The Effect of Dimethyl Sulfoxide on the Lysozyme Unfolding Kinetics, Thermodynamics, and Mechanism
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. The Differential Scanning Calorimetry (DSC) Method
2.3. Circular Dichroism (CD) Spectroscopy Study of Unfolding Equilibrium
2.4. Spectrofluorimetric Study of Unfolding Equilibrium
2.5. Spectrofluorimetric Study of Unfolding Kinetics
3. Results
3.1. DSC Experiments
3.2. Circular Dichroism Spectroscopy
3.3. Spectrofluorimetry
3.4. Notes on Kinetic Models of Denaturation
3.5. Isothermal Kinetics of Lysozyme Unfolding
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Castro, G.R.; Knubovets, T. Homogeneous Biocatalysis in Organic Solvents and Water-Organic Mixtures. Crit. Rev. Biotechnol. 2003, 23, 195–231. [Google Scholar] [CrossRef] [PubMed]
- Gerhards, T.; Mackfeld, U.; Bocola, M.; von Lieres, E.; Wiechert, W.; Pohl, M.; Rother, D. Influence of Organic Solvents on Enzymatic Asymmetric Carboligations. Adv. Synth. Catal. 2012, 354, 2805–2820. [Google Scholar] [CrossRef] [PubMed]
- Versari, A.; Ménard, R.; Lortie, R. Enzymatic hydrolysis of nitrides by an engineered nitrile hydratase (Papain Gln19Glu) in aqueous-organic media: Hydrolysis of Nitriles Using Papain Mutant. Biotechnol. Bioeng. 2002, 79, 9–14. [Google Scholar] [CrossRef] [PubMed]
- Spinozzi, F.; Ortore, M.G.; Sinibaldi, R.; Mariani, P.; Esposito, A.; Cinelli, S.; Onori, G. Microcalorimetric study of thermal unfolding of lysozyme in water/glycerol mixtures: An analysis by solvent exchange model. J. Chem. Phys. 2008, 129, 035101. [Google Scholar] [CrossRef] [PubMed]
- Gekko, K.; Timasheff, S.N. Thermodynamic and kinetic examination of protein stabilization by glycerol. Biochemistry 1981, 20, 4677–4686. [Google Scholar] [CrossRef] [PubMed]
- Griebenow, K.; Klibanov, A.M. On Protein Denaturation in Aqueous−Organic Mixtures but Not in Pure Organic Solvents. J. Am. Chem. Soc. 1996, 118, 11695–11700. [Google Scholar] [CrossRef]
- Romero, C.M.; Lozano, J.M.; Sancho, J.; Giraldo, G.I. Thermal stability of β-lactoglobulin in the presence of aqueous solution of alcohols and polyols. Int. J. Biol. Macromol. 2007, 40, 423–428. [Google Scholar] [CrossRef] [PubMed]
- Cinelli, S.; Onori, G.; Santucci, A. Effect of Aqueous Alcohol Solutions on the Thermal Transition of Lysozyme: A Calorimetric Study. J. Phys. Chem. B 1997, 101, 8029–8034. [Google Scholar] [CrossRef]
