Zhankuic Acids A, B and C from Taiwanofungus camphoratus Act as Cytotoxicity Enhancers by Regulating P-Glycoprotein in Multi-Drug Resistant Cancer Cells
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals and Reagents
2.2. Cell Lines
2.3. SRB Cytotoxicity Assay
2.4. Intracellular Calcein Accumulation Assay
2.5. Real-Time Quantitative RT-PCR
2.6. MDR1 Shift Assay
2.7. Rhodamine123 and Doxorubicin Efflux Assay
2.8. P-gp ATPase Activity Assay
2.9. Cell Cycle Analysis
2.10. Apoptosis Assay
2.11. Statistical Analysis
3. Results
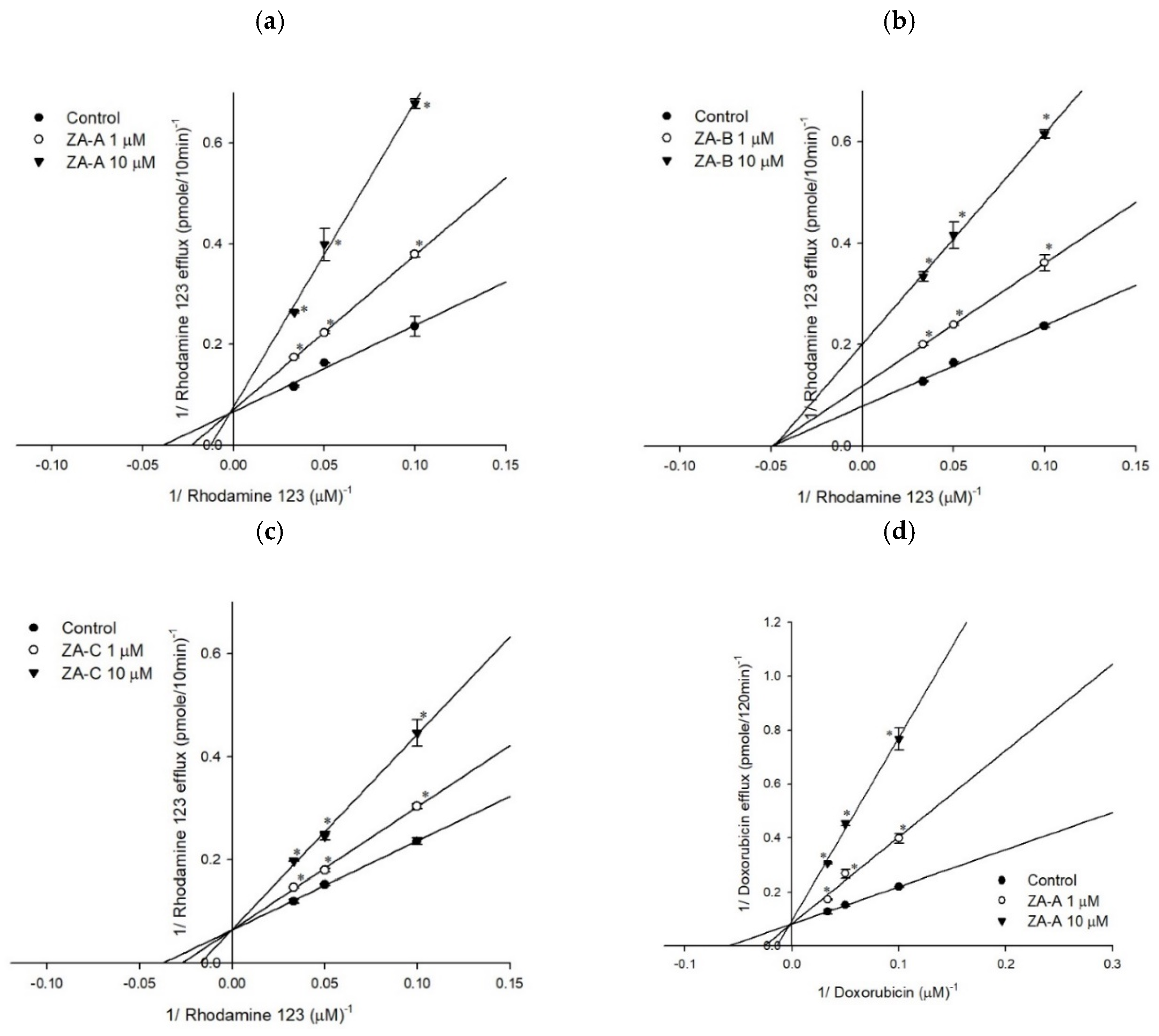
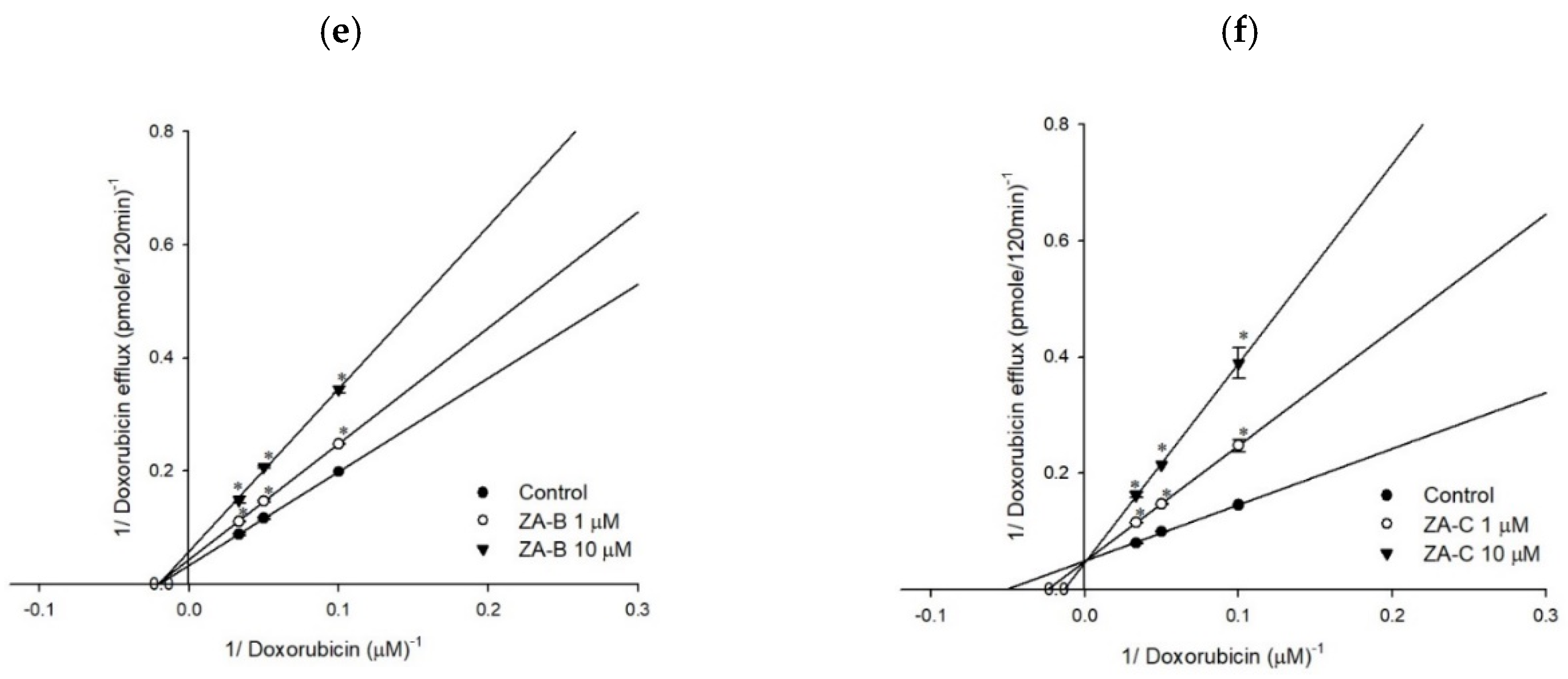
3.1. P-gp Efflux Function was Inhibited by ZA-A, ZA-B and ZA-C
3.2. ZA-A, ZA-B and ZA-C were not P-gp Substrates and the ABCB1 mRNA Levels were not Influenced by These Triterpenoids
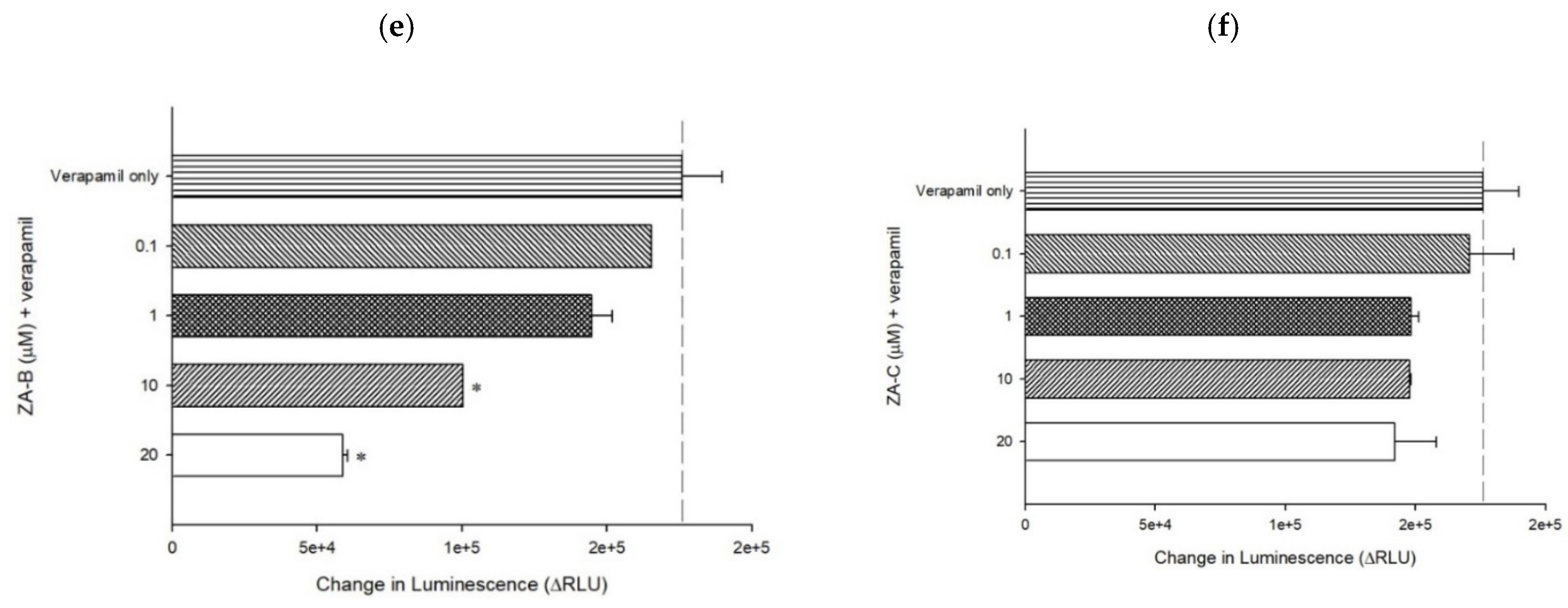
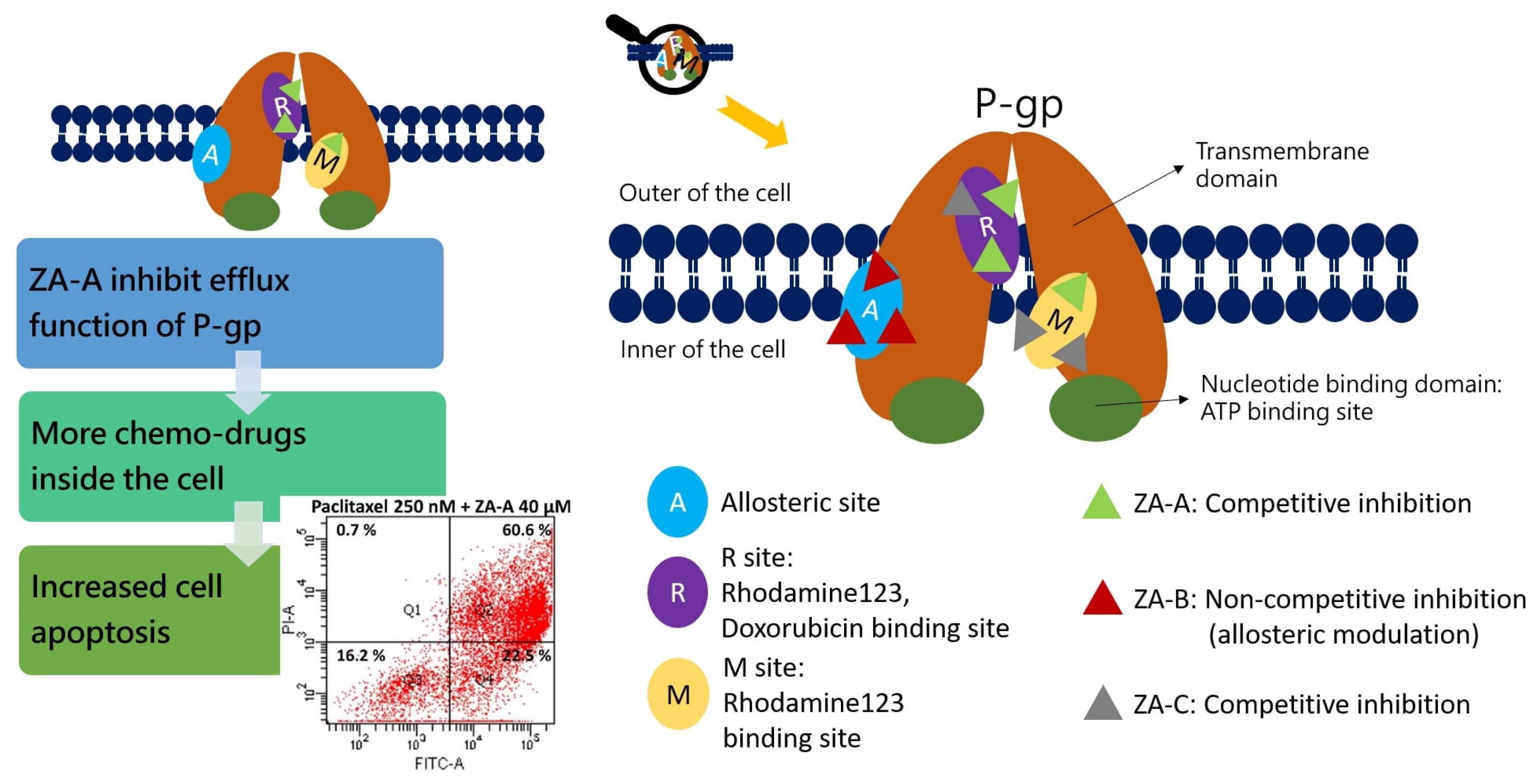
3.3. The Inhibitory Mechanisms and ATPase Interactions between P-gp and ZA-A, ZA-B and ZA-C
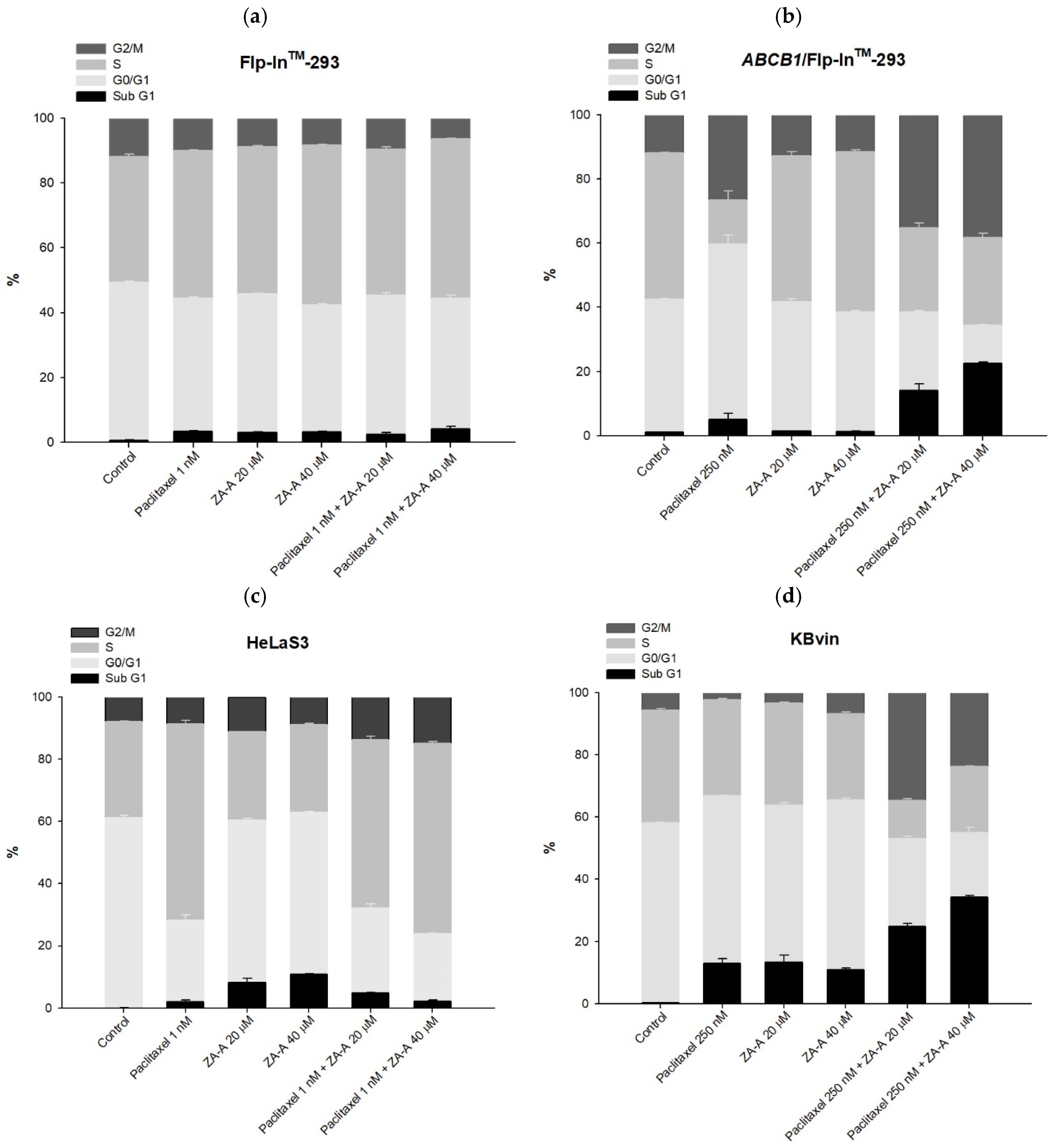
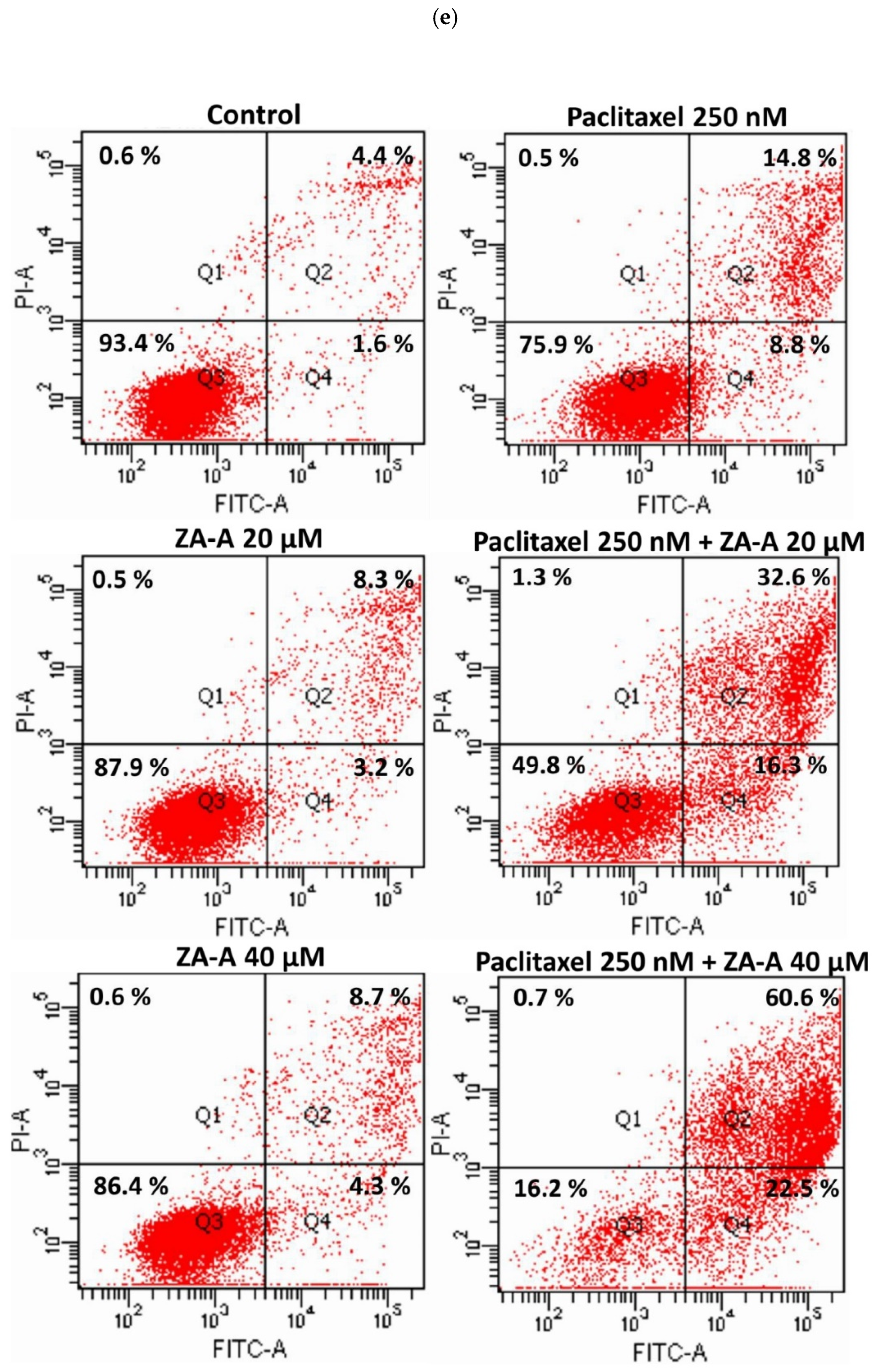
3.4. The MDR Reversal Effects and Molecular Mechanisms of ZA-A, ZA-B and ZA-C
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Holohan, C.; Van Schaeybroeck, S.; Longley, D.B.; Johnston, P.G. Cancer drug resistance: An evolving paradigm. Nat. Rev. Cancer 2013, 13, 714–726. [Google Scholar] [CrossRef] [PubMed]
- Szakacs, G.; Paterson, J.K.; Ludwig, J.A.; Booth-Genthe, C.; Gottesman, M.M. Targeting multidrug resistance in cancer. Nat. Rev. Drug Discov. 2006, 5, 219–234. [Google Scholar] [CrossRef] [PubMed]
