSanto, K.; Chastain, D.B.; Sillau, S.; Montoya, J.G.; Franco-Paredes, C.; et al. Proinflammatory cytokines levels in sepsis and healthy volunteers, and tumor necrosis factor-alpha associated sepsis mortality: A systematic review and meta-analysis. Cytokine 2022, 158, 156006. [Google Scholar] [CrossRef]
- Bozza, F.A.; Salluh, J.I.; Japiassu, A.M.; Soares, M.; Assis, E.F.; Gomes, R.N.; Bozza, M.T.; Castro-Faria-Neto, H.C.; Bozza, P.T. Cytokine profiles as markers of disease severity in sepsis: A multiplex analysis. Crit. Care 2007, 11, R49. [Google Scholar] [CrossRef] [Green Version]
- Bubici, C.; Papa, S.; Dean, K.; Franzoso, G. Mutual cross-talk between reactive oxygen species and nuclear factor-kappa B: Molecular basis and biological significance. Oncogene 2006, 25, 6731–6748. [Google Scholar] [CrossRef] [Green Version]
- Liu, S.; Lu, C.; Liu, Y.; Zhou, X.; Sun, L.; Gu, Q.; Shen, G.; Guo, A. Hyperbaric Oxygen Alleviates the Inflammatory Response Induced by LPS Through Inhibition of NF-kappaB/MAPKs-CCL2/CXCL1 Signaling Pathway in Cultured Astrocytes. Inflammation 2018, 41, 2003–2011. [Google Scholar] [CrossRef]
- Sakoda, M.; Ueno, S.; Kihara, K.; Arikawa, K.; Dogomori, H.; Nuruki, K.; Takao, S.; Aikou, T. A potential role of hyperbaric oxygen exposure through intestinal nuclear factor-kappaB. Crit. Care Med. 2004, 32, 1722–1729. [Google Scholar] [CrossRef]
- Lu, Z.; Ma, J.; Liu, B.; Dai, C.; Xie, T.; Ma, X.; Li, M.; Dong, J.; Lan, Q.; Huang, Q. Hyperbaric oxygen therapy sensitizes nimustine treatment for glioma in mice. Cancer Med. 2016, 5, 3147–3155. [Google Scholar] [CrossRef]
- De Wolde, S.D.; Hulskes, R.H.; Weenink, R.P.; Hollmann, M.W.; Van Hulst, R.A. The Effects of Hyperbaric Oxygenation on Oxidative Stress, Inflammation and Angiogenesis. Biomolecules 2021, 11, 1210. [Google Scholar] [CrossRef]
- Weisz, G.; Lavy, A.; Adir, Y.; Melamed, Y.; Rubin, D.; Eidelman, S.; Pollack, S. Modification of in vivo and in vitro TNF-alpha, IL-1, and IL-6 secretion by circulating monocytes during hyperbaric oxygen treatment in patients with perianal Crohn’s disease. J. Clin. Immunol. 1997, 17, 154–159. [Google Scholar] [CrossRef]
- Warburg, O.; Wind, F.; Negelein, E. The Metabolism of Tumors in the Body. J. Gen. Physiol. 1927, 8, 519–530. [Google Scholar] [CrossRef] [Green Version]
- Srivastava, A.; Mannam, P. Warburg revisited: Lessons for innate immunity and sepsis. Front. Physiol. 2015, 6, 70. [Google Scholar] [CrossRef] [Green Version]
- Cheng, S.C.; Quintin, J.; Cramer, R.A.; Shepardson, K.M.; Saeed, S.; Kumar, V.; Giamarellos-Bourboulis, E.J.; Martens, J.H.; Rao, N.A.; Aghajanirefah, A.; et al. mTOR- and HIF-1alpha-mediated aerobic glycolysis as metabolic basis for trained immunity. Science 2014, 345, 1250684. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bar-Or, D.; Carrick, M.; Tanner, A., 2nd; Lieser, M.J.; Rael, L.T.; Brody, E. Overcoming the Warburg Effect: Is it the key to survival in sepsis? J. Crit. Care 2018, 43, 197–201. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.; Xie, M.; Yang, M.; Yu, Y.; Zhu, S.; Hou, W.; Kang, R.; Lotze, M.T.; Billiar, T.R.; Wang, H.; et al. PKM2 regulates the Warburg effect and promotes HMGB1 release in sepsis. Nat. Commun. 2014, 5, 4436. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, R.; Green, D.R. Metabolic checkpoints in activated T cells. Nat. Immunol. 2012, 13, 907–915. [Google Scholar] [CrossRef]
- Madsen, M.B.; Skrede, S.; Perner, A.; Arnell, P.; Nekludov, M.; Bruun, T.; Karlsson, Y.; Hansen, M.B.; Polzik, P.; Hedetoft, M.; et al. Patient’s characteristics and outcomes in necrotising soft-tissue infections: Results from a Scandinavian, multicentre, prospective cohort study. Intensive Care Med. 2019, 45, 1241–1251. [Google Scholar] [CrossRef]
- Casserly, B.; Phillips, G.S.; Schorr, C.; Dellinger, R.P.; Townsend, S.R.; Osborn, T.M.; Reinhart, K.; Selvakumar, N.; Levy, M.M. Lactate measurements in sepsis-induced tissue hypoperfusion: Results from the Surviving Sepsis Campaign database. Crit. Care Med. 2015, 43, 567–573. [Google Scholar] [CrossRef]
- Mikkelsen, M.E.; Miltiades, A.N.; Gaieski, D.F.; Goyal, M.; Fuchs, B.D.; Shah, C.V.; Bellamy, S.L.; Christie, J.D. Serum lactate is associated with mortality in severe sepsis independent of organ failure and shock. Crit. Care Med. 2009, 37, 1670–1677. [Google Scholar] [CrossRef]