- Kamal, M.Z.; Yedavalli, P.; Deshmukh, M.V.; Rao, N.M. Lipase in aqueous-polar organic solvents: Activity, structure, and stability: Lipase in Organic Solvents. Protein Sci. 2013, 22, 904–915. [Google Scholar] [CrossRef] [PubMed]
- Jacobson, A.L.; Turner, C.L. Specific solvent effects on the thermal denaturation of ribonuclease. Effect of dimethyl sulfoxide and p-dioxane on thermodynamics of denaturation. Biochemistry 1980, 19, 4534–4538. [Google Scholar] [CrossRef] [PubMed]
- Azevedo, A.M.; Prazeres, D.M.; Cabral, J.M.; Fonseca, L.P. Stability of free and immobilised peroxidase in aqueous–organic solvents mixtures. J. Mol. Catal. B Enzym. 2001, 15, 147–153. [Google Scholar] [CrossRef]
- Mozhaev, V.V.; Khmelnitsky, Y.L.; Sergeeva, M.V.; Belova, A.B.; Klyachko, N.L.; Levashov, A.V.; Martinek, K. Catalytic activity and denaturation of enzymes in water/organic cosolvent mixtures. alpha-Chymotrypsin and laccase in mixed water/alcohol, water/glycol and water/formamide solvents. Eur. J. Biochem. 1989, 184, 597–602. [Google Scholar] [CrossRef] [PubMed]
- Simon, L.M.; Kotormán, M.; Szabó, A.; Nemcsók, J.; Laczkó, I. The effects of organic solvent/water mixtures on the structure and catalytic activity of porcine pepsin. Process Biochem. 2007, 42, 909–912. [Google Scholar] [CrossRef]
- Torreggiani, A.; Di Foggia, M.; Manco, I.; De Maio, A.; Markarian, S.A.; Bonora, S. Effect of sulfoxides on the thermal denaturation of hen lysozyme: A calorimetric and Raman study. J. Mol. Struct. 2008, 891, 115–122. [Google Scholar] [CrossRef]
- Velicelebi, G.; Sturtevant, J.M. Thermodynamics of the denaturation of lysozyme in alcohol-water mixtures. Biochemistry 1979, 18, 1180–1186. [Google Scholar] [CrossRef]
- Fujita, Y.; Miyanaga, A.; Noda, Y. Effect of Alcohols on the Thermal Denaturation of Lysozyme as Measured by Differential Scanning Calorimetry. Bull. Chem. Soc. Jpn. 1979, 52, 3659–3662. [Google Scholar] [CrossRef]
- Brandts, J.F.; Hunt, L. Thermodynamics of protein denaturation. III. Denaturation of ribonuclease in water and in aqueous urea and aqueous ethanol mixtures. J. Am. Chem. Soc. 1967, 89, 4826–4838. [Google Scholar] [CrossRef]
- Lai, B.; Cao, A.; Lai, L. Organic cosolvents and hen egg white lysozyme folding. Biochim. Biophys. Acta BBA Protein Struct. Mol. Enzymol. 2000, 1543, 115–122. [Google Scholar] [CrossRef]
- Blumlein, A.; McManus, J.J. Reversible and non-reversible thermal denaturation of lysozyme with varying pH at low ionic strength. Biochim. Biophys. Acta BBA Proteins Proteom. 2013, 1834, 2064–2070. [Google Scholar] [CrossRef]
