Abstract
The heartwood of the Swiss Stone Pine, Pinus cembra L., has been scarcely investigated for secondary metabolites for a long period of time. Considering age and relative simplicity of heartwood investigations dating back to the 1940s to 1960s, we conducted the first investigation of P. cembra heartwood by HPLC, using UHPLC-DAD-ESI-MSn and HPLC-DAD techniques in combination with isolation and NMR spectroscopy, with focus on stilbenes, bibenzyls and flavonoids. Analytical problems in the HPLC analysis of Pinus stilbenes and flavonoids on reversed stationary phases were also challenged, by comparing HPLC on pentafluorophenyl (PFP) and C18 stationary phases. Seven flavonoids (1, 2, 3, 7, 8, 11, 12), four stilbenes (4, 6, 10, 13), two bibenzyls (5, 9), three fatty acids (14, 16, 17) and one diterpenic acid (15) were detected in an ethanolic extract of Pinus cembra heartwood. HPLC comparison of reversed stationary phases in HPLC showed that the antifungal, antibacterial and chemosensitizing dihydropinosylvin monomethyl ether (9) and pinosylvin monomethyl ether (10) can be separated on PFP, but not on C18 material, when eluting with a screening gradient of 20–100% acetonitrile. Flavonoid separation showed additional benefits of combining analyses on different stationary phases, as flavonoids 7 and 8 could only be separated on one of two C18 stationary phases. Earlier phytochemical results for heartwood investigations were shown to be mostly correct, yet expandable. Substances 5 to 12 were found in alignment with these references, proving remarkable phytochemical analyses at the time. Evidence for the described presence of pinobanksin could not be found. Substances 1 to 4 and 13 have to our knowledge not yet been described for P. cembra.
Keywords:
Pinus cembra; heartwood; phytochemistry; flavonoids; stilbenes; bibenzyls; UHPLC-MS; HPLC-DAD; isolation; NMR 1. Introduction
The Swiss Stone Pine, Pinus cembra L., is a prominent coniferous tree in the family Pinaceae. It is distributed in the Alps and Carpathian Mountains and has a long history of use for its wood as crafting and building material. The seeds are very nutritious and have culinary uses [1]. Essential oils and preparations produced from different Pinus cembra parts and liquors aromatized with fresh cones are commercially available. However, the phytochemistry of secondary metabolites in Pinus cembra, especially in wood samples, has been rather scarcely investigated for a long period of time. Results in literature have also never been compiled appropriately, even more so while credibly excluding results for Pinus sibirica Du Tour, which is often incorporated into the species as Pinus cembra var. sibirica or Pinus cembra subsp. sibirica [2,3]. Therefore, a chronological summary of phytochemical research concerning secondary metabolites in Pinus cembra L. is appropriate and provided by us in the following. Here, special focus is given to substance groups of stilbenes, bibenzyls and flavonoids. In earlier studies of P. cembra heartwood, the isolation of the stilbenes pinosylvin (PS), pinosylvin monomethyl ether (PSM), the corresponding bibenzyls dihydropinosylvin (DHPS), and dihydropinosylvin monomethyl ether (DHPSM) and the flavonoids chrysin, tectochrysin, pinocembrin, pinostrobin and pinobanksin was reported [4,5,6]. Griesbach and Santamour investigated anthocyanins in male cones of P. cembra by HPLC-UV, reporting cyanidin-3-glucoside, delphinidin-3-glucoside and peonidin-3-glucoside [7]. Slimestad published results for flavonoids in buds and young needles investigated by HPLC-DAD-MS, giving kaempferol-3-glucoside, isorhamnetin-3-glucoside, kaempferol-3-(6″-coumaroyl-glucoside) and kaempferol-3-(3″, 6″-di-coumaroyl-glucoside) [8]. For some years, scientific focus shifted to knotwood investigations, with the main stilbene and bibenzyl being PSM and DHPSM [9] and pinocembrin as major flavonoid [10,11]. Others reported total phenolics, flavonoids and proanthocyanidins in bark and needles [12], and group investigations with reference standards for fatty acids, resin acids, terpenes, phenolic compounds and saccharides [13], however, without identification of flavonoids, stilbenes and bibenzyls. Also, P. cembra softwood lignins were investigated by NMR spectroscopy [14]. HPLC techniques in the analysis of P. cembra wood samples have only been reintroduced in recent years. Patyra et al. investigated branch wood methanolic extracts by LC-DAD-ESI-MSn to find fourteen compounds, of which six catechin derivatives and procyanidins, as well as taxifolin, a taxifolin hexoside, eriodictyol and pinobanksin can be assigned to the substance group of flavonoids. PSM was the only reported stilbene in P. cembra extracts [15]. Finally, Coniglio et al. detected PS, DHPS, chrysin and pinocembrin in P. cembra knotwood extracts by LC-ESI-MS [16].
As given here, composition of stilbenes, bibenzyls and flavonoids seems to vary between wood samples of different morphological parts. Considering these variances, we aim for the first investigation of Pinus cembra heartwood by HPLC, using UHPLC-DAD-ESI-MSn and HPLC-DAD techniques, with focus on stilbenes, bibenzyls and flavonoids, compound classes of numerous biological and pharmacological effects. Thereby we also revisit some results from Erdtman et al. and Lindstedt et al. [4,5,6] with modern instrument-based chromatographic possibilities in combination with isolation and NMR spectroscopy. In course of this study, we also challenge major analytical problems in the HPLC analysis of Pinus stilbenes and flavonoids on reversed stationary phases. New findings in method development are of interest for the development of fast and reliable natural product isolation by HPLC.