- Fletcher, J.I.; Williams, R.T.; Henderson, M.J.; Norris, M.D.; Haber, M. ABC transporters as mediators of drug resistance and contributors to cancer cell biology. Drug Resist. Updat. 2016, 26, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Dean, M.; Rzhetsky, A.; Allikmets, R. The human ATP-binding cassette (ABC) transporter superfamily. Genome Res. 2001, 11, 1156–1166. [Google Scholar] [CrossRef]
- Ozols, R.F.; Cunnion, R.E.; Klecker, R.W., Jr.; Hamilton, T.C.; Ostchega, Y.; Parrillo, J.E.; Young, R.C. Verapamil and adriamycin in the treatment of drug-resistant ovarian cancer patients. J. Clin. Oncol. 1987, 5, 641–647. [Google Scholar] [CrossRef]
- Sharom, F.J. ABC multidrug transporters: Structure, function and role in chemoresistance. Pharmacogenomics 2007, 9, 105–127. [Google Scholar] [CrossRef]
- Leonard, G.D.; Fojo, T.; Bates, S.E. The role of ABC transporters in clinical practice. Oncologist 2003, 8, 411–424. [Google Scholar] [CrossRef]
- Li, W.; Zhang, H.; Assaraf, Y.G.; Zhao, K.; Xu, X.; Xie, J.; Yang, D.H.; Chen, Z.S. Overcoming ABC transporter-mediated multidrug resistance: Molecular mechanisms and novel therapeutic drug strategies. Drug Resist. Updat. 2016, 27, 14–29. [Google Scholar] [CrossRef]
- Teng, Y.N.; Hsieh, Y.W.; Hung, C.C.; Lin, H.Y. Demethoxycurcumin modulates human P-glycoprotein function via uncompetitive inhibition of ATPase hydrolysis activity. J. Agric. Food Chem. 2015, 63, 847–855. [Google Scholar] [CrossRef]
- Sheu, M.J.; Teng, Y.N.; Chen, Y.Y.; Hung, C.C. The functional influences of common ABCB1 genetic variants on the inhibition of P-glycoprotein by Antrodia cinnamomea extracts. PLoS ONE 2014, 9, e89622. [Google Scholar] [CrossRef]