- Babu, K.R.; Bhakuni, V. Ionic-Strength-Dependent Transition of Hen Egg-White Lysozyme at Low PH to a Compact State and its Aggregation on Thermal Denaturation. Eur. J. Biochem. 1997, 245, 781–789. [Google Scholar] [CrossRef]
- Hédoux, A.; Guinet, Y.; Paccou, L. Analysis of the Mechanism of Lysozyme Pressure Denaturation from Raman Spectroscopy Investigations, and Comparison with Thermal Denaturation. J. Phys. Chem. B 2011, 115, 6740–6748. [Google Scholar] [CrossRef] [PubMed]
- Ortore, M.G.; Mariani, P.; Carsughi, F.; Cinelli, S.; Onori, G.; Teixeira, J.; Spinozzi, F. Preferential solvation of lysozyme in water/ethanol mixtures. J. Chem. Phys. 2011, 135, 245103. [Google Scholar] [CrossRef]
- Hédoux, A.; Krenzlin, S.; Paccou, L.; Guinet, Y.; Flament, M.-P.; Siepmann, J. Influence of urea and guanidine hydrochloride on lysozyme stability and thermal denaturation; a correlation between activity, protein dynamics and conformational changes. Phys. Chem. Chem. Phys. 2010, 12, 13189. [Google Scholar] [CrossRef] [PubMed]
- Kovrigin, E.L.; Potekhin, S.A. On the stabilizing action of protein denaturants: Acetonitrile effect on stability of lysozyme in aqueous solutions. Biophys. Chem. 2000, 83, 45–59. [Google Scholar] [CrossRef]
- Kamiyama, T.; Liu, H.L.; Kimura, T. Preferential solvation of lysozyme by dimethyl sulfoxide in binary solutions of water and dimethyl sulfoxide. J. Therm. Anal. Calorim. 2009, 95, 353–359. [Google Scholar] [CrossRef]
- Voets, I.K.; Cruz, W.A.; Moitzi, C.; Lindner, P.; Arêas, E.P.G.; Schurtenberger, P. DMSO-Induced Denaturation of Hen Egg White Lysozyme. J. Phys. Chem. B 2010, 114, 11875–11883. [Google Scholar] [CrossRef] [PubMed]
- Bhattacharjya, S.; Balaram, P. Effects of organic solvents on protein structures: Observation of a structured helical core in hen egg-white lysozyme in aqueous dimethylsulfoxide. Proteins Struct. Funct. Bioinform. 1997, 29, 492–507. [Google Scholar] [CrossRef]
- Fujita, Y.; Izumiguchi, S.; Noda, Y. Effect of dimethylsulfoxide and its homologues on the thermal denaturation of lysozyme as measured by differential scanning calorimetry. Int. J. Pept. Protein Res. 1982, 19, 25–31. [Google Scholar] [CrossRef]
- Ayad, M.; Eskioglu, E.; Mericle, R.A. Onyx®: A unique neuroembolic agent. Expert Rev. Med. Devices 2006, 3, 705–715. [Google Scholar] [CrossRef]
- Wiedenmann, R.; Lau, H. A gel containing dexpanthenol, heparin and dimethyl sulfoxide in accident and sport medicine. Therapiewoche 1984, 34, 1840–1842. [Google Scholar]