2. Results and Discussion
2.1. UHPLC-DAD-ESI-MSn Analysis
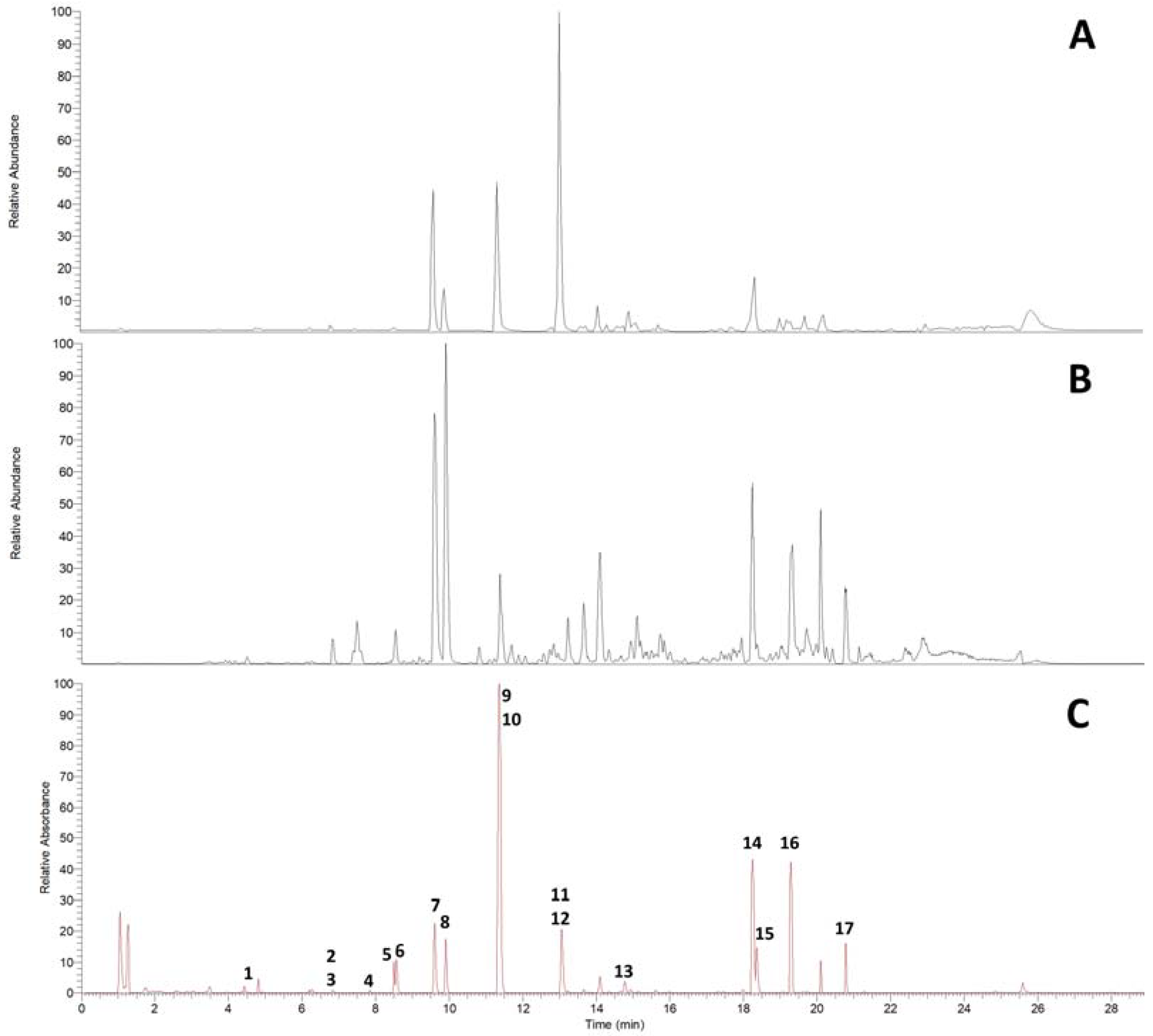
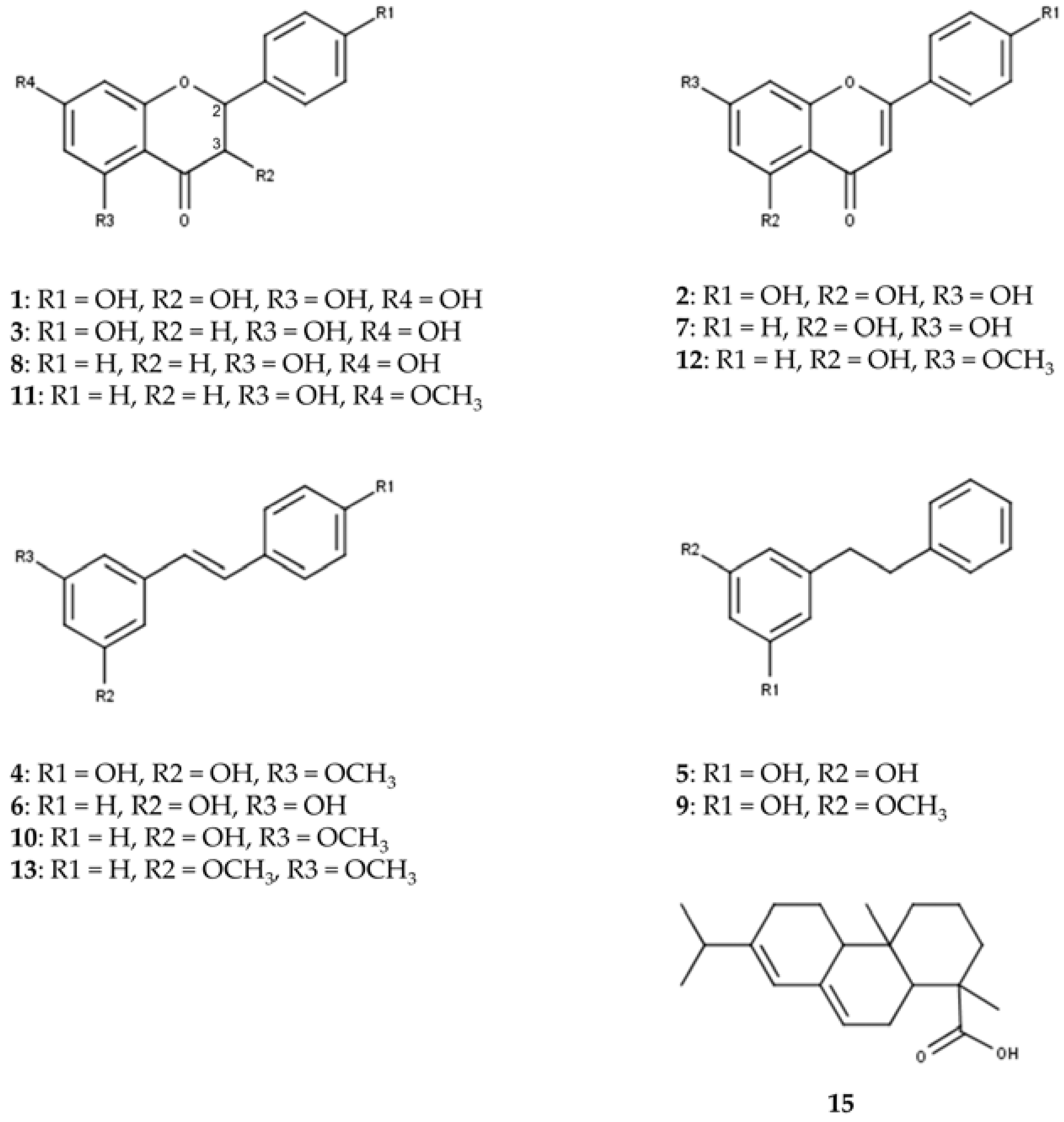
UHPLC on an RP18 stationary phase was employed for the initial qualitative analysis of a Pinus cembra heartwood ethanolic extract. Seventeen substances were characterized by interpretation of DAD-UV data and MSn in positive and negative ionization mode, of which six could further be confirmed by comparison with authentic reference standards and four by subsequent isolation (2.2), as given in Table 1. A chromatogram of MS in positive and negative ionization mode and DAD-UV (190–700 nm) can be found in Figure 1; Figure 2 gives structures of all characterized substances except for common fatty acids 14, 16 and 17.