- Revelly, J.P.; Tappy, L.; Martinez, A.; Bollmann, M.; Cayeux, M.C.; Berger, M.M.; Chiolero, R.L. Lactate and glucose metabolism in severe sepsis and cardiogenic shock. Crit. Care Med. 2005, 33, 2235–2240. [Google Scholar] [CrossRef]
- Tezgin, D.; Giardina, C.; Perdrizet, G.A.; Hightower, L.E. The effect of hyperbaric oxygen on mitochondrial and glycolytic energy metabolism: The caloristasis concept. Cell Stress Chaperones 2020, 25, 667–677. [Google Scholar] [CrossRef]
- Zhang, L.; Ke, J.; Min, S.; Wu, N.; Liu, F.; Qu, Z.; Li, W.; Wang, H.; Qian, Z.; Wang, X. Hyperbaric Oxygen Therapy Represses the Warburg Effect and Epithelial-Mesenchymal Transition in Hypoxic NSCLC Cells via the HIF-1alpha/PFKP Axis. Front. Oncol. 2021, 11, 691762. [Google Scholar] [CrossRef]
- Poff, A.M.; Ward, N.; Seyfried, T.N.; Arnold, P.; D’Agostino, D.P. Non-Toxic Metabolic Management of Metastatic Cancer in VM Mice: Novel Combination of Ketogenic Diet, Ketone Supplementation, and Hyperbaric Oxygen Therapy. PLoS ONE 2015, 10, e0127407. [Google Scholar] [CrossRef] [Green Version]
- Poff, A.M.; Ari, C.; Seyfried, T.N.; D’Agostino, D.P. The ketogenic diet and hyperbaric oxygen therapy prolong survival in mice with systemic metastatic cancer. PLoS ONE 2013, 8, e65522. [Google Scholar] [CrossRef] [Green Version]
- Fink, M. Cytopathic hypoxia in sepsis. Acta Anaesthesiol. Scand. Suppl. 1997, 110, 87–95. [Google Scholar] [CrossRef]
- Tenhunen, R.; Marver, H.S.; Schmid, R. The enzymatic conversion of heme to bilirubin by microsomal heme oxygenase. Proc. Natl. Acad. Sci. USA 1968, 61, 748–755. [Google Scholar] [CrossRef]
- Ferreira, B.L.; Leite, G.G.F.; Brunialti, M.K.C.; Assuncao, M.; Azevedo, L.C.P.; Freitas, F.; Salomao, R. HIF-1alpha and Hypoxia Responsive Genes are Differentially Expressed in Leukocytes from Survivors and Non-Survivors Patients During Clinical Sepsis. Shock 2021, 56, 80–91. [Google Scholar] [CrossRef]
- Seixas, E.; Gozzelino, R.; Chora, A.; Ferreira, A.; Silva, G.; Larsen, R.; Rebelo, S.; Penido, C.; Smith, N.R.; Coutinho, A.; et al. Heme oxygenase-1 affords protection against noncerebral forms of severe malaria. Proc. Natl. Acad. Sci. USA 2009, 106, 15837–15842. [Google Scholar] [CrossRef]
- Liu, Q.; Wu, J.; Zhang, X.; Wu, X.; Zhao, Y.; Ren, J. Iron homeostasis and disorders revisited in the sepsis. Free Radic. Biol. Med. 2021, 165, 1–13. [Google Scholar] [CrossRef]
- Prawan, A.; Kundu, J.K.; Surh, Y.J. Molecular basis of heme oxygenase-1 induction: Implications for chemoprevention and chemoprotection. Antioxid. Redox Signal. 2005, 7, 1688–1703. [Google Scholar] [CrossRef]
- Huang, T.Y.; Tsai, P.S.; Wang, T.Y.; Huang, C.L.; Huang, C.J. Hyperbaric oxygen attenuation of lipopolysaccharide-induced acute lung injury involves heme oxygenase-1. Acta Anaesthesiol. Scand. 2005, 49, 1293–1301. [Google Scholar] [CrossRef]
- Li, Q.; Li, J.; Zhang, L.; Wang, B.; Xiong, L. Preconditioning with hyperbaric oxygen induces tolerance against oxidative injury via increased expression of heme oxygenase-1 in primary cultured spinal cord neurons. Life Sci. 2007, 80, 1087–1093. [Google Scholar] [CrossRef]
- He, X.; Xu, X.; Fan, M.; Chen, X.; Sun, X.; Luo, G.; Chen, L.; Mu, Q.; Feng, Y.; Mao, Q.; et al. Preconditioning with hyperbaric oxygen induces tolerance against renal ischemia-reperfusion injury via increased expression of heme oxygenase-1. J. Surg. Res. 2011, 170, e271–e277. [Google Scholar] [CrossRef]
- Speit, G.; Dennog, C.; Eichhorn, U.; Rothfuss, A.; Kaina, B. Induction of heme oxygenase-1 and adaptive protection against the induction of DNA damage after hyperbaric oxygen treatment. Carcinogenesis 2000, 21, 1795–1799. [Google Scholar] [CrossRef] [Green Version]
- Rothfuss, A.; Radermacher, P.; Speit, G. Involvement of heme oxygenase-1 (HO-1) in the adaptive protection of human lymphocytes after hyperbaric oxygen (HBO) treatment. Carcinogenesis 2001, 22, 1979–1985. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yin, X.; Wang, X.; Fan, Z.; Peng, C.; Ren, Z.; Huang, L.; Liu, Z.; Zhao, K. Hyperbaric Oxygen Preconditioning Attenuates Myocardium Ischemia-Reperfusion Injury Through Upregulation of Heme Oxygenase 1 Expression: PI3K/Akt/Nrf2 Pathway Involved. J. Cardiovasc. Pharmacol. Ther. 2015, 20, 428–438. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- van der Poll, T.; van Deventer, S.J. Cytokines and anticytokines in the pathogenesis of sepsis. Infect. Dis. Clin. N. Am. 1999, 13, 413–426. [Google Scholar] [CrossRef] [PubMed]