- Barua, J.M.; Arance, I.; Angulo, J.C.; Riedl, C.R. A systematic review and meta-analysis on the efficacy of intravesical therapy for bladder pain syndrome/interstitial cystitis. Int. Urogynecol. J. 2016, 27, 1137–1147. [Google Scholar] [CrossRef]
- Segawa, S.-I.; Sugihara, M. Characterization of the transition state of Lysozyme unfolding. I. Effect of protein-solvent interactions on the transition state. Biopolymers 1984, 23, 2473–2488. [Google Scholar] [CrossRef] [PubMed]
- Pohl, F.M. Kinetics of Reversible Denaturation of Trypsin in Water and Water-Ethanol Mixtures. Eur. J. Biochem. 1968, 7, 146–152. [Google Scholar] [CrossRef] [PubMed]
- Armarego, W.L.F. Purification of Laboratory Chemicals, 8th ed.; Butterworth-Heinemann: Amsterdam, The Netherlands, 2017; ISBN 978-0-12-805457-4. [Google Scholar]
- Pace, C.N.; Vajdos, F.; Fee, L.; Grimsley, G.; Gray, T. How to measure and predict the molar absorption coefficient of a protein. Protein Sci. 1995, 4, 2411–2423. [Google Scholar] [CrossRef] [PubMed]
- Sedov, I.A.; Muhametzyanov, T.A.; Solomonov, B.N. A procedure for calibration of differential scanning calorimeters. Thermochim. Acta 2016, 639, 10–13. [Google Scholar] [CrossRef]
- Wu, S.; Ding, Y.; Zhang, G. Mechanic Insight into Aggregation of Lysozyme by Ultrasensitive Differential Scanning Calorimetry and Sedimentation Velocity. J. Phys. Chem. B 2015, 119, 15789–15795. [Google Scholar] [CrossRef] [PubMed]
- Clever, H.L.; Pigott, S.P. Enthalpies of mixing of dimethylsulfoxide with water and with several ketones at 298.15 K. J. Chem. Thermodyn. 1971, 3, 221–225. [Google Scholar] [CrossRef]
- Cao, X.M.; Tian, Y.; Wang, Z.Y.; Liu, Y.W.; Wang, C.X. Effects of protein and phosphate buffer concentrations on thermal denaturation of lysozyme analyzed by isoconversional method. Bioengineered 2016, 7, 235–240. [Google Scholar] [CrossRef] [PubMed]
- Privalov, G.; Kavina, V.; Freire, E.; Privalov, P.L. Precise Scanning Calorimeter for Studying Thermal Properties of Biological Macromolecules in Dilute Solution. Anal. Biochem. 1995, 232, 79–85. [Google Scholar] [CrossRef]
- Burova, T.V.; Grinberg, N.V.; Grinberg, V.Y.; Rariy, R.V.; Klibanov, A.M. Calorimetric evidence for a native-like conformation of hen egg-white lysozyme dissolved in glycerol. Biochim. Biophys. Acta BBA Protein Struct. Mol. Enzymol. 2000, 1478, 309–317. [Google Scholar] [CrossRef]