Table 1.
Qualitative analysis of Pinus cembra heartwood ethanolic extract.
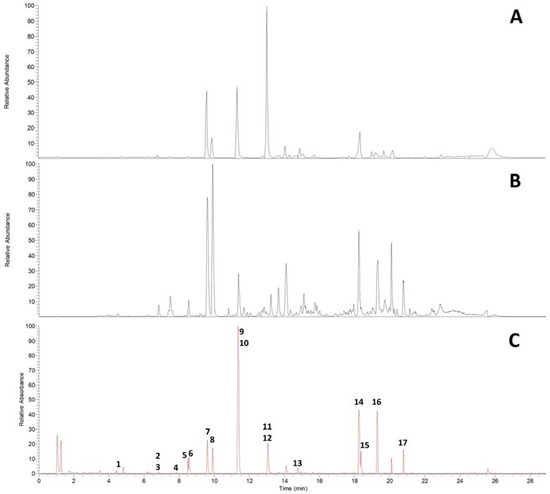
Figure 1.
UHPLC-DAD-ESI-MSn chromatograms of Pinus cembra heartwood ethanolic extract on RP18 stationary phase. (A) ESI positive mode; (B) ESI negative mode; (C) DAD-UV (Spectrum maxi-mum 190–700 nm).
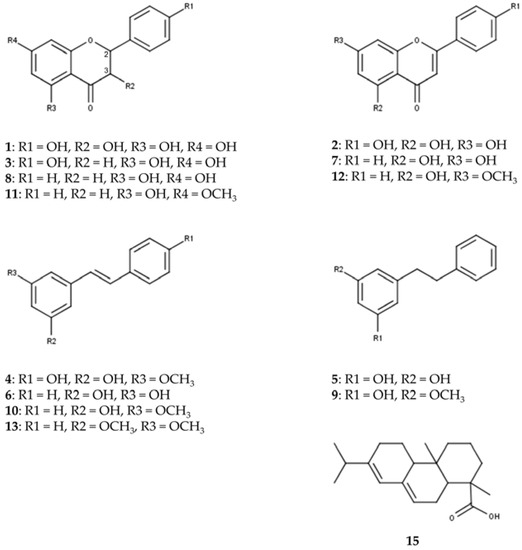
Figure 2.
Structures of characterized substances except for common fatty acids 14, 16 and 17.
The constituents can be grouped into seven flavonoids (1, 2, 3, 7, 8, 11, 12), four stilbenes (4, 6, 10, 13), two bibenzyls (5, 9), three fatty acids (14, 16, 17) and one diterpenic acid (15). Additional data for MSn in positive and negative ionization mode are given in Supplementary Materials (Tables S1 and S2).