- Shi, L.S.; Chao, C.H.; Shen, D.Y.; Chan, H.H.; Chen, C.H.; Liao, Y.R.; Wu, S.J.; Leu, Y.L.; Shen, Y.C.; Kuo, Y.H.; et al. Biologically active constituents from the fruiting body of Taiwanofungus camphoratus. Bioorg. Med. Chem. 2011, 19, 677–683. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.H.; Yang, S.W.; Shen, Y.C. New steroid acids from Antrodia cinnamomea, a fungal parasite of Cinnamomum micranthum. J. Nat. Prod. 1995, 58, 1655–1661. [Google Scholar] [CrossRef] [PubMed]
- Geethangili, M.; Tzeng, Y.M. Review of Pharmacological Effects of Antrodia camphorata and Its Bioactive Compounds. Evid. Based Complement. Alternat. Med. 2011, 2011, 212641. [Google Scholar] [CrossRef] [PubMed]
- Yeh, C.T.; Rao, Y.K.; Yao, C.J.; Yeh, C.F.; Li, C.H.; Chuang, S.E.; Luong, J.H.; Lai, G.M.; Tzeng, Y.M. Cytotoxic triterpenes from Antrodia camphorata and their mode of action in HT-29 human colon cancer cells. Cancer Lett. 2009, 285, 73–79. [Google Scholar] [CrossRef]
- Liu, D.Z. A review of ergostane and cucurbitane triterpenoids of mushroom origin. Nat. Prod. Res. 2014, 28, 1099–1105. [Google Scholar] [CrossRef]
- Wu, S.J.; Leu, Y.L.; Chen, C.H.; Chao, C.H.; Shen, D.Y.; Chan, H.H.; Lee, E.J.; Wu, T.S.; Wang, Y.H.; Shen, Y.C.; et al. Camphoratins A-J, potent cytotoxic and anti-inflammatory triterpenoids from the fruiting body of Taiwanofungus camphoratus. J. Nat. Prod. 2010, 73, 1756–1762. [Google Scholar] [CrossRef]
- Hung, C.C.; Chiou, M.H.; Teng, Y.N.; Hsieh, Y.W.; Huang, C.L.; Lane, H.Y. Functional impact of ABCB1 variants on interactions between P-glycoprotein and methadone. PLoS ONE 2013, 8, e59419. [Google Scholar] [CrossRef]
- Kai, W.; Yating, S.; Lin, M.; Kaiyong, Y.; Baojin, H.; Wu, Y.; Fangzhou, Y.; Yan, C. Natural product toosendanin reverses the resistance of human breast cancer cells to adriamycin as a novel PI3K inhibitor. Biochem. Pharmacol. 2018, 152, 153–164. [Google Scholar] [CrossRef]