- Kovrigin, E.L.; Potekhin, S.A. Preferential Solvation Changes upon Lysozyme Heat Denaturation in Mixed Solvents. Biochemistry 1997, 36, 9195–9199. [Google Scholar] [CrossRef]
- Sedov, I.A.; Magsumov, T.I. Molecular dynamics study of unfolding of lysozyme in water and its mixtures with dimethyl sulfoxide. J. Mol. Graph. Model. 2017, 76, 466–474. [Google Scholar] [CrossRef] [PubMed]
- Ahern, T.; Klibanov, A. The mechanisms of irreversible enzyme inactivation at 100C. Science 1985, 228, 1280–1284. [Google Scholar] [CrossRef]
- Daniel, R.M.; Dines, M.; Petach, H.H. The denaturation and degradation of stable enzymes at high temperatures. Biochem. J. 1996, 317, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Knubovets, T.; Osterhout, J.J.; Connolly, P.J.; Klibanov, A.M. Structure, thermostability, and conformational flexibility of hen egg-white lysozyme dissolved in glycerol. Proc. Natl. Acad. Sci. USA 1999, 96, 1262–1267. [Google Scholar] [CrossRef] [PubMed]
- Jha, S.K.; Marqusee, S. Kinetic evidence for a two-stage mechanism of protein denaturation by guanidinium chloride. Proc. Natl. Acad. Sci. USA 2014, 111, 4856–4861. [Google Scholar] [CrossRef] [PubMed]
- Newcomer, R.L.; Fraser, L.C.R.; Teschke, C.M.; Alexandrescu, A.T. Mechanism of Protein Denaturation: Partial Unfolding of the P22 Coat Protein I-Domain by Urea Binding. Biophys. J. 2015, 109, 2666–2677. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Giugliarelli, A.; Paolantoni, M.; Morresi, A.; Sassi, P. Denaturation and Preservation of Globular Proteins: The Role of DMSO. J. Phys. Chem. B 2012, 116, 13361–13367. [Google Scholar] [CrossRef] [PubMed]
- García-Hernández, E.; Hernández-Arana, A.; Zubillaga, R.; Rojo-Domínguez, A. Spectroscopic and thermodynamic evidence for a complex denaturation mechanism of bovine β-lactoglobulin A. IUBMB Life 1998, 45, 761–768. [Google Scholar] [CrossRef]
- Seelig, J.; Schönfeld, H.-J. Thermal protein unfolding by differential scanning calorimetry and circular dichroism spectroscopy Two-state model versus sequential unfolding. Q. Rev. Biophys. 2016, 49, e9. [Google Scholar] [CrossRef]
- Esposito, A.; Comez, L.; Cinelli, S.; Scarponi, F.; Onori, G. Influence of Glycerol on the Structure and Thermal Stability of Lysozyme: A Dynamic Light Scattering and Circular Dichroism Study. J. Phys. Chem. B 2009, 113, 16420–16424. [Google Scholar] [CrossRef] [PubMed]