To our knowledge, trace flavonoids 1 (aromadendrin), 2 (apigenin) and 3 (naringenin) are described for Pinus cembra for the first time. Previous literature mentions the occurrence of 1 and 2 in Pinus sibirica [17]. For flavonoid 1, comparison with MS data in literature supports the characterization of aromadendrin rather than eriodictyol [18,19,20]. Evidence for the presence of pinobanksin could not be identified, as MS data for flavonoid 3 rather suggest the detection of naringenin [15,19,20,21,22,23]. For flavonoid 1, indicative losses of H2O (18 amu) and CO (28 amu) are detected in positive ionization mode, to form [M+H-H2O]+ (m/z 271), [M+H-H2O-CO]+ (m/z 243) and [M+H-H2O-2CO]+ (m/z 215). C-ring cleavage leads to the detection of an 1,3A+-fragment (m/z 153); also, the loss of the B-ring can be recognized as [M+H-B-ring]+ (m/z 195). Besides several losses of small groups in negative ionization mode, the 1,3A−-fragment (m/z 151) is also detected. Flavonoid 2 shows the combined loss of H2O and CO in positive ionization mode to give [M+H-H2O-CO]+ (m/z 225), as well as the 1,3A+-fragment (m/z 153). The 1,3A−-fragment (m/z 151) is again detected in negative ionization mode. In positive ionization mode, the fragmentation of flavonoid 3 shows the 1,3A+-fragment (m/z 153) and 0,4B+-fragment (m/z 147) at high intensities after C-ring cleavage. In negative ionization mode, the 1,3A−-fragment (m/z 151) is detected as the most abundant fragment ion in MS2. Flavonoids 7 (chrysin), 8 (pinocembrin), 11 (pinostrobin) and 12 (tectochrysin) were found in accordance with older references for Pinus cembra [4,5,6,10,11,16].
Stilbenes 6 (pinosylvin, PS) and 10 (pinosylvin monomethyl ether, PSM) as well as corresponding bibenzyls 5 (dihydropinosylvin, DHPS) and 9 (dihydropinosylvin monomethyl ether, DHPSM) were previously described for Pinus cembra heartwood [4,5,6,9,15,16]. Stilbenes 4 (pinostilbene) and 13 (pinosylvin dimethyl ether) were only found in other Pinus species, including old references of occurrence in Pinus sibirica [24,25]. ESI-MS in positive ionization mode proved to give more spectral information for stilbenes than negative ionization. In positive ionization mode, stilbene 4 shows the loss of a phenolic ring [M+H-C6H6O]+ (m/z 149) as the most abundant fragment ion in MS2. This is followed by a fragmentation of −28 amu in MS3 (m/z 149 to 121), which could be interpreted as [M+H-C6H6O-CH2CH2]+ or [M+H-C6H6O-CO]+. In negative ionization mode, the loss of a methyl group (15 amu) gives the most abundant fragment ion [M-H-CH3]- (m/z 226) in MS2. Stilbene 13 shares the most fragments in MS2 of the positive ionization mode with stilbene 10, with the molecular ion [M+H]+ and the two heaviest fragment ions of 13 (m/z 241, 223 and 213, respectively) showing +14 amu, which corresponds to an additional methoxy- instead of hydroxy-group in stilbene 13.
2.2. Isolation of Major Compounds
For further confirmation of UHPLC-DAD-ESI-MSn results, isolation of several main compounds in Pinus cembra heartwood was performed. Open-column chromatography (CC) of the ethanolic extract on silica led to 67 fractions. Chrysin (7) directly precipitated in F28 without further processing. Four major components were isolated by semipreparative reversed phase HPLC after evaluation of all combined fractions by LC-MS. Partial separation on silica facilitated the consecutive isolation of the main bibenzyl 9 and stilbene 10 in Pinus cembra heartwood, as well as a mixture of both. When a combination of both substances was present in the same CC fraction (F20–22), they could not be isolated separately, as they eluted in one peak on RP18 phase at identical retention times. DHPSM (9) and PSM (10) proved to have interesting pharmacological potential in the past, showing anticancer and chemosensitizing effects of different potencies against human cancer cell lines [26,27]. Both substances also showed a wide spectrum of antibiotic and antifungal properties [11,28]. Two major flavonoids 8 and 12 could also be isolated. Results as confirmed by NMR (Supplementary Materials Figures S1–S4, Tables S3–S6) are given in Table 2.

Table 2.
Substances isolated from CC fractions of Pinus cembra heartwood ethanolic extract by semi-preparative HPLC on RP18 stationary phase. Identification was confirmed by NMR analysis.
2.3. Chromatographic Optimization for HPLC Separation of Isolated Compounds
Qualitative UHPLC analysis showed that bibenzyl 9 and its corresponding stilbene 10 were not distinguishable by their retention behavior on a Zorbax Eclipse Plus C18 column (EclC18) when eluting with a linear gradient of increasing concentrations of acetonitrile, as is common practice for HPLC analytical screenings of plant extracts (Figure 1). This poses an analytical problem that, to our knowledge, has not been tackled in literature before and could potentially lead to difficulties in detecting pairs of stilbenes and bibenzyls in the analysis of plant extracts via HPLC. The mixture of 9 and 10 isolated from F20–22 was therefore used for further analytical optimization by variation of stationary phase and mobile phase gradient.
Three different HPLC columns were selected for testing and optimizing separation of substances 9 and 10: EclC18, that was also used for qualitative analysis of the ethanolic extract and fractions, a Kinetex C18 (KtxC18) and a Kinetex PFP (KtxPFP) of identical dimensions and core shell particle size, but with a pentafluorophenyl reversed stationary phase. The results are summarized in Table 3, with resolution values calculated from full width at half maximum (FWHM). A detection wavelength of 210 nm was chosen in accordance with strong UV absorption presented by both substances. Three-dimensional chromatograms (190–500 nm) and chromatograms at 210 nm are given in Supplementary Materials (Figures S5–S10).

Table 3.
HPLC studies on mixture of isolated compounds 9 and 10, UV-detection at λ = 210 nm.
As expected, gradient elution of 9 and 10 on EclC18 showed no separation. Gradient elution on KtxC18 led to similar results. However, a peak shoulder was visible at λ = 210 nm, which indicates slight separation. On KtxPFP, it was possible to separate 9 and 10 using gradient elution (RS = 1.90). Isocratic elution on EclC18 and KtxC18 facilitated peak separation at 30% acetonitrile (RS = 1.68, RS = 1.57), although retention times were high, doubling from KtxC18 (24.787, 27.267 min) to EclC18 (51.243, 55.020 min). Elution on KtxPFP at 40% acetonitrile gave peak separation at remarkably low retention times (5.620, 6.237 min) with high resolution (RS = 2.55). According to information by the manufacturer, a pentafluorophenyl stationary phase offers high degrees of steric interaction and can therefore improve the separation of structural isomers. Furthermore, fluorine groups with high electronegativity are present. It can be assumed that a combination of steric and polar interactions leads to the separation of compounds 9 and 10, which only differ by one central double bond in the molecules.