- Abraham, I.; Jain, S.; Wu, C.P.; Khanfar, M.A.; Kuang, Y.; Dai, C.L.; Shi, Z.; Chen, X.; Fu, L.; Ambudkar, S.V.; et al. Marine sponge-derived sipholane triterpenoids reverse P-glycoprotein (ABCB1)-mediated multidrug resistance in cancer cells. Biochem. Pharmacol. 2010, 80, 1497–1506. [Google Scholar] [CrossRef]
- Liu, D.L.; Li, Y.J.; Yao, N.; Xu, J.; Chen, Z.S.; Yiu, A.; Zhang, C.X.; Ye, W.C.; Zhang, D.M. Acerinol, a cyclolanstane triterpenoid from Cimicifuga acerina, reverses ABCB1-mediated multidrug resistance in HepG2/ADM and MCF-7/ADR cells. Eur. J. Pharmacol. 2014, 733, 34–44. [Google Scholar] [CrossRef]
- Chang, C.Y.; Huang, Z.N.; Yu, H.H.; Chang, L.H.; Li, S.L.; Chen, Y.P.; Lee, K.Y.; Chuu, J.J. The adjuvant effects of Antrodia Camphorata extracts combined with anti-tumor agents on multidrug resistant human hepatoma cells. J. Ethnopharmacol. 2008, 118, 387–395. [Google Scholar] [CrossRef] [PubMed]
- Li, S.L.; Huang, Z.N.; Hsieh, H.H.; Yu, W.C.; Tzeng, W.Y.; Lee, G.Y.; Chen, Y.P.; Chang, C.Y.; Chuu, J.J. The Augmented Anti-Tumor Effects of Antrodia camphorata Co-Fermented with Chinese Medicinal Herb in Human Hepatoma Cells. Am. J. Chin. Med. 2009, 37, 771–783. [Google Scholar] [CrossRef] [PubMed]
- Montanari, F.; Ecker, G.F. Prediction of drug-ABC-transporter interaction--Recent advances and future challenges. Adv. Drug Deliv. Rev. 2015, 86, 17–26. [Google Scholar] [CrossRef] [PubMed]
- Ambudkar, S.V.; Dey, S.; Hrycyna, C.A.; Ramachandra, M.; Pastan, I.; Gottesman, M.M. Biochemical, Cellular, and Pharmacological Aspects of the Multidrug Transporter. Annu. Rev. Pharmacol. Toxicol. 1999, 39, 361–398. [Google Scholar] [CrossRef]