- Poklar, N.; Petrovčič, N.; Oblak, M.; Vesnaver, G. Thermodynamic stability of ribonuclease A in alkylurea solutions and preferential solvation changes accompanying its thermal denaturation: A calorimetric and spectroscopic study. Protein Sci. 1999, 8, 832–840. [Google Scholar] [CrossRef] [PubMed]
- Fink, A.L.; Painter, B. Characterization of the unfolding of ribonuclease A in aqueous methanol solvents. Biochemistry 1987, 26, 1665–1671. [Google Scholar] [CrossRef] [PubMed]
- Luo, J.-J.; Wu, F.-G.; Yu, J.-S.; Wang, R.; Yu, Z.-W. Denaturation Behaviors of Two-State and Non-Two-State Proteins Examined by an Interruption–Incubation Protocol. J. Phys. Chem. B 2011, 115, 8901–8909. [Google Scholar] [CrossRef]
- Ghosh, S.; Chattoraj, S.; Chowdhury, R.; Bhattacharyya, K. Structure and dynamics of lysozyme in DMSO–water binary mixture: Fluorescence correlation spectroscopy. RSC Adv. 2014, 4, 14378. [Google Scholar] [CrossRef]
- Privalov, P.L.; Khechinashvili, N.N. A thermodynamic approach to the problem of stabilization of globular protein structure: A calorimetric study. J. Mol. Biol. 1974, 86, 665–684. [Google Scholar] [CrossRef]
- Mazurenko, S.; Kunka, A.; Beerens, K.; Johnson, C.M.; Damborsky, J.; Prokop, Z. Exploration of Protein Unfolding by Modelling Calorimetry Data from Reheating. Sci. Rep. 2017, 7, 16321. [Google Scholar] [CrossRef]
- Xing, L.; Lin, K.; Zhou, X.; Liu, S.; Luo, Y. Multistate Mechanism of Lysozyme Denaturation through Synchronous Analysis of Raman Spectra. J. Phys. Chem. B 2016, 120, 10660–10667. [Google Scholar] [CrossRef]
- Kiefhaber, T. Kinetic traps in lysozyme folding. Proc. Natl. Acad. Sci. USA 1995, 92, 9029–9033. [Google Scholar] [CrossRef]
- Muttathukattil, A.N.; Singh, P.C.; Reddy, G. Role of Disulfide Bonds and Topological Frustration in the Kinetic Partitioning of Lysozyme Folding Pathways. J. Phys. Chem. B 2019, 123, 3232–3241. [Google Scholar] [CrossRef]
- Mukhametzyanov, T.A.; Sedov, I.A.; Solomonov, B.N.; Schick, C. Fast scanning calorimetry of lysozyme unfolding at scanning rates from 5 K/min to 500,000 K/min. Biochim. Biophys. Acta BBA Gen. Subj. 2018, 1862, 2024–2030. [Google Scholar] [CrossRef] [PubMed]
- Splinter, R.; van Herwaarden, A.W.; van Wetten, I.A.; Pfreundt, A.; Svendsen, W.E. Fast differential scanning calorimetry of liquid samples with chips. Thermochim. Acta 2015, 603, 162–171. [Google Scholar] [CrossRef]
- Lepock, J.R.; Ritchie, K.P.; Kolios, M.C.; Rodahl, A.M.; Heinz, K.A.; Kruuv, J. Influence of transition rates and scan rate on kinetic simulations of differential scanning calorimetry profiles of reversible and irreversible protein denaturation. Biochemistry 1992, 31, 12706–12712. [Google Scholar] [CrossRef] [PubMed]