2.4. Application to Extract Sample
As separation of isolated substances 9 and 10 had been achieved using gradient elution on KtxPFP, it was then tested on an additional Pinus cembra heartwood ethanolic extract and produced according to the method given under 3.3. Elution with the beforementioned linear gradient of 20 to 100% acetonitrile simulated a real-life sample in plant extract screening via HPLC and analyses were performed on all three columns (EclC18, KtxC18, KtxPFP), to work out potential differences.
All comparative results for the main bibenzyl and stilbene 9 and 10, as well as retention of major flavonoids 7, 8, 11 and 12 at a detection wavelength of 210 nm are given in Table 4. Three-dimensional chromatograms (190–500 nm) and chromatograms at 210 nm can be found in Supplementary Materials (Figures S11–S13).

Table 4.
HPLC screening of Pinus cembra heartwood ethanolic extract on different columns using gradient elution, UV-detection at λ = 210 nm.
In the more complex matrix of the extract solution, separation of 9 and 10 by gradient elution was only successful on KtxPFP, comparable to results for isolated substances (RS = 1.90). Additionally, effects on flavonoid separation became obvious, with 11 and 12 being separated (RS = 1.25), which was also not possible on EclC18 during initial qualitative analysis. On KtxC18, which has identical column parameters to KtxPFP except for stationary phase type, 11 and 12, differing by a double bond between C-2 and C-3, were also separated, but with lower resolution (RS = 0.88). Slight separation of 9 and 10 within the same peak could be visualized in 3D chromatograms. However, out of all three columns, 7 and 8, differing from each other by a double bond between C-2 and C-3, were only separated on EclC18 (RS = 2.44), with no separation for 9 and 10 as well as 11 and 12. EclC18 is a double-endcapped C18 stationary phase on a special porous silica support, which according to the manufacturer is designed to reduce strong absorption of basic and highly polar compounds. This could have improved the separation of 7 and 8, having no methoxyl moiety in contrast to 9–12, in comparison to other columns.
3. Materials and Methods
3.1. Plant Material
All the investigations were based on a complete radial cut disk of a Pinus cembra trunk, with approximate thickness of 4.5 cm and a diameter of 26.6 to 28.3 cm. The age of the tree was about 54 years. Material was obtained from the sawmill “Sägewerk und Hobelwerk Seebacher” (Himmelberg, Carinthia, Austria) in July 2021. A certificate of origin for the region of Nockberge, Austria, was given in written form. A voucher specimen was deposited at the Department of Pharmacognosy, University of Graz.
For analytical investigations, two sets of eight drillings through heartwood areas were made with a flat milling drill and wood chips combined to yield 45.79 g and 45.18 g of heartwood, respectively. Wood chips were freeze-dried using a VirTis BenchTop Pro freeze-drier (SP Industries, Warminster, PA, USA) for 24 h before grinding in a Retsch ZM100 centrifugal mill with 2.0 mm mesh (Retsch, Haan, Nordrhein-Westfalen, Germany).
3.2. Solvents and Reference Substances
Solvents for different steps in analyses were partially denatured ethanol (AustrAlco, Spillern, Austria) for extraction, hexane, ethyl acetate (both Carl Roth, Karlsruhe, Baden-Württemberg, Germany) and methanol (VWR, Radnor, PA, USA) for open column chromatography and TLC, chloroform-d1 (VWR) and pyridine-d5 (Armar, Döttingen, Aargau, Switzerland) for NMR.
HPLC mobile phases consisted of ultrapure water prepared from deionized water with a Barnstead MicroPure system (Thermo Fisher Scientific, Waltham, MA, USA), HPLC-grade acetonitrile (VWR) and formic acid for LC-MS (Honeywell, Charlotte, NC, USA).
Reference substances were abietic acid contributed by the Department of Pharmaceutical Chemistry, University of Graz, Austria, apigenin (Sigma-Aldrich, St. Louis, MO, USA), chrysin, pinocembrin-7-methyl ether (pinostrobin), linoleic acid (all Carl Roth) and oleic acid (Fluka, Buchs, St. Gallen, Switzerland).
3.3. Extraction and Fractionation
Two separate Soxhlet extractions of 40.95 g and 41.36 g of plant material were carried out with 500 mL of ethanol each for 24 h. The native extracts were evaporated to dryness on a rotary evaporator (Büchi R-100, Büchi, Flawil, St. Gallen, Switzerland). A total of 2.63 g of dry extract yielded from the first Soxhlet extraction were separated by open-column chromatography on 25 g of silica (40–63 µm, VWR) with hexane, ethyl acetate and methanol in solvent systems of rising polarity, leading to 67 fractions.
TLC analyses of fractions were carried out on Silica Gel 60 F254 aluminium plates (Merck, Darmstadt, Hessen, Germany) with mobile phase systems hexane/ethyl acetate/methanol = 75/20/5 (v/v) for apolar fractions and hexane/ethyl acetate/methanol = 60/20/20 (v/v) for polar fractions. The staining reagent consisted of 0.5 mL of 4-anisaldehyde, 10 mL of acetic acid (both Carl Roth), 85 mL of methanol and 5 mL of sulfuric acid (Carl Roth). After spraying, the plates were heated at 105 °C for 5 min.