- Hack, C.E.; Aarden, L.A.; Thijs, L.G. Role of cytokines in sepsis. Adv. Immunol. 1997, 66, 101–195. [Google Scholar] [CrossRef]
- Schulte, W.; Bernhagen, J.; Bucala, R. Cytokines in sepsis: Potent immunoregulators and potential therapeutic targets—An updated view. Mediators Inflamm. 2013, 2013, 165974. [Google Scholar] [CrossRef] [Green Version]
- Howard, M.; O’Garra, A.; Ishida, H.; de Waal Malefyt, R.; de Vries, J. Biological properties of interleukin 10. J. Clin. Immunol. 1992, 12, 239–247. [Google Scholar] [CrossRef]
- Howard, M.; Muchamuel, T.; Andrade, S.; Menon, S. Interleukin 10 protects mice from lethal endotoxemia. J. Exp. Med. 1993, 177, 1205–1208. [Google Scholar] [CrossRef]
- Opal, S.M.; DePalo, V.A. Anti-inflammatory cytokines. Chest 2000, 117, 1162–1172. [Google Scholar] [CrossRef] [Green Version]
- Marie, C.; Cavaillon, J.M.; Losser, M.R. Elevated levels of circulating transforming growth factor-beta 1 in patients with the sepsis syndrome. Ann. Intern. Med. 1996, 125, 520–521. [Google Scholar] [CrossRef]
- Buras, J.A.; Holt, D.; Orlow, D.; Belikoff, B.; Pavlides, S.; Reenstra, W.R. Hyperbaric oxygen protects from sepsis mortality via an interleukin-10-dependent mechanism. Crit. Care Med. 2006, 34, 2624–2629. [Google Scholar] [CrossRef]
- Bai, X.; Song, Z.; Zhou, Y.; Pan, S.; Wang, F.; Guo, Z.; Jiang, M.; Wang, G.; Kong, R.; Sun, B. The apoptosis of peripheral blood lymphocytes promoted by hyperbaric oxygen treatment contributes to attenuate the severity of early stage acute pancreatitis in rats. Apoptosis 2014, 19, 58–75. [Google Scholar] [CrossRef]
- Yoshinoya, Y.; Bocker, A.H.; Ruhl, T.; Siekmann, U.; Pallua, N.; Beier, J.P.; Kim, B.S. The Effect of Hyperbaric Oxygen Therapy on Human Adipose-Derived Stem Cells. Plast. Reconstr. Surg. 2020, 146, 309–320. [Google Scholar] [CrossRef]
- Romero-Valdovinos, M.; Cardenas-Mejia, A.; Gutierrez-Gomez, C.; Flisser, A.; Kawa-Karasik, S.; Ortiz-Monasterio, F. Keloid skin scars: The influence of hyperbaric oxygenation on fibroblast growth and on the expression of messenger RNA for insulin like growth factor and for transforming growth factor. In Vitro Cell Dev. Biol. Anim. 2011, 47, 421–424. [Google Scholar] [CrossRef]
- Oscarsson, N.; Ny, L.; Molne, J.; Lind, F.; Ricksten, S.E.; Seeman-Lodding, H.; Giglio, D. Hyperbaric oxygen treatment reverses radiation induced pro-fibrotic and oxidative stress responses in a rat model. Free Radic. Biol. Med. 2017, 103, 248–255. [Google Scholar] [CrossRef]
- Wee, H.Y.; Lim, S.W.; Chio, C.C.; Niu, K.C.; Wang, C.C.; Kuo, J.R. Hyperbaric oxygen effects on neuronal apoptosis associations in a traumatic brain injury rat model. J. Surg. Res. 2015, 197, 382–389. [Google Scholar] [CrossRef]
- Shapira, R.; Solomon, B.; Efrati, S.; Frenkel, D.; Ashery, U. Hyperbaric oxygen therapy ameliorates pathophysiology of 3xTg-AD mouse model by attenuating neuroinflammation. Neurobiol. Aging 2018, 62, 105–119. [Google Scholar] [CrossRef]
- Wang, C.H.; Shan, M.J.; Liu, H.; Hao, Y.; Song, K.X.; Wu, H.W.; Meng, T.; Feng, C.; Qi, Z.; Wang, Z.; et al. Hyperbaric oxygen treatment on keloid tumor immune gene expression. Chin. Med. J. 2021, 134, 2205–2213. [Google Scholar] [CrossRef]
- Geng, C.K.; Cao, H.H.; Ying, X.; Zhang, H.T.; Yu, H.L. The effects of hyperbaric oxygen on macrophage polarization after rat spinal cord injury. Brain Res. 2015, 1606, 68–76. [Google Scholar] [CrossRef]
- Bonello, S.; Zahringer, C.; BelAiba, R.S.; Djordjevic, T.; Hess, J.; Michiels, C.; Kietzmann, T.; Gorlach, A. Reactive oxygen species activate the HIF-1alpha promoter via a functional NFkappaB site. Arterioscler. Thromb. Vasc. Biol. 2007, 27, 755–761. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rius, J.; Guma, M.; Schachtrup, C.; Akassoglou, K.; Zinkernagel, A.S.; Nizet, V.; Johnson, R.S.; Haddad, G.G.; Karin, M. NF-kappaB links innate immunity to the hypoxic response through transcriptional regulation of HIF-1alpha. Nature 2008, 453, 807–811. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Choudhury, R. Hypoxia and hyperbaric oxygen therapy: A review. Int. J. Gen. Med. 2018, 11, 431–442. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Weidemann, A.; Johnson, R.S. Biology of HIF-1alpha. Cell Death Differ. 2008, 15, 621–627. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vanderhaeghen, T.; Vandewalle, J.; Libert, C. Hypoxia-inducible factors in metabolic reprogramming during sepsis. FEBS J. 2020, 287, 1478–1495. [Google Scholar] [CrossRef] [Green Version]