- Guise, A.D.; Chaudhuri, J.B. Initial protein concentration and residual denaturant concentration strongly affect the batch refolding of hen egg white lysozyme. Biotechnol. Bioprocess Eng. 2001, 6, 410–418. [Google Scholar] [CrossRef]
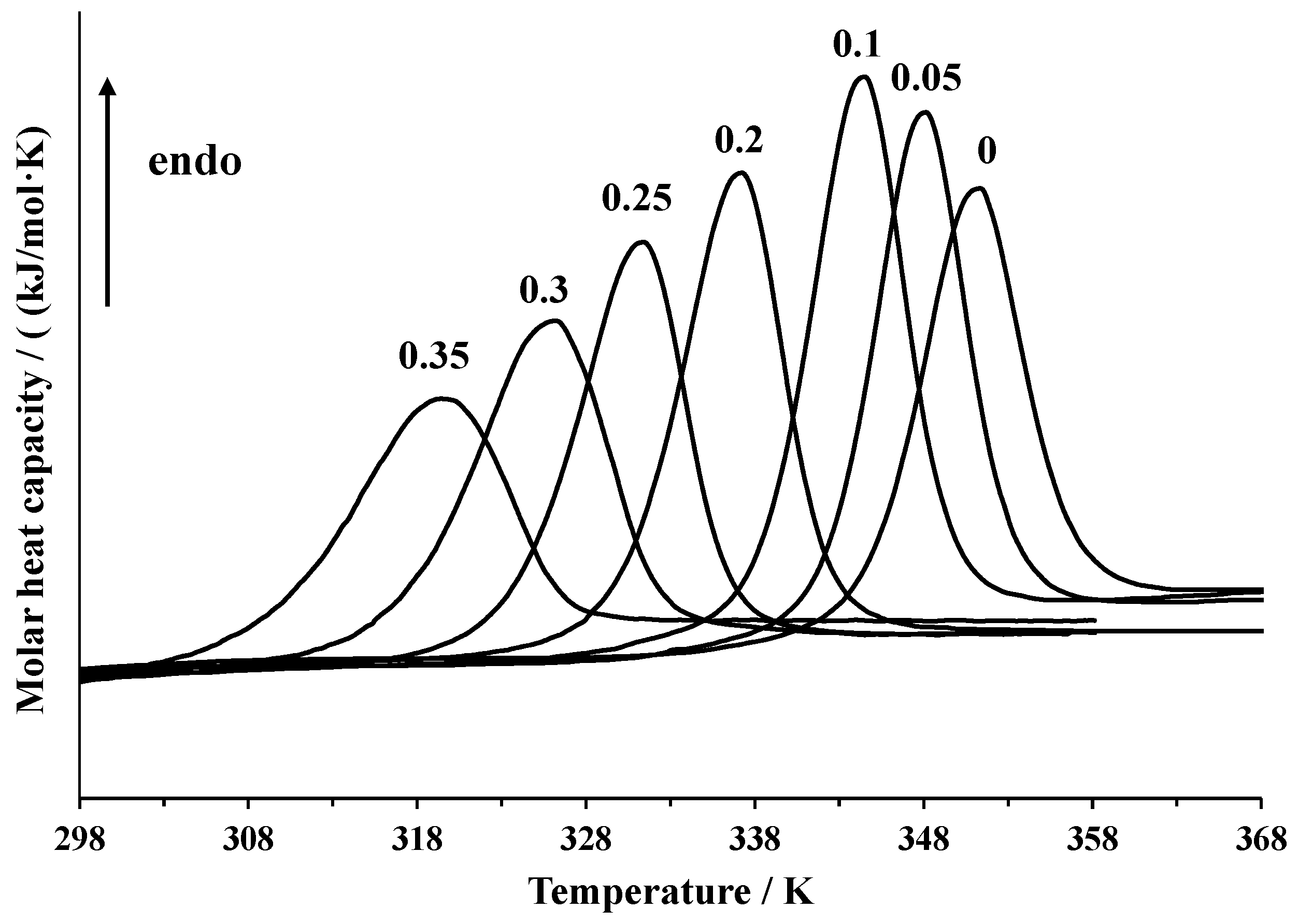








| x1 | Td (K) | ΔdHcal (kJ·mol−1) |
|---|---|---|
| 0 | 351.0 | 494 ± 10 |
| 0.05 | 347.6 | 539 ± 9 |
| 0.1 | 344.7 | 569 ± 10 |
| 0.2 | 337.2 | 508 ± 9 |
| 0.25 | 331.3 | 476 ± 9 |
| 0.3 | 326.0 | 415 ± 12 |
| 0.35 | 319.4 | 337 ± 18 |
| x1 | v (K·min−1) | Td (K) | Td(eq). (K) |
|---|---|---|---|
| 0 | 0.1 | 350.4 | 350.3 |
| 0.25 | 350.4 | ||
| 0.5 | 350.6 | ||
| 1.0 | 351.0 | ||
| 2.0 | 351.4 | ||
| 0.05 | 0.5 | 347.2 | 347.0 |
| 1.0 | 347.6 | ||
| 2.0 | 348.1 | ||
| 0.10 | 0.5 | 343.9 | 343.7 |
| 1.0 | 344.7 | ||
| 2.0 | 345.2 | ||
| 0.20 | 0.5 | 336.5 | 336.1 |
| 1.0 | 337.2 | ||
| 2.0 | 338.1 | ||
| 0.25 | 0.5 | 330.2 | 329.4 |
| 1.0 | 331.3 | ||
| 2.0 | 332.9 | ||
| 0.30 | 0.5 | 324.4 | 323.8 |
| 1.0 | 326.0 | ||
| 2.0 | 327.2 | ||
| 0.35 | 0.5 | 316.1 | 312.8 |
| 1.0 | 319.4 |
| x1 | Td − TCD (K) |
|---|---|
| 0 | 0.6 |
| 0.1 | 1.7 |
| 0.2 | 3.6 |
| 0.25 | 4.4 |
| 0.3 | 7.3 |
| 0.35 | 11.7 |
| x1 | E1 (kJ·mol−1) | ln(A1/min−1) |
|---|---|---|
| 0.25 | 231 ± 20 | 86.6 ± 4 |
| 0.30 | 233 ± 18 | 88.5 ± 4 |
| 0.35 | 222 ± 15 | 85.7 ± 2 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Magsumov, T.; Fatkhutdinova, A.; Mukhametzyanov, T.; Sedov, I. The Effect of Dimethyl Sulfoxide on the Lysozyme Unfolding Kinetics, Thermodynamics, and Mechanism. Biomolecules 2019, 9, 547. https://doi.org/10.3390/biom9100547
Magsumov T, Fatkhutdinova A, Mukhametzyanov T, Sedov I. The Effect of Dimethyl Sulfoxide on the Lysozyme Unfolding Kinetics, Thermodynamics, and Mechanism. Biomolecules. 2019; 9(10):547. https://doi.org/10.3390/biom9100547
Chicago/Turabian StyleMagsumov, Timur, Alisa Fatkhutdinova, Timur Mukhametzyanov, and Igor Sedov. 2019. "The Effect of Dimethyl Sulfoxide on the Lysozyme Unfolding Kinetics, Thermodynamics, and Mechanism" Biomolecules 9, no. 10: 547. https://doi.org/10.3390/biom9100547
APA StyleMagsumov, T., Fatkhutdinova, A., Mukhametzyanov, T., & Sedov, I. (2019). The Effect of Dimethyl Sulfoxide on the Lysozyme Unfolding Kinetics, Thermodynamics, and Mechanism. Biomolecules, 9(10), 547. https://doi.org/10.3390/biom9100547