3.4. UHPLC-DAD-ESI-MSn
Analyses of the first ethanolic extract, combined fractions and reference substances were conducted on a Dionex UltiMate 3000 RS system equipped with a DAD detector and coupled to an LTQ XL linear ion-trap mass spectrometer with ESI ion source (allThermo Fisher Scientific, Waltham, MA, USA). A Zorbax Eclipse Plus C18 column, 2.1 × 100 mm and 1.8 µm particle size (Agilent, Santa Clara, CA, USA) served as stationary phase, while the mobile phase was made up of water +0.1% formic acid (A) and acetonitrile (B). Gradient elution at a flow rate of 0.25 mL/min started at 20% B, rising to 100% B at 20.0 min, followed by a plateau of 100% B to 23.0 min and a drop back to 20% B at 23.5 min, which was kept stable until 29 min for re-equilibration. Column temperature was 35 °C. Injection volumes were 2 µL for dry ethanolic extract in methanol (5 mg/mL) and 1 µL for reference substances dissolved in methanol (1 mg/mL) as well as subfractions of varying concentrations.
DAD-UV spectra were recorded in a wavelength range of 190 to 700 nm. Mass spectral detection was performed in the range of m/z 50 to 2000 amu, with conditions set as follows: source heater temperature 250 °C, sheath gas flow 50 arb (arbitrary units), auxiliary gas flow 8 arb, source voltage 4.0 kV for ESI negative mode and 4.2 kV for ESI positive mode.
3.5. HPLC-DAD
A separate Dionex UltiMate 3000 RS system was used for analytical optimization without coupling to MS. Chromatographic conditions were the same as for qualitative UHPLC analysis, with the additional use of a Kinetex C18 column, 100 × 2.1 mm, 2.6 μm column and a Kinetex PFP column, 100 × 2.1 mm, 2.6 μm (both Phenomenex, Torrance, CA, USA). Samples of isolated compounds dissolved in methanol (1 mg/mL) were injected at 1 µL; a second dry ethanolic extract in methanol (5 mg/mL) had an injection volume of 2 µL.
3.6. Semipreparative HPLC
Semipreparative HPLC was carried out on a Shimadzu system consisting of DGU-20A5R degassing unit, LC-20AT solvent delivery pump, SIL-10AF autosampler, CBM-20A controller, CTO-20AC column oven, SPD-M20A diode array detector and FRC-10A fraction collector (all Shimadzu, Kyoto, Kinki, Japan). Stationary phase was a Luna C-10(2) column with 250 × 10 mm and 10 µm particle size (Phenomenex). Mobile phase consisted of water (A) and acetonitrile (B). Elution at 4 mL/min and 35 °C column temperature started at 40% B, rising to 100% B at 20.0 min. All compounds of interest eluted within this gradient, followed by varying plateaus of 100% B (0–5 min duration) for column cleaning and re-equilibration at 40% B. Injection volume was 200 µL.
3.7. NMR
Isolated substances 9 and 10 were measured on a Bruker NEO NMR-spectrometer (Bruker Corporation; Billerica, MA, USA) at 400 MHz in chloroform-d1. Substances 8 and 12 were measured on a Bruker Advance II NMR-spectrometer (Bruker Corporation) at 700 MHz in pyridine-d5. All measurements were performed with an internal standard of TMS. Recorded data sets were 1H and 13C, as well as 2D experiments COSY, HSQC and HMBC.
4. Conclusions
The present study investigating constituents of Pinus cembra heartwood with a focus on flavonoids, stilbenes and bibenzyls has shown early results for equivalent morphological plant material, generated in the 1940s to 1960s to be mostly true, yet expandable. We employed modern instrument-based HPLC techniques in combination with isolation and NMR spectroscopy to find substances 5 to 12 in alignment with these literature references, proving remarkable phytochemical analyses at the time [4,5,6].
However, we could not find evidence for the presence of pinobanksin, as described by Erdtman et al. in 1966 [6]. In addition, substances 1 to 4 and 13 have, to our knowledge, not yet been described for Pinus cembra.
This contribution to the knowledge of Pinus cembra heartwood phytochemistry also brings some clarity in consideration of reported variances in composition of different morphological parts of Pinus cembra. As for stilbenes and bibenzyls, Coniglio et al. find PS (6) and DHPS (5) in knotwood, while Patyra et al. report only PSM (10) in branch wood [15,16]. Both detect no DHPSM (9), while Erdtman et al., Lindstedt et al. and Willför et al. find it in knot- and heartwood [4,5,6,9]. For flavonoids, pinocembrin (8) aligns for knotwood and heartwood investigations, while other components vary as well.
In consecutive analyses, HPLC comparisons using different reversed phase materials as stationary phases showed a pentafluorophenyl reversed phase (KtxPFP) to be able to separate bibenzyl DHPSM (9) and stilbene PSM (10), which was not possible on two different C18 stationary phases (EclC18, KtxC18). Flavonoids pinostrobin (11) and tectochrysin (12) could be separated on KtxPFP and KtxC18. However, flavonoids chrysin (7) and pinocembrin (8) were only separated on EclC18.