- Fitzpatrick, S.F. Immunometabolism and Sepsis: A Role for HIF? Front. Mol. Biosci. 2019, 6, 85. [Google Scholar] [CrossRef]
- Fitzpatrick, S.F.; Gojkovic, M.; Macias, D.; Tegnebratt, T.; Lu, L.; Samen, E.; Rundqvist, H.; Johnson, R.S. Glycolytic Response to Inflammation Over Time: Role of Myeloid HIF-1alpha. Front. Physiol. 2018, 9, 1624. [Google Scholar] [CrossRef] [Green Version]
- Peyssonnaux, C.; Cejudo-Martin, P.; Doedens, A.; Zinkernagel, A.S.; Johnson, R.S.; Nizet, V. Cutting edge: Essential role of hypoxia inducible factor-1alpha in development of lipopolysaccharide-induced sepsis. J. Immunol. 2007, 178, 7516–7519. [Google Scholar] [CrossRef] [Green Version]
- Davenport, E.E.; Burnham, K.L.; Radhakrishnan, J.; Humburg, P.; Hutton, P.; Mills, T.C.; Rautanen, A.; Gordon, A.C.; Garrard, C.; Hill, A.V.; et al. Genomic landscape of the individual host response and outcomes in sepsis: A prospective cohort study. Lancet Respir. Med. 2016, 4, 259–271. [Google Scholar] [CrossRef] [Green Version]
- Schafer, S.T.; Frede, S.; Winning, S.; Bick, A.; Roshangar, P.; Fandrey, J.; Peters, J.; Adamzik, M. Hypoxia-inducible factor and target gene expression are decreased in patients with sepsis: Prospective observational clinical and cellular studies. Anesthesiology 2013, 118, 1426–1436. [Google Scholar] [CrossRef] [Green Version]
- Cimino, F.; Balestra, C.; Germonpre, P.; De Bels, D.; Tillmans, F.; Saija, A.; Speciale, A.; Virgili, F. Pulsed high oxygen induces a hypoxic-like response in human umbilical endothelial cells and in humans. J. Appl. Physiol. (1985) 2012, 113, 1684–1689. [Google Scholar] [CrossRef] [Green Version]
- Balestra, C.; Germonpre, P. Hypoxia, a multifaceted phenomenon: The example of the “normobaric oxygen paradox”. Eur. J. Appl. Physiol. 2012, 112, 4173–4175. [Google Scholar] [CrossRef]
- Rocco, M.; D’Itri, L.; De Bels, D.; Corazza, F.; Balestra, C. The "normobaric oxygen paradox”: A new tool for the anesthetist? Minerva Anestesiol. 2014, 80, 366–372. [Google Scholar]
- D’Ignazio, L.; Bandarra, D.; Rocha, S. NF-kappaB and HIF crosstalk in immune responses. FEBS J. 2016, 283, 413–424. [Google Scholar] [CrossRef] [Green Version]
- D’Ignazio, L.; Rocha, S. Hypoxia Induced NF-kappaB. Cells 2016, 5, 10. [Google Scholar] [CrossRef] [Green Version]
- Eltzschig, H.K.; Carmeliet, P. Hypoxia and inflammation. N. Engl. J. Med. 2011, 364, 656–665. [Google Scholar] [CrossRef] [Green Version]
- Lin, H.C.; Wan, F.J.; Wu, C.C.; Tung, C.S.; Wu, T.H. Hyperbaric oxygen protects against lipopolysaccharide-stimulated oxidative stress and mortality in rats. Eur. J. Pharmacol. 2005, 508, 249–254. [Google Scholar] [CrossRef]
- Cash, T.P.; Pan, Y.; Simon, M.C. Reactive oxygen species and cellular oxygen sensing. Free Radic. Biol. Med. 2007, 43, 1219–1225. [Google Scholar] [CrossRef] [Green Version]
- Movafagh, S.; Crook, S.; Vo, K. Regulation of hypoxia-inducible factor-1a by reactive oxygen species: New developments in an old debate. J. Cell Biochem. 2015, 116, 696–703. [Google Scholar] [CrossRef]
- Hagen, T. Oxygen versus Reactive Oxygen in the Regulation of HIF-1alpha: The Balance Tips. Biochem. Res. Int. 2012, 2012, 436981. [Google Scholar] [CrossRef] [Green Version]
- van Uden, P.; Kenneth, N.S.; Rocha, S. Regulation of hypoxia-inducible factor-1alpha by NF-kappaB. Biochem. J. 2008, 412, 477–484. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Salmón-González, Z.; Anchuelo, J.; Borregán, J.C.; Del Real, A.; Sañudo, C.; García Ibarbia, C.; Pérez Nuñez, M.I.; Riancho, J.A.; Valero, C. Influence of high-concentration hyperbaric oxygen therapy on bone metabolism. Rev. Osteoporos. Metab. Miner. 2020, 12, 28–31. [Google Scholar] [CrossRef]
- Ren, P.; Kang, Z.; Gu, G.; Liu, Y.; Xu, W.; Tao, H.; Zhang, J.H.; Sun, X.; Ji, H. Hyperbaric oxygen preconditioning promotes angiogenesis in rat liver after partial hepatectomy. Life Sci. 2008, 83, 236–241. [Google Scholar] [CrossRef] [PubMed]
- Ravaioli, M.; Baldassare, M.; Vasuri, F.; Pasquinelli, G.; Laggetta, M.; Valente, S.; De Pace, V.; Neri, F.; Siniscalchi, A.; Zanfi, C.; et al. Strategies to Restore Adenosine Triphosphate (ATP) Level After More than 20 Hours of Cold Ischemia Time in Human Marginal Kidney Grafts. Ann. Transplant. 2018, 23, 34–44. [Google Scholar] [CrossRef]