- Li, P.; Chen, S.Y.; Shen, S.X.; Liu, L.X.; Xu, J.H.; Zhang, Z.Q. Ethyl lucidenates A reverses P-glycoprotein mediated vincristine resistance in K562/A02 cells. Nat. Prod. Res. 2019, 33, 732–735. [Google Scholar] [CrossRef]
- Xie, G.; Yu, X.; Liang, H.; Chen, J.; Tang, X.; Wu, S.; Liao, C. Pristimerin overcomes adriamycin resistance in breast cancer cells through suppressing Akt signaling. Oncol. Lett. 2016, 11, 3111–3116. [Google Scholar] [CrossRef][Green Version]
- Du, J.R.; Long, F.Y.; Chen, C. Research Progress on Natural Triterpenoid Saponins in the Chemoprevention and Chemotherapy of Cancer. Enzymes 2014, 36, 95–130. [Google Scholar] [CrossRef]
- Xu, X.H.; Li, T.; Fong, C.M.; Chen, X.; Chen, X.J.; Wang, Y.T.; Huang, M.Q.; Lu, J.J. Saponins from Chinese Medicines as Anticancer Agents. Molecules 2016, 21. [Google Scholar] [CrossRef]
- Shen, Y.C.; Wang, Y.H.; Chou, Y.C.; Chen, C.F.; Lin, L.C.; Chang, T.T.; Tien, J.H.; Chou, C.J. Evaluation of the anti-inflammatory activity of zhankuic acids isolated from the fruiting bodies of Antrodia camphorata. Planta Med. 2004, 70, 310–314. [Google Scholar] [CrossRef]
- Chen, Y.F.; Shiau, A.L.; Wang, S.H.; Yang, J.S.; Chang, S.J.; Wu, C.L.; Wu, T.S. Zhankuic acid A isolated from Taiwanofungus camphoratus is a novel selective TLR4/MD-2 antagonist with anti-inflammatory properties. J. Immunol. 2014, 192, 2778–2786. [Google Scholar] [CrossRef]


| Cell Line | Flp-InTM293 | ABCB1/Flp-InTM293 | ||
| Compound | IC50 ± S.E. (nM) | RF | IC50 ± S.E. (nM) | RF |
| Doxorubicin | 84.783 ± 0.856 | 1.0 | 767.708 ± 135.973 | 1.0 |
| + ZA-A (10 μM) | 53.276 ± 1.913 | 1.6 | 462.964 ± 27.210 | 1.7 |