Results show that a combination of different reversed phase columns could be recommended for HPLC screening of plant extracts using gradient elution, as good chromatographic resolution for several substances of interest is desired. This should especially be considered for UV detection, as overlays of coeluting substances make them hard to identify by UV-fingerprint, or when classifying substance groups through UV spectra. Quantification and isolation of individual components will also not be possible. In MS detection, coeluting substances can often be differentiated by their molecular ion when tracking specific m/z values using single ion monitoring. For screening purposes, good chromatographic resolution of substances should still be aimed for. Furthermore, chromatographic techniques coupled with mass spectrometry are destructive and therefore not useful for isolation of natural products. DHPSM (9) and PSM (10) proved to have interesting pharmacological potential in the past, showing anticancer and chemosensitizing effects of different potencies against human cancer cell lines [26,27]. Both substances also showed a wide spectrum of antibiotic and antifungal properties [11,28]. Still, repeated CC and HPLC were often necessary for isolation before biological testing. This could be improved by implementing HPLC method development employing different stationary phases in the fast and cost-effective isolation of these substances for further pharmacological testing.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/plants12193388/s1, Figure S1: Structure of substance 9 Dihydropinosylvin monomethyl ether; Figure S2: Structure of substance 10 Pinosylvin monomethyl ether; Figure S3: Structure of substance 12 Tectochrysin; Figure S4: Structure of substance 8 Pinocembrin; Figure S5: HPLC chromatogram (5–15 min) of a mixture of 9 and 10, gradient elution (20–100% B), column EclC18; A: 210 nm, B: 3D field (190–500 nm); Figure S6: HPLC chromatogram of a mixture of 9 and 10, isocratic elution (30% B), column EclC18; A: 210 nm, B: 3D field (190–500 nm); Figure S7: HPLC chromatogram (5–15 min) of a mixture of 9 and 10, gradient elution (20–100% B), column KtxC18; A: 210 nm, B: 3D field (190–500 nm); Figure S8: HPLC chromatogram of a mixture of 9 and 10, isocratic elution (30% B), column KtxC18; A: 210 nm, B: 3D field (190–500 nm); Figure S9: HPLC chromatogram (5–15 min) of a mixture of 9 and 10, gradient elution (20–100% B), column KtxPFP; A: 210 nm, B: 3D field (190–500 nm); Figure S10: HPLC chromatogram of a mixture of 9 and 10, isocratic elution (40% B), column KtxPFP; A: 210 nm, B: 3D field (190–500 nm); Figure S11: HPLC chromatogram (8–18 min) of a Pinus cembra heartwood ethanolic extract, gradient elution (20–100% B), column EclC18; A: 210 nm, B: 3D field (190–500 nm); Figure S12: HPLC chromatogram (8–18 min) of a Pinus cembra heartwood ethanolic extract, gradient elution (20–100% B), column KtxC18; A: 210 nm, B: 3D field (190–500 nm); Figure S13: HPLC chromatogram (5–15 min) of a Pinus cembra heartwood ethanolic extract, gradient elution (20–100% B), column KtxPFP; A: 210 nm, B: 3D field (190–500 nm); Table S1: Qualitative analysis of Pinus cembra heartwood ethanolic extract. MSn data in ESI positive mode; Table S2: Qualitative analysis of Pinus cembra heartwood ethanolic extract. MSn data in ESI negative mode; Table S3: NMR-data of substance 9 Dihydropinosylvin monomethyl ether in CDCl3 (400 MHz); Table S4: NMR-data of substance 10 Pinosylvin monomethyl ether in CDCl3 (400 MHz); Table S5: NMR-data of substance 12 Tectochrysin in pyridin-d5 (700 MHz); Table S6: NMR-data of substance 8 Pinocembrin in pyridin-d5 (700 MHz).
Author Contributions
Conceptualization, F.A. and F.B.; methodology, F.A.; investigation, A.S., F.A. and O.K.; resources, A.S. and F.B.; writing—original draft preparation, F.A.; writing—review and editing, F.B., O.K. and A.S.; visualization, F.A.; supervision, F.B. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Data Availability Statement
Data are included in this article.
Acknowledgments
Open Access Funding by the University of Graz. The authors acknowledge financial support by the University of Graz. We thank Sägewerk und Hobelwerk Seebacher for providing Pinus cembra wood samples.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Schütt, P. Pinus cembra. In Enzyklopädie der Holzgewächse: Handbuch und Atlas der Dendrologie; Roloff, A., Weisgerber, H., Lang, U.M., Stimm, B., Eds.; Wiley-VCH: Weinheim, Germany, 2014; pp. 1–12. ISBN 9783527678518. [Google Scholar]
- World Flora Online. Pinus cembra L. Available online: https://worldfloraonline.org/taxon/wfo-0000481525 (accessed on 3 August 2023).
- World Flora Online. Pinus sibirica Du Tour. Available online: https://worldfloraonline.org/taxon/wfo-0000482273 (accessed on 3 August 2023).
- Erdtman, H. The phenolic constituents of pine heartwood. VI. The heartwood of Pinus cembra L. Sven. Kem. Tidskr. 1944, 56, 26–31. [Google Scholar]
- Lindstedt, G.; Misiorny, A. Constituents of Pine Heartwood: XXV. Investigation of Forty-Eight Pinus Species by Paper Partition Chromatography. Acta Chem. Scand. 1951, 5, 121–128. [Google Scholar] [CrossRef]