- Chen, L.; Gajendrareddy, P.K.; DiPietro, L.A. Differential expression of HIF-1alpha in skin and mucosal wounds. J. Dent. Res. 2012, 91, 871–876. [Google Scholar] [CrossRef] [Green Version]
- Huang, X.; Liang, P.; Jiang, B.; Zhang, P.; Yu, W.; Duan, M.; Guo, L.; Cui, X.; Huang, M.; Huang, X. Hyperbaric oxygen potentiates diabetic wound healing by promoting fibroblast cell proliferation and endothelial cell angiogenesis. Life Sci. 2020, 259, 118246. [Google Scholar] [CrossRef]
- Mihaljevic, Z.; Matic, A.; Stupin, A.; Frkanec, R.; Tavcar, B.; Kelava, V.; Tartaro Bujak, I.; Kolobaric, N.; Kibel, A.; Drenjancevic, I. Arachidonic Acid Metabolites of CYP450 Enzymes and HIF-1alpha Modulate Endothelium-Dependent Vasorelaxation in Sprague-Dawley Rats under Acute and Intermittent Hyperbaric Oxygenation. Int. J. Mol. Sci. 2020, 21, 6353. [Google Scholar] [CrossRef]
- Arienti, C.; Pignatta, S.; Zanoni, M.; Zamagni, A.; Cortesi, M.; Sarnelli, A.; Romeo, A.; Arpa, D.; Longobardi, P.; Bartolini, D.; et al. High-pressure oxygen rewires glucose metabolism of patient-derived glioblastoma cells and fuels inflammasome response. Cancer Lett. 2021, 506, 152–166. [Google Scholar] [CrossRef]
- Zhou, Y.; Liu, X.H.; Qu, S.D.; Yang, J.; Wang, Z.W.; Gao, C.J.; Su, Q.J. Hyperbaric oxygen intervention on expression of hypoxia-inducible factor-1alpha and vascular endothelial growth factor in spinal cord injury models in rats. Chin. Med. J. 2013, 126, 3897–3903. [Google Scholar]
- Novak, S.; Drenjancevic, I.; Vukovic, R.; Kellermayer, Z.; Cosic, A.; Tolusic Levak, M.; Balogh, P.; Culo, F.; Mihalj, M. Anti-Inflammatory Effects of Hyperbaric Oxygenation during DSS-Induced Colitis in BALB/c Mice Include Changes in Gene Expression of HIF-1alpha, Proinflammatory Cytokines, and Antioxidative Enzymes. Mediators Inflamm. 2016, 2016, 7141430. [Google Scholar] [CrossRef] [Green Version]
- Al Hadi, H.; Smerdon, G.R.; Fox, S.W. Hyperbaric oxygen therapy suppresses osteoclast formation and bone resorption. J. Orthop. Res. 2013, 31, 1839–1844. [Google Scholar] [CrossRef]
- Ergözen Serkan, A.H. The Effects of Hyperbaric Oxygen Treatment on Hypoxia Inducible Factor-1α, Inducible Nitric Oxide Synthase and Vascular Endothelial Growth Factor Levels in Patients with Diabetic Foot Wound. Med. J. Mugla Sitki Kocman Univ. 2019, 6, 142–146. [Google Scholar]
- Peng, Z.; Ren, P.; Kang, Z.; Du, J.; Lian, Q.; Liu, Y.; Zhang, J.H.; Sun, X. Up-regulated HIF-1alpha is involved in the hypoxic tolerance induced by hyperbaric oxygen preconditioning. Brain Res. 2008, 1212, 71–78. [Google Scholar] [CrossRef]
- Gu, G.J.; Li, Y.P.; Peng, Z.Y.; Xu, J.J.; Kang, Z.M.; Xu, W.G.; Tao, H.Y.; Ostrowski, R.P.; Zhang, J.H.; Sun, X.J. Mechanism of ischemic tolerance induced by hyperbaric oxygen preconditioning involves upregulation of hypoxia-inducible factor-1alpha and erythropoietin in rats. J. Appl. Physiol. (1985) 2008, 104, 1185–1191. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Milovanova, T.N.; Bhopale, V.M.; Sorokina, E.M.; Moore, J.S.; Hunt, T.K.; Hauer-Jensen, M.; Velazquez, O.C.; Thom, S.R. Hyperbaric oxygen stimulates vasculogenic stem cell growth and differentiation in vivo. J. Appl. Physiol. (1985) 2009, 106, 711–728. [Google Scholar] [CrossRef]
- Zembrzuska, K.; Ostrowski, R.P.; Matyja, E. Hyperbaric oxygen increases glioma cell sensitivity to antitumor treatment with a novel isothiourea derivative in vitro. Oncol. Rep. 2019, 41, 2703–2716. [Google Scholar] [CrossRef] [Green Version]
- Hu, Q.; Liang, X.; Chen, D.; Chen, Y.; Doycheva, D.; Tang, J.; Tang, J.; Zhang, J.H. Delayed hyperbaric oxygen therapy promotes neurogenesis through reactive oxygen species/hypoxia-inducible factor-1alpha/beta-catenin pathway in middle cerebral artery occlusion rats. Stroke 2014, 45, 1807–1814. [Google Scholar] [CrossRef]
- Ostrowski, R.P.; Colohan, A.R.; Zhang, J.H. Mechanisms of hyperbaric oxygen-induced neuroprotection in a rat model of subarachnoid hemorrhage. J. Cereb. Blood Flow Metab. 2005, 25, 554–571. [Google Scholar] [CrossRef] [Green Version]
- Li, Y.; Zhou, C.; Calvert, J.W.; Colohan, A.R.; Zhang, J.H. Multiple effects of hyperbaric oxygen on the expression of HIF-1 alpha and apoptotic genes in a global ischemia-hypotension rat model. Exp. Neurol. 2005, 191, 198–210. [Google Scholar] [CrossRef]