| + ZA-A (20 μM) | 44.824 ± 0.436 | 1.9 | 433.570 ± 9.658 | 1.8 |
| + ZA-A (40 μM) | 37.224 ± 2.369 * | 2.3 | 124.221 ± 14.013 * | 6.2 |
| + ZA-B (10 μM) | 50.806 ± 7.431 | 1.7 | 462.296 ± 39.402 | 1.7 |
| + ZA-B (20 μM) | 48.319 ± 6.729 | 1.8 | 414.329 ± 18.101 | 1.9 |
| + ZA-B (40 μM) | 42.509 ± 2.711 * | 2.0 | 208.154 ± 3.263 * | 3.7 |
| + ZA-C (10 μM) | 64.002 ± 1.282 | 1.3 | 555.031 ± 6.192 | 1.4 |
| + ZA-C (20 μM) | 56.275 ± 2.034 | 1.5 | 507.168 ± 26.490 | 1.5 |
| + ZA-C (40 μM) | 45.570 ± 0.215 | 1.9 | 215.486 ± 105.299 * | 3.6 |
| Cell Line | Flp-InTM293 | ABCB1/Flp-InTM293 | ||
| Compound | IC50 ± S.E. (nM) | RF | IC50 ± S.E. (nM) | RF |
| Paclitaxel | 6.556 ± 0.037 | 1.0 | 381.099 ± 19.440 | 1.0 |
| + ZA-A (10 μM) | 6.342 ± 0.336 | 1.0 | 119.886 ± 1.945 * | 3.2 |
| + ZA-A (20 μM) | 4.649 ± 0.314 | 1.4 | 33.885 ± 7.803 * | 11.2 |
| + ZA-A (40 μM) | 1.607 ± 0.638 * | 4.1 | 4.099 ± 1.729 * | 93.0 |
| + ZA-B (10 μM) | 5.749 ± 0.021 | 1.1 | 243.946 ± 26.032 | 1.6 |
| + ZA-B (20 μM) | 4.586 ± 0.182 | 1.4 | 172.364 ± 9.353 * | 2.2 |
| + ZA-B (40 μM) | 4.362 ± 0.650 | 1.5 | 27.096 ± 2.799 * | 14.1 |
| + ZA-C (10 μM) | 5.089 ± 0.752 | 1.3 | 312.457 ± 3.101 | 1.2 |
| + ZA-C (20 μM) | 4.534 ± 0.063 | 1.4 | 285.500 ± 3.030 | 1.3 |
| + ZA-C (40 μM) | 3.844 ± 0.297 | 1.7 | 201.582 ± 8.884 | 1.9 |
| Cell Line | Flp-InTM293 | ABCB1/Flp-InTM293 | ||
| Compound | IC50 ± S.E. (nM) | RF | IC50 ± S.E. (nM) | RF |
| Vincristine | 83.822 ± 1.350 | 1.0 | 3858.781 ±217.537 | 1.0 |
| + ZA-A (10 μM) | 85.570 ± 0.120 | 1.0 | 1868.840 ± 12.138 * | 2.1 |
| + ZA-A (20 μM) | 76.274 ± 9.852 | 1.1 | 1200.585 ± 112.243 * | 3.2 |
| + ZA-A (40 μM) | 3.732 ± 0.040 * | 22.5 | 134.419 ± 4.400 * | 28.7 |