- Erdtman, H.; Kimland, B.; Norin, T. Pine Phenolics and Pine Classification. Bot. Mag. 1966, 79, 499–505. [Google Scholar] [CrossRef]
- Griesbach, R.J.; Santamour, J.F. Anthocyanins in cones of Abies, Picea, Pinus, Pseudotsuga and Tsuga (Pinaceae). Biochem. Syst. Ecol. 2003, 31, 261–268. [Google Scholar] [CrossRef]
- Slimestad, R. Flavonoids in buds and young needles of Picea, Pinus and Abies. Biochem. Syst. Ecol. 2003, 31, 1247–1255. [Google Scholar] [CrossRef]
- Willför, S.M.; Ahotupa, M.O.; Hemming, J.E.; Reunanen, M.H.T.; Eklund, P.C.; Sjöholm, R.E.; Eckerman, C.S.E.; Pohjamo, S.P.; Holmbom, B.R. Antioxidant Activity of Knotwood Extractives and Phenolic Compounds of Selected Tree Species. J. Agric. Food Chem. 2003, 51, 7600–7606. [Google Scholar] [CrossRef]
- Pietarinen, S.P.; Willför, S.M.; Ahotupa, M.O.; Hemming, J.E.; Holmbom, B.R. Knotwood and bark extracts: Strong antioxidants from waste materials. J. Wood Sci. 2006, 52, 436–444. [Google Scholar] [CrossRef]
- Välimaa, A.-L.; Honkalampi-Hämäläinen, U.; Pietarinen, S.; Willför, S.; Holmbom, B.; von Wright, A. Antimicrobial and cytotoxic knotwood extracts and related pure compounds and their effects on food-associated microorganisms. Int. J. Food Microbiol. 2007, 115, 235–243. [Google Scholar] [CrossRef]
- Apetrei, C.L.; Tuchilus, C.; Aprotosoaie, A.C.; Oprea, A.; Malterud, K.E.; Miron, A. Chemical, Antioxidant and Antimicrobial Investigations of Pinus cembra L. Bark and Needles. Molecules 2011, 16, 7773–7788. [Google Scholar] [CrossRef]
- Piccand, M.; Bianchi, S.; Halaburt, E.I.; Mayer, I. Characterization of extractives from biomasses of the alpine forests and their antioxidative efficacy. Ind. Crop. Prod. 2019, 142, 111832. [Google Scholar] [CrossRef]
- Faleva, A.V.; Kozhevnikov, A.Y.; Pokryshkin, S.A.; Falev, D.I.; Shestakov, S.L.; Popova, J.A. Structural characteristics of different softwood lignins according to 1D and 2D NMR spectroscopy. J. Wood Chem. Technol. 2020, 40, 178–189. [Google Scholar] [CrossRef]
- Patyra, A.; Dudek, M.K.; Kiss, A.K. LC-DAD-ESI-MS/MS and NMR Analysis of Conifer Wood Specialized Metabolites. Cells 2022, 11, 3332. [Google Scholar] [CrossRef] [PubMed]
- Coniglio, R.; Gaschler, W.; Dieste, A. Knot extractives responsible for the yellowing of white-coated pine wood. Eur. J. Wood Prod. 2023, 81, 1109–1117. [Google Scholar] [CrossRef]
- Lutskii, V.I.; Gromova, A.S.; Tyukavkina, N.A. Aromadendrin, apigenin, and kaempferol from the wood of Pinus sibirica. Chem. Nat. Compd. 1971, 7, 197–198. [Google Scholar] [CrossRef]
- Zhou, S.; Shao, Y.; Fu, J.; Xiang, L.; Zheng, Y.; Li, W. Characterization and Quantification of Taxifolin Related Flavonoids in Larix olgensis Henry Var. koreana Nakai Extract Analysis and its Antioxidant Activity Assay. Int. J. Pharmacol. 2018, 14, 534–545. [Google Scholar] [CrossRef]
- Zhang, Y.; Cao, C.; Yang, Z.; Jia, G.; Liu, X.; Li, X.; Cui, Z.; Li, A. Simultaneous determination of 20 phenolic compounds in propolis by HPLC-UV and HPLC-MS/MS. J. Food Compos. Anal. 2023, 115, 104877. [Google Scholar] [CrossRef]
- Tsimogiannis, D.; Samiotaki, M.; Panayotou, G.; Oreopoulou, V. Characterization of flavonoid subgroups and hydroxy substitution by HPLC-MS/MS. Molecules 2007, 12, 593–606. [Google Scholar] [CrossRef]
- Fang, T.; Wang, Y.; Ma, Y.; Su, W.; Bai, Y.; Zhao, P. A rapid LC/MS/MS quantitation assay for naringin and its two metabolites in rats plasma. J. Pharm. Biomed. Anal. 2006, 40, 454–459. [Google Scholar] [CrossRef]
- Falcão, S.I.; Vilas-Boas, M.; Estevinho, L.M.; Barros, C.; Domingues, M.R.M.; Cardoso, S.M. Phenolic characterization of Northeast Portuguese propolis: Usual and unusual compounds. Anal. Bioanal. Chem. 2010, 396, 887–897. [Google Scholar] [CrossRef]
- Piccinelli, A.L.; Lotti, C.; Campone, L.; Cuesta-Rubio, O.; Campo Fernandez, M.; Rastrelli, L. Cuban and Brazilian red propolis: Botanical origin and comparative analysis by high-performance liquid chromatography-photodiode array detection/electrospray ionization tandem mass spectrometry. J. Agric. Food Chem. 2011, 59, 6484–6491. [Google Scholar] [CrossRef]
- Tyukavkina, N.A.; Gromova, A.S.; Lutskii, V.I.; Voronov, V.K. Hydroxystilbenes from the bark of Pinus sibirica. Chem. Nat. Compd. 1972, 8, 570–572. [Google Scholar] [CrossRef]
- Lisina, A.I.; Kashtanova, N.K.; Dzizenko, A.K.; Pentegova, V.A. Composition of the neutral fraction of the oleoresin from Pinus sibirica. V. Agathadienediol and 3,5-dimethoxystilbene. Izv. Sib. Otd. Akad. Nauk. SSSR Seriya Khimicheskikh Nauk 1967, 1, 165–166. [Google Scholar]
- Sa, N.H.; Tam, N.T.; Anh, N.T.H.; Quan, T.D.; Thien, D.D.; Phong, D.T.; van Sung, T.; Thuy, T.T. Chemical constituents from the leaves of Pinus dalatensis Ferré. Nat. Prod. Res. 2018, 32, 341–345. [Google Scholar] [CrossRef] [PubMed]
- Sein, K.L.; Lertnitikul, N.; Suttisri, R.; Jianmongkol, S. Anticancer and chemosensitizing activities of stilbenoids from three orchid species. Naunyn Schmiedebergs Arch. Pharmacol. 2023, 396, 749–758. [Google Scholar] [CrossRef]
- Li, X.; Yao, L.; Xiong, B.; Wu, Y.; Chen, S.; Xu, Z.; Qiu, S.-X. Inhibitory Mechanism of Pinosylvin Monomethyl Ether against Aspergillus flavus. J. Agric. Food Chem. 2022, 70, 15840–15847. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).