- Parra, R.S.; Lopes, A.H.; Carreira, E.U.; Feitosa, M.R.; Cunha, F.Q.; Garcia, S.B.; Cunha, T.M.; da Rocha, J.J.; Feres, O. Hyperbaric oxygen therapy ameliorates TNBS-induced acute distal colitis in rats. Med. Gas Res. 2015, 5, 6. [Google Scholar] [CrossRef]
- Calvert, J.W.; Cahill, J.; Yamaguchi-Okada, M.; Zhang, J.H. Oxygen treatment after experimental hypoxia-ischemia in neonatal rats alters the expression of HIF-1alpha and its downstream target genes. J. Appl. Physiol. (1985) 2006, 101, 853–865. [Google Scholar] [CrossRef] [Green Version]
- Sun, L.; Marti, H.H.; Veltkamp, R. Hyperbaric oxygen reduces tissue hypoxia and hypoxia-inducible factor-1 alpha expression in focal cerebral ischemia. Stroke 2008, 39, 1000–1006. [Google Scholar] [CrossRef] [Green Version]
- Huang, Z.X.; Kang, Z.M.; Gu, G.J.; Peng, G.N.; Yun, L.; Tao, H.Y.; Xu, W.G.; Sun, X.J.; Zhang, J.H. Therapeutic effects of hyperbaric oxygen in a rat model of endothelin-1-induced focal cerebral ischemia. Brain Res. 2007, 1153, 204–213. [Google Scholar] [CrossRef]
- Zhou, W.; Marinescu, M.; Veltkamp, R. Only very early oxygen therapy attenuates posthemorrhagic edema formation and blood-brain barrier disruption in murine intracerebral hemorrhage. Neurocrit. Care 2015, 22, 121–132. [Google Scholar] [CrossRef]
- Zhang, Q.; Chang, Q.; Cox, R.A.; Gong, X.; Gould, L.J. Hyperbaric oxygen attenuates apoptosis and decreases inflammation in an ischemic wound model. J. Investig. Dermatol. 2008, 128, 2102–2112. [Google Scholar] [CrossRef] [Green Version]
- Yang, Y.; Zhang, Y.G.; Lin, G.A.; Xie, H.Q.; Pan, H.T.; Huang, B.Q.; Liu, J.D.; Liu, H.; Zhang, N.; Li, L.; et al. The effects of different hyperbaric oxygen manipulations in rats after traumatic brain injury. Neurosci. Lett. 2014, 563, 38–43. [Google Scholar] [CrossRef]
- Lin, P.Y.; Sung, P.H.; Chung, S.Y.; Hsu, S.L.; Chung, W.J.; Sheu, J.J.; Hsueh, S.K.; Chen, K.H.; Wu, R.W.; Yip, H.K. Hyperbaric Oxygen Therapy Enhanced Circulating Levels of Endothelial Progenitor Cells and Angiogenesis Biomarkers, Blood Flow, in Ischemic Areas in Patients with Peripheral Arterial Occlusive Disease. J. Clin. Med. 2018, 7, 548. [Google Scholar] [CrossRef] [Green Version]
- Xue, R.; Pan, S.; Guo, D. Effect of Hyperbaric oxygen on myelin injury and repair after hypoxic-ischemic brain damage in adult rat. Neurosci. Lett. 2023, 794, 137015. [Google Scholar] [CrossRef]
- Yamamoto, N.; Oyaizu, T.; Enomoto, M.; Horie, M.; Yuasa, M.; Okawa, A.; Yagishita, K. VEGF and bFGF induction by nitric oxide is associated with hyperbaric oxygen-induced angiogenesis and muscle regeneration. Sci. Rep. 2020, 10, 2744. [Google Scholar] [CrossRef] [Green Version]
- Salhanick, S.D.; Belikoff, B.; Orlow, D.; Holt, D.; Reenstra, W.; Buras, J.A. Hyperbaric oxygen reduces acetaminophen toxicity and increases HIF-1alpha expression. Acad. Emerg. Med. 2006, 13, 707–714. [Google Scholar] [CrossRef]
- Sunkari, V.G.; Lind, F.; Botusan, I.R.; Kashif, A.; Liu, Z.J.; Yla-Herttuala, S.; Brismar, K.; Velazquez, O.; Catrina, S.B. Hyperbaric oxygen therapy activates hypoxia-inducible factor 1 (HIF-1), which contributes to improved wound healing in diabetic mice. Wound Repair Regen. 2015, 23, 98–103. [Google Scholar] [CrossRef] [PubMed]
- Hsu, S.L.; Yin, T.C.; Shao, P.L.; Chen, K.H.; Wu, R.W.; Chen, C.C.; Lin, P.Y.; Chung, S.Y.; Sheu, J.J.; Sung, P.H.; et al. Hyperbaric oxygen facilitates the effect of endothelial progenitor cell therapy on improving outcome of rat critical limb ischemia. Am. J. Transl. Res. 2019, 11, 1948–1964. [Google Scholar] [PubMed]
- Bai, X.; Sun, B.; Pan, S.; Jiang, H.; Wang, F.; Krissansen, G.W.; Sun, X. Down-regulation of hypoxia-inducible factor-1alpha by hyperbaric oxygen attenuates the severity of acute pancreatitis in rats. Pancreas 2009, 38, 515–522. [Google Scholar] [CrossRef] [PubMed]
- Harnanik, T.; Prihartono, S.; Juliandhy, T. Hyperbaric oxygen in animal model of rheumatoid arthritis: Analysis of HIF-1alpha, ACPA and IL-17a. Infect. Dis. Rep. 2020, 12 (Suppl. S1), 8766. [Google Scholar] [CrossRef]
- Sakata, N.; Chan, N.K.; Ostrowski, R.P.; Chrisler, J.; Hayes, P.; Kim, S.; Obenaus, A.; Zhang, J.H.; Hathout, E. Hyperbaric oxygen therapy improves early posttransplant islet function. Pediatr. Diabetes 2010, 11, 471–478. [Google Scholar] [CrossRef] [Green Version]