| + ZA-B (10 μM) | 43.234 ± 1.928 | 1.9 | 1698.434 ± 325.352 * | 2.3 |
| + ZA-B (20 μM) | 36.645 ± 12.895 * | 2.3 | 1475.014 ± 111.837 * | 2.6 |
| + ZA-B (40 μM) | 4.459 ± 0.455 * | 18.8 | 399.390 ± 30.490 * | 9.7 |
| + ZA-C (10 μM) | 69.245 ± 0.871 | 1.2 | 2995.200 ± 51.279 | 1.3 |
| + ZA-C (20 μM) | 54.267 ± 1.594 | 1.5 | 2274.225 ± 75.201 | 1.7 |
| + ZA-C (40 μM) | 7.801 ± 1.350 * | 10.7 | 1553.927 ± 33.018 * | 2.5 |
| Cell Line | HeLaS3 | KB/VIN | ||
| Compound | IC50 ± S.E. (nM) | RF | IC50 ± S.E. (nM) | RF |
| Doxorubicin | 101.088 ± 1.283 | 1.0 | 3819.266 ± 98.110 | 1.0 |
| + ZA-A (10 μM) | 75.675 ± 0.732 | 1.3 | 427.791 ± 49.019 * | 8.9 |
| + ZA-A (20 μM) | 51.914 ± 1.216 | 1.9 | 76.419 ± 0.151 * | 50.0 |
| + ZA-B (10 μM) | 105.041 ± 6.633 | 1.0 | 2109.439 ± 24.634 | 1.8 |
| + ZA-B (20 μM) | 69.211 ± 5.204 | 1.5 | 1310.133 ± 1.797 * | 2.9 |
| + ZA-C (10 μM) | 99.561 ± 8.025 | 1.0 | 2146.430 ± 129.070 | 1.8 |
| + ZA-C (20 μM) | 83.740 ± 2.555 | 1.2 | 621.683 ± 17.651 * | 6.1 |
| Cell Line | HeLaS3 | KB/VIN | ||
| Compound | IC50 ± S.E. (nM) | RF | IC50 ± S.E. (nM) | RF |
| Paclitaxel | 4.994 ± 0.077 | 1.0 | 510.512 ± 11.020 | 1.0 |
| + ZA-A (10 μM) | 1.509 ± 0.032 * | 3.3 | 141.745 ± 0.293 * | 3.6 |
| + ZA-A (20 μM) | 0.793 ± 0.028 * | 6.3 | 45.807 ± 0.128 * | 11.1 |
| + ZA-B (10 μM) | 2.053 ± 0.002 * | 2.4 | 225.581 ± 1.992 * | 2.3 |
| + ZA-B (20 μM) | 0.609 ± 0.004 * | 8.2 | 139.363 ± 2.287 * | 3.7 |
| + ZA-C (10 μM) | 3.657 ± 0.031 | 1.4 | 247.282 ± 7.276 * | 2.1 |
| + ZA-C (20 μM) | 3.238 ± 0.146 | 1.5 | 215.503 ± 5.563 * | 2.4 |
| Cell Line | HeLaS3 | KB/VIN | ||
| Compound | IC50 ± S.E. (nM) | RF | IC50 ± S.E. (nM) | RF |