- Utami, P.D.; Hadi, U.; Dachlan, Y.P.; Suryokusumo, G.; Fitri, L.E. Hyperbaric Oxygen Exposure Reduces ICAM-1 And HIF-1α Expression in Brain Endothelial Cells from Experimental Cerebral Malaria Mice. Indian J. Forensic Med. Toxicol. 2020, 14, 3347–3352. [Google Scholar]
- Yuan, Y.; Li, Y.; Qiao, G.; Zhou, Y.; Xu, Z.; Hill, C.; Jiang, Z.; Wang, Y. Hyperbaric Oxygen Ameliorates Bleomycin-Induced Pulmonary Fibrosis in Mice. Front. Mol. Biosci. 2021, 8, 675437. [Google Scholar] [CrossRef]
- Syahrizal, D.; Mustika, C.; Ayu Puspita, N.; Guritno Suryokusumo, M.; Hendarto, H. The effect of hyperbaric oxygen therapy in the inflammatory response in a mouse model of endometriosis: An experimental study. Int. J. Reprod. Biomed. 2022, 20, 347–356. [Google Scholar] [CrossRef]
- Harnanik, T.; Soeroso, J.; Suryokusumo, M.G.; Juliandhy, T. Effects of Hyperbaric Oxygen on T helper 17/regulatory T Polarization in Antigen and Collagen-induced Arthritis: Hypoxia-inducible Factor-1alpha as a Target. Oman. Med. J. 2020, 35, e90. [Google Scholar] [CrossRef]
- Harnanik, T.; Soeroso, J.; Suryokusumo, M.G.; Juliandhy, T. Hyperbaric Oxygen Effect on Switching Phenotype M1-M2 Macrophage Expression in Antigen and Collagen-Induced Arthritis. Int. Med. J. 2019, 29, 479–482. [Google Scholar]
- Zhang, M.; Liu, S.; Guan, E.; Liu, H.; Dong, X.; Hao, Y.; Zhang, X.; Zhao, P.; Liu, X.; Pan, S.; et al. Hyperbaric oxygen therapy can ameliorate the EMT phenomenon in keloid tissue. Medicine 2018, 97, e11529. [Google Scholar] [CrossRef]
- Chen, C.A.; Huang, Y.C.; Lo, J.J.; Wang, S.H.; Huang, S.H.; Wu, S.H. Hyperbaric Oxygen Therapy Attenuates Burn-Induced Denervated Muscle Atrophy. Int. J. Med. Sci. 2021, 18, 3821–3830. [Google Scholar] [CrossRef]
- Song, K.X.; Liu, S.; Zhang, M.Z.; Liang, W.Z.; Liu, H.; Dong, X.H.; Wang, Y.B.; Wang, X.J. Hyperbaric oxygen therapy improves the effect of keloid surgery and radiotherapy by reducing the recurrence rate. J. Zhejiang Univ. Sci. B 2018, 19, 853–862. [Google Scholar] [CrossRef]
- Li, Z.; Li, M.; Li, X.; Zhang, M.; Zhao, Y.; Ren, W.; Cheng, J.; Wang, X. Hyperbaric oxygen inhibits venous neointimal hyperplasia following arteriovenous fistulization. Int. J. Mol. Med. 2017, 39, 1299–1306. [Google Scholar] [CrossRef] [Green Version]
- Wang, G.; Wu, Z.R.; Wang, R.; Li, J.; Huang, S.G. Effect of hyperbaric oxygen on HIF-1α expression in rat experimental periodontitis with psychological stress. Int. J. Clin. Exp. Med. 2017, 10, 11866–11875. [Google Scholar]
- Ding, J.B.; Chen, J.R.; Xu, H.Z.; Qin, Z.Y. Effect of Hyperbaric Oxygen on the Growth of Intracranial Glioma in Rats. Chin. Med. J. 2015, 128, 3197–3203. [Google Scholar] [CrossRef]
- Anguiano-Hernandez, Y.M.; Contreras-Mendez, L.; de Los Angeles Hernandez-Cueto, M.; Muand Oz-Medina, J.E.; Santillan-Verde, M.A.; Barbosa-Cabrera, R.E.; Delgado-Quintana, C.A.; Trejo-Rosas, S.; Santacruz-Tinoco, C.E.; Gonzalez-Ibarra, J.; et al. Modification of HIF-1alpha, NF-akappaB, IGFBP-3, VEGF and adiponectin in diabetic foot ulcers treated with hyperbaric oxygen. Undersea Hyperb. Med. 2019, 46, 35–44. [Google Scholar] [CrossRef]
- Capó, X.; Monserrat-Mesquida, M.; Quetglas-Llabrés, M.; Batle, J.M.; Tur, J.A.; Pons, A.; Sureda, A.; Tejada, S. Hyperbaric Oxygen Therapy Reduces Oxidative Stress and Inflammation, and Increases Growth Factors Favouring the Healing Process of Diabetic Wounds. Int. J. Mol. Sci. 2023, 24, 7040. [Google Scholar] [CrossRef]
- Madden, L.A.; Vince, R.V.; Laden, G. The effect of acute hyperoxia in vivo on NF kappa B expression in human PBMC. Cell Biochem. Funct. 2011, 29, 71–73. [Google Scholar] [CrossRef]
- Ko, S.F.; Chen, K.H.; Wallace, C.G.; Yang, C.C.; Sung, P.H.; Shao, P.L.; Li, Y.C.; Chen, Y.T.; Yip, H.K. Protective effect of combined therapy with hyperbaric oxygen and autologous adipose-derived mesenchymal stem cells on renal function in rodent after acute ischemia-reperfusion injury. Am. J. Transl. Res. 2020, 12, 3272–3287. [Google Scholar]
- Liang, F.; Kang, N.; Liu, X.; Yang, J.; Li, Z.; Tan, J.W. Effect of HMGB1/NF-kappaB in hyperbaric oxygen treatment on decreasing injury caused by skin flap grafts in rats. Eur. Rev. Med. Pharmacol. Sci. 2013, 17, 2010–2018. [Google Scholar] [PubMed]
- Yang, J.; Liu, X.; Zhou, Y.; Wang, G.; Gao, C.; Su, Q. Hyperbaric oxygen alleviates experimental (spinal cord) injury by downregulating HMGB1/NF-kappaB expression. Spine 2013, 38, E1641–E1648. [Google Scholar] [CrossRef] [PubMed]
- Tan, J.; Zhang, F.; Liang, F.; Wang, Y.; Li, Z.; Yang, J.; Liu, X. Protective effects of hyperbaric oxygen treatment against spinal cord injury in rats via toll-like receptor 2/nuclear factor-kappaB signaling. Int. J. Clin. Exp. Pathol. 2014, 7, 1911–1919. [Google Scholar]
- Meng, X.E.; Zhang, Y.; Li, N.; Fan, D.F.; Yang, C.; Li, H.; Guo, D.Z.; Pan, S.Y. Hyperbaric Oxygen Alleviates Secondary Brain Injury After Trauma Through Inhibition of TLR4/NF-kappaB Signaling Pathway. Med. Sci. Monit. 2016, 22, 284–288. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sun, L.; Zhao, L.; Li, P.; Liu, X.; Liang, F.; Jiang, Y.; Kang, N.; Gao, C.; Yang, J. Effect of hyperbaric oxygen therapy on HMGB1/NF-kappaB expression and prognosis of acute spinal cord injury: A randomized clinical trial. Neurosci. Lett. 2019, 692, 47–52. [Google Scholar] [CrossRef]
- Kang, N.; Hai, Y.; Yang, J.; Liang, F.; Gao, C.J. Hyperbaric oxygen intervention reduces secondary spinal cord injury in rats via regulation of HMGB1/TLR4/NF-kappaB signaling pathway. Int. J. Clin. Exp. Pathol. 2015, 8, 1141–1153. [Google Scholar]
- Yu, X.; Li, Y.G.; He, X.W.; Li, X.R.; Din, B.N.; Gan, Y.; Xu, M. Hyperbaric oxygen reduces inflammatory response in acute pancreatitis by inhibiting NF-kappaB activation. Eur. Surg. Res. 2009, 42, 130–135. [Google Scholar] [CrossRef]
- Liang, F.; Sun, L.; Yang, J.; Liu, X.H.; Zhang, J.; Zhu, W.Q.; Yang, L.; Nan, D. The effect of different atmosphere absolute hyperbaric oxygen on the expression of extracellular histones after traumatic brain injury in rats. Cell Stress Chaperones 2020, 25, 1013–1024. [Google Scholar] [CrossRef]
- Wang, G.; Wang, X.; Zheng, X.; Sun, S.; Zhao, J.; Long, Y.; Mao, Y. Acidic oligosaccharide sugar chain combined with hyperbaric oxygen delays D-galactose-induced brain senescence in mice via attenuating oxidative stress and neuroinflammation. Neurosci. Res. 2022, 185, 40–48. [Google Scholar] [CrossRef]
- Yildirim, A.O.; Eryilmaz, M.; Kaldirim, U.; Eyi, Y.E.; Tuncer, S.K.; Eroglu, M.; Durusu, M.; Topal, T.; Kurt, B.; Dilmen, S.; et al. Effectiveness of hyperbaric oxygen and ozone applications in tissue healing in generated soft tissue trauma model in rats: An experimental study. Ulus Travma Acil. Cerrahi. Derg. 2014, 20, 167–175. [Google Scholar] [CrossRef] [Green Version]
- Wang, C.; Niu, F.; Ren, N.; Wang, X.; Zhong, H.; Zhu, J.; Li, B. Hyperbaric Oxygen Improves Cerebral Ischemia/Reperfusion Injury in Rats Probably via Inhibition of Autophagy Triggered by the Downregulation of Hypoxia-Inducing Factor-1 Alpha. BioMed Res. Int. 2021, 2021, 6615685. [Google Scholar] [CrossRef]
- Oter, S.; Edremitlioglu, M.; Korkmaz, A.; Coskun, O.; Kilic, D.; Kisa, U.; Yaren, H.; Bilgic, H. Effects of hyperbaric oxygen treatment on liver functions, oxidative status and histology in septic rats. Intensive Care Med. 2005, 31, 1262–1268. [Google Scholar] [CrossRef]
- Lima, F.L.; Joazeiro, P.P.; Lancellotti, M.; de Hollanda, L.M.; de Araujo Lima, B.; Linares, E.; Augusto, O.; Brocchi, M.; Giorgio, S. Effects of hyperbaric oxygen on Pseudomonas aeruginosa susceptibility to imipenem and macrophages. Future Microbiol. 2015, 10, 179–189. [Google Scholar] [CrossRef]
- Tsuneyoshi, I.; Boyle, W.A., 3rd; Kanmura, Y.; Fujimoto, T. Hyperbaric hyperoxia suppresses growth of Staphylococcus aureus, including methicillin-resistant strains. J. Anesth. 2001, 15, 29–32. [Google Scholar] [CrossRef]
- Van, U. Inhibition of Toxin Production in Clostridium Perfringens In Vitro by Hyperbaric Oxygen. Antonie Van Leeuwenhoek 1965, 31, 181–186. [Google Scholar] [CrossRef]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vinkel, J.; Arenkiel, B.; Hyldegaard, O. The Mechanisms of Action of Hyperbaric Oxygen in Restoring Host Homeostasis during Sepsis. Biomolecules 2023, 13, 1228. https://doi.org/10.3390/biom13081228
Vinkel J, Arenkiel B, Hyldegaard O. The Mechanisms of Action of Hyperbaric Oxygen in Restoring Host Homeostasis during Sepsis. Biomolecules. 2023; 13(8):1228. https://doi.org/10.3390/biom13081228
Chicago/Turabian StyleVinkel, Julie, Bjoern Arenkiel, and Ole Hyldegaard. 2023. "The Mechanisms of Action of Hyperbaric Oxygen in Restoring Host Homeostasis during Sepsis" Biomolecules 13, no. 8: 1228. https://doi.org/10.3390/biom13081228