| Vincristine | 17.326 ± 0.645 | 1.0 | 15730.719 ± 2402.023 | 1.0 |
| + ZA-A (10 μM) | 6.661 ± 0.820 * | 2.6 | 2007.168 ± 51.760 * | 7.8 |
| + ZA-A (20 μM) | 4.463 ± 0.549 * | 3.9 | 304.132 ± 28.753 * | 51.7 |
| + ZA-B (10 μM) | 8.838 ± 0.420 * | 2.0 | 1967.489 ± 98.547 * | 8.0 |
| + ZA-B (20 μM) | 6.590 ± 0.236 * | 2.6 | 1018.696 ± 138.704 * | 15.4 |
| + ZA-C (10 μM) | 17.004 ± 1.944 | 1.0 | 6952.094 ± 54.567 * | 2.3 |
| + ZA-C (20 μM) | 15.795 ± 0.928 | 1.1 | 2938.459 ± 874.146 * | 5.4 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Teng, Y.-N.; Wang, Y.-H.; Wu, T.-S.; Hung, H.-Y.; Hung, C.-C. Zhankuic Acids A, B and C from Taiwanofungus camphoratus Act as Cytotoxicity Enhancers by Regulating P-Glycoprotein in Multi-Drug Resistant Cancer Cells. Biomolecules 2019, 9, 759. https://doi.org/10.3390/biom9120759
Teng Y-N, Wang Y-H, Wu T-S, Hung H-Y, Hung C-C. Zhankuic Acids A, B and C from Taiwanofungus camphoratus Act as Cytotoxicity Enhancers by Regulating P-Glycoprotein in Multi-Drug Resistant Cancer Cells. Biomolecules. 2019; 9(12):759. https://doi.org/10.3390/biom9120759
Chicago/Turabian StyleTeng, Yu-Ning, Yen-Hsiang Wang, Tian-Shung Wu, Hsin-Yi Hung, and Chin-Chuan Hung. 2019. "Zhankuic Acids A, B and C from Taiwanofungus camphoratus Act as Cytotoxicity Enhancers by Regulating P-Glycoprotein in Multi-Drug Resistant Cancer Cells" Biomolecules 9, no. 12: 759. https://doi.org/10.3390/biom9120759
APA StyleTeng, Y.-N., Wang, Y.-H., Wu, T.-S., Hung, H.-Y., & Hung, C.-C. (2019). Zhankuic Acids A, B and C from Taiwanofungus camphoratus Act as Cytotoxicity Enhancers by Regulating P-Glycoprotein in Multi-Drug Resistant Cancer Cells. Biomolecules, 9(12), 759. https://doi.org/10.3390/biom9120759